The Pediatric Airway and Rapid Sequence Intubation in Trauma
November 1, 2017
Reprints
AUTHORS
Carmen D. Sulton, MD, FAAP, Assistant Professor of Pediatrics and Emergency Medicine, Emory University School of Medicine, Atlanta, GA
Taryn R. Taylor, MD, FAAP, FACEP, Assistant Professor of Pediatrics and Emergency Medicine, Emory University School of Medicine, Atlanta, GA
PEER REVIEWER
Mary Jo A. Bowman, MD, FAAP, Associate Professor of Clinical Pediatrics, Ohio State University College of Medicine, Attending Physician, Emergency Department, Nationwide Children’s Hospital, Columbus, OH
EXECUTIVE SUMMARY
- The pediatric airway differs greatly from the adult airway: It is shorter and more anterior. This often can make direct visualization challenging.
- Assume difficult airways in children with significant head, face, or neck trauma. This includes children with cervical spine immobilization. Additionally, children with craniofacial abnormalities and syndromes are at higher risk for difficult airways and difficult intubations.
- Hypoxia is a precursor to cardiac arrest in children. Preoxygenation is necessary prior to securing an airway.
- Oral airways and nasopharyngeal airways, when properly sized, are helpful adjuncts to prevent upper airway obstruction.
- The goal of rapid sequence intubation is to emergently secure an airway safely. There are a variety of sedation options in pediatrics to achieve this goal.
- Nondepolarizing paralytic agents, such as rocuronium, are increasing in popularity in pediatric emergency medicine because of their shorter half-life and more favorable safety profile.
- Complications of RSI, such as missed intubations and multiple airway attempts, should be considered carefully. Consultation with subspecialists, such as anesthesia if needed, should be considered.
Trauma remains the most common cause of injury and death among children between 1 and 19 years of age. Injuries are the leading cause of emergency department visits, and nearly 22 million children are injured every year. Motor vehicle collisions continue to be the most common cause of injury-related death in the pediatric population, and blunt trauma is the predominant mechanism.1 Understanding the pediatric airway is a critical part of trauma resuscitation.
Pediatric trauma can range from minor to life and limb threatening. Therefore, rapid recognition of major injuries is paramount. Establishing a patent airway to provide adequate oxygenation is of critical importance. A rapid and reproducible scheme to evaluate the pediatric trauma patient is the core of initial management and stabilization. The consistent order of priority is airway, breathing, and circulation, followed by disability and exposure/environment. This article will discuss in detail the approach to the pediatric trauma patient, with a strict focus on the pediatric airway.
Pediatric Upper Airway: Basic Anatomy
The pediatric airway can be divided into three basic components/segments: the supraglottic segment, the glottic segment, and the intrathoracic segment. The supraglottic component is the most collapsible component of the upper airway. The glottis or laryngeal component of the upper airway consists of the vocal cords and trachea. The intrathoracic segment consists of the thoracic trachea and bronchi.
Several factors fundamentally distinguish the pediatric airway from the adult airway. First, the pediatric airway is shorter in length and smaller in diameter. This often makes the field of vision narrow when assessing the airway. Children have large occiputs, which predisposes to airway obstruction. The large occiput combined with the short neck makes laryngoscopy more difficult. The vocal cords also are narrowed somewhat. The adenoids may be hypertrophied. The tongue in children often is noted to be large relative to the size of the oropharynx.2 In addition, the larynx is located more anteriorly in infants and children. The epiglottis is narrow, curved, and floppy, and is described as omega-shaped. This can make this structure difficult to mobilize and control with the laryngoscope blade. The trachea is more compressible and, thus, cricoid pressure often is less helpful when accessing the airway. In children who are younger than 10 years of age, the narrowest portion of the airway is at the level of the cricoid cartilage, below the level of the glottis. This is at approximately the level of C4 in children and at the level of C6-C7 in adults.3 (See Figure 1.) Finally, pediatric patients have developing teeth that are easily injured, avulsed, and/or aspirated. The underlying alveolar ridge contains tooth buds that are susceptible to injury and disruption.4
Figure 1. Diagram of Adult vs. Infant Airway
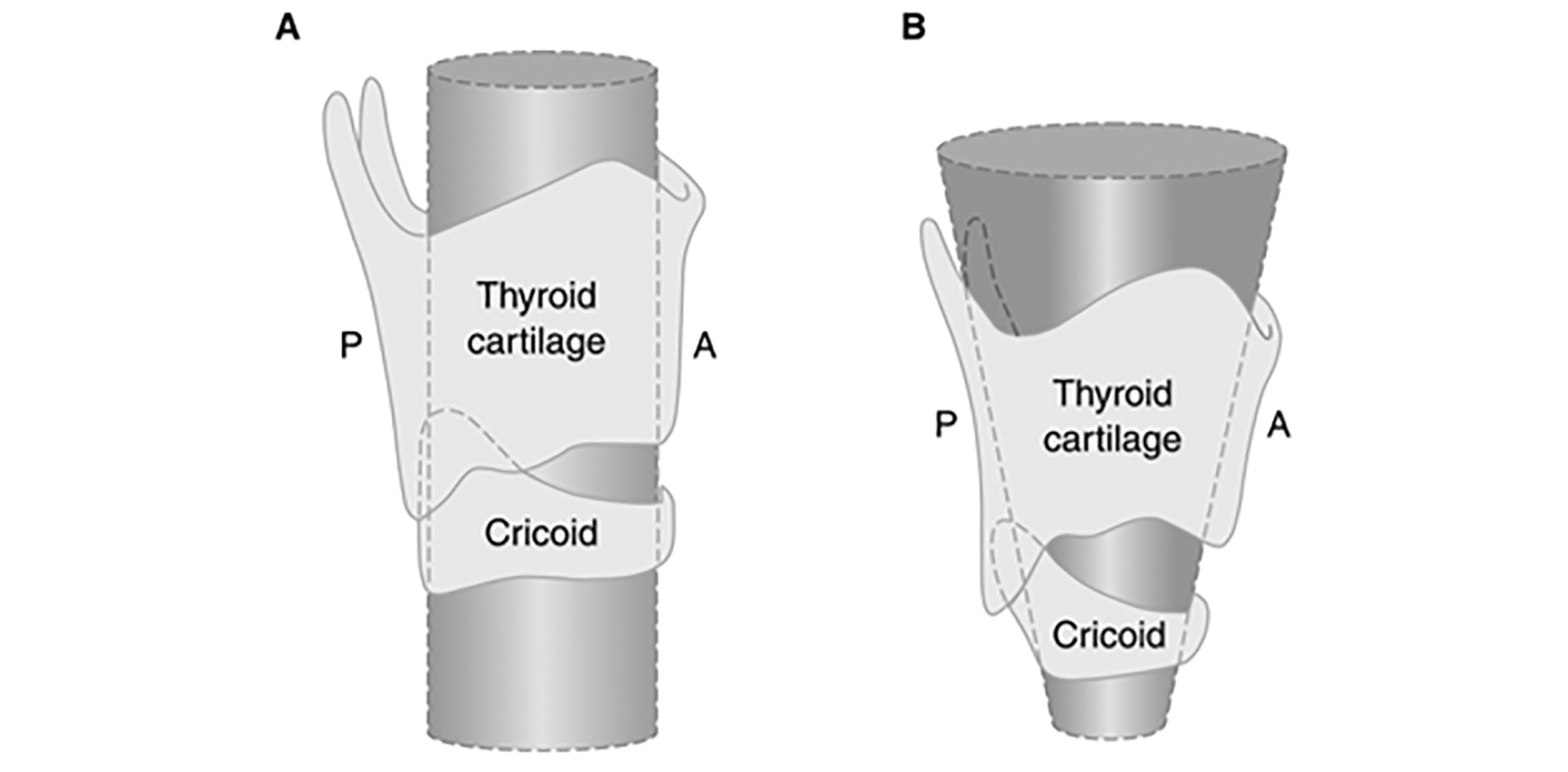
Schematic of an adult (A) and infant (B) airway. Note the comparison between the cylindrically chaped airway with uniform diameters in the adult and the conically shaped airway of the child with the narrowest region at the cricoid. A, anterior; P, posterior. Reprinted with permission from: Cote CJ, Todres ID. The pediatric airway. In: Ryan JF, Todres ID, Cote CJ, et al., eds. A Practice of Anesthesia for Infants and Children. Philadelphia: WB Saunders, 1986;35-58, with permission from Elsevier.
