A Review of Heart Failure and Current Therapeutic Strategies
March 1, 2018
Reprints
AUTHORS
Hunter Mwansa, MD, St. Vincent Charity Medical Center, Case Western Reserve University, Cleveland, OH
Sula Mazimba, MD, MPH, Division of Cardiovascular Medicine, University of Virginia Health System, Charlottesville, VA
PEER REVIEWER
Glen D. Solomon, MD, FACP, Professor and Chair, Department of Internal Medicine, Wright State University, Boonshoft School of Medicine, Dayton, OH
EXECUTIVE SUMMARY
The management of heart failure falls primarily on the primary care physician. Because of the Medicare financial penalty on hospitals for readmissions, primary care physicians need to be cognizant of the updated treatment options and work with colleagues across multiple disciplines to prevent unnecessary hospitalizations and improve outcomes.
- Heart failure is a common disease. It is the primary diagnosis in more than 1 million hospitalizations and contributes to 2-3 million additional hospitalizations.
- Screening at-risk patients with B-type natriuretic peptide or N-terminal pronatriuretic peptide has reduced onset of left ventricular dysfunction, symptomatic heart failure, and cardiac events.
- Multiple pharmacological and device interventions need to be partnered with control of comorbid risk factors such as diabetes, hypertension, and obesity.
Heart failure (HF) is a complex clinical syndrome that is characterized by the heart’s inability to meet the body’s metabolic demands because of an underlying structural and/or functional impairment of ventricular filling or ejection of blood. Patients with HF usually present with effort intolerance, dyspnea on exertion or at rest, and, in some cases, symptoms of tissue congestion. (See Table 1.) HF is the phenotypic manifestation of injury to the cardiac components (endocardium, myocardium, pericardium, valvular, conduction system, and the great vessels).
HF commonly is stratified using a noninvasive contractile imaging parameter, ejection fraction (EF). With this construct, HF is stratified into HF with reduced ejection fraction (HFrEF) vs. HF with preserved ejection fraction (HFpEF), with EF thresholds of ≤ 40% and ≥ 50%, respectively. Using these definitions, about 50% of patients with HF have HFpEF.1 More recently, the European HF consensus guidelines proposed another HF category with mid-range EF (HFmrEF), with an EF 41-49%.2 Patients with HFmrEF share similar characteristics with HFrEF and HFpEF, and HFmrEF is considered a subcategory of HFpEF. Interestingly, there is a subset of patients in this range of EF (> 40%) who may have improved from a previously low EF (< 40%). These patients are referred to more appropriately as HFpEF with recovered EF.2
Table 1. Clinical Presentation of HF |
Symptoms |
|
Signs |
|
Supportive Findings |
|
Heart Failure Epidemiology
HF is a very common disease condition whose prevalence is increasing, in part, due to the aging U.S. population.3,4 HF is the primary diagnosis in more than 1 million hospitalizations in the United States, and it contributes to 2 to 3 million additional hospitalizations.5 The increased survival of the general population is attributed to advances in the medical treatment of cardiovascular diseases, especially coronary artery disease (CAD).6 From a public health standpoint, about 5.7 million U.S. adults are estimated to be living with HF.7 The lifetime risk of developing HF among Americans older than 40 years of age is about 20%.8 It is estimated that more than 650,000 new HF cases are diagnosed every year,9,10 accounting for more than 15 million physician office visits annually.11 As such, HF poses a huge financial burden on the U.S. economy, with an estimated annual cost of care in excess of $40 billion.9 HF is widely prevalent in older individuals, with the incidence rising from about 20 per 1,000 persons in adults 65 to 69 years of age to more than 85 per 1,000 persons in those 85 years of age or older.10 HF risk and mortality are disproportionately higher among African Americans compared to whites.12 For example, the prevalence of HF in African-American males and females is 4.3% and 3.8%, respectively, compared to 2.7% and 1.8 %, respectively, in demographics for whites.
HF contributes significantly to the burden of morbidity and mortality in the United States. HF contributes to one in every nine deaths. Furthermore, 50% of patients with HF die within five years of diagnosis.7 Fortunately, there has been a progressive trend toward improved prognosis in HF that partly may explain the increased prevalence of the disease.13,14
Heart Failure Risk Factors
There are multiple risk factors for HF. By far, hypertension (HTN) is the most common cardiovascular disease risk factor in the United States. HTN substantially increases the risk of developing HF in both men and women.15 Both systolic and diastolic blood pressure elevations are associated with an increased risk of HF.15,16 Effective blood pressure control can reduce the risk of HF by as much as 50%.17,18 Among hypertensive subjects, antecedent myocardial infarction also predicts an increased risk of HF.15 Recent guidelines on HTN management have called for tighter goals of blood pressure levels.19
Diabetes mellitus is another common condition that significantly increases the risk of developing HF independent of the structural heart aberrations.20 Not surprisingly, the risk increases substantially with the conjoining of other traditional risk factors. For instance, the annual risk of HF among diabetics in isolation has been reported in one study to be 3.0%, which rises to 8.2% when clustering of other traditional risk factors is present.21
The compounding effect of these risk factors on the development of HF also is observed in patients with metabolic syndrome (abdominal adiposity, hypertriglyceridemia, low high-density lipoprotein, elevated fasting glucose, and HTN), which is significantly associated with incipient HF. Importantly, the effective treatment and control of these comorbid conditions can attenuate the risk of developing HF.22 Commonly encountered risk factors for HF are noted in Table 2. It is well recognized that treatment of risk factors also can modify the natural history and overall trajectory of HF. This understanding of the inciting role of risk factors in the development of HF formed the basis for the development of the American College of Cardiology/American Heart Association (ACC/AHA) stages of HF classification system. In this classification system, patients with HF risk factors and no overt symptoms were grouped in Stage A HF. The intent of this nomenclature is to foster early recognition and treatment of risk factors before structural heart damage ensues. This cuts across the entire spectrum of HF phenotypes.
Table 2. Common Risk Factors for Heart Failure |
|
|
a Most common risk factor in the United States |
Classification of Heart Failure
There are several HF classification schemas (e.g., acute vs. chronic). However, the New York Heart Association (NYHA) functional classification, based on symptoms and functional capacity, has been the most enduring and has been employed in major clinical trials.23 As mentioned, the ACC/AHA classification system considers the risk factors, development, and progression of HF. The ACC/AHA staging and NYHA functional classification systems complement each other in the evaluation and management of HF. (See Table 3.) The NYHA classification is clinician dependent and can be subjective, but has been widely adopted and has been shown to predict mortality independently.24
Table 3. ACC/AHA Staging and NYHA Classification of HF |
|||
ACC/AHA Stages of HF |
NYHA Classes of HF |
||
|
A |
At risk for HF but without structural heart disease or symptoms of HF |
None |
|
|
B |
Structural heart disease but without signs or symptoms of HF |
I |
No limitation of physical activity. Ordinary physical activity does not cause symptoms of HF |
|
C |
Structural heart disease with prior or current symptoms of HF |
I |
No limitation of physical activity. Ordinary physical activity does not cause symptoms of HF |
|
II |
Slight limitation of physical activity. Comfortable at rest, but ordinary physical activity results in symptoms of HF |
||
|
III |
Marked limitation of physical activity. Comfortable at rest, but less than ordinary activity causes symptoms of HF |
||
|
D |
Refractory HF requiring specialized interventions |
IV |
Unable to carry on any physical activity without symptoms of HF, or symptoms of HF at rest |
|
ACC, American College of Cardiology; AHA, American Heart Association; HF, heart failure; and NYHA, New York Heart Association |
|||
The ACC/AHA staging categorizes HF into four stages: A, B, C, and D. Stage A comprises patients who have risk factors for developing HF, are asymptomatic, and have no structural heart disease. These risk factors include hypertension, coronary artery disease, alcoholism, diabetes, and cardiotoxin exposure, among others. Stage B HF includes patients with evidence of structural heart disease, such as left ventricular (LV) systolic dysfunction, prior myocardial infarction, dilated cardiomyopathy, valvular heart disease, and left ventricular hypertrophy (LVH) who have no symptoms of HF. Stages C and D constitute patients with current or prior symptoms of HF in the setting of underlying structural heart disease and those with refractory symptoms of HF, respectively.
Etiologies of Heart Failure
Causes of HF are classified into ischemic or nonischemic etiologies.25 CAD accounts for about 70% of cases of HF in the developed world. Nonischemic causes vary broadly and include cardiomyopathies (dilated, restrictive, and hypertrophic), valvular abnormalities, arrhythmias, and pericardial diseases.
Dilated cardiomyopathy (DCM) is a common structural cause of HF that is characterized by ventricular dilatation and compromised myocardial contractility in the absence of abnormal loading conditions like HTN and valvular heart disease.26,27 About 20-35% of cardiomyopathies have underlying genetic abnormalities.28
Figure 1 shows nonischemic dilated cardiomyopathy. Genetic counseling and testing may be appropriate in suspected familial cardiomyopathies, which account for one-third of the HF cases with DCM.23 Symptomatic HF patients with underlying DCM have a poor prognosis, with an estimated 25% dying at one year from diagnosis.29 (See Table 4.)
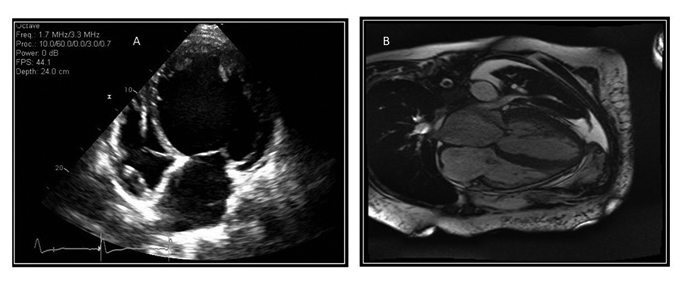
Figure 1. Nonischemic Dilated Cardiomyopathy |
 |
|
A: Two-D-echocardiographic image from the apical 4 chamber view demonstrating a dilated cardiomyopathy. Note the dilated left ventricular cavity. |
Table 4. Causes of Dilated Cardiomyopathy |
|
1. Familial cardiomyopathies: Familial and idiopathic 2. Endocrine and metabolic factors: Obesity, diabetes, thyroid disease, acromegaly, and growth hormone deficiency 3. Toxins: Alcohol, cocaine, chemotherapeutic agents (e.g., anthracyclines and trastuzumab) 4. Nutritional deficiencies (e.g., thiamine) 5. Tachycardia-induced (e.g., supraventricular and ventricular tachycardia) 6. Infectious myocarditis: Chagas, human immunodeficiency virus, and other viruses 7. Inflammatory myocarditis: Connective tissues diseases (e.g., systemic lupus erythematosus and scleroderma) and hypersensitivity 8. Peripartum cardiomyopathy 9. Iron overload as occurs with hemochromatosis 10. Amyloidosis 11. Stress (Takotsubo) cardiomyopathy 12. Cardiac sarcoidosis |
Ischemic cardiomyopathy is notably the most common cause of HF in the United States and other Western countries.20 Patients with ischemic cardiomyopathy have significant coronary artery disease and may have had prior myocardial infarction.2
Restrictive cardiomyopathies are a heterogeneous group of disorders characterized by stiffened myocardium resulting from infiltration, storage, or deposition of substances. This leads to increased cardiac filling pressures and diastolic abnormalities. Some conditions that lead to restrictive cardiomyopathies include cardiac amyloidosis (see Figure 2), sarcoidosis, and hemochromatosis. Importantly, patients with restrictive cardiomyopathy have been excluded from most HF clinical trials, and the standard therapies used in HFrEF even may be detrimental in this group.
