Targeted Management of Headaches for Emergency Physicians
December 1, 2017
Reprints
AUTHORS
Larry B. Mellick, MD, MS, FAAP, FACEP, Professor of Emergency Medicine and Pediatrics, Augusta University, Augusta, GA
Erin K. Purdy-Payne, MD, Emergency Medicine Resident, Department of Emergency Medicine, Augusta University, Augusta, GA
PEER REVIEWER
Jonathan Glauser, MD, FACEP, Professor, Emergency Medicine, Case Western Reserve University, Faculty, Residency Program in Emergency Medicine, MetroHealth Medical Center, Cleveland, OH
EXECUTIVE SUMMARY
- Migraine headaches are best treated in the ED with neuroleptic agents. Opioids should be avoided.
- Patients who experience aura with a migraine headache have an increased risk of stroke.
- Patients with cluster headaches are treated with 100% oxygen.
- Treatment of medication overuse headache starts with discontinuation of analgesics.
Headaches are one of the most common emergency department (ED) complaints,1 and they also can be one of the most frustrating, for both patients and providers. Most of our patients come to the ED because they are suffering from an attack that is either more severe than usual or refractory to their usual treatment. Headache patients tend to be discharged from the ED with vague diagnoses, such as “headache not otherwise specified,” despite meeting criteria for specific diagnoses such as migraine or cluster headache. Furthermore, this diagnostic non-specificity often is reflected in our management choices, as these patients usually are treated with a one-size-fits-all cocktail,2 and are 20 times more likely to be given opioids than other more tailored and effective treatments.1
Headaches are one of the most prevalent and disabling categories of disorders worldwide.3 Emergency healthcare providers are in a unique position to provide management of acute attacks, exclude emergent causes, educate patients, initiate some forms of preventive treatment, refer patients for appropriate outpatient workup and management of chronic headaches, and clarify and provide feedback regarding diagnostic criteria. The good news is that there are underutilized, evidence-based resources available to help busy ED providers make specific primary headache diagnoses, and choose specific therapies that not only will help headache patients return to baseline and maintain a better quality of life, but also will reduce repeat visits and associated healthcare costs.2
This review will discuss different headache diagnoses as defined by the third edition of the International Classification of Headache Disorders (ICHD-3), as well as evidence-based treatments for each type of headache. In most cases described below, patients may be diagnosed with a “probable” version of each headache (e.g., “probable migraine”) if the patient meets many, but not all, of the diagnostic features. In all cases, the headaches must not be better accounted for by another diagnosis. Universal themes for management of all headaches include identifying and treating underlying causes if applicable, educating patients about the underlying mechanisms and potential triggers for their type of headache, counseling patients about self-care for headaches, and encouraging patients to keep a journal to help them identify and avoid or manage their triggers.4 For a headache decision tree that compares selected categories of headaches by their presenting characteristics, see Figure 1.
Figure 1. Headache Decision Tree
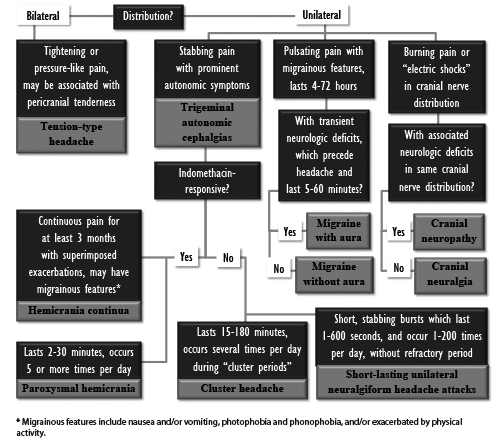
Primary Headaches
Migraine Headaches
Diagnosing Migraines. Migraines have a tendency to be underdiagnosed in spite of their high individual and societal impact.5 The ICDH-3 defines migraines as moderate to severe intensity, with unilateral location, and pulsating quality. They must be accompanied by either nausea and vomiting or photophobia and phonophobia, and they may be aggravated by, or cause avoidance of, routine physical activity. With a few exceptions, migraine duration is between four and 72 hours, and the frequency over time may be chronic or episodic. Patients must have had at least five such attacks before these headaches are defined as migraines, but may be diagnosed as “probable migraines” before this point.
Any migraine headache may be accompanied by premonitory symptoms, such as fatigue or difficulty concentrating, which are not considered auras, and which may begin up to two days prior to the headache. Patients also may present to the ED with extracranial migraine symptoms, such as abdominal pain or vomiting, which are cyclical, with complete return to baseline between episodes.4
Migraines without auras occur due to increased sensitization of pain tracts and neurotransmitters,4 and are not associated with cerebral blood flow changes or increased stroke risk. Conversely, migraines with auras have shown an association with cerebral blood flow changes, and confer a twofold risk of ischemic stroke. Such strokes are more likely to occur in the posterior circulation, and have a much higher predominance in women.4,6 The association between migraine with aura and stroke also is more pronounced in younger patients who otherwise would be unlikely to suffer a stroke,7 and is positively correlated with headache frequency among migraine sufferers.6,7 This risk is compounded by other risk factors, including smoking and oral contraceptive use, which these patients should be counseled to avoid.7 In addition to ischemic stroke, migraines with auras are associated with other rare complications such as infarction or seizure.4
An aura is a syndrome of fully reversible transient neurologic symptoms, at least one of which must be unilateral. By definition, auras onset over five or more minutes, last between five and 60 minutes, and precede the headache onset by up to 60 minutes. Typical auras include visual effects, such as scintillating scotoma (present in a majority of cases), “pins and needles”-type paresthesias, and aphasia (considered to be a unilateral symptom). Less common auras include hemiplegic motor symptoms (which may last up to 72 hours); retinal migraines, which present with a monocular (not hemianopic) visual field defect; and brainstem migraines, which may include symptoms such as alterations in level of consciousness, ataxia, diplopia, vertigo, tinnitus, hyperacusis, or dysarthria. Note that many of these symptoms mimic other, more serious disorders, such as intracranial hemorrhage, transient ischemic attack (TIA), or amaurosis fugax. These symptoms, particularly less common auras or those that the patient has not experienced before, will require further workup to exclude more critical diagnoses.4
Managing Migraines. Patients presenting to the ED with migraines should receive aggressive and targeted management early in their visit.2 The best approach to this aggressive and targeted management, however, is still up for debate.