Cervical Spine Considerations
One notable trauma consideration involves the protection of the cervical spine. While pediatric neck trauma and cervical spine trauma are uncommon, if unrecognized they can be life-threatening. Most cervical spine injuries and neck trauma are due to either blunt or penetrating trauma. Motor vehicle crashes, falls, and nonaccidental trauma are the most common etiologies of blunt trauma. Penetrating trauma leading to cervical spine injury is uncommon in children but may be caused by gunshots, dog bites, and other sharp objects. Pediatric patients with cervical spine trauma often have subtle neurologic findings, so reassessments must be frequent and an index of suspension must be high. Clinical signs such as neurologic dysfunction, audible stridor, subcutaneous emphysema, hoarseness, and neck hematomas are concerning for possible neck trauma. This also should raise concerns for possible cervical spine injury and the potential for a difficult airway. Oral intubation is the preferred method for securing an airway in the setting of cervical spine injury. However, this should be performed by a skilled provider and only with preparation for a surgical airway if needed. Care should be taken not to hyperextend the neck.4 All airway maneuvers in a multiple trauma or unresponsive patient should be performed with manual in-line stabilization. For airway-opening maneuvers use a jaw thrust or chin lift; do not perform a head tilt maneuver.
Airway Adjuncts
There are several measures that may serve effectively and efficiently as adjuncts. The most common are oropharyngeal airways (OPA) and nasopharyngeal airways. These devices may relieve upper airway obstruction caused by the tongue.
An OPA can be used in an unconscious child with no gag reflex. It is a helpful tool when airway readjustment or jaw thrust have failed and an unobstructed airway is needed. The OPA is made of hard plastic and consists of a bite block as well as a conduit for suction. The use of a tongue blade may aid in successful insertion. The practice of inserting the OPA backward and then rotating it 180 degrees to fit in the mouth, as often is performed in adults, may cause soft tissue trauma in children. The OPA is measured from the corner of the mouth to the angle of the jaw. It is critical to select the correct size of OPA. If it is too large, the OPA can obstruct the airway and cause trauma to the larynx. If it is too small, the OPA can push the tongue back into the airway, also contributing to obstruction.5 OPAs come in a variety of sizes from 4 cm to 10 cm.
Nasopharyngeal (NP) tubes can be used in a similar fashion and also are helpful for upper airway obstruction. They can be used in conscious or semiconscious patients. NP tubes often are made of soft plastic, and are best measured from the nares to the tragus of the ear. NP tubes may lacerate adenoidal tissue, so adenoidal hypertrophy is a relative contraindication for NP tube placement. NP tubes come in a variety of sizes from 12F to 30F.5 NPA should be avoided in children with suspicion of a basilar skull fracture.
Rapid Sequence Intubation: Goals
The goals of intubation in the setting of trauma are to place an artificial and secure airway in the safest and most efficient manner as possible. Rapid sequence intubation (RSI) is defined as the simultaneous administration of a sedative agent for emergent intubation.6 Tracheal intubation is indicated in the following clinical scenarios: 1) respiratory failure due to inadequate oxygenation; 2) respiratory failure due to inadequate ventilation; 3) lack of neuromuscular respiratory drive/impending airway compromise; and 4) lack of airway protective reflexes.1,4 Within the pediatric population, the most common indications for intubation are trauma and primary respiratory failure. Examples include cardiac arrest, traumatic brain injury, and status epilepticus. Additionally, in the setting of trauma, patients with a Glasgow Coma Scale score of 8 or less should be evaluated for immediate airway protection and intubation.7 The establishment and maintenance of a definitive airway is an essential skill for emergency medicine and pediatric emergency medicine providers.1
The most common route of intubation in the emergency setting is orally, namely direct laryngoscopy, which will be referenced most commonly throughout this article. Other methods and devices used in RSI and artificial airways include video-assist methods (i.e., GlideScope and C-MAC) and surgical airways (surgical cricothyroidotomy, needle cricothyroidotomy, and tracheostomy). Surgical airways are beyond the scope of this article.
Preoxygenation
Before attempts are made to establish an airway, all children should be preoxygenated by the delivery of high-flow 100% oxygen prior to intubation. The goal is to prevent complications during the apneic phase of RSI. Without this practice, nearly one-third of patients will have a desaturation episode to < 90%.8
In the setting of trauma, children have a higher metabolic rate and resultant oxygen requirement. Poor ventilation and increased demands can result in hypoxia. Even transient episodes of hypoxia or desaturation can have devastating consequences. Untreated hypoxia often is a precursor to cardiac arrest in children.1 Other complications from hypoxia during RSI include hemodynamic instability, dysrhythmias, and anoxic brain injury.
