Chronic Abdominal Pain Disorders: Chronic Pancreatitis and Cyclic Vomiting Syndrome
August 1, 2018
Reprints
AUTHORS
Caleb P. Canders, MD, Department of Emergency Medicine, UCLA Ronald Reagan Medical Center, Los Angeles
Brian V. Truong, MD, Department of Emergency Medicine, UCLA Ronald Reagan Medical Center, Los Angeles
Kellie M. Kitamura, MD, Department of Emergency Medicine, UCLA Ronald Reagan Medical Center, Los Angeles
PEER REVIEWER
Steven M. Winograd, MD, Attending Physician, Mt. Sinai Queens Hospital Center, Assistant Clinical Professor of Emergency Medicine, Mt. Sinai Medical School, Jamaica Queens, New York
EXECUTIVE SUMMARY
- Causes of chronic pancreatitis include alcohol and/or tobacco use, pancreatic duct obstruction, heredity, or autoimmune disease.
- Complications of chronic pancreatitis include diabetes, steatorrhea, splanchnic vein thrombosis with gastric varices, pseudocysts, pancreatic duct stricture, and pancreatic cancer.
- Cyclic vomiting syndrome was described first in children. Many cases are associated with migraine, anxiety, and/or depression. More recently, there has been an increase seen with frequent use of cannabis.
- Acute symptoms often respond to treatment with sumatriptan. Patients with cyclic vomiting due to cannabis should abstain from its use. Others find relief with hot showers, use of capsaicin cream, haloperidol, or benzodiazepines.
Introduction
Disorders such as chronic pancreatitis and cyclic vomiting syndrome are characterized by chronic abdominal pain, nausea, and vomiting. The pain associated with these disorders often is refractory to analgesics, leaving emergency providers with the difficult task of relieving pain while simultaneously attempting to limit the use of opiates. In addition, some cases of chronic abdominal pain are secondary to substance abuse (e.g., alcohol in chronic pancreatitis, cannabis in cyclic vomiting syndrome), leading to frustration among some providers, who may perceive the disorders as self-inflicted. Moreover, the association of chronic abdominal pain disorders with comorbid psychiatric disorders, including anxiety and depression, can make the physician-patient interaction complex. Such challenges can lead to low patient satisfaction and medication noncompliance among patients and contribute to burnout among emergency providers.1,2
However, many of the perceptions of these pain disorders are misguided. For example, cyclic vomiting syndrome was recognized first in children, and the majority of cases are unrelated to substance use.3 Many non-opiate treatments have been shown to be effective at managing symptoms in patients. Therefore, it is important for emergency providers to understand the pathophysiology and management of chronic abdominal pain disorders. This article will review two common chronic abdominal pain disorders encountered in the ED: chronic pancreatitis and cyclic vomiting syndrome.
Chronic Pancreatitis
Epidemiology
Both acute and chronic pancreatitis are characterized by inflammation and injury to the pancreas, although the progression and outcomes of the two diseases are significantly different. Less than 20% of patients who experience an episode of acute pancreatitis will develop recurrent episodes, and less than 10% will develop chronic pancreatitis.4 “Recurrent acute pancreatitis” refers to separate episodes of pancreatitis due to an identifiable cause (e.g., gallstones) with a period of resolution between episodes.5 “Chronic pancreatitis,” in most consensus definitions, requires irreversible histologic or morphologic changes in the pancreas, usually with accompanying pain and long-term exocrine and endocrine dysfunction.5 Most patients with chronic pancreatitis will have at least one preceding episode of acute pancreatitis, although one-third of patients lack a history of acute pancreatitis.6 The histologic and morphologic changes needed to diagnose chronic pancreatitis can take months or years to develop, so it often is difficult to diagnose at an early stage.
