All That Is Round Is Not Fungus: A Differential Diagnosis of Annular Lesions
July 1, 2018
Reprints
AUTHORS
Karl Kellawan, MD, Forefront Dermatology, Centerville, OH
Wyatt Andrasik, BS, MD Candidate, Wright State University Boonshoft School of Medicine, Dayton, OH
PEER REVIEWER
Richard A. Miller, DO, Program Director, Dermatology Residency Program, Nova Southeastern University College of Osteopathic Medicine/Largo Medical Center, Largo, FL
Executive Summary
Annular skin lesions commonly plague many primary care patients, but not all that is round is fungus. This article highlights the differential diagnosis of conditions that can mimic tinea.
- Annular lesions are round with central clearing, whereas nummular lesions are coin-shaped with discrete margins without central clearing.
- Tinea infections result from close contact with infected people, animals, and soils and are promoted by humidity, warmth, and poor hygiene.
- Infections are more frequent in young adults and in men.
- Other etiologies of annular lesions include granuloma annulare, pityriasis rosea, erythema annulare centrifugum, lichen planus, and psoriasis.
Primary care physicians often are sought to diagnose a wide variety of dermatologic conditions. Among the common presentations seen on a day-to-day basis are annular lesions. The term annular is derived from the Latin word “annulus,” meaning ring shaped.1 These lesions have a characteristic appearance, described as round to oval-shaped patches or plaques with central clearing. In contrast, the term nummular, derived from the Latin word “nummulus,” meaning coin shaped, is used to describe sharply marginated, round to disc-shaped lesions without central clearing. While these terms describe similar lesions, they are not interchangeable. Common etiologies of nummular lesions include parapsoriasis and lichen aureus, among others. In adults, the most common etiology of annular lesions is tinea, a superficial fungal infection of keratinized tissue. While tinea may be readily identifiable, a wide variety of clinical entities mimic these infections, leading to confusion and, often, misdiagnosis.
When diagnosing annular lesions, a broad differential must be considered. Subtleties among the various etiologies can be used to exclude potential diagnoses, but perhaps may be too numerous or nuanced to recall. A discussion emphasizing key morphological characteristics may improve accuracy of diagnosis, limit unnecessary treatment, and lead to faster resolution of disease. This review will provide guidance for the primary care provider in the recognition, diagnosis, and management of dermatological entities that present with annular lesions. (See Table 1.) This review may be especially helpful in the case of suspected tinea that is refractory to treatment.
Table 1. Summary of Diseases, Clinical Features, and Treatment |
||
Diagnosis |
Clinical Features |
Treatment |
Tinea corporis/cruris |
Asymmetric, well-demarcated, erythematous lesion with or without central clearing. Often associated with a scaly, palpable edge. KOH microscopy positive. |
Topical or systemic antifungal agents |
Granuloma annulare |
Non-scaly, erythematous to violaceus colored papules or plaques with a thin, smooth border. Favors the dorsal aspects of the extremities. |
Topical, intralesional, or oral corticosteroids |
Pityriasis rosea |
Can begin with a herald patch, followed by a diffuse eruption involving the trunk and proximal extremities. Lesions are oval-shaped, scaly, skin to salmon-colored papules or plaques. Described as a “Christmas-tree distribution” when on the back. |
Oral corticosteroids if significant pruritus is present |
Erythema annulare centrifugum |
Erythematous annular or polycyclic plaques with a trailing scale inside an erythematous border. Favors the trunk and proximal extremities. |
Topical corticosteroid creams |
Erythema chronicum migrans |
Large, evolving erythematous plaque without scale. May have a characteristic “bull’s-eye” appearance. |
Doxycycline (adults)/amoxicillin (pregnant women and children) |
Urticaria |
Well-circumscribed, non-scaly edematous papules or plaques with blanched centers, surrounded by a red flare. |
First- or second-generation antihistamines |
Lichen planus |
Small, violaceous, polygonal-shaped papules and plaques with a diffuse network of white streaking. |
Topical or intralesional corticosteroids; oral corticosteroids if severe |
Cutaneous larva migrans |
Erythematous, serpiginous tracts associated with intense pruritus. |
Ivermectin (adults)/albendazole (children) |
Porokeratosis of Mibelli |
Annular, skin- to brown-colored plaque with a raised, ridge-like border. |
Topical 5-fluorouracil with or without topical retinoids; cryosurgery |
Psoriasis |
Well-circumscribed, erythematous dry plaques with silvery scale. |
Topical corticosteroids |
Nummular eczema |
Well-demarcated, erythematous or hyperpigmented scaly or crusty coin-shaped plaques. |
Topical corticosteroids |
Subacute cutaneous lupus erythematosus |
Red to pink plaques with raised borders and central clearing. Occurs following sun exposure. Favors the face, upper trunk, and backs of arms. |
Sun protection |
Sarcoidosis |
Multiple, firm, red to purple to brown colored papules, nodules, or plaques. Favor the face, neck, and areas of trauma. Can have annular lesions. |
Intralesional triamcinolone. Oral corticosteroids for systemic disease |
Seborrheic dermatitis |
Sharply demarcated red to pink to brown patches or thin plaques with greasy scale. |
Topical ketoconazole cream or shampoo |
Erythema multiforme |
Edematous plaques with three distinct zones. |
Valacyclovir daily |
Hansen’s disease |
Tuberculoid — erythematous plaques with a raised, sharply defined borders and atrophic center. Characteristically anesthetic. Lepromatous — numerous, poorly defined, hypopigmented macules that are symmetrically distributed. |
Dapsone and rifampin Dapsone, rifampin, and clofazimine |
The Superficial Mycoses
Superficial mycotic infections are those limited to keratinized environments such as the skin, hair, and nails. Notoriously, these infections are caused by dermatophytes, a collective term that describes three genera — Microsporum, Trichophyton, and Epidermophyton. In naming dermatophyte infections, the term “tinea” is used, followed by the Latin name for the anatomical area infected.2 Subtypes include tinea corporis (body), cruris (groin), capitis (scalp), faciei (face), barbae (beard), unguium (nail), and manuum (hand).
Tinea infections are caused by close contact with infected persons, animals, or, occasionally, soil, and are spread to multiple body areas via autoinoculation when an infected region is scratched.1 Most infections occur in young adults and affect men more often than women.3 Warm environments, poor hygiene, contact sports, prolonged use of potent topical corticosteroids, and diseases that cause defects in the skin barrier facilitate these infections.4 Additionally, practices such as sharing towels, clothing, and toiletries, commonly seen in young athletes, are other important risk factors.
Tinea can be diagnosed accurately on visual exam. When available, confirming the diagnosis by observing branching hyphae under the microscope using a potassium hydroxide (KOH) preparation is recommended. Tinea corporis and tinea cruris, the most common subtypes of dermatophytosis, are discussed briefly in this report. A more detailed review outlining tinea subtypes, clinical variants, diagnosis, and treatment is covered in the August 2017 issue of Primary Care Reports.