Preoxygenation with 100% oxygen for about three minutes via either a nasal cannula,9 bag-valve-mask, or a non-rebreather mask can decrease the chance of rapid desaturation.10 With preoxygenation, an apneic child is significantly less likely to have significant desaturation during RSI.11,12
Pediatric Airway Management Equipment
The first step in assessing and managing the pediatric airway involves having the necessary equipment available and appropriately sized. Supplemental oxygen with a bag and mask is crucial prior to the establishment of any advanced airway. The pediatric mask should measure from the bridge of the nose to the cleft of the chin, forming a tight seal. A poorly aligned face mask can contribute to ineffective ventilation and oxygenation. There are two commonly used ventilation devices — the self-inflating bag and the flow-inflating bag. Self-inflating bags often are used for initial resuscitations and fill with either oxygen or room air. Flow-inflating bags require an oxygen source for operation. Being trained and experienced at bag-mask ventilation is the first step in successful management of the pediatric airway.5
For most children, the size of endotracheal tubes (ETT) can be calculated using standard formulas and calculations. In the past, uncuffed tubes were the standard of care for younger children (< 8 years old) because of concerns about airway trauma and post-extubation stridor. However, most standard cuffed ETTs have low-pressure cuffs, with minimal risk for airway trauma, airway edema, or need for post-extubation steroids.13 Specific preferences for cuffed tubes include concern for aspiration and the ability to ventilate at lower peak pressures. Calculating ETT sizes is standard by age. They are measured by diameter in millimeters. The common formula is: (age/4) + 4 for uncuffed tubes and (age/4) + 3.5 for cuffed tubes. Length-based pediatric emergency measurement tapes (i.e., Broselow®) are helpful for determining appropriate ETT sizes. Regardless of the measured size, it is crucial to have one size larger and one size smaller at the bedside when securing an airway in case one size does not have an appropriate seal. Research shows that ETT cuffs should be inflated to no more than 20-25 cm H2O.5 Some have found that < 15 cm H2O also is acceptable.14 (See Table 1.)
Table 1. Equipment Sizing Chart (PICU Chart)
Weight |
Oral Airway |
Laryngoscope Blade (size) |
ET Tube (mm) † |
ET Tube Insertion Length |
|
Abbreviations: ET, endotracheal; mm, millimeters; cm, centimeters. † Per 2015 AHA Guidelines, in the hospital cuffed or uncuffed tubes may be used. Adapted from University of Iowa Stead Family Children’s Hospital https://uichildrens.org/health-library/equipment-sizing-chart-picu-chart |
||||
|
< 3 kg |
< 1 kg: 2.5 uncuffed 1-2 kg: 3.0 uncuffed |
0.5 kg: 6.5 cm 1 kg: 7 cm 2 kg: 8 cm |
||
|
3-5 kg |
2-4 kg: 3.5 uncuffed 5 kg: 3.5 uncuffed |
3 kg: 9-9.5 cm 4 kg: 9.5-10 cm 5 kg: 10-10.5 cm |
||
|
Small infant 6-7 kg |
50 mm |
Miller 1 (straight) |
3.5 uncuffed 3.0 cuffed |
10.5-11 cm |
|
Infant 8-9 kg |
60 mm |
Miller 1 (straight) |
3.5 uncuffed 3.0 cuffed |
10.5-11 cm |
|
Toddler 10-11 kg |
60 mm |
Miller 1 (straight) |
4.0 uncuffed 3.5 cuffed |
11-12 cm |
|
Small child 12-14 kg |
60 mm |
Miller 2 (straight) |
4.5 uncuffed 4.0 cuffed |
13.5 cm |
|
Child 15-18 kg |
70 mm |
Miller 2 (straight) |
5.0 uncuffed 4.5 cuffed |
14-15 cm |
|
Child 19-23 kg |
80 mm |
Miller 2 (straight) or Macintosh 2 (curved) |
5.5 uncuffed 5.0 cuffed |
16.5 cm |
|
Large child 24-29 kg |
80 mm |
Miller 2 (straight) or Macintosh 2 (curved) |
6.0 cuffed |
17-18 cm |
The goal of selecting the laryngoscope blade should be for the operator to reach the glottic structures while controlling the tongue effectively. There are two components of the laryngoscope: the handle and the blade.4 Although smaller handles often are preferred for smaller blades, this selection is ultimately provider preference. Laryngeal blades can be curved (Macintosh) or straight (Miller). Curved blades should follow the tongue and end in the epiglottic vallecula. Straight blades allow lifting of the epiglottis to expose the glottic opening.15 Blade sizes and choices are described in Table 2.
Table 2. Laryngoscope Blades by Age
Age |
Blade |
|
Premature neonate |
Miller 0 |
|
Term neonate |
Miller 0 or 1 |
|
6 to 12 months |
Miller 1 |
|
1 to 2 years |
Miller 1, Macintosh 2 |
|
2 to 8 years |
Miller 2, Macintosh 2 |
|
> 8 years |
Miller 2, Macintosh 3 |
|
Key: Miller (straight), Macintosh (curved) |
|
Additionally, it is important to have all necessary advanced airway equipment at the bedside. This includes airway adjuncts as described above, oxygen sources, bag and mask, and intubation medications. There are several mnemonics to help organize the necessary airway equipment prior to the intubation procedure. One common mnemonic is SOAPME, which is described as:
- Suction: appropriate sized suction catheter and suction apparatus (i.e., Yankauer suction).
- Oxygen: adequate oxygen supply, functioning flow meters, and appropriate oxygen delivery devices.
- Airway: appropriate sized airway equipment. This includes nasopharyngeal airways, oral airways, laryngoscope blades (which have been checked, light working), endotracheal tubes, laryngeal mask airways, bag-valve-mask (checked for size).
- Pharmacy: all resuscitation medications needed for an emergency and all reversal medications, as well as sedative medications and sedative antagonists.
- Monitors: pulse oximeter with size-appropriate probes as well as blood pressure cuffs, ECG and stethoscopes, and end-tidal monitoring as needed.
- Extra Equipment: any extra equipment as needed.