In the United States, the incidence of chronic pancreatitis is four cases per 100,000 person-years. A higher incidence has been reported worldwide.7 Chronic pancreatitis is more common in men and African Americans and usually is diagnosed in the fifth or sixth decade of life, but it can develop at younger ages.5 Patients with chronic pancreatitis have high rates of healthcare utilization and medical costs, mainly because of the chronic pain associated with the disease and its complications.8 In the United States, chronic pancreatitis results in an aggregate cost of more than $150 million annually.9
Etiology
The pancreas has endocrine and exocrine functions. Its endocrine function involves secreting insulin and glucagon, and its exocrine function is to secrete digestive enzymes.10 In patients with chronic pancreatitis, there is repeated stress or injury to the pancreas, which leads to destruction of acinar cells and subsequent release of pancreatic enzymes into the interstitium of the pancreas. Eventually, this leads to necrosis and fibrosis of the organ.11,12 An episode of acute pancreatitis may or may not serve as the sentinel event in the development of chronic pancreatitis.13 Some common inciting factors are described in the following section.
Alcohol. Alcohol is the most common cause of chronic pancreatitis and accounts for 50% of cases.5 The non-esterified esters in alcohol induce acinar cell injury and inflammation.14 Cumulative exposure to alcohol, including the amount and duration of alcohol use, is the most important determinant for the development of chronic pancreatitis. Consuming four to five alcoholic drinks daily doubles the risk of developing chronic pancreatitis.15,16 Because of gender differences in alcohol absorption and metabolism, women have a higher risk of developing chronic pancreatitis at lower levels of alcohol intake.17,18 On average, it takes 18 years between the start of excess alcohol intake and the diagnosis of chronic pancreatitis.19 In patients with acute pancreatitis secondary to alcohol, 25-50% will develop recurrent pancreatitis and, among those patients, 42-80% will develop chronic pancreatitis.20,21 However, many patients who develop chronic pancreatitis secondary to alcohol lack a history of acute pancreatitis.22 Progression of established chronic pancreatitis can be halted or slowed if a patient stops drinking alcohol.5
Tobacco. Smoking tobacco increases the risk of developing chronic pancreatitis in a dose-dependent manner independently from alcohol, although there is a cumulative risk in patients who smoke and drink alcohol.23 Nicotine induces oxidative stress in the pancreas via several pathways, which damages acinar cells and leads to fibrosis.24 Smoking cessation has been shown to decrease disease progression in patients with established chronic pancreatitis.25
Pancreatic Duct Obstruction. Pancreatic duct obstruction, secondary to strictures, stones, or extrinsic compression, causes increased pressure in the pancreatic duct and can lead to fibrosis and chronic pancreatitis.26 Pancreatic duct strictures may form after an acute episode of gallstone-induced pancreatitis, endoscopic/surgical procedure, or blunt injury.5 Pancreatic ducts also can be blocked by stones (often the consequence of already established chronic pancreatitis) or extrinsic compression from a pancreatic or intestinal tumor.27,28 It is controversial whether pancreas divisum, a common and congenital anatomic variation of the pancreas, can cause pancreatic duct obstruction and subsequent chronic pancreatitis.5
Hereditary Chronic Pancreatitis. A number of genetic mutations, including PRSS1, CFTR, and SPINK, induce stress in the acinar cells and have been connected with development of chronic pancreatitis.14,29-34 Patients with hereditary pancreatitis usually experience episodes of acute pancreatitis in childhood and develop signs of chronic pancreatitis by the third decade of life.35
Chronic Autoimmune Pancreatitis. The most common type of autoimmune pancreatitis is associated with a rare inflammatory multiorgan syndrome known as immunoglobulin G4 (IgG4)-related inflammatory disease.7 Rather than presenting with abdominal pain, many patients with autoimmune pancreatitis initially present with painless jaundice.36 Often it can be difficult to distinguish chronic autoimmune pancreatitis from pancreatic cancer.37 Serologic tumor markers (e.g., Ca 19-9), imaging, and pancreatic biopsy may help to differentiate autoimmune pancreatitis from pancreatic cancer. In addition, unlike pancreatic cancer and most forms of chronic pancreatitis, autoimmune pancreatitis responds rapidly to steroids, and disease progression often can be halted.
