Advanced Imaging in Acute Ischemic Stroke
December 15, 2017
Reprints
AUTHORS
A. John Tsiouris, MD, Associate Professor of Clinical Radiology, Director of MRI, Department of Radiology, Weill Cornell Medicine, New York, NY
Jenny Qian, MD, Department of Radiology, Weill Cornell Medicine, New York, NY
PEER REVIEWER
Benjamin R. Kummer, MD, Clinical Informatics Fellow, New York-Presbyterian Hospital, New York, NY; Assistant Clinical Professor of Neurology, Mount Sinai Health System, New York, NY
EXECUTIVE SUMMARY
- During initial non-contrast computed tomography (CT) imaging of a suspected stroke, consider obtaining a CT angiogram and perfusion study, if additional time will not delay administration of indicated systemic tPA therapy.
- The non-contrast CT scan can be used to calculate the Alberta Stroke Program Early CT Score (ASPECTS), a method to quantify the extent of middle cerebral artery territory infarction to predict outcome after systemic thrombolytic therapy.
- Magnetic resonance imaging with diffusion-weighted imaging is the gold standard for acute ischemic stroke.
- Magnetic resonance angiography of the intracranial vessels can be obtained without the use of contrast using the time-of-flight gradient recall echo technique.
- CT perfusion imaging can identify “at risk” or salvageable tissue (the ischemic penumbra) potentially recoverable with reperfusion therapy.
Overview
Acute ischemic stroke is a leading cause of death in the United States, accounting for approximately one in every 20 deaths,1,2 and is a major cause of long-term disability.3 Every year, approximately 795,000 people will experience a new or recurrent acute ischemic stroke, and an estimated 6.6 million Americans older than 20 years of age already have experienced one.2 Acute ischemic stroke occurs when there is a disruption of the blood supply to the brain due to an acute vessel occlusion, which may be either embolic in nature or form in situ at a ruptured arterial plaque. Therefore, treatments are aimed at reperfusing the affected brain parenchyma to salvage at-risk tissue before it infarcts in order to improve clinical outcomes. Timely workup, including imaging, and management of a patient presenting with stroke are paramount.
Timely systemic intravenous administration of recombinant tissue plasminogen activator (tPA) remains the mainstay of treatment for acute ischemic stroke. Although systemic tPA increases survival with favorable functional outcomes, still more than half of treated patients either will fail to recover completely or will die.4,5 Early treatment, particularly within three hours of symptom onset, is associated with the best clinical outcomes,4-6 although current guidelines allow for the treatment window to be extended to 4.5 hours in selected patients.7 In a patient who is eligible for systemic thrombolytic therapy, early imaging evaluation is crucial to avoid delaying effective treatment.
In recent years, newer-generation thrombectomy devices have come into use, and there are robust clinical trials supporting their use in selected patients.8-12 Thus, advanced imaging plays an increasingly important role in the initial evaluation of acute ischemic stroke to identify patients who are the most likely to benefit from the intervention and who are the least likely to experience potentially catastrophic adverse events. Advanced imaging evaluation may be particularly important in extending the treatment window for these selected patients, many of whom will present outside of the treatment window for systemic thrombolytic therapy. Any clinician who may have a role in the initial triage and management of these patients would be well-served to have an understanding of the currently available imaging modalities and techniques, and the applications of each in the evaluation of acute ischemic stroke.
Structural Imaging
For the initial evaluation and diagnosis of acute stroke, structural imaging is key to characterize the nature and extent of the insult as well as to identify or exclude alternate etiologies for an acute neurologic deficit, such as an intracranial mass, extra-axial collection, or hemorrhage. (See Figure 1.) Both computed tomography (CT) and magnetic resonance (MR) may be used; each has its distinct strengths and weaknesses.
Figure 1. Acute Stroke
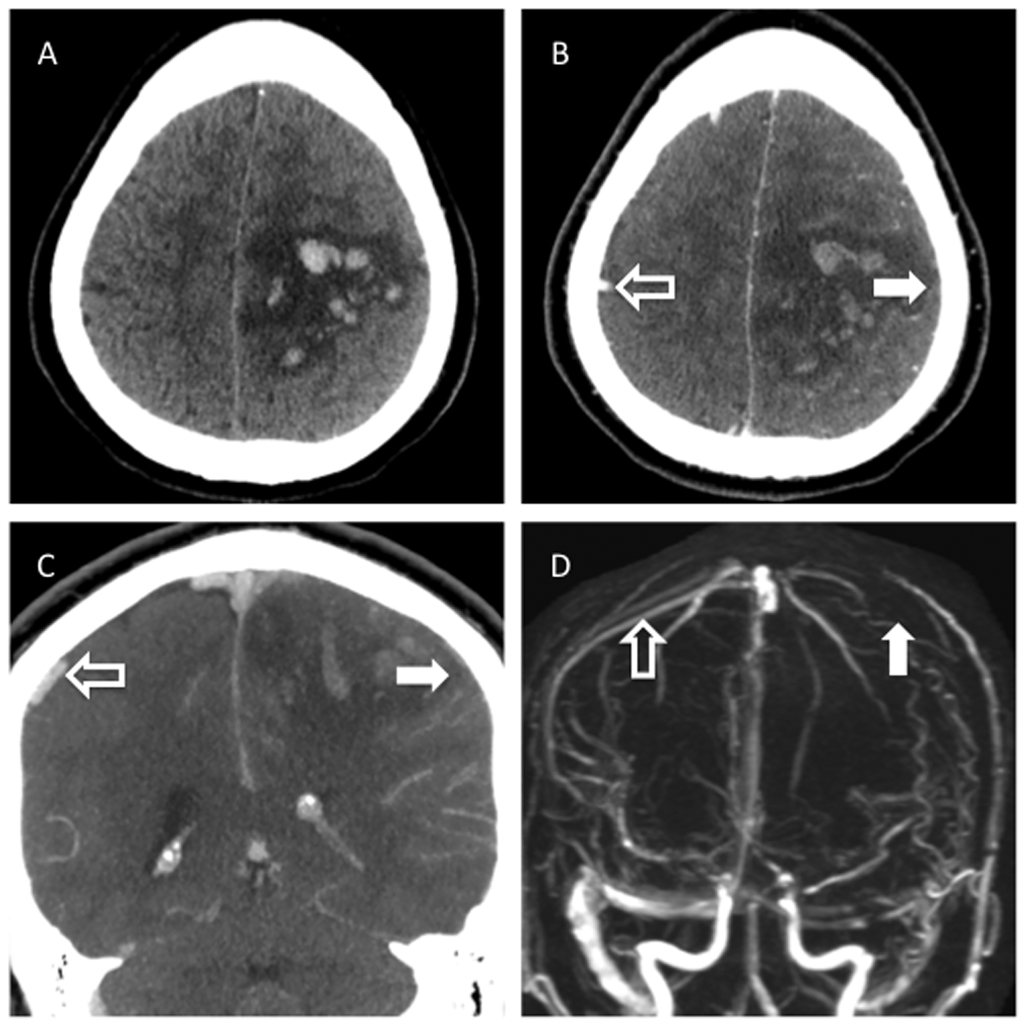
A patient who presented with an acute stroke. A) Axial NCCT shows an area of hemorrhage and edema in the left frontoparietal region. B, C) Axial and coronal CT venography demonstrates lack of contrast within the left vein of Trolard (solid white arrows), a major superficial cerebral vein. Note the contrast within the contralateral vein of Trolard (outlined arrows). D) 3D maximum-intensity reconstruction from MR venography demonstrates the same finding — a normal vein of Trolard on the right (outlined arrow), which is missing from the symptomatic side (solid white arrow). Imaging in this patient confirmed the patient’s stroke was related to a large cortical vein thrombosis resulting in hemorrhagic venous infarction, and not an arterial acute ischemic stroke. tPA would be contraindicated.
Non-contrast Computed Tomography
Non-contrast computed tomography (NCCT) remains the mainstay of first-line imaging for acute stroke. It is widely available, can be performed and interpreted expeditiously, and can detect the presence of acute hemorrhage with high sensitivity13 — almost 100% in the setting of acute subarachnoid hemorrhage14 — and is excellent at depicting calcifications. (See Figure 2.) In addition, there are almost no contraindications for its use.
A major disadvantage of NCCT in the evaluation of acute ischemic stroke is that in the hyperacute phase, signs of early ischemia may be subtle and difficult to detect or altogether absent, with a poor sensitivity reported at 58% in one study.15 The earliest sign of acute ischemic stroke on NCCT may be a hyperdense middle cerebral artery (MCA) segment, indicating the presence of a thrombus or thromboembolism. (See Figure 3.)
Figure 2. Calcified Embolus in Middle Cerebral Artery
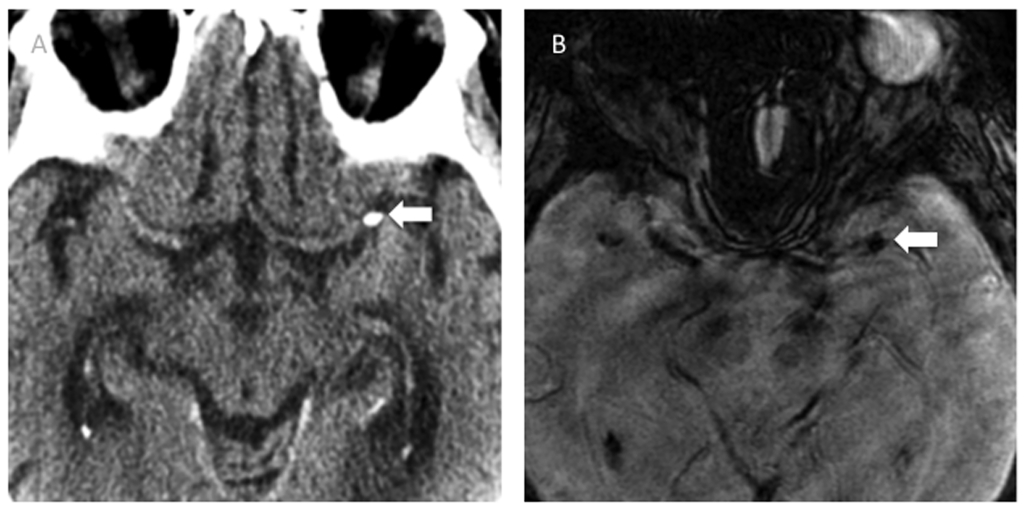
After a transcatheter aortic valve replacement for severe aortic stenosis, this patient was noted by a nurse to have expressive aphasia. A) NCCT demonstrated a heavily calcified embolus in the left MCA. B) “Blooming” artifact can be seen on MR susceptibility-weighted imaging corresponding to the calcification, but the finding is clearly more conspicuous on CT.
Figure 3. Hyperdense Middle Cerebral Artery Sign
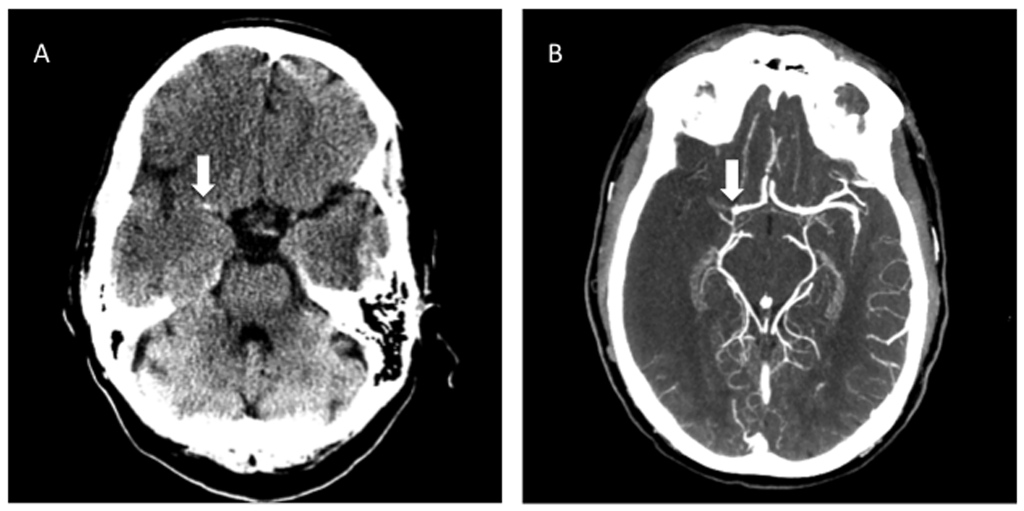
A) Non-contrast head CT showing a hyperdense proximal right M1 segment representing acute thrombus in a patient presenting with acute stroke. B) CTA demonstrates the corresponding cutoff of the right MCA.
The earliest indication of acute ischemia in the brain parenchyma is the loss of differentiation of the gray and white matter in the affected region, which is due to decreased blood flow to the relatively vascular and, thus, hyperdense cortex, rendering it indistinct from the adjacent, typically more hypodense white matter.16,17 This is termed the “insular ribbon sign” when it occurs in the insula, and the “disappearing basal ganglia” sign when it occurs in the basal ganglia. Classically peaking two to five days after the onset of a cerebral infarction, increasing hypoattenuation and mass effect from progressive vasogenic edema are present within the affected vascular territory.18 Beyond three to four weeks, in the chronic stage, the infarcted region evolves into a hypoattenuating region with gliosis and volume loss.16,19 (See Figure 4.)