Identifying Difficult Airways
Assessing for a difficult airway can be challenging in children, namely because of lack of patient cooperation.16 This makes airway prediction tools, such as Mallampati scoring, problematic and less predictive than in adults. Commonly used in adults, Mallampati scoring is attractive because it is noninvasive and quick. This scoring system describes visualization of the tonsils and uvula. It has been accepted by specialists, such as anesthesiologists, as a guide for assessing ease of intubation; however, the correlation to pediatrics is limited to cooperative patients, where it has been found to be effective.17 The Wilson Risk score also often is used in the adult population. It describes weight, head and neck movement, jaw movement, receding mandible, and “buck teeth” as predictive risk factors. There is a growing trend toward combining airway scoring systems in an effort to improve their sensitivity and specificity.18
Pediatric airways can be even more challenging in the setting of trauma, when blood, vomit, or excess secretions can obstruct direct visualization of the airway. Patients younger than 1 year of age also are at higher risk of difficult airways and difficult intubations.3,19 In addition, a difficult airway always should be anticipated in pediatric patients with baseline craniofacial abnormalities.19 This includes a variety of conditions, such as Pierre Robin, Down, Treacher Collins, Cornelia de Lange, and Goldenhar syndromes. Other challenging anatomic considerations include macroglossia, midface hypoplasia, and cleft palate. Additionally, a history of a difficult intubation or a difficult airway in the past should prompt expert consultation (i.e., pediatric anesthesia).4,15,20
In the setting of facial trauma, the potential for a difficult airway should be considered. Inability to open the mouth because of pain will make accessing the airway problematic. Loose teeth, blood in the airway, and pharyngeal edema can increase the risk of possible airway obstruction.4 Cervical spinal immobilization also is noteworthy, in that pediatric patients often have limited mobility, making visualization of the airway compromised. It is of the utmost importance that patients with possible neck trauma have their head and neck maintained in a neutral position so as to minimize the risk of possible cervical spine compression and further injury.3
An alternative to traditional endotracheal intubation and direct laryngoscopy after multiple attempts is a laryngeal mask airway (LMA). The LMA is a supraglottic device that can be placed blindly. Although not a definitive airway device, an LMA can be placed with minimal training and has a low complication rate.21 This is an option when a patient cannot be intubated and a bag-mask is ineffective.22 The LMA is teardrop-shaped and has an inflatable cuff.2,13 It is placed in the hypopharynx and sits above the glottis. Air flow is directed through the glottis and then into the lungs. LMAs are available in a variety of sizes for infants to teenagers. (See Table 3.) A variety of other supraglottic devices can be used when intubation and ventilation are challenging, with blind placement in the esophagus. They include the CombiTube, the laryngeal tube, and the perilaryngeal airway.4
Table 3. Laryngeal Mask Airway Sizes by Weight for Pediatric Patients
Weight |
Laryngeal Mask Airway Size |
|
< 5 kg |
1 |
|
5 to 10 kg |
1.5 |
|
10 to 20 kg |
2 |
|
20 to 30 kg |
2.5 |
|
30 to 50 kg |
3 |
Monitors
Pulse Oximeters. Throughout assessment and management of the pediatric airway, it is imperative that oxygen saturations are monitored closely.23 It is well documented that hypoxemia often is a precursor to cardiac arrest.1 Pulse oximetry is a simple method of continuously measuring oxygen saturations in a sedated and/or intubated patient.23 Most pulse oximeters also give feedback about the patient’s heart rate as well.
Pulse oximeters measure hemoglobin oxygen saturation via an external probe. The light of two varying wavelengths passes through the skin, and the difference in absorbance by arterial blood of these two wavelengths by the photodetector within the pulse oximeter is used to calculate the level of oxygenation.18 Hypoxemia is assessed more readily by monitoring devices such as pulse oximetry rather than physical exam alone.
End-tidal Carbon Dioxide Monitoring. Capnography, or continuous monitoring of expired carbon dioxide (CO2), is noninvasive and can provide an early indication of inadequate ventilation.24 Capnography uses infrared technology to measure the amount of CO2 exhaled.18 Additionally, capnography provides a visual depiction of CO2 exhalation over time, which provides insight into ventilation and respiratory function. Although capnography is standard practice in anesthesia, it has not been implemented routinely in standard pediatric airway management.24
Specifically, in pediatric patients, capnography has several advantages. It has been shown to be superior in detecting ETT displacement. It also has been shown to be superior to physical exam at identifying apnea and gives the earliest warning signs of ETT obstruction and esophageal intubation.24-26
Rapid Sequence Intubation: Sedation Options
The goal of RSI, which is the cornerstone of airway management, is to secure the airway safely with as few attempts as possible in a controlled manner.27 There are two primary classes of medications used for RSI.
The first class of medications is sedatives, which are used to mitigate the physiologic consequences of intubation. The second class is the paralytics. These two classes of medications will be discussed in further detail below. (See Tables 4 and 5.)
Table 4. Sedatives Used in Pediatric Rapid Sequence Intubation
Medication |
Dose |
Onset |
Duration |
Clinical Effects |
Adverse Events |
|
Etomidate |
0.3 mg/kg IV |
5 to 15 seconds |
5 to 15 minutes |
Provides sedation with minimal hemodynamic changes |
Possible adrenal suppression; contraindicated in sepsis |
|
Ketamine |
1 to 2 mg/kg IV |
45 to 60 seconds |
10 to 20 minutes |
Provides sedation with minimal hemodynamic changes; bronchodilator |
Use with caution in hypertensive patients |
|
Propofol |
1 to 1.5 mg/kg IV |
15 to 45 seconds |
5 to 10 minutes |
Provides sedation with rapid onset and short duration of action; inhibits bronchoconstriction |
Reduced cerebral perfusion pressure; hypotension; myocardial depression; pain on injection |
|
Midazolam |
0.2 to 0.3 mg/kg IV |
60 to 90 seconds |
15 to 30 minutes |
Anticonvulsant |
Respiratory depression; apnea, hypotension |
|
Fentanyl |
2 to 4 mcg/kg IV |
1 to 2 minutes |
30 minutes |
Analgesia |
Respiratory depression; apnea; hypotension; chest wall rigidity |
|
Morphine |
0.1 to 0.2 mg/kg IV |
3 to 5 minutes |
4 to 6 hours |
Analgesia |
Respiratory depression; hypotension; pruritus |
Table 5. Neuromuscular Blocking Agents Used in Pediatric Rapid Sequence Intubation
Medication |
Dose |
Onset |
Duration |
Adverse Events |
|
Succinylcholine * Contraindicated in children with or suspected of having muscular dystrophy or neuromuscular disease |
2 mg/kg IV for children younger than 2 years of age and 1 to 1.5 mg/kg IV for children older than 2 years of age |
45 seconds |
5 to 10 minutes |
Malignant hyperthermia, bradycardia, hyperkalemia, increased intraocular pressure, muscle fasciculation, jaw rigidity |
|
Rocuronium |
1 mg/kg IV |
60 seconds |
45 minutes |
Allergy (rare) |
|
Vecuronium |
0.15 to 0.2 mg/kg IV |
120 to 180 seconds |
45 to 60 minutes |
Long acting, which can be disadvantageous in difficult airway |
Sedatives
Etomidate. Etomidate is an imidazole, a non-barbiturate, and a gamma-aminobutyric acid receptor (GABA)-mediated sedative hypnotic. It has very potent sedative features but no analgesic properties. One of its key features is its very minimal effect on heart rate and blood pressure. Also, apnea is noted significantly less with etomidate as compared to propofol or barbiturates. The cerebral perfusion pressure is maintained during etomidate administration, while the intracranial pressure, cerebral blood flow, and metabolic rates are decreased. Since etomidate does not release histamine, allergic reactions rarely are seen.28 Etomidate has a relatively quick onset of action of 5 to 15 seconds, with a short duration of sedation of 5 to 15 minutes. The dose for pediatric RSI that has been shown to be safe is 0.3 mg/kg intravenously (IV).28,29
Since hemodynamic instability is not a significant consequence of etomidate, its use is favorable in patients who are experiencing decompensated shock, particularly those in a trauma setting who have sustained a large volume of blood loss. Both Sokolove et al30 in their retrospective review and Zuckerbraun et al29 in their prospective study found that hypotension after induction with etomidate was uncommon.31 Further, the neuroprotective properties of etomidate make it a popular choice for RSI of pediatric patients who have sustained a traumatic head injury. However, it is important to note that etomidate has no analgesic properties; thus, in the global management of a trauma patient, additional therapies may be warranted in the peri-intubation setting.
