Pediatric Abdominal Ultrasound: A Comprehensive Guide to Making the Diagnosis — Part II
August 1, 2018
Reprints
AUTHORS
Kimberly M. Fender, MD, Emergency Medicine Resident, PGY 1, Department of Emergency Medicine, University of North Carolina, Chapel Hill
Daniel B. Park, MD, Associate Medical Director, Pediatric Emergency Medicine; Director, Pediatric Emergency Ultrasound; Assistant Professor, Departments of Pediatrics and Emergency Medicine, University of North Carolina School of Medicine, Chapel Hill
Daniel Migliaccio, MD, Clinical Assistant Professor, Department of Emergency Medicine, University of North Carolina, Chapel Hill
PEER REVIEWER
Aaron Leetch, MD, Assistant Professor, Departments of Emergency Medicine and Pediatrics; Program Director, Combined Emergency Medicine and Pediatrics Residency, University of Arizona, Tucson
EXECUTIVE SUMMARY
- The incidence of renal colic in the pediatric population is increasing and mostly affects adolescents. Ultrasound can be used to diagnose stones by direct visualization or by evidence of obstructive uropathy.
- The twinkling artifact is a multicolor, high-intensity signal produced behind highly reflective surfaces, like calculi, when color Doppler is applied. Several studies have shown that the sensitivity of ultrasound for detecting lithiasis increases when this artifact is present compared to using B-mode alone.
- Ovarian torsion (OT) is rare, with an estimated incidence of 4.9 per 100,000 females between 1 and 20 years of age and an average age of 14.5 years, and occurs in both premenarchal and postmenarchal girls. Ultrasound is the primary imaging modality to evaluate pelvic pain, with a sensitivity and specificity of 90.9% and 68.7% for OT, respectively. The presence of an ovarian mass, most commonly a functional cyst, mature teratoma, or serous cystadenoma, is associated with OT. OT occurs more frequently on the right side.
- Testicular torsion is a urological emergency that accounts for approximately 23% of acute scrotums in boys and requires a prompt and accurate diagnosis to guide management to promote testicular salvage. Torsion occurs when the testis twists around the vascular pedicle, also known as the spermatic cord. Ultrasound is the main diagnostic modality used to differentiate between various scrotal pathologies.
- Although the diagnosis of small bowel obstruction (SBO) occurs less frequently in children compared to adults, the incorporation of point-of-care ultrasound into the assessment of children who present with abdominal pain, vomiting, and/or abdominal distention to identify SBO should be considered. Plain abdominal films often are nondiagnostic and have been found to be inferior to ultrasound in the evaluation of SBO, despite the use of X-ray as a standard initial imaging technique. In a systematic review and meta-analysis, researchers concluded that ultrasound was 92% sensitive and 96% specific for the diagnosis of small bowel obstruction.
- Children of all ages are subject to gallbladder disease. Although many cases of pediatric cholelithiasis are idiopathic, there is a wide spectrum of risk factors, including obesity, pregnancy, hemolytic disease, prolonged total parenteral nutrition, and use of oral contraceptives. Ultrasound is the favored imaging modality to identify gallbladder pathology. Detection of cholecystitis by ultrasound by emergency medicine physicians shares similar accuracy to that of radiologists.
Ultrasound is evolving rapidly as the ideal imaging modality for many common pediatric complaints. In the second part of this series, the authors discuss point-of-care use of ultrasound for concerns regarding the kidneys, ovaries, testicles, gallbladder, and small bowel obstruction. The ability to make critical diagnoses safely and rapidly with ultrasound is an invaluable clinical tool to facilitate and improve pediatric care.
— Ann M. Dietrich, MD, FAAP, FACEP, Editor
To avoid ionizing radiation in support of the ALARA (as low as reasonably achievable) concept in pediatric imaging, ultrasound frequently is the preferred imaging modality in the pediatric population.1 Ultrasound is noninvasive, cost-effective, easy to use, portable, and requires no sedation; however, the reliability of ultrasound is highly operator dependent. Point-of-care ultrasound performed and interpreted in the emergency department (ED) can reduce the time to disposition and expedite definitive patient care.2-4 This two-part series reviews the applications of ultrasound to diagnose common pediatric abdominal pathologies, as well as discusses the basic technique for each ultrasound exam. Part I included discussion of pyloric stenosis, acute appendicitis, intussusception, the Pediatric Focused Assessment with Sonography for Trauma (FAST) exam, and imaging of the inferior vena cava and aorta. Part II will include renal, ovarian, testicular, and biliary ultrasound focusing on diagnosing obstructive uropathy, ovarian torsion, testicular torsion, small bowel obstruction, and cholecystitis. Emergency medicine physicians should be proficient in using point-of-care pediatric ultrasound while having a thorough understanding of the limitations and common pitfalls.
Renal, Testicular, and Ovarian Ultrasound: From the Beans to the Groin and Everything in Between
Renal Ultrasound: Time Traveling Back to the Stone Age
A 12-year-old girl presents to the ED with right-sided flank pain and nausea. Her creatinine is within normal limits and her urinalysis is significant for hematuria. There is concern that she may have an obstructing ureteral stone. The incidence of renal colic in the pediatric population is rising and mostly affects adolescents.5 Stone disease is related to one in 685 pediatric hospitalizations in the United States.6 Stones that are more distal in location and 5 mm or less in size have a greater probability of passing and are unlikely to require urologic intervention.7
Unenhanced computed tomography (CT) is the preferred imaging method to detect nephrolithiasis in patients with suspected renal colic; however, ultrasound is a useful alternative in the emergency setting to lower cost and limit exposure to ionizing radiation. Ultrasound can be useful for diagnosing kidney stones either via direct visualization or by evidence of obstructive uropathy, particularly in the setting of a high clinical suspicion for the presence of obstructing stone disease. Point-of-care ultrasound can identify signs of obstruction secondary to urolithiasis, including hydronephrosis, hydroureter, and decreased or absent ureteral jets on the affected side.8 Although ureteral stones often are obscured by bowel gas, ultrasound is best at detecting stones that are either proximal to the ureteropelvic junction or distal to the ureterovesical junction.9 Stones are hyperechoic with associated posterior shadowing. CT is both highly sensitive and specific for diagnosing kidney stones with a sensitivity of 95-98% and specificity of 95-100%.10-12 Authors of one study found that the preferential use of ultrasound as the initial imaging modality vs. CT reduced cumulative radiation exposure; they found no significant difference in complications, hospitalizations, pain scores, serious adverse events, or return ED visits.13
Initiate the exam with the patient in the supine position. The curvilinear or phase-array transducer should be used to perform the exam. The smaller footprint of the phase-array probe may assist in intercostal imaging to avoid rib shadowing. The right kidney typically is found posterior to the right midaxillary line between the ninth and 10th intercostal space. The probe indicator should be directed toward the patient’s head. Adjusting either the angle of the probe to facilitate imaging between the intercostal spaces or directing the probe cephalad or caudal may be necessary to bring the kidney, with its characteristic bean shape, into view. Both the upper and lower poles of the kidney should be visualized.14,15 A thin, linear capsule separates the kidney from the surrounding echogenic perirenal fat. The kidney is divided into the renal parenchyma, which is composed of the cortex and more hypoechoic medullary pyramids, and the hyperechoic renal sinus, which includes the calyces, renal pelvis, and major intrarenal vessels.16 Nomograms exist for estimating pediatric kidney size based on height and weight.17 Fan the probe anteriorly and posteriorly in the coronal plane to evaluate the entire long axis of the kidney. Evaluate the structure of the kidney while looking for evidence of hydronephrosis, stones, masses, cysts, or other pathology. (See Figure 1.)