Figure 4. Evolution of MCA Infarction
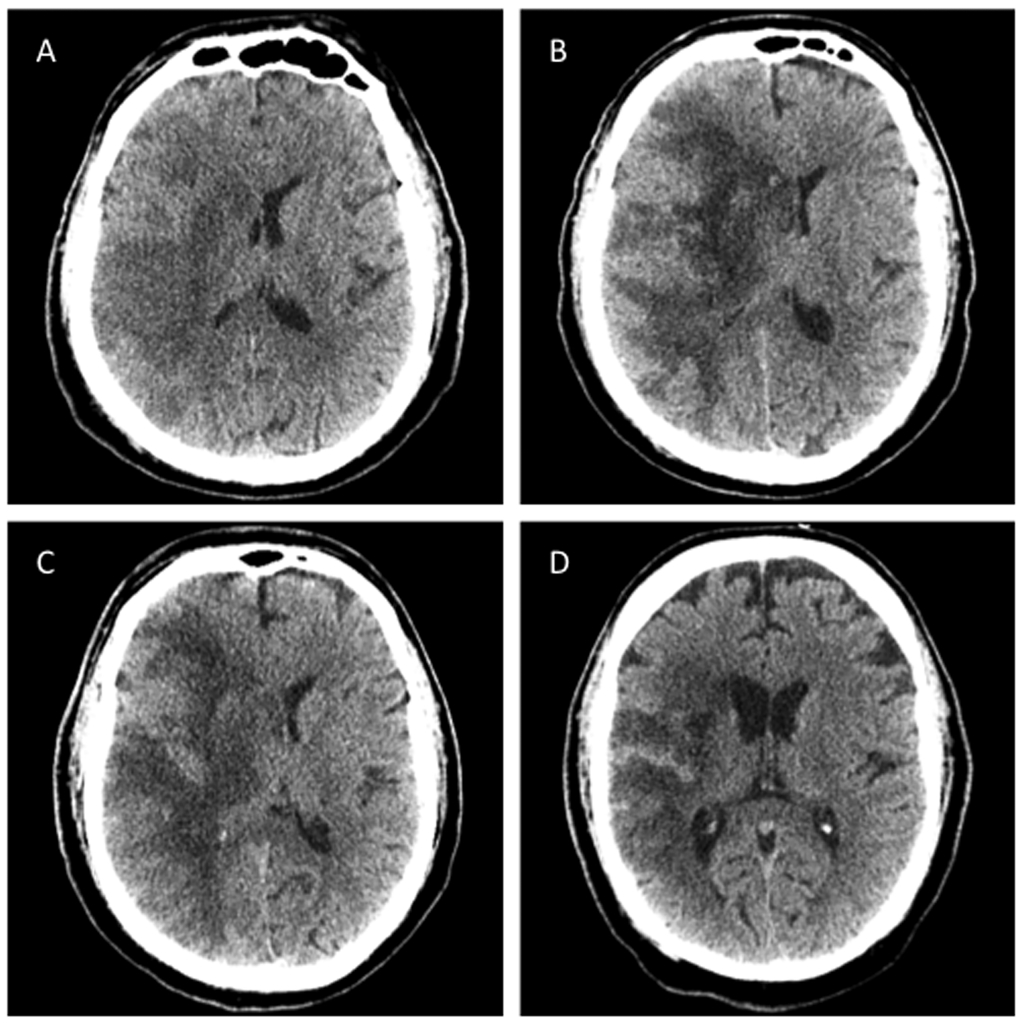
NCCT images from a patient who presented initially to an outside hospital showing temporal evolution of his large right MCA infarction. A) Significant mass effect already is appreciable by two days. B) Peak mass effect occurred at day 4. C) Significant mass effect persisted through day 10. D) Six weeks later, mass effect has resolved and volume loss is appreciable, as evidenced by mild ex-vacuo dilation of the right lateral ventricle.
Although primarily used to exclude intracranial hemorrhage, findings on NCCT also may help evaluate the severity of infarction and have some prognostic value. The Alberta Stroke Program Early CT Score (ASPECTS) is a scoring system devised to quantify the extent of MCA territory infarction to predict outcome after systemic thrombolytic therapy.20 It is relatively simple and reliable to use, requiring training but no additional resources beyond those required to perform an NCCT.21 The patient starts with 10 points, and one point is deducted for each preset area of interest in the MCA territory in which early ischemic change is identified. The areas of interest include the caudate, lentiform nucleus, internal capsule, insula, and six sections of MCA cortex divided between two levels, demarcated by the caudate head.20,21 (See Figure 5.) ASPECTS has been shown to correlate with prognosis, clinical stroke severity, the presence and location of an intra-arterial occlusion, and has good interobserver agreement.20-23 However, the ability of the ASPECTS to predict individual outcomes is modest, and, thus, care must be taken when interpreting results for prognostication on an individual basis.21
Currently there is no evidence to show that ASPECTS modifies the effect of intravenous tPA therapy within a six-hour time window, and, thus, eligible patients should not be excluded from receiving systemic thrombolytic treatment based on ASPECTS score alone.21,23,24 It has been suggested that ASPECTS may be useful in extending the treatment window for select patients, and that patients who have a clinically or functionally significant neurologic deficit but good ASPECTS score (greater than 7) may benefit from delayed thrombolytic therapy, although this has not been validated.21
Figure 5. Alberta Stroke Program Early CT Score (ASPECTS)
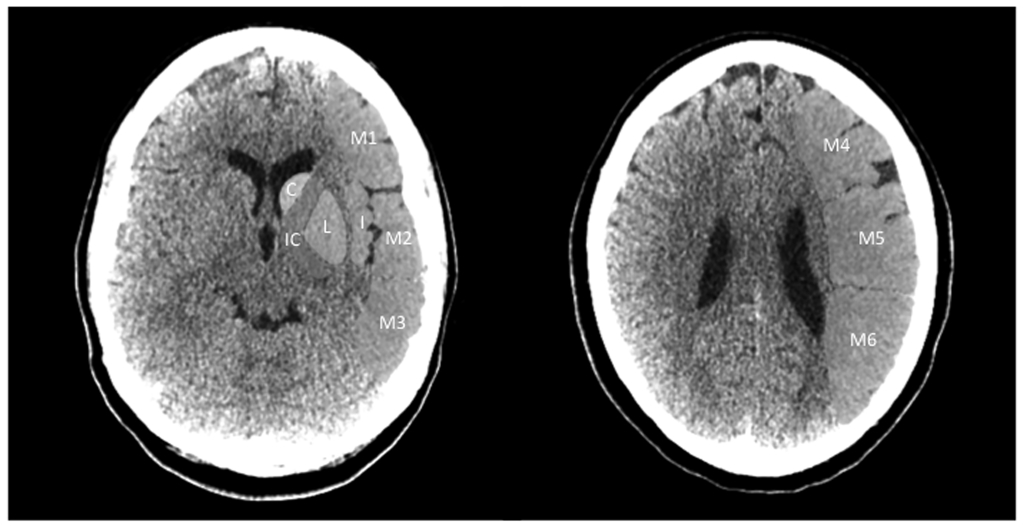
The Alberta Stroke Program Early CT Score (ASPECTS) scoring system. Starting with 10 points – one for each predefined region – a point is subtracted for each region that displays signs of early ischemia such as loss of gray-white matter differentiation, hypoattenuation, or mass effect.
Key: C = caudate; IC = internal capsule; I = Insula; M1 = MCA cortex anterior to insula; M2 = MCA cortex lateral to insula; M3 = MCA cortex posterior to insula; M4 = MCA cortex superior to M1; M5 = MCA cortex superior to M2; M6 = MCA cortex superior to M3.
Of the five major prospective randomized, controlled trials in favor of endovascular intervention (EXTEND-IA, MR CLEAN, SWIFT PRIME, ESCAPE, and REVASCAT), all excluded patients with a large infarction core, and some used ASPECTS to do so.8-12 It is interesting to note that on later central adjudication of imaging studies in the REVASCAT trial, 25% of study patients actually were categorized as having ASPECTS scores of 6 or less (below the cutoff for exclusion). This may explain in part why the overall treatment effect in REVASCAT was lower than in many of the other studies; however, this also suggests the possibility that discrimination based on ASPECTS would exclude some patients who otherwise would have benefited from treatment.10
Of course, ASPECTS is limited in that it was devised only to evaluate MCA territory infarctions. A similar scoring system for posterior circulation system infarction (pc-ASPECTS) has been devised, but it has not been studied as well to date for its ability to predict outcomes and help select patients for intervention.25
Magnetic Resonance Imaging
Structural magnetic resonance imaging (MRI) with diffusion-weighted imaging (DWI) is exquisitely sensitive in the detection of acute ischemic stroke.17,19,26,27 DWI is currently the gold standard for imaging of acute infarction. Regions of acute ischemia are represented as areas of bright signal on the final images, the so-called “lightbulb sign,” corresponding to restricted diffusion of water molecules due to the presence of cytotoxic edema. DWI is an early marker for ischemia,28 and has high reliability for the detection of acute infarction, with a sensitivity of 88-100% and specificity of 86-100%.17,19,26,27 (See Figures 6 and 7.) It also has high utility in situations such as assessing for the presence of acute ischemia in a patient with an unclear clinical presentation or detection of acute ischemia on a background of chronic white matter disease, which significantly reduces the sensitivity of CT.29
Figure 6. Acute Thalamic Infarction
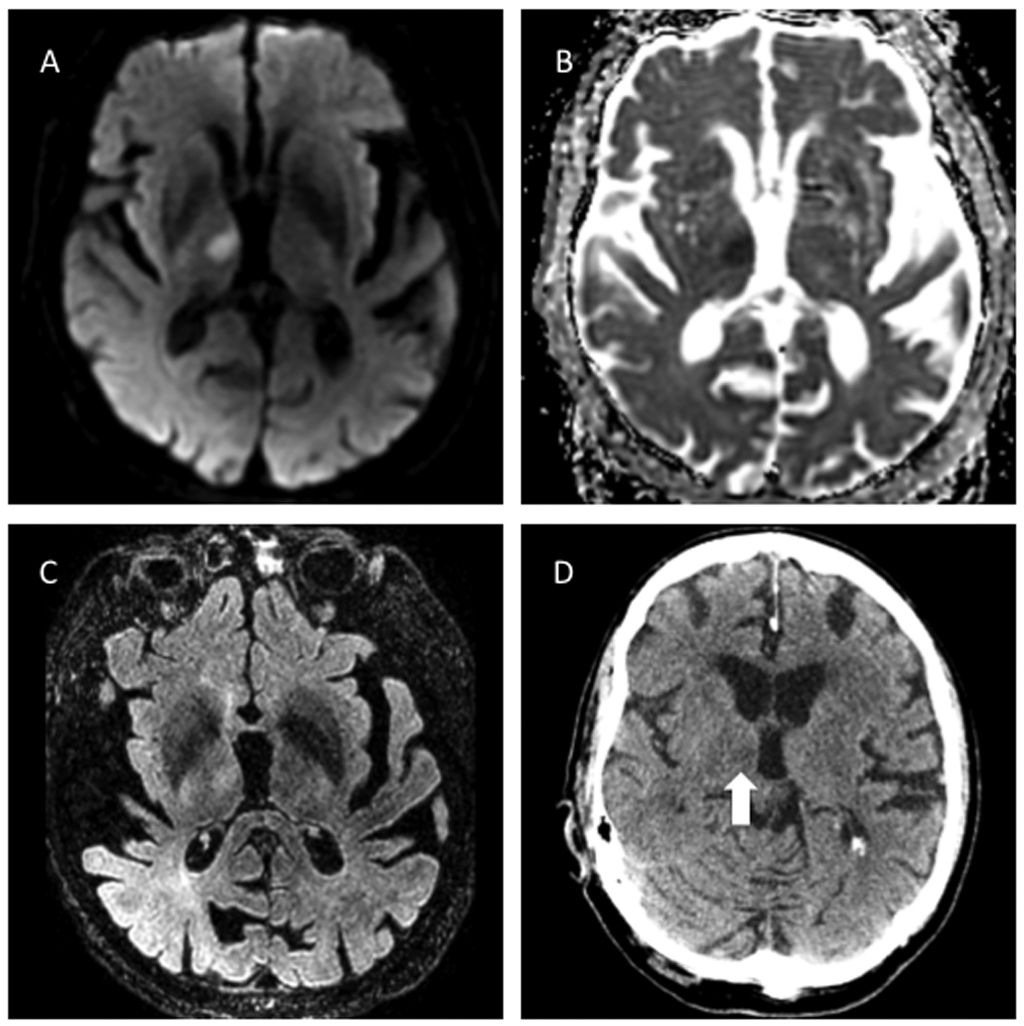
A, B) Acute right thalamic infarction seen axial MR DWI and ADC images. C) There is a small corresponding focus of T2 hyperintensity. D) In retrospect, the infarction is visible as a subtle, ill-defined hypodensity in the right thalamus on the patient’s initial NCCT.
Figure 7. Acute Posterior Cerebral Artery Infarctions
A) Axial image from a non-contrast head CT demonstrating a region of hypoattenuation and loss of gray-white matter differentiation in the right PCA territory, compatible with acute infarction. B) 3D maximum-intensity image from TOF MR angiography shows a cutoff of the right PCA. C, D) Axial MR DWI and ADC images not only confirm the diagnosis, but also show additional small infarctions in the contralateral left occipital lobe, which were not appreciated on the initial CT.