Tinea Corporis
Tinea corporis is a superficial dermatophyte infection of the body, not involving the scalp, face, hands, feet, or groin.4 Classically, tinea corporis is characterized by an asymmetric, well-demarcated, erythematous, dry, and scaly patch with a raised, red advancing border.2,4 Centrally, lesions clear, forming an annular pattern.4 Individual eruptions vary in size, enlarge over time, and often are associated with mild to moderate pruritus, although smaller lesions may be asymptomatic.4
Tinea Cruris
Tinea cruris, or “jock itch,” is dermatophytosis of the groin. Lesions are similar to those of tinea corporis, characterized by an asymmetric, well-defined, mildly erythematous patch with associated scale. The advancing border is raised and may contain vesicles, pustules, or papules.4 Typically, infection begins unilaterally and extends to the medial thighs, perineum, and anus, sparing the scrotum.5 Tinea cruris is commonly found in conjunction with tinea pedis and/or tinea unguium, as the fungus is spread from the infected foot to the groin when clothing is pulled to the waist.2
Treatment of Tinea
For localized tinea corporis, cruris, pedis, and faciei, topical antifungals, such as imidazoles (clotrimazole, miconazole, ketoconazole, econazole, oxiconazole, sulconazole) or allylamines (naftifine, terbinafine), should be used once to twice daily for two to six weeks, including two weeks following clearance.2,6 Tinea capitis and tinea barbae should be treated with the systemic antifungal agent terbinafine. Patients with tinea manuum, tinea unguium, and extensive or refractory disease require systemic antifungal agents such as terbinafine, fluconazole, itraconazole, or griseofulvin.4,5 Nystatin, commonly used to treat Candida infections, should not be used to treat tinea.3
Other Etiologies of Annular Lesions
Granuloma Annulare
Granuloma annulare (GA) is a relatively common, self-limiting disorder of the dermis that affects women twice as often as men.7 The eruption has a predilection for young adults and children, with most cases presenting before the third decade of life.1,7 Although the etiology remains unknown, GA is controversially associated with diabetes mellitus and hyperlipidemia.8 Several clinical variants of GA exist; however, localized disease primarily affects the dorsal surfaces of the fingers, hands, elbows, feet, and ankles.1,7 Lesions are characterized by asymptomatic, erythematous to violaceus colored papules or plaques with a thin, smooth border.9 Lesions develop slowly, spread peripherally, and involute centrally, conferring an annular appearance.9 An isolated (< 5 cm) lesion on the hand or arm represents most cases.7
Granuloma annulare can be diagnosed clinically by its unique distribution and morphology. Asymptomatic lesions with smooth, non-scaly contours are differentiated easily from the pruritic, scaly, and rough lesions of tinea. Biopsy with histopathologic correlation can be used to confirm or establish the diagnosis when not clinically obvious.7
Reassurance and observation is appropriate, as GA is benign and self-limiting. If left alone, one half of cases will resolve within two years.8 If treatment is preferred, high-potency topical corticosteroids or intralesional triamcinolone can be used.8,9 Although most cases heal without residual skin findings, recurrence is common regardless of treatment.9
Pityriasis Rosea
Pityriasis rosea is a common eruption primarily seen in adolescents and young adults.10 The etiology remains unknown, but given its seasonal variation, occurring mostly in the spring and fall, a viral cause is suspected.11 The eruption is aptly named — pityriasis, meaning scaly, and rosea, meaning pink. About half of cases will begin with a “herald patch,” a 2 to 4 cm lesion that occurs mainly on the trunk or neck.11 The herald patch is a pink to brown oval-shaped patch or plaque with slightly raised margins and collarette scale at the periphery.10,12 One to two weeks following the herald patch, numerous, fine scaling papules and patches arise on the trunk and proximal extremities, sparing the face, palms, and soles.10 Subsequent lesions are smaller, oval-shaped, and salmon-colored, although they may be hyperpigmented in darker skinned individuals.10,12 (See Figure 1.) Multiple lesions may coalesce, forming a diffuse rash that, when present on the back, exhibits a “Christmas tree” pattern along Langer’s lines, the natural folds of the body. Lesions are mildly pruritic and oriented along cleavage lines.12 When stretched, scale hangs, resulting in the pathognomonic “hanging curtain sign.” A small subset of patients will experience a prodrome of headache, fever, and generalized malaise days to weeks prior to the initial outbreak.13
Figure 1. Multiple, Oval-shaped Lesions of Pityriasis Rosea |
 |
Source: Karl Kellawan, MD |
When the rash is localized to the trunk, axillae, or groin, it commonly is mistaken for tinea. Tinea rarely is as widespread, and its lesions generally exude more significant central clearing.12 As always, a negative KOH preparation can be used to exclude tinea.
The classic presentation often is alarming to patients, prompting medical evaluation. However, treatment generally is not required, as the eruption spontaneously remits in three to eight weeks.12 If patients experience intense pruritus, second-generation antihistamines, topical corticosteroids, a short course of systemic corticosteroids, or narrowband ultraviolet B (UVB) phototherapy can provide symptomatic relief.10
Erythema Annulare Centrifugum
Erythema annulare centrifugum (EAC) belongs to a group of disorders characterized by raised, erythematous lesions that form annular, polycyclic, or arcuate arrangements.14 Although the etiology is unclear and most patients do not have an identifiable trigger, an association with leukemias and lymphomas has been reported.15 The eruption is seen in both men and women and peaks in the fifth decade of life.16 Lesions begin as firm, pink papules that expand slowly over weeks, clear centrally, and progress to annular, erythematous, minimally elevated lesions that remain relatively asymptomatic beyond mild pruritis.14,16 Just inside the erythematous border is a trailing white scale that represents desquamation at the inner margin, characteristic of EAC. (See Figure 2.) Lesions have a predilection for the trunk and proximal extremities, sparing the hands, feet, face, and mucosa.14 When pressed, lesions should blanch.
Figure 2. Erythema Annulare Centrifugum With Faint Trailing White Scale Inside the Erythematous Border |
 |
Source: Karl Kellawan, MD |
Diagnosis relies on clinical presentation. Centrally, lesions lack the associated crusts or vesicles commonly seen with tinea. Annular psoriasis may present similarly, but these lesions have diffuse, thick scaling throughout as opposed to the fine trailing scale around the margin in EAC.
EAC often has a waxing and waning course that persists for about nine months.1,14 Most cases will subside spontaneously without treatment, leaving no residual scar. While rare, a basic cancer workup is warranted to rule out a paraneoplastic cause. If pruritus is present or treatment is desired, corticosteroids often are effective, but topical applications are preferred, as recurrence upon discontinuation of systemic agents is common.14,16
Erythema Chronicum Migrans