Early studies have reported myoclonus and vomiting as potential side effects of etomidate. However, more recent studies found these effects to be uncommon, particularly when etomidate was used in conjunction with a paralytic for RSI.29,32,33 Injection site pain and thrombophlebitis also are concerns if etomidate is administered through a peripheral vein.18 A more concerning potential disadvantage of etomidate is its suppression of adrenal cortical function by inhibiting 11-β-hydroxylase. In light of this, providers should consider other sedative agents for RSI in patients at risk for adrenal insufficiency, including those presenting in septic shock, as etomidate is contraindicated.34 The use of etomidate in pediatric trauma patients is not as clearly delineated in the literature. However, a recent survey of pediatric trauma programs found that its use is relatively common.35
Ketamine. Ketamine is a dissociative anesthetic that interacts with the N-methyl D-aspartate (NMDA) receptors. It is also an α- and β-adrenergic receptor agonist, and blocks the reuptake of catecholamines.36 Clinical effects include anesthesia, analgesia, amnesia, and suppression of fear and anxiety.36 Ketamine is unique in that it possesses both analgesic and amnestic properties.37 The sympathomimetic effects of ketamine on the cardiovascular and respiratory systems result in increased heart rate, blood pressure, and bronchodilation. Although ketamine administration results in increased salivary and tracheobronchial secretions, airway protective reflexes, such as coughing, sneezing, and swallowing, are maintained.36 Ketamine is highly lipid soluble; thus, it crosses the blood brain-barrier quickly, leading to rapid onset of action. The onset of sedation with IV dosing is 45 to 60 seconds, with duration of 10 to 20 minutes.38 The RSI dose is 1 to 2 mg/kg IV.18
Secondary to its bronchodilatory effects, ketamine is an ideal sedative for RSI in patients with severe bronchospasm. Because of the sympathomimetic properties of ketamine, it also may be useful for patients in shock. Unlike etomidate, ketamine also may be considered in patients with septic shock, as it does not cause adrenocortical suppression. However, every effort should be made to reverse hypovolemia and hypotension prior to RSI in these patients. Ketamine’s hemodynamic stability and anticonvulsant properties may benefit the hypo- or normotensive patient with traumatic brain injury.39
Early studies of ketamine suggested that it was contraindicated in patients with traumatic brain injury because of the side effect of increased intracranial pressure (ICP). More recent studies suggest that the evidence for this is minimal.40 Bar-Joseph et al further disproved this notion, finding that it is safe for use in patients with traumatic brain injury.41 Similar to early studies relating ketamine to increased ICP, ketamine previously has been thought to cause mild increased intraocular pressure.42 At doses commonly used for RSI, this has been found not to be the case.43 Another potential side effect of ketamine is excessive salivation, for which atropine pretreatment previously was recommended. In their prospective observational study, Brown et al found that this is a rare complication of ketamine at RSI doses, and the routine use of atropine was not warranted.44 Other side effects of ketamine administration include transient oxygen desaturations, laryngospasm, tachycardia, emesis, emergence reactions, and myoclonic jerks. These effects are not seen commonly in the setting of ketamine use for RSI.
Propofol. Propofol is a highly lipid-soluble agent that acts at the GABA and sodium blockade. It is a non-barbiturate sedative hypnotic anesthetic agent that causes sedation by direct suppression of brain activity, while the amnestic effect is due to interference with long-term memory creation.45 However, it does not provide any analgesia. Propofol contains egg lecithin and soybean oil and, thus, is absolutely contraindicated in patients with egg allergy. However, fewer allergic reactions have been reported with newer preparations of the medication. The initial bolus dose is 1 to 1.5 mg/kg IV, with extremely rapid onset generally in 15 to 45 seconds and duration of action of 5 to 10 minutes. Studies have suggested a slightly longer time to peak effect in children.46 Infants also are noted to require higher doses than older children.
Because of its suppression of the sympathetic activity, propofol causes both vasodilation and myocardial suppression. Therefore, its use as an RSI agent should be limited to those patients who are hemodynamically stable. Although it does have neuroinhibitory effects that make it a good RSI choice for patients in status epilepticus, the associated drop in mean arterial pressure and subsequent cerebral perfusion pressure limit its use in patients with traumatic brain injury. Propofol has been shown to inhibit bronchoconstriction, and can be useful for RSI in patients who have bronchospasm. It also has been shown to have modest antiemetic properties. Respiratory events, such as central apnea and hypoxia, are not uncommon and should be expected. These occur most commonly with bolus doses. The safety profile of propofol has been well-studied in pediatrics, outside of the operating room.47 Serious events, such as cardiac arrest, unplanned anesthesia consult, and increased level of medical care, are noted to be minimal when propofol is used in healthy children.48
Midazolam. Midazolam is a short-acting benzodiazepine that, like propofol, binds the GABA receptor. It has amnestic, anxiolytic, and anticonvulsant properties. The anticonvulsant effect lends itself to usefulness as a sedative for RSI for patients in status epilepticus. Also, similar to propofol, midazolam decreases systemic vascular resistance, resulting in hypotension; therefore, it should not be used in hemodynamically unstable patients. Respiratory depression and apnea also are side effects seen with midazolam use, making preoxygenation prior to paralytic administration less effective. These side effects are augmented when midazolam is combined with other sedative agents, particularly opioids. Because of the above noted side effects, midazolam usually is not recommended as a first-line sedative agent for RSI. The induction dose is 0.2 to 0.3 mg/kg IV, the onset of action is 60 to 90 seconds, and the effect lasts for 15 to 30 minutes.
Narcotics. Fentanyl and morphine are narcotics that also can be used for sedation during RSI. Unlike the other sedatives, these have analgesic properties that can be useful in the trauma setting. Morphine is associated with greater histamine release, resulting in pruritus, while fentanyl can be associated with chest wall rigidity when rapidly infused. Both fentanyl and morphine can cause hypotension, and so should be given with caution. The RSI induction dose of fentanyl is 2 to 4 mcg/kg IV and morphine is 0.1 to 0.2 mg/kg IV. The onset of action of fentanyl is 1 to 2 minutes as compared to morphine, which usually takes effect in 3 to 5 minutes. Fentanyl duration is generally 30 minutes as compared to the longer duration of morphine of 4 to 6 hours.
