Joint Arthrocentesis in the Emergency Department
October 1, 2017
Reprints
AUTHORS
Brian L. Springer, MD, FACEP, Associate Professor, Department of Emergency Medicine, Wright State University, Kettering, OH.
Brian M. Pennington, MD, Assistant Professor, Ultrasound Director, Department of Emergency Medicine, Wright State University, Kettering, OH.
PEER REVIEWER
Andrew Perron, MD, FACEP, Residency Program Director, Maine Medical Center, Portland.
EXECUTIVE SUMMARY
- The primary indication for arthrocentesis is evaluation of acute monoarticular arthritis.
- Disability and mortality are increased with delayed diagnosis of bacterial arthritis.
- There is no evidence that arthrocentesis through possibly cellulitic tissue causes a joint infection.
- Ultrasound-guided arthrocentesis may be less time consuming, less technically difficult, and less painful for the patient compared to the landmark technique.
- Peripheral blood tests lack adequate accuracy to include or exclude septic arthritis.
- There often is an overlap in the results of synovial fluid analysis between the various causes of monoarticular arthritis.
Introduction
Arthrocentesis, the technique of aspirating fluid from a joint capsule for diagnostic or therapeutic reasons, traces its origins back as far as the 15th century. At that time, the evacuation of body fluids to re-establish equilibrium among the four humors (air, fire, earth, and water) was the target of most all therapeutic procedures.1 Now, the ability to rapidly recognize infection, as well as distinguish inflammatory arthropathy from infection, crystal arthropathy, and osteoarthritis, makes arthrocentesis a valuable and sometimes essential emergency medicine procedure. Therapeutic removal of fluid from a tense joint effusion improves mobility and comfort, and provides access for the injection of analgesics, anesthetics, and corticosteroids. With the advent of bedside ultrasound, traditional identification of external landmarks to guide needle placement is being replaced by ultrasonic identification of joint effusions and real-time visualization of joint aspiration, improving procedural success rates.
This article will review the indications for arthrocentesis, explain how to perform the procedure with and without ultrasound guidance, and discuss how to interpret synovial fluid analysis when considering the differential diagnosis of acute monoarticular arthritis.
Indications
The presence of acute monoarticular arthritis in patients presenting to the emergency department has multiple potential etiologies. These include infection, crystalloid arthropathy, rheumatoid arthritis, osteoarthritis, lupus, and trauma. The removal of fluid from a joint effusion can provide significant pain relief and improve range of motion and mobility. For additional consideration, intra-articular morphine or lidocaine can provide significant pain relief.2,3 (See Table 1.)
Table 1. Indications for Arthrocentesis
- Evaluate for monoarticular arthritis.
- Identify cause of unexplained joint effusion.
- Rule out joint infection/limit joint damage from an infectious process.
- Identify intra-articular fractures or disruption of intra-articular structures.
- Provide symptomatic relief of pain from a tense effusion.
- Identify crystal-induced arthropathy.
- Inject therapeutic agents.
Adapted from: Boss SE, Mehta A, Maddow C, Luber SD. Critical orthopedic skills and procedures. Emerg Clin North Am 2013;31:261-290. Zuber TJ. Knee joint aspiration and injection. Am Fam Physician 2002;66:1497-1500.
Septic Arthritis
Septic arthritis is the most serious cause of monoarthritis in adults. Bacteria, fungus, mycobacteria, and viruses all can result in joint infection, although bacteria remain the most common cause of infection causing acute arthritis. Between 25-50% of patients with bacterial arthritis will sustain irreversible loss of joint function, and up to 75% will be left with significant functional disabilities. Mortality rates are 5-15%, highlighting the voracity of disease. Both disability and mortality are associated with delayed presentation and diagnosis.4,5
Elderly patients have a significantly higher risk for development of septic arthritis, given the prevalence of other disease states that can lead to seeding and infection of the joints. These include any acquired disease that results in immunosuppression, including diabetes, cancer, and chronic liver and kidney disease. Joint damage associated with systemic connective tissue disease or chronic arthritis increases the risk of bacterial seeding of the joint space. Intravenous drug abusers also are at increased risk, from direct inoculation, extension of nearby localized infection, or seeding from systemic bacteremia in cases of endocarditis.5 Patients with HIV have a greater risk for development of nongonococcal septic arthritis.4
Hematogenous spread of bacteria with subsequent joint seeding is seen most commonly in IV drug abusers, patients with endocarditis, and those with infected indwelling catheters. The synovial membrane is highly vascularized and lacks a basement membrane, facilitating bacterial entry into the joint.
Direct inoculation results from joint penetration from foreign bodies. This includes the dreaded “fight bite” in which teeth penetrate the joint spaces of the digits. Diabetics are at greater risk of extension of adjacent infection into the joint space. Presence of ulcers, cellulitis, bursitis, or osteomyelitis in the vicinity of a painful swollen joint should raise suspicion for joint infection.5
Once the joint has been seeded with bacteria, joint destruction can occur rapidly. Synovial necrosis extends into the articular cartilage, abetted by pressure generated from purulent synovial fluid. Inflamed synovium results in the release of proteolytic enzymes and acute inflammatory mediators that create progressive articular cartilage destruction that can continue even without viable organisms being present.
Although any joint in the body has the potential to become infected, the knee is most commonly affected by septic arthritis, accounting for approximately 50% of cases, followed by the hip, shoulder, and elbow.4
The clinical manifestations of joint infection include pain, joint swelling, and presence of effusion. Joint effusions may be difficult to recognize on clinical exam, although bedside ultrasound may facilitate detection. Joint pain with motion and limited range of motion on exam are both highly sensitive findings, although lack of empirical data means that specificities and likelihood ratios cannot be determined. Presence of fever is neither sensitive nor specific for septic arthritis, and may be present in crystal-induced arthritis as well. Use of antipyretic analgesics can further obscure this finding. Given limited studies, there is no finding from the patient’s history that will decrease the probability that an acutely painful joint is septic arthritis; recent joint surgery or a prosthesis with overlying infection increases probability.4,6
Periprosthetic Infection
Periprosthetic infections are a leading cause of morbidity in patients following total joint arthroplasty, and one of the most common reasons for early revision. Since cartilage, which is highly susceptible to damage in native joint infection, has been replaced by prosthesis, periprosthetic infections tend to be more indolent. They may occur in the acute postoperative period or be a chronic infection that develops months to years postoperatively. Both direct inoculation and hematogenous seeding can cause infection. A recent systemic illness, dental work, or urological or gastrointestinal procedure can cause acute hematogenous seeding and infection.
Risk factors for the development of a periprosthetic joint infection include smoking, obesity, diabetes, and the use of corticosteroids or other immunosuppressive medications. Intravenous drug use, psoriasis, venous stasis, and active skin ulcerations increase the risk of bacterial entry into the circulation. Infection of the surgical site in the perioperative period provides a source for direct inoculation of a prosthetic joint.6
The presence of a prosthetic joint is not a contraindication for arthrocentesis, although the indications for the procedure are somewhat more conservative. Testing strategies are based on history, exam, and patient risk factors. Joint aspiration is recommended if the erythrocyte sedimentation rate and C-reactive protein (CRP) levels are elevated, or when clinical suspicion for infection is high. Aspiration of the knee can be performed in the emergency department, while hip aspiration usually requires fluoroscopic guidance. Bedside ultrasound-guided arthrocentesis of prosthetic hips is not well documented in the literature and should not be attempted except by experienced providers in consultation with an orthopedic surgeon.6
Crystal-induced Arthropathies
Gout and pseudogout are the two most common crystal-induced arthropathies. Gout is caused by monosodium urate monohydrate crystals; pseudogout is caused by calcium pyrophosphate crystals. Acute onset of pain, edema, and inflammation in the metatarsal-phalangeal joint of the great toe (known as podagra) is highly suggestive of gout. Podagra is the initial joint manifestation in 50% of gout cases and is seen at some time in 90% of cases. The most common sites of pseudogout are large joints such as the knee, wrist, elbow, or ankle.7
As previously discussed, the cause of new-onset acute monoarticular arthritis cannot be determined reliably from the history and physical examination alone. Septic arthritis, gout, and pseudogout may present in similar fashions. Certain clinical presentations are highly characteristic of gout, and attempts have been made to diagnose or exclude gout without joint aspiration. Patient and clinical factors such as male sex, previous arthritis attack, onset within one day, joint redness, first metatarsophalangeal joint involvement, and elevated serum uric acid content make gout more likely.7
However, these factors still cannot be used to rule out septic arthritis. In fact, the two entities may coexist. There are documented cases in which arthrocentesis and synovial fluid analysis have demonstrated both the presence of crystals and a positive synovial fluid culture. The exact mechanism as to why these may occur simultaneously is not well understood. Theories include the release of crystal particles into the synovium prompted by infection-mediated inflammation, and the possibility that joint damage from crystal-induced arthritis is a predisposing factor to the development of intra-articular infection.8
The bottom line: The criterion standards for the diagnosis of gout and pseudogout are the demonstration of intracellular crystals and the exclusion of infection in the synovial fluid from the inflamed joint. Patients who present with acute inflammatory arthritis need to undergo arthrocentesis to exclude septic arthritis, especially if this is a first-time event.