Figure 1. Ureterovesical Junction Stone |
|
A stone (indicated by the arrowheads) is present at the ureterovesical junction (or UVJ). The asterisk denotes the ureteral dilation proximal to the stone. |
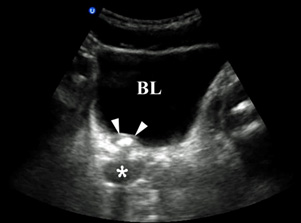 |
Rotate the probe by 90 degrees to view the kidney in a transverse orientation. Fan the ultrasound probe in the cephalad to caudal direction to view the entire kidney. The left kidney’s anatomical placement differs from that of the right kidney. The left kidney is located more superior and posterior compared to the right kidney. To visualize the left kidney, place the probe, with the probe indicator toward the head of the patient, between the seventh and eighth intercostal spaces at the posterior axillary line. Perform the same movements to obtain the images of the left kidney in both the coronal and transverse planes. Sometimes overlying bowel gas may obscure the view of the kidney. If the kidneys are difficult to visualize, ask the patient to take a deep breath in and hold. The position of the kidney will change with inspiration and may provide a superior imaging window. Furthermore, rotating the patient in the lateral decubitus or prone position may be helpful.14,15
Renal point-of-care ultrasound is used commonly in the ED setting to evaluate for secondary signs of nephrolithiasis, including hydronephrosis. (See Figure 2.) A grading system ranging from mild to severe was created to classify the degree of dilation of the intrarenal collecting system. In a normal kidney or grade 0, no dilatation of the collecting system should be present. Grade I is minimal dilation of the renal pelvis only. Grade II is present when the pelvis and most, but not all, calyces are dilated. Grade III is complete pelvocaliectasis with uniformly dilated calyces with sparing of the parenchyma. Grade IV is significant dilation of the entire collecting system with parenchymal thinning.16,18
Figure 2. Progressive Severity of Hydronephrosis |
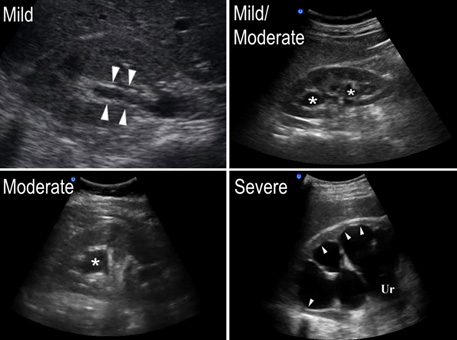 |
The twinkling artifact is a multicolor, high-intensity signal produced behind highly reflective surfaces, like calculi, when color Doppler is applied. Several studies have shown that the sensitivity of ultrasound for detecting lithiasis increases when this artifact is present compared to using B-mode alone.19,20 The sensitivity, specificity, and accuracy of ultrasound for detecting lithiasis are 90-98.3%, 100%, and 92-98.4%, respectively.21,22 The twinkling artifact may assist in localizing smaller stones and stones positioned in the ureter, often one of the most challenging areas to visualize with ultrasound.22
Ureteral jet flow can provide useful information about the integrity and flow dynamics of the urinary tract system. Studies have shown that the ureteral jet frequency, duration, and peak velocity are decreased in an obstructed ureter when compared to an unobstructed ureter.23 Peak flow velocity is related inversely to stone size, and can be used to predict spontaneous passage.24 Perform the exam by obtaining a transverse image of the bladder that encompasses both of the ureteral orifices at the level of the trigone. Apply color Doppler to view the ureteral jets. Most studies in the literature suggest observing for ureteral jets for 5-10 minutes.23,25
Ovarian Torsion: Twister Warning
A 15-year-old female with a recent diagnosis of polycystic ovarian syndrome enters the ED with a two-day history of right lower pelvic pain. She reports one episode of vomiting. She is afebrile and has right pelvic tenderness to palpation. Laboratory results reveal leukocytosis. A large differential diagnosis exists for pelvic and lower abdominal pain in children; however, ovarian torsion (OT) is a possible suspect. (See Table 1.)
Table 1. Findings on Point-of-care Ultrasound |
||
Pathology |
Findings |
Comments |
|
Ovarian torsion |
Enlarged peripherally displaced cysts, free fluid, whirlpool sign, medialization of the ovary, absence of color Doppler (typically a late finding) |
Ultrasound is a good indicator, but is not definitive to exclude torsion. Must consult gynecology if there is a high degree of suspicion. |
|
Testicular torsion |
Heterogenous appearance, enlarged, lack of color Doppler, whirlpool sign (spermatic cord twisting) |
Ultrasound is a good indicator, but is not definitive to exclude torsion. Must consult urology if there is a high degree of suspicion |
OT is a challenging, time-sensitive, and feared gynecologic emergency to miss among emergency medicine physicians. OT is defined by a partial or complete rotation of the ovary and adnexal components around the ligamentous structures that results in compromised circulation and lymphatic obstruction.26,27 It is referred to as adnexal torsion if the fallopian tube is affected as well.27 Often, children are unable to provide an adequate history, and the presenting symptoms frequently are nonspecific; however, nausea and vomiting are more likely to be reported in OT.28-31 Peritoneal signs are elicited more often in patients with torsion.29
OT is rare, with an estimated incidence of 4.9 per 100,000 females between 1 and 20 years of age, with an average age of 14.5 years.32 Both premenarchal and postmenarchal girls are at risk for OT.27,33 Ultrasound is the primary imaging modality to evaluate pelvic pain, with a sensitivity and specificity of 90.9% and 68.7% for OT, respectively.28 The presence of an ovarian mass, most commonly a functional cyst, mature teratoma, or serous cystadenoma, is associated with OT.28 OT has been found to occur more frequently on the right side.34
Authors of several recent studies have advocated for ovarian detorsion, with the goal to spare the ovary rather than perform definitive operative management with oophorectomy.30,35 Walker et al found that more than 70% of children and adolescents who underwent ovarian-sparing operations had signs of subsequent development of ovarian follicles in the previously torsed ovary.30 Laparoscopy should be considered in children with high clinical suspicion for ovarian torsion, despite a negative ultrasound.36
The most common sonographic finding of OT is an enlarged ovarian mass.28,34 An ovarian mass > 5 cm in the setting of pain is highly determinant for OT.28 The presence of ovarian stromal edema and the peripheral distribution of follicles has been found to be predictive of OT.28 Linam et al found ovarian torsion in menarchal females is unlikely if the adnexal volume is less than 20 mL,37 while Servaes et al found that the median volume of the torsed adnexa was 12 times that of the contralateral normal ovary when both ovaries were visualized.34 Assessment of the contralateral ovary is essential when evaluating for torsion. In torsion, medialization of the affected ovary may occur. That is, when the adnexal structures become twisted, the distance between the ovary and the uterus shortens and the ovary becomes more mid-line.38
A high-frequency or curvilinear probe should be used to perform the exam. The transabdominal approach is preferred in premenarchal children. It is recommended that the patient have a full bladder during the assessment to create an acoustic window to improve visualization of the ovaries.27 Place the probe superior to the pubic symphysis midline on the abdominal wall with the probe angled into the pelvis; the probe indicator should be pointed toward the patient’s head. Assess the uterus, ovaries, and the adnexal structures in both the longitudinal and transverse orientations. A normal ovary should appear as an ellipsoid structure containing hypoechoic follicles; the size of the ovary varies with age, pubertal status, and menstruation.