According to the National Institute for Health and Care Excellence (NICE) guidelines, patients suffering from migraine should be offered a triptan, a nonsteroidal anti-inflammatory drug (NSAID), or acetaminophen, and an antiemetic (regardless of the presence of nausea and vomiting). These guidelines are heavily researched and frequently updated.8 However, they are not specific to emergency healthcare providers or the ED setting, and despite these recommendations, triptans have not been widely accepted for ED use.2,9 Triptans work by causing intracranial vasoconstriction to disrupt the neurovascularly mediated pain cascade. Therefore, triptans should be avoided or used with caution in patients for whom vasoconstriction potentially would be harmful, including patients with coronary artery disease or Prinzmetal’s angina, peripheral artery disease, or any other condition that may affect end-organ perfusion, such as uncontrolled hypertension or sepsis.10,11 In some settings, triptans are not an ideal option for the ED patient population, whose management may be complicated by one or more of the aforementioned comorbidities. Additionally, when used in the ED, triptans are less effective than most other medications, including neuroleptics and other antidopaminergic agents, opioids, and even NSAIDs.12 This may be because triptans work best if taken very early in the headache course,10,11 and many patients present to the ED after hours or days have passed, or after their usual rescue medication has failed.2
The American Headache Society (AHS) also publishes recommendations for acute migraine management, and has found that there is level A evidence for a wide variety of treatments, including triptans, dihydroergotamine, opioids, and over-the-counter analgesics such as acetaminophen, NSAIDs, and combination formulations containing both of these with caffiene.13 However, these recommendations are not specific to the ED patient population or setting, and are based on the evidence for each treatment rather than the quality of the treatments themselves. AHS publishes summaries on “level of evidence” for various therapies. Despite listing opiates, among many other therapies, as having level A evidence (due to the extensive amount of research that has been done on them), neither AHS nor NICE recommend opioids to be applied for actual use in this situation. This is likely due to the fact that they are habit-forming and we are in a national crisis requiring us to curtail opioids except when they are absolutely necessary, as well as the fact that they have been shown to be less effective than other agents and, therefore, not worth the risk of spurring an addiction.
Ergotamines are not recommended by NICE for acute migraine,8 and are associated with an adverse safety profile and side effects, such as nausea and vomiting, not seen with other, more effective medications.12 Likewise, neither NICE nor AHS recommends opioids.8,13 Not only do opioids have a higher propensity to cause medication overuse headaches, which add to the patient’s headache burden as well as abuse potential,5 but they also have demonstrated substantially lower efficacy than other recommended agents,12,14 and a higher rate of return visits to the ED.12 Despite the recommendations and evidence against their use, many patients who present to the ED with migraines continue to receive hydromorphone or other potent opioids.9
Instead of turning to opioids for patients who do not respond to or cannot take triptans, NICE recommends that a parenteral antidopaminergic agent be administered alongside the antiemetic and an NSAID or acetaminophen.8 Antidopaminergic medications encompass a very broad spectrum that includes neuroleptic agents, such as haloperidol, as well as other agents, such as metoclopramide. As a category, neuroleptics have demonstrated a better efficacy for migraine headaches, even with monotherapy, than any other class or combination of agents. Among antidopaminergic agents, neuroleptic or otherwise, the side effect profile is similar, with the most notable effect being akathisia.12 Among neuroleptics, many studies have attempted to differentiate the safest and most efficacious individual agents, with overall equivocal results.12,15 Although some studies have shown droperidol to be the most efficacious,12 others have shown prochlorperazine to be superior.15 The rate of recurrence appears to be similar among all neuroleptics, including haloperidol and chlorpromazine.12
When administering antidopaminergic medications, many providers include adjunct diphenhydramine to reduce the incidence of akathisia. However, a recent randomized, controlled trial of patients receiving metoclopramide for migraine in the ED failed to show that adjunct diphenhydramine either reduced the rate of this side effect or provided more efficacious headache relief.16 Furthermore, a large retrospective study of pediatric migraine patients treated in the ED suggested that diphenhydramine actually may increase the rate of return visits.17
Several other treatment options have been used with varying degrees of success, but require more evidence before they can be recommended. For example, intravenous corticosteroids may be effective in preventing relapse for up to three days after discharge in patients with prolonged migraine symptoms at the time of presentation,12 but there is not sufficient evidence of benefit for this to be routine practice.13 Any perceived benefit should be weighed against the risk posed by recurrent exposure upon repeat visits.18 In particularly refractory cases, there is early evidence that sub-dissociative ketamine infusions may be safe and effective, but more evidence is needed to evaluate this potential strategy as well.19 Finally, several new receptor-targeted therapies are being investigated for treatment of acute migraine, but so far they have not shown enough clinical benefit to be put into use.20
When patients are discharged home, they should be counseled that over-the-counter analgesics can be used for less severe migraines, but rescue medications, such as triptans, should be used only two to three times per week to avoid inducing medication overuse headaches. Therefore, rescue medications should be reserved for severe attacks.10,11
Discharged patients also should be referred to a primary physician or headache specialist for further management. In particular, patients who suffer from headaches more than four days per month should be evaluated for preventive treatment.5
Tension-type Headaches
Diagnosing Tension-type Headaches. Tension-type headaches (TTH) are the most common type of headache, with an estimated lifetime prevalence of up to 78%,4 and the greatest worldwide disease burden of any of the headache disorders.3 They are characterized by mild to moderate intensity pressure- or tightening-type pain in a bilateral distribution. Unlike migraine headaches, tension-type headaches are not associated with physical activity or nausea and vomiting, although they may be associated with photophobia or phonophobia. These headaches may occur in close association with another known causative condition, in which case they should be coded as secondary headaches. They also may be preexisting but increase in frequency or severity in close association with another known causative condition, in which case they should be dual coded as primary tension-type headaches and secondary headaches.4
TTH may or may not be associated with pericranial tenderness of the frontal, temporal, masseter, pterygoid, sternocleidomastoid, splenius, and/or trapezius regions. When pericranial tenderness is present, it commonly is present at baseline and increases during the episode, and may be elicited via manual palpation of the affected areas.4 On examination, the pericranial muscles also may be tense and firm. There is some evidence that in patients prone to TTH, there is a higher level of baseline muscle tension, compounded by a lower threshold and/or increased sensitization that leads to the headache pain.24
Managing Tension-type Headaches. According to the NICE guidelines, these patients should be offered an NSAID or acetaminophen.8 Many studies have shown these treatments to provide only modest benefits for TTHs, and high-quality evidence is surprisingly scant.3,25,26 Over-the-counter formulations combining an NSAID, acetaminophen, and caffeine generally are well tolerated and provide pain relief within 30 minutes for most patients with TTHs,27 but should remain as second-line therapy because of the potential for caffeine withdrawal to cause rebound headaches.28 Multiple guidelines advise against the use of opioids, sedatives, and muscle relaxants because of their potential to cause both dependence and medication overuse headaches.8,28 Guidelines even warn against overuse of over-the-counter analgesics, which also can lead to chronic headaches.