Pain Control
Large joint effusions, especially secondary to traumatic injury, can cause significant patient discomfort and limit range of motion. Additionally, the presence of hemarthrosis following a traumatic injury can indicate the presence of a fracture or other significant anatomic disruption (such as an anterior cruciate ligament [ACL] tear of the knee.) The presence of fat globules confirms the presence of an intra-articular fracture as opposed to disruption of an intra-articular ligament. Drainage of a tense joint effusion can provide pain relief and improve range of motion and mobility. In hemophilia patients who develop acute hemarthrosis, aspiration of blood from the joint space following replacement of clotting factors can provide significant pain relief.9
Injection of a local anesthetic can relieve pain and facilitate procedures such as joint reduction, as well as facilitate a more accurate physical examination of a painful joint. The use intra-articular lidocaine has been well studied for joint reduction, particularly following glenohumeral dislocation, and has equal efficacy to intravenous (IV) sedation with a lower complication rate. Intra-articular morphine has been shown to reduce analgesic requirements following surgery, and may be of value in the treatment of acute knee pain.10,11,12
Corticosteroid injections into the synovial fluid are designed to maximize local benefits of the steroid while minimizing systemic effects. Corticosteroids have both anti-inflammatory and analgesic properties, and can provide significant relief and improved functional ability in patients with acute exacerbations of osteoarthritis. Complications such as steroid-induced arthropathy are rare, especially when injections into weight-bearing joints are limited to no more than three or four per year.13
Lyme Arthritis
Lyme disease is a disorder with multisystem manifestations. Lyme arthritis, a late disease manifestation, occurs months later and is accompanied by intense adaptive immune responses. It develops in approximately 60% of untreated Lyme patients. Following the initial tick bite, Borrelia burgdorferi spirochetes invade synovial joints, inducing an inflammatory response in synovial tissue consisting of synovial hypertrophy, vascular proliferation, and infiltration of mononuclear cells.14
Episodes of Lyme arthritis typically are brief and involve a single large joint. The knee is affected most commonly, with involvement in up to 90% of patients. Fever and other systemic symptoms usually are absent. Diagnosis of Lyme arthritis is based on characteristic clinical findings, history of exposure in an endemic area, and confirmatory serologic testing. On presentation, joint aspiration usually is performed for diagnostic purposes to rule out the presence of crystalline arthropathy or septic arthritis. Joint fluid white cell counts usually are in the range of 10,000 to 25,000, but cell counts from as low as 500 to as high as 100,000 have been reported.15
Contraindications and Complications
Arthrocentesis is relatively contraindicated when there is overlying skin or soft tissue infection. If possible, the provider should use an entry site that appears clear of obvious infection. The fear is that the procedure may introduce infection into an underlying joint that was not infected initially. For the same reasons, bacteremia and sepsis are considered relative contraindications, and ideally needles should not traverse skin lesions such as psoriasis or acne.
However, the risk of seeding a joint with bacteria after passing through possibly cellulitic tissue is mostly theoretical and is not backed up in the medical literature.16 Given the potential complications, including death, of undiagnosed septic arthritis, some authors advise diagnostic arthrocentesis despite the previous findings, and suggest those patients be admitted subsequently for IV antibiotics.9,17
The literature reports that the risk of iatrogenic infection is low overall but can occur. Estimates of risk are from 0.01% to 0.05%, with the greatest risk occurring in immunocompromised patients.4
Anticoagulation has been suggested as a relative contraindication to arthrocentesis, although little data exist regarding the actual risk of harm from bleeding. Proposed strategies include stopping anticoagulation several days prior to the procedure, reversing anticoagulation at the time of the procedure, or avoiding it altogether. Stopping or reversing the procedure puts patients at risk of a thromboembolic event or other complication.
Several studies have looked at performance of arthrocentesis in therapeutic and supratherapeutic patients and found little to no risk of periprocedural bleeding.9,18 A PubMed search by the authors did not find any studies looking specifically at bleeding risk associated with antiplatelet medications or the novel oral anticoagulants.
Other relative contraindications to arthrocentesis and intra-articular injection include adjacent osteomyelitis, impending joint replacement surgery, and uncontrolled diabetes if intraarticular steroids are to be administered. Osteonecrosis is a rare complication of steroid injections. Emergency physicians can avoid potential pitfalls by performing a good history and physical examination, explaining potential harms and benefits of joint aspiration or injection, and obtaining informed consent.19
General Techniques
The first step is to obtain informed consent from the patient or his or her caregiver. In general, assemble the necessary equipment on a bedside table or tray and position the patient at a comfortable height within reach of the tools. Ultimately, specific positioning depends on the joint being assessed.
Palpate the affected joint, identifying appropriate landmarks, and mark the needle insertion site. (See Table 2.) Alternatively, point-of-care ultrasound can be used to evaluate the joint, which will be discussed in more detail in later sections. Once the optimal site for needle insertion has been identified, proper sterile technique must be utilized and the site cleansed and draped. Sterile gloves should be worn. Allow the skin to dry after sterilization before injecting an anesthetic.
Table 2. Landmarks for Arthrocentesis
|
Joint |
Approach |
Key Landmarks |
|
Shoulder |
Posterior approach |
|
|
Anterior approach |
|
|
|
Elbow |
Lateral/radial approach or Posterolateral approach |
|
|
Wrist |
Dorsal approach |
|
|
Knee |
Parapatellar approach |
|
|
Suprapatellar approach |
|
|
|
Infrapatellar approach |
|
|
|
Ankle |
Anterolateral approach |
|
|
Anteromedial approach |
|
|
|
MCP/MTP/IP |
Dorsal approach |
|
Deep needle insertion during arthrocentesis causes significant pain in the majority of patients, but it can be mitigated effectively with local anesthesia use. There is concern that the bacteriostatic nature of lidocaine and bupivacaine may compromise culture results, and some authors advise against local anesthetic use, especially in assessment for periprosthetic joint infection.6,20,21 We believe that local anesthesia is appropriate in most cases, and recommend consulting with the orthopedic surgeon (if available) before aspirating joint fluid in all cases of painful joint arthroplasty.
Use either a 25- or 27-gauge needle to form a skin wheal around the needle insertion site using an appropriate anesthetic such as lidocaine 1% or bupivacaine 0.5%. Take care to avoid injecting local anesthetic directly into the joint space to prevent possible interference with synovial fluid analysis. A topical anesthetic or coolant can be applied to the skin as an alternate to local anesthetic as well, although they have only minimal effect on pain in deeper structures.20 In extremely painful joints, a regional nerve block may be appropriate. In rare instances, procedural sedation may be required in young or uncooperative patients.
In the majority of cases, aspiration of the viscous synovial fluid requires an 18-gauge or larger needle; however, in morbidly obese patients, a 21-gauge spinal needle may be necessary to reach the joint space. Although damage to the articular cartilage is unlikely, halting the advancement of the needle once fluid is aspirated can reduce risk of striking the bone or cartilage. If the syringe must be changed during the procedure, hemostats should be used to firmly grasp and stabilize the needle hub while the syringe is exchanged.22
Use of Ultrasound
Since 1895, when Wilhelm Rontgen discovered the principles of medical X-rays, physicians have been using diagnostic imaging to visualize structures deep to the skin. Despite the advent of new technological modalities such as MRI and ultrasound, X-ray imaging remains the standard initial, and sometimes only, study used to evaluate musculoskeletal problems. Although MRI is considered the best imaging modality for evaluation of the muscles, ligaments, and other soft tissues, its cost and availability restrict its use. Ultrasonography is becoming more cost-effective, portable, and available in multiple clinical settings. One critical advantage of ultrasound is that it allows a dynamic examination of the musculoskeletal system.23
Ultrasound has been used widely to evaluate arthropathies in multiple different joints. In the emergency department, bedside ultrasound can be used to identify the presence of clinically significant joint effusions and potentially help avoid unnecessary procedures. If there is doubt whether there is a significant effusion, comparison views of the contralateral joint can be helpful.
Often, multiple views of the affected joint are obtained to identify the optimal site for needle insertion. Additionally, ultrasound can be used to guide the needle directly during arthrocentesis, reducing the number of attempts needed to aspirate synovial fluid.24
Ultrasound has other applications in the diagnosis and management of musculoskeletal disorders such as identification of bone fractures; evaluation of the bursa, tendons, ligaments, muscles, and other soft tissue; as well as identification of nerve morphology and the guidance of nerve blocks. Ultrasound reportedly has been used to identify monosodium urate and calcium pyrophosphate crystals in the setting of gout and pseudogout.25,26
There are no absolute contraindications to the use of ultrasound for joint assessment or arthrocentesis, and relative contraindications are similar to those of the blind procedure. Compared to the blind technique, the use of ultrasound-guided arthrocentesis by emergency physicians may be less time consuming, less technically difficult, and less painful for the patient.27
Most commonly, the high frequency linear array transducer with the musculoskeletal exam type is used to evaluate the joints. Maintain a proper sterile technique throughout the procedure, including a sterile cover over the ultrasound transducer. Once the effusion and needle insertion site are identified, the ultrasound can be used for real-time guidance of the needle tip into the synovial space. It also is reasonable to use ultrasound simply to mark the location of the effusion and optimal entry site, and then perform the procedure blindly.
For direct needle guidance, insert the needle into the skin adjacent to the ultrasound transducer and slowly advance both the probe and the needle, maintaining visualization of the needle tip until entry into the synovial space. The needle will appear brightly hyperechoic in comparison to the surrounding tissue.