The ovary has a dual blood supply from both the ovarian and uterine arteries. There is conflicting evidence regarding the ability of color Doppler to predict ovarian vascular compromise in torsion. Several studies have found a poor correlation between actual vascular sufficiency and color Doppler tracings. Color flow, either venous or arterial in origin, was found to be present in 62% of OT cases.34 A Doppler signal in an abnormal ovary does not eliminate the possibility of OT. Likewise, the absence of color Doppler signals as a lone ultrasound finding does not necessarily translate into a diagnosis of torsion every time.27,39
Free fluid in the pelvis may be present, but often is small in quantity and nonspecific.29,34 The “spiral” or “whirlpool” sign is a sonographic finding indicative of adnexal torsion;40 this sign refers to the appearance of the twisted ovarian pedicle together with a characteristic Doppler sign that resembles a whirlpool or spiral.41 To produce this sign, move the probe back and forth along the axis of the ovarian pedicle.40 This sign can be found lateral or medial to the ovary.40 Although it is sensitive for torsion, it is not always present.41,42 Of note, this sign is used in the testicular torsion literature as well. Be aware that hemorrhagic cysts can share a similar appearance to OT and may result in false-positive results.28,38 (See Figure 3.)
Figure 3. Ovarian Ultrasound |
|
|
The first image shows the peripheral distribution of the hypoechoic ovarian follicles, a common finding in ovarian torsion. The second image depicts a longitundinal view of the bladder, uterus, and posterior cul-de-sac. |
|
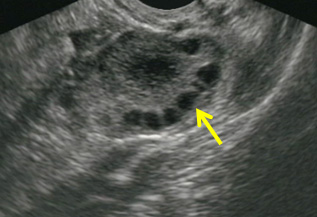 |
 |
Special consideration must be made with both ovarian and testicular torsion, as these are diagnoses that cannot be excluded with ultrasound alone. A high degree of suspicion for either ovarian or testicular torsion, even with a negative ultrasound, warrants a consultation with a gynecologist or urologist, respectively. While point-of-care ultrasound can serve as an adjunct to a more rapid diagnosis, emergency providers must not rely solely on a negative study to rule out these pathologies if the clinical presentation is supportive of the diagnosis.
Testicular Torsion: Pain and Suffering
A previously healthy, fully immunized 9-year-old male has experienced a two-hour history of acute onset of right-sided scrotal pain and nausea that began during play. No history of trauma is mentioned. He appears uncomfortable during the exam and has right scrotal tenderness. The cremasteric reflex is difficult to elicit on the right, and there is concern for a high-riding right testicle. (See Table 1.)
There is a broad differential diagnosis for acute scrotal pain in children. Epididymitis and torsion of the testicle, appendix testis, and appendix epididymis are common culprits, with varying levels of morbidity (i.e., testicular loss). Testicular torsion is a urological emergency that accounts for approximately 23% of acute scrotums in boys and requires a prompt and accurate diagnosis to guide management to promote testicular salvage.43,44 Torsion occurs when the testis twists around the vascular pedicle, also known as the spermatic cord. Ultrasound is the main diagnostic modality used to differentiate between various scrotal pathologies.45 (See Figure 4.)
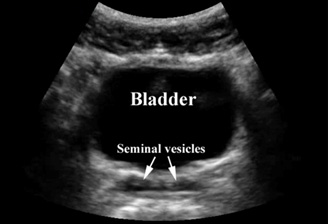
Figure 4. Testicular Torsion |
|
|
The first color Doppler image illustrates compromised vascular flow observed in testicular torsion. The second image of the male pelvis demonstrates the bladder and seminal vesicles in the transverse view. |
|
 |
 |
Normally, the testes appear as symmetric, oval-shaped, and homogenous structures within the scrotum. The testes are separated by a highly reflective raphe. The tunica albuginea that encases each testicle is easily differentiated from the surrounding structures because of its hyperechoic appearance. The mediastinum testis, rete testis, appendix testis, and the epididymis should be identified. The mediastinum testis can be seen as a linear, thin echogenic band traversing the testis near the midline, whereas the nearby rete testis can be visualized as a collection of hypoechoic tubular structures. The isoechoic oval-shaped appendix testis lies between the epididymis and testis. The epididymis is a curved structure that extends over the posterior border of the testicle.46-49 The head of the epididymis often appears isoechoic or slightly hyperechoic or hypoechoic compared to the testicle, while the body and tail tend to be isoechoic.50 The spermatic cord is highly echogenic and can be found within the testicle extending into the inguinal canal.46-49
Always perform genital exams with a chaperone present. Prior to examination, ensure that the patient is comfortable. Placing a towel under the scrotum provides elevation and support. Use a high-frequency, linear transducer. Ask the patient to locate the point of maximal pain. First, examine both testicles side-by-side in the transverse view to document lie and obtain both static and real-time color Doppler images. Each testis should be scanned in both the longitudinal and transverse orientation. Measure each testicle in three dimensions to calculate the testis volume. Scan the unaffected testicle first to provide a normal reference point to compare against the symptomatic testicle.46,47,51
Assess the flow pattern of each testis using color or power Doppler (calculate a testicular arterial resistive index if possible). Power Doppler often is preferred over color Doppler because of its greater sensitivity.46 Scan the testis thoroughly again in multiple orientations; note the echotexture of the normal testicle, as this should be compared against the abnormal side. Identify and evaluate the orientation and echogenicity of the epididymis. In the longitudinal plane, follow the spermatic cord from its origin through the inguinal canal to the internal ring. Furthermore, survey the testicle and extra-testicular space for additional pathology or incidental findings (e.g., hydrocele, mass, appendages.)46,47,51 Repeat the exam on the affected side documenting any pathology.