Pathophysiology
In patients with chronic pancreatitis, oxidative stress either from exogenous (e.g., alcohol) or endogenous (e.g., genetic) sources induces injury to the pancreatic acinar cells. The cells release calcium and pancreatic enzymes, which cause destruction and fibrosis of the adjacent parenchyma. Subsequent inflammation also can cause pancreatic duct obstruction, leading to pancreatic ductal hypertension, resultant hypoperfusion, and further injury to pancreatic cells.7 Typically, this inflammation and fibrosis cause abdominal pain, although some patients may develop chronic pancreatitis without experiencing symptoms. Destruction of pancreatic cells leads to early endocrine dysfunction and the development of diabetes. Exocrine dysfunction develops later in the disease and presents with steatorrhea and diarrhea.38
Clinical Features
The clinical features of chronic pancreatitis vary depending on its etiology. Chronic pancreatitis secondary to alcohol, for example, typically has an aggressive course, whereas chronic pancreatitis due to other etiologies has a more indolent course.
Abdominal Pain. Abdominal pain is the presenting symptom in 90% of patients with chronic pancreatitis.39 (See Table 1.) A minority of patients will have no pain. The abdominal pain usually is epigastric, worse with meals, and partially relieved with leaning forward. The pain associated with chronic pancreatitis is multifactorial in nature and may be due to inflammation of the parenchymal nerve sheaths, increased pressure in the pancreatic ducts, tension in the pancreatic capsule, or complications of the disease, such as gastric ulcers.40
Diabetes. Endocrine insufficiency develops in 80% of patients with chronic pancreatitis, leading to pancreatogenous or “type 3c” diabetes.41 Approximately 5% of all diabetes is attributed to type 3c diabetes, which is diagnosed by excluding other sources of diabetes and fulfilling criteria for both chronic pancreatitis and diabetes.42 Types 1 and 2 diabetes are not independent risk factors for chronic pancreatitis. Patients with type 3c diabetes are at high risk of developing episodes of both hyperglycemia and hypoglycemia, due to concomitant loss of glucagon and other glucose-regulating hormones that are produced by the pancreas.7
Table 1. Complications of Chronic Pancreatitis |
|
Steatorrhea. Steatorrhea, defined as greater than 7 grams of fat content in the stool per day, occurs if lipase levels drop to less than 10% of normal.43 It is usually a late manifestation of chronic pancreatitis and can result in deficiencies of fat-soluble vitamins (vitamins A, D, E, and K) and malnutrition.