The appearance of the fluid on ultrasound can differ depending on the nature of the fluid. Simple fluid, such as in a non-inflammatory effusion, will appear anechoic, or dark black, within the joint space. Complex fluid, such as in a traumatic effusion or an infected joint with purulent debris, will be heterogeneous in appearance, a mixture of black/gray fluid with gray or even white hyperechoic particles within its contents. Bone will appear as bright white, or hyperechoic, while the tendons and soft tissue will appear more gray or hypoechoic when compared to the bone.21
Shoulder Arthrocentesis
Posterior Approach
The accuracy of needle placement and successful shoulder arthrocentesis is reportedly higher when utilizing the posterior approach compared to the anterior approach.28 Place the patient in a comfortable seated position with the hand of the affected limb held across the body and placed on the opposite shoulder. Palpate the posterior tip of the acromion and palpate the joint space typically 1-2 cm inferior and medial to the tip of acromion.
Anesthetize the soft tissue and mark the optimal needle insertion site. Use a 20-gauge or larger needle for aspiration attached to at least a 10 cc syringe. The needle should be inserted 1-2 cm inferior to the posterior end of the acromion just medial to the humeral head and directed in a horizontal plane anteriorly and medially toward the coracoid. Continue to aspirate as you advance the needle until synovial fluid is obtained. When the procedure is complete, withdraw the needle and apply slight pressure and a bandage to the entry site.29
Anterior Approach
For the anterior approach, place the patient in a comfortable seated position with the hand of the affected limb resting palm down on his or her lap, which will keep the shoulder internally rotated. Palpate the coracoid process and the humeral head. With the limb internally rotated, the joint space can be felt as a groove lateral to the coracoid process. Use an 18-gauge or larger needle attached to at least a 10 cc syringe for the procedure.
Insert the needle medial to the humeral head just inferior and lateral to the coracoid process and direct it posteriorly into the glenohumeral joint space. Continue to aspirate until synovial fluid is obtained. When the procedure is complete, withdraw the needle and apply slight pressure and a bandage to the entry site.29
Bedside Ultrasound for Shoulder Arthrocentesis
Ultrasound can be used to evaluate the shoulder from either the anterior or posterior location; the anterior view often is easier for visualization while the posterior approach often is preferred for aspiration.
For the anterior view, place the patient in a comfortable seated position with the elbow held against the patient’s body and the hand on the affected side in supination, with the palm upward and the back of the hand resting on the thigh. Align the probe in a transverse plane, with the probe marker toward the patient’s right, over the biceps groove and identify the biceps tendon. The synovial sheath of the long head of the biceps tendon communicates with the glenohumeral joint, so fluid often is seen tracking into the biceps tendon sheath. (See Figure 1.) Ultrasound is very sensitive for the detection of fluid, and a small amount of physiologic fluid often is seen. However, excess fluid, especially fluid noted circumferentially around the biceps tendon, is abnormal.
Figure 1. Shoulder Ultrasound: Site of Potential Effusion (Gray Arrow)
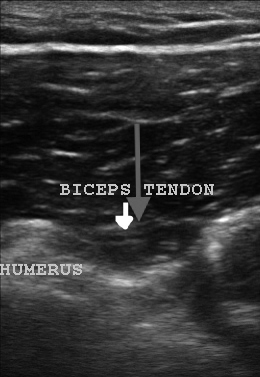
Insert the needle at a steep angle aligned with the center of the transducer but out-of-plane of the transducer beam. Maintain visualization of the needle tip until it enters the center of the effusion around the biceps tendon, at which point aspiration is performed.
For the posterior view, place the patient either prone or seated but leaning forward with the hand of the affected limb grasping the contralateral shoulder. Place the transducer posteriorly over the head of the humerus with the probe marker toward the lateral side. Identify the glenohumeral joint space beneath the infraspinatus muscle and the glenoid labrum with the hyperechoic humeral head as the bony landmark.
Insert the needle just proximal to the transducer opposite the probe marker. Slowly advance the needle, which will appear as a bright hyperechoic linear structure, to lie in plane to the transducer and advance under direct visualization until the tip of the needle is seen within the joint space and fluid can be aspirated. When the procedure is complete, withdraw the needle and apply slight pressure and a bandage to the entry site.21
Knee Arthrocentesis
Position the patient supine in the bed at a comfortable height with a towel roll below the affected knee to flex the joint about 20 degrees, keeping the foot flat on the bed perpendicular to the floor. Knee arthrocentesis can be performed from a parapatellar, suprapatellar, or infrapatellar approach, with parapatellar being preferred. Each approach can be performed from either the medial or lateral side. If the patient is unable to flex the knee and the procedure must be done with the knee in full extension, then a medial approach often is easier.
Maintain proper sterile technique throughout the procedure and anesthetize with local anesthetic to facilitate procedure and minimize discomfort. Knee arthrocentesis often requires at least an 18-gauge needle for fluid aspiration and a large syringe such as a 20 cc or 30 cc; however, at times larger (e.g., 60 cc) or multiple syringes may be needed with large effusions.
For the parapatellar approach, identify the tibial plateau and the midpoint of either the medial or lateral border of the patella. Slightly sublux the patella toward the side of entry and insert the needle perpendicular to the skin roughly 0.5 cm beneath the patellar border above the tibial plateau. Direct the needle perpendicular to the long axis of the femur parallel to the bed. Continue to aspirate until synovial fluid is obtained.
For the suprapatellar approach, first identify the superomedial or superolateral border of the patella. Insert the needle just inferior to the patellar border, directing the tip of the needle toward the intercondylar notch of the femur. (See Figure 2.) In this approach, the needle actually inserts into the suprapatellar bursa, which openly communicates with the knee joint. One disadvantage of the suprapatellar approach is that in up to 10% of patients, the bursa does not communicate with the joint space, therefore yielding inaccurate results.
Figure 2. Knee Arthrocentesis from the Suprapatellar Lateral Approach
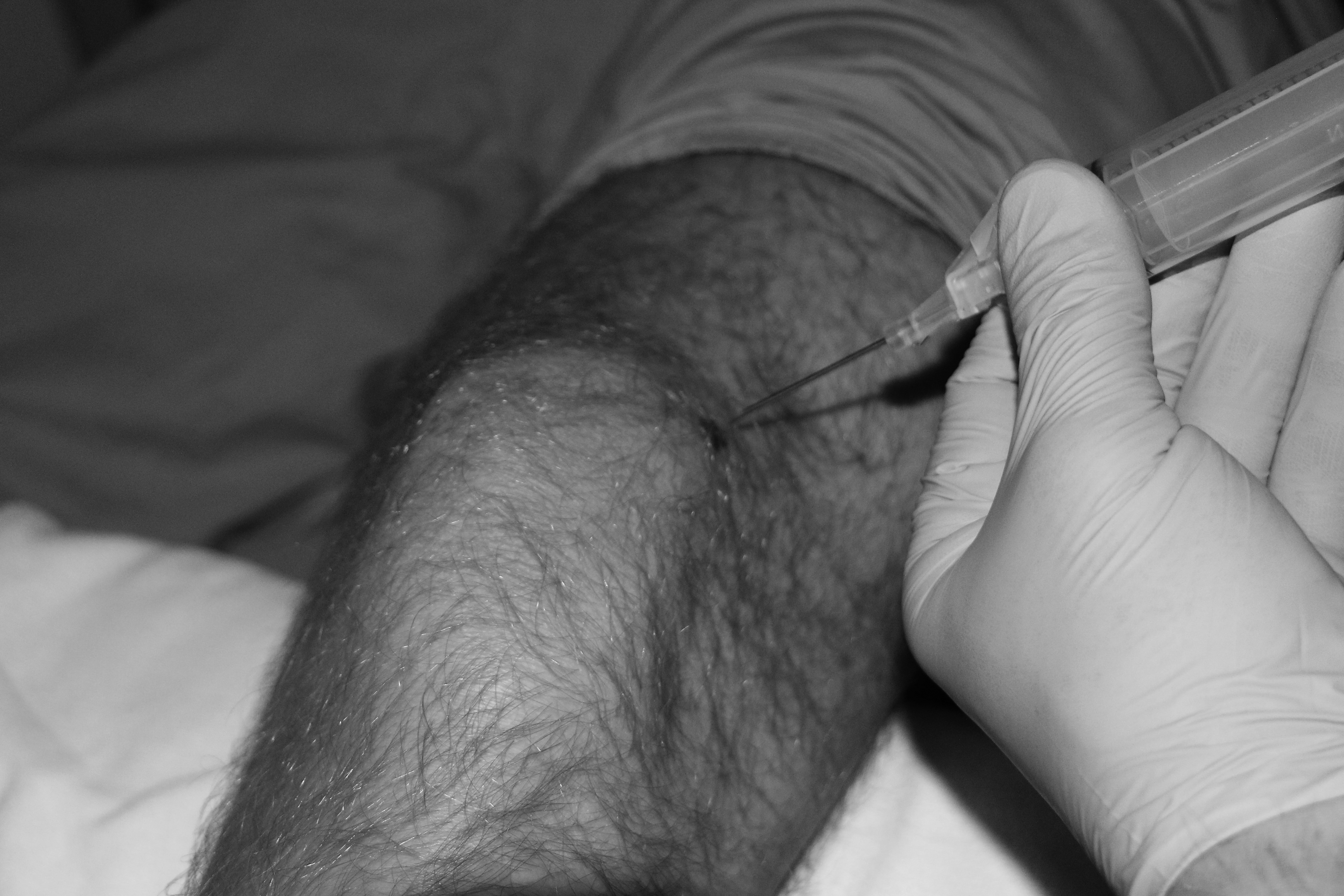
Needle being inserted in the superolateral border and directed toward the intercondylar notch of the femur.