Impedance of the venous, arterial, and lymphatic system of both the testis and epididymis may develop. Consequently, this process can lead to ischemia, infarction, and eventually necrosis of the testis, which translates into highly variable sonographic findings.52 Although some studies have demonstrated that color Doppler ultrasound has an ability to detect testicular torsion with a 96-100% sensitivity and 75-95% specificity,53-55 there is evidence in the literature to suggest that color Doppler waveform should not be the sole determinant of the presence or absence of torsion, particularly in the setting of early torsion or partial torsion in which testicular flow still may be present, leading to false-negative results.43,53-62 In the case of complete torsion, arterial and venous flow often are absent or reduced.46,63
The torsed testicle may assume a heterogeneous and hypoechoic or hyperechoic appearance that results from arterial insufficiency and subsequent ischemia.46,64 Concomitantly, the testicle may become enlarged secondary to vascular congestion, and a significant size discrepancy between the affected and unaffected testicle may be notable. A healthy testis typically is positioned in the vertical orientation, while a horizontal or oblique lie is a concerning feature.65 Scrotal edema and reactive hydroceles comprised of anechoic fluid on ultrasound may occur.64
Abnormal morphology and displacement of the epididymis, and/or an enlarged, rotated, redundant, or an abnormal echo texture or compromised vascular flow of the spermatic cord suggests torsion.62,66 The classic “whirlpool sign” signifies a twisting of the spermatic cord creating a spiral-like pattern.67-69 A recent systematic review and meta-analysis found the “whirlpool sign” to be highly prognostic for torsion of the testicle and spermatic cord in children, with the exception of neonates.49 Unfortunately, the “whirlpool sign” is not reliably observed in all cases.70
Small Bowel Obstruction: Blocks Are Not Fun for Children
A 6-year-old female with a past surgical history of appendectomy reports a three-day history of paroxysmal abdominal pain. She has had several episodes of nonbilious emesis. Multiple children have been ill at her school recently. While her symptoms easily could be attributed to gastroenteritis or even constipation, it is important to include small bowel obstruction (SBO) as part of the differential diagnosis. Fifteen percent of hospital admissions for acute abdominal pain are attributable to SBO.71 The common culprits of SBO in children include intestinal adhesions, Meckel’s diverticulum, and internal hernia.72 If not recognized in a timely manner, the complications of SBO, such as ischemia, necrosis, and perforation, could result in significant morbidity and mortality.73
Ultrasound is valuable in the diagnosis of SBO. Most studies in the literature primarily have focused on the adult population, while the existing data in the pediatric population are limited.74,75 Although the diagnosis of SBO occurs less frequently in children compared to adults, the incorporation of point-of-care ultrasound into the assessment of children who present with abdominal pain, vomiting, and/or abdominal distention to identify SBO should be considered. Plain abdominal films often are nondiagnostic and have been found to be inferior to ultrasound in the evaluation of SBO, despite the use of X-ray as a standard initial imaging technique.76-78 X-ray has a sensitivity of 46% and specificity of 67% for SBO.78
In a systematic review and meta-analysis, Gottlieb et al concluded that ultrasound was 92% sensitive and 96% specific for the diagnosis of SBO.74 Although the authors did not exclude the pediatric population from this study, the mean age of participants was 50 years, which raises concerns about the generalizability of using ultrasound to diagnose SBO in children. However, several clinical cases have demonstrated the successful use of point-of-care ultrasound in the ED to diagnose SBO in children.75 Emergency medicine residents are able to diagnose SBO accurately using point-of-care ultrasound, comparable with that of radiology residents, after a brief training period.76,78
Both phase array and curvilinear probes are acceptable to evaluate for SBO.78 The patient should be in the supine position. Scan in the colic gutters, epigastrium, and suprapubic region, observing for fluid-filled dilated bowel, abnormal peristalsis, and collapse of the colonic lumen.76,78 (See Figure 5.) A dilated bowel extending over three bowel loop segments that measures ≥ 25 mm in the jejunum and ≥ 15 mm in the ileum has a sensitivity of 91-94% and specificity of 84-94% for SBO.76,78 The intestinal fold pattern and location of dilated loops should aid in the discernment of the location of the bowel obstruction.76 The jejunum has prominent and numerous valvulae conniventes, also known as plicae circulares, which decrease substantially in the ileum. Abnormal peristalsis has been associated with SBO.78
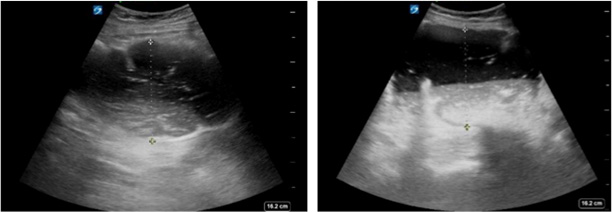
Figure 5. Small Bowel Obstruction |
|
Dilated loops of fluid-filled bowel present in small bowel obstruction. |
 |
Frequently, abnormal peristalsis is visualized in SBO and has been described as “to-and-fro,” “bounce-back,” or “back and forth” movements with echoes present within the fluid-filled bowel.76 Increased or decreased to absent peristalsis also has been reported in SBO.75,76,78 Decreased peristalsis alone has a sensitivity of 27%, while increased peristalsis has a reported sensitivity of 72%.76,78 Combining both the presence of dilated bowel and decreased peristalsis increased the sensitivity to 94%, improving the sensitivity of either finding alone.78 A collapsed colon is 85% sensitive for SBO.76 Additionally, the presence of ascites in the setting of SBO is suggestive of intestinal strangulation, a complication of SBO with high morbidity.72 An increase in bowel wall thickness and an increase in the intestinal content may be found as well.71
Pediatric Cholecystitis: A Condition Gallingly on the Rise
A 6-year-old, afebrile male is being evaluated for a one-day history of abdominal pain. His mother denies fever, vomiting, diarrhea, constipation, dysuria, or contact with sick individuals. His exam is remarkable for voluntary right-sided abdominal guarding. Labs are normal. After a period of observation and a normal ultrasound of the appendix, his abdominal tenderness remains concerning. Although cholecystitis primarily is considered an adult disease, the prevalence of cholecystitis and biliary tract pathology in the pediatric population has been on the rise.79 The etiology of the increase is not fully understood; however, the rising childhood obesity rate and more widespread use of ultrasound, leading to a higher rate of diagnosis, have been proposed as factors.80