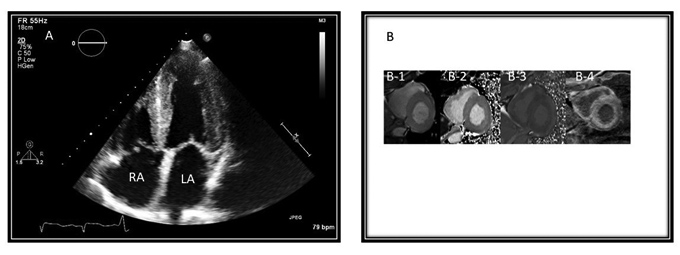
Figure 2. Cardiac Amyloidosis |
 |
|
A: Two-D-echocardiographic image from the apical 4 chamber view demonstrating biatrial enlargement and hyper-enhanced myocardium consistent with amyloidosis. RA = right atrium; LA = Left atrium |
Pathophysiology of HF
Several conceptual models have been advanced in an attempt to explain HF, but since HF is a heterogeneous syndrome, a single unifying model across the spectrum of disease has been elusive. Earlier conceptual paradigms, such as the cardiorenal model, viewed HF as a primary problem involving sodium and water retention. The cardiocirculatory or hemodynamic model30 conceived HF as a consequence of pathologic abnormalities of myocardial contractility. Neither of these models was able to account for the progressive nature of the disease even after a seemingly trivial insult.30
A relatively more recent and widely embraced conceptual paradigm is the neurohormonal model. This model conceptualizes HF as a consequence of a cascade of neurohormonal mechanisms that are initiated following a seminal cardiac insult. Myocardial injury results in adaptive activation of the neurohormonal systems, including the adrenergic nervous system (ANS) and the renin-angiotensin aldosterone system (RAAS). Activation of these systems provokes the release of biomolecules such as norepinephrine, angiotensin II, and aldosterone that are initially compensatory but when perpetuated become deleterious to the overall cardiac function. Adrenergic system activation results in increased myocardial contractility, heart rate, and circulatory volume (via nonosmotic beta-1-mediated vasopressin release), which is augmented by the effects of the RAAS mediated through angiotensin II (vasoconstriction effects) and aldosterone (sodium/water retention). These effects help a failing heart maintain adequate cardiac output to preserve end-organ perfusion. With time, the sustained activation of these neuro-endocrine pathways results in maladaptive remodeling of the cardiovascular system.30 (See Figure 3.) For example, elevated levels of aldosterone have been shown to cause direct damage to cardiac myocytes and vascular endothelial tissue, contributing to the remodeling process that occurs in HF.31 Other molecules also act in an autocrine/paracrine signaling manner. These molecules include cytokines, tumor necrosis factor (TNF), and natriuretic peptides, which are released by cardiac tissue. Endothelin, cytokines/TNF, and natriuretic peptides mediate endothelial dysfunction, inflammatory changes including fibrosis (part of the remodeling process), and vasodilation (to counteract excessive vasoconstriction resulting from overactivation of the ANS and RAAS), respectively. Targeted modulation of these biological pathways has informed the development of therapeutic armamentarium now referred to as guideline-directed medical therapies (GDMT).32-37 Unfortunately, the aforementioned models have not been adequate in guiding a unified conceptual understanding of HFpEF, which may explain the paucity of effective therapies for this disease.
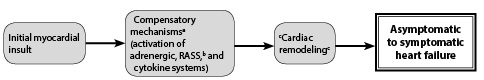
Figure 3. Simplified Model of Heart Failure |
 |
|
a Initial compensatory mechanisms if sustained and persistent become maladaptive, leading to secondary damage. |
Evaluation for HF
When evaluating patients with HF symptoms, it is important to elicit a detailed history, which might help uncover preexisting comorbid conditions and risk factors for HF. (See Tables 2 and 3.) Obtaining a three-generation family history also might help in identifying patients with heritable causes of HF. Not infrequently, patients with established HF also might present with worsening symptoms such as dyspnea suspicious for an acute exacerbation. These patients must be queried on potential triggers of the exacerbation. Common triggers of HF exacerbation include nonadherence to medications and sodium/water restriction; uncontrolled blood pressure; alcohol and recreational drug use; arrhythmias (e.g., atrial fibrillation); acute myocardial ischemia; concurrent infections (especially pulmonary infections); medications such as beta-blockers and calcium channel blockers, especially when initiation is ill-timed or done without appropriate titration; nonsteroidal anti-inflammatory drugs (NSAIDs) and steroids; and valvular and pericardial conditions. Ascertaining a patient’s baseline NYHA functional class helps in risk stratification and assessment of the condition’s severity, which also may provide a benchmark for assessment of interventions.
A thorough physical examination is equally vital in the diagnosis of HF. Physical signs may aid in the diagnosis and risk stratification of HF patients. (See Table 1.) The presence of morbid obesity might raise the concern for pre-existing metabolic syndrome or sleep-disordered breathing. Characterization of the pulse might help identify underlying rhythm abnormalities such as atrial fibrillation. Pericardial rubs or knocks and murmurs might signal the presence of pericardial disease and functional and/or structural valvular abnormalities, respectively. An S3 gallop has moderate correlation with increased cardiac filling pressures and might indicate poor prognosis in those patients with established HFrEF.38 Other important signs of volume overload include exaggeration of jugular venous distension (hepatojugular reflux) on application of abdominal pressure, rales, liver enlargement, ascites, and extremity edema. Cold extremities might be indicative of significant hypoperfusion and the need for inotropic support or the need for mechanical circulatory support.2,23
Diagnostic tests can help establish the diagnosis and cause of HF. Valuable routine laboratory tests include a complete blood count, complete metabolic panel, magnesium levels, lipid panel, and thyroid function tests. (See Table 5.)
Table 5. Laboratory Tests Useful in Initial Evaluation |
|
|
Laboratory Test |
Importance |
|
Complete blood count (CBC) |
|
|
Complete metabolic panel (CMP) |
|
|
Serum magnesium |
|
|
Natriuretic peptide levels |
|
|
Cardiac troponins |
|
|
Lipid panel |
|
|
Thyroid function tests |
|
Role of Biomarkers in HF
Biomarkers increasingly have become useful in the diagnosis, prognostication, and most recently prevention of HF. The most commonly used are the B-type natriuretic peptide (BNP) and its counterpart, N-terminal pronatriuretic peptide (NT-proBNP). These biomolecules are secreted primarily by the over-stretched ventricular heart muscle. BNP’s main effects are natriuresis, diuresis, and smooth muscle relaxation, whereas NT-proBNP is an inactive molecule. These biomolecules correlate with increased filling pressures or ventricular load in HF and are significantly elevated in decompensated HF.39,40 Both markers have fairly high sensitivities for HF. They both have very poor positive predictive values but high negative predictive values. Used in the right clinical setting, both markers can aid in the diagnosis of HF. In patients presenting with clinical symptoms of HF like dyspnea and leg swelling, a low BNP can be used to exclude the possibility of HF.41,42 It is important to understand that there are several cardiac and noncardiac conditions that might cause elevation in BNP and NT-proBNP. (See Table 6.)
Table 6. Causes of Elevated BNP and NT-proBNP |
|
Cardiac Causes |
Noncardiac Causes |
|
|
More recently, natriuretic peptides have been shown to have potential benefit in preventing development of LV dysfunction and HF in at-risk patients, such as those with hypertension, diabetes, and dyslipidemia. In two recent studies, researchers demonstrated that screening at-risk patients with BNP or NT-proBNP and referring those with elevated levels to cardiologists for appropriate evaluation and timely treatment reduced the onset of asymptomatic LV dysfunction and symptomatic HF43 and cardiac events.44 Currently, there are no standardized screening recommendations, so applying these new insights will require further studies on long-term patient outcomes, evaluation of risks to patients, as well as cost-benefit analysis.
Elevated natriuretic peptide levels portend poor clinical outcomes and increased mortality.39,45-51 There is evidence demonstrating prognostic value in the measurement of natriuretic peptide levels during admission.45,50,51 Some observational studies have suggested that predischarge natriuretic peptide levels can be useful in establishing postdischarge prognosis in HF.47,52,53 Other biomarkers of myocardial fibrosis, such as soluble ST2 receptor, galectin-3, and high-sensitivity cardiac troponin, have been studied, and there is evidence to suggest increased predictive value when used in combination with cardiac natriuretic peptides and troponins.54,55
Other useful diagnostic tests include an electrocardiogram that might show evidence of myocardial ischemia, arrhythmias like atrial fibrillation, LVH, and other nonspecific changes indicative of a possible etiology of HF.
Imaging with a chest X-ray (CXR) might reveal pulmonary congestion and cardiomegaly. (See Figure 4.) It is important to realize that the CXR has poor sensitivity and specificity for HF, and can be negative in 20% of patients with chronic HF because of compensatory lymphatic drainage of fluid.56 Other nonspecific findings, such as valvular calcifications and pericardial calcifications, might be present on CXR. The CXR also helps exclude other pulmonary causes of dyspnea, such as pneumonia.
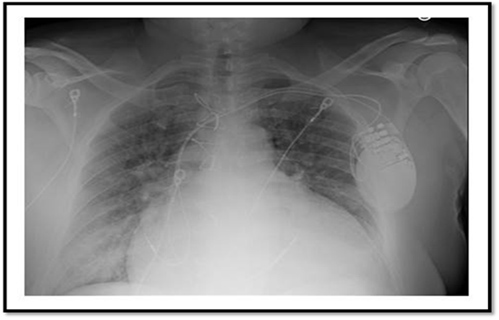
Figure 4. Chest X-ray Showing Cardiomegaly |
 |
|
A chest X-ray showing cardiomegaly (enlarged cardiac silhouette) with prominent pulmonary vascular markings consistent with pulmonary venous congestion. A dual chamber implantable cardioverter defibrillator is also seen. |
Two-dimensional echocardiography (2-D echo) with Doppler is one of the most useful noninvasive cardiovascular imaging tools for patients with suspected HF. Echocardiography helps in the anatomic and functional evaluation of the heart. Systolic and diastolic contractile abnormalities might point to possible etiology of HF. Other abnormalities that can be detected on 2-D echo include valvular abnormalities, myocardial infiltrative processes like amyloidosis, and pericardial disease.
In patients with HF of unknown etiology and in whom myocarditis and infiltrative processes like amyloidosis are considered a possible cause, cardiac magnetic resonance imaging (CMRI) can be useful.57 CMRI is able to provide details of cardiac function, myocardial perfusion, viability, fibrosis, and cardiac anatomic abnormalities. However, it is expensive and is of limited utility in patients with significant tachycardia. Rarely, endomyocardial biopsies are performed in patients with rapidly progressive and refractory acute HF and when giant cell myocarditis and infiltrative myocardial processes associated with ventricular arrhythmias and/or atrioventricular conduction delays are suspected.58 Results of myocardial biopsy might help influence treatment decisions in patients with primary cardiac amyloidosis. CMRI is valuable as a noninvasive diagnostic modality in patients suspected of having cardiac amyloidosis. (See Figure 3.)
Coronary angiography has an important role in patients suspected of having an underlying ischemic heart disease.
Management of HF
Treatment of Stage A HF: Management of stage A HF involves control of risk factors (see Table 2) to prevent and/or delay remodeling that eventually would lead to overt HF. Diabetes leads to morphological, biochemical, and functional cardiac abnormalities independent of the acceleration of atherosclerosis. This may explain the increased lifetime risk of HF among diabetics.59 This risk is higher among women with diabetes and those with poor glycemic control with glycated hemoglobin greater than 10.5%.60 Treatment of diabetes and incident diabetic renal disease might help reduce the risks of developing HF.