Splanchnic Vein Thrombosis. Splanchnic vein thrombosis (most commonly the splenic vein) can occur in both acute and chronic pancreatitis. Most patients are asymptomatic, although some develop gastric varices and subsequent gastrointestinal bleeding. Splanchnic vein thrombosis can be diagnosed with abdominal ultrasound or computed tomography with intravenous contrast. Anticoagulation is not started, although elective embolization of the spleen may be performed to prevent the development of gastric varices.44
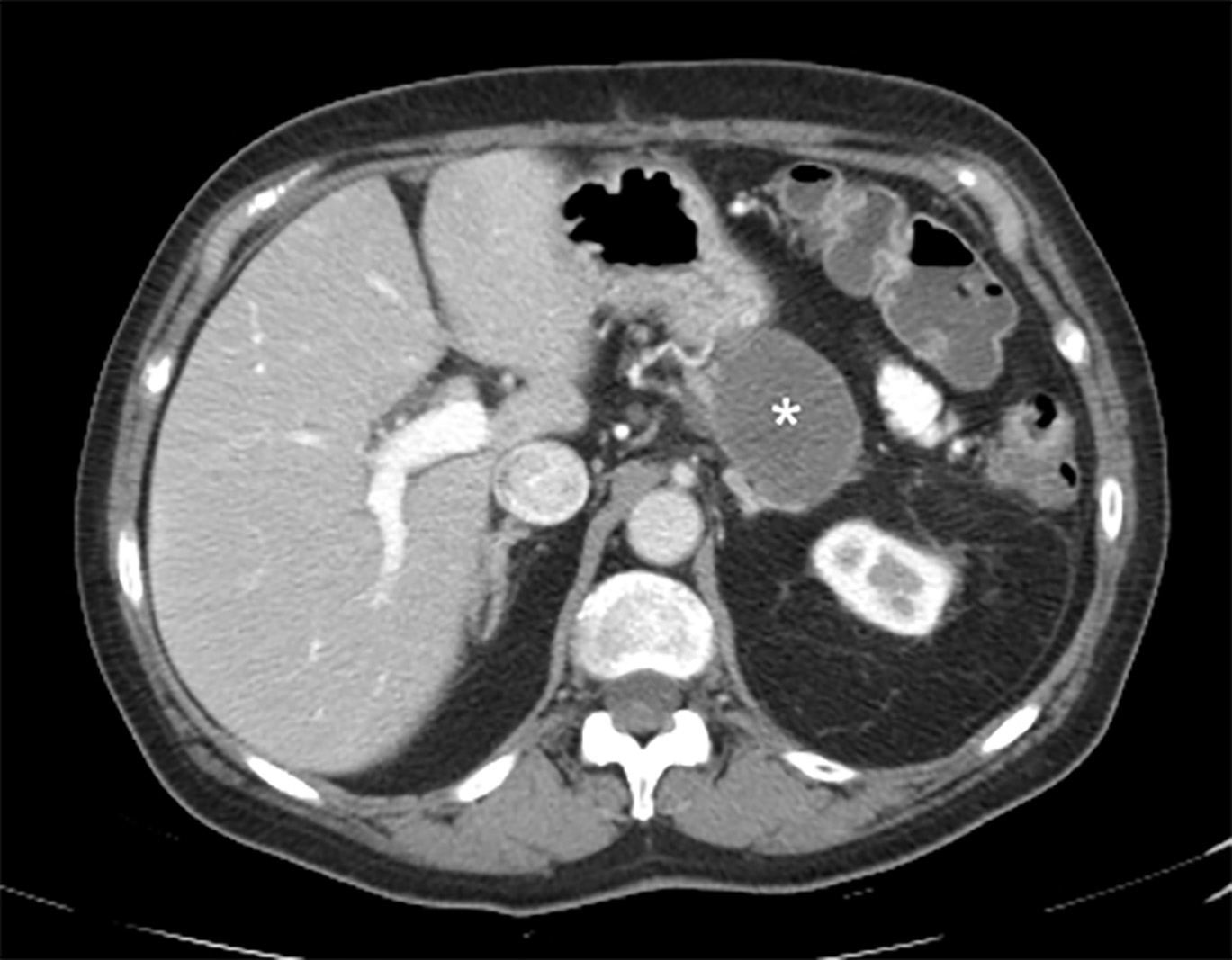
Pancreatic Pseudocysts. Approximately 20-40% of patients with chronic pancreatitis develop pancreatic pseudocysts, which are collections of pancreatic fluid encased in inflammatory walls.45 (See Figure 1.) The majority of pseudocysts remain asymptomatic and can be observed.46 Intervention is warranted in patients with pseudocysts who develop chronic abdominal pain, early satiety with weight loss, gastric outlet obstruction, biliary obstruction, infection, fistula formation, or pancreatic duct stones.47 Typically, patients develop symptoms or complications from pseudocysts that are > 5 cm in diameter.40
Figure 1. Computed Tomography Demonstrating a Pseudocyst (Star) in a Patient With Chronic Pancreatitis |
 |
|
Image used with permission from: David Guez, MD |
Pancreatic Duct Strictures. Many patients with chronic pancreatitis develop pancreatic duct strictures, which can lead to increased pressure in the pancreatic duct and subsequent pain.51,52 Most pancreatic duct strictures are benign; however, malignancy should be excluded. Approximately 10% of patients with pancreatic duct strictures develop biliary cirrhosis or cholangitis.52 Endoscopic treatment of benign pancreatic duct strictures is indicated if patients develop secondary biliary cirrhosis, common bile duct stones, persistent jaundice, or significant pain.53
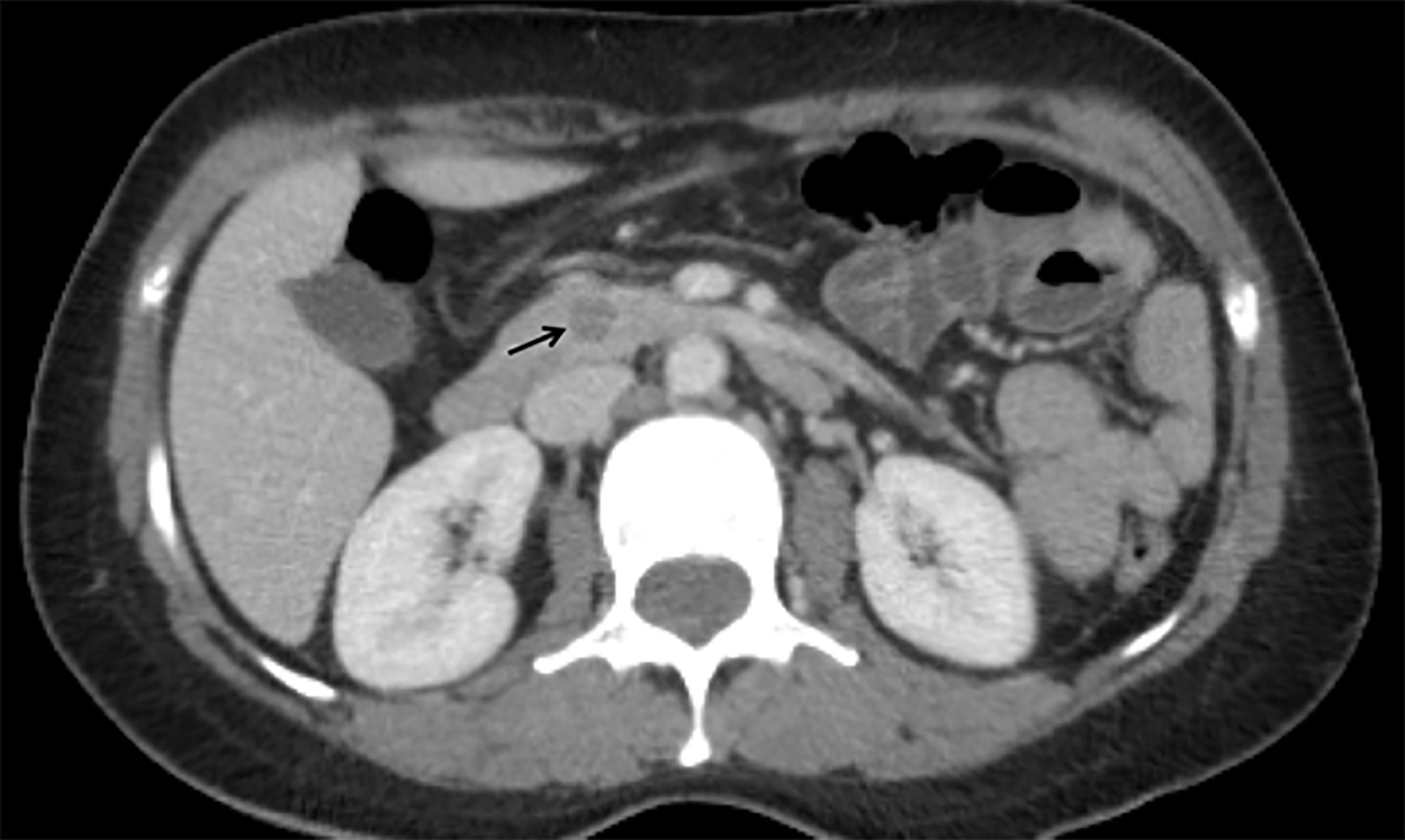
Pancreatic Cancer. About 5% of patients who have chronic pancreatitis will develop pancreatic cancer.54-58 (See Figure 2.) The cumulative incidence of pancreatic cancer is 2% per decade after diagnosis of chronic pancreatitis.56 Higher rates of alcohol use and tobacco smoking in these patients likely contribute to the increased overall risk of cancer.57,59 Without surgical resection, the life expectancy of patients with chronic pancreatitis and pancreatic cancer is less than one year. If a tumor is found and resected in a timely manner, five-year survival improves to 20-25%.40