Children of all ages are subject to gallbladder disease.80 Although many cases of pediatric cholelithiasis are idiopathic, there is a wide spectrum of risk factors, including obesity, pregnancy, hemolytic disease, prolonged total parenteral nutrition, and use of oral contraceptives.81-86 In addition, chronic cholecystitis and acute acalculous cholecystitis (ACC) account for a subset of pediatric inflammatory gallbladder disease.87,88 ACC can result in significant morbidity and mortality, and it has been found to be an unfortunate complication of the course of various illnesses, including infectious disease (e.g., hepatitis A, Epstein-Barr virus, human herpes virus-type 6), systemic disease (e.g., systemic lupus erythematosus, Kawasaki disease, cystic fibrosis), critical illness (e.g., sepsis), trauma, and the postoperative period.88-94 ACC can occur in otherwise healthy children.95 In contrast, hemoglobinopathies, such as sickle cell disease, predispose children to gallstones from disproportionate bilirubin production secondary to recurrent hemolysis.85 Ultrasound is the favored imaging modality to identify gallbladder pathology. Detection of cholecystitis by ultrasound by emergency medicine physicians shares similar accuracy to that of radiologists.96
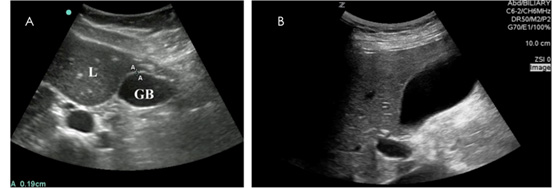
The gallbladder is a hypoechoic, oblong structure surrounded by a thin echogenic wall.93 The normal anterior gallbladder wall thickness is < 3 mm. (See Figure 6.) Gallbladder wall thickening alone is not definitive for cholecystitis; wall thickening can be manifested in multiple other disease processes, including bacterial or viral infections (e.g., hepatitis A), systemic disease (e.g., renal failure, heart failure, liver disease, or systemic lupus erythematosus), and pancreatitis.97-99 The normal common bile duct has a diameter up to 6 mm in older children and adults, while the standard is considered to be < 3.3 mm in children younger than 13 years of age and < 1.2 mm in children younger than 3 months of age.100
Figure 6. Transverse and Longitudinal Views of a Normal Gallbladder |
 |
Sonographically, cholelithiasis will appear as hyperechoic foci within the gallbladder or ducts.93 Most gallstones will produce a posterior acoustic shadowing artifact, but stones < 5 mm may not.93 The wall-echo-shadow complex, or WES sign, is produced when the gallbladder is collapsed and filled with a large or multiple stones.101 The WES complex is composed of the highly echogenic, thin curvilinear wall of the gallbladder with a subjacent echo formed from the reflective gallstones with the accompanying underlying shadowing.101 Pericholecystic fluid and gallbladder distention are additional secondary findings. (See Figure 7.)
Figure 7. Gallbladder |
|
A. Gallbladder wall thickening. B. Pericholecystic fluid. C. Stone in the neck of the gallbladder. D. Multiple stones in the gallbladder. |
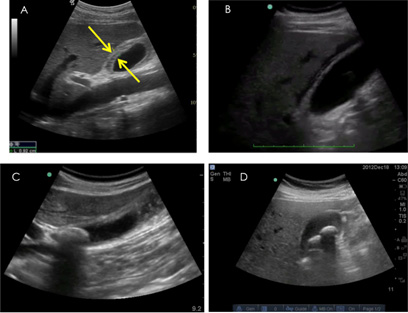 |
A positive sonographic Murphy sign, a key diagnostic marker, occurs when pain is elicited as downward pressure is placed directly over the gallbladder by the ultrasound probe.93 The sonographic Murphy sign detected by emergency medicine physicians has been shown to have superior sensitivity for diagnosing acute cholecystitis compared to a formal ultrasound by radiology.102 Tsai et al suggested that ultrasound findings described in acute cholecystitis have lower sensitivities and positive predictive values in the pediatric population compared to those reported in adults.103
Perform the examination with the patient in the supine position, starting in either the sagittal or transverse orientation with either the curvilinear or phase array probe.80,93 Ideally, the exam should be performed after the patient has fasted to avoid contraction of the gallbladder, which naturally occurs after meal consumption. Several different techniques can be used to locate the gallbladder, including the “subcostal sweep,” “X-minus 7,” and “flattening the probe.” In the subcostal approach, the probe is oriented in the longitudinal direction with the probe indicator toward the patient’s head subjacent to the xiphoid process. The probe then is swept inferiorly and laterally along the subcostal margin. Alternatively, the probe is placed 7 cm to the right of the xiphoid process in the closest intercostal space in the “X-minus 7” approach. Furthermore, it may be helpful to flatten and direct the probe subcostally while aiming toward the patient’s shoulder and fanning anteriorly and posteriorly.104 Rotating the patient in the left lateral decubitus position, scanning in the oblique plane, and having the patient take a deep breath can improve visualization of the gallbladder.80 The main lobar fissure and portal triad are useful landmarks.80
Obtain images in the longitudinal, oblique, and transverse plane to evaluate the entire gallbladder and portal triad. Scan through the entire gallbladder from the fundus to the narrow neck. The anterior gallbladder wall should be measured from outer-to-outer wall in the transverse plane. Follow the echogenic line of the main hepatic fissure, which extends from the neck of the gallbladder to the portal triad. The gallbladder, hepatic fissure, and portal triad resemble an “exclamation point.” The portal triad consists of the portal vein, common bile duct (CBD), and the hepatic artery. The CBD has a “double-barrel shot-gun” or “tram track” appearance. The CBD is located parallel and anterior to the right of the portal vein in the oblique long axis. The diameter of the CBD should be measured from inner-to-inner wall.93 Color Doppler is beneficial to differentiate the CBD from the hepatic artery, which lies anterior and to the left of the portal vein.93
CONCLUSION
Ultrasound is instrumental in the diagnosis of many pediatric abdominal diseases and may be used to guide patient care. Emergency medicine physicians should have a foundation in the basics of point-of-care ultrasound. Being adept at using ultrasound is an invaluable skill in expediting patient care by providing a quick diagnosis and disposition while avoiding ionizing radiation. Although ultrasound is a very effective and efficient means to diagnose pediatric abdominal pathologies, providers must be attentive to cautions and limitations for each exam. While the diagnostic parameters in this article are widely accepted in the literature, it is important to verify the values used with the guidelines of each institution’s radiology department.
The authors would like to thank Geoffrey E. Hayden, MD, FACEP, for his assistance in creating some of the ultrasound images.