Long-standing hypertension inevitably leads to LVH, subsequent HF, and cardiovascular death. Studies have shown a reduction in the incidence of LVH and HF by 50% with use of antihypertensive therapy, a trend that also translated into reduced cardiovascular mortality.18,61 Most recently, a large randomized, controlled trial (RCT) showed that in patients with increased cardiovascular risk (defined as those older than 75 years of age and with pre-existing vascular disease, chronic renal disease, or a Framingham Risk Score >15%), intensive blood pressure control to a goal systolic blood pressure (SBP) < 120 mmHg was associated with significant reduction in incidence of HF.62 Therefore, the ACC/AHA recommend treatment of hypertension to a goal blood pressure of 130/80 mmHg or less in those with stage A HF.63 The choice of antihypertensive agents should be based on guidelines,19 but may be modified according to specific indications like coexisting CAD, kidney disease, and diabetes. Diuretic-based antihypertensive therapies consistently have shown a benefit in preventing incident HF.64 Angiotensin-converting enzyme (ACE) inhibitor treatment of asymptomatic diabetics and patients with vascular disease without overt HF symptoms demonstrated improved outcomes in terms of death, myocardial infarction, and stroke.65 Use of losartan, an angiotensin II type 1 receptor blocker, showed delay to the first hospitalization for HF in patients with diabetic nephropathy.66
The treatment of hyperlipidemia, especially in patients with existing CAD using statins, might be beneficial in some patients. In patients at risk for HF, particularly those with dyslipidemia, statin therapy has been shown to be effective in preventing HF.67,68
Obesity must be addressed with lifestyle modification measures that promote weight loss, such as a low-calorie diet and exercise. Sodium intake is associated with hypertension, LV hypertrophy, and cardiovascular disease.69,70 Paradoxically, patients with HF with obesity have better prognosis.71 Therefore, patients with stage A HF must keep sodium intake to less than 1,500 mg per day.23 Patients also must be counseled on the dangers of excessive alcohol consumption, tobacco use, and recreational drug use, and helped with cessation methods. NSAIDs have been associated with increased hospitalization for HF, increased incidence of new-onset HF, and decompensation of chronic HF.72 These effects mainly are related to fluid retention associated with these medications. Patients with prior or current use of cardiotoxic chemotherapeutic agents must be evaluated for possible LV dysfunction.
Treatment of Stage B HF: Treatment of HF patients in stage B focuses on risk factor control to delay and reverse the disease progression. Treatment goals also include incorporating recommendations for stage A. Therapies aimed at promoting reverse remodeling include the use of ACE inhibitors, angiotensin II receptor blockers (ARBs), beta-blockers, and mineralocorticoid receptor antagonists in patients with structural heart disease (underlying CAD and ventricular hypertrophy and dysfunction ). These medications have been shown to help delay the onset of symptomatic HF as well as reduce mortality. ACE inhibitors, or ARBs for those intolerant to ACE inhibitors, are effective in preventing progression to symptomatic HF and reduce mortality in post-myocardial infarction (MI) patients with reduced EF.73 Evidence-based beta-blockers, such as carvedilol, bisoprolol, and metoprolol, have also been shown to reduce onset and/or progression of HF and overall cardiovascular mortality in post-MI patients with reduced EF.74,75 Statins confer a mortality benefit in post-MI patients and can prevent progression of stage B HF.22,76-78 Patients with asymptomatic LV dysfunction with reduced EF should be treated with ACE inhibitors or ARBs if intolerant to ACE inhibitors. ACE inhibitors have been shown to reduce onset of overt HF, rates of hospitalizations, and cardiovascular mortality.79,80 Evidence-based beta-blockers also help reduce morbidity and mortality in asymptomatic patients with reduced LVEF.79
Implantable cardioverter-defibrillator (ICD) placement is beneficial in asymptomatic patients with ischemic cardiomyopathy who are at least 40 days post-MI, have LVEF
≤ 30%, are already on guideline-directed medical therapy (GDMT), and have good functional capacity and life expectancy greater than one year.81
Pharmacologic treatments for stage C and D HF with reduced EF (HFrEF): Treatment goals for stage C and D HFrEF include delay of disease progression, symptomatic relief, and improvement of survival. Pharmacotherapy is complemented by some of the supportive measures already discussed under stage A and B HF. Other nonpharmacological interventions include education, social support, and cardiac rehabilitation with exercise. Education on self-care involves getting patients to understand their medical condition, including monitoring their symptoms and identifying ways to stay healthy with medication, dietary adherence, and exercise programs. HF patients who receive self-care education overall have an improved understanding of the disease process, medication adherence, and time to hospitalization and duration of hospital stay.82
Nonpharmacological interventions may be beneficial and complementary in HF management. For example, there is evidence that disease-specific education at discharge leads to lower six-month mortality.83 A lack of social support has been associated with poor adherence to medical care, higher hospitalization rates,84,85 and mortality risk.86
Weight management presents a delicate balance in HF patients. Those who are morbidly obese and cachexic have worse outcomes compared to those whose body mass index (BMI) is between 30 and 35 kg/m2.87
Cardiac rehabilitation through supervised exercise programs is beneficial in patients who are able to participate in exercise.88 A meta-analysis of RCTs showed that supervised exercise not only improved survival and exercise capacity but also extended the time to admission in patients with HFrEF.89,90 This possibly might be related to reverse remodeling effects of exercise on the heart.91
Management of common comorbid disorders provides incremental benefit in HF. For example, patients with iron deficiency anemia and evidence of low iron have been shown to benefit from intravenous iron repletion.63 Patients at risk for sleep-disordered breathing should undergo sleep studies and be allowed to use continuous positive airway pressure devices, given their benefit in improving symptoms of daytime sleepiness and occurrence of atrial fibrillation.92
Stage D HF represents “truly refractory” heart failure, with patients having persistent severe symptoms despite optimal GDMT. These patients should be evaluated for potentially reversible or alternative etiologies for worsening or persistent symptoms before being labeled stage D HF. In addition, they should be evaluated for candidacy for advanced treatment strategies, including but not limited to mechanical circulatory support and heart transplantation, as well as novel experimental surgical procedures. Palliative and hospice care should be considered early in those who are not candidates for these advanced therapies.23 Patients with stage D HF typically will have severe symptoms, such as dyspnea and/or fatigue at rest or with minimal exertion, fluid retention, echocardiographic or other objective evidence of severe cardiac impairment, severe functional impairment, and more than one hospitalization in less than six months.93 Specific agents used in the treatment of stage C and D HFrEF are summarized in Tables 7 and 8 and are discussed in detail below.
Table 7. Common Medications Used in Patients With HFrEF |
|
Drug |
Dose Range |
|
Angiotensin-converting Enzyme Inhibitora |
|
|
Captopril |
6.25 mg TID to 50 mg TID |
|
Enalapril |
2.5 mg BID to 10 to 20 mg BID |
|
Fosinopril |
5 to 10 mg QD to 40 mg QD |
|
Lisinopril |
2.5 to 5 mg QD to 20 to 40 mg QD |
|
Perindopril |
2 mg QD to 8 to 16 mg QD |
|
Quinapril |
5 mg BID to 20 mg BID |
|
Ramipril |
1.25 to 2.5 mg to QD 10 mg QD |
|
Trandolapril |
1 mg QD to 4 mg QD |
|
Angiotensin Receptor Blockersa |
|
|
Candesartan |
4 to 8 mg QD to 32 mg QD |
|
Losartan |
25 to 50 mg QD to 50 to 150 mg QD |
|
Valsartan |
20 to 40 mg BID to 160 mg BID |
|
Angiotensin Receptor Neprilysin Inhibitor |
|
|
Sacubitril/valsartan |
49/51 mg BID (sacubitril/valsartan) to 97/103 mg BID (sacubitril/valsartan) |
|
Beta-blockersb |
|
|
Bisoprolol |
1.25 mg QD to 10 mg QD |
|
Carvedilol |
3.125 mg BID to 50 mg BID |
|
Carvedilol CR |
10 mg QD to 80 mg QD |
|
Metoprolol succinate extended release (metoprolol CR/XL) |
12.5 to 25 mg QD to 200 mg QD |
|
If Channel Inhibitor |
|
|
Ivabradine |
5 mg BID to 7.5 mg BID |
|
Isosorbide Dinitrate and Hydralazine |
|
|
Fixed-dose combination |
20 mg isosorbide dinitrate/ 37.5 mg hydralazine TID to 40 mg isosorbide dinitrate/ 75 mg hydralazine TID |
|
Isosorbide dinitrate and hydralazine |
20 to 30 mg isosorbide dinitrate/25 to 50 mg hydralazine TID or QID 40 mg isosorbide dinitrate TID with 100 mg hydralazine TID |
|
Cardiac Glycosidesc |
|
|
Digoxin |
0.125 mg QD to 0.25 mg QD |
|
a Medication morbidity and mortality benefit not dose dependent. |
|
ACE inhibitors: ACE inhibitors cause vasodilation and reverse remodeling through suppression of the RAAS, thereby attenuating their deleterious effects on the cardiovascular system. They have been shown to improve symptoms, HF hospitalization, and cardiovascular mortality in patients with mild, moderate, or severe symptomatic HFrEF and in patients with or without CAD.73,94-96 These beneficial effects of ACE inhibitors are not dose-dependent, and no significant difference in mortality benefit has been demonstrated between low-dose and high-dose ACE inhibitor therapy in most RCTs.95,97,98 It is desirable to attempt to titrate doses equivalent to those used in clinical trials.
Angioedema is a rare but potential serious adverse effect related to ACE inhibitor therapy. These medications should be started at low doses to assess for tolerability before being titrated up. Caution must be exercised in patients with hypotension, renal insufficiency, and elevated serum potassium. A dry cough can be seen in 20% of patients taking these medications.
ARBs: ARBs block the effects of angiotensin II at their receptors (type 1). These medications have comparable efficacy to ACE inhibitors but are not superior.99,100,101 ARBs are recommended for patients who are intolerant to ACE inhibitors and those who develop HF while on an ARB for other reasons (i.e., hypertension).63
Angiotensin receptor neprilysin inhibitors (ARNI): ARNI combine the effects of an ARB and an inhibitor of neprilysin, an enzyme that degrades natriuretic peptides, bradykinin, adrenomedullin, and other vasoactive peptides, thereby enhancing vasodilation. A recent RCT (PARADIGM-HF) showed that an ARNI (sacubitril/valsartan) significantly reduced the composite endpoint of cardiovascular death or HF hospitalization by 20% when compared to enalapril in patients with symptomatic HFrEF tolerating an adequate dose of either an ACE inhibitor or an ARB.102 The updated ACC/AHA guidelines incorporated ARNIs among first-line therapies, alongside ACE inhibitors and ARBs, for stage C (symptomatic) HF. Appropriately selected patients with chronic HFrEF NYHA class II-III whose symptoms are refractory on maximal tolerable doses of either an ACE inhibitor or an ARB may be considered as candidates for switching to an ARNI.63
ARNI therapy is associated with hypotension and infrequently angioedema.102 An ARNI should not be administered to patients with a history of angioedema. Concomitant administration with an ACE inhibitor is associated with significantly high risks of angioedema and therefore is contraindicated. A washout period of 36 hours should be allowed from the last dose of an ACE inhibitor before initiation of an ARNI to avoid this potentially serious adverse effect. The ARNI should be started at the lowest dose to assess for tolerability then titrated up to the dose that was tested in the clinical trial.103 As is the case with ACE inhibitors and ARB therapy, patients should be monitored closely for hypotension, renal insufficiency, and hyperkalemia within one to two weeks of initiating them on therapy with an ARNI or whenever there is a change in dosing.
Beta-blockers: Beta-blockers have shown improved survival, event-free survival, quality of life, and rates of hospitalization and sudden cardiac death in patients with mild, moderate, and severe HF.104,105 The beneficial effects of beta-blocker therapy possibly may be related to structural and functional effects of these medications on the heart, their effects on the maladaptive adrenergic drive, and the consequent structural changes imposed on a failing heart. There is evidence to confirm that beta-blockers cause reverse remodeling and improvement in systolic function as early as three to four months from initiation of therapy.106,107 One study showed mortality and hospitalization benefits as early as two to three weeks following initiation of therapy.108
Beta-blockers should be started only in hemodynamically stable patients. Initial doses should be low with a gradual titration up after proven tolerance (generally over two to four weeks or whenever the adverse effects to a lower dose resolve) to low doses to reduce rates of adverse effects and unnecessary withdrawal of therapy. This cautious approach allowed for about 85% of patients enrolled in clinical trials to tolerate short- and long-term treatment to maximum planned trial doses.23 Initiating beta-blocker therapy before discharge for hospitalized HF patients who are not on intravenous inotropic therapy can be done safely.109
Patients should be educated on the benefits of beta-blockers to avoid discontinuation of medications for tolerable side effects. Worsening of HF, fatigue, varying degrees of heart block, and hypotension are potential adverse effects of beta-blocker therapy. Caution should be exercised in patients with reactive airway disease and diabetics with frequent hypoglycemic spells.