Chronic tension-type headaches may be managed prophylactically with antidepressants, such as tricyclics or selective serotonin reuptake inhibitors (SSRIs), in addition to nonpharmacologic strategies, which tend to target the musculoskeletal component of these headaches.
Trigeminal Autonomic Cephalgias
The trigeminal autonomic cephalgias (TACs) are a group of primary headache disorders with shared characteristics and ipsilateral parasympathetic autonomic symptoms. Specifically, they are defined by sharp, severe, unilateral pain that usually is periorbital or temporal. They are associated with ipsilateral conjunctival injection; pupil constriction; drooping or edema of the eyelid; tearing, congestion, or rhinorrhea; flushing; and/or perspiration. TACs include cluster headaches, paroxysmal hemicrania, hemicrania continua, and short-lasting unilateral neuralgiform headache attacks.4 These conditions are differentiated predominantly by the timing of the attacks and their treatment responses, although there may be overlap of one or both features. These disorders are highly distressing, highly disabling, and fraught with diagnostic delay and treatment failure.29
Cluster headache is the most common type of TAC.30 The attacks last 15-180 minutes, during which patients typically are restless, agitated, unable to sit still, and may pace. The attacks occur several times per day during cluster periods of weeks to months, and frequently are triggered during sleep. Many patients, but not all, experience remission and return to baseline for months to years in between these cluster periods.4
According to the NICE guidelines, patients suffering from acute cluster headache attacks should be offered 100% oxygen and/or a subcutaneous or nasal triptan. They should not be offered other treatments, including over-the-counter analgesics, opioids, or ergots.8 The AHS is mostly in agreement, reporting that level A evidence exists only for 100% oxygen and certain triptan formulations for management of acute attacks, and suboccipital corticosteroid injections for prophylaxis to reduce future attack frequency.30 While NICE recommends 100% oxygen at a rate of at least 12 liters per minute via non-rebreather mask with reservoir bag,8 the AHS systematic review reports efficacy at rates of 6-12 liters per minute.30 NICE also recommends that patients should be discharged with home oxygen for use in future attacks.8
Differentiating between the TACs is important because cluster headaches are the only type that responds to oxygen and triptans. Paroxysmal hemicrania and hemicrania continua respond both acutely and prophylactically to indomethacin with such reliability that the response to this medication is considered a diagnostic feature.4,29 In paroxysmal hemicrania, the attacks last two to 30 minutes and typically occur more than five times per day. In hemicrania continua, the pain is continuous for at least three months, with waxing and waning fluctuations in intensity. As with migraines, exacerbations may be triggered by movement and associated with photophobia and phonophobia. As with cluster headaches, patients may be restless and agitated.4
Short-lasting unilateral neuralgiform headache attacks occur up to 200 times per day, do not have a refractory period (unlike trigeminal neuralgia), and last only 1-600 seconds in a series of short, stabbing bursts.4 They do not respond to oxygen, triptans, or indomethacin. Instead, IV lidocaine or glucocorticoids have been shown to be effective for acute exacerbations, while lamotrigine, topiramate, or gabapentin can be used as preventive maintenance therapy.31 In addition to the periorbital and temporal regions affected by other TACs, the trigeminal distribution may be involved.4 In fact, it has been proposed that these headaches belong in the trigeminal neuralgia family rather than to the trigeminal autonomic cephalgia family. Like trigeminal neuralgia, this condition may be triggered by structural abnormalities, such as microvascular compression of the trigeminal nerve, in which case surgery can be beneficial.29,32
Other Primary Headaches
Primary stabbing headaches present with short bursts of severe stabbing-type sensations that last only a few seconds. They may be bilateral or unilateral and have been reported in a variety of locations, including trigeminal, frontal, temporal, occipital, and nuchal. They may be associated with visual disturbances, nausea and vomiting, or photophobia and phonophobia. Primary stabbing headaches are not associated with cranial autonomic symptoms that define the trigeminal autonomic cephalgias, and likewise do not meet diagnostic criteria for any other types of headaches.4 They also are known by other names, such as “ice pick headache” or “needle in the eye syndrome.”