REFERENCES
- The ALARA (as low as reasonably achievable) concept in pediatric CT intelligent dose reduction. Multidisciplinary conference organized by the Society of Pediatric Radiology. August 18-19, 2001. Pediatr Radiol 2002;32:217-313.
- Thamburaj R, Sivitz A. Does the use of bedside pelvic ultrasound decrease length of stay in the emergency department? Pediatr Emerg Care 2013;29:67-70.
- Hall MK, Taylor RA, Luty S, et al. Impact of point-of-care ultrasonography on ED time to disposition for patients with nontraumatic shock. Am J Emerg Med 2016;34:1022-1030.
- Park YH, Jung RB, Lee YG, et al. Does the use of bedside ultrasonography reduce emergency department length of stay for patients with renal colic?: A pilot study. Clin Exp Emerg Med 2016;3:197-203.
- Kairam N, Allegra JR, Eskin B. Rise in emergency department visits of pediatric patients for renal colic from 1999 to 2008. Pediatr Emerg Care 2013;29:462-464.
- Bush NC, Xu L, Brown BJ, et al. Hospitalizations for pediatric stone disease in United States, 2002-2007. J Urol 2010;183:1151-1156.
- Moore CL, Scoutt L. Sonography first for acute flank pain? J Ultrasound Med 2012;31:1703-1711.
- Ng C, Tsung JW. Avoiding computed tomography scans by using point-of-care ultrasound when evaluating suspected pediatric renal colic. J Emerg Med 2015;49:165-171.
- Dalziel PJ, Noble VE. Bedside ultrasound and the assessment of renal colic: A review. Emerg Med J 2013;30:3-8.
- Niemann T, Kollmann T, Bongartz G. Diagnostic performance of low-dose CT for the detection of urolithiasis: A meta-analysis. AJR Am J Roentgenol 2008;191:396-401.
- Smith RC, Verga M, McCarthy S, Rosenfield AT. Diagnosis of acute flank pain: Value of unenhanced helical CT. AJR Am J Roentgenol 1996;166:97-101.
- Fielding JR, Steele G, Fox LA, et al. Spiral computerized tomography in the evaluation of acute flank pain: A replacement for excretory urography. J Urol 1997;157:2071-2073.
- Smith-Bindman R, Aubin C, Bailitz J, et al. Ultrasonography versus computed tomography for suspected nephrolithiasis. N Engl J Med 2014;371:1100-1110.
- Melvin JE, Ost MC, Marin JR. Hydro-nephrosis from ureteropelvic junction obstruction discovered on point-of-care ultrasound in patients with trauma. Pediatr Emerg Care 2018;34:365-367.
- Seif D, Swadron SP. Chapter 12. Renal. In: Ma OJ, Mateer JR, Reardon RF, Joing SA, eds. Ma and Mateer’s Emergency Ultrasound, 3e. New York: The McGraw-Hill Companies; 2014.
- Hansen KL, Nielsen MB, Ewertsen C. Ultrasonography of the kidney: A pictorial review. Diagnostics (Basel) 2015;6: pii: E2. doi: 10.3390/diagnostics6010002.
- Dinkel E, Ertel M, Dittrich M, et al. Kidney size in childhood. Sonographical growth charts for kidney length and volume. Pediatr Radiol 1985;15:38-43.
- Fernbach SK, Maizels M, Conway JJ. Ultrasound grading of hydronephrosis: Introduction to the system used by the Society for Fetal Urology. Pediatr Radiol 1993;23:478-480.
- Aytac SK, Ozcan H. Effect of color Doppler system on the twinkling sign associated with urinary tract calculi. J Clin Ultrasound 1999;27:433-439.
- Mitterberger M, Aigner F, Pallwein L, et al. Sonographic detection of renal and ureteral stones. Value of the twinkling sign. Int Braz J Urol 2009;35:532-539; discussion 540-531.
- Park SJ, Yi BH, Lee HK, et al. Evaluation of patients with suspected ureteral calculi using sonography as an initial diagnostic tool: How can we improve diagnostic accuracy? J Ultrasound Med 2008;27:1441-1450.
- Ripolles T, Martinez-Perez MJ, Vizuete J, et al. Sonographic diagnosis of symptomatic ureteral calculi: Usefulness of the twinkling artifact. Abdom Imaging 2013;38:863-869.
- Jandaghi AB, Falahatkar S, Alizadeh A, et al. Assessment of ureterovesical jet dynamics in obstructed ureter by urinary stone with color Doppler and duplex Doppler examinations. Urolithiasis 2013;41:159-163.
- Ongun S, Teken A, Yilmaz O, Suleyman S. Relationship between ureteral jet flow, visual analogue scale, and ureteral stone size. Urology 2017;104:36-39.
- de Bessa J Jr., Denes FT, Chammas MC, et al. Diagnostic accuracy of color Doppler sonographic study of the ureteric jets in evaluation of hydronephrosis. J Pediatr Urol 2008;4:113-117.
- Chang HC, Bhatt S, Dogra VS. Pearls and pitfalls in diagnosis of ovarian torsion. Radiographics 2008;28:1355-1368.
- Riccabona M, Lobo ML, Ording-Muller LS, et al. European Society of Paediatric Radiology abdominal imaging task force recommendations in paediatric uroradiology, part IX: Imaging in anorectal and cloacal malformation, imaging in childhood ovarian torsion, and efforts in standardising paediatric uroradiology terminology. Pediatr Radiol 2017;47:1369-1380.
- Jourjon R, Morel B, Irtan S, et al. Analysis of clinical and ultrasound determinants of adnexal torsion in children and adolescents. J Pediatr Adolesc Gynecol 2017;30:582-590.
- Melcer Y, Maymon R, Pekar-Zlotin M, et al. Clinical and sonographic predictors of adnexal torsion in pediatric and adolescent patients. J Pediatr Surg 2018;53:1396-1398.
- Walker SK, Lal DR, Boyd KP, Sato TT. Management of pediatric ovarian torsion: Evidence of follicular development after ovarian preservation. Surgery 2018;163:547-552.
- Bolli P, Schadelin S, Holland-Cunz S, Zimmermann P. Ovarian torsion in children: Development of a predictive score. Medicine (Baltimore) 2017;96:e8299.
- Guthrie BD, Adler MD, Powell EC. Incidence and trends of pediatric ovarian torsion hospitalizations in the United States, 2000-2006. Pediatrics 2010;125:532-538.
- Schuh AM, Klein EJ, Allred RJ, et al. Pediatric adnexal torsion: Not just a postmenarchal problem. J Emerg Med 2017;52:169-175.
- Servaes S, Zurakowski D, Laufer MR, et al. Sonographic findings of ovarian torsion in children. Pediatr Radiol 2007;37:446-451.