Aldosterone Antagonists: The activation of the RAAS in HF results in increased aldosterone production. Aldosterone has multiple biological and physiological effects, including salt and water retention, collagen deposition, direct vascular damage, vascular fibrosis, myocardial hypertrophy, and fibrosis (remodeling).110,111 These effects are inhibited by aldosterone receptor blockade, thereby preventing the remodeling process that characterizes HF.111 When added to standard therapy including an ACE inhibitor, low-dose aldosterone antagonists decrease both morbidity and mortality in patients with severe HF (NYHA III-IV) and EF no more than 35%.37 There is also evidence of morbidity and mortality benefit in post-MI patients with LV dysfunction and/or severe HF treated with a selective aldosterone antagonist eplerenone.112 The EMPHASIS-HF trial demonstrated that eplerenone reduced mortality and rates of hospitalization even in patients with mild symptoms of HF (NYHA II).113 These medications can have considerable effects at doses as low as 12.5 mg daily since their beneficial effects were seen without much effect on water retention.
Aldosterone antagonists can cause hyperkalemia and, therefore, should be given with caution in patients with renal insufficiency and those on ACE inhibitors/ARB therapy or potassium replacement. They generally are contraindicated in patients with a serum creatinine ≥ 2.5 mg/dL and those with serum potassium ≥ 5 mEq/L. Patients with glomerular filtration rates (GFR) between 30 and 49 can be started on these medications once every other day and watched closely for hyperkalemia before titration up.23 Patients should be advised to avoid NSAIDs and watch intake of food rich in potassium after initiation of these drugs.
Hydralazine and Isosorbide Dinitrate: The vasodilator combination of hydralazine and isosorbide dinitrate added to optimal GDMT (including an ACE inhibitor or ARB, beta-blocker, and aldosterone antagonist) in African-American patients with advanced HF (NYHA III-IV HFrEF) was found to improve mortality, morbidity, and quality of life.114 This combination also proved to reduce mortality but not rates of hospitalization in patients with current or prior symptomatic HFrEF not on an ACE inhibitor or beta-blocker but on digoxin and diuretics.115 Therefore, the ACC/AHA recommend treatment of symptomatic patients with HFrEF who cannot tolerate an ACE inhibitor or ARB and beta-blocker with the combination of hydralazine and isosorbide dinitrate, provided there are no contraindications.23 This combination can cause significant dizziness, headaches, hypotension, and gastrointestinal disturbances.
Ivabradine: Ivabradine is a relatively new medication that selectively inhibits the If current in the sinoatrial node, resulting in heart rate reduction. Patients with NYHA II-III HFrEF (EF ≤ 35%) on standard GDMT, including a beta-blocker, were found to benefit from reduced HF hospitalization when ivabradine was added to therapy.116 The study cohort predominantly included patients in sinus rhythm with a resting heart rate of 70 beats per minute or more. Therefore, it is imperative to optimize beta-blocker therapy to maximal tolerable doses before considering ivabradine as an add-on therapy.
Diuretics: Diuretics are indicated for symptomatic relief in patients with HF who have evidence of fluid retention.23 This class of therapeutic agents inhibits the reabsorption of sodium chloride and water in the renal tubes, thereby improving the fluid retention associated with HF. Clinical trials have shown that diuretics lead to improvement in symptoms and exercise tolerance in patients with HF.117,118 Diuretics help reduce volume overload and pulmonary vascular congestion.119 However, there is no evidence of mortality benefit from diuretic use.
In an outpatient setting, diuretics generally should be started at lower doses and titrated up to achieve a goal weight reduction of 0.5 to 1.0 kg daily.23 Therefore, daily weight measurement is vital in the management of patients on diuretic therapy. A key element in the use of diuretics is the judicious use of adequate doses so as to optimize patient comfort while minimizing adverse effects. In some cases, patients may be trained to self-titrate diuretic doses based on symptoms.
A recent RCT showed no major difference in patient’s global assessment of symptoms or renal function when diuretic therapy was administered by continuous infusion as compared to bolus infusion or when a high dose was compared with a low dose diuretic strategy in patients with acute decompensated HF.120 Resistance to diuretics is not uncommon in patients on NSAIDs because of their renal effects;121 this relative unresponsiveness also might occur in patients taking diets rich in salt and those with significant renal dysfunction.122 Use of intravenous diuretics or a combination of different diuretic classes for synergy might overcome this resistance.123,124,125
These medications can cause electrolyte abnormalities, including hypokalemia and hypomagnesemia, especially if used in combination. Therefore, electrolytes should be checked and appropriately replaced. Table 8 shows diuretics commonly used in the treatment of HF.
Table 8. Diuretics Commonly Used in HF Patients |
||
|
Diuretic |
Initial Doses |
Maximum Total Daily Dose |
|
Loop diuretica |
||
|
Bumetanide |
0.5 mg to 1.0 mg QD or BID |
10 mg |
|
Furosemide |
20 mg to 40 mg QD or BID |
600 mg |
|
Torsemide |
10 mg to 20 mg QD |
200 mg |
|
Thiazide diureticsb |
||
|
Chlorothiazide |
250 mg to 500 mg QD or BID |
1,000 mg |
|
Chlorthalidone |
12.5 to 25.0 mg QD |
100 mg |
|
Hydrochlorothiazide |
25 mg QD or BID |
200 mg |
|
Indapamide |
2.5 mg QD |
5 mg |
|
Metolazone |
2.5 mg QD |
20 mg |
|
Potassium-sparing diuretic |
||
|
Amiloride |
5 mg QD |
20 mg |
|
Eplerenonec |
25 mg QD |
50 mg |
|
Spironolactonec |
12.5 mg to 25.0 mg QD |
50 mg |
|
Triamterene |
100 mg to 300 mg BID |
300 mg |
|
a Considered mainstay of diuretic therapy for relief of volume overload. |
||
Digoxin: Digoxin can be used as add-on therapy in patients with HFrEF who are symptomatic despite being on optimal GDMT. It also can be beneficial in patients with severe symptoms not yet responsive to initial GDMT.23 Lower doses, with goal plasma levels of 0.5 to 0.9 ng/mL, are safer than and just as effective as higher doses.126 Several clinical trials have shown that digoxin improves symptoms, exercise tolerance, and health-related quality of life in patients with mild to moderate HF.127-130 Notwithstanding its positive effect on symptoms and hospitalization rates, digoxin confers no mortality benefit.131 Digoxin also is beneficial in chronic HF patients with concomitant atrial fibrillation.
Patients on digoxin need close monitoring, especially elderly patients and those with renal insufficiency, because of the increased occurrence of digitalis toxicity in such patients. Medications like amiodarone and other antiarrhythmic agents, macrolides (e.g., clarithromycin, azithromycin), and itraconazole increase plasma levels of digoxin, thereby increasing the risk for digoxin toxicity.132,133 Major adverse effects of digoxin include nausea, vomiting, arrhythmias, visual disturbances, and neurologic manifestations such as confusion. It should be avoided in patients with second- and third-degree AV block who have no pacemakers and those with significant renal disease.
Other Medications Used in Heart Failure
Inotropes: Hemodynamically unstable patients with HF can benefit from inotropic support. Despite their favorable hemodynamic effects, inotropes have not been shown to alter mortality in patients with advanced HF.134,135,136 In fact, there is evidence of increased mortality associated with chronic oral inotrope use.136 The adverse effects associated with these agents are mostly a result of increased arrhythmias.
However, intravenous inotropes can be useful in HF patients with severe systolic dysfunction associated with significant end-organ hypoperfusion. Ideal patients for intravenous inotropic support typically will manifest with marked symptoms of congestion, hypotension, low cardiac index, and evidence of end-organ hypoperfusion, such as renal dysfunction and congestive hepatopathy. The ACC/AHA recommend use of temporal parenteral inotropic therapy in patients with cardiogenic shock awaiting definitive therapy.23 Patients with stage D HF might benefit from continuous therapy with inotropes, either as a bridge to definitive intervention and/or transplantation or as part of palliative care.23 Common inotropes in use include the following:
• Adrenergic Agents (dopamine and dobutamine): These medications have dose-dependent effects and can help improve renal perfusion and enhance diuresis. However, they can cause hypotension, headaches, and tachyarrhythmias. Furthermore, dopamine might cause tissue necrosis, while dobutamine can result in hypersensitivity reactions. Caution should be exercised in patients on monoamine inhibitors.
• Milrinone: Milrinone is a phosphodiesterase inhibitor that causes significant vasodilation. Potential adverse effects include hypotension and hepatic dysfunction. It requires renal dosing in patients with renal dysfunction.
Intravenous Vasodilators: These drugs generally are used to acutely relieve dyspnea in hospitalized HF patients. Data on their impact on HF outcomes are lacking.23 They include the following drugs:
• Nitroglycerin: Nitroglycerin is an intravenous venodilator that lowers preload, leading to rapid improvement in pulmonary congestion.137,138 It is particularly helpful in patients with elevated blood pressure, acute coronary syndromes, and mitral regurgitation in the setting of acutely decompensated HF. Decreased response to nitroglycerin due to tachyphylaxis can occur with 24 hours of infusion.139,140 It is not uncommon to see this lack of responsiveness even at higher doses of nitroglycerin.141
• Sodium Nitroprusside: Sodium nitroprusside is a potent venodilator and arterio-dilator and, therefore, has both preload and afterload reduction properties. Additionally, it causes vasodilation of the pulmonary vasculature. Like intravenous nitroglycerin, sodium nitroprusside can help relieve symptoms of congestion in HF patients with hypertension and/or hemodynamically significant mitral regurgitation.142 This drug can cause severe hypotension, and cyanide toxicity might occur with prolonged infusion periods, especially in those with renal insufficiency. It must be administered in intensive care unit settings with invasive monitoring.
• Nesiritide: Nesiritide is a form of human B-type natriuretic peptide that causes preload reduction through natriuresis while decreasing afterload through its vasodilatory effects. Studies on nesiritide have yielded mixed results. A large RCT found no benefit from nesiritide in both mortality and hospitalization, and it showed a statistically nonsignificant improvement in dyspnea.143 Further, the study found increased risks for prolonged hypotension with nesiritide use.143
• Arginine Vasopressin Antagonists: These drugs block V1 and/or V2 vasopressin receptors, resulting in electrolyte free water excretion by the kidneys. These drugs are effective in normalizing serum sodium in patients with hypervolemic hyponatremia and, therefore, may be beneficial in normalizing sodium and improving mental performance in patients with refractory hyponatremia despite water restriction and optimal GDMT.144,145 Because of the neurohumoral imbalance that characterizes HF, patients with HF frequently present with hyponatremia. Hyponatremia actually is an indicator of poor outcomes in HF.146 Despite these effects on sodium, long-term therapy with V2-selective vasopressin antagonists showed no mortality benefit in HF.147,148 Conivaptan and tolvaptan currently are available for the treatment of hypervolemic hyponatremic patients. They are relatively new on the market, and their long-term safety and benefit have yet to be established.