Paralytics
Depolarizing Agents. Succinylcholine is the only depolarizing paralytic agent that is used commonly in pediatric RSI. Its rapid onset and short duration of action make it an ideal neuromuscular blocking agent for RSI in patients with presumed difficult airways. Succinylcholine is distributed rapidly into an enlarged volume of extracellular fluid in younger patients, leading to the necessity of higher doses for RSI.49 For children younger than 2 years of age, the recommended dose is 2 mg/kg IV. The dose is 1 to 1.5 mg/kg IV for children older than 2 years of age. Bradycardia can be seen with administration of succinylcholine, and is seen more commonly in younger children. Asystole also can be seen with repeated doses of succinylcholine; however, administration of multiple doses is rare in RSI. To combat these effects, it has been recommended that children younger than 5 years of age and older children who require a second dose of succinylcholine be pretreated with atropine; however, this practice has been called into question more recently.50
An additional adverse effect associated with succinylcholine includes hyperkalemia, which results from the widespread state of muscle depolarization. Therefore, administration is contraindicated in patients with preexisting hyperkalemia, malignant hyperthermia, extensive crush injuries with rhabdomyolysis, within 48 to 72 hours after burns or acute spinal cord injury, as well as in patients with muscular dystrophy.51 Relative contraindications include administration in patients with increased ICP and intraocular pressure.51
Nondepolarizing Agents. Paralytics in this category include rocuronium and vecuronium. Rocuronium is most commonly used for pediatric RSI, as it has the shortest onset of action of less than one minute.52 It acts on the nicotinic cholinergic receptor and has a shorter duration of action than the other nondepolarizing agents.53 It has minimal cardiovascular side effects, with only a minimal reported incidence of tachycardia and mild hypertension.53 The pediatric RSI dose is 1 mg/kg IV. The average time to onset for vecuronium is 120 to 180 seconds, and the duration of action is 45 to 60 minutes. It generally is not used for RSI unless succinylcholine is contraindicated and rocuronium is unavailable. The recommended dose is 0.15 to 0.2 mg/kg IV. (See Table 5.)
Complications
Missed Intubations. RSI has been shown to be well within the scope of practice and training of pediatric as well as emergency medicine staff. Specifically, first-attempt direct laryngoscopy success rates have been shown to be about 77% for pediatric emergency medicine fellows and emergency medicine residents. Pediatrics residents have a slightly lower success rate at about 50%. Overall, the first-attempt success rate has been shown to be about 80-90%. The difficulty of the airway, as previously discussed, also is predictive of first-attempt success.54
Mainstem/Endobronchial Intubation. One of the most common technical complications of RSI is a mainstem, or endobronchial, intubation. Achieving the appropriate ETT position can be challenging for the intubating provider because the pediatric trachea is much shorter in length than an adult trachea. When intubated in the emergency department, about 30% of children likely will have an ETT that is placed incorrectly. There also is some evidence that female patients have a higher rate of misplaced ETT compared to males, possibly because of the shorter distance between the lips and the carina.55 Female patients also have a higher rate of first-attempt missed intubations, likely for similar reasons.52 Tracheas can be as short as 5 cm in neonates. Younger age and lower weight are associated with a higher rate of misplaced ETT. Additionally, the ETT position often is changed easily by subtle alterations in head position, such as flexion and rotation.56 This often leads to over-ventilation of the intubated side and hypoventilation and atelectasis of the contralateral side. This can lead to severe hypoxia if not recognized promptly.56,57
The ETT should be advanced as little as 2.5 cm in neonates, as the distance between the glottis and the carina can be as little as 5 cm. In older children, advance only enough for the cuffed ETT to disappear. Most commercial ETTs have an indicator mark that helps denote proper positioning or a visual cue for proper placement. However, generally, most ETTs can be secured at the gum based on the formula: ETT size × 3.55
Colorimetric CO2 monitoring devices also are adjuncts to help suggest proper intubation, but they do not ensure correct placement. Generally, colorimetric monitoring devices have been popular during RSI because they are fast, easily portable, highly sensitive, and do not require a monitor source.58 However, now they are being replaced largely by end-tidal capnography for a variety of reasons, which include improved accuracy and the ability to measure ventilation.24
Most airway lengths vary in size greatly, so one should not rely completely on any one formula for depth of insertion. It is common practice to observe for adequate chest rise as well as equal breath sounds. Chest radiograph after intubation also is necessary and is the most accurate method of assessing appropriate ETT position.
Pneumothorax and Aspiration. Although rare, two physical complications of RSI are pneumothoraces and aspiration events. Pneumothorax results from air entering the potential space between the visceral and parietal pleura. In the setting of trauma, a simple or asymptomatic pneumothorax can progress to a tension pneumothorax with bag-valve-mask and positive pressure ventilation.1 Overall, pneumothoraces are reported to occur in < 1% of RSI encounters.52 Aspiration generally is defined as food or food particles in the airway along with concerning sequelae: change in chest radiograph or change in oxygen requirement.59 Although these events are rare in the setting of RSI, they must be recognized promptly and treated appropriately.
Specific Pediatric Trauma Considerations
Injuries and trauma occur frequently in pediatric patients. However, providers should consider a number of factors, as they represent key differences between the management of adults and children. In the setting of blood loss, children remain normotensive much longer than adults. Further, since cardiac output in children is so dependent on their ability to increase their heart rate, procedures and medications that cause bradycardia can have significant deleterious effects on tissue perfusion.60 The pediatric airway, as discussed previously, has characteristic anatomic differences that put the patients at increased risk for airway obstruction and limited visualization during advanced airway maneuvers.
In addition to the above noted pediatric considerations, it is important to note that RSI is not without inherent risk for adverse events. These risks are compounded in a trauma setting. One must consider the potential for foreign objects such as teeth, vomitus, blood, and other debris in the airway obstructing visualization of the vocal cords. Trauma patients must be assumed to have a full stomach; pain, additional injuries, and airway maneuvers that stimulate the gag reflex can lead to vomiting and subsequent aspiration. Additional adverse events include the necessity for multiple attempts, right mainstem bronchus, or esophageal intubation, as discussed previously.