Primary attacks can occur spontaneously or may be provoked by Valsalva maneuvers, bright lights, or a variety of other triggers. Secondary attacks may be associated with (and in some cases, may be the sentinel symptom of) a variety of other conditions, including intracerebral tumors such as meningiomas, autoimmune conditions such as multiple sclerosis, or cerebrovascular events such as ischemic stroke or hemorrhage. These headaches usually respond to indomethacin, although this response is much less reliable than it is with paroxysmal hemicrania and hemicrania continua. Refractory cases usually are responsive to cyclooxygenase-2 (COX-2) inhibitors, gabapentin, or tricyclic antidepressants.33,34
Primary thunderclap headaches are severe, onset abruptly, and reach their maximum intensity within one minute of onset. While these headaches are diagnosed in the absence of any underlying pathology,4 they mimic several emergent conditions, including subarachnoid hemorrhage, cerebral venous sinus thrombosis, cervical artery dissection, unruptured intracranial aneurysm, reversible cerebral vasoconstriction, or spontaneous intracranial hypotension. Therefore, they must be evaluated via emergent computed tomography scan with computed tomography angiography and/or analysis of the cerebrospinal fluid. Only after these studies are negative should the headache be diagnosed as a primary thunderclap headache.35 There is a paucity of data regarding treatment of the headache pain itself.
Nummular headaches are characterized by pain in a small, round or elliptical, well-circumscribed area of the scalp, in the absence of any structural lesion at the site.4 The pain varies in quality, is of mild to moderate intensity, may be episodic or chronic in its timing, and frequently is associated with focal sensory disturbances in the same well-demarcated area. Secondary nummular headaches may occur as a result of underlying superficial, bony, or intracranial lesions, which should be excluded in an outpatient setting unless other alarming features are present. Primary nummular headaches may be left untreated if symptoms are mild; they may be treated episodically with acetaminophen or NSAIDs; or they may be managed preventively with botulinum injections, tricyclic antidepressants, or gabapentin for severe or refractory cases.36
Hypnic headaches are rare. These headaches begin exclusively during sleep,4 most commonly between 2 a.m. and 4 a.m. They cause the patient to awaken with variable quality pain, which usually is bilateral and of moderate intensity.37 The headaches may be associated with migraine-like features,4 and have a high rate of co-occurrence with migraines.37,38 According to the ICHD-3 diagnostic criteria, hypnic headaches are not associated with cranial autonomic symptoms;4 however, other studies have shown that mild bilateral autonomic symptoms occur.37,38 The ICHD-3 defines hypnic headaches as recurring at least 10 nights per month and lasting up to four hours,4 but most cases occur fewer than 10 times per month, lasting between 15 minutes and eight hours.
Hypnic headaches may be chronic for several years.38 These headaches occur more commonly in patients older than 50 years of age,4 and according to recent MRI data, may be associated with gray matter volume reduction in the hypothalamus4,39 and other areas related to centrally mediated pain.39 The diagnosis of hypnic headache should be made after structural lesions or physiologic disturbances, such as nocturnal arterial hypertension, have been ruled out.37
High-quality data are lacking for management of acute attacks, but anecdotally, many patients report modest relief if they use caffeine upon being awakened by the headache. Lithium seems to be the most effective and well-tolerated agent for preventing the recurrent attacks, and because of its slow onset, should be tried for approximately two months before any decision is made about its efficacy. Many patients also report that mild to moderate exercise decreases the frequency of the attacks. Melatonin, topiramate, and indomethacin have been used, but more data are needed to evaluate these treatments.38
New daily persistent headaches have a distinct starting point in patients with limited or no prior history of headaches, from which point the headaches occur daily for at least three months. They may be migraine-like, tension-type, or have features of both, but are not better accounted for by another headache diagnosis or underlying cause.4 A new structural lesion should be excluded.
This disorder is notoriously difficult to treat, but typically is managed similarly to chronic migraine headaches.40,41 Acute agents include over-the-counter analgesics or triptans, depending on the headache severity, used in such a way as to minimize the risk of inciting medication overuse headaches.
The “other primary headaches” category also includes headaches that have been induced by cough or other type of Valsalva maneuver (which has a higher prevalence in patients with Arnold Chiari type I malformations), strenuous exercise (which is more likely in hot weather or at high altitude), or sexual activity (in which headache intensity increases dramatically just before or simultaneously with orgasm) for which increased intracranial pressure should be excluded.
Other triggers include cold stimulus (commonly referred to as an “ice cream headache”) and external compression or traction (such as due to tight headbands or hair braids). They are classified as primary rather than secondary headaches because their triggers are normal physiologic stimuli that do not cause injury. Treatment of these headaches includes recognition and avoidance or management of the inciting factors, coupled with over-the-counter analgesics as needed for acute episodes.4 For a comparison of selected primary headaches, with key points regarding their characteristics, management, and mimics, see Table 1.