Erythema chronicum migrans (ECM) is the cutaneous manifestation of Lyme disease. In the United States, Borrelia spirochetes are transmitted to humans by an infected Ixodes tick. For most patients, ECM at the site of the tick bite is the first sign of infection.17 However, only about half of patient’s presenting with ECM will recall a recent bite or exposure to ticks. As with most tick bites, an erythematous, small, round papule will appear at the site of the bite, and within days, the surrounding erythema will migrate peripherally, forming a large, erythematous plaque.17 The expanding border will be warm and slightly raised without an associated scale, while centrally, a ring may clear, resulting in an annular or pathognomonic bull’s-eye appearance.18,19 (See Figure 3.) Primary lesions can reach 15 cm in diameter and commonly are found on the trunk, axilla, groin, and popliteal fossa.17,18 Days to weeks following the primary eruption, a subset of patients will develop secondary lesions that are smaller and less pronounced than the primary lesion. Generally, these secondary lesions will not have the targetoid appearance of the primary lesion and will spare the palms and soles.19 The cutaneous manifestations are self-limiting and, if untreated, will fade within six weeks.17
Figure 3. Classic Bull’s-eye Rash of Erythema Chronicum Migrans on the Lower Extremity |
 |
Source: Getty Images |
While the skin manifestations are mostly asymptomatic, accompanying symptoms of Lyme disease are common. Early in the disease course, a mild flu-like illness consisting of general malaise, fever, headache, nausea, vomiting, arthralgias, myalgias, and photophobia can occur.19 If untreated, the disease can progress over the course of weeks to months, resulting in significant complications such as chronic arthritis, facial nerve palsy, and varying degrees of atrioventricular block.19
Early recognition and accurate diagnosis can prevent progression of disease and subsequent lifelong rheumatologic, neurologic, and cardiac complications. In a case of suspected Lyme disease, cutaneous findings are the most sensitive sign of early infection.18 Tick bites not associated with infection will begin to regress within a few days, while ECM will persist and progress. A detailed travel history, recent bites, or outdoor exposures should raise clinical suspicion. Lab findings may include an elevated erythrocyte sedimentation rate, elevated liver function tests, and mild anemia.18 Serologic evidence of infection is most sensitive between weeks three and six and commonly involves detection of anti-Borrelia antibodies using enzyme-linked immunosorbent assay (ELISA), followed by a Western blot for confirmation.17
Adults should be treated with a 21-day course of doxycycline.18 Amoxicillin, cefuroxime, and penicillin can be used in pregnant women and children younger than 8 years of age.18 If significant rheumatologic, neurologic, or cardiac complications are present, more aggressive regimens consisting of intravenous antibiotics should be used.18
Urticaria
Often referred to as hives, urticaria is an extremely common skin eruption that occurs in up to 20% of the population.20 Most cases are benign and resolve spontaneously. Rarely, patients experience a chronic course lasting longer than six weeks or progress to anaphylaxis.21 In acute cases, a trigger such as infection or allergic reaction may be identified. Chronic urticaria almost always occurs in adults and often does not have an obvious trigger.20 The eruption is composed of wheals, well-defined superficial swellings of the dermis that are characterized by raised, erythematous papules or plaques surrounded by a red flare.21 (See Figure 4.) Shape varies, but lesions are often are round and blanch centrally, conferring an annular appearance.14 Lesions range from a few millimeters to more than 10 cm in size and are accompanied by intense itching, stinging, or burning. The hallmark finding in urticaria is its transient course. As a general rule, individual wheals do not last longer than 24 hours.20 Urticaria may be accompanied by deeper, ill-defined swellings of the dermis and subcutaneous tissue, termed angioedema.21
Figure 4. Edematous Plaque With Red Flare Characteristic of Urticaria |
 |
Source: Karl Kellawan, MD |
Urticaria can be classified as allergic, physical, or idiopathic. Allergic urticaria can be IgE-mediated, occurring in response to foods (milk, eggs, wheat, shellfish, nuts), inhalants (pollen, dander), medications (penicillin), or complement-mediated in the case of serum sickness. Physical urticarias include dermatographic urticaria, occurring where skin has been stroked; cholinergic urticaria, occurring in response to fever and hot baths; cold urticaria, occurring on distal extremities upon rewarming; pressure urticaria, often occurring on the feet and buttocks hours after pressure has been applied; solar urticaria, occurring after exposure to light; and exercise-induced urticaria, which presents with large lesions five to 30 minutes into exercise.14
Urticaria is a clinical diagnosis based on history and physical exam. Laboratory tests rarely are indicated and generally are not recommended. Difficulty arises when patients present asymptomatically after an eruption, limiting the exam. As mentioned earlier, individual lesions should not last longer than 24 hours, although an attack may last much longer.21 If lesions persist beyond 24 hours, other causes must be considered and a biopsy may be warranted.22 If food is a suspected trigger, a food diary may be helpful. If a physical cause is suspected, provocative testing can uncover the offending stimuli.14
Acute urticaria can be managed medically; however, chronic cases should be referred to a specialist for further workup.20 First- and second-generation antihistamines or montelukast can be used as a bridge until a specialist can be seen. Treatment for acute urticaria involves avoidance of identifiable triggers and use of first- or second-generation antihistamines.14,21 If the patient fails to respond, a three-week tapered course of systemic corticosteroids is effective and less associated with rebound than shorter courses.14
Lichen Planus
Lichen planus (LP) is an idiopathic, inflammatory disease of the skin and mucus membranes that primarily affects middle-aged adults.23 Mediated by T-lymphocytes, the reaction can be triggered by medications, vaccinations, or viral infections, especially hepatitis C.24 The eruption begins as small, pinpoint papules that expand into plaques that are classically shiny, violaceous, and polygonal-shaped.25 Wickham striae, a diffuse network of white streaking, can be seen embedded throughout the surface.23 Lesions commonly are found on the dorsal hands, flexor wrists, forearms, shins, vulva, and glans penis, but also can be seen in the mouth.23 When lesions are present on the body, patients may be asymptomatic or complain of intense pruritus. Oral lesions typically are painful, especially when ulcerated.
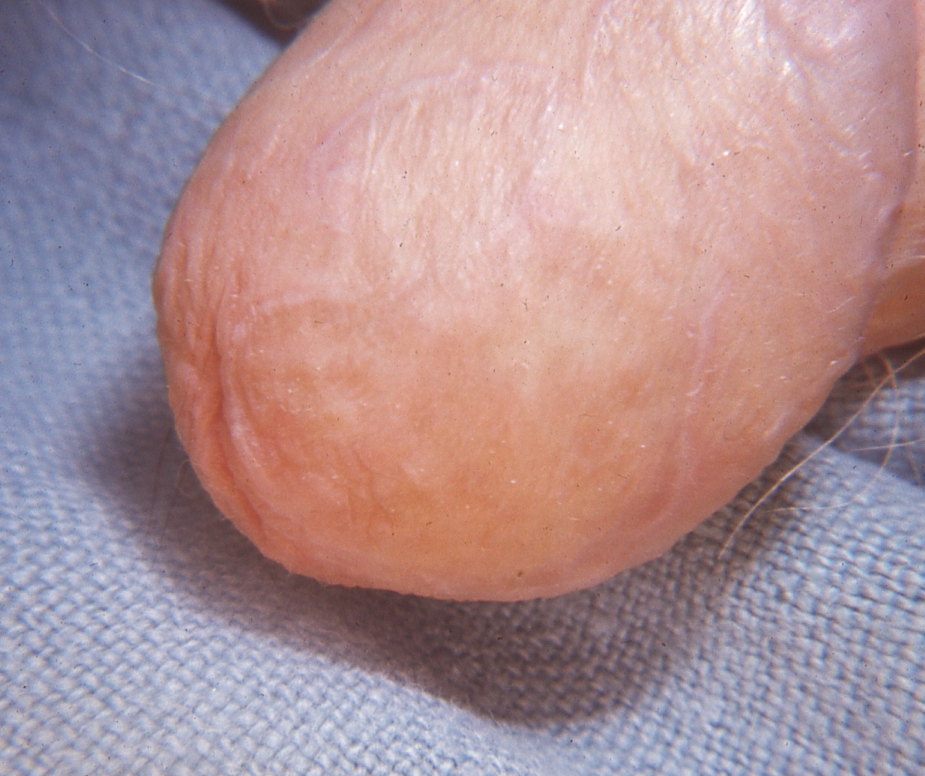
Annular LP is a subtype of LP present in 10% of cases and seen scattered among typical lesions described above. Lesions are characterized by asymptomatic, small, annular papules or plaques with raised borders, typically purple to white in color.24,25 (See Figure 5.) Centrally, lesions may be hyperpigmented or skin-colored.24 LP may mimic tinea, granuloma annulare, or porokeratosis of Mibelli (discussed later). Tinea is classically more erythematous and scaly than LP, while lesions of GA are less numerous and have smooth borders.