The use of filtered apparent diffusion coefficient (ADC) mapping in conjunction with DWI can aid in confirming true diffusion restriction. In fact, hypointensity on ADC maps may precede hyperintensity on DWI in hyperacute infarction.30 At one to two weeks, ADC hypointensity may “pseudonormalize” on its way to eventual hyperintensity in the chronic phase. DWI hyperintensity typically resolves in the early chronic phase, and will convert to low signal in the presence of cystic encephalomalacia.30 The characteristic appearance of a region of ischemia on different MR sequences also can be used to determine the age of an infarction.30 (See Table 1.)
Intravenous gadolinium-based contrast material is not required in the evaluation of an acute infarction, but may be useful in determining the age of an infarction. In early ischemia, even within two hours, an intrasulcal arterial pattern of enhancement may be seen, related to slow flow within arteries distal to the occluded artery. This gives way to parenchymal enhancement after approximately three days to a week, depending on the size and extent of infarction. Parenchymal enhancement rarely persists beyond four months.30,31
Table 1. Evolution of Imaging Findings of Ischemic Stroke on CT and MR16,19,30
Time From Symptom Onset |
CT Findings |
MR Findings |
|
Early hyperacute (0-6 hours) |
• Normal • Hyperdense artery • Subtle loss of gray-white differentiation |
• DWI hyperintensity (“lightbulb” sign) with corresponding ADC hypointensity |
|
Late hyperacute (6-24 hours) |
• Increasing hypoattenuation • Loss of gray-white differentiation • Developing mass effect and sulcal effacement |
• DWI hyperintensity with corresponding ADC hypointensity • T2 hyperintensity • T1 hypointensity (after 16 hours) • Developing mass effect and sulcal effacement |
|
Acute (24 hours - 1 week) |
• Pronounced hypoattenuation • Increasing mass effect; peak mass effect at 2-5 days • Acute hemorrhage may be present |
• DWI hyperintensity with corresponding ADC hypointensity • Increasing T2 hyperintensity • Increasing T1 hyperintensity • Increasing mass effect and sulcal effacement • Acute hemorrhage may be present |
|
Subacute (1-3 weeks) |
• Decreasing mass effect • Continued hypoattenuation |
• ADC pseudonormalization at 10-15 days, then ADC hyperintensity • DWI iso- or hypointensity at 10-14 days, or persistent hyperintensity due to T2 sine-through • T2 hyperintensity • Decreasing mass effect • Subacute hemorrhage may be present |
|
Chronic (> 3 weeks) |
• Volume loss • Hypodensity reflecting gliosis |
• T2 hyperintensity and volume loss reflecting gliosis • Possible DWI hyperintensity due to T2 shine-through without corresponding ADC hypointensity • Chronic hemorrhage may be present |
Susceptibility weighted imaging (SWI) is a high-resolution, two or three-dimensional gradient-recalled echo (GRE) sequence and is extremely sensitive to the presence of magnetic field inhomogeneities, including those caused by paramagentic agents, such as deoxyhemoglobin, and ferromagnetic agents, such as ferritin and hemosiderin.32,33 These areas of magnetic field inhomogeneity appear as regions of signal dropout on the final images. While NCCT long has been the imaging modality of choice for detecting acute hemorrhage, SWI has been shown to be as accurate as CT for detecting hyperacute hemorrhage, and superior to CT for chronic hemorrhage.13,36
However, the ability of SWI to detect deoxyhemoglobin has applications beyond the evaluation of hemorrhage. Deoxyhemoglobin in a clot may be evident as a focus of hypointensity on SWI that is greater in diameter than the contralateral side, due to what is termed “blooming” artifact, and may help to localize an occlusion.37,38 Similarly, SWI may aid in detection of acute venous thrombosis, an important mimic of acute ischemic stroke, which translates as pronounced asymmetric hypointensity on the affected vein.39,40 In addition, it has been suggested that because ischemic tissue has a higher oxygen extraction fraction (OEF), transmedullary veins in the area of ischemia may contain increased deoxyhemoglobin and appear more prominent on SWI. (See Figure 8.) This finding may demarcate the ischemic penumbra and correlate with risk of hemorrhagic transformation.32,33,41-43 It also has been suggested that SWI may allow for the visualization of leptomeningeal collaterals by a similar mechanism demonstrating decreased signal, indicating a greater proportion of deoxygenated hemoglobin, within small arterial collaterals,32,44 the significance of which is discussed in more detail below under vessel imaging. It is clear that SWI may provide useful corollary information in conjunction with other MR sequences that have the potential to affect treatment decision-making in acute ischemic stroke.33,45
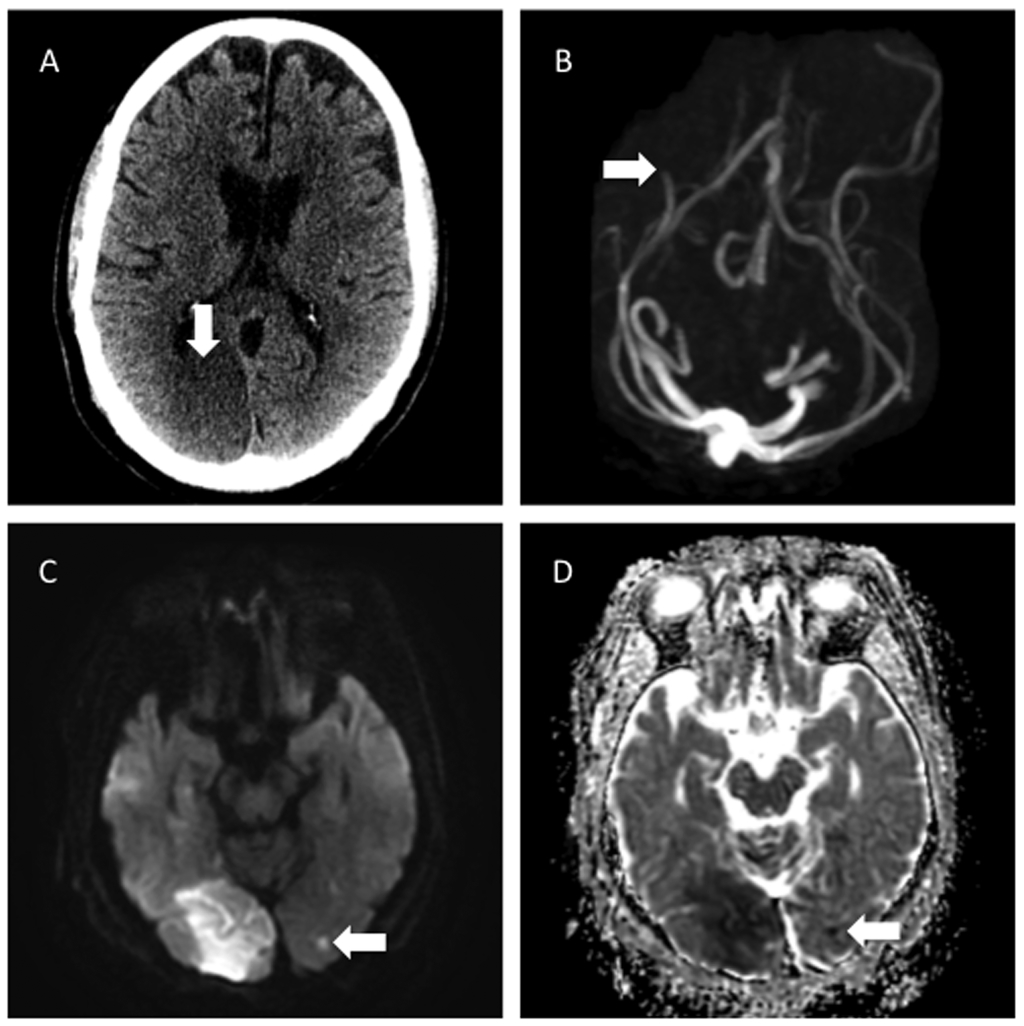
Figure 8. Middle Cerebral Artery Occlusion With Dilated Transmedullary Veins on SWI
A) 3D maximum-intensity reconstruction from an MR angiogram in a patient presenting with acute stroke showing a cutoff of the right MCA. B) Axial MR SWI in the same patient showing dilated right transmedullary veins in the region of infarction, which indicates increased deoxyhemoglobin due to increased oxygen extraction by the ischemic tissue.
Vessel Imaging
Given that acute ischemic stroke is a disease of vascular etiology, it is not surprising that vessel lumen imaging of the head and neck can be of value in its evaluation. Imaging the vessel lumen can detect the presence of an acute occlusion, assess for additional lesions such as stenoses or dissection, assess the quality of collateral flow, and aid in planning for possible interventions.
In the 2015 American Heart Association/American Stroke Association Focused Update of the 2013 Guidelines for the Early Management of Patients With Acute Ischemic Stroke Regarding Endovascular Treatment, there is a strong recommendation for noninvasive intracranial vascular study during the initial imaging evaluation of an acute stroke patient. But such noninvasive vascular imaging should not delay intravenous tPA if indicated.
There are several different modalities available for noninvasive vascular imaging of the head and neck, each with corresponding advantages and drawbacks.
Conventional Angiography
Digital subtraction angiography (DSA), or conventional catheter angiography, is the gold standard against which other vessel lumen imaging modalities are compared when it comes to assessment of the neurovasculature. However, this technique is invasive, is resource-intensive (including high use of skilled labor), and is associated with significant radiation exposure. As a purely diagnostic modality, it has little role in the modern clinical setting in the initial evaluation of acute ischemic stroke.
CT Angiography
CT angiography (CTA) is an imaging technique that requires the administration of intravenous iodine-based CT contrast. Image acquisition then is timed for maximal contrast opacification of the arteries for a single-phase CTA. This technique has the advantages of being widely available (most centers that can perform NCCT also can perform CTA), cost effective compared to MR as well as conventional angiography, and fast to perform. In fact, when combined with NCCT, a CTA adds only two to five minutes to time on the CT table.46 It can provide high-resolution anatomic information that can be extremely useful in planning interventional procedures.47-52 CTA has been demonstrated to have higher sensitivity, positive predictive value, and inter-rater reliability compared to time-of-flight (TOF) MR angiography for identifying arterial occlusion, and has even been shown to be superior to DSA in the evaluation of posterior circulation steno-occlusive disease in the presence of slow flow.53 (See Figure 9.)
When an arterial occlusion is present, CTA can help define clot characteristics that have predictive value regarding the likelihood of reperfusion after the administration of systemic tPA. Clot location and length, as well as the presence of residual blood flow around or through the occlusion, can be determined easily.54 (See Figure 10.) Not surprisingly, patients with shorter clots (less than 15 mm) and residual blood flow are more likely to have early reperfusion after systemic tPA therapy.54 It has been shown that proximal large vessel occlusions are relatively resistant to systemic thrombolysis; patients with proximal M1 segment clots without residual flow will only reperfuse 8% of the time, and patients with carotid terminus “T” or “L” type occlusions only reperfuse 1.7% of the time.54-58 In patients with so-called “tandem” lesions, where there is both a proximal internal carotid lesion as well as an MCA occlusion, patients whose M1 segment clot was less than 10 mm from the M1 origin reperfused only 8% of the time, as compared to 25% who had early reperfusion with an M1 clot more than 10 mm from the M1 origin.54
Of note, each of the five recent major randomized clinical trials that showed significant benefit for endovascular intervention required definitive identification of a proximal large artery occlusion for patients to be included in the trial. This was done largely via CTA.8-12
Figure 9. Severe Internal Carotid Artery Stenosis With String Sign
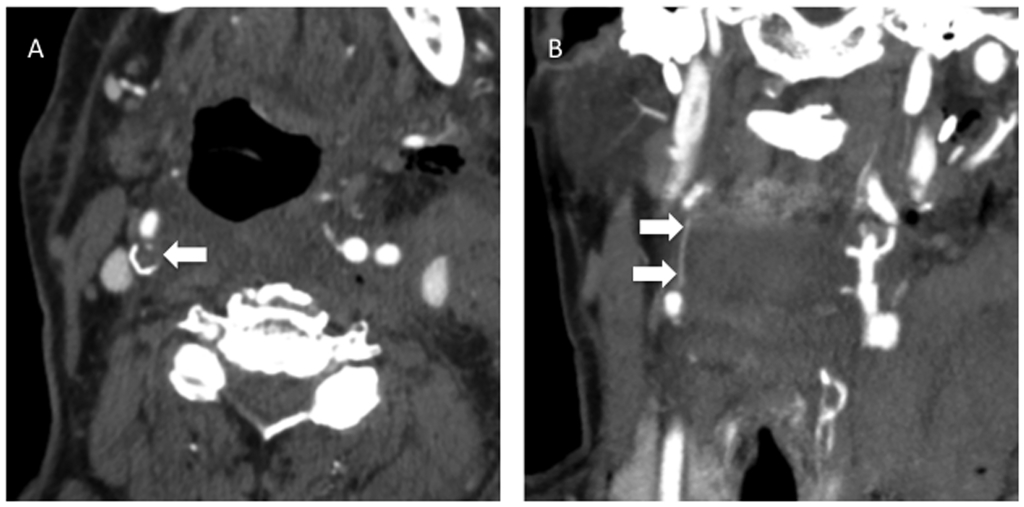
A) Axial CTA image showing severe stenosis of the right ICA by mixed fibrofatty and calcified plaque. B) Coronal reformation from the same study demonstrates the “string sign” of attenuated flow beyond a point of luminal stenosis.