The infrapatellar approach requires the patient to be seated upright with the knee bent 90 degrees. Identify the inferior border of the patella and the patellar tendon on either the medial or lateral side and insert the needle below the border of the patella lateral to the edge of the patellar tendon. Care must be taken to avoid puncturing the patellar tendon. While performing knee arthrocentesis, pressure can be applied to the suprapatellar bursa in an attempt to milk the fluid if flow stops. When the procedure is complete, withdraw the needle and apply slight pressure and a bandage to the entry site.29
Bedside Ultrasound for Knee Arthrocentesis
To perform ultrasound-guided arthrocentesis of the knee via the suprapatellar approach, position the patient similarly to the blind procedure with the knee flexed to about 20 degrees and place a towel roll behind the knee. Begin with the probe in a longitudinal axis with the probe marker toward the patient’s head. Identify the quadriceps femoris muscle and tendon and the hyperechoic patella, which should be on the right side of the screen. Look for fluid in the suprapatellar bursa, which is just below the quadriceps tendon. (See Figure 3.) The deepest landmark is the femur, which should appear as a linear hyperechoic structure similar to the patella.
Figure 3. Knee Ultrasound: Arrow Indicates Location of Joint Space
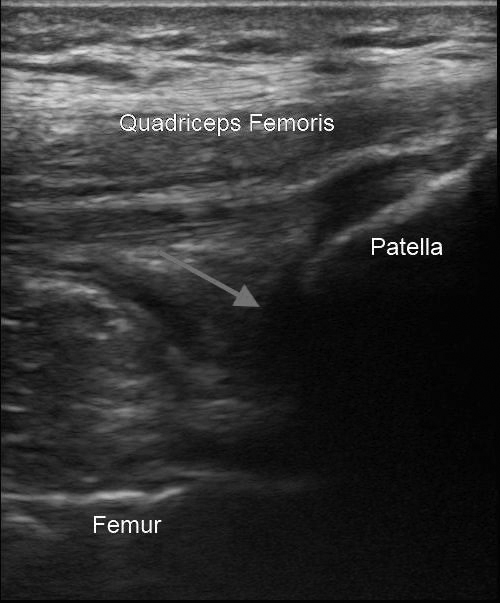
Next, rotate the probe 90 degrees, keeping the suprapatellar bursa centered on the screen. The needle can be inserted from either the medial or lateral aspect, with the lateral approach reportedly the preferred method. On the lateral approach, insert the needle just proximal to the transducer in between the vastus lateralis and the iliotibial band and slowly advance, maintaining the hyperechoic linear appearance of the needle in plane with the acoustic window. Advance until the tip of the needle is identified within the bursa, at which point aspiration is completed. When the procedure is complete, withdraw the needle and apply slight pressure and a bandage to the entry site.29,30
Ankle Arthrocentesis
Position the patient either seated or supine with the knee flexed at 90 degrees, the ankle plantar flexed, and the heel resting flat against the bed perpendicular to the floor. The ankle joint space can be entered from an anterolateral or anteromedial approach. The anterolateral approach is preferred, as it reduces risk of injury to the dorsalis pedis and posterior tibial blood vessels as well as the deep peroneal nerve, all of which travel through the medial aspect of the ankle. Ankle arthrocentesis typically requires an 18-gauge needle and at least a 10 cc syringe.
Anterolateral Approach
For the anterolateral approach, identify the ankle joint line, the lateral malleolus, and lateral border of the extensor digitorum longus. Having the patient dorsiflex the ankle against some resistance or actively flexing and extending the ankle can help identify these landmarks more consistently. Insert the needle in the space between the base of the lateral malleolus and the lateral border of the extensor digitorum longus at the joint line, directing the needle perpendicular to the shaft of the fibula. (See Figure 4.)
Figure 4. Arthrocentesis of the Ankle Via the Anterolateral Approach
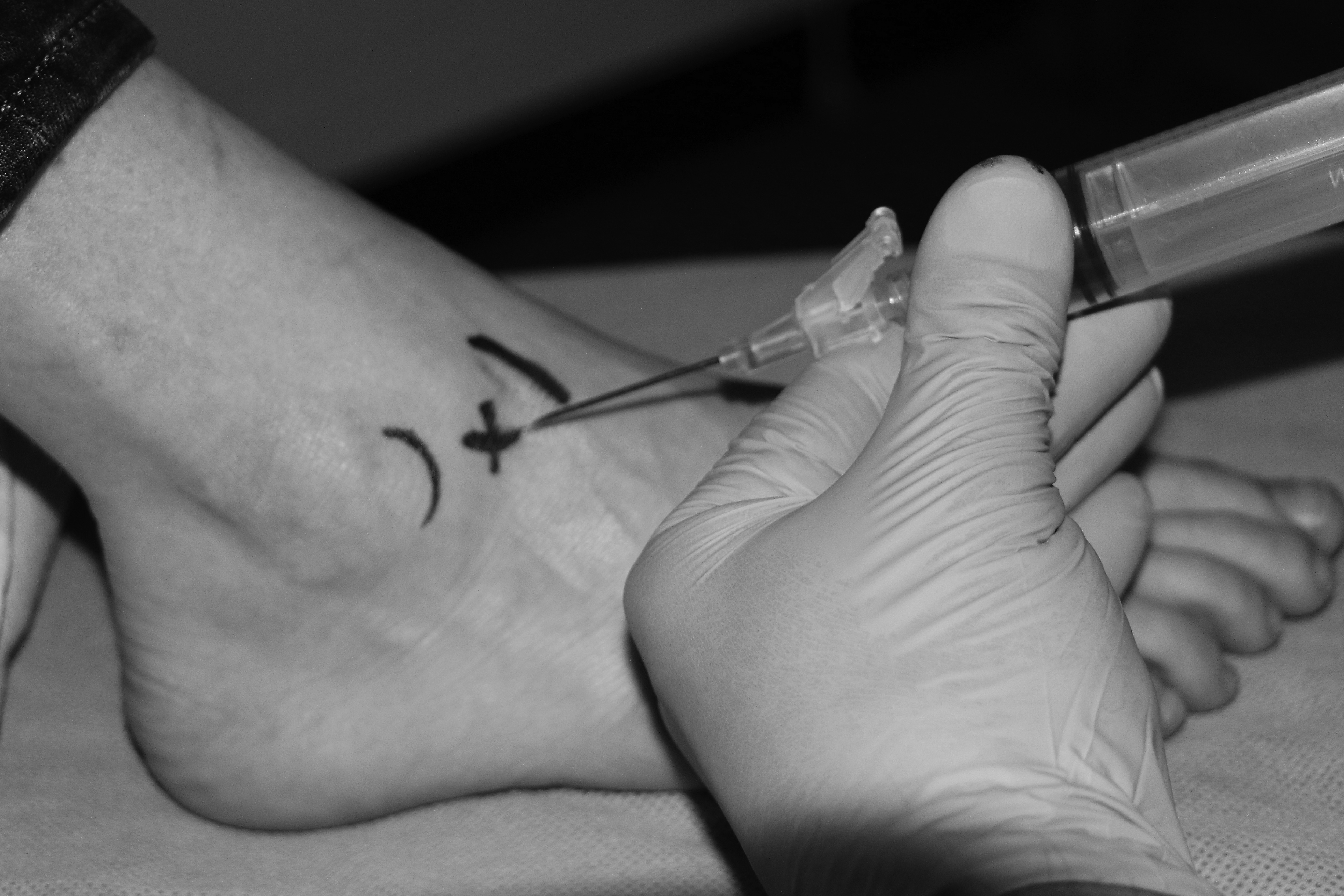
The joint space is located in between the borders of the lateral malleolus and the lateral border of the extensor digitorum longus.
Anteromedial Approach
For the anteromedial approach, take care to avoid injury to the nerve and vessels that are directly below and lateral to the extensor hallucis longus tendon. Identify the ankle joint line, the medial malleolus, and the extensor hallucis longus and tibialis anterior tendons. Having the patient dorsiflex the ankle against resistance or actively flexing and extending the ankle can help identify these landmarks more consistently.