Figure 5. Annular Lichen Planus of the Glans Penis |
 |
Source: Karl Kellawan, MD |
Cutaneous LP often is self-limiting; most cases resolve spontaneously within one year.24 For cutaneous disease, topical corticosteroids under occlusion can be used.24,25 When lesions are symptomatic or when oral lesions are present, intralesional triamcinolone is helpful.25 Widespread disease can be treated with systemic corticosteroids, although relapse may occur when the dose is tapered.25
Cutaneous Larva Migrans
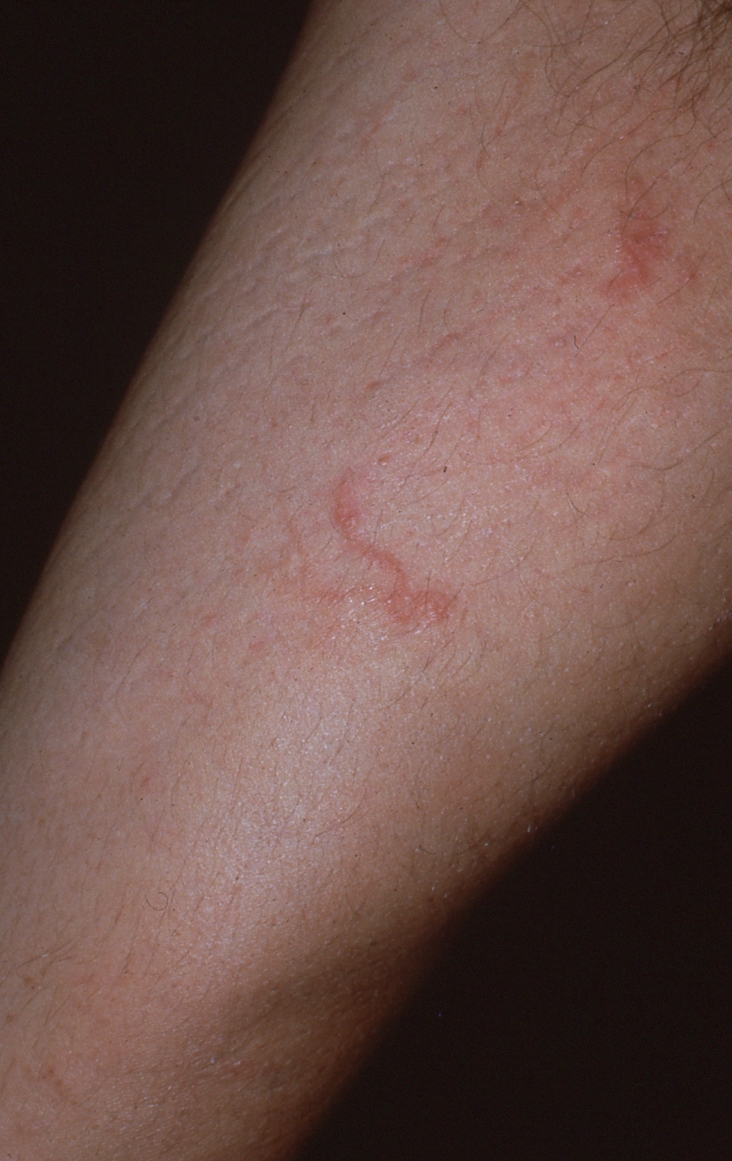
Cutaneous larva migrans is a serpiginous eruption caused by animal hookworm larvae as they migrate through the epidermis.26 In the United States, this disease is most common in warm, southeastern states.27 Percutaneous infection often occurs in those who walk barefoot and children who play in sandboxes. Most cases occur on the feet, hands, and buttocks where larvae can penetrate the skin easily.28 Shortly after infection, patients experience intense pruritus localized to the area of inoculation. Within days, erythematous papules followed by characteristic thin, red, torturous tracts begin to appear.26 (See Figure 6.) Many isolated tracts may be present, each representing the course of an individual larva. Eventually, tracts can coalesce to form annular or ring-shaped lesions. Tracts migrate up to several centimeters a day and may disappear then reappear throughout its course.27 Systemic manifestations are not seen, as larvae are unable to invade deeper tissues.27
Figure 6. Serpiginous Tracts of Cutaneous Larva Migrans on the Proximal Lower Extremity |
 |
Source: Karl Kellawan, MD |
Diagnosis is based on clinical findings and supported by a recent history of potential exposure. The eruption can be differentiated from tinea by its lack of scale and distinctive migrating course.
The cutaneous infestation is self-
limiting in that humans are “dead-end” hosts. Most eruptions spontaneously resolve in two to eight weeks.28 Treatment with systemic agents, such as ivermectin, albendazole, or thiabendazole, is highly effective and preferred over topical preparations.27
Porokeratosis of Mibelli
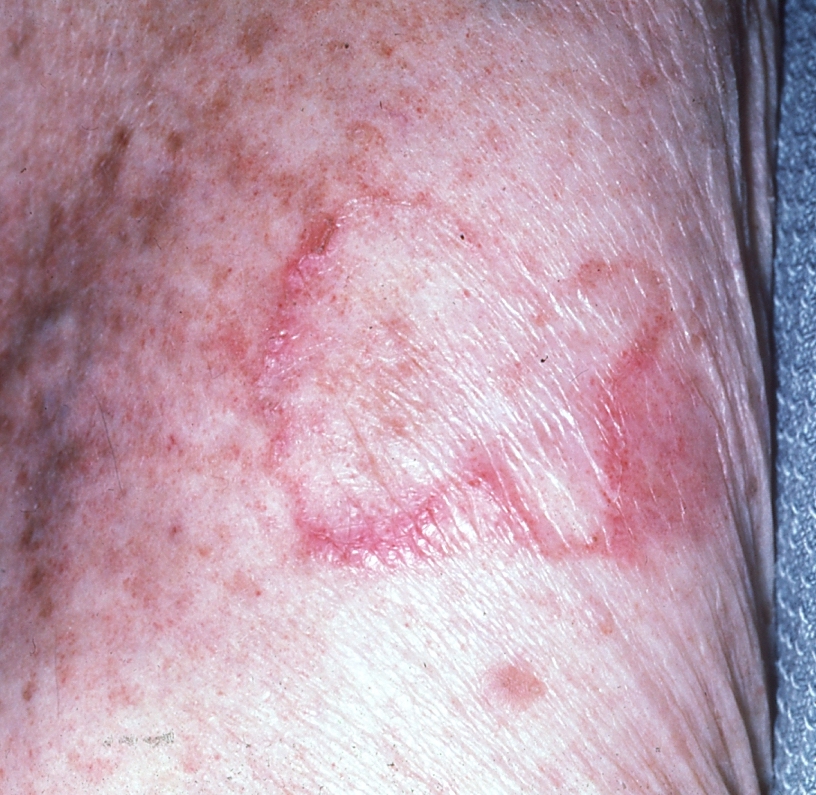
Porokeratosis of Mibelli is a rare, genetically inherited disorder of epidermal keratinization that arises during childhood or adolescence.29 Lesions begin as small, skin- to brown-colored papules that enlarge over the course of years, forming plaques with raised, ridge-like borders and atrophic, hyper- or hypopigmented centers.30 (See Figure 7.) Lesions are asymptomatic, measuring up to several centimeters in diameter and having a predilection for the hands, fingers, feet, and ankles.31 When multiple lesions are present, they are almost always unilateral and localized.30
Figure 7. Enlarging Plaque With Raised Border of Porokeratosis of Mibelli |
 |
Source: Karl Kellawan, MD |
Focusing on the furrowed rim formed by the prominent border and depressed center is key in differentiating the lesion from other annular lesions like tinea. The large, ridge-like border can be accentuated with the application of colored dye, such as crystal violet or povidone-iodine, followed by removal with alcohol.31 In addition, questioning may reveal a family history of similar lesions. Definitive diagnosis can be made with biopsy and histopathological examination showing a cornoid lamella.29
Treatment consists of topical
5-fluorouracil alone or in combination with topical retinoids.30,31 Other possible approaches include cryotherapy with liquid nitrogen.29
Psoriasis
Psoriasis is one of the most common skin conditions encountered by primary care physicians. The immune-mediated, hyperproliferative disorder affects approximately 2% of the U.S. population with equal frequency in men and women.32 Psoriasis can begin at any age, although the majority of cases occur in patients younger than 40 years of age.33
Many different types of psoriasis exist. The most common type, chronic plaque psoriasis, accounts for approximately 90% of cases.34 In this variant, lesions present as well-circumscribed, erythematous, dry plaques covered by a silvery scale. During an eruption, lesions begin small then extend peripherally, becoming more erythematous over time.32 As lesions mature, desquamation occurs, leaving a collarette of scale at the margin, which often is accompanied by intense pruritis and burning.32,33