Figure 10. Hemorrhagic Transformation of Anterior MCA Territory Infarction Following IV tPA
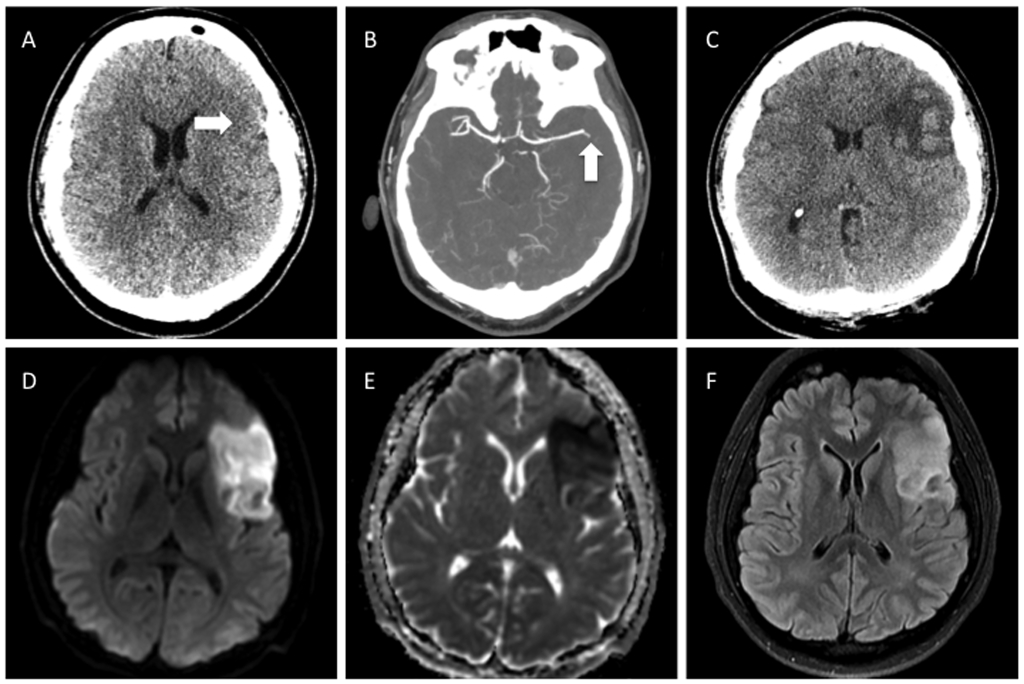
A) Axial image from an initial NCCT in a patient presenting with acute stroke, showing subtle loss of gray-white matter differentiation in the left frontal operculum. B) CT angiography demonstrated a vessel cutoff sign in a left M2 segment. C) Following IV tPA therapy, the NCCT a few days later showed hemorrhagic transformation. D, E, F) MR DWI, ADC, and T2 FLAIR images demonstrated a region of restricted diffusion and T2 hyperintensity corresponding to the original finding on CT.
Robust collateral filling vial leptomeningeal (pial) collaterals as assessed by CTA has been shown to be associated with reduced infarction growth, smaller final infarction volume, and improved patient outcomes.59-64 Additionally, angiographic grade of collateral flow has been linked to a likelihood of hemorrhagic transformation of acute ischemic stroke, with more significant risk shown in patients with poor leptomeningeal collaterals, particularly if the patients achieve recanalization.65,66 In fact, it is worth noting that the ESCAPE trial excluded patients with absent or poor pial collaterals on CTA.12 The IMS-III trial, an earlier trial that failed to show an overall benefit for endovascular treatment, demonstrated that the quality of collateral flow on baseline CTA was a good predictor of final clinical outcome, and that the most benefit of endovascular treatment was seen in patient with intermediate collaterals.67
MR Angiography
There are a number of techniques by which MR angiography (MRA) may be performed, which may be flow-dependent or rely on the administration of an intravenous contrast agent. Three-dimensional TOF GRE imaging relies on the contrast between the non-saturated proton spins in blood entering the imaging plane and the stationary saturated protons in the adjacent tissue, which results in the only signal on the image derived from flow within the vessels.68,69 It is currently the preferred flow-dependent MRA sequence for imaging of intracranial arteries, given its relatively rapid acquisition time and high spatial resolution.68 Phase-contrast MRA, another flow-dependent technique, takes blood flow measurements through velocity encoding, but has poorer spatial resolution than TOF angiography.68
While contrast-enhanced MRA may provide better anatomic detail than TOF angiography, its sensitivity for detecting intracranial lesions is lower than that for extracranial steno-occlusive disease.70 Contrast-enhanced MRA can be used to detect and characterize luminal stenoses and occlusions of both the intracranial and extracranial vessels (see Figures 11 and 12), but is less accurate in more distal arterial branches as compared to CTA and is more susceptible to imaging artifacts.68 In addition, literature regarding the utility of contrast-enhanced MRA to assess collateral flow to at-risk tissue is currently lacking.68
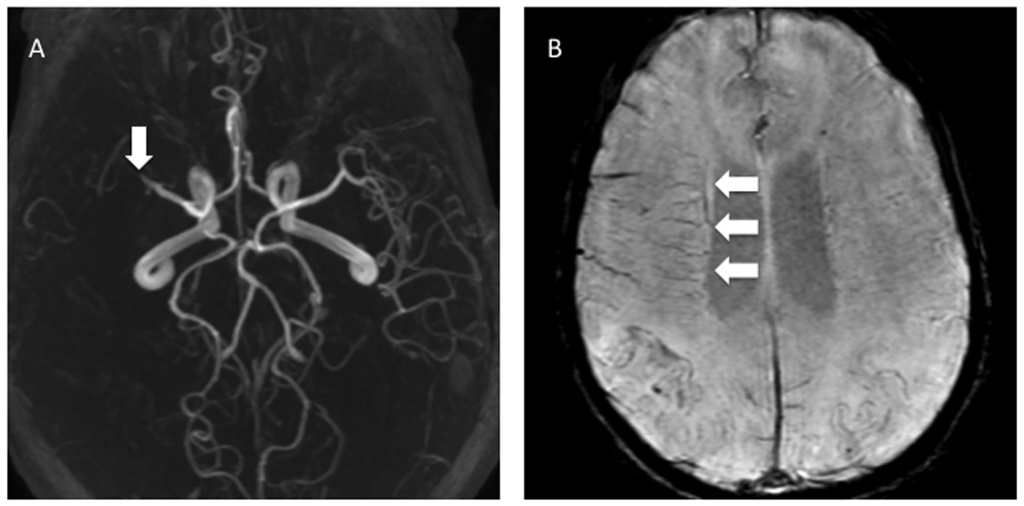
Figure 11. Acute Infarction With Proximal Internal Artery Stenosis
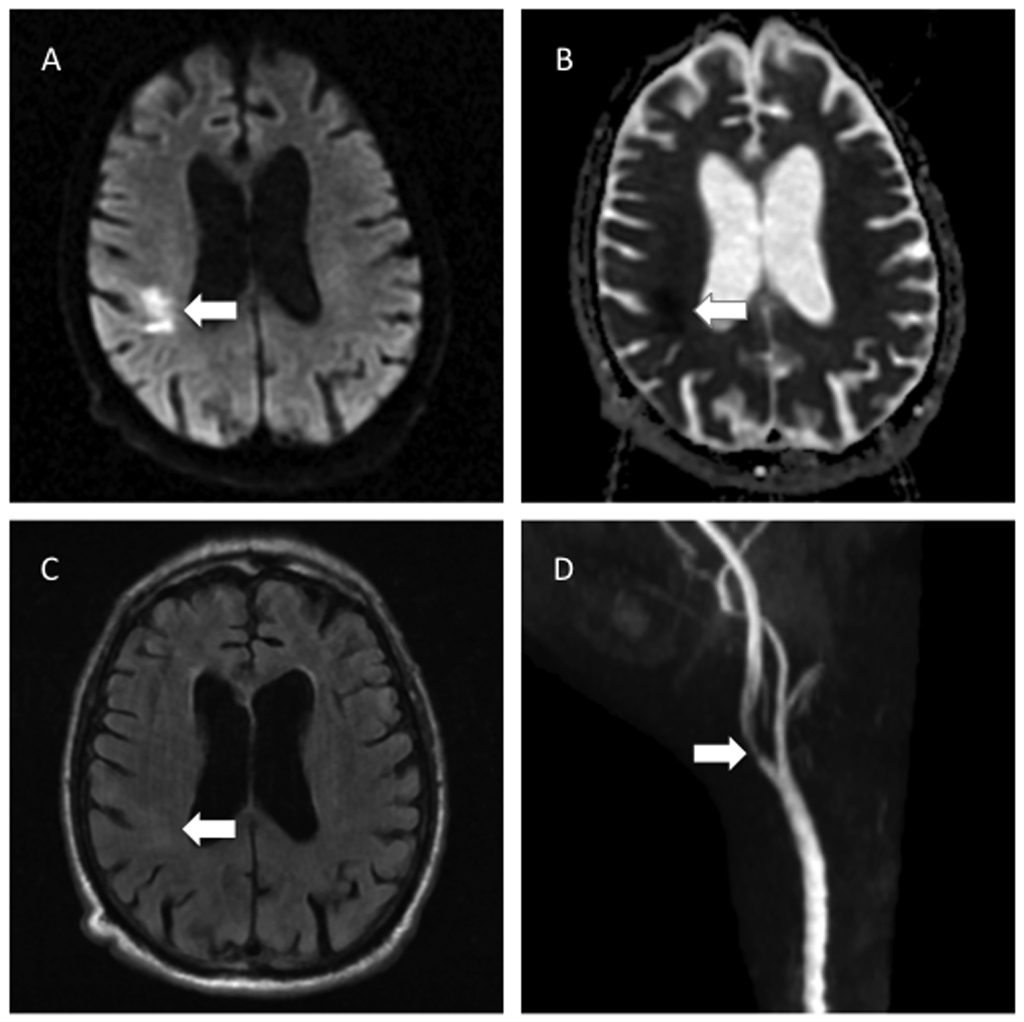
A) Hyperintensity on DWI (the “lightbulb” sign).
B) Corresponding hypointensity on ADC confirms true diffusion restriction, consistent with acute infarction. C) T2-weighted FLAIR imaging shows only minimal corresponding hyperintensity, indicating the age of the stroke is less than 6 hours old. D) Contrast-enhanced MR angiography demonstrates severe stenosis of the proximal right internal carotid artery, likely the source of an artery-to-artery embolus.
Figure 12. Cervical ICA Dissection Resulting in Infarction
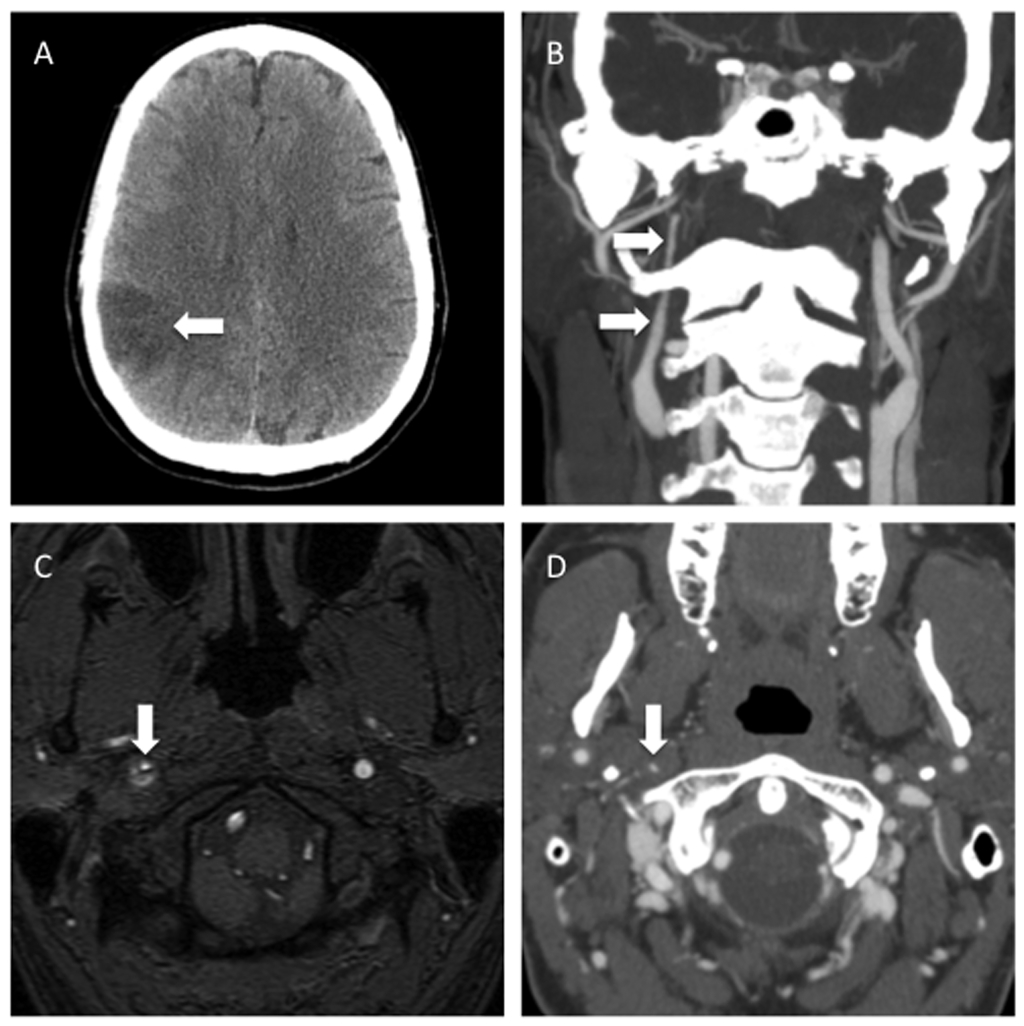
A) NCCT showing a focal region of hypoattenuation in the right parietal lobe, consistent with an MCA territory infarction. B, D) CTA in the same patient showed long segmental narrowing of the right internal carotid artery seen on the coronal maximum-intensity reformation and axial plane, consistent with dissection. C) MR angiography confirmed the diagnosis, demonstrating crescentic hyperintensity within the false lumen of the dissection compatible with subintimal thrombus around the narrowed true vessel lumen. Normal flow can be seen in the contralateral left internal carotid artery on both CT and MR angiography images.