Palpate the space either between the medial border of the extensor hallucis longus and the lateral border of the tibialis anterior tendon or between the base of the medial malleolus and the medial border of the tibialis anterior tendon. Insert the needle in either space, directing the needle perpendicular to the shaft of the tibia. Continue to aspirate as you advance the needle until synovial fluid is obtained. When the procedure is complete, withdraw the needle and apply slight pressure and a bandage to the entry site.29
Bedside Ultrasound for Ankle Arthrocentesis
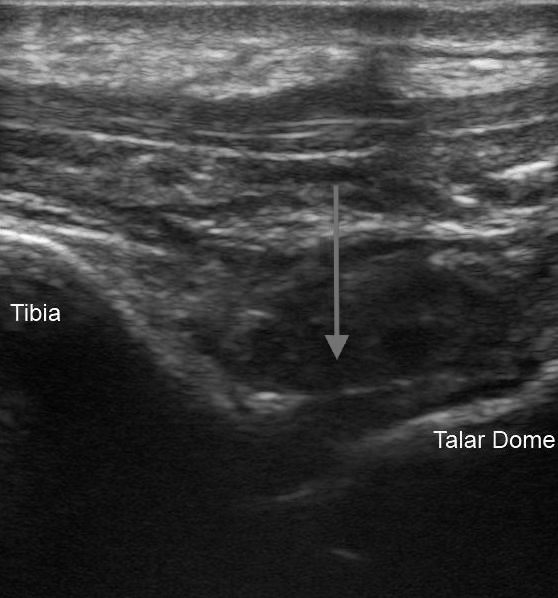
For ultrasound-guided arthrocentesis of the ankle, position the patient so that the foot can be plantar flexed and stable against the bed. Ultrasound has been reported as able to identify as little as 2 cc of fluid in the ankle21 with the most sensitive view over the anterior recess of the tibiotalar joint. Align the transducer in a longitudinal plane, with the probe marker aimed cephalad, over the distal anterior tibia to identify this hyperechoic bony landmark and then slide distally toward the foot until the joint space between the distal tibia and the talar dome is identified. An effusion will appear in the recess between the tibia and the talar dome. (See Figure 5.)
Insert the needle from either the medial or lateral side at a steep angle aligned with the center of the transducer but out-of-plane of the transducer beam. Whichever approach is taken, take care to avoid the extensor hallucis longus and tibialis anterior tendons. Maintain visualization of the needle tip until it enters the center of the effusion, at which point aspiration is performed.21,31,32
Figure 5. Ankle Ultrasound: Arrow Indicates Location of Joint Space
Wrist Arthrocentesis
Place the patient in a seated, reclined or recumbent position, whichever is most comfortable, with the wrist easily accessible and braced on the bed, table, or tray. It may be helpful for an assistant to hold the hand of the affected limb and apply slight traction and ulnar deviation of the wrist, opening the joint space. Hanging finger traps also can be used if an assistant is unavailable. Slight volar flexion also can help facilitate needle insertion into the joint space.
When performing arthrocentesis of the wrist, a 22-gauge needle can be used; however, in some cases a larger needle may be required for more viscous synovial fluid. A 5-10 cc syringe often is sufficient for fluid aspiration. The skin and subcutaneous tissue can be infiltrated with local anesthetic; however, in some cases a topical anesthetic can be used or anesthetic may be avoided altogether. Patients may find a single needle insertion for the arthrocentesis less painful than the administration of local anesthetic.
On the dorsal surface of the wrist, palpate Lister’s tubercle, the bony prominence found in the center of the distal end of the radius. Next identify the extensor pollicis longus tendon, which runs on the radial side of Lister’s tubercle and makes up the ulnar border of the anatomic snuffbox. Insert the needle perpendicular to the wrist on the ulnar side of the extensor pollicis longus tendon and just distal to the distal radius. (See Figure 6.) If bone is struck, withdraw the needle slightly and redirect slightly more distally. Continue to aspirate as you advance the needle until synovial fluid is obtained. When the procedure is complete, withdraw the needle and apply slight pressure and a bandage to the entry site.29
Figure 6. Arthrocentesis of the Wrist Via the Dorsal Approach
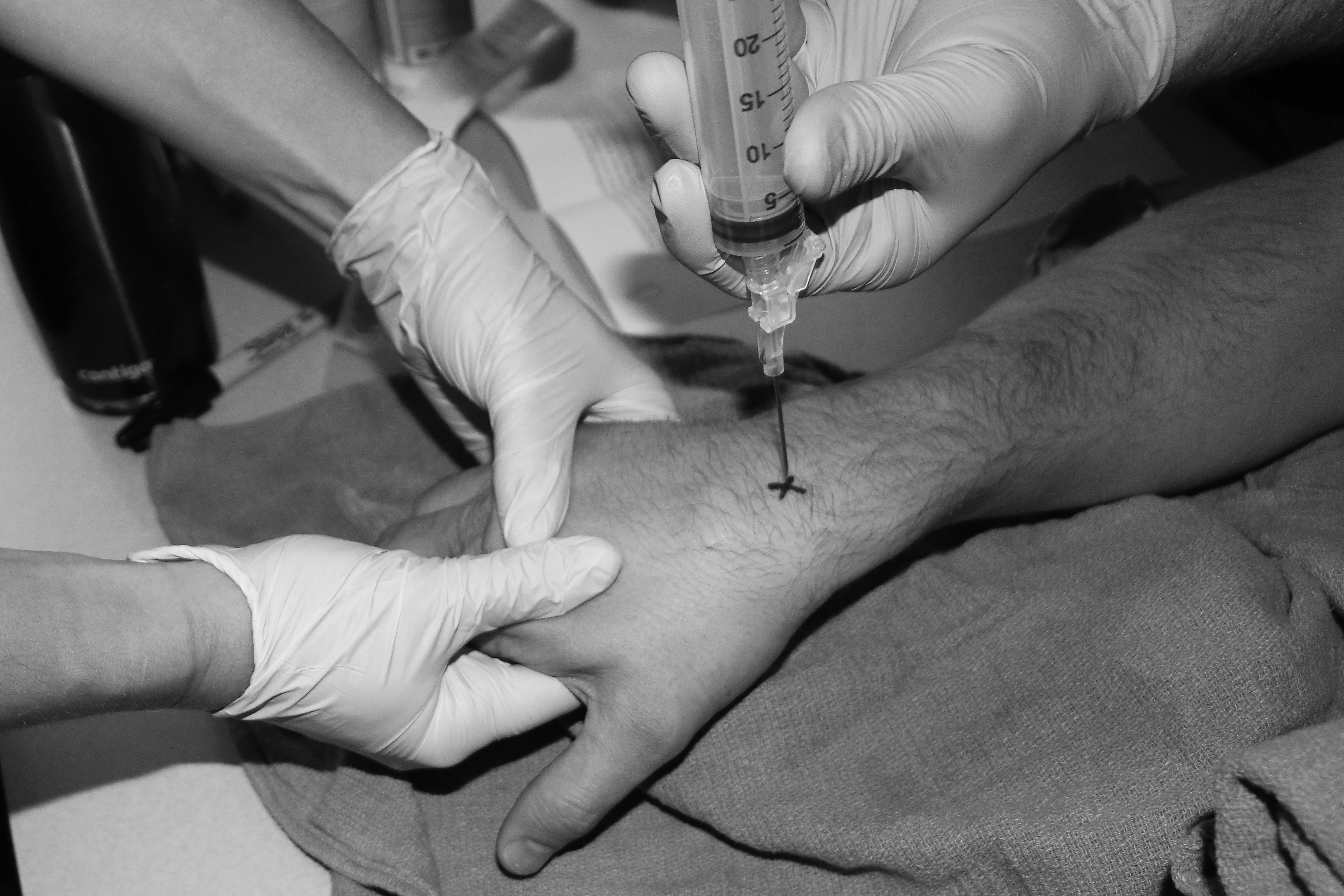
An assistant applies slight traction and ulnar deviation to facilitate opening of the joint space.
Bedside Ultrasound for Wrist Arthrocentesis
For ultrasound-guided arthrocentesis of the wrist, position the patient similar to when the procedure is performed blindly. Place the linear transducer in a longitudinal axis on the dorsal surface of the distal radius with probe marker pointed cephalad. It may be helpful for an assistant to hold the hand of the affected limb and apply slight traction and ulnar deviation of the wrist, opening the joint space. Hanging finger traps also can be used if an assistant is unavailable. Applying volar flexion also can help facilitate needle insertion into the joint space.
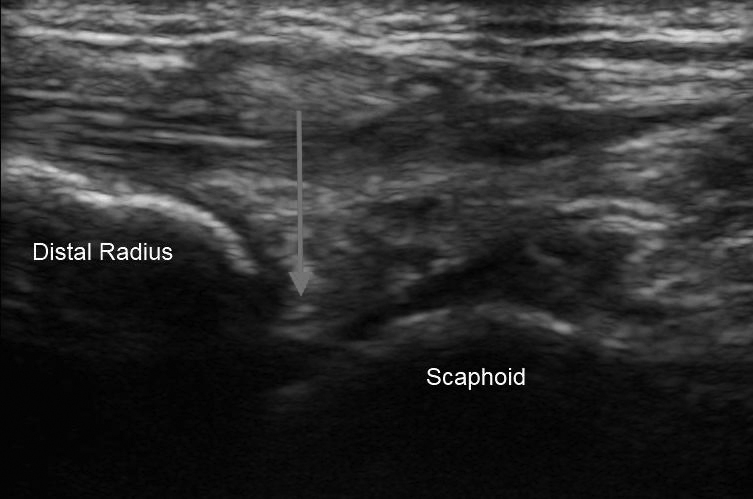
Slide the probe until the distal radius and the carpal bone are identified. The effusion will appear as a hypoechoic region between the distal radius and scaphoid. (See Figure 7.) Insert the needle on the ulnar side of the extensor pollicis longus adjacent to the probe at a steep angle aligned with the center but out-of-plane with the transducer beam. Maintain visualization of the needle tip until it enters the center of the effusion, at which point aspiration is performed. Alternatively, the transducer can be rotated 90 degrees over the joint space and a similar out-of-plane approach can be performed, again inserting the needle on the ulnar side of the extensor pollicis longus.21
Figure 7. Wrist Ultrasound: Arrow Indicates Location of Joint Space
Elbow Arthrocentesis
The patient should be seated upright with his or her back against the stretcher or chair with the arm abducted. Bend the patient’s elbow to 90 degrees and pronate the forearm to place the palm down on a table or tray at a height of greatest comfort.