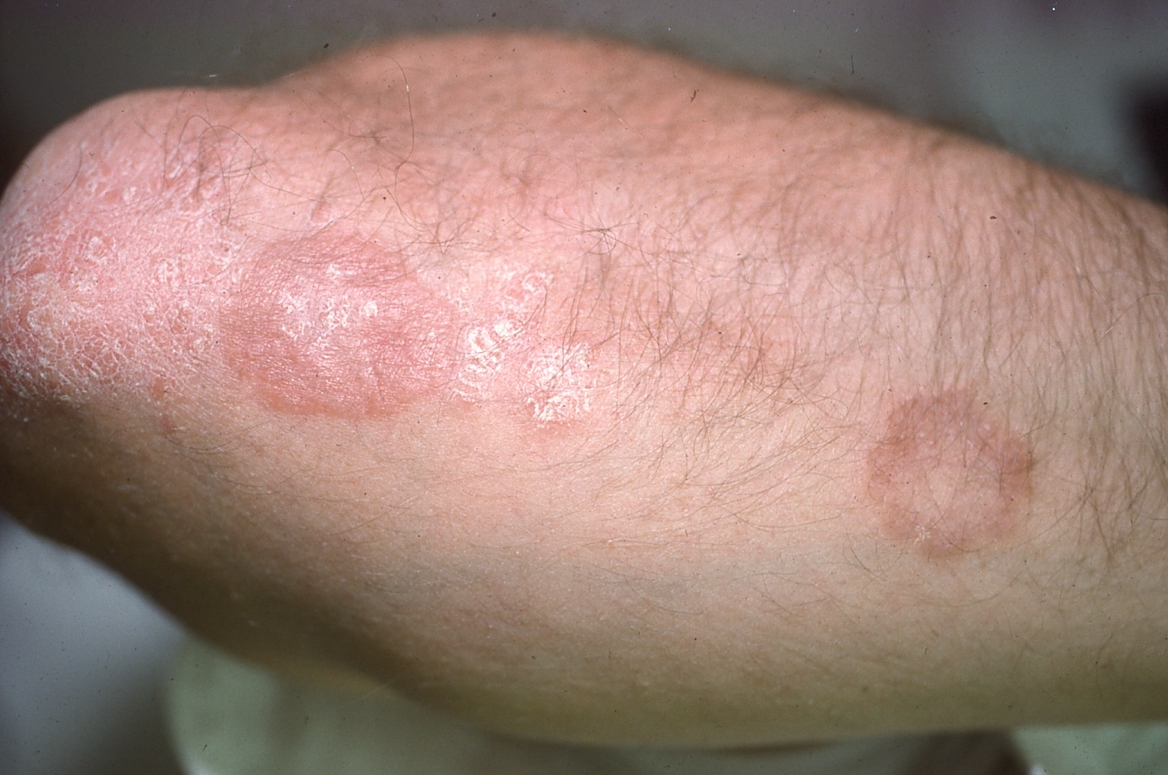
Although characteristic plaques predominate, variations in morphology are not uncommon. Lesions may appear annular when they develop rapidly, forming the active outer margin first, leaving relatively normal skin at the center.34 As plaques regress, margins persist while the center heals, again resulting in annular-shaped lesions.35 (See Figure 8.) A rare form of psoriasis, termed annular pustular psoriasis, also is considered when annular lesions are present. In this variant, lesions are characterized by well-demarcated, erythematous plaques with an active border composed of pustules.
Figure 8. Healing Lesions of Chronic Plaque Psoriasis |
 |
Source: Karl Kellawan, MD |
In many cases, the diagnosis may not be obvious, even for the experienced clinician. Family history may be helpful in revealing relatives with the disease. The location is suggestive when plaques are confined to the extensor surfaces of limbs, umbilical region, and sacrum.33 Physically removing scale can produce pinpoint bleeding, referred to as the “Auspitz sign,” characteristic of psoriasis.32 Extracutaneous manifestations including nail changes and asymmetric, oligoarthritis of the hands and feet commonly are seen in conjunction with cutaneous findings.34
The chronic nature of the disease often leads to complex treatment regimens requiring combination therapy to achieve control. Therefore, referral to a dermatologist is appropriate. For small, localized lesions, potent topical corticosteroids and vitamin D derivatives in creams, lotions, or ointments can be used.32 When plaques persist, intralesional injections of triamcinolone can be tried.32 Another commonly used, cost-effective modality is phototherapy.32 When the disease is severe or widespread, biologic and oral immunosuppressive agents are options.
Nummular Eczema
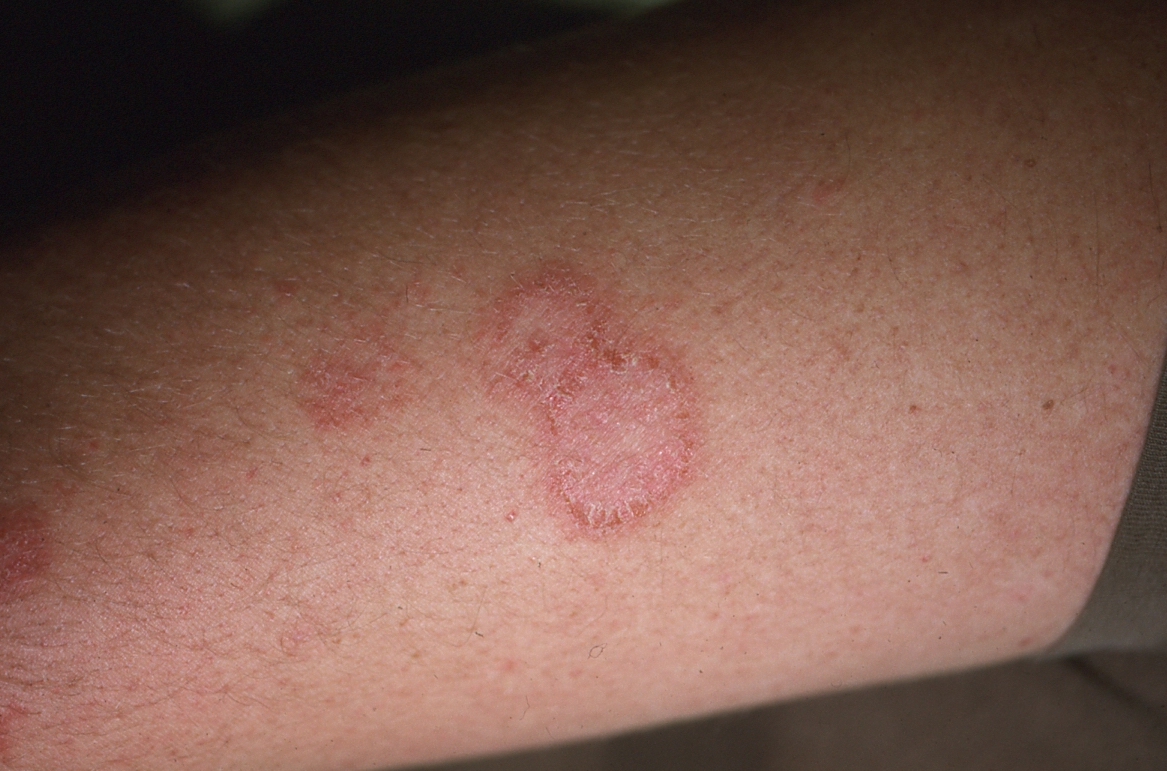
Nummular eczema (NE) is a relatively common, inflammatory dermatitis that occurs in the form of coin-shaped lesions.36 The pathogenesis remains poorly understood, but often it is seen in atopic individuals. Clinically, NE follows a chronic course characterized by well-demarcated, erythematous, or hyperpigmented coin-shaped plaques with diffuse crust.37 (See Figure 9.) Acutely, lesions may ooze and have associated vesicles.38 Most will lack central clearing, although in some cases, plaques can expand and clear centrally, conferring an annular appearance.1 Lesions are especially common during winter months when skin is dry and primarily present on the lower legs, dorsal hands, and extensor surface of the arms.39 Excoriations often are prominent because of intense pruritus.36
Figure 9. Nummular Eczema of the Lower Extremity |
 |
Source: Karl Kellawan, MD |
Nummular eczema should be differentiated from tinea corporis and psoriasis. Tinea corporis is associated with a fine scale and commonly affects the trunk, unlike NE, which has a thick crust and occurs almost exclusively on the extremities. Lesions of psoriasis often are larger and associated with a more silvery scale compared to NE.40 In addition, nail changes and arthritis would not be seen in conjunction with NE.
Treatment consists of potent topical corticosteroids supplemented by daily soaking and greasing with occlusive ointments.36,40 In refractory cases, phototherapy can be used.36
Subacute Cutaneous Lupus Erythematosus
Systemic lupus erythematosus can have dramatic effects on the skin. Skin involvement comes in three variants — acute, subacute, and discoid. Acute cutaneous lupus erythematosus refers to the notorious malar rash that is almost always associated with systemic disease.41 Subacute cutaneous lupus erythematosus (SCLE) describes a photosensitive eruption that commonly forms annular lesions, but is associated with systemic disease only occasionally. Lesions of discoid lupus erythematosus vary greatly; however, they rarely are annular.