MR Vessel Wall Imaging
Where MR does hold many advantages is in vessel wall imaging (VWI) and the assessment of intra-arterial plaque composition. With advances in MR, evaluation for vulnerable carotid and intracranial plaques is possible.71,72,73 While cervical carotid steno-occlusive disease has been studied extensively, much of the research has focused on the degree of luminal stenosis as a marker for ischemic stroke risk, and, thus, its indications for intervention.74,75 In recent years, there has been a surge in interest in the characterization of plaque composition, and whether such characterization can help risk-stratify patients.
A 2017 meta-analysis of studies looking at carotid plaque composition demonstrated that the presence of intraplaque hemorrhage, a lipid-rich necrotic core, and thinning or rupture of the fibrous cap on MR carotid plaque imaging significantly correlates with an increased risk for future cerebrovascular events.76 (See Figures 13 and 14.) In addition, intracranial atherosclerotic disease is a well-known cause of stroke,77-81 and high-resolution intracranial VWI may allow for the assessment of intracranial atherosclerotic plaques for the presence of similar high-risk characteristics.69,82,83
Figure 13. Carotid Bulb Stenosis With Intraplaque Hemorrhage
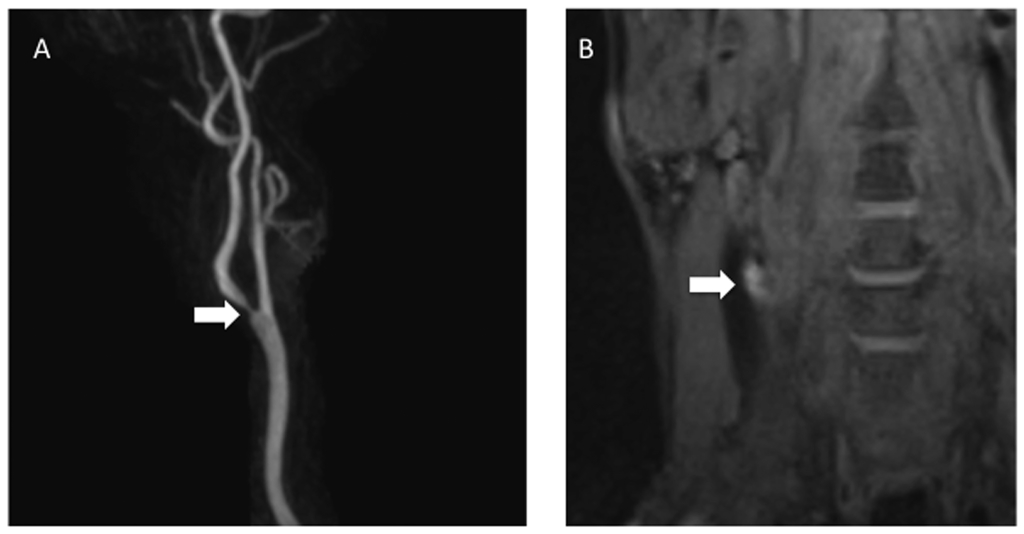
A) MR angiography in a patient showing stenosis of the right internal carotid artery at its origin due to atherosclerotic plaque. B) Black-blood imaging shows high signal within the plaque, suspicious for intra-plaque hemorrhage
Figure 14. Artery Schematics
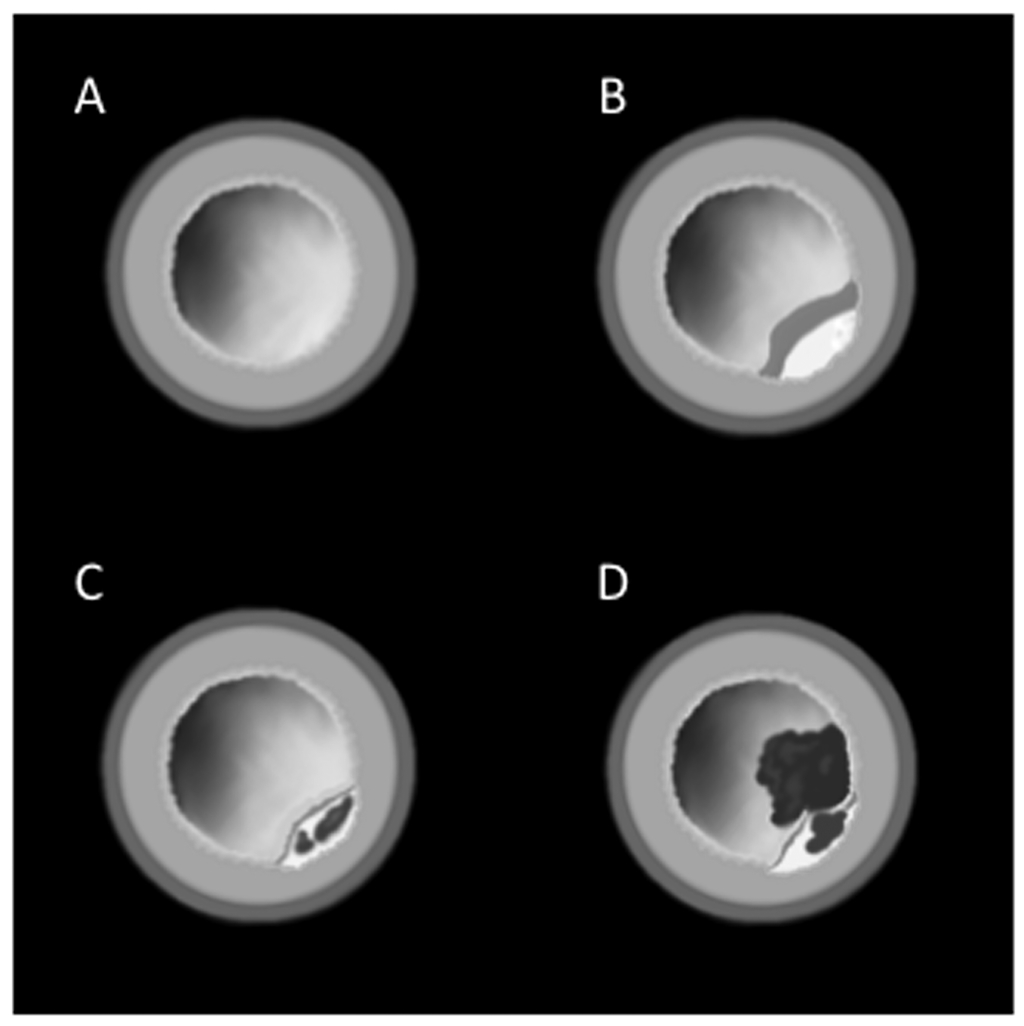
Schematics showing A) normal artery, B) stable plaque with thick fibrous cap and calcification, C) high-risk plaque with thinned fibrous plaque and intra-plaque hemorrhage, and D) ruptured plaque with non-occlusive thrombus.
Transcranial Sonography, Carotid Sonography
Sonographic imaging of intracranial and extracranial vessels has many potential advantages: There is no exposure to ionizing radiation with almost no contraindication for use, and theoretically, the studies can be performed rapidly, potentially at the bedside. B-mode (or 2D) sonography uses information from reflected ultrasound waves to construct a grayscale image, and Doppler sonography takes advantage of the Doppler effect and uses the phase shift in reflected ultrasound waves to determine the velocity and direction of blood flow within a vessel.83
Transcranial Doppler (TCD) is an ultrasound technique using four different acoustic windows in the skull to transmit ultrasound waves to acquire arterial waveform Dopplers.68 Transcranial color-coded sonography (TCCS) uses a similar technique to visualize the proximal intracranial arteries by color-coding blood velocity.68 Transcranial sonography is complemented by information from the more ubiquitous carotid Doppler sonography, which can provide anatomic and structural (B-mode) as well as hemodynamic (Doppler) information about the extracranial vessels.68
The major advantage of sonographic evaluation is the capability of real-time characterization of flow information, such as direction of flow, collateralization, microembolism, and steal phenomena.84 Unfortunately, sonographic vessel imaging has many limitations as well. Sonographic evaluation of extracranial vessels may be suboptimal or difficult to interpret in the presence of patient factors such as tortuous vessels, cardiac arrhythmias, or ventricular assist devices.85 For intracranial vessel imaging, the available acoustic windows through the cranium limit visualization to the proximal vessels — the distal internal carotid (except the carotid terminus) and much of the vertebral artery course cannot be evaluated,68 and transcranial Doppler has limited accuracy compared to CTA.86,87 In addition, sonographic studies are extremely operator-dependent and require a skilled technologist to perform; therefore, they may not be a realistic option in many settings.68,87
Perfusion Weighted Imaging
The routine use of perfusion imaging in the evaluation of acute ischemic stroke is not currently ubiquitous. Perfusion imaging is time-resolved imaging that provides information about the quantity and quality of cerebral perfusion in an area of interest and can help determine whether tissue has been infarcted irreversibly or is potentially salvageable with reperfusion. In the setting of acute ischemic stroke, this may identify patients who most likely will benefit from treatment or intervention.
Several studies have demonstrated the value or potential value of CT and MR perfusion imaging in selecting patients for both treatment with systemic thrombolytic therapy in a prolonged treatment window, and in selecting patients for endovascular intervention.8-12,88-90 A recently terminated large, randomized clinical trial (DEFUSE3) used perfusion mismatch on either CT or MR perfusion to select patients for delayed thrombectomy between 6-16 hours from symptom onset. The final results of the study have not been published, but interim analysis reportedly revealed a high likelihood of benefit in the intervention group (NCT02586415).
CT Perfusion
Like CTA, CT perfusion (CTP) requires the administration of intravenous CT contrast. Also similar to CTA, the resources required to perform CTP imaging are widely available, it can be performed rapidly, and it is low cost when compared to MR.91 After receiving a bolus of intravenous contrast, a portion of the patient’s brain is imaged repeatedly over a short period of time, and perfusion maps are generated by post-processing software. Multiple parameters are assessed, with an individual map generated for each: cerebral blood volume (CBV), which is a measure of the total blood volume per unit of brain tissue; cerebral blood flow (CBF), which is a measure of blood volume per unit of brain tissue per unit of time; mean transit time (MTT), which is a measure of the average amount of time it takes blood to travel through a given unit of brain; and time to peak (TTP), a measure of the time it takes for maximum contrast opacification to occur for a given unit of brain.
The amount of at-risk or salvageable tissue (the ischemic penumbra) identified on CTP that becomes revascularized after treatment has been shown to correlate positively with clinical outcome. It also has been shown that regions identified as the infarction core on CTP correlate closely to end-infarcted regions in patients who do get revascularized.92-94
Acutely infarcted tissue (i.e., that which is damaged irreversibly) is represented by regions with diminished CBF and CBV, and increased MTT and TTP.91,92,95,96 (See Figures 15 and 16.) In contrast, the ischemic penumbra may be represented as “mismatched” regions with decreased CBF, prolonged MTT, and relatively normal to elevated CBV.91 (See Figure 17.)