Figure 2. Computed Tomography Demonstrating Pancreatic Cancer (Arrow) |
 |
Morbidity and Mortality. Patients with chronic pancreatitis typically report a decreased quality of life, which is thought to contribute to high rates of unemployment and low income in these patients.60 Patients with chronic pancreatitis have 2.5-times higher mortality than sex- and age-matched controls.61-63 Part of the increased mortality is due to the comorbidities associated with chronic pancreatitis, such as diabetes and peptic ulcer disease. The most common causes of death in patients with chronic pancreatitis are malignancy (22-23%) and cardiovascular disease (12-21%).61,63
Diagnostic Studies
Given that acinar cell destruction leads to decreased enzyme production in patients with chronic pancreatitis, serum levels of pancreatic enzymes, such as lipase, are unreliable markers of disease. As a result, imaging generally is required to diagnose chronic pancreatitis and its complications.
Endoscopic ultrasound, with or without contrast, is considered the best imaging modality for chronic pancreatitis, although it generally is not available in the ED.10,64,65 Transabdominal ultrasound can be used to detect large pseudocysts. Computed tomography and magnetic resonance imaging are insensitive for identification of early stages of chronic pancreatitis, although they may detect complications of late disease, including pancreatic calcifications, ductal changes, pancreatic atrophy, pseudocysts, and malignancy.10,66 Endoscopic retrograde cholangiopancreatography (ERCP) rarely is used for diagnostic purposes and largely has been replaced by endoscopic ultrasound.10
Management
In most cases of chronic pancreatitis, there is no specific treatment to halt disease progression aside from alcohol and smoking cessation. Management is mainly symptom control. Medical management, consisting of analgesics and pancreatic enzyme supplementation, is effective at treating symptoms in approximately 50% of patients.14 It is less effective in patients who continue to drink alcohol or smoke.67 Patients with diabetes will require insulin, and pancreatic enzyme-replacement therapy usually is started in patients with exocrine insufficiency.7