SCLE is characterized by the rapid appearance of small or large, erythematous to pink-colored plaques with raised borders, central clearing, and an easily detached scale.41,42 The eruption is extremely photosensitive and, thus, most commonly will present on the face, upper trunk, and the backs of the arms.41
In differentiating SCLE from other annular lesions, a history of sun exposure in correlation with the appropriate distribution supports the diagnosis. Patients often are Caucasian women aged 15 to 40 years.42 Nearly 70% will have anti-SSA/Ro autoantibodies and up to 50% will fulfill four or more of the American College of Rheumatology's criteria for systemic lupus erythematous.1,41,42
Because of the transient nature of the eruption, treatment is centered around sun protection.42 If medical therapy is preferred, topical corticosteroids or antimalarial agents, such as hydroxychloroquine, can be used.41
Sarcoidosis
Sarcoidosis is an idiopathic systemic disease characterized by non-caseating granulomas in multiple organ systems — mainly the lungs, but also the skin, lymph nodes, eyes, and salivary glands.7,9 The disease is more common in women and typically begins between the ages of 20 and 40 years.9 Interestingly, in the United States, there is marked racial variation, with sarcoidosis affecting African Americans more often than Caucasians.9 Skin involvement is seen in up to one-third of those affected and may be the first and/or only clinical sign of disease.7,9 Lesion morphology varies, presenting as multiple round papules, patches, or plaques that favor the face, neck, and areas of prior injury such as tattoos and scars.7 Color ranges from red to purple to brown.7 Annular configurations may be present when multiple papules coalesce or when plaques clear centrally.7 Applying pressure to the firm lesions causes blanching, sometimes revealing a yellow-brown (apple-jelly) color.7 Lesions are almost always asymptomatic, although rarely may itch.9
Diagnosis may be straightforward in a patient with a known history of sarcoidosis; however, with no prior history, a biopsy of the lesion will reveal non-
caseating epithelioid granulomas.7 In newly diagnosed cases, a chest X-ray is warranted and, if abnormal, patients should be referred for pulmonary evaluation.9
Treating the underlying systemic disease often clears cutaneous lesions.9 Limited cutaneous eruptions can be treated with intralesional triamcinolone.9 Systemic corticosteroids are effective but generally are reserved for active pulmonary, ocular, cardiac, and central nervous system involvement or when skin disease is widespread.9
Seborrheic Dermatitis
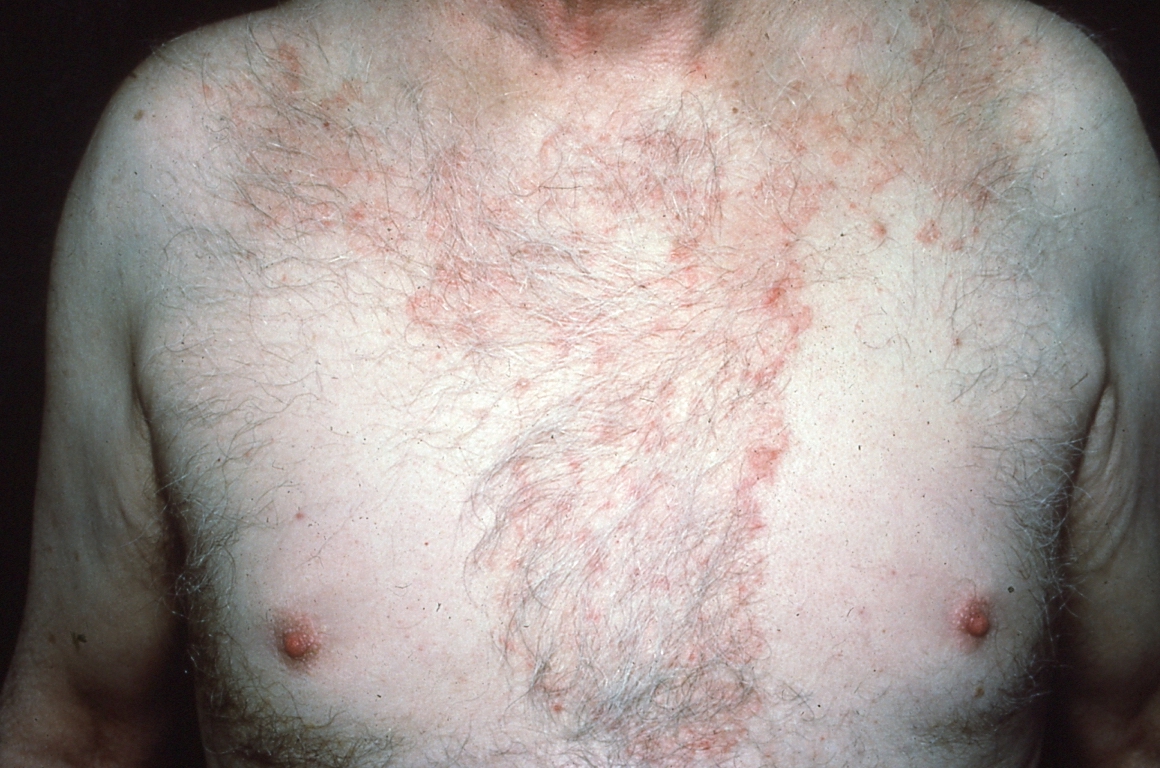
Seborrheic dermatitis is a common, chronic dermatosis present in up to 5% of the population.32 The pathogenesis has been linked to the overproduction of sebum and Malassezia yeasts. Therefore, eruptions occur in regions in which sebaceous glands are most active, such as the scalp, face, eyebrows, ears, and presternal area (see Figure 10), as well as large body folds such as the axillae and groin.36 Lesions are more common in men and are characterized by gradual onset of sharply demarcated patches or thin plaques that vary in color from red to orange to yellow. Flaky, visibly greasy, white to yellow scaling overlies affected areas.36 Most cases are pruritic, and symptoms generally intensify with sweat.32,36
Figure 10. Sharply Demarcated, Scaly Patch of Seborrheic Dermatitis Affecting the Presternal Area |
 |
Source: Karl Kellawan, MD |
Seborrheic dermatitis may closely resemble tinea or psoriasis, especially in the groin. Distribution is helpful in distinguishing seborrheic dermatitis from tinea. When seborrheic dermatitis is present on the face, lesions are remarkably symmetric, mostly affecting the forehead, eyebrows, and retroauricular areas.36 When present in large folds, seborrheic dermatitis tends to be centered along the crease, whereas tinea is less often as symmetric.36 In addition, patches and plaques of seborrheic dermatitis typically are more numerous and appear greasier than lesions of tinea. Plaques of psoriasis are associated with a heavier scale that, unlike seborrheic dermatitis, bleeds when removed.32
Treatment consists of imidazoles, mainly ketoconazole, as a shampoo when present on the scalp or cream when present on the face and body.36 Topical steroids work quickly and are effective but should be limited because of the risk of steroid rosacea.32 Once cleared, daily maintenance therapy is important to prevent relapse.