Figure 15. Large Matched MCA Core Infarction on CT Perfusion
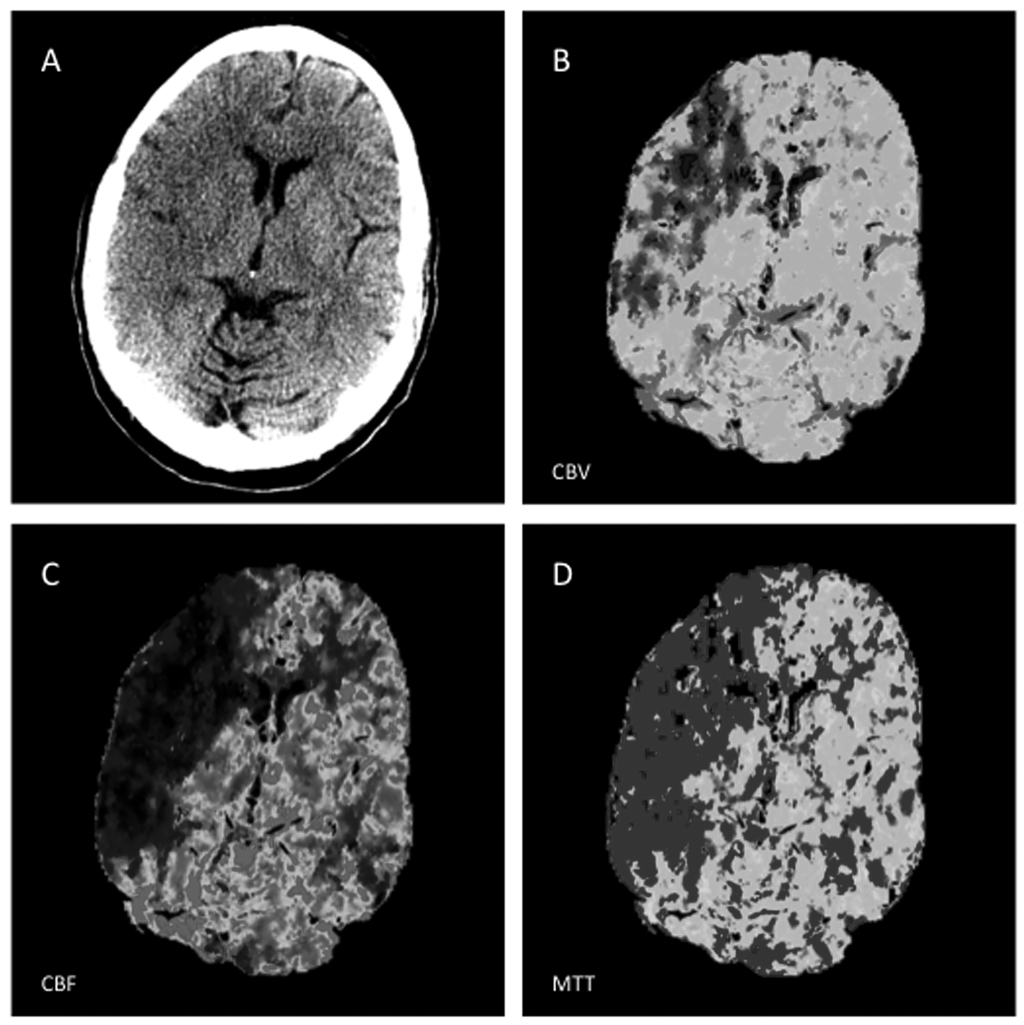
A) NCCT showed a large region of hypoattenuation with loss of gray-white matter differentiation and early mass effect evidenced by sulcal effacement. CT perfusion showed a matched deficit in cerebral blood volume (B), cerebral blood flow (C), and mean transit time functional maps (D), indicating a large ischemic core of irreversibly damaged tissue.
Figure 16. Large Core Infarction on CT Perfusion
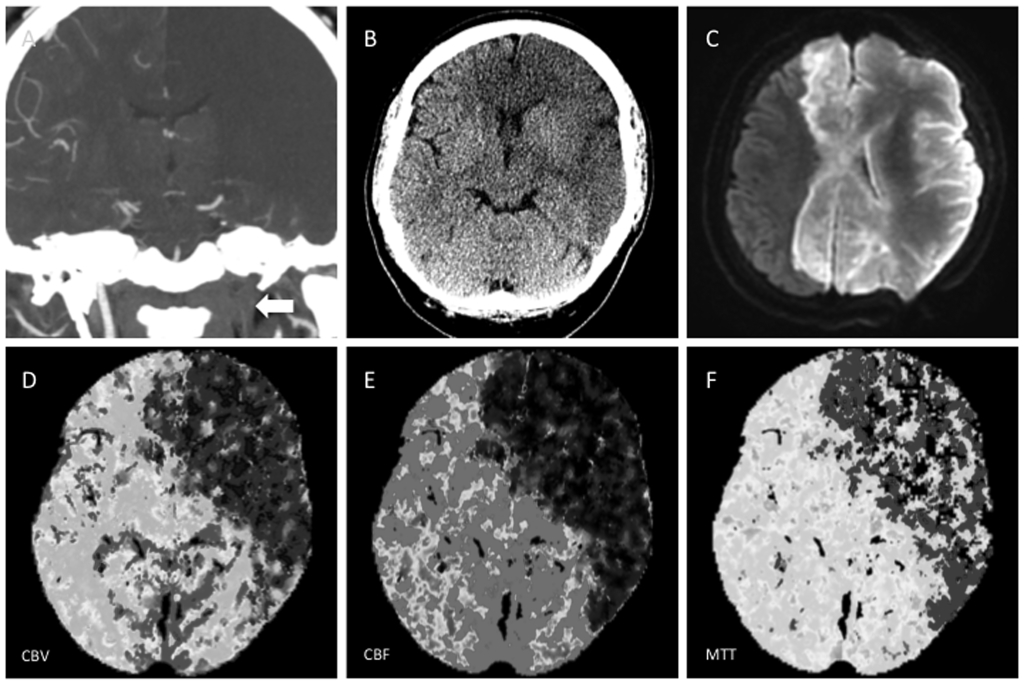
A) Coronal reformation from a CT angiogram showing occlusion of the left ICA; note the contrast opacification of the normal contralateral right ICA. B) A concurrent CT head demonstrated striking loss of gray-white matter differentiation. C) A diffusion-weighted image from a subsequent MR demonstrates striking diffusion restriction involving the left MCA as well as the bilateral ACA and PCA territories. D, E, F) CT perfusion images show a corresponding large matched deficit in cerebral blood volume, cerebral blood flow, and prolonged mean transit time indicating irreversibly damaged tissue without a significant penumbra of salvageable tissue.
Figure 17. Large Penumbra on CT Perfusion
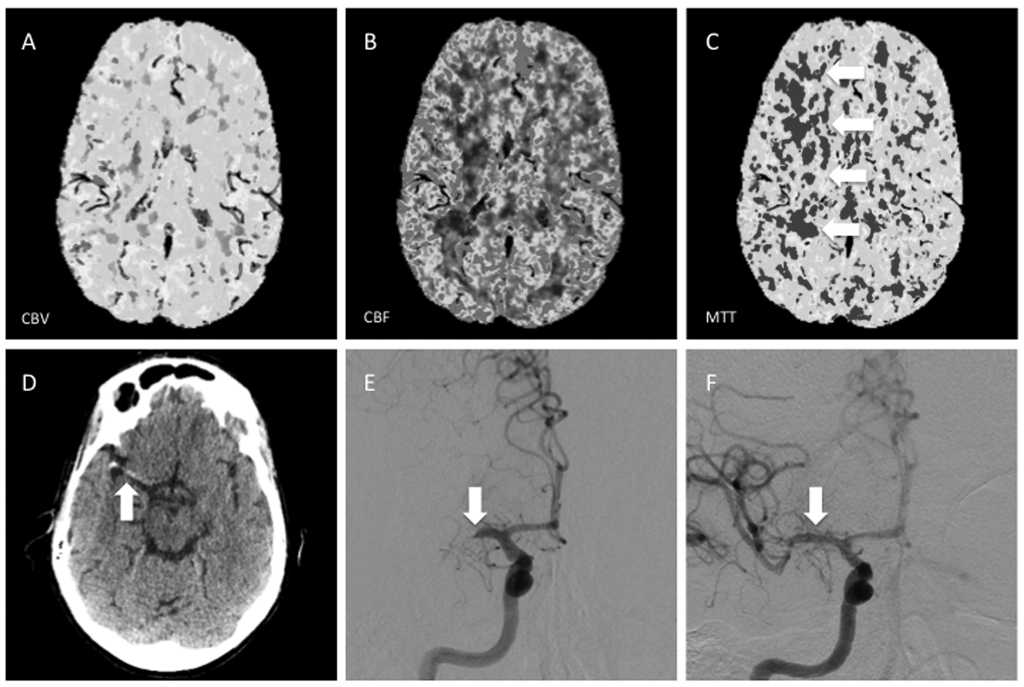
A, B, C) CT perfusion images showing preserved cerebral blood volume and cerebral blood flow, with slightly elevated mean transit time in the right MCA distribution, indicating a large ischemic penumbra without a significant core of infarcted tissue. D) A NCCT head in the same patient shows a hyperdense right M1 segment, corresponding to acute thrombus. E) The patient was taken for endovascular intervention, where a proximal M1 occlusion was confirmed. F) The patient underwent successful thrombectomy with excellent restoration of flow within the right MCA.
A major challenge with CTP is the lack of standardization in post-processing data and the lack of a clear definition of threshold values at which infarction core and ischemic penumbra are differentiated.94 However, both the EXTEND-IA and SWIFT PRIME trials used CTP in their selection of patients for the study. Both studies also used operator-independent post-processing software to standardize interpretation of CTP,8,11 which has been shown to increase reproducibility and may facilitate the implementation of CTP in healthcare settings that are not highly specialized academic centers.95,98 Of note, the recently terminated DEFUSE 3 trial also required the use of special automated perfusion software to calculate mismatch volume.
MR Perfusion-weighted Imaging
Perfusion-weighted imaging (PWI) can be performed with the use of intravenous contrast, a technique called dynamic susceptibility contrast imaging, or by a newer technique called arterial spin labeling, which does not require the use of contrast. Perfusion maps are generated demonstrating similar parameters as those used for CTP. Thus, a thorough MR-based stroke evaluation can be performed without the use of contrast at all.
The infarction core may be identified using similar parameters as those used in CTP, and should correspond to the region of DWI abnormality.98-100 Thus, the identification of at-risk tissue may be represented as a mismatch between perfusion abnormalities and diffusion abnormalities, with penumbral tissue identified by a TTP Tmax of 4-6 seconds.95,101,102
Many of the earlier studies investigating the expansion of the treatment window in stroke used MR perfusion-diffusion mismatch to select patients for treatment. For example, the EPITHET and DEFUSE trials used mismatch to select patients for systemic thrombolytic therapy outside of the three-hour treatment window, both of which showed promising but not definitive results.88,90 The DEFUSE2 trial was a prospective cohort study that followed patients undergoing endovascular intervention, and suggested that there was a strong correlation between perfusion mismatch and favorable outcomes.90
Current available evidence suggests that MR diffusion-perfusion mismatch may remain slightly superior to CTP in identifying the infarction core and delineating tissue at risk, although CTP can provide similar information and is much more readily and usually rapidly available.95 However, if evaluation is less urgent, or if CT imaging is not feasible, then MR PWI may be the modality of choice for characterizing stroke.
Considerations: CT vs. MR Imaging
CT and MR techniques are capable of providing similar information for structural, vessel, and perfusion imaging, although with distinct strengths and weaknesses for each type of assessment. However, there are intrinsic advantages and disadvantages to each modality. (See Table 2.)
Table 2. Comparison of CT and MR15,17,19,26,27,95
Computed Tomography |
Magnetic Resonance |
|
|
Cost |
Relatively inexpensive |
Relatively costly |
|
Ionizing radiation exposure |
Yes, although modern scanners capable of acquiring studies with minimal radiation |
No |
|
Time to perform |
Rapidly acquired images requiring only minutes on the table, even if contrast is used |
Requires longer time investment, 20 to 40 minutes |
|
Evaluation for hemorrhage |
Acute hemorrhage readily identified |
Extremely sensitive with SWI |
|
Stroke detection |
Less sensitive for hyperacute/acute stroke |
Extremely sensitive for hyperacute/acute stroke |
|
Contraindications |
Few contraindications |
Contraindications for many patients with implanted devices, such as pacemakers |
|
Availability |
Widely and rapidly available |
Less widely available |
The clear advantages of CT imaging are its widespread availability, rapidity of image acquisition, and relative low cost compared to MR imaging. Although MRI may be more sensitive for the detection of acute ischemic stroke, a non-contrast head CT can be performed expeditiously to exclude hemorrhage or other potential etiologies for a stroke-like syndrome, without the burden of delays intrinsic to MR, such as metal screening, metal removal, and individual sequence acquisition, which can delay treatment by as much as 20 to 50 minutes.103,104 Similarly, in patients who may be candidates for stent-retriever thrombectomy, CTA is much more accessible and rapidly acquired than MRA for the detection of arterial occlusions and interventional planning.
However, a potential pitfall of single-phase CTA is that with maximization of contrast opacification of the proximal large arteries, smaller distal collateral vessels may be opacified suboptimally and, therefore, may not be apparent.68 In addition, slow flow within the proximal large arteries, for example due to a high-grade proximal stenosis, may falsely give the impression of an occlusion. Multiphase time-resolved CTA is an alternative technique that may overcome some of these pitfalls, typically adding equilibrium and late venous phases to the arterial phase, thus allowing time for contrast filling of collateral vessels and of large vessels with slow flow due to proximal stenosis.68 However, the added radiation exposure from multiple phases is a significant clinical concern. In fact, one of the greatest downsides of CT imaging in general is ionizing radiation exposure, although newer scanners can acquire diagnostic quality images with minimal overall radiation, even with added CTA/CTP.68,91 (See Table 3.) Additionally, the projected lifetime risk of death from cancer attributable to a CT angiography study is estimated at a mere 0.02-0.04%,106 which is minute compared to the much more tangible risk of death or disability in a patient with major stroke who may have benefited from treatment facilitated by CT imaging.
Table 3. Radiation Dose Associated With CT Neuroimaging for Acute Stroke105
Average Radiation Dose |
Equivalent Amount of Background Radiation at Sea Level |
|
|
CT head |
2.0 mSV |
8 months |
|
CT angiography head/neck with perfusion |
16.4 mSV |
5 years |
Although MRA and PWI may be performed without the use of intravenous contrast, both CTA and CTP require intravenous iodinated contrast injection. Administering intravenous contrast always carries a risk of contrast reaction, although serious reactions are rare — as low as one to two per 10,000 examinations with low-osmolarity contrast agents107,108 — and the majority happen immediately or soon after injection, and are readily identified and treated.68 Clinicians and patients alike also may be concerned about the risk of contrast-induced nephropathy from iodinated contrast injection, particularly in patients who already have chronic renal disease. Although caution still is suggested, particularly for patients with known severe renal insufficiency who are not yet dialysis-dependent, recent studies suggest that the clinical significance of renal dysfunction attributable to intravenous iodinated contrast is at best minimal, if at all present, even for patients undergoing both contrast-enhanced CT imaging and endovascular intervention, where there is an additional dye load to consider.109-112
Endovascular Intervention
Earlier studies on endovascular intervention vs. standard of care medical therapy failed to show significant benefit, and one, the IMS-III trial, was stopped early for futility.67,92,113 These studies primarily used early-generation thrombectomy devices, and most did not have stringent imaging criteria for patient selection.