Maintain proper sterile technique throughout the procedure. The skin and subcutaneous tissue may be infiltrated with local anesthetic. For elbow arthrocentesis, an 18- or 20-gauge needle should be used and attached to a 5-10 cc syringe.
Palpate the bony prominences of the elbow and identify the olecranon process, lateral epicondyle, and the head of the radius. Visualize a triangle formed by these three structures and palpate the depression at the center of the triangle. It may help to extend the elbow and palpate the depression between the radial head and lateral epicondyle, then maintain this location while flexing the elbow. If a large effusion is present, this may be palpated as a bulge in the skin rather than a depression. In the lateral, or radial, approach, insert the needle from the lateral side perpendicular to the skin and humeral head. (See Figure 8.) Continue to aspirate as you advance the needle until synovial fluid is obtained. Avoid a medial, or ulnar, approach, as it incurs a greater risk of injury to the ulnar nerve and superior ulnar collateral artery. A posterolateral approach can be used if the effusion is palpated inferior to the lateral epicondyle; however, there is greater risk of injury to the radial nerve and triceps tendon. In this approach, the position and identified landmarks are similar, but the needle is inserted from the posterior elbow perpendicular to the skin parallel to the shaft of the radius. When the procedure is complete, withdraw the needle and apply slight pressure and a bandage to the entry site.29
Figure 8. Arthrocentesis of the Elbow
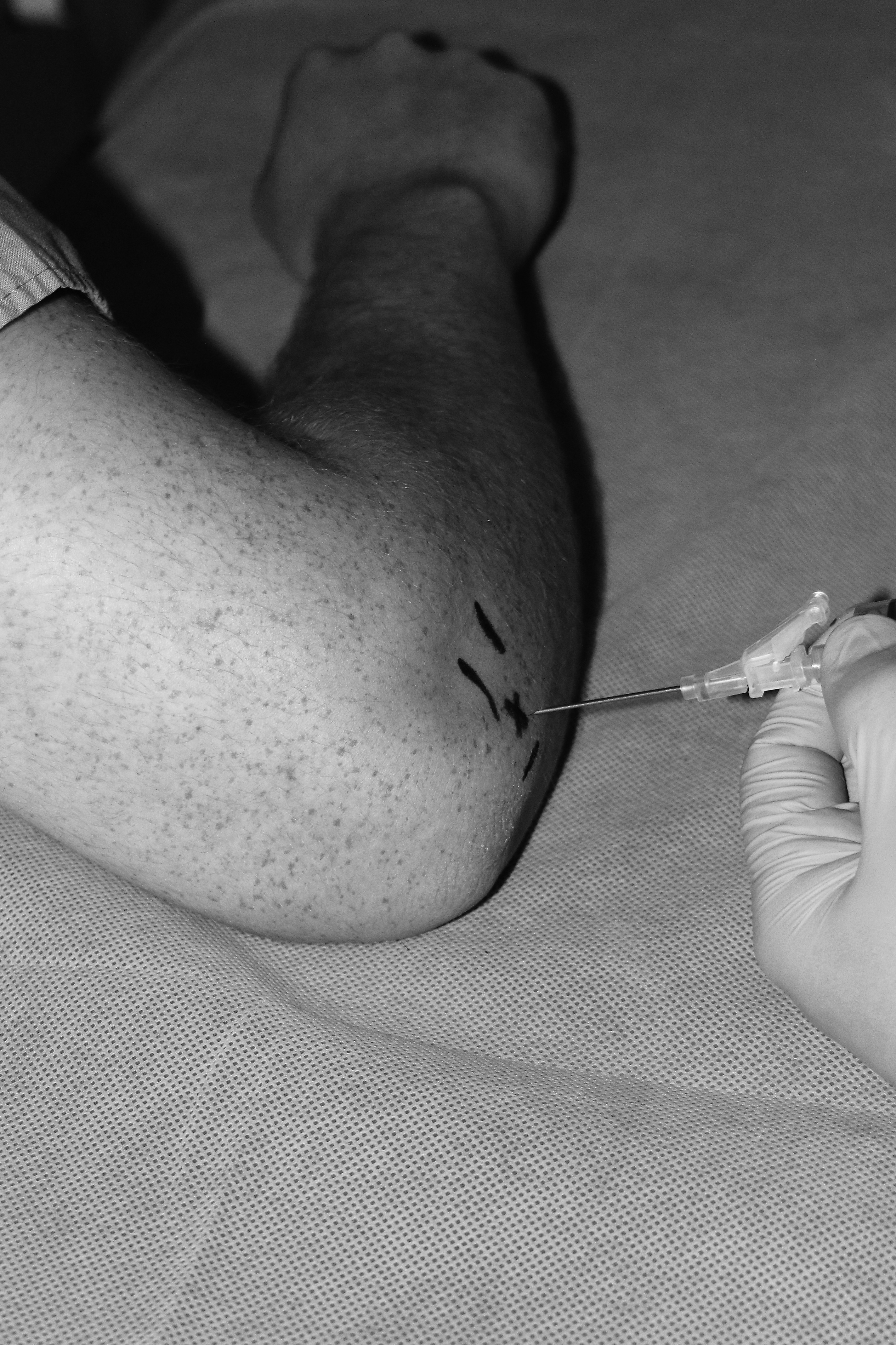
Visualize a triangle formed by the olecranon process (inferior line), the lateral epicondyle (proximal line), and the head of the radius (distal line). Palpate the depression at the center of the triangle and insert the needle.
Bedside Ultrasound for Elbow Arthrocentesis
Ultrasound-guided arthrocentesis of the elbow typically is performed from the lateral approach with the patient in a seated position. Abduct the affected arm and bend the elbow to 90 degrees, pronating the forearm to place the palm down on a table or tray at a height of greatest comfort. Place the transducer in a longitudinal view over the proximal forearm parallel to the radius with the probe marker cephalad. Scan proximally until the head of the radius and the capitellum with which it articulates are identified. Also, identify the extensor tendon, which lies superficial to the radio-capitellar joint and must be avoided when inserting the needle. An elbow effusion will appear as a hypoechoic area between the capitellum and radial head. (See Figure 9.)
Figure 9. Elbow Ultrasound: Arrow Indicates Location of Joint Space
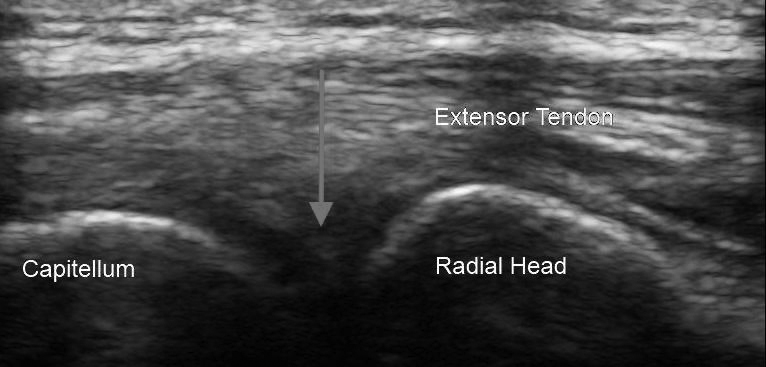
Insert the needle from the lateral side at a steep angle aligned with the center of the transducer but out-of-plane of the transducer beam. Maintain visualization of the needle tip until it enters the center of the effusion, at which point aspiration is performed. When the procedure is complete, withdraw the needle and apply slight pressure and a bandage to the entry site. A posterior approach for ultrasound-guided arthrocentesis of the elbow also has been described as an alternative approach.21,33
Arthrocentesis of the Metatarsophalangeal, Metacarpophalangeal, and Interphalangeal Joints
Arthrocentesis of the small joints in the hands or feet is performed from the dorsal side. When performing the procedure on the hand, place the patient in a comfortable seated position with the affected hand and forearm pronated and the palm flat on a table or stand. For the foot, place the patient in either a seated or supine position on the bed with knees bent and the affected foot plantar flexed and the sole flat on the bed.
The skin and subcutaneous tissue can be infiltrated with local anesthetic; however, in some cases the patient may prefer a topical anesthetic or avoidance of anesthesia with a single needle insertion for the aspiration. Because of the relatively small joint space, a 21-gauge needle attached to a 5 cc syringe is typically sufficient for aspiration.
Hold the affected joint in slight flexion to about 20 degrees and palpate the joint line. It is imperative to identify the extensor tendon and avoid puncturing this structure during the procedure. If swelling obscures tendon visualization, have the patient extend the digit against resistance to improve visualization of the tendon. Gentle traction of the affected digit can be used to widen the joint space and improve needle insertion.