Given the specific pediatric anatomic and physiologic differences, as well as the potential for adverse events during RSI in a trauma patient, careful consideration should be given to the management of the pediatric airway. The decision to intubate children in a trauma setting should not be taken lightly, and one must consider the potential side effects of the medications administered. It is appropriate to choose an opiate as a sedative, as these agents provide pain management, which ultimately will decrease morbidity and mortality.61 However, it is important to note that both fentanyl and morphine can result in hypotension, which may compound the hemodynamic effects of extensive blood loss. Both ketamine and etomidate provide sedation with minimal hemodynamic effects and subsequently are the most favorable sedatives for RSI in the trauma setting. Further, etomidate may have a neuroprotective effect for patients who have sustained traumatic brain injury. The ideal agent for neuromuscular blockade in the trauma RSI setting is one that has rapid onset, short duration of action, and minimal side effects. Of those available, succinylcholine and rocuronium are used most frequently. A recent Cochrane Review article compared both medications and found that succinylcholine was a superior RSI agent.62 Although succinylcholine produced more favorable intubation conditions, there are a great deal of contraindications to its use, which must be recognized.
Summary/Pearls
- The pediatric airway differs greatly from the adult airway: It is shorter and more anterior. This often can make direct visualization challenging.
- Assume difficult airways in children with significant head, face, or neck trauma. This includes children with cervical spine immobilization. Additionally, children with craniofacial abnormalities and syndromes are at higher risk for difficult airways and difficult intubations.
- Hypoxia is a precursor to cardiac arrest in children. Preoxygenation is necessary prior to securing an airway.
- Oral airways and nasopharyngeal airways, when properly sized, are helpful adjuncts to prevent upper airway obstruction.
- The goal of rapid sequence intubation is to emergently secure an airway safely. There are a variety of sedation options in pediatrics to achieve this goal.
- Nondepolarizing paralytic agents, such as rocuronium, are increasing in popularity in pediatric emergency medicine because of their shorter half-life and more favorable safety profile.
- Complications of RSI, such as missed intubations and multiple airway attempts, should be considered carefully. Consultation with subspecialists, such as anesthesia if needed, should be considered.
REFERENCES
- American College of Surgeons Committee on Trauma. Advanced Trauma Life Support. 9E ed. Chicago, IL: American College of Surgeons; 2012.
- Miller R, Eriksson LI, Flesher LA, et al. Miller’s Anesthesia. 8th ed. Philadelphia: Elsevier Inc; 2015.
- Belanger J, Kossick M. Methods of identifying and managing the difficult airway in the pediatric population. AANA J 2015;83:35-41.
- Shaw KN, Bachur RG, eds. Fleisher & Ludwig’s Textbook of Pediatric Emergency Medicine. 7th ed. Philadelphia: Wolters Kluwer; 2016.
- American Heart Association. Pediatric Advanced Life Support: Provider Manual. Dallas, TX: American Heart Association; 2016.
- Sagarin MJ, Chiang V, Sakles JC, et al. Rapid sequence intubation for pediatric emergency airway management. Pediatr Emerg Care 2002;18:417-423.
- Marvez-Valls E, Houry D, Ernst AA, et al. Protocol for rapid sequence intubation in pediatric patients — a four-year study. Med Sci Monit 2002;8:CR229-34.
- Sakles JC. Improving the safety of rapid sequence intubation in the emergency department. Ann Emerg Med 2017;69:7-9.
- Hayes-Bradley C, Lewis A, Burns B, Miller M. Efficacy of nasal cannula oxygen as a preoxygenation adjunct in emergency airway management. Ann Emerg Med 2016;68:174-180.
- Pourmand A, Robinson C, Dorwart K, O’Connell F. Pre-oxygenation: Implications in emergency airway management. Am J Emerg Med 2017;35:1177-1183.
- Sakles JC, Mosier JM, Patanwala AE, Dicken JM. Apneic oxygenation is associated with a reduction in the incidence of hypoxemia during the RSI of patients with intracranial hemorrhage in the emergency department. Intern Emerg Med 2016;11:983-992.
- Videira RL, Neto PP, do Amaral RV, Freeman JA. Preoxygenation in children: For how long? Acta Anaesthesiol Scand 1992;36:109-111.
- Miller RD, Pardo M. Basics of Anesthesia. 6th ed. Philadelphia: Elsevier Inc; 2011.
- Murat I. Cuffed tubes in children: A 3-year experience in a single institution. Paediatr Anaesth 2001;11:748-749.
- Longnecker DE, Brown DL, Newman MF, Zapol WM. Anesthesiology. New York: McGraw Hill; 2008.
- Karsli C. Managing the challenging pediatric airway: Continuing Professional Development. Can J Anesth 2015;62:1000-1016.
- Kumar HV, Schroeder JW, Gang Z, Sheldon SH. Mallampati score and pediatric obstructive sleep apnea. J Clin Sleep Med 2014;10:985-990.
- Tobias JD, Cravero JP. Procedural Sedation for Infants, Children, and Adolescents. Elk Grove Village, IL: American Academy of Pediatrics; 2016.
- Karsli C, Pehora C, Al-Izzi A, Mathew P. A retrospective review of pediatric difficult airways: Once easy, not always easy. Can J Anaesth 2016;63:776-777.
- Bai W, Golmirzaie K, Burke C, et al. Evaluation of emergency pediatric tracheal intubation by pediatric anesthesiologists on inpatient units and the emergency department. Paediatr Anaesth 2016;26:384-391.
- Shafi Ahmed MA, Al-Ghamdi AA, Mowafi HA, et al. The use of laryngeal mask airway during transesophageal echocardiography in pediatric patients. Saudi J Anaesth 2014;8:489.
- Chen L, Hsiao AL. Randomized trial of endotracheal tube versus laryngeal mask airway in simulated prehospital pediatric arrest. Pediatrics 2008;122:e294-e297.
- Coté CJ, Goldstein EA, Coté MA, et al. A single-blind study of pulse oximetry in children. Anesthesiology 1988;68:184-188.
- Langhan ML, Shabanova V, Li FY, et al. A randomized controlled trial of capnography during sedation in a pediatric emergency setting. Am J Emerg Med 2015;33:25-30.
- Langhan ML, Chen L. Current utilization of continuous end-tidal carbon dioxide monitoring in pediatric emergency departments. Pediatr Emerg Care 2008;24:211-213.
- Langhan ML, Auerbach M, Smith AN, Chen L. Improving detection by pediatric residents of endotracheal tube dislodgement with capnography: A randomized controlled trial. J Pediatr 2012;160:1009-1014.
- Reid C, Chan L, Tweeddale M. The who, where, and what of rapid sequence intubation: Prospective observational study of emergency RSI outside the operating theatre. Emerg Med J 2004;21:296-301.
- Forman S. Clinical and molecular pharmacology of etomidate. Anesthesiology 2011;114:695-707.
- Zuckerbraun NS, Pitetti RD, Herr SM, et al. Use of etomidate as an induction agent for rapid sequence intubation in a pediatric emergency department. Acad Emerg Med 2006;13:602-609.