Table 1. Primary Headaches
|
Headache Type |
Characteristics |
Emergency Department Management |
Prophylaxis |
Mimics |
|
|
Migraine headache |
Unilateral, pulsating, severe, 4-72 hours, migrainous features, with or without aura |
Neuroleptics (e.g. droperidol, prochlorperazine) or other anti-dopaminergics, combined with another analgesic (e.g., acetaminophen or an NSAID) and an antiemetic (e.g., promethazine or ondansetron) |
Antiepileptics, botulinum toxin injections |
Ischemic stroke or TIA, intracranial hemorrhage, giant cell arteritis, central venous sinus thrombosis |
|
|
Management alternatives to consider: sub-dissociative ketamine infusions, triptans Management adjuncts to consider: diphenhydramine (for prevention or treatment of akathisia), corticosteroids (for prevention of recurrence) |
|||||
|
Tension-type headache |
Bilateral, tightening or pressure-like, with or without pericranial tenderness |
Over-the-counter analgesics (e.g., an NSAID or acetaminophen), consider a combination formulation with caffeine |
Antidepressants, biofeedback or physical therapy |
Cervicogenic headaches, medication overuse headaches, and many other types of secondary headaches |
|
|
Paroxysmal hemicrania |
Severe, stabbing, with ipsilateral parasympathetic autonomic symptoms |
Lasts 2-30 minutes, occurs several times per day |
Indomethacin |
Indomethacin |
Cluster headaches |
|
Hemicrania continua |
Continuous pain with waxing and waning fluctuations |
||||
|
Cluster headache |
15-180 minutes, several times per day during “cluster periods” |
100% oxygen, subcutaneous or nasal triptan |
Suboccipital corticosteroid injections |
Migraine, paroxysmal hemicrania |
|
|
Short-lasting unilateral neuralgiform headache attacks |
Short stabbing bursts which last a few seconds and occur many times per day |
IV lidocaine or corticosteroids |
Anti-epileptics, gabapentin |
Primary stabbing headaches, trigeminal neuralgia |
|
|
Primary stabbing headaches |
Short stabbing bursts without autonomic symptoms |
Indomethacin, COX-2 inhibitors |
Indomethacin, gabapentin, tricyclic antidepressants |
Short-lasting unilateral neuralgiform headache attacks, trigeminal or other cranial neuralgia |
|
|
Primary thunderclap headache |
Severe, sudden onset, maximal at onset |
Rule out emergent causes |
Intracranial hemorrhage, central venous sinus thrombosis, other acute intracranial vascular problems |
||
|
Nummular headache |
Small, well-circumscribed area of mild pain |
Over-the-counter analgesics (e.g., an NSAID or acetaminophen) |
Only for severe or refractory cases |
Underlying structural lesion |
|
|
Hypnic headache |
Onsets exclusively during sleep (2-4 am), usually moderate bilateral pain |
Analgesic formulations containing caffeine |
Lithium |
Variable, depending on features |
|
|
New daily persistent headaches |
Occur daily for at least 3 months in patients with little to no prior headache history, variable features |
Similar to migraine headaches |
Similar to migraine headaches |
Underlying structural lesion |
|
Secondary Headaches
Secondary headaches are those that occur as a result of another underlying medical problem, and thus are largely outside the scope this paper. While the discussion of these headaches will be limited in this review, considering and ruling out secondary causes are some of the most important first steps in managing headaches in the ED.4
Many secondary headaches occur as a result of other, less emergent conditions, such as infections like sinusitis or influenza, systemic diseases such as hypertensive crisis or diabetic ketoacidosis, or simple cases of caffeine withdrawal, dehydration, or hangover. In these cases, a thorough history and physical exam will allow the emergency provider to identify and treat the underlying cause appropriately. Additionally, in patients with new onset of persistent or otherwise atypical headaches that have not been worked up, intracranial disorders such as neoplasm or unruptured vascular malformation should be considered. Systemic illnesses may predispose some patients to cerebral vasculitis.4
A few secondary headaches will be discussed in more detail. For a comparison of these disorders, with key points regarding their mimics, differentiating features, and management, see Table 2.
Table 2. Secondary Headaches
Headache |
May Mimic |
Differentiating Features |
Emergency Department Management |
|
Giant cell arteritis |
Migraine, certain cranial neuropathies |
Ipsilateral monocular vision changes (very rare with migraine); may also have scalp tenderness, jaw claudication, fevers, polymyalgia |
High-dose corticosteroids, arrange for temporal artery biopsy |
|
Central venous sinus thrombosis |
Migraine, encephalopathy, intracranial hemorrhage, ischemic stroke, or other intracranial vascular problems |
Highly variable, may have neurologic findings including seizures, vision changes, focal deficits, alterations of mental status or level of consciousness |
Anticoagulation, antiepileptic prophylaxis, monitoring and management of intracranial pressure |
|
Medication overuse headache |
Migraine or tension-type headaches |
Worsening or new type of headache in the setting of chronic overuse of medication intended for acute headache management |
Tapered or abrupt withdrawal of acute medications, optimize preventative management of chronic headaches |
|
Cervicogenic headache |
Tension-type headaches, occipital neuralgia |
Headache onsets in neck before spreading to head, may have bony tenderness and/or signs of cervical nerve root compression |
Consider imaging of cervical spine to evaluate for underlying problem if acute |
Giant Cell Arteritis
Giant cell arteritis (GCA) is a form of granulomatous vasculitis that affects small vessels in the head and neck, particularly branches of the external carotid artery, and frequently the temporal artery. Temporal GCA usually is characterized by a temporal headache associated with ipsilateral monocular vision changes, but also should be considered for any elderly patient with new persistent headache or recurrent episodes of amaurosis fugax. It may mimic other disorders, including optic neuritis and retinal migraine, although the latter is very rare and should not be diagnosed until an extensive workup has ruled out other conditions.4 GCA also may involve scalp tenderness, claudication of the jaw or tongue, and systemic symptoms, such as fever or polymyalgia, although these symptoms are not always present, making diagnosis very difficult and frequently delayed.42 GCA is two times more common in women than in men. It also is rare before 50 years of age.4,42 When GCA is suspected, high-dose steroids should be initiated as soon as possible to avoid permanent vision loss. Diagnosis should be confirmed later via temporal arterial biopsy. While GCA may present unilaterally or bilaterally, temporal arteritis typically affects both sides, with contralateral vision loss usually occurring within one week.4
Cerebral Venous Sinus Thrombosis
Cerebral venous sinus thrombosis most frequently presents with headache, which may be mistaken for other types of headaches. Duration of onset varies, from “thunderclap” to gradually over several days, and in presentation, from unilateral and migraine-like to diffuse and poorly localized. Less than half of patients with this condition will present with other symptoms such as encephalopathy, seizures, vision changes, or stroke-like symptoms, including unilateral weakness of the face or extremities. Less commonly, signs of increased intracranial pressure may be present. As with many conditions, elderly patients may show only nonspecific decline in mentation or level of consciousness, but with no other symptoms.4 The condition affects patients at a median age of 37 years, and, like many other headache disorders, is far more common in females. It is associated with known risk factors for venous thromboembolic events, including oral contraceptive use.43
Cerebral venous sinus thrombosis is diagnosed by magnetic resonance venogram, although other advanced imaging with venography can be used. It is treated with anticoagulation, antiepileptic prophylaxis, and monitoring and management of any complications, such as increased intracranial pressure. In certain cases, thrombolysis and even endovascular intervention also may be considered.44