Insert the needle perpendicular to the skin on either side of the extensor tendon but avoiding the neurovascular bundle running along either side of the digit. Direct the needle toward the midline of the joint and aspirate during insertion until fluid is obtained. When the procedure is complete, withdraw the needle and apply slight pressure and a bandage to the entry site.29
Bedside Ultrasound for Metatarsophalangeal, Metacarpophalangeal, and Interphalangeal Arthrocentesis
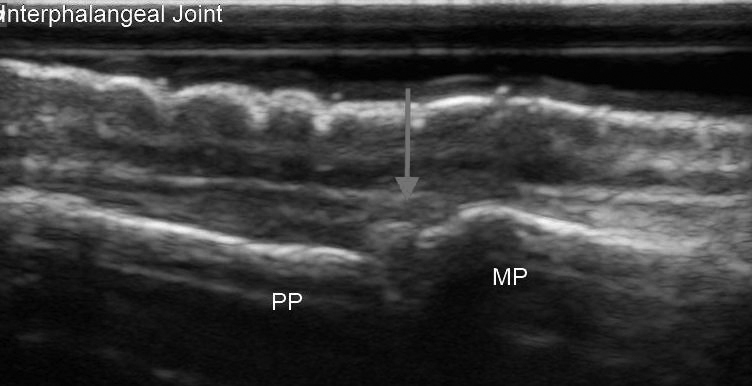
Ultrasound evaluation of the metatarsophalangeal, metacarpophalangeal, and interphalangeal joints can be performed using the high frequency linear array probe, although a specialized small footprint transducer can be used if available. When evaluating the superficial structures such as the joints of the digits, a water bath can be used to better delineate landmarks and identify an effusion. A water bath replaces ultrasound gel and can avoid contact between the ultrasound transducer and the patient’s skin. Ultrasound often is used to identify and evaluate for joint effusions (see Figures 10 and 11), but given the relatively small joint size and narrow windows, the procedure rarely is performed under direct real-time ultrasound guidance.34
Figure 10. Distal Interphalangeal Joint Ultrasound: Arrow Indicates Location of Joint Space
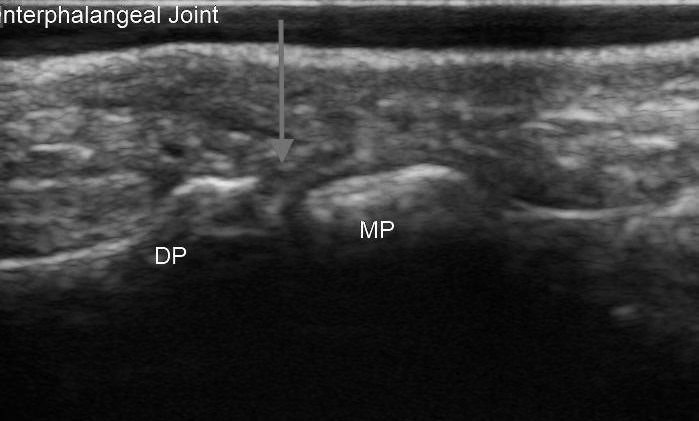
Figure 11. Proximal Interphalangeal Joint Ultrasound: Arrow Indicates Location of Joint Space
Interpretation of Results
The diagnosis of septic arthritis, as well as the diagnosis of gout, pseudogout, or post-traumatic effusion, is made based on a combination of historical, clinical, imaging, and laboratory findings. Given the scope of this review, we are going to focus specifically on the analysis of synovial fluid and how that can help narrow the differential and provide a definitive diagnosis. However, two questions that often arise when a patient presents with acute monoarticular arthritis are: 1) Can the emergency physician rule out a septic joint based on laboratory findings alone? 2) Is imaging useful in the evaluation of a suspected septic joint?
Serum Studies
Multiple studies have looked at the peripheral white blood cell (WBC) count for diagnosis of septic arthritis, using multiple cutoff thresholds. Sensitivities ranged from as low as 19% to as high as 77%, lacking acceptable diagnostic accuracy.
Erythrocyte sedimentation rate (ESR) has been found to be more sensitive than peripheral WBC when using cutoff values of > 30 mm/hr, reporting a sensitivity of about 90%. However, findings varied markedly between trials. ESR also is of limited utility because of its poor specificity.
Although serum CRP is significantly more elevated in septic arthritis than other inflammatory joint disorders (like gout), it lacks sufficient sensitivity or specificity to increase or decrease the post-test probability of septic arthritis.
Combining WBC, ESR, and synovial WBC counts significantly enhances sensitivity, although the combined specificity is low. Of the three, joint fluid WBC appears to be the best test for the initial diagnosis, although even it may be rated only as fair when likelihood ratios are calculated.4,35,36
When there is suspicion of an infected prosthetic joint, serum markers may be of greater utility. Especially in the first six weeks postoperatively, differentiation of a superficial skin infection from a periprosthetic joint infection is facilitated by the CRP, with an optimal cutoff of 100 mg/L. Patients with a CRP at or very near this level should have arthrocentesis performed. When the CRP is abnormal (> 10 mg/L) but not approaching the threshold, consideration can be given to close observation or a course of antibiotics to treat obvious cellulitis. The approach to postoperative complications should be determined in consultation with the patient’s orthopedic surgeon.6
Imaging
Radiographic imaging of septic arthritis has low sensitivity, especially for changes in the early stages of disease. The presence of soft-tissue swelling may represent underlying synovial inflammation and joint effusion. A joint effusion may be noted on radiographs of the elbow, knee, and ankle. In more advanced disease, cartilage destruction may cause joint space narrowing, and bone erosion may become evident at the joint margins where the capsule attaches and the cartilage is thinnest.5
CT scanning and MRI are of relatively low utility in the initial evaluation of a septic joint, especially when joint aspiration will be performed. They are of greater use when looking for osteomyelitis or an abscess, with an edge given to MRI because of its greater soft tissue detail. These tests may be helpful in patients with sacroiliac or sternoclavicular joint infection, helping look for extension into the mediastinum or pelvis.37 MRI features of septic arthritis include joint effusion, joint-space narrowing, and bone marrow edema surrounding the joint, starting in the marginal areas.5
Synovial Fluid Analysis
Synovial fluid should be inspected by the emergency physician for color, clarity, viscosity, and presence of abnormal inclusions. Normal synovial fluid is straw-colored and clear enough to read newsprint through. An elevated synovial fluid WBC count will result in a specimen that is more opaque. Bloody fluid may be seen in fracture or ligamentous disruption; fat globules may be seen in fracture.33 When performed by an experienced examiner, gross inspection alone is 94% sensitive for differentiating inflammatory from noninflammatory causes of acute arthritis.4 Following gross inspection, fluid should be sent to the laboratory for cell count with differential, Gram stain, culture, and crystal analysis. (See Table 3.)
Table 3. Synovial Fluid4,9,35,36,40,41,42
|
Condition |
Color |
Clarity |
Viscosity |
WBC/mm 3 |
PMN |
Crystal |
Gram Stain |
Culture |
|
PMN = Polymorphonuclear leukocytes *Routine bacterial culture techniques often do not detect Borrelia burgdorferi. † Should demonstrate a predominance of red blood cells |
||||||||
|
Normal |
Yellow |
Translucent |
High |
< 200 |
< 25% |
Negative |
Negative |
Negative |
|
Non-inflammatory |
Straw/yellow |
Translucent |
High |
200-2,000 |
< 25% |
Negative |
Negative |
Negative |
|
Inflammatory |
||||||||
|
Rheumatoid Arthritis |
Yellow to green |
Translucent to turbid |
Low |
3,000-50,000 |
> 50% |
Negative |
Negative |
Negative |
|
Gout |
Yellow to white |
Translucent to opaque |
Low |
2,000-50,000 |
> 50% |
Negative birefringence needle-shaped |
Negative |
Negative |
|
Pseudogout |
Yellow to white |
Translucent to opaque |
Low |
2,000-50,000 |
> 50% |
Positive birefringence rhomboid-shaped |
Negative |
Negative |
|
Septic |
||||||||
|
Bacterial |
Yellow to white |
Cloudy to opaque |
Low |
50,000-200,000 |
> 90% |
Negative |
Positive |
Positive |
|
Spirochetal (e.g. Lyme) |
Yellow to white |
Yellow/cloudy |
Low |
3,000-100,000 (mean 25,000) |
> 50% |
Negative |
Negative |
Negative* |
|
Hemorrhagic |
Pink to red |
Translucent to bloody |
Variable |
200-10,000† |
25-75% |
Negative |
Negative |
Negative |
In gout, monosodium urate crystals appear as needle-shaped intracellular and extracellular crystals. Negatively birefringent urate crystals are seen on polarizing examination in up to 85% of specimens. This means that when examined with polarizing and red filters, they appear yellow when aligned parallel to the slow axis of the red compensator and turn blue when aligned across the direction of polarization.
Pseudogout shows calcium pyrophosphate crystals, which are shorter than monosodium urate crystals and often are rhomboid-shaped. Under a polarizing filter, they display weak positive birefringence, appearing blue when aligned parallel with the slow axis of the compensator and yellow when perpendicular.