In 2015, five randomized clinical trials comparing endovascular intervention to medical management alone were published, each of which showed significant benefit of treatment for patients randomized to intervention — EXTEND-IA, MR CLEAN, REVASCAT, SWIFT PRIME, ESCAPE.8-12 (See Table 4 in online supplement.) The studies shared many similarities in that they used new-generation, more effective thrombectomy devices — predominantly the Solitaire stent retriever (Medtronic, previously Covidien) — and had strict imaging criteria for patient enrollment, mostly using advanced imaging techniques, including confirmed proximal occlusion on vessel imaging and a small infarction core. Several of these studies were stopped early for efficacy, and, therefore, were insufficiently powered for meaningful subgroup analyses.
Investigators from the trials pooled individual patient data for analysis in the HERMES meta-analysis, and were able to assess for benefit of endovascular therapy for particular subgroups of interest.114 The results demonstrated that across all studies and patients, endovascular therapy was associated with significantly reduced disability at 90 days compared to control. They found the number needed to treat to reduce disability by at least one point on the modified Rankin scale for one patient was only 2.6, and that for every 100 patients treated, 38 would have a less disabled outcome compared to if they had received best medical therapy alone, and that 20 more would achieve functional independence.114 Subgroup analysis of pooled patient data demonstrated no heterogeneity of treatment effect (reduced disability by modified Rankin score), including in patients 80 years of age or older, those who were randomized more than 300 minutes after symptom onset (but primarily before 420 minutes), and those who did or could not receive systemic thrombolytic therapy. The individual studies were insufficiently powered for assessment of the efficacy of endovascular intervention in patients ineligible for systemic tPA; pooled data from 188 intravenous tPA-ineligible patients showed substantial benefit with endovascular therapy, suggesting that endovascular perfusion should be considered for large anterior circulation vessel occlusions regardless of eligibility for systemic thrombolytic therapy.114 (See Figure 18.)
Figure 18. MCA Recanalization With Stent Retriever
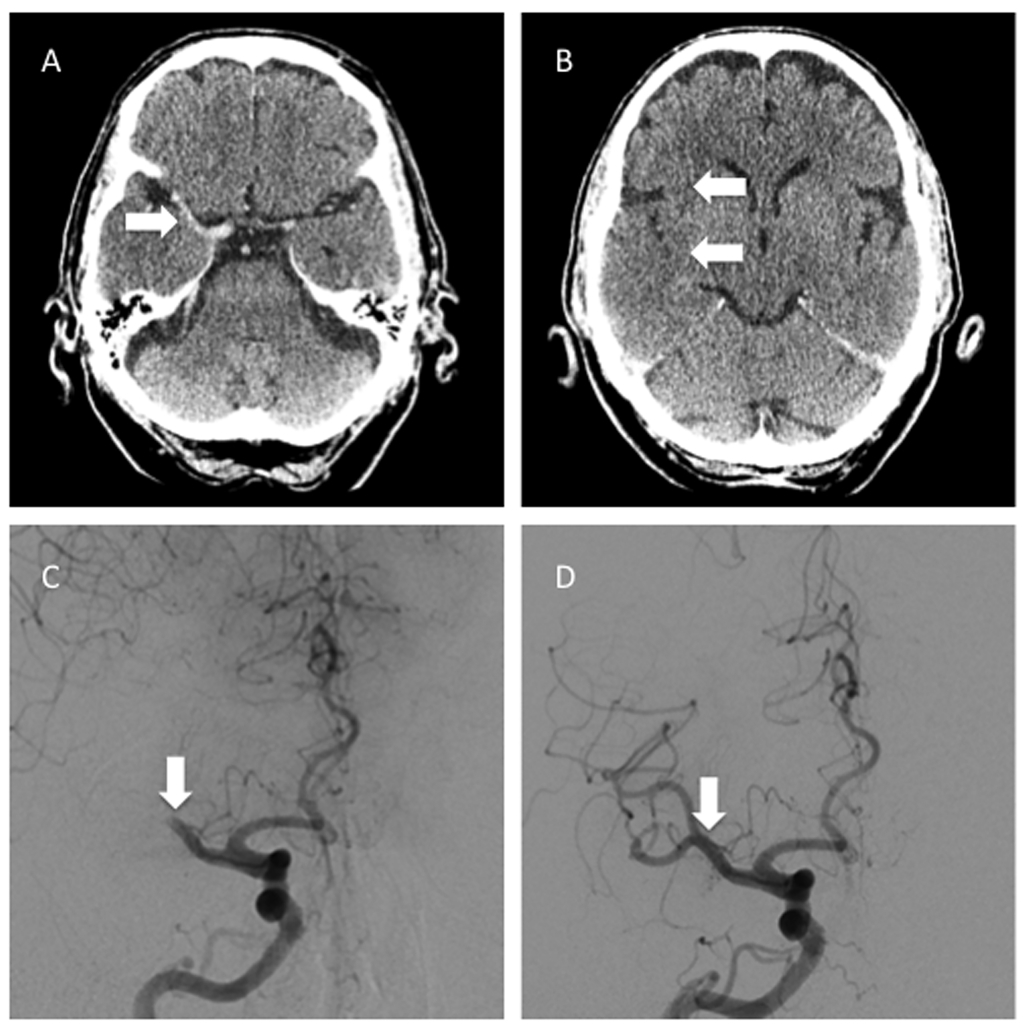
A, B) Axial images from an NCCT in a patient presenting with acute stroke showing a hyperdense MCA and loss of gray-white matter differentiation in the insular cortex, frontal operculum, and temporal operculum. Because of recent major surgery, the patient was not a candidate for IV tPA; however, the patient was able to go for endovascular intervention. Angiographic images from the procedure demonstrate C) the cutoff of the right MCA prior to recanalization and D) restoration of flow through the right MCA post embolectomy with a stent retriever.
Summary and Recommendations
The landscape of management for acute ischemic stroke has changed considerably in recent years, given the recent robust evidence in favor of endovascular intervention for carefully selected patients. Thus, the initial evaluation of patients presenting with acute ischemic stroke must be directed appropriately to identify this population, and this evaluation almost invariably will need to include advanced imaging techniques. In almost every institution, non-contrast head CT will remain the first-line imaging study to exclude hemorrhage to determine patient eligibility for systemic thrombolytic therapy. In institutions that offer or can transfer to institutions that offer endovascular treatment, rapidly obtained vessel lumen imaging, and possibly perfusion imaging, becomes essential. This is especially true for patients who are ineligible for systemic thrombolytic therapy or who present outside of a 4.5-hour or even six-hour time frame. While the ideal imaging protocol for the initial evaluation of acute ischemic stroke may vary depending on the resources available to any given institution, included is a sample “brain attack” protocol (see Figure 19 in online supplement), which illustrates succinctly the role each imaging technique and modality may have to play in triaging and aiding decision-making in the treatment of acute ischemic stroke.
References
- Xu J, et al. Mortality in the United States, 2015. NCHS Data Brief 2016;(267):1-8.
- Mozzafarian D, et al; American Heart Association Statistics Committee; Stroke Statistics Subcommittee. Heart disease and stroke statistics — 2016 update: A report from the American Heart Association. Circulation 2016;133:e38-60.
- Centers for Disease Control and Prevention (CDC). Prevalence and most common causes of disability among adults — United States, 2005. MMWR Morb Mortal Wkly Rep 2009;58:421-426.
- Wardlaw JM, et al. Recombinant tissue plasminogen activator for acute ischaemic stroke: An updated systematic review and meta-analysis. Lancet 2012;379:2364-2362.
- Wardlaw JM, et al. Thrombolysis for acute ischaemic stroke. Cochrane Database Syst Rev 2014;7:CD000213.
- The National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group. Tissue plasminogen activator for acute ischemic stroke. N Engl J Med 1995;333:1581-1587.
- Powers WJ, et al; American Heart Association Stroke Council. 2015 American Heart Association/American Stroke Association focused update of the 2013 guidelines for the early management of patients with acute ischemic stroke regarding endovascular treatment: A guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2015;46:3030-3035.
- Campbell BC, et al; EXTEND-IA Investigators. Endovascular therapy for ischemic stroke with perfusion-imaging selection. N Engl J Med 2015;372:1009-1018.
- Berkhemer OA, et al. A randomized trial of intraarterial treatment for acute ischemic stroke. N Engl J Med 2015;372:11-20.
- Jovin TG, et al. Thrombectomy within 8 hours after symptom onset in ischemic stroke. N Engl J Med 2015;372:2296-2306.
- Saver JL, et al. Stent-retriever thrombectomy after intravenous t-PA vs. t-PA alone in stroke. N Engl J Med 2015;372:2285-2295.
- Goyal M, et al. Randomized assessment of rapid endovascular treatment of ischemic stroke. N Engl J Med 2015;372:1019-1030.
- Kidwell CS, et al. Comparison of MRI and CT for the detection of acute intracerebral hemorrhage. JAMA 2004;292:1823-1830.
- Perry JJ, et al. Sensitivity of computed tomography performed within six hours of onset of headache for diagnosis of subarachnoid hemorrhage: Prospective cohort study. BMJ 2011;343:d4277.
- Bryan R, et al. Diagnosis of acute cerebral infarction: Comparison of CT and MR imaging. AJNR Am J Neuroradiol 1991;12:611-620.
- Kanekar SG, , et al. Imaging of stroke: Part 2, pathophysiology at the molecular and cellular levels and corresponding imaging changes. AJR Am J Roentgenol 2012;198:63-74.
- Marks MP, et al. Acute and chronic stroke: Navigated spin-echo diffusion-weighted MR imaging. Radiology 1996;199:403-408.
- Cheng-Mei S, et al. Swelling of the brain following ischemic infarction with arterial occlusion. AMA Arch Neurol 1959;1:161-177.
- Schaefer PW, et al. Diffusion-weighted MR imaging of the brain. Radiology 2000;217:331-345.
- Barber PA, et al. Validity and reliability of a quantitative computed tomography score in predicting outcome of hyperacute stroke before thrombolytic therapy. ASPECTS Study Group. Alberta Stroke Programme Early CT Score. Lancet 2000;355:1670-1674.
- Puetz V, et al. The Alberta Stroke Program Early CT Score in clinical practice: What have we learned? Int J Stroke 2009;4:354-364.
- Hill MD, Buchan AM; Canadian Alteplase for Stroke Effectiveness Study Investigators. Thrombolysis for acute ischemic stroke: Results of the Canadian Alteplase for Stroke Effectiveness Study. CMAJ 2005;172:1307-1312.
- Demchuk AM, et al. Importance of early ischemic computed tomography changes using ASPECTS in NINDS rtPA Stroke Study. Stroke 2005;36:2110-2115.
- 2Dzialowski I, et al. Extent of early ischemic changes on computed tomography (CT) before thrombolysis: Prognostic value of the Alberta Stroke Program Early CT Score in ECASS II. Stroke 2006;37:973-978.
- 2Puetz V, et al. Extent of hypoattenuation on CT angiography source images predicts functional outcome in patients with basilar artery occlusion. Stroke 2008;39:2485-2490.
- 2Lovblad KO, et al. Clinical experience with diffusion-weighted MR in patients with acute stroke. AJNR Am J Neuroradiol 1998;19:1061-1066.
- 2Gonzalez RG, et al. Diffusion-weighted MR imaging: Diagnostic accuracy in patients imaged within 6 hours of stroke symptom onset. Radiology 1999;201:155-162.
- Sorensen AG, et al. Hyperacute stroke: Evaluation with combined multisection diffusion-weighted and hemodynamically weighted echo-planar MR imaging. Radiology 1996;199:391-401.
- Caldwell J, et al. Imaging in acute ischaemic stroke: Pearls and pitfalls. Pract Neurol 2017;17:349-358.
- Allen LM, et al. Sequence-specific MR imaging findings that are useful in dating ischemic stroke. Radiographics 2012;32:1285-1297.
- Grossman RI, Yousem DM. Neuroradiology: The Requisites. Philadelphia, PA: Mosby; 2003: 183-196, 217.
- Hermier M, Nighogossian N. Contribution of susceptibility-weighted imaging to acute stroke assessment. Stroke 2004;35:1989-1994.
- Santhosh K, et al. Susceptibility weighted imaging: A new tool in magnetic resonance imaging of stroke. Clin Radiol 2009;64:74-83.
- Cheng AL, et al. Susceptibility-weighted imaging is more reliable than T2*-weighted gradient-recalled echo MRI for detecting microbleeds. Stroke 2013;44:2782-2786.
- Fazekas F, et al. Histopathologic analysis of foci of signal loss on gradient-echo T2*-weighted MR images in patients with spontaneous intracerebral hemorrhage: Evidence of microangiopathy-related microbleeds. AJNR Am J Neuroradiol 1999;20:637-642.
- Chalela JA, et al. Magnetic resonance imaging and computed tomography in emergency assessment of patients with suspected acute stroke: A prospective comparison. Lancet 2007;369:293-298.
- Rovira A, et al. Hyperacute ischemic stroke: Middle cerebral artery susceptibility sign at echo-planar gradient-echo MR imaging. Radiology 2004;232:466-473.