Medication Overuse Headache
Medication overuse headache is diagnosed when there is a new type or worsening of a preexisting headache, which occurs at least 15 days per month, in the setting of at least three months of chronic overuse of medication intended for management of acute headaches.4 Today, chronic migraines are the most common underlying headache disorder, and triptans or over-the-counter analgesics are the most commonly associated overused acute medications. Research has shown an association with structural and functional changes in several parts of the brain responsible for mediating and transmitting pain signals, suggesting that this disorder is a result of pain processing dysregulation.46
Treatment includes discontinuing acute headache medications; initiating better management of the underlying chronic headache disorder, including prophylactic medications as well as nonpharmacologic treatments; and counseling patients on the nature of this disorder and the optimal use of targeted rescue medications for breakthrough headaches.46,47 While tapered withdrawal of headache medicines is associated with milder symptoms, abrupt withdrawal is favored as it produces faster return to baseline.45 Conversely, it has been suggested that withholding pain-relieving medicines from patients with headaches is irresponsible and may cause more harm than benefit.48
Cervicogenic Headaches
Many headache patients report associated neck symptoms.49 Headaches originating from other disorders of the cervical spine, discs, tendons, or ligaments are classified as cervicogenic headaches, a subcategory of secondary headaches.4 In primary headache disorders, the headache begins first, with associated neck muscle tightness and spasms developing later. Conversely, in cervicogenic headaches, the neck pain tends to precede the headache and is associated with ipsilateral shoulder or arm pain, and the headache tends to begin in the occipital region before generalizing anteriorly.
Management of cervicogenic headaches begins with identification of the underlying cervical disorder, which ultimately may be corrected with surgery or managed symptomatically with over-the-counter analgesics, topical menthol creams or heating pads, physical therapy, and trigger point or greater occipital nerve injections of local anesthetic and/or corticosteroid. Patients also should be educated on techniques to minimize further neck strain or injury.49
Cranial Neuropathies and Other Facial Pains
The cranial neuralgias and neuropathies are neuropathic pain disorders of the head and face characterized by short bursts of intense, sharp, unilateral pain, said to feel like electric shocks, and which recur in an episodic fashion. The term cranial neuralgia is used when there is only neuropathic pain without any accompanying neurologic deficits. When neurologic deficits are present, this is referred to as a cranial neuropathy. Such neurologic deficits are indicative of axonal damage due to infectious etiologies such as herpes zoster, autoimmune disorders such as multiple sclerosis, or mass effect from space-occupying lesions.4
The most well-known of the cranial neuralgias is trigeminal neuralgia. Unlike the trigeminal autonomic cephalalgias, trigeminal neuralgia attacks usually are followed by a refractory period during which another attack cannot be triggered, and are not usually associated with autonomic manifestations, although mild symptoms such as eye redness and tearing may be present. These attacks may occur spontaneously or in response to a benign external stimulus, and may or may not be superimposed on a baseline of constant, moderate facial pain. Over time, they may become longer-lasting and more severe. The classic form usually occurs in the distribution of the second or third branches of the trigeminal nerve, and is caused by compression of the trigeminal nerve via nearby structures, such as the superior cerebellar artery.4
Similar syndromes may occur in other cranial nerve distributions. For example, glossopharyngeal neuropathy causes symptoms in the ear, angle of the jaw, and other pharyngeal tissues innervated by the glossopharyngeal nerve; occipital neuropathy, in the posterior scalp; and facial nerve neuropathy, deep in the external auditory canal. Periorbital pain exacerbated by extraocular movements may be indicative of optic neuritis, which may be associated with an underlying multiple sclerosis lesion; ischemic oculomotor nerve palsy, which occurs secondary to microvascular disease caused by systemic illnesses such as poorly controlled diabetes mellitus; Tolosa-Hunt syndrome, which is caused by granulomatous infiltration of one or more cranial nerves; or recurrent painful ophthalmoplegic neuropathy, previously called ophthalmoplegic migraine, which affects one or more ocular motor nerves and is not associated with an underlying lesion. In contrast, paratrigeminal oculosympathetic syndrome causes constant pain in the ophthalmic division of the trigeminal nerve and also is associated with Horner’s syndrome. It is caused by underlying pathology of this nerve branch involving the carotid artery or elsewhere in the middle cranial fossa.4
Facial pain syndromes that do not follow cranial nerve distributions include burning mouth syndrome, a superficial burning-type glossodynia, and persistent idiopathic facial pain, an aching-type, poorly localized facial pain. In both cases, symptoms occur for more than two hours per day every day for at least three months. Finally, central neuropathic pain is a nebulously defined pain syndrome in which patients experience neuropathic-type pain that is temporally attributed to an underlying central condition, such as multiple sclerosis or stroke. The distribution of the pain is not necessarily dependent on the location of the underlying lesion, and may be unilateral or bilateral, with or without sensory changes.4
Management of cranial neuralgias and neuropathies should include referral for outpatient identification and management of underlying causes. Management of pain includes topical anesthetics for acute flares, and tricyclic antidepressants or antiepileptics, sometimes in conjunction with psychotherapy, for prophylaxis.50
Common Themes and New Directions in Headache Management
Despite the multitude of different definitions and etiologies for various headache disorders, there clearly are shared pathophysiologic mechanisms. Many headaches appear to share common pathophysiologic pathways in the trigeminovascular system and dysregulation of nociceptive signaling, in addition to other characteristics such as their relapsing-remitting episodic nature, their higher prevalence in female patients, and their cross-association with other types of headaches.51
A popular theory that suggests all headaches share a common pathway has led to research into various treatments that may work across different categories of headache types. For example, while botulinum toxin injections have been investigated and used primarily as a prophylactic treatment for chronic migraine, there is emerging evidence that they also may be effective in reducing the frequency of many other chronic primary and secondary headaches.53 While surgical decompression of the trigeminal nerve historically has been a treatment for trigeminal neuralgia, up to 90% of patients with refractory migraine headaches experience a reduction in symptoms by surgical decompression of the trigeminal, cervical spinal, or other peripheral nerves, suggesting that these disorders also may have more in common than we realize.54 Other authors point out that there is significant overlap and that, for example, tension headaches and migraine headaches are distinguished in diagnosis by nuances of questionable reliability.59 The abundant similarities in the epidemiology, clinical features, and treatment response patterns between the two types of headaches suggests that they are sharing common pathophysiology.59
While the purpose of this paper is to discuss the targeted approaches that currently are superior for most specific headache diagnoses, there may come a day when a greater understanding of the shared pathophysiology of headaches allows for greater optimization of their management.