Endoscopic Treatments. Endoscopic techniques to decompress pancreatic duct stones and strictures and drain pseudocysts have been shown to relieve pain in 65% of patients.51 Endoscopic treatment of pancreatic duct strictures typically consists of pancreatic sphincterectomy, followed by stent placement.68,69 The duration of stent therapy is controversial, and stent occlusion is common.70 Endoscopic techniques to remove pancreatic duct stones via retrograde access of the pancreatic duct include pancreatic sphincterotomy, forceps, or employment of extraction balloons or baskets. 26,71,72 Simultaneous endoscopic extracorporeal shock wave lithotripsy is also performed to clear stones.73,74 Complications of these procedures include bleeding, iatrogenic pancreatitis, duct perforation, stent migration, infection, and sphincter stenosis.26
Surgical Treatments. Surgical options in patients with severe pain related to chronic pancreatitis include partial pancreatic resection and total pancreatectomy, with or without islet cell auto-transplantation. Studies regarding the optimal timing for surgery to treat painful chronic pancreatitis are limited, although there is some evidence that surgery is more effective if performed early in the disease course.76,77 Cases of infected pancreatic necrosis may be managed conservatively with antibiotics or surgical debridement.40
Novel Therapies. Some novel therapies to decrease inflammation and subsequent fibrosis are in the experimental phase.14 Cannabinoid-2-receptor agonists have been shown to decrease pain associated with chronic pancreatitis and protect tissue from fibrosis.78 Some immunomodulatory agents are effective in animal models but are not yet available for humans.79,80 Spinal cord stimulation has shown promise in decreasing pain and daily opioid requirement in patients with chronic pancreatitis.81
Additional Aspects
Management of Chronic Pain. Chronic stimulation of pancreatic pain receptors in patients with chronic pancreatitis is thought to lead to abnormal pain processing and neural remodeling in the central nervous system, known as “central sensitization.”82-85 As a result, patients develop a maladaptive response to pain that can be difficult to manage. Acetaminophen is recommended as the first-line analgesic for chronic pain.86 Other non-opiate medications shown to be effective in the treatment of pain associated with chronic pancreatitis include gabapentin, pregabalin, selective serotonin reuptake inhibitors, and tricyclic antidepressants (TCA).7,87 Sub-anesthetic doses of ketamine have been used in children and adults, although evidence for its efficacy is limited.88-90 There is conflicting evidence about whether antioxidants are effective at relieving pain.91,92 Nonsteroidal anti-inflammatory drugs should be avoided because of the risk of stomach ulcers in patients. Alcohol and smoking cessation also have been shown to decrease pain and should be encouraged.25