- Sokolove PE, Price DD, Okada P. The safety of etomidate for emergency rapid sequence intubation of pediatric patients. Pediatr Emerg Care 2000;16:18-21.
- Baxter AL, Mallory MD, Spandorfer PR, et al. Etomidate versus pentobarbital for computed tomography sedations: Report from the Pediatric Sedation Research Consortium. Pediatr Emerg Care 2007;23:690-695.
- Guldner G, Schultz J, Sexton P, et al. Etomidate for rapid-sequence intubation in young children: Hemodynamic effects and adverse events. Acad Emerg Med 2003;10:134-139.
- Smith DC, Bergen JM, Smithline H, Kirschner R. A trial of etomidate for rapid sequence intubation in the emergency department. J Emerg Med 2000;18:13-16.
- Cushman JT, Fairbanks RJ, O’Gara KG, et al. Ambulance personnel perceptions of near misses and adverse events in pediatric patients. Prehosp Emerg Care 2010;14:477-484.
- Cies JJ, Moront ML, Moore WS, et al. Use of etomidate for rapid sequence intubation (RSI) in pediatric trauma patients: An exploratory national survey. Pharmacy 2015;3:197-209.
- Bergman SA. Ketamine: Review of its pharmacology and its use in pediatric anesthesia. Anesth Prog 1999;46:10-20.
- Scheier E, Gadot C, Leiba R, Shavit I. Sedation with the combination of ketamine and propofol in a pediatric ED: A retrospective case series analysis. Am J Emerg Med 2015;33:815-817.
- Cotsen MR, Donaldson JS, Uejima T, Morello FP. Efficacy of ketamine hydrochloride sedation in children for interventional radiologic procedures. AJR Am J Roentgenol 1997;169:1019-1022.
- Al-Nahhas S. Towards evidence based emergency medicine: Best BETs from the Manchester Royal Infirmary. BET 2: Do wrist splints need to have a thumb extension when immobilising suspected scaphoid fractures? Emerg Med J 2011;28:1075-1076.
- Green SM, Andolfatto G, Krauss BS. Ketamine and intracranial pressure: No contraindication except hydrocephalus. Ann Emerg Med 2015;65:52-54.
- Bar-Joseph G, Guilburd Y, Tamir A, Guilburd JN. Effectiveness of ketamine in decreasing intracranial pressure in children with intracranial hypertension. J Neurosurg Pediatr 2009;4:40-46.
- Wadia S, Bhola R, Lorenz D, et al. Ketamine and intraocular pressure in children. Ann Emerg Med 2014;64:385-388.e1.
- Drayna PC, Estrada C, Wang W, et al. Ketamine sedation is not associated with clinically meaningful elevation of intraocular pressure. Am J Emerg Med 2012;30:1215-1218.
- Brown L, Christian-Kopp S, Sherwin TS, et al. Adjunctive atropine is unnecessary during ketamine sedation in children. Acad Emerg Med 2008;15:314-318.
- Veselis RA, Reinsel RA, Feshchenko VA, Johnson R Jr. Information loss over time defines the memory defect of propofol: A comparative response with thiopental and dexmedetomidine. Anesthesiology 2004;101:831-841.
- Muñoz HR, Cortínez LI, Ibacache ME, Altermatt FR. Estimation of the plasma effect site equilibration rate constant (ke0) of propofol in children using the time to peak effect: Comparison with adults. Anesthesiology 2004;101:1269-1274.
- Kamat PP, McCracken CE, Gillespie SE, et al. Pediatric critical care physician-administered procedural sedation using propofol: A report from the Pediatric Sedation Research Consortium Database. Pediatr Crit Care Med 2015;16:11-20.
- Cravero JP, Beach ML, Blike GT, et al; Pediatric Sedation Research Consortium. The incidence and nature of adverse events during pediatric sedation/anesthesia with propofol for procedures outside the operating room: A report from the Pediatric Sedation Research Consortium. Anesth Analg 2009;108:795-804.
- Meakin GH. Neuromuscular blocking drugs in infants and children. Contin Educ Anaesthesia, Crit Care Pain 2007;7:143-147.
- Fleming B, McCollough M, Henderson HO. Myth: Atropine should be administered before succinylcholine for neonatal and pediatric intubation. CJEM 2005;7:114-117.
- Orebaugh SL. Succinylcholine: Adverse effects and alternatives in emergency medicine. Am J Emerg Med 1999;17:715-721.
- Pallin DJ, Dwyer RC, Walls RM, Brown CA 3rd; NEAR III Investigators. Techniques and trends, success rates, and adverse events in emergency department pediatric intubations: A report from the National Emergency Airway Registry. Ann Emerg Med 2016;67:610-615.e1.
- Mendez DR, Goto CS, Abramo TJ, Wiebe RA. Safety and efficacy of rocuronium for controlled intubation with paralytics in the pediatric emergency department. Pediatr Emerg Care 2001;17:233-236.
- Kim C, Kang HG, Lim TH, et al. What factors affect the success rate of the first attempt at endotracheal intubation in emergency departments? Emerg Med J 2013;30:888-892.
- Miller KA, Kimia A, Monuteaux MC, Nagler J. Factors associated with misplaced endotracheal tubes during intubation in pediatric patients. J Emerg Med 2016;51:9-18.
- Verghese ST, Hannallah RS, Slack MC, et al. Auscultation of bilateral breath sounds does not rule out endobronchial intubation in children. Anesth Analg 2004;99:56-58.
- Mansy HA, O’Connor CJ, Balk RA, Sandler RH. Breath sound changes associated with malpositioned endotracheal tubes. Med Biol Eng Comput 2005;43:206-211.
- Goldberg JS, Rawle PR, Zehnder JL, Sladen RN. Colorimetric end-tidal carbon dioxide monitoring for tracheal intubation. Anesth Analg 1990;70:191-194.
- Beach ML, Cohen DM, Gallagher SM, Cravero JP. Major adverse events and relationship to nil per os status in pediatric sedation/anesthesia outside the operating room: A Report of the Pediatric Sedation Research Consortium. Anesthesiology 2016;124:80-88.
- Overly FL, Wills H, Valente JH. “Not just little adults” — a pediatric trauma primer. R I Med J (2013). 2014;97:27-30.
- Ahmadi A, Bazargan-Hejazi S, Heidari Zadie Z, et al. Pain management in trauma: A review study. J Inj Violence Res 2016;8:89-98.
- Tran DT, Newton EK, Mount VA, et al. Rocuronium versus succinylcholine for rapid sequence induction intubation. Cochrane Database Syst Rev 2015;(10):CD002788.
This article will discuss in detail the approach to the pediatric trauma patient, with a strict focus on the pediatric airway.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