Erythema Multiforme
Erythema multiforme (EM) is an acute and often recurrent eruption that commonly affects young adults.14 The reaction pattern represents an immune-mediated response to a variety of antigenic agents, most notably herpes simplex. Other triggers include drugs (penicillin, phenytoin, allopurinol, sulfonamides, barbiturates) and Mycoplasma infections.14 Erythema multiforme can be separated into EM minor and EM major, the more severe form with mucosal involvement. The minor variant is not associated with systemic symptoms, but EM major is accompanied by symptoms such as fevers and arthralgias.14
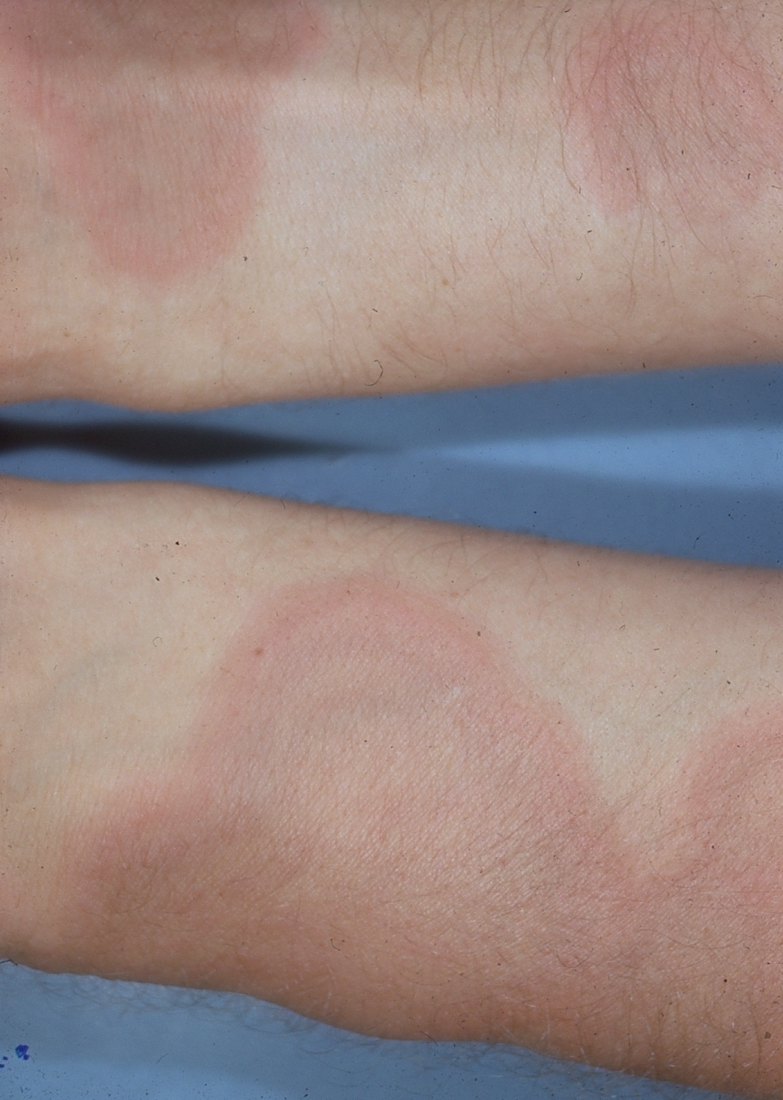
EM minor is characterized by distinctive “target-shaped” or “iris-shaped” lesions.14 Lesions evolve over several days, beginning as round, sharply demarcated erythematous macules that progress to raised, edematous papules.4 Mature lesions have three distinct zones — a dull red center, a pale outlining ring, and an encircling macular erythema.14 (See Figure 11.) Occasionally, atypical target lesions that are round, edematous, and composed of only two distinct zones are present.43 Lesions may sting, burn, and even blister at the center.43 Sites of predilection include the palms, dorsal hands, elbows, backs of the arms, knees, shins, dorsal feet, and soles.14,43
Figure 11. Target-shaped Lesions of Erythema Multiforme Affecting the Forearms Bilaterally |
 |
Source: Karl Kellawan, MD |
EM major occurs primarily as a drug reaction. The eruption presents with the same target-shaped lesions seen in EM minor but is accompanied by systemic symptoms and mucosal involvement.14 Erosions and even ulcerations may involve the oral, genital, and ocular mucosa.14,43
EM often is confused with large urticarial lesions. To diagnose EM, several lesions must evolve into the classic, target-shaped ones described above.43 In addition, EM lesions remain fixed for multiple days, whereas individual urticarial lesions last less than 24 hours. Erythema chronicum migrans also presents with a target-shaped lesion, but the presence of numerous, small lesions and potential mucosal involvement seen in EM separates the two eruptions. EM is self-limiting and usually resolves within weeks without complications.14 For limited cutaneous disease, symptomatic treatment with oral antihistamines or low-potency topical corticosteroids is sufficient.43 In recurrent cases precipitated by herpes simplex virus eruptions, chronic suppressive antiviral therapy with valacyclovir is effective.14 When oral lesions are present, topical corticosteroid gels and “swish and spit” rinses containing a mixture of lidocaine, diphenhydramine, and kaolin are helpful.14 In severely ill patients, systemic corticosteroids should be considered.43
Hansen’s Disease
Hansen’s disease, also known as leprosy, is a rare, chronic granulomatous disease caused by Mycobacterium leprae.44 Although mostly a disease of the developing world, cases in the United States occur in those who have resided in foreign countries or those living in southeastern states with exposure to armadillos, a natural host.1,45 In almost all new cases, a history of close contact with an untreated, infected individual is obtainable.45 Once a patient is infected, the disease has an insidious onset and an incubation period that ranges from three to 20 years.1 Hansen’s disease primarily presents in two forms, tuberculoid leprosy and lepromatous leprosy.45 In both forms, cooler areas of the body such as the skin, peripheral nerves, upper respiratory tract, and eyes are affected.1,45 Warmer areas such as the axilla, groin, and scalp usually are spared. Often, peripheral nerves are affected first, resulting in paresthesias and numbness of the infected area; however, symptoms may be mild and go undiagnosed until cutaneous lesions appear.45
The tuberculoid form represents a strong cell-mediated immune response.45 Patients present with localized disease, consisting of fewer than five asymmetrically distributed lesions characterized by large plaques with a raised, sharply defined border and an atrophic, depressed center.45 Typically, the borders are erythematous to purple in color while the centers often are hypopigmented. Lesions may be dry, scaly, and hairless, but most importantly, anesthetic.44,45 The superficial nerves supplying the area may be enlarged, tender, and palpable.1,45
The lepromatous form represents a poor cell-mediated immune response.45 Patients present with generalized disease, consisting of numerous, symmetrically distributed lesions characterized by small, poorly defined hypopigmented macules without associated anesthesia.45 Eventually, nodules and raised plaques of the face (leonine facies), ears, elbows, hands, and knees may appear.45 Nerve involvement can occur, but will present as a symmetric stocking-glove pattern neuropathy.45
Diagnosis of Hansen’s disease is made by identifying the infectious acid-fast bacilli in skin or nerve lesions with biopsy.45 In the United States, tuberculoid leprosy is treated with a combination of dapsone and rifampin for 12 months.45 Lepromatous leprosy is treated with a combination of dapsone, rifampin, and clofazimine for two years.45
Summary
Annular skin lesions are among the most common dermatologic presentations encountered by primary care physicians. Although tinea is often to blame, many other entities mimic these infections. Establishing a broad differential and focusing on key morphological characteristics will improve accuracy of diagnosis, limit unnecessary treatment, and lead to faster resolution of disease.
Want to earn free Dermatology CME courses? With FreeCME you can earn accredited CME courses online for free. Get started today!
REFERENCES
- Hsu S, Le EH, Khoshevis MR. Differential diagnosis of annular lesions. Am Fam Physician 2001;64:289-296.
- Elewski BE, et al. Fungal Diseases. In: Bolognia JL, Schaffer JV, Cerroni L. Dermatology. 4th ed. Elsevier Limited; 2018:1329-1363.
- Perkins J, Buckland M, Miller RA. Fungus among us? Dermatophyte infections, mimickers, and treatment options. Primary Care Reports 2017;23:77-87.
- James WD, Berger TG, Elston DM. Diseases Resulting From Fungi and Yeasts. In: James WD, Berger TG, Elston DM. Andrews’ Diseases of the Skin: Clinical Dermatology. 12th ed. Philadelphia: Elsevier; 2016:285-318.
- Kaushik N, Pujalte GG, Reese ST. Superficial fungal infections. Prim Care 2015;42:501-516.
- Moriary B, Hay R, Morris-Jones R. The diagnosis and management of tinea. BMJ 2012;345:e4380.
- Rosenbach M, et al. Non-infectious Granulomas. In: Bolognia JL, Schaffer JV, Cerroni L. Dermatology. 4th ed. Elsevier Limited; 2018:1644-1663.