Patients with chronic pancreatitis often require oral opiates for severe pain; however, only 25% of patients are likely to benefit from opioid treatment.93 Tramadol has been shown to be as effective as morphine, although with a better side-effect profile, making it the preferred first-line opioid.94
Pediatric Chronic Pancreatitis. Acute pancreatitis that occurs in children usually is secondary to biliary obstruction, systemic illnesses, or medications. Up to 30% of cases are idiopathic.101 Of children with acute pancreatitis, 15-35% will develop recurrent or chronic pancreatitis; the most common predisposing factor is genetic mutation.102 Similar to adults, children with chronic pancreatitis experience significant pain and may develop type 3c diabetes and exocrine insufficiency. Transabdominal ultrasound is the preferred imaging modality in children because of the lack of ionizing radiation.101 There is increased emphasis on opioid-free management of the condition in children.
Disposition
In patients with chronic pancreatitis who present to the ED with abdominal pain, it is important to ascertain the etiology of the pain. If the pain is secondary to an exacerbation of chronic pain and can be managed successfully with oral analgesics, patients may be discharged home. Patients with clinical or radiologic signs of infection, such as infected pancreatic necrosis or an infected pseudocyst, require admission for antibiotics and potential debridement or drainage. Antibiotics usually are not given to patients with sterile necrosis or prophylatically.103 Unless there is an associated paralytic ileus, enteral nutrition should be encouraged in admitted patients, as parenteral nutrition can lead to intestinal villous atrophy, bacterial translocation, and subsequent infection of necrotic tissue.104,105 Stress ulcer prophylaxis is recommended in admitted patients, given the risk of gastrointestinal ulcers.40
Patients who are discharged should be counseled on alcohol and smoking cessation. Patients typically are referred to primary care for preventive care (e.g., cancer screening), gastroenterology, and a pain management specialist.