Other Nonpharmacological Approaches for Stage C and D HFrEF: Broadly, these include device therapy, revascularization, and surgical intervention. HFrEF is associated with significant risk for sudden cardiac death (SCD) because of the fibrosis that characterizes the remodeling process. Widespread use of neurohormonal antagonists that protect from arrhythmias through cardiac remodeling has significantly reduced rates of SCD. Nonetheless, the risk for SCD still exists.23 Several interventions are discussed below:
• Implantable Cardioverter-defibrillator (ICD): This device reduces rates of SCD from ventricular arrhythmias. It does not alter the disease course. RCTs have demonstrated significant reduction in rates of SCD in symptomatic HF patients (NYHA II and III) on optimal chronic GDMT who have LVEF
≤ 35%.149 Increased occurrence of other events in the first 40 days post-MI preclude early ICD use.150 Therefore, the ACC/AHA strongly recommend ICD for primary prevention of SCD in selected patients with nonischemic dilated cardiomyopathy or ischemic heart disease who are at least 40 days post-MI with LV dysfunction and EF ≤ 35% and mild to moderate symptoms of HF while on optimal GDMT (must have a meaningful survival and life expectancy of more than one year).23
• Cardiac Resynchronization Therapy (CRT): Several RCTs have demonstrated that CRT in appropriately selected patients leads to reduction in hospitalization rates and improvement in HF symptoms, functional capacity, overall quality of life, and mortality.151,152 These observed results likely are the result of CRT’s ability to reverse cardiac remodeling and improve ventricular contractility, ultimately leading to improved LVEF.153,154 A third of HF patients may have accompanying QRS prolongation as the disease progresses. A widened QRS portends poor outcomes.155 The strongest indication for CRT appears to be for patients with NYHA II/III and ambulatory IV with LVEF ≤ 35% with QRS ≥ 150 ms who have a left bundle branch block pattern and are in sinus rhythm.156,157 Accumulating evidence now suggests benefit from CRT even in patients with milder symptoms of HF.158,159 Table 9 includes a summary of compelling indications for CRT in HF patients.
Table 9. Compelling Indications for CRT |
||
Treatment Effect (Benefits Compared to Risks) |
NYHA Class |
|
NYHA Class I |
NYHA Class III and Ambulatory Class IV |
|
|
Benefit >>> Risk |
LVEF ≤ 35%, QRS ≥ 150 ms, LBBB pattern, sinus rhythm |
LVEF ≤ 35%, QRS ≥ 150 ms, LBBB pattern, sinus rhythm |
|
Benefit >> Risk |
LVEF ≤ 35%, QRS 120-149 ms, LBBB pattern, sinus rhythm |
LVEF ≤ 35%, QRS 120-149 ms, LBBB pattern, sinus rhythm |
|
LVEF ≤ 35%, QRS ≥ 150 ms, non-LBBB pattern, sinus rhythm |
||
|
Note: Patients with atrial fibrillation who have undergone atrioventricular nodal ablation and have LVEF ≤ 35% with anticipated good quality of life and lifespan also are likely to benefit from CRT. |
||
Some patients with HF might benefit from angioplasty and coronary artery bypass graft surgery. These interventions can lead to reduced rates of SCD and improvement in symptoms of ischemia and LV function, especially in those with coronary lesions amenable to revascularization.160-163
There has been increasing interest in the role of surgery, including mitral valve repair, aortic valve repair, and surgical myomectomy in the management of certain patients with HF.164-167 In patients with nonischemic advanced HF and severe LV dysfunction accompanied by significant mitral insufficiency, mitral valve repair surgery was shown to promote reversal of LV remodeling and resulted in improved functional capacity.167,168 There also is evidence indicating that combined bypass surgery and mitral valve repair surgery in HF patients with ischemic cardiomyopathy results in improved quality of life.169-171
Some patients with refractory cardiogenic shock in the setting of acute decompensated systolic HF and those with cardiogenic shock following a myocardial infarction might benefit from intra-aortic balloon pump (IABP) placement.172 An IABP is a mechanical device that uses counterpulsation to increase diastolic aortic pressure and reduce systolic aortic pressure, thereby increasing coronary perfusion and reducing myocardial oxygen demand, respectively. This device showed evidence of lower in-hospital mortality in patients with myocardial infarction complicated by cardiogenic shock who underwent thrombolysis and revascularization.173 However, recent evidence suggests that IABPs do not significantly reduce 30-day mortality in patients in whom an early revascularization strategy with PCI or bypass surgery is planned.174 IABPs provide temporal mechanical circulatory support and only serve as a bridge to definitive therapy.
Left ventricular assist devices (LVADs) provide another form of mechanical circulatory support in patients with advanced HF. Mechanical circulatory support with LVAD can be indicated as a bridge to definitive therapy like transplantation or as destination therapy in stage D HF refractory to optimal GDMT in patients who are not suitable candidates for heart transplantation.175-177
Management of Stage C HFpEF
The prevalence of HFpEF has been increasing, with about 50% of all patients with HF having HFpEF.1 The condition is most prevalent in the elderly (most commonly women) with HTN, diabetes, and atrial fibrillation.178 Mortality and hospitalization rates among patients with HFpEF are similar to those observed in patients with HFrEF.179 Nevertheless, deaths from noncardiovascular causes are more prevalent in patients with HFpEF than in patients with HFrEF, in whom death from cardiovascular causes seem predominant.180
Ventricular diastolic dysfunction is characterized by impaired ventricular relaxation, and increased diastolic stiffness at rest or with stress is the hallmark disturbance in HFpEF.181-183 Evidence suggests that the presence of impaired ventricular response to exercise and systolic abnormalities sometimes is evident on echocardiographic measures.179,184 Other findings characteristic of HFpEF include endothelial dysfunction and arterial stiffening, which, along with the ventricular stiffness, cause a heightened sensitivity to pressure or volume loading. This means that HFpEF easily can become symptomatic with small fluid volumes.185 Furthermore, HFpEF patients exhibit significant intolerance to exercise due to a combination of impaired chronotropic and vasodilatory responses in the setting of diminutive ventricular systolic and diastolic reserve.186,187 Significant changes in heart rate like tachycardia and bradycardia also might provoke symptoms in these patients. Little is known on the pathophysiology of HFpEF, and evidence-based therapeutic strategies still are lacking. Traditionally, uncontrolled hypertension leading to vascular dysfunction, concentric LVH, and eventual remodeling has been implicated in its etiology.179 More recently, however, systemic microvascular endothelial inflammation leading to myocardial inflammation and fibrosis (remodeling) has been suggested. This inflammatory cascade is related to underlying conditions like hypertension, diabetes, the metabolic syndrome, smoking, lung disease, and iron deficiency anemia.188,189
HFpEF presents a unique diagnostic challenge. Supportive tests like natriuretic peptide levels may be normal in up to 30% of patients, especially the obese and those with only exertional symptoms.179,190 Furthermore, this condition exists predominantly in elderly patients who might have some diastolic dysfunction but not necessarily HFpEF. Morbidly obese patients also might present with symptoms of fatigue and dyspnea but not necessarily have HFpEF. Therefore, its diagnosis requires a high index of suspicion, appropriate interpretation of clinical and laboratory data, and demonstration of objective measures of diastolic dysfunction on echocardiography in the presence of HF symptoms. A normal resting echocardiography does not necessarily exclude HFpEF (in some cases, exercise may unmask diastolic dysfunction on echocardiogram). Cardiopulmonary exercise testing and invasive measures like right-heart catheterization might aid in diagnosing indeterminate cases of HFpEF.179
Currently, no therapies have been shown to improve outcomes in patients with HFpEF. Therapy in these patients is aimed at symptomatic relief of congestion, improvement of exercise tolerance, and control of coexisting medical conditions. Acute symptoms of volume overload are treated with gentle diuresis. Hypertension must be controlled according to guideline-directed recommendations.19 Most HF patients with hypertension will require a diuretic. Those with stable kidney disease should be considered for ACE inhibitors or ARBs, provided there are no compelling contraindications or intolerance to therapy. Beta-blockers and other antihypertensive agents are equally reasonable options for control of blood pressure when indications are present. ACE inhibitors reduce rates of hospitalization in HFpEF patients, despite their lack of mortality benefit.191 Aldosterone receptor antagonists have been shown to lower rates of hospitalization in appropriately selected patients with HFpEF,192,193 typically those with elevated BNP levels or HF admission within one year, GFR > 30 mL/min/1.73 m2, serum creatinine < 2.5 mg/dL, and potassium < 5.0 mEq/L. CAD should be managed according to current clinical guidelines.194 Coronary revascularization can be used for symptom relief in those with underlying CAD with angina or dyspnea attributable to myocardial ischemia.23 Patients with atrial fibrillation initially must be managed with rate control and anticoagulation, and rhythm control should be attempted in those with refractory symptoms despite rate control.23 Weight loss in obese patients with a BMI ≥ 35% and HFpEF might help improve exercise tolerance.195 Coexisting lung and sleep disorders must be treated to improve symptoms of fatigue and dyspnea. Exercise training also helps improve symptoms of dyspnea and exercise intolerance.196
Summary
HF is a relentlessly progressive disease condition that carries a large burden of morbidity and mortality. It is very costly and remains a major public health challenge. The prevalence of HF remains high and is expected to increase with the aging population. Focused efforts increasingly must address prevention of HF in those with risk factors. Optimal GDMT for patients with established HF is key to improving overall outcomes. There is need for further understanding of the HF syndrome, especially HFpEF, and development of newer therapies to further reduce morbidity and mortality.
REFERENCES
- Gladden JD, Linke WA, Redfield MM. Heart failure with preserved ejection fraction. Pflugers Arch 2014;466:1037-1053.
- Ponikowski P, Voors AA, Anker SD, et al. 2016 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J 2016;37:2129-2200.
- McMurray JJ, Stewart S. Epidemiology, aetiology, and prognosis of heart failure. Heart 2000;83:596-602.
- Mendez GF, Cowie MR. The epidemiological features of heart failure in developing countries: A review of the literature. Int J Cardiol 2001;80:213-219.
- Butler J, Fonarow GC, Gheorghiade M. Need for increased awareness and evidence-based therapies for patients hospitalized for heart failure. JAMA 2013;310:2035-2036.
- Senni M, Tribouilloy CM, Rodeheffer RJ, et al. Congestive heart failure in the community: Trends in incidence and survival in a 10-year period. Arch Intern Med 1999;159:29-34.
- Mozzafarian D, Benjamin EJ, Go AS, et al. On behalf of the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics—2016 update: A report from the American Heart Association. Circulation 2016;133:e38-e360.
- Djousse L, Driver JA, Gaziano JM. Relation between modifiable lifestyle factors and lifetime risk of heart failure. JAMA 2009;302:394-400.
- Go AS, Mozaffarian D, Roger VL, et al. Heart disease and stroke statistics—2013 update: A report from the American Heart Association. Circulation 2013;127:e6-245.
- Curtis LH, Whellan DJ, Hammill BG, et al. Incidence and prevalence of heart failure in elderly persons, 1994-2003. Arch Intern Med 2008;168:418-424.
- O’Connell JB, Bristow MR. Economic impact of heart failure in the United States: Time for a different approach. J Heart Lung Transplant 1994;13:S107-S12.
- Bahrami H, Kronmal R, Bluemke DA, et al. Differences in the incidence of congestive heart failure by ethnicity: The multi-ethnic study of atherosclerosis. Arch Intern Med 2008;168:2138-2145.
- Chen J, Normand S-LT, Wang Y, Krumholz HM. National and regional trends in heart failure hospitalization and mortality rates for Medicare beneficiaries, 1998-2008. JAMA 2011;306:1669-1678.
- Levy D, Jenchaiah S, Larson MG, et al. Long term trends in the incidence of and survival with heart failure. N Engl J Med 2002;347:1397–1402.
- Levy D, Larson MG, Vasan RS, et al. The progression from hypertension to congestive heart failure. JAMA 1996;275:1557-1562.
- Wilhelmsen L, Rosengren A, Eriksson H, et al. Heart failure in the general population of men — morbidity, risk factors and prognosis. J Intern Med 2001;249:253-261.
- Izzo JL, Jr., Gradman AH. Mechanisms and management of hypertensive heart disease: From left ventricular hypertrophy to heart failure. Med Clin North Am 2004;88:1257-1271.
- Baker DW. Prevention of heart failure. J Card Fail 2002;8:333-346.
- Whelton PK, Carey RM, Aronow WS, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/AphA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults: A Case Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol 2017; Nov. 13. doi: 10.1016/j.jacc.2017.11.006. [Epub ahead of print].
- He J, Ogden LG, Bazzano LA, et al. Risk factors for congestive heart failure in US men and women: NHANES I epidemiologic follow-up study. Arch Intern Med 2001;161:996-1002.
- Bibbins-Domingo K, Lin F, Vittinghoff E, et al. Predictors of heart failure among women with coronary disease. Circulation 2004;110:1424-1430.