REFERENCES
- Burch RC, Loder S, Loder E, Smitherman TA. The prevalence and burden of migraine and severe headache in the United States: Updated statistics from government health surveillance studies. Headache 2015;55:21-34.
- Cerbo R, Villani V, Bruti G, et al. Primary headache in emergency department: Prevalence, clinical features and therapeutical approach. J Headache Pain 2005;6:287-289.
- Stovner L, Hagen K, Jensen R, et al. The global burden of headache: A documentation of headache prevalence and disability worldwide. Cephalalgia 2007;27:193-210.
- Headache Classification Committee of the International Headache Society. The International Classification of Headache Disorders, 3rd edition (beta version). Cephalalgia 2013;33:629-808.
- Miller S, Matharu MS. Migraine is underdiagnosed and undertreated. Practitioner 2014;258:19-24,2-3.
- Ferrari MD. Headache: The changing migraine brain. Lancet Neurol 2013;12:6-8.
- Kurth T, Chabriat H, Bousser MG. Migraine and stroke: A complex association with clinical implications. Lancet Neurol 2012;11:92-100.
- National Institute for Health and Care Excellence. Headaches in over 12s: Diagnosis and management. Clinical guideline (CG150). 2015.
- Friedman BW, West J, Vinson DR, et al. Current management of migraine in US emergency departments: An analysis of the National Hospital Ambulatory Medical Care Survey. Cephalalgia 2015;35:301-309.
- Silberstein SD. Migraine. Lancet 2004;363:381-391.
- Cameron C, Kelly S, Hsieh SC, et al. Triptans in the acute treatment of migraine: A systematic review and network meta-analysis. Headache 2015;55Suppl 4:221-235.
- Sumamo Schellenberg E, Dryden DM, Pasichnyk D, et al. Acute Migraine Treatment in Emergency Settings [Internet]. Rockville (MD): Agency for Healthcare Research and Quality (US). 2012 Nov. Report. No. 12(13)-EHC142-EF.
- Marmura MJ, Silberstein SD, Schwedt TJ. The acute treatment of migraine in adults: The American Headache Society evidence assessment of migraine pharmacotherapies. Headache 2015;55:3-20.
- Friedman BW, Irizarry E, Solorzano C, et al. Randomized study of IV prochlorperazine plus diphenhydramine vs IV hydromorphone for migraine. Neurology 2017;89:2075-2082.
- Kelley NE, Tepper DE. Rescue therapy for acute migraine part 2: Neuroleptics, antihistamines, and others. Headache 2012;52:292-306.
- Friedman BW, Cabral L, Adewunmi V, et al. Diphenhydramine as adjuvant therapy for acute migraine: An emergency department-based randomized clinical trial. Ann Emerg Med 2016;67:32-39.e3.
- Bachur RG, Monuteaux MC, Neuman MI. A comparison of acute treatment regimens for migraine in the emergency department. Pediatrics 2015;135:232-238.
- Gefland AA, Goadsby PJ. A neurologist’s guide to acute migraine therapy in the emergency room. Neurohospitalist 2012;2:51-59.
- Diener HC, Charles A, Goadsby PJ, Holle D. New therapeutic approaches for the prevention and treatment of migraine. Lancet Neurol 2015;14:1010-1022.
- Lauritsen C, Mazuera S, Lipton RB, Ashina S. Intravenous ketamine for subacute treatment of refractory chronic migraine: A case series. J Headache Pain 2016;17:106.
- Magis D, Schoenen J. Advances and challenges in neurostimulation for headaches. Lancet Neurol 2012;11:708-719.
- Aurora SK, Winner P, Freeman MC, et al. OnabotulinumtoxinA for treatment of chronic migraine: Pooled analyses of the 56-week PREEMPT clinical program. Headache 2011;51:1358-1373.
- Blumenfeld A, Silberstein SD, Dodick DW, et al. Method of injection of OnabotulinumtoxinA for chronic migraine: A safe, well-tolerated, and effective treatment paradigm based on the PREEMPT clinical program. Headache 2010;50:1406-1418.
- Bendtsen L, Ashina S, Moore A, Steiner TJ. Muscles and their implications for episodic tension-type headache: Implications for treatment. Eur J Pain 2016;20:166-175.
- Barbanti P, Egeo G, Aurilia C, Fofi L. Treatment of tension-type headache: From old myths to modern concepts. Neurol Sci 2014;35Suppl 1:17-21.
- Moore RA, Derry S, Wiffen PJ, et al. Evidence for efficacy of acute treatment of episodic tension-type headache: Methodological critique of randomised trials for oral treatments. Pain 2014;155:2220-2228.
- Gaul C, Grater H, Weiser T. Results from a pharmacy-based patient survey on the use of a fixed combination of analgesic containing acetylsalicylic acid, paracetamol and caffeine by self-diagnosing and self-treating patients. Springerplus 2016;5:721.