- Flacke S, et al. Middle cerebral artery (MCA) susceptibility sign at susceptibility-based perfusion MR imaging: Clinical importance and comparison with hyperdense MCA sign at CT. Radiology 2000;215:476-482.
- Selim M, et al. Diagnosis of cerebral venous thrombosis with echo-planar T2*-weighted magnetic resonance imaging. Arch Neurol 2002;59:1021-1026.
- Singh R, et al. Isolated cortical vein thrombosis: Case series. J Neurosurg 2015;123:427-433.
- Hermier M, et al. Hypointense transcerebral veins at T2*-weighted MRI: A marker of hemorrhagic transformation risk in patients treated with intravenous tissue plasminogen activator. J Cereb Blood Flow Metab 2003;23:1362-1370.
- Viallon M, et al. Combined use of pulsed arterial spin-labeling and susceptibility-weighted imaging in stroke at 3T. Eur Neurol 2010;64:286-296.
- Kesavadas C, et al. Susceptibility weighted imaging: Does it give information similar to perfusion weighted imaging in acute stroke? J Neurol 2011;258:932-934.
- Liebeskind DS, et al. Intravascular deoxygenation of leptomeningeal collaterals detected with gradient-echo MRI. Stroke 2004;35:266.
- Huang P, et al. Clinical applications of susceptibility weighted imaging in patients with major stroke. J Neurol 2012;259:1426-1432.
- Chakraborty S, et al. Beating the clock: Time delays to thrombolytic therapy with advanced imaging and impact of optimized workflow. J Stroke Cerebrovasc Dis 2015;24:1270-1275.
- Power S, et al. Value of CT angiography in anterior circulation large vessel occlusive stroke: Imaging findings, pearls, and pitfalls. Eur J Radiol 2015;84:1333-1344.
- Faggioli G, et al. Atherosclerotic aortic lesions increase the risk of cerebral embolism during carotid stenting in patients with complex aortic arch anatomy. J Vasc Surg 2009;49:80-85.
- Faggioli GL, et al. Aortic arch anomalies are associated with increased risk of neurological events in carotid stent procedures. Eur J Vasc Endovasc Surg 2007;33:436-441.
- Faggioli G, et al. Measurement and impact of proximal and distal tortuosity in carotid stenting procedures. J Vasc Surg 2007;46:1119-1124.
- Goyal M, et al. Consistently achieving computed tomography to endovascular recanalization < 90 minutes: Solutions and innovations. Stroke 2014;45:e252-e256.
- Almekhlafi MA, et al. Ultrashort imaging to reperfusion time interval arrests core expansion in endovascular therapy for acute ischemic stroke. J Neurointerv Surg 2013;5:58-61.
- Bash S, et al. Intracranial vascular stenosis and occlusive disease: Evaluation with CT angiography, MR angiography, and digital subtraction angiography. AJNR Am J Neuroradiol 2005;26:1012-1021.
- Mishra SM, et al. Early reperfusion rates with IV tPA are determined by CTA clot characteristics. AJNR Am J Neuroradiol 2014;35:2265-2272.
- Furlan A, et al. Intra-arterial prourokinase for acute ischemic stroke. The PROACT II Study: A randomized controlled trial. Prolyse in Acute Cerebral Thromboembolism. JAMA 1999;282:2003-2011.
- Riedel C, et al. The importance of size: Successful recanalization by intravenous thrombolysis in acute anterior stroke depends on thrombus length. Stroke 2011;42:1775-1777.
- Saqqur M, et al. Site of arterial occlusion identified by transcranial Doppler predicts the response to intravenous thrombolysis for stroke. Stroke 2007;38:948-950.
- Mattle HP, et al. Comparison of intraarterial and intravenous thrombolysis for ischemic stroke with hyperdense middle cerebral artery sign. Stroke 2008;39:379-380.
- Miteff F, et al. The independent predictive utility of computed tomography angiographic collateral status in acute ischaemic stroke. Brain 2009;132(Pt 8):2231-2238.
- Maas MB, et al. Collateral vessels on CT angiography predict outcome in acute ischemic stroke. Stroke 2009;40:3001-3005.
- Menon BK, et al. Regional leptomeningeal score on CT angiography predicts clinical and imaging outcomes in patients with acute anterior circulation occlusions. AJNR Am J Neuroradiol 2011;32:1640-1645.
- Tan IY, et al. CT angiography clot burden score and collateral score: Correlation with clinical and radiologic outcomes in acute middle cerebral artery infarct. AJNR Am J Neuroradiol 2009;30:525-531.
- Schramm P, et al. Comparison of CT and CT angiography source images with diffusion-weighted imaging in patients with acute stroke within 6 hours after onset. Stroke 2002;33:2426-2432.
- Rosenthal ES, et al. Role of recanalization in acute stroke outcome: Rationale for a CT angiogram-based “benefit of recanalization” model. AJNR Am J Neuroradiol 2008;29:1471-1475.
- Christoforidis GA, et al. Predictors of hemorrhage following intra-arterial thrombolysis for acute ischemic stroke: The role of pial collateral formation. AJNR Am J Neuroradiol 2009;30:165-170.
- Bang OY, et al; UCLA-Samsung Stroke Collaborators. Collateral flow averts hemorrhagic transformation after endovascular therapy for acute ischemic stroke. Stroke 2011;42:2235-2239.
- Broderick JP, et al. Endovascular therapy after intravenous t-PA versus t-PA alone for stroke. N Engl J Med 2013;368:893-903.
- Demchuk AM, et al. Comparing vessel imaging: Noncontrast computed tomography/computed tomographic angiography should be the new minimum standard in acute disabling stroke. Stroke 2016;47:273-281.
- Degnan AJ, et al. MR angiography and imaging for the evaluation of middle cerebral artery atherosclerotic disease. AJNR Am J Neuroradiol 2012;33:1427-1435.
- van Laar PJ, et al. Magnetic resonance imaging of the cerebral circulation in obstructive arterial disease. Cerebrovasc Dis 2006;21:297-306.
- Grimm JM, et al. Computed tomography angiography vs 3 T black-blood cardiovascular magnetic resonance for identification of symptomatic carotid plaques. J Cardiovasc Magn Reson 2014;16:84.
- Brinjikji W, et al. Contemporary carotid imaging: From degree of stenosis to plaque vulnerability. J Neurosurg 2016;124:27-42.
- Yang H, et al. Clinical value of black-blood high-resolution magnetic resonance imaging for intracranial atherosclerotic plaques. Exp Ther Med 2015;10:231-236.
- Halliday A, et al; Asymptomatic Carotid Surgery Trial (ACST) Collaborative Group. 10-year stroke prevention after successful carotid endarterectomy for asymptomatic stenosis (ACST-1): A multicentre randomised trial. Lancet 2010;376:1074-1084.
- North American Symptomatic Carotid Endarterectomy Trial Collaborators. Beneficial effect of carotid endarterectomy in symptomatic patients with high-grade carotid stenosis. N Engl J Med 1991;325:445-453.
- Gupta A, et al. Carotid plaque MRI and stroke risk: A systematic review and meta-analysis. Stroke 2013;44:3071-3077.
- Qureshi AI, et al. Intracranial atherosclerotic disease: An update. Ann Neurol 2009;66:730-738.
- Arenillas JF. Intracranial atherosclerosis: Current concepts. Stroke 2011;42(1 Suppl):S20-23.
- Klein IF, et al. Is moderate atherosclerotic stenosis in the middle cerebral artery a cause of or a coincidental finding in ischemic stroke? Cerebrovasc Dis 2010;29:140-145.
- Mazighi M, et al. Autopsy prevalence of intracranial atherosclerosis in patients with fatal stroke. Stroke 2008;39:1142-1147.
- Wong LK. Global burden of intracranial atherosclerosis. Int J Stroke 2006;1:158-159.
- Leng X, et al. Evaluating intracranial atherosclerosis rather than intracranial stenosis. Stroke 2014;45:645-651.
- Tahmasebpour HR, et al. Sonographic examination of the carotid arterires. Radiographics 2005;25:1561-1575.
- Feldmann E, et al. The Stroke Outcomes and Neuroimaging of Intracranial Atherosclerosis (SONIA) trial. Neurology 2007;68:2099-2106.
- Horrow MM, et al. The limitations of carotid sonography: Interpretave and technology-related errors. AJR Am J Roentgenol 2000;174:189-194.
- Demchuk AM, et al. Accuracy and criteria for localizing arterial occlusion with transcranial Doppler. J Neuroimaging 2000;10:1-12.
- Guan J, et al. The diagnostic accuracy of TCD for intracranial arterial stenosis/occlusion in patients with acute ischemic stroke: The importance of time interval between detection of TCD and CTA. Neurol Res 2013;35:930-936.
- Davis SM, et al. Effects of alteplase beyond 3 h after stroke in the Echoplanar Imaging Thrombolytic Evaluation Trial (EPITHET): A placebo-controlled randomised trial. Lancet Neurol 2008;7:299-309.
- Albers GW, et al. Magnetic resonance imaging profiles predict clinical response to early reperfusion: The Diffusion and Perfusion Imaging Evaluation for Understanding Stroke Evolution (DEFUSE) study. Ann Neurol 2006;60:508-17.
- Lansberg MG, et al. MRI profile and response to endovascular reperfusion after stroke (DEFUSE 2): A prospective cohort study. Lancet Neurol 2012;11:860-867.
- Allmendinger AM, et al. Imaging of stroke: Part I, Perfusion CT — overview of imaging technique, interpretation pearls, and common pitfalls. AJR Am J Roentgenol 2012;198:52-62.
- Kidwell CS, et al. A trial of imaging selection and endovascular treatment of ischemic stroke. N Engl J Med 2013;368:914-923.
- Bivard A, et al. Perfusion computer tomography: Imaging and clinical validation in acute ischaemic stroke. Brain 2011;134:3408-3416.
- Bivard A, et al. Perfusion CT in acute stroke: A comprehensive analysis of infarct and penumbra. Radiology 2013;267:543-550.
- Campbell BC, et al. Comparison of computed tomography perfusion and magnetic resonance imaging perfusion-diffusion mismatch in ischemic stroke. Stroke 2012;43:2648-2653.
- Campbell BC, et al. Cerebral blood flow is the optimal CT perfusion parameter for assessing infarct core. Stroke 2011;42:3435-3440.
- Campbell BC, et al. Imaging selection in ischemic stroke: Feasibility of automated CT-perfusion analysis. Int J Stroke 2015;10:51-54.
- Campbell BC, et al. The infarct core is well represented by the acute diffusion lesion: Sustained reversal is infrequent. J Cereb Blood Flow Metab 2012;32:50-56.
- Chemmanam T, et al. Ischemic diffusion lesion reversal is uncommon and rarely alters perfusion-diffusion mismatch. Neurology 2010;75:1040-1047.
- Schellinger PD, et al. Evidence-based guideline: The role of diffusion and perfusion MRI for the diagnosis of acute ischemic stroke: Report of the Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology. Neurology 2010;75:177-185.
- Olivot JM, et al. Optimal Tmax threshold for predicting penumbral tissue in acute stroke. Stroke 2009;40:469-475.
- Zaro-Weber O, et al. Maps of time to maximum and time to peak for mismatch definition in clinical stroke studies validated with positron emission tomography. Stroke 2010;41:2817-2821.
- Cramer BC, et al. Renal function following infusion of radiologic contrast material. A prospective controlled study. Arch Intern Med 1985;145:87-89.
- Heller CA, et al. Failure to demonstrate contrast nephrotoxicity. Med J Aust 1991;155:329-332.
- X-ray Risk: Risk calculator. Available at: http://www.xrayrisk.com/calculator/calculator.php. Accessed Sept. 6, 2017.
- Stewart FA, et al. ICRP publication 118: ICRP statement on tissue reactions and early and late effects of radiation in normal tissues and organs — threshold doses for tissue reactions in a radiation protection context. Ann ICRP 2012;41:1-322.
- Wang CL, et al. Frequency, outcome, and appropriateness of treatment of nonionic iodinated contrast media reactions. AJR Am J Roentgenol 2008;191:409-415.
- Hunt CH, et al. Frequency and severity of adverse effects of iodinated and gadolinium contrast materials: Retrospective review of 456,930 doses. AJR Am J Roentgenol 2009;193:1124-1127.
- Brinjikji W, et al. Neurons over nephrons: Systematic review and meta-analysis of contrast-induced nephropathy in patients with acute stroke. Stroke 2017;48:1862-1868.
- Hinson JS, et al. Risk of acute kidney injury after intravenous contrast media administration. Ann Emerg Med 2017;69:577-586.
- Loh Y, et al. The risk of acute radiocontrast-mediated kidney injury following endovascular therapy for acute ischemic stroke is low. AJNR Am J Neuroradiol 2010;31:1584-1587.
- Sharma J, et al. Risk of contrast-induced nephropathy in patients undergoing endovascular treatment of acute ischemic stroke.
J Neurointerv Surg 2013;5:543-545. - Ciccone A, et al. Endovascular treatment for acute ischemic stroke. N Engl J Med 2013;386:904-913.
- Goyal M, et al; HERMES collaborators. Endovascular thrombectomy after large-vessel ischaemic stroke: A meta-analysis of individual patient data from five randomised trials. Lancet 2016;387:1723-1731.
Any clinician who may have a role in the initial triage and management of these patients would be well-served to have an understanding of the currently available imaging modalities and techniques, and the applications of each in the evaluation of acute ischemic stroke.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