- Piette EW, Rosenbach M. Granuloma annulare: Pathogenesis, disease associations and triggers, and therapeutic options. J Am Acad Dermatol 2016;75:467-479.
- James WD, Berger TG, Elston DM. Macrophage/Monocyte Disorders. In: James WD, Berger TG, Elston DM. Andrews’ Diseases of the Skin: Clinical Dermatology. 12th ed. Philadelphia: Elsevier; 2016:699-725.
- Wood GS, Reizner GT. Other Papulosquamous Disorders. In: Bolognia JL, Schaffer JV, Cerroni L. Dermatology. 4th ed. Elsevier Limited; 2018:161-174.
- Eisman S, Sinclair R. Pityriasis rosea. BMJ 2015;351;h5233.
- James WD, Berger TG, Elston DM. Pityriasis Rosea, Pityriasis Rubra Pilaris, and Other Papulosquamous and Hyperkeratotic Diseases. In: James WD, Berger TG, Elston DM. Andrews’ Diseases of the Skin: Clinical Dermatology. 12th ed. Philadelphia: Elsevier; 2016:199-208.
- Drago F, Ciccarese G, Rebora A, et al. Pityriasis rosea: A comprehensive classification. Dermatology 2016;232:431-437.
- James WD, Berger TG, Elston DM. Erythema and Urticaria. In: James WD, Berger TG, Elston DM. Andrews’ Diseases of the Skin: Clinical Dermatology. 12th ed. Philadelphia: Elsevier; 2016:136-152.
- Mu EW, Sanchez M, Mir A, et al. Paraneoplastic erythema annulare centrifugum eruption (PEACE). Dermatol Online J 2015;21:pii: 13030/qt6053h29n.
- España A. Figurate Erythemas. In: Bolognia JL, Schaffer JV, Cerroni L. Dermatology. 4th ed. Elsevier Limited; 2018:320-331.
- Sommer LL, Reboli AC, Heymann WR. Bacterial Diseases. In: Bolognia JL, Schaffer JV, Cerroni L. Dermatology. 4th ed. Elsevier Limited; 2018:1259-1295.
- James WD, Berger TG, Elston DM. Bacterial Infections. In: James WD, Berger TG, Elston DM. Andrews’ Diseases of the Skin: Clinical Dermatology. 12th ed. Philadelphia: Elsevier; 2016:136-152.
- Nadelman RB. Erythema migrans. Infect Dis Clin North Am 2015;29:211-239.
- Peroni A, Colato C, Schena D, Girolomoni G. Urticarial lesions: If not urticarial, what else? The differential diagnosis of urticaria. J Am Acad Dermatol 2010;62:541-555.
- Grattan CE, Saini SS. Urticaria and Angioedema. In: Bolognia JL, Schaffer JV, Cerroni L. Dermatology. 4th ed. Elsevier Limited; 2018:304-319.
- Rudikoff D. Differential diagnosis of round or discoid lesions. Clin Dermatol 2011;29:489-497.
- Weston G, Payette M. Update of lichen planus and its clinical variants. Int J Womens Dermatol 2015;1:140-149.
- Shiohara T, Mizukawa Y. Lichen planus and lichenoid dermatoses. In: Bolognia JL, Schaffer JV, Cerroni L. Dermatology. 4th ed. Elsevier Limited; 2018:188-207.
- James WD, Berger TG, Elston DM. Lichen Planus and Related Conditions. In: James WD, Berger TG, Elston DM. Andrews’ Diseases of the Skin: Clinical Dermatology. 12th ed. Philadelphia: Elsevier; 2016:209-224.
- Feldmeier H, Schuster A. Mini review: Hookworm-related cutaneous larva migrans. Eur J Clin Microbiol Infect Dis 2012;31:915-918.
- Bravo FG. Protozoa and Worms. In: Bolognia JL, Schaffer JV, Cerroni L. Dermatology. 4th ed. Elsevier Limited; 2018:1470-1502.
- James WD, Berger TG, Elston DM. Parasitic Infestations, Stings, and Bites. In: James WD, Berger TG, Elston DM. Andrews’ Diseases of the Skin: Clinical Dermatology. 12th ed. Philadelphia: Elsevier; 2016:418-450.
- Ferreira FR, Santos LD, Tagliarini FA, Lira ML. Porokeratosis of Mibelli — literature review and a case report. An Bras Dermatol 2013;88(6 Suppl 1):179-182.
- Requena L, Requena C, Cockerell CJ. Benign Epidermal Tumors and Proliferations. In: Bolognia JL, Schaffer JV, Cerroni L. Dermatology. 4th ed. Elsevier Limited; 2018:1894-1916.
- James WD, Berger TG, Elston DM. Genodermatoses and Congenital Anomalies. In: James WD, Berger TG, Elston DM. Andrews’ Diseases of the Skin: Clinical Dermatology. 12th ed. Philadelphia: Elsevier; 2016:542-578.
- James WD, Berger TG, Elston DM. Seborrheic Dermatitis, Psoriasis, Recalcitrant, Palmoplantar Eruptions, Pustular Dermatitis, and Erythroderma. In: James WD, Berger TG, Elston DM. Andrews’ Diseases of the Skin: Clinical Dermatology. 12th ed. Philadelphia: Elsevier; 2016:185-198.
- van de Kerkhof PCM, Nestle FO. Psoriasis. In: Bolognia JL, Schaffer JV, Cerroni L. Dermatology. 4th ed. Elsevier Limited; 2018:138-160.
- Griffiths CEM, Barker JNWN. Pathogenesis and clinical features of psoriasis. Lancet 2007;370:263-271.
- Naldi L, Gambini D. The clinical spectrum of psoriasis. Clin Dermatol 2007;25:510-518.
- Reider N, Fritsch PO. Other eczematous eruptions. In: Bolognia JL, Schaffer JV, Cerroni L. Dermatology. 4th ed. Elsevier Limited; 2018:228-241.
- Barrett M, Luu M. Differential diagnosis of atopic dermatitis. Immunol Allergy Clin North Am 2017;37:11-34.
- Silverberg NB. Typical and atypical clinical appearance of atopic dermatitis. Clin Dermatol 2017;35:354-359.
- Suh KS, Park JB, Yang MH, et al. Diagnostic usefulness of dermoscopy in differentiating lichen aureus from nummular eczema. J Dermatol 2017;44:533-537.
- James WD, Berger TG, Elston DM. Atopic Dermatitis, Eczema, and Noninfectious Immunodeficiency Disorders. In: James WD, Berger TG, Elston DM. Andrews’ Diseases of the Skin: Clinical Dermatology. 12th ed. Philadelphia: Elsevier; 2016:62-89.
- Lee LA, Werth VP. Lupus Erythematosus. In: Bolognia JL, Schaffer JV, Cerroni L. Dermatology. 4th ed. Elsevier Limited; 2018:662-680.
- James WD, Berger TG, Elston DM. Connective Tissue Diseases. In: James WD, Berger TG, Elston DM. Andrews’ Diseases of the Skin: Clinical Dermatology. 12th ed. Philadelphia: Elsevier; 2016:153-178.
- Hotzenecker W, Prins C, French LE. Erythema Multiforme, Stevens-Johnson Syndrome, and Toxic Epidermal Necrolysis. In: Bolognia JL, Schaffer JV, Cerroni L. Dermatology. 4th ed. Elsevier Limited; 2018:332-347.
- Ramos-e-Silva M, Ribeiro de Castro MC. Mycobacterial Infections. In: Bolognia JL, Schaffer JV, Cerroni L. Dermatology. 4th ed. Elsevier Limited; 2018:1296-1318.
- James WD, Berger TG, Elston DM. Hansen’s Disease. In: James WD, Berger TG, Elston DM. Andrews’ Diseases of the Skin: Clinical Dermatology. 12th ed. Philadelphia: Elsevier; 2016:331-342.
Annular skin lesions commonly plague many primary care patients, but not all that is round is fungus. This article highlights the differential diagnosis of conditions that can mimic tinea.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