- Bendtsen L, Evers S, Linde M, et al. EFNS guideline on the treatment of tension-type headache — Report of an EFNS task force. Eur J Neurol 2010;17:1318-1325.
- Barloese MCJ. The pathophysiology of the trigeminal autonomic cephalalgias, with clinical implications. Clin Auton Res 2017 Sep 23. doi: 10.1007/s10286-017-0468-9. [Epub ahead of print.]
- Robbins MS, Starling AJ, Pringsheim TM, et al. Treatment of cluster headache: The American Headache Society evidence-based guidelines. Headache 2016;56:1093-1106.
- Baraldi C, Pellesi L, Guerzoni S, et al. Therapeutical approaches to paroxysmal hemicrania, hemicrania continua and short lasting unilateral neuralgiform headache attacks: A critical appraisal. J Headache Pain 2017;18:71.
- Benoliel R, Sharav Y, Haviv Y, Almoznino G. Tic, triggering, and tearing: From CTN to SUNHA. Headache 2017;57:997-1009.
- Chua AL, Nahas S. Ice pick headache. Curr Pain Headache Rep 2016;20:30.
- Hagler S, Ballaban-Gil K, Robbins MS. Primary stabbing headache in adults and pediatrics: A review. Curr Pain Headache Rep 2014;18:450.
- Schwedt TJ, Matharu MS, Dodick DW. Thunderclap headache. Lancet Neurol 2006;5:621-631.
- Schwartz DP, Robbins MS, Grosberg BM. Nummular headache update. Curr Pain Headache Rep 2013;17:340.
- Ruiz M, Mulero P, Pedraza MI, et al. From wakefulness to sleep: Migraine and hypnic headache association in a series of 23 patients. Headache 2015;55:167-173.
- Tariq N, Estemalik E, Vij B, et al. Long-term outcomes and clinical characteristics of hypnic headache syndrome: 40 patient series from a tertiary referral center. Headache 2016;56:714-724.
- Holle D, Naegel S, Krebs S, et al. Hypothalamic gray matter volume loss in hypnic headache. Ann Neurol 2011;69:533-539.
- Robbins MS, Vanast WJ, Allan Purdy R. New daily persistent headache: Historical review and an interview with Dr. Walter Vanast. Headache 2017;57:926-934.
- Couch JR. Update on chronic daily headache. Curr Treat Options Neurol 2011;13:41-55.
- Prior JA, Ranjbar H, Belcher M, et al. Diagnostic delay for giant cell arteritis — a systematic review and meta-analysis. BMC Med 2017;15:120.
- Ferro JM, Canhao P, Stam J, et al; ISCVT Investigators. Prognosis of cerebral vein and dural sinus thrombosis: Results of the International Study on Cerebral Vein and Dural Sinus Vein Thrombosis (ISCVT). Stroke 2004;35:664-670.
- Saposnik G, Barinagarrementeria F, Brown RD Jr, et al; American Heart Association Stroke Council and the Council on Epidemiology and Prevention. Diagnosis and management of cerebral veous thrombosis: A statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2011;42:1158-1192.
- Evers S, Marziniak M. Clinical features, pathophysiology, and treatment of medication-overuse headache. Lancet Neurol 2010;9:391-401.
- Krymchantowski AV, Jevoux CDC. Medication-overuse headache. Despite the advances in understanding it, treatment evidence still lacks. Expert Rev Neurother 2017;17:1055-1058.
- Negro A, Curto M, Lionetto L, et al. A critical evaluation on MOH current treatments. Curr Treat Options Neurol 2017;19:32.
- Scher AI, Rizzoli PB, Loder EW. Medication overuse headache: An entrenched idea in need of scrutiny. Neurology 2017;89:1296-1304.
- Gallagher RM. Cervicogenic headache. Expert Rev Neurother 2007;7:1279-1283.
- Shephard MK, Macgregor EA, Zakrzewska JM. Orofacial pain: A guide for the headache physician. Headache 2014;54:22-39.
- Tassorelli C. The grand challenge in cranial pain — From migraine to cranial neuralgias: Understanding differences and similarities to advance knowledge and management. Front Neurol 2017;8:19.
- Forderreuther S, Ruscheweyh R. From ophthalmoplegic migraine to cranial neuropathy. Curr Pain Headache Rep 2015;19:21.
- Sandrini G, De Icco R, Tassorelli C, et al. Botulinum neurotoxin type A for the treatment of pain: Not just in migraine and trigeminal neuralgia. J Headache Pain 2017;18:38.
- Janus JE, Barker JC, Javadi C, et al. A review of current evidence in the surgical treatment of migraine headaches. Plast Reconstr Surg 2014;134(4 Suppl 2):131S-41S.
- Levin M. Nerve blocks in the treatment of headache. Neurotheraputics 2010;7:197-203.
- Mellick LB, McIlrath ST, Mellick GA. Treatment of headaches in the ED with lower cervical intramuscular bupivacaine injections: A 1-year retrospective review of 417 patients. Headache 2006;46:1441-1449.
- Mellick LB, Pleasant MR. Do pediatric headaches respond to bilateral lower cervical paraspinous bupivaicaine injections? Pediatr Emerg Care 2010;26:192-196.
- Ahmed HE, White PF, Craig WF, et al. Use of percutaneous electrical nerve stimulation (PENS) in the short-term management of headache. Headache 2000;40:311-315.
- Cady R, Schreiber C, Farmer K, Sheftell F. Primary headaches: A convergence hypothesis. Headache 2002;42:204-216.
Headaches are one of the most prevalent and disabling categories of disorders worldwide. Emergency healthcare providers are in a unique position to provide management of acute attacks, exclude emergent causes, educate patients, initiate some forms of preventive treatment, refer patients for appropriate outpatient workup and management of chronic headaches, and clarify and provide feedback regarding diagnostic criteria.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