- Grundy SM, Cleeman JI, Merz CN, et al. Implications of recent clinical trials for the National Cholesterol Education Program Adult Treatment Panel III Guidelines. J Am Coll Cardiol 2004;44:720-732.
- Yancy CW, Jessup M, Bozkurt B, et al. 2013 ACCF/AHA guideline for the management of heart failure: A report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Circulation 2013;128:e240-327.
- Goldman L, Hashimoto B, Cook EF, et al. Comparative reproducibility and validity of systems for assessing cardiovascular functional class: Advantages of a new specific activity scale. Circulation 1981;64:1227-1234.
- Fox KF, Cowie MR, Wood DA, et al. Coronary artery disease as the cause of incident heart disease in the population. Eur Heart J 2001;22;228-236.
- Manolio TA, Baughman KL, Rodeheffer R, et al. Prevalence and etiology of idiopathic dilated cardiomyopathy (summary of a National Heart, Lung, and Blood Institute workshop). Am J Cardiol 1992;69:1458-1466.
- Adams KF, Jr., Fonarow GC, Emerman CL, et al. Characteristics and outcomes of patients hospitalized for heart failure in the United States: Rationale, design, and preliminary observations from the first 100,000 cases in the Acute Decompensated Heart Failure National Registry (ADHERE). Am Heart J 2005;149:209-216.
- Francis GS, Pierpont GL. Pathophysiology of congestive heart failure secondary to congestive and ischemic cardiomyopathy. Cardiovasc Clin 1988;19:57-74.
- Dec GW, Fuster V. Idiopathic dilated cardiomyopathy. N Engl J Med 1994;331:1564-1575.
- Mann DL, Bristow MR. Mechanisms and models in heart failure: The biomechanical model and beyond. Circulation 2005;111:2837-2849.
- Klug D, Robert V, Swynghedauw B. Role of mechanical and hormonal factors in cardiac remodeling and the biologic limits of myocardial adaptation. Am J Cardiol 1993;71(Suppl):46A-54A.
- Tan LB, Jalil JE, Pick R, et al. Cardiac myocyte necrosis induced by angiotensin II. Circ Res 1991;69:1185-1195.
- Mann DL, Kent RL, Parsons B, Cooper GIV. Adrenergic effects on the biology of the adult mammalian cardiocyte. Circulation 1992;85:790-804.
- Bozkurt B, Kribbs S, Clubb FJ Jr, et al. Pathophysiologically relevant concentrations of tumor necrosis factor-α promote progressive left ventricular dysfunction and remodeling in rats. Circulation 1998;97:1382-1391.
- The SOLVD Investigators. Effect of enalapril on survival in patients with reduced left ventricular ejection fractions and congestive heart failure. N Engl J Med 1991;325:293-302.
- Bristow MR, Gilbert EM, Abraham WT, et al. Carvedilol produces dose-related improvements in left ventricular function and survival in subjects with chronic heart failure. Circulation 1996;94:2807-2816.
- Pitt B, Zannad F, Remme WJ, et al; for the Randomized Aldactone Evaluation Study Investigators. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. N Engl J Med 1999;341:709-717.
- Drazner MH, Rame JE, Stevenson LW, et al. Prognostic importance of elevated jugular venous pressure and a third heart sound in patients with heart failure. N Engl J Med 2001;345:574-581.
- Cheng V, Kazanagra R, Garcia A, et al. A rapid bedside test for B-type peptide predicts treatment outcomes in patients admitted for decompensated heart failure: A pilot study. J Am Coll Cardiol 2001;37:386-391.
- Maisel A. B-type natriuretic peptide levels, a potential novel “white count” for congestive heart failure. J Card Failure 2001;7:183-193.
- Zaphiriou A, Robb S, Murray-Thomas T, et al. The diagnostic accuracy of plasma BNP and NTproBNP in patients referred from primary care with suspected heart failure: Results of the UK natriuretic peptide study. Eur J Heart Fail 2005;7:537-541.
- Kelder JC, Cramer MJ, Van WJ, et al. The diagnostic value of physical examination and additional testing in primary care patients with suspected heart failure. Circulation 2011;124:2865-2873.
- Ledwidge M, Gallagher J, Conlon C, et al. Natriuretic peptide-based screening and collaborative care for heart failure: The STOP-HF randomized trial. JAMA 2013;310:66-74.
- Huelsmann M, Neuhold S, Resl M, et al. PONTIAC (NT-proBNP selected prevention of cardiac events in a population of diabetic patients without a history of cardiac disease): A prospective randomized controlled trial. J Am Coll Cardiol 2013;62:1365-1372.
- Van Kimmenade RR, Pinto YM, Bayes-Genis A, et al. Usefulness of intermediate amino-terminal pro-brain natriuretic peptide concentrations for diagnosis and prognosis of acute heart failure. Am J Cardiol 2006;98:386-390.
- Santaguida PL, Don-Wauchope AC, Ali U, et al. Incremental value of natriuretic peptide measurement in acute decompensated heart failure (ADHF): A systematic review. Heart Fail Rev 2014;19:507-519.
- Bettencourt P, Azevedo A, Pimenta J, et al. N-terminal-pro-brain natriuretic peptide predicts outcome after hospital discharge in heart failure patients. Circulation 2004;110:2168-2174.
- Fonarow GC, Peacock WF, Horwich TB, et al. Usefulness of B-type natriuretic peptide and cardiac troponin levels to predict in-hospital mortality from ADHERE. Am J Cardiol 2008;101:231-237.
- Logeart D, Thabut G, Jourdain P, et al. Predischarge B-type natriuretic peptide assay for identifying patients at high risk of readmission after decompensated heart failure. J Am Coll Cardiol 2004;43:635-641.
- Zairis MN, Tsiaousis GZ, Georgilas AT, et al. Multimarker strategy for the prediction of 31 days cardiac death in patients with acutely decompensated chronic heart failure. Int J Cardiol 2010;141:284-290.
- Lee DS, Stitt A, Austin PC, et al. Prediction of heart failure mortality in emergent care: A cohort study. Ann Intern Med 2012;156:767-775.
- Cohen-Solal A, Logeart D, Huang B, et al. Lowered B-type natriuretic peptide in response to levosimendan or dobutamine treatment is associated with improved survival in patients with severe acutely decompensated heart failure. J Am Coll Cardiol 2009;53:2343-2348.
- Bayes-Genis A, Lopez L, Zapico E, et al. NTProBNP reduction percentage during admission for acutely decompensated heart failure predicts long-term cardiovascular mortality. J Card Fail 2005;11:S3-8.
- Fermann GJ, Lindsell CJ, Storrow AB, et al. Galectin 3 complements BNP in risk stratification in acute heart failure. Biomarkers 2012;17:706-713.
- Lassus J, Gayat E, Mueller C, et al. Incremental value of biomarkers to clinical variables for mortality. J Am Coll Cardiol 2017;70.
- Stevenson LW, Perloff JK. The limited reliability of physical signs for estimating hemodynamics in chronic heart failure. JAMA 1989;261:884-888.
- Syed IS, Glockner JF, Feng D, et al. Role of cardiac magnetic resonance imaging in the detection of cardiac amyloidosis. JACC Cardiovasc Imaging 2010;3:155-164.
- Cooper LT, Baughman KL, Feldman AM, et al. The role of endomyocardial biopsy in the management of cardiovascular disease: A scientific statement from the American Heart Association, the American College of Cardiology, and the European Society of Cardiology. Circulation 2007;116:2216-2233.
- Taegtmeyer H, McNulty P, Young ME. Adaptation and maladaptation of the heart in diabetes. I. General concepts. Circulation 2002;105:1727-1733.
- Lind M, Bounias I, Olsson M, et al. Glycaemic control and incidence of heart failure in 20,985 patients with type 1 diabetes: An observational study. Lancet 2011;378:140-146.
- Mosterd A, D’Agostino RB, Silbershatz H, et al. Trends in the prevalence of hypertension, antihypertensive therapy, and left ventricular hypertrophy from 1950 to 1989. N Engl J Med 1999;340:1221-127.
- Wright JT Jr, Williamson JD, Whelton PK, et al. A randomized trial of intensive versus standard blood pressure control. N Engl J Med 2015;373:2103-2116.
- Yancy CW, Jessup M, Bozkurt B, et al. 2017 ACC/AHA/HFSA Focused Update of the 2013 ACCF/AHA Guideline for the Management of Heart Failure. J Am Coll Cardiol 2017;70:776-803.
- Staessen JA, Wang JG, Thijs L. Cardiovascular prevention and blood pressure reduction: A quantitative overview updated until 1 March 2003. J Hypertens 2003;21:1055-1076.
- Heart Outcomes Prevention Evaluation Study Investigators. Effects of ramipril on cardiovascular and microvascular outcomes in people with diabetes mellitus: Results of the HOPE study and MICRO-HOPE substudy. Lancet 2000;355:253-259. [Erratum: Lancet 2000;356:860.]
- Brenner BM, Cooper ME, de Zeeuw D, et al. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med 2001;345:861-869.
- Mills EJ, Rachlis B, Wu P, et al. Primary prevention of cardiovascular mortality and events with statin treatments: A network meta-analysis involving more than 65,000 patients. J Am Coll Cardiol 2008;52:1769-1781.
- Taylor F, Ward K, Moore TH, et al. Statins for the primary prevention of cardiovascular disease. Cochrane Database Syst Rev 2011;CD004816.
- Gupta D, Georgiopoulou VV, Kalogeropoulos AP, et al. Dietary sodium intake in heart failure. Circulation 2012;126:479-485.
- He FJ, MacGregor GA. Effect of longer-term modest salt reduction on blood pressure. Cochrane Database Syst Rev 2004;CD004937.
- Nagarajan V, Kohan L, Holland E et al. Obesity paradox in heart failure: A heavy matter. ESC Heart Fail 2016;3:227-234.
- Page J, Henry D. Consumption of NSAIDs and the development of congestive heart failure in elderly patients: An underrecognized public health problem. Arch Intern Med 2000;160:777-784.
- Pfeffer MA, Braunwald E, Moye LA, et al. Effect of captopril on mortality and morbidity in patients with left ventricular dysfunction after myocardial infarction. Results of the survival and ventricular enlargement trial. The SAVE Investigators. N Engl J Med 1992;327:669-677.
- Dargie HJ. Effect of carvedilol on outcome after myocardial infarction in patients with left-ventricular dysfunction: The CAPRICORN randomised trial. Lancet 2001;357:1385-1390.
- Vantrimpont P, Rouleau JL, Wun CC, et al. Additive beneficial effects of beta-blockers to angiotensin-converting enzyme inhibitors in the Survival and Ventricular Enlargement (SAVE) Study. SAVE Investigators. J Am Coll Cardiol 1997;29:229-236.
- Scirica BM, Morrow DA, Cannon CP, et al. Intensive statin therapy and the risk of hospitalization for heart failure after an acute coronary syndrome in the PROVE IT-TIMI 22 study. J Am Coll Cardiol 2006;47:2326-2331.
- Afilalo J, Majdan AA, Eisenberg MJ. Intensive statin therapy in acute coronary syndromes and stable coronary heart disease: A comparative meta-analysis of randomised controlled trials. Heart 2007;93:914-921.
- Sacks FM, Pfeffer MA, Moye LA, et al. The effect of pravastatin on coronary events after myocardial infarction in patients with average cholesterol levels. Cholesterol and Recurrent Events Trial investigators. N Engl J Med 1996;335:1001-1009.
- Effect of enalapril on mortality and the development of heart failure in asymptomatic patients with reduced left ventricular ejection fractions. The SOLVD Investigators. N Engl J Med 1992;327:685-691.
- Jong P, Yusuf S, Rousseau MF, et al. Effect of enalapril on 12-year survival and life expectancy in patients with left ventricular systolic dysfunction: A follow-up study. Lancet 2003;361:1843-1848.
- Moss AJ, Zareba W, Hall WJ, et al. Prophylactic implantation of a defibrillator in patients with myocardial infarction and reduced ejection fraction. N Engl J Med 2002;346:877-883.