- Dasgupta R, Renaud E, Goldin AB, et al. Ovarian torsion in pediatric and adolescent patients: A systematic review. J Pediatr Surg 2018;53:1387-1391.
- Melcer Y, Maymon R, Pekar-Zlotin M, et al. Does she have adnexal torsion? Prediction of adnexal torsion in reproductive age women. Arch Gynecol Obstet 2018;297:685-690.
- Linam LE, Darolia R, Naffaa LN, et al. US findings of adnexal torsion in children and adolescents: Size really does matter. Pediatr Radiol 2007;37:1013-1019.
- Ngo AV, Otjen JP, Parisi MT, et al. Pediatric ovarian torsion: A pictorial review. Pediatr Radiol 2015;45:1845-1855; quiz 1842-1844.
- Lourenco AP, Swenson D, Tubbs RJ, Lazarus E. Ovarian and tubal torsion: Imaging findings on US, CT, and MRI. Emerg Radiol 2014;21:179-187.
- Navve D, Hershkovitz R, Zetounie E, et al. Medial or lateral location of the whirlpool sign in adnexal torsion: Clinical importance. J Ultrasound Med 2013;32:1631-1634.
- Valsky DV, Esh-Broder E, Cohen SM, et al. Added value of the gray-scale whirlpool sign in the diagnosis of adnexal torsion. Ultrasound Obstet Gynecol 2010;36:630-634.
- Lee EJ, Kwon HC, Joo HJ, et al. Diagnosis of ovarian torsion with color Doppler sonography: Depiction of twisted vascular pedicle. J Ultrasound Med 1998;17:83-89.
- Kalfa N, Veyrac C, Lopez M, et al. Multicenter assessment of ultrasound of the spermatic cord in children with acute scrotum. J Urol 2007;177:297-301; discussion 301.
- Cattolica EV, Karol JB, Rankin KN, Klein RS. High testicular salvage rate in torsion of the spermatic cord. J Urol 1982;128:66-68.
- Boettcher M, Bergholz R, Krebs TF, et al. Differentiation of epididymitis and appendix testis torsion by clinical and ultrasound signs in children. Urology 2013;82:899-904.
- Wright S, Hoffmann B. Emergency ultrasound of acute scrotal pain. Eur J Emerg Med 2015;22:2-9.
- Kuhn AL, Scortegagna E, Nowitzki KM, Kim YH. Ultrasonography of the scrotum in adults. Ultrasonography 2016;35:180-197.
- Boettcher M, Krebs T, Bergholz R, et al. Clinical and sonographic features predict testicular torsion in children: A prospective study. BJU Int 2013;112:1201-1206.
- McDowall J, Adam A, Gerber L, et al. The ultrasonographic “whirlpool sign” in testicular torsion: Valuable tool or waste of valuable time? A systematic review and meta-analysis. Emerg Radiol 2018;25:281-292.
- Alkhori NA, Barth RA. Pediatric scrotal ultrasound: Review and update. Pediatr Radiol 2017;47:1125-1133.
- Bandarkar AN, Blask AR. Testicular torsion with preserved flow: Key sonographic features and value-added approach to diagnosis. Pediatr Radiol 2018;48:735-744.
- Basta AM, Courtier J, Phelps A, et al. Scrotal swelling in the neonate. J Ultrasound Med 2015;34:495-505.
- Altinkilic B, Pilatz A, Weidner W. Detection of normal intratesticular perfusion using color coded duplex sonography obviates need for scrotal exploration in patients with suspected testicular torsion. J Urol 2013;189:1853-1858.
- Waldert M, Klatte T, Schmidbauer J, et al. Color Doppler sonography reliably identifies testicular torsion in boys. Urology 2010;75:1170-1174.
- Pepe P, Panella P, Pennisi M, Aragona F. Does color Doppler sonography improve the clinical assessment of patients with acute scrotum? Eur J Radiol 2006;60:120-124.
- Kalfa N, Veyrac C, Baud C, et al. Ultrasonography of the spermatic cord in children with testicular torsion: Impact on the surgical strategy. J Urol 2004;172(4 Pt 2):1692-1695; discussion 1695.
- Bader TR, Kammerhuber F, Herneth AM. Testicular blood flow in boys as assessed at color Doppler and power Doppler sonography. Radiology 1997;202:559-564.
- Sanelli PC, Burke BJ, Lee L. Color and spectral doppler sonography of partial torsion of the spermatic cord. AJR Am J Roentgenol 1999;172:49-51.
- Lee FT, Jr, Winter DB, Madsen FA, et al. Conventional color Doppler velocity sonography versus color Doppler energy sonography for the diagnosis of acute experimental torsion of the spermatic cord. AJR Am J Roentgenol 1996;167:785-790.
- Karmazyn B, Steinberg R, Kornreich L, et al. Clinical and sonographic criteria of acute scrotum in children: A retrospective study of 172 boys. Pediatr Radiol 2005;35:302-310.
- Dogra VS, Gottlieb RH, Oka M, Rubens DJ. Sonography of the scrotum. Radiology 2003;227:18-36.
- Galina P, Dermentzoglou V, Baltogiannis N, Zarifi M. Sonographic appearances of the epididymis in boys with acute testicular torsion but preserved testicular blood flow on color Doppler. Pediatr Radiol 2015;45:1661-1671.
- Fujita N, Tambo M, Okegawa T, et al. Distinguishing testicular torsion from torsion of the appendix testis by clinical features and signs in patients with acute scrotum. Res Rep Urol 2017;9:169-174.
- Sung EK, Setty BN, Castro-Aragon I. Sonography of the pediatric scrotum: Emphasis on the Ts—torsion, trauma, and tumors. AJR Am J Roentgenol 2012;198:996-1003.
- Eaton SH, Cendron MA, Estrada CR, et al. Intermittent testicular torsion: Diagnostic features and management outcomes. J Urol 2005;174(4 Pt 2):1532-1535; discussion 1535.
- Arce JD, Cortes M, Vargas JC. Sonographic diagnosis of acute spermatic cord torsion. Rotation of the cord: A key to the diagnosis. Pediatr Radiol 2002;32:485-491.
- Prando D. Torsion of the spermatic cord: The main gray-scale and doppler sonographic signs. Abdom Imaging 2009;34:648-661.
- Baud C, Veyrac C, Couture A, Ferran JL. Spiral twist of the spermatic cord: A reliable sign of testicular torsion. Pediatr Radiol 1998;28:950-954.
- Vijayaraghavan SB. Sonographic differential diagnosis of acute scrotum: Real-time whirlpool sign, a key sign of torsion. J Ultrasound Med 2006;25:563-574.
- Munden MM, Williams JL, Zhang W, et al. Intermittent testicular torsion in the pediatric patient: Sonographic indicators of a difficult diagnosis. AJR Am J Roentgenol 2013;201:912-918.