Even when asymptomatic or “intercritical,” patients with a history of diagnosed or clinically suspected gout are likely to have monosodium urate crystals present in their synovial fluid. During acute gout attacks, synovial WBC may be elevated, with the number ranging from as low as 1,000 to as high as 100,000.7,38
As mentioned earlier, there are no reliable clinical cues that differentiate crystal arthropathy from a septic joint. Lab findings are variable in each and they occasionally may coexist. Therefore, a Gram stain and culture should be performed in all cases of crystal-induced arthritis, and the patient’s full clinical picture, including symptoms, risk factors, and examination, should be considered prior to ruling out septic arthritis.8
Joint fluid WBC count traditionally is used to differentiate between inflammatory, noninflammatory, and septic causes of acute arthritis. (See Table 3.) However, the ranges cited in the literature are highly variable, with significant overlap seen within the cutoffs. Typical guidelines suggest the following interpretations of synovial WBC count: < 200 is normal; 200-2,000 noninflammatory; 2,000-50,000 inflammatory; and > 50,000 infectious, although the upper limit cutoff in septic arthritis has not been clearly established. Relatively few trials have looked at both the sensitivity and specificity of the synovial WBC count; heterogeneous findings among those trials are present because of variation in the population studied, interval between disease onset and testing, equipment used to quantify cell counts, and the gold standard used. Neither the percentage of polymorphonuclear cells, synovial glucose levels, or synovial protein levels increase or decrease the probability of septic arthritis.4,9,35,36
Gram stain sensitivity estimates range from 29% to 75%, and specificity has not been determined by trials.4,39 Sensitivity of synovial fluid culture, sometimes cited as the gold standard for diagnosis, has a sensitivity ranging from 75% to 95%. Of note, sensitivities of Gram stain and culture in disseminated gonococcal infection are much lower (< 10% and 10% to 50%, respectively.) If gonococcus is suspected, cultures of the blood and of urethral, rectal, or oropharyngeal swabs should be obtained.39
Summary
No validated clinical decision tools exist to assist with the diagnosis of septic arthritis. No single element is sufficiently sensitive and specific when looking for the cause of an acutely hot joint. The emergency physician is best served by combining elements of the history, physical exam, patient factors, and lab analysis to help make the diagnosis and determine the next steps in patient management.4 When a septic joint is part of the differential diagnosis, arthrocentesis must be performed.
REFERENCES
- Aceves-Avila FJ, Delgadillo-Ruano MA, Ramos-Remus C, et al. The first descriptions of therapeutic arthrocentesis: A historical note. Rheumatology 2003;42:180-183.
- Dooley P, Martin R. Corticosteroid injections and arthrocentesis. Can Fam Physician 2002;48:285-292.
- Beyaz SG. Comparison of efficacy of intra-articular morphine and steroid in patients with knee osteoarthritis. J Anaesthesiol Clin Pharmacol 2012;28:496-500.
- Carpenter CR, Schuur JD, Everett WW, Pines JM. Evidence-based diagnostics: Adult septic arthritis. Acad Emerg Med 2011;18:781-796.
- Lin HM, Learch TJ, White EA, Gottsegen CJ. Emergency joint aspiration: A guide for radiologists on call. Radiographics 2009;29:1139-1158.
- Luthringer TA, Fillingham YA, Okroj K, et al. Periprosthetic joint infection after hip and knee arthroplasty: A review for emergency care providers. Ann Emerg Med 2016;68:324-334.
- Rothschild BM. Gout and pseudogout. eMedicine 2017. Available at: http://emedicine.medscape.com/article/329958. Accessed Sept. 14, 2017.
- Ungprasert P, Kaewpoowat Q, Ratapano S, et al. Presence of crystals is not evidence of absence of infection. Am J Emerg Med 2013;31:455.e1-455.e2.
- Boss SE, Mehta A, Maddow C, Luber SD. Critical orthopedic skills and procedures. Emerg Med Clin North Am 2013;31:261-290.
- Fitch RW, Kuhn JE. Intraarticular lidocaine versus intravenous procedural sedation with narcotics and benzodiazepines for reduction of the dislocated shoulder: A systematic review. Acad Emerg Med 2008;15:703-708.
- Yari M, Saeb M, Golfam P, Makhloogh Z. Analgesic efficacy of intra-articular morphine after arthroscopic knee surgery in sport injury patients. J Inj Violence Res 2013;5:84-88.
- Graham RF, Hughes J, Johnson A, et al. Intra-articular morphine versus lidocaine for acute knee pain. J Spec Oper Med 2014;14:74-79.
- Zuber TJ. Knee joint aspiration and injection. Am Fam Physician 2002;66:1497-1500.
- Smith BG, Cruz AI Jr, Milewski MD, Shapiro ED. Lyme disease and the orthopaedic implications of Lyme arthritis. J Am Acad Orthop Surg 2011;19:91-100.
- Arvikar SL, Steere AC. Diagnosis and treatment of Lyme arthritis. Infect Dis Clin North Am 2015;29:269-280.
- Dooley DP. Aspiration of the possibly septic joint through potential cellulitis: Just do it! J Emerg Med 2002;23:210.
- Bettencourt RB, Linder MM. Arthrocentesis and therapeutic joint injection: An overview for the primary care physician. Prim Care 2010;37:691-702.
- Ahmed I, Gertner E. Safety of arthrocentesis and joint injection in patients receiving anticoagulation at therapeutic levels. Am J Med 2012;125:265-269.
- Kontovazenitis PI, Starantzis KA, Soucacos PN. Major complication following minor outpatient procedure: Osteonecrosis of the knee after intraarticular injection of cortisone for treatment of knee arthritis. J Surg Orthop Adv 2009;18:42-44.
- Park KS, Peisajovich A, Michael AA, et al. Should local anesthesia be used for arthrocentesis and joint injections? Rheumatol Int 2009;29:721-723.
- Lewis MF. Joint assessment using bedside ultrasonography. eMedicine 2015. Available at: http://emedicine.medscape.com/article/1380545. Accessed Sept. 14, 2017.
- Berney SN. Joint aspiration. eMedicine 2015. Available at: http://emedicine.medscape.com/article/2094114. Accessed Sept. 14, 2017.
- Cardinal E, Chhem RK, Beauregard CG. Ultrasound-guided interventional procedures in the musculoskeletal system. Radiol Clin North Am 1998;36:597-604.
- Balint PV, Kane D, Hunter J, et al. Ultrasound guided versus conventional joint and soft tissue fluid aspiration in rheumatology practice: A pilot study. J Rheumatol 2002;29:2209-2213.
- Ruta S, Catay E, Marin J, et al. Knee effusion: Ultrasound as a useful tool for the detection of calcium pyrophosphate crystals. Clin Rheumatol 2016;35:1087-1091.
- Punzi L, Oliviero F. Arthrocentesis and synovial fluid analysis in clinical practice: Value of ultrasonography in difficult cases. Ann NY Acad Sci 2009;1154:152-158.
- Royall NA, Farrin E, Bahner DP, Stawicki SP. Ultrasound-assisted musculoskeletal procedures: A practical overview of the current literature. World J Orthop 2011;2:57-66.
- Daley EL, Bajaj S, Bisson LJ, Cole BJ. Improving injection accuracy of elbow, knee, and shoulder: Does injection site and imaging make a difference? A systematic review. Am J Sports Med 2011;39:656-662.
- Hansford BG, Stacy GS. Musculoskeletal aspiration procedures. Semin Intervent Radiol 2012;29:270-285.
- Bum Park Y, Ah Choi W, Kim YK, et al. Accuracy of blind versus ultrasound-guided suprapatellar bursal injection. J Clin Ultrasound 2012;40:20-25.
- Roy S, Dewitz A, Paul I. Ultrasound-assisted ankle arthrocentesis. Am J Emerg Med 1999;17:300-301.
- Wisniewski SJ, Smith J, Patterson DG, et al. Ultrasound-guided versus nonguided tibiotalar joint and sinus tarsi injections: A cadaveric study. PM R 2010;2:277-281.
- Boniface KS, Ajmera K, Cohen JS, et al. Ultrasound-guided arthrocentesis of the elbow: A posterior approach. J Emerg Med 2013;45:698-701.
- Raza K, Lee CY, Pilling D, et al. Ultrasound guidance allows accurate needle placement and aspiration from small joints in patients with early inflammatory arthritis. Rheumatology 2003;42:976-979.
- Li SF, Henderson J, Dickman E, Darzynkiewicz R. Laboratory tests in adults with monoarticular arthritis: Can they rule out a septic joint? Acad Emerg Med 2004;11:276-280.
- Li SF, Cassidy C, Chang C, et al. Diagnostic utility of laboratory tests in septic arthritis. Emerg Med J 2007;24:75-77.
- Brusch JL. Septic arthritis. eMedicine 2016. Available at: http://emedicine.medscape.com/article/236299. Accessed Sept. 14, 2017.
- Pascual E, Batlle-Gualda E, Martinez A, et al. Synovial fluid analysis for diagnosis of intercritical gout. Ann Intern Med 1999;131:756-759.
- Thomsen TW, Shen S, Shaffer RW, Setnik GS. Videos in clinical medicine. Arthrocentesis of the knee. N Engl J Med 2006;354:e19.
- Smith BG, Cruz AI Jr, Milewski MD, Shapiro ED. Lyme disease and the orthopaedic implications of Lyme arthritis. J Am Acad Orthop Surg 2011;19:91-100.
- Abdullah S, Young‐Min SA, Hudson SJ, et al. Gross synovial fluid analysis in the differential diagnosis of joint effusion. J Clin Pathol 2007;60:1144-1147.
- Cho HJ, Burke LA, Lee M. Septic arthritis. Hospital Med Clin 2014;3:494-503.
The ability to rapidly recognize infection, as well as distinguish inflammatory arthropathy from infection, crystal arthropathy, and osteoarthritis, makes arthrocentesis a valuable and sometimes essential emergency medicine procedure.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