- Boren SA, Wakefield BJ, Gunlock TL, et al. Heart failure self-management education: A systematic review of the evidence. Int J Evid Based Healthc 2009;7:159-168.
- Koelling TM, Johnson ML, Cody RJ, et al. Discharge education improves clinical outcomes in patients with chronic heart failure. Circulation 2005;111:179-185.
- Gallagher R, Luttik ML, Jaarsma T. Social support and self-care in heart failure. J Cardiovasc Nurs 2011;26:439-445.
- Luttik ML, Jaarsma T, Moser D, et al. The importance and impact of social support on outcomes in patients with heart failure: An overview of the literature. J Cardiovasc Nurs 2005;20:162-169.
- Murberg TA. Long-term effect of social relationships on mortality in patients with congestive heart failure. Int J Psychiatry Med 2004;34:207-217.
- Habbu A, Lakkis NM, Dokainish H. The obesity paradox: Fact or fiction? Am J Cardiol 2006;98:944-948.
- Hambrecht R, Gielen S, Linke A, et al. Effects of exercise training on left ventricular function and peripheral resistance in patients with chronic heart failure: A randomized trial. JAMA 2000;283:3095-3101.
- Piepoli MF, Davos C, Francis DP, et al. Exercise training meta-analysis of trials in patients with chronic heart failure (ExTraMATCH). BMJ 2004;328:189.
- O’Connor CM, Whellan DJ, Lee KL, et al. Efficacy and safety of exercise training in patients with chronic heart failure: HF-ACTION randomized controlled trial. JAMA 2009;301:1439-1450.
- Giannuzzi P, Temporelli PL, Corra U, et al. Attenuation of unfavorable remodeling by exercise training in postinfarction patients with left ventricular dysfunction: Results of the Exercise in Left Ventricular Dysfunction (ELVD) trial. Circulation 1997;96:1790-1797.
- McEvoy RD, Antic NA, Heeley E, et al. CPAP for prevention of cardiovascular events in obstructive sleep apnea. N Engl J Med 2016;375:919-931.
- Metra M, Ponikowski P, Dickstein K, et al. Advanced chronic heart failure: A position statement from the Study Group on Advanced Heart Failure of the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail 2007;9:684-694.
- SOLVD Investigators; Yusuf S, Pitt B, et al. Effect of enalapril on survival in patients with reduced left ventricular ejection fractions and congestive heart failure. N Engl J Med 1991;325:293-302.
- Packer M, Poole-Wilson PA, Armstrong PW, et al. Comparative effects of low and high doses of the angiotensin-converting enzyme inhibitor, lisinopril, on morbidity and mortality in chronic heart failure. ATLAS Study Group. Circulation 1999;100:2312-2318.
- Effect of ramipril on mortality and morbidity of survivors of acute myocardial infarction with clinical evidence of heart failure. The Acute Infarction Ramipril Efficacy (AIRE) Study Investigators. Lancet 1993;342:821-828.
- Gullestad L, Aukrust P, Ueland T, et al. Effect of high- versus low-dose angiotensin converting enzyme inhibition on cytokine levels in chronic heart failure. J Am Coll Cardiol 1999;34:2061-2067.
- Tang WH, Vagelos RH, Yee YG, et al. Neurohormonal and clinical responses to high- versus low-dose enalapril therapy in chronic heart failure. J Am Coll Cardiol 2002;39:70-78. [Erratum: J Am Coll Cardiol 2002;39:746.]
- Cohn J N, Tognoni G; Valsartan heart failure trial investigators. A randomized trial of the angiotensin-receptor blocker valsartan in chronic heart failure. N Engl J Med 2001;345:1667-1675.
- McMurray JJ, Pteffer MA, et al. Which inhibitor of the renin angiotensin system should be used in chronic heart failure and left ventricular systolic dysfunction: Results of the CHARM low left ventricular ejection fraction trials. Circulation 2004;110:2618-2626.
- Pitt B, Poole-Wilson PA, Segal R, et al. Effects of losartan compared with captopril on mortality in patients with symptomatic heart failure: Randomised trial — the Losartan Heart Failure Survival Study ELITE II. Lancet 2000;355:1582-1587.
- McMurray JJV, Packer M, Desai AS, et al. Angiotensin-neprilysin inhibition versus enalapril in heart failure. N Engl J Med 2014;371:993-1004.
- Entresto package insert. Hanover, NJ: Novartis Pharmaceuticals Corporation; 2015.
- Bristow M. Beta-adrenergic receptor blockade in chronic heart failure. Circulation 2000;101:558-569.
- Packer M, Bristow MR, Cohn JN, et al. The effect of carvedilol on morbidity and mortality in patients with chronic heart failure. US Carvedilol Heart Failure Study Group. N Engl J Med 1996;334:1349-1355.
- Groenning B, Nilsson J, Sondergaard L, et al. Anti-remodeling effects on the left ventricle during beta-blockade with metoprolol in the treatment of chronic heart failure. J Am Coll Cardiol 2000,36:2072-2080.
- Hall S, Cigarroa C, Marcoux L, et al. Time course of improvement in left ventricular function, mass and geometry in patients with congestive heart failure treated with beta-adrenergic blockade. J Am Coll Cardiol 1995;25;1154-1161.
- Krum H, Roecker EB, Mohacsi P, et al. Effects of initiating carvedilol in patients with severe chronic heart failure: Results from the COPERNICUS study. JAMA 2003;289:712-718.
- Gattis WA, O’Connor CM, Gallup DS, et al. Predischarge initiation of carvedilol in patients hospitalized for decompensated heart failure: Results of the Initiation Management Predischarge: Process for Assessment of Carvedilol Therapy in Heart Failure (IMPACT-HF) trial. J Am Coll Cardiol 2004;43:1534-1541.
- Brilla CG, Matsubara LS, Weber KT. Anti-aldosterone treatment and the prevention of myocardial fibrosis in primary and secondary hyperaldosteronism. J Mol Cell Cardiol 1993;25:563-575.
- Weber KT, Brilla CG. Pathological hypertrophy and cardiac interstitium: Fibrosis and renin-angiotensin-aldosterone system. Circulation 1991;83:1849-1865.
- Pitt B, Remme W, Zannad F, et al; Eplerenone Post–Acute Myocardial Infarction Heart Failure Efficacy and Survival Study Investigators. Eplerenone, a selective aldosterone blocker, in patients with left ventricular dysfunction after myocardial infarction. N Engl J Med 2003;348:1309-1321.
- Zannad F, McMurray JJV, Krum H, et al. Eplerenone in patients with systolic heart failure and mild symptoms. N Engl J Med 2011;364:11-21.
- Taylor AL, Ziesche S, Yancy C, et al. Combination of isosorbide dinitrate and hydralazine in blacks with heart failure. N Engl J Med 2004;351:2049-2057.
- Cohn JN, Archibald DG, Ziesche S, et al. Effect of vasodilator therapy on mortality in chronic congestive heart failure. Results of a Veterans Administration Cooperative Study. N Engl J Med 1986;314:1547-1552.
- Swedberg K, Komajda M, Böhm M, et al. Ivabradine and outcomes in chronic heart failure (SHIFT): A randomised placebo-controlled study. Lancet 2010;376:875-885.
- Wilson JR, Reichek N, Dunkman WB, et al. Effect of diuresis on the performance of the failing left ventricle in man. Am J Med 1981;70:234-239.
- Parker JO. The effects of oral ibopamine in patients with mild heart failure—a double blind placebo controlled comparison to furosemide. The Ibopamine Study Group. Int J Cardiol 1993;40:221-227.
- Faris R, Flather M, Purcell H, et al. Current evidence supporting the role of diuretics in heart failure: A meta analysis of randomized control trials. Int J Cardiol 2002;82:149-158.
- Felker GM, Lee KL, Bull DA, et al. Diuretic strategies in patients with acute decompensated heart failure. N Engl J Med 2011;364:797-805.
- Herchuelz A, Derenne F, Deger F, et al. Interaction between nonsteroidal anti-inflammatory drugs and loop diuretics: Modulation by sodium balance. J Pharmacol Exp Ther 1989;248:1175-1181.
- Risler T, Schwab A, Kramer B, et al. Comparative pharmacokinetics and pharmacodynamics of loop diuretics in renal failure. Cardiology 1994;84(Suppl 2):155-161.
- Brater DC. Diuretic therapy. N Engl J Med 1998;339:387-395.
- Epstein M, Lepp B, Hoffman D. Potentiation of furosemide by metolazone in refractory edema. Curr Ther Res 1977;656-667.
- Ellison DH. The physiologic basis of diuretic synergism: Its role in treating diuretic resistance. Ann Intern Med 1991;114:886-894.
- Adams KF, Gheorghiade M, Uretsky BF, et al. Clinical benefits of low serum digoxin concentrations in heart failure. J Am Coll Cardiol 2002;39:946-953.
- Comparative effects of therapy with captopril and digoxin in patients with mild to moderate heart failure. The Captopril-Digoxin Multicenter Research Group. JAMA 1988;259:539-544.
- Lee DC, Johnson RA, Bingham JB, et al. Heart failure in outpatients: A randomized trial of digoxin versus placebo. N Engl J Med 1982;306:699-705.
- Guyatt GH, Sullivan MJ, Fallen EL, et al. A controlled trial of digoxin in congestive heart failure. Am J Cardiol 1988;61:371-375.
- Packer M, Gheorghiade M, Young JB, et al. Withdrawal of digoxin from patients with chronic heart failure treated with angiotensin-converting-enzyme inhibitors. RADIANCE Study. N Engl J Med 1993;329:1-7.
- The Digitalis Investigation Group. The effect of digoxin on mortality and morbidity in patients with heart failure. N Engl J Med 1997;336:525-533.
- Hager WD, Fenster P, Mayersohn M, et al. Digoxin-quinidine interaction pharmacokinetic evaluation. N Engl J Med 1979;300:1238-1241.
- Bizjak ED, Mauro VF. Digoxin-macrolide drug interaction. Ann Pharmacother 1997;31:1077-1079.
- Cohn JN, Goldstein SO, Greenberg BH, et al. A dose-dependent increase in mortality with vesnarinone among patients with severe heart failure. Vesnarinone Trial Investigators. N Engl J Med 1998;339:1810-1816.
- Hampton JR, van Veldhuisen DJ, Kleber FX, et al. Randomised study of effect of ibopamine on survival in patients with advanced severe heart failure. Second Prospective Randomised Study of Ibopamine on Mortality and Efficacy (PRIME II) Investigators. Lancet 1997;349:971-977.
- Packer M, Carver JR, Rodeheffer RJ, et al. Effect of oral milrinone on mortality in severe chronic heart failure. The PROMISE Study Research Group. N Engl J Med 1991;325:1468-1475.
- Elkayam U, Akhter MW, Singh H, et al. Comparison of effects on left ventricular filling pressure of intravenous nesiritide and high-dose nitroglycerin in patients with decompensated heart failure. Am J Cardiol 2004;93:237-240.
- Cotter G, Metzkor E, Kaluski E, et al. Randomised trial of high-dose isosorbide dinitrate plus low-dose furosemide versus high-dose furosemide plus low-dose isosorbide dinitrate in severe pulmonary oedema. Lancet 1998;351:389-393.
- Fung HL, Bauer JA. Mechanisms of nitrate tolerance. Cardiovasc Drugs Ther 1994;8:489-499.
- Elkayam U, Kulick D, McIntosh N, et al. Incidence of early tolerance to hemodynamic effects of continuous infusion of nitroglycerin in patients with coronary artery disease and heart failure. Circulation 1987;76:577-584.
- Dupuis J, Lalonde G, Lemieux R, et al. Tolerance to intravenous nitroglycerin in patients with congestive heart failure: Role of increased intravascular volume, neurohumoral activation and lack of prevention with N-acetylcysteine. J Am Coll Cardiol 1990;16:923-931.
- Mullens W, Abrahams Z, Francis GS, et al. Sodium nitroprusside for advanced low-output heart failure. J Am Coll Cardiol 2008;52:200-207.