- Guttman J, Stone MB, Kimberly HH, Rempell JS. Point-of-care ultrasonography for the diagnosis of small bowel obstruction in the emergency department. CJEM 2015;17:206-209.
- Chang YJ, Yan DC, Lai JY, et al. Strangulated small bowel obstruction in children. J Pediatr Surg 2017;52:1313-1317.
- Paulson EK, Thompson WM. Review of small-bowel obstruction: The diagnosis and when to worry. Radiology 2015;275:332-342.
- Gottlieb M, Peksa GD, Pandurangadu AV, et al. Utilization of ultrasound for the evaluation of small bowel obstruction: A systematic review and meta-analysis. Am J Emerg Med 2018;36:234-242.
- James V, Alsani FS, Fregonas C, et al. Point-of-care ultrasound in pediatric small bowel obstruction: An ED case series. Am J Emerg Med 2016;34:2464.e1-2464.e2. doi: 10.1016/j.ajem.2016.06.021.
- Unluer EE, Yavasi O, Eroglu O, et al. Ultrasonography by emergency medicine and radiology residents for the diagnosis of small bowel obstruction. Eur J Emerg Med 2010;17:260-264.
- Taylor MR, Lalani N. Adult small bowel obstruction. Acad Emerg Med 2013;20:528-544.
- Jang TB, Schindler D, Kaji AH. Bedside ultrasonography for the detection of small bowel obstruction in the emergency department. Emerg Med J 2011;28:676-678.
- Bailey PV, Connors RH, Tracy TF, Jr, et al. Changing spectrum of cholelithiasis and cholecystitis in infants and children. Am J Surg 1989;158:585-588.
- Tsung JW, Raio CC, Ramirez-Schrempp D, Blaivas M. Point-of-care ultrasound diagnosis of pediatric cholecystitis in the ED. Am J Emerg Med 2010;28:338-342.
- Walker SK, Maki AC, Cannon RM, et al. Etiology and incidence of pediatric gallbladder disease. Surgery 2013;154:927-931; discussion 931-923.
- Mehta S, Lopez ME, Chumpitazi BP, et al. Clinical characteristics and risk factors for symptomatic pediatric gallbladder disease. Pediatrics 2012;129:e82-88.
- Komura J, Yano H, Tanaka Y, Tsuru T. Increased incidence of cholestasis during total parenteral nutrition in children — factors affecting stone formation. Kurume Med J 1993;40:7-11.
- Clements RH, Holcomb GW, 3rd. Laparoscopic cholecystectomy. Curr Opin Pediatr 1998;10:310-314.
- Suell MN, Horton TM, Dishop MK, et al. Outcomes for children with gallbladder abnormalities and sickle cell disease. J Pediatr 2004;145:617-621.
- Murphy PB, Vogt KN, Winick-Ng J, et al. The increasing incidence of gallbladder disease in children: A 20 year perspective. J Pediatr Surg 2016;51:748-752.
- P Blackwood B, Grabowski J. Chronic cholecystitis in the pediatric population: An underappreciated disease process. Gastroenterol Hepatol Bed Bench 2017;10:125-130.
- Yi DY, Chang EJ, Kim JY, et al. Age, predisposing diseases, and ultrasonographic findings in determining clinical outcome of acute acalculous inflammatory gallbladder diseases in children. J Korean Med Sci 2016;31:1617-1623.
- Tsakayannis DE, Kozakewich HP, Lillehei CW. Acalculous cholecystitis in children. J Pediatr Surg 1996;31:127-130; discussion 130-121.
- Imamoglu M, Sarihan H, Sari A, Ahmetoglu A. Acute acalculous cholecystitis in children: Diagnosis and treatment. J Pediatr Surg 2002;37:36-39.
- Gora-Gebka M, Liberek A, Bako W, et al. Acute acalculous cholecystitis of viral etiology — a rare condition in children? J Pediatr Surg 2008;43:e25-27.
- Gomes MM, Antunes H, Lobo AL, et al. Acute alithiasic cholecystitis and human herpes virus type-6 infection: First case. Case Rep Pediatr 2016;2016:9130673.
- Shihabuddin B, Sivitz A. Acute acalculous cholecystitis in a 10-year-old girl with cystic fibrosis. Pediatr Emerg Care 2013;29:117-121.
- Lu YA, Chiu CH, Kong MS, et al. Risk factors for poor outcomes of children with acute acalculous cholecystitis. Pediatr Neonatol 2017;58:497-503.
- McNaughton C, Morrow S, Cico SJ. Acalculous cholecystitis in a healthy 4-year-old boy. Am J Emerg Med 2011;29:962.e1-2. doi: 10.1016/j.ajem.2010.08.022.
- Summers SM, Scruggs W, Menchine MD, et al. A prospective evaluation of emergency department bedside ultrasonography for the detection of acute cholecystitis. Ann Emerg Med 2010;56:114-122.
- Basiratnia M, Vasei M, Bahador A, et al. Acute acalculous cholecystitis in a child with systemic lupus erythematosus. Pediatr Nephrol 2006;21:873-876.
- Lee JH, No YE, Lee YJ, et al. Acalculous diffuse gallbladder wall thickening in children. Pediatr Gastroenterol Hepatol Nutr 2014;17:98-103.
- Sanches BF, Martins T, Santos MJ, Azeredo P. Acute acalculous cholecystitis in a patient with juvenile dermatomyositis. BMJ Case Rep 2014;2014; pii: bcr2014205066. doi: 10.1136/bcr-2014-205066.
- Hernanz-Schulman M, Ambrosino MM, et al. Common bile duct in children: Sonographic dimensions. Radiology 1995;195:193-195.
- George N, Dawkins A, DiSantis D. The wall-echo-shadow (WES) sign. Abdom Imaging 2015;40:2903.
- Kendall JL, Shimp RJ. Performance and interpretation of focused right upper quadrant ultrasound by emergency physicians. J Emerg Med 2001;21:7-13.
- Tsai J, Sulkowski JP, Cooper JN, et al. Sensitivity and predictive value of ultrasound in pediatric cholecystitis. J Surg Res 2013;184:378-382.
- Fox JC, Scruggs W. Ultrasound Guide for Emergency Physicians: Biliary Ultrasound. 2008. Available at: https://www.acep.org/sonoguide/biliary.html. Accessed March 30, 2018.
Ultrasound is evolving rapidly as the ideal imaging modality for many common pediatric complaints. In the second part of this series, the authors discuss point-of-care use of ultrasound for concerns regarding the kidneys, ovaries, testicles, gallbladder, and small bowel obstruction. The ability to make critical diagnoses safely and rapidly with ultrasound is an invaluable clinical tool to facilitate and improve pediatric care.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