- O’Connor CM, Starling RC, Hernandez AF, et al. Effect of nesiritide in patients with acute decompensated heart failure. N Engl J Med 2011;365:32-43.
- Schrier RW, Gross P, Gheorghiade M, et al. Tolvaptan, a selective oral vasopressin V2-receptor antagonist, for hyponatremia. N Engl J Med 2006;355:2099-2112.
- Ghali JK, Koren MJ, Taylor JR, et al. Efficacy and safety of oral conivaptan: A V1A/V2 vasopressin receptor antagonist, assessed in a randomized, placebo-controlled trial in patients with euvolemic or hypervolemic hyponatremia. J Clin Endocrinol Metab 2006;91:2145-2152.
- Gheorghiade M, Abraham WT, Albert NM,et al. Relationship between admission serum sodium concentration and clinical outcomes in patients hospitalized for heart failure: An analysis from the OPTIMIZE-HF registry. Eur Heart J 2007;28:980-988.
- Gheorghiade M, Konstam MA, Burnett JC, Jr., et al. Short-term clinical effects of tolvaptan, an oral vasopressin antagonist, in patients hospitalized for heart failure: The EVEREST Clinical Status Trials. JAMA 2007;297:1332-1343.
- Konstam MA, Gheorghiade M, Burnett JC, Jr., et al. Effects of oral tolvaptan in patients hospitalized for worsening heart failure: The EVEREST Outcome Trial. JAMA 2007;297:1319-1331.
- Bardy GH, Lee KL, Mark DB, et al. Amiodarone or an implantable cardioverter-defibrillator for congestive heart failure. N Engl J Med 2005;352:225-237.
- Steinbeck G, Andresen D, Seidl K, et al. Defibrillator implantation early after myocardial infarction. N Engl J Med 2009;361:1427-1436.
- Cleland JG, Daubert JC, Erdmann E, et al. The effect of cardiac resynchronization on morbidity and mortality in heart failure. N Engl J Med 2005;352:1539-1549.
- Saxon LA, Ellenbogen KA. Resynchronization therapy for the treatment of heart failure. Circulation 2003;108:1044-1048.
- Stellbrink C, Breithardt O-A, Franke A, et al. Impact of cardiac resynchronization therapy using hemodynamically optimized pacing on left ventricular remodeling in patients with congestive heart failure and ventricular conduction disturbances. J Am Coll Cardiol 2001;38:1957-1965.
- Kerwin WF, Botvinick EH, O’Connell JW, et al. Ventricular contraction abnormalities in dilated cardiomyopathy: Effect of biventricular pacing to correct interventricular dyssynchrony. J Am Coll Cardiol 2000;35:1221-1227.
- Young JB, Abraham WT, Smith AL, et al. Combined cardiac resynchronization and implantable cardioversion defibrillation in advanced chronic heart failure: The MIRACLE ICD Trial. JAMA 2003;289:2685-2694.
- Stavrakis S, Lazzara R, Thadani U. The benefit of cardiac resynchronization therapy and QRS duration: A meta-analysis. J Cardiovasc Electrophysiol 2012;23:163-168.
- Bilchick KC, Kamath S, DiMarco JP, et al. Bundle-branch block morphology and other predictors of outcome after cardiac resynchronization therapy in Medicare patients. Circulation 2010;122:2022-2030.
- Tang AS, Wells GA, Talajic M, et al. Cardiac-resynchronization therapy for mild-to-moderate heart failure. N Engl J Med 2010;363:2385-2395.
- Linde C, Abraham WT, Gold MR, et al. Randomized trial of cardiac resynchronization in mildly symptomatic heart failure patients and in asymptomatic patients with left ventricular dysfunction and previous heart failure symptoms. J Am Coll Cardiol 2008;52:1834-1843.
- Hillis LD, Smith PK, Anderson JL, et al. 2011 ACCF/AHA Guideline for Coronary Artery Bypass Graft Surgery. A report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Circulation 2011;124:e652-735.
- Levine GN, Bates ER, Blankenship JC, et al. 2011 ACCF/AHA/SCAI Guideline for Percutaneous Coronary Intervention. A report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines and the Society for Cardiovascular Angiography and Interventions. Circulation 2011;124:e574-651.
- Patel MR, Dehmer GJ, Hirshfeld JW, et al. ACCF/SCAI/STS/AATS/AHA/ASNC 2009 Appropriateness Criteria for Coronary Revascularization: A report by the American College of Cardiology Foundation Appropriateness Criteria Task Force, Society for Cardiovascular Angiography and Interventions, Society of Thoracic Surgeons, American Association for Thoracic Surgery, American Heart Association, and the American Society of Nuclear Cardiology. Circulation 2009;119:1330-1352.
- Velazquez EJ, Lee KL, Deja MA, et al. Coronary-artery bypass surgery in patients with left ventricular dysfunction. N Engl J Med 2011;364:1607-1616.
- Leon MB, Smith CR, Mack M, et al. Transcatheter aortic-valve implantation for aortic stenosis in patients who cannot undergo surgery. N Engl J Med 2010;363:1597-1607.
- Gersh BJ, Maron BJ, Bonow RO, et al. 2011 ACCF/AHA Guideline for the Diagnosis and Treatment of Hypertrophic Cardiomyopathy A Report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines Developed in Collaboration With the American Association for Thoracic Surgery, American Society of Echocardiography, American Society of Nuclear Cardiology, Heart Failure Society of America, Heart Rhythm Society, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. Circulation 2011;124:e783-831.
- Raman JS, Hata M, Storer M, et al. The mid-term results of ventricular containment (ACORN WRAP) for end-stage ischemic cardiomyopathy. Ann Thorac Cardiovasc Surg 2001;7:278-281.
- Acker MA, Bolling S, Shemin R, et al. Mitral valve surgery in heart failure: Insights from the Acorn Clinical Trial. J Thorac Cardiovasc Surg 2006;132:568-577.
- Bishay ES, McCarthy PM, Cosgrove DM, et al. Mitral valve surgery in patients with severe left ventricular dysfunction. Eur J Cardiothorac Surg 2000;17:213-221.
- Braun, et al. Long-term follow-up confirms left ventricular reverse remodeling following restrictive mitral annuloplasty and CABG in ischemic MR. Presented at the AATS 87th Annual Meeting, San Diego, Calif. May 2007.
- DiDonato M, Frigiola A, Menicanti L, et al. Moderate ischemic MR and CABG: Effect of mitral repair on clinical outcome. J Heart Valve Dis 2003;12:272–279.
- Geidel S, Schneider C, Lass M, et al. Changes of myocardial function after combined coronary revascularization and mitral valve downsizing in patients with ischemic mitral regurgitation and advanced cardiomyopathy. Thorac Cardiov Surg 2007;55:106.
- Braunwald E, Antman EM, Beasley JW, et al. ACC/AHA Guidelines for the Management of Patients With Unstable Angina and Non–ST-Segment Elevation Myocardial Infarction: Executive Summary and Recommendations. A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee on the Management of Patients With Unstable Angina). Circulation 2000;102:1193-1209.
- Sanborn TA, Sleeper LA, Bates ER, et al. Impact of thrombolysis, intra-aortic balloon pump counterpulsation, and their combination in cardiogenic shock complicating acute myocardial infarction: A report from the SHOCK Trial Registry. Should we emergently revascularize occluded coronaries for cardiogenic shock? J Am Coll Cardiol 2000;36(3 Suppl A):1123-1129.
- Thiele H, Zeymer U, Neumann FJ, et al. Intraaortic balloon support for myocardial infarction with cardiogenic shock. N Engl J Med 2012;367:1287-1296.
- Burkhoff D, Cohen H, Brunckhorst C, et al. A randomized multicenter clinical study to evaluate the safety and efficacy of the TandemHeart percutaneous ventricular assist device versus conventional therapy with intraaortic balloon pumping for treatment of cardiogenic shock. Am Heart J 2006;152:469-468.
- Slaughter MS, Rogers JG, Milano CA, et al. Advanced heart failure treated with continuous-flow left ventricular assist device. N Engl J Med 2009;361:2241-2251.
- Rogers JG, Butler J, Lansman SL, et al. Chronic mechanical circulatory support for inotrope-dependent heart failure patients who are not transplant candidates: Results of the INTrEPID Trial. J Am Coll Cardiol 2007;50:741-747.
- Gaasch WH, Zile MR. Left ventricular diastolic dysfunction and diastolic heart failure. Annu Rev Med 2004;55:373-394.
- Redfield MM. Heart failure with preserved ejection fraction. N Engl J Med 2016;375:1868-1877.
- Chan MM, Lam CS. How do patients with heart failure with preserved ejection fraction die? Eur J Heart Fail 2013;15:604-613.
- Borlaug BA. Mechanisms of exercise intolerance in heart failure with preserved ejection fraction. Circ J 2014;78:20-32.
- Borlaug BA, Nishimura RA, Sorajja P, et al. Exercise hemodynamics enhance diagnosis of early heart failure with preserved ejection fraction. Circ Heart Fail 2010;3:588-595.
- Zile MR, Baicu CF, Gaasch WH. Diastolic heart failure — abnormalities in active relaxation and passive stiffness of the left ventricle. N Engl J Med 2004;350:1953-1959.
- Borlaug BA, Lam CS, Roger VL, et al. Contractility and ventricular systolic stiffening in hypertensive heart disease: Insights into the pathogenesis of heart failure with preserved ejection fraction. J Am Coll Cardiol 2009;54:410-418.
- Solomon GC. Clinical practice: Heart failure with preserved ejection fraction. N Engl J Med 2016;375:1868-1877.
- Dhakal BP, Malhotra R, Murphy RM, et al. Mechanisms of exercise intolerance in heart failure with preserved ejection fraction: The role of abnormal peripheral oxygen extraction. Circ Heart Fail 2015;8:286-294.
- Haykowsky MJ, Tomczak CR, Scott JM, et al. Determinants of exercise intolerance in patients with heart failure and reduced or preserved ejection fraction. J Appl Physiol 2015;119:739-744.
- Mohammed SF, Hussain S, Mirzoyev SA, et al. Coronary microvascular rarefaction and myocardial fibrosis in heart failure with preserved ejection fraction. Circulation 2015;131:550-559.
- Mohammed SF, Majure DT, Redfield MM. Zooming in on the microvasculature in heart failure with preserved ejection fraction. Circ Heart Fail 2016;9:e003272.
- Anjan VY, Loftus TM, Burke MA, et al. Prevalence, clinical phenotype, and outcomes associated with normal B-type natriuretic peptide levels in heart failure with preserved ejection fraction. Am J Cardiol 2012;110:870-876.
- Yusuf S, Pfeffer MA, Swedberg K, et al. Effects of candesartan in patients with chronic heart failure and preserved left ventricular ejection fraction: The CHARM-Preserved Trial. Lancet 2003;362:777-781.
- Pfeffer MA, Claggett B, Assmann SF, et al. Regional variation in patients and outcomes in the Treatment of Preserved Cardiac Function Heart Failure With an Aldosterone Antagonist (TOPCAT) trial. Circulation 2015;131:34-42.
- Pitt B, Pfeffer MA, Assmann SF, et al. Spironolactone for heart failure with preserved ejection fraction. N Engl J Med 2014;370:1383-1392.
- Ohman EM. Chronic stable angina. N Engl J Med 2016;374:1167-1176.
- Kitzman DW, Brubaker P, Morgan T, et al. Effect of caloric restriction or aerobic exercise training on peak oxygen consumption and quality of life in obese older patients with heart failure with preserved ejection fraction: A randomized clinical trial. JAMA 2016;315:36-46.
- Pandey A, Parashar A, Kumbhani DJ, et al. Exercise training in patients with heart failure and preserved ejection fraction: Meta-analysis of randomized control trials. Circ Heart Fail 2015;8:33-40.
The management of heart failure falls primarily on the primary care physician. Because of the Medicare financial penalty on hospitals for readmissions, primary care physicians need to be cognizant of the updated treatment options and work with colleagues across multiple disciplines to prevent unnecessary hospitalizations and improve outcomes.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