Cyclic Vomiting Syndrome
Epidemiology
Cyclic vomiting syndrome is characterized by recurrent, self-limited, and often incapacitating episodes of nausea and vomiting that are separated by periods free of symptoms.106,107 It was described first in children in 1882.3 The prevalence of cyclic vomiting syndrome is estimated to be 2% in school-age children and is unknown in adults, although it may account for up to 14% of cases of unexplained nausea and vomiting in adults.107-109 Patients most commonly are white (63%) and female (63%).106,110 The mean age of onset in children is approximately 5 years of age, although diagnosis typically is not made until age 9 years.111 In adults, the mean age of onset and diagnosis is 20 to 30 years.106,107
The average cost of an inpatient admission for cyclic vomiting syndrome is nearly $20,000, which does not include costs related to missed work or outpatient studies.110 High rates of medical recidivism have been associated with difficulties managing the disorder and frustration among patients and physicians regarding its often-unclear etiology.110 Despite a recent increase in published literature about the disorder in both children and adults, the diagnosis often is missed or misdiagnosed in the ED.107,112
Etiology
The etiology of cyclic vomiting syndrome likely is multifactorial and variable across patients, as many conditions and comorbidities have been linked to the disorder. Triggers of cyclic vomiting syndrome are identified more commonly in children and can be found in 80% of patients.108
Comorbidities
Headache. Patients with cyclic vomiting syndrome often have a personal or family history of headaches.108,110,111,113 Many children and adults also eventually develop migraines, and studies have demonstrated similar electroencephalogram changes in patients with cyclic vomiting syndrome and migraines.114 It is thought that cyclic vomiting syndrome and migraines may exist on a spectrum of pathology; cyclic vomiting syndrome is considered a subgroup of migraines.107,115 (See Table 2.)
Table 2. Differential Diagnosis of Patients Presenting With Cyclic Vomiting Syndrome |
|
|
Gastrointestinal disorders |
|
|
Neurologic disorders |
|
|
Metabolic disorders |
|
|
Medications |
|
|
Urologic and gynecologic disorders |
|
Substance Use. Recently, there have been increased reports of cyclic vomiting syndrome related to cannabis use, although opiates and nicotine have been implicated in the disorder as well.110 Cannabis has been used for brief periods to treat nausea in some patients. However, its long-term use has been shown to induce cyclic vomiting syndrome. It is thought that chronic use leads to fat tissue accumulation of δ-9-tetrahydrocannabinol (THC), a lipophilic compound in cannabis. During times of stress, lipolysis releases a large amount of THC into the circulation and induces nausea.115 Abstinence from cannabis resolves symptoms in some, but not all, patients.115
Psychiatric Disease. Patients with cyclic vomiting syndrome often have concurrent anxiety or depression, although a causal relationship has not been identified.108,110,114,116 An estimated 44% of cases of cyclic vomiting syndrome can be attributed to psychological stress.108 Treating comorbid anxiety has been shown to improve clinical outcomes in patients with cyclic vomiting syndrome.117 It is recommended that patients who present with symptoms of cyclic vomiting syndrome be screened for psychiatric illness.
Other Associations. Cyclic vomiting syndrome is associated with gastroesophageal reflux disease, gastroparesis, infections (most commonly upper respiratory infections), diabetes mellitus, sleep deprivation, medications, menses, and pregnancy.107,109-111,115,119 Excitement and emotional stress are common triggers in children.114
Pathophysiology
The exact pathophysiology of cyclic vomiting syndrome is unclear.113,115 Given the similarities with migraines (e.g., episodic attacks, stereotyped onset), cyclic vomiting syndrome, like migraines, may be the result of autonomic dysfunction.108,109 Another theory implicates the brain-gut neuroendocrine axis.107,108,115,118 Normally, the hypothalamus releases corticotropin-releasing factor (CRF), which stimulates downstream release of adrenocorticotropic hormone (ACTH) and antidiuretic hormone (ADH). In patients with cyclic vomiting syndrome, elevated ACTH and ADH levels are seen, which is thought to be due to stress-induced release of CRF and leads to delayed gastric emptying and vomiting.108,113 TCAs are known to inhibit the CRF gene and have been shown to be effective in the treatment of cyclic vomiting syndrome.108 Another theory has linked mitochondrial DNA mutations with cyclic vomiting syndrome in children, although this has not been demonstrated in adults with the disorder.115
Clinical Features
Often it is difficult to diagnose cyclic vomiting syndrome in the ED. Up to one-third of patients with cyclic vomiting syndrome also develop low-grade fever and diarrhea, making it difficult to distinguish from gastroenteritis.119 Up to 70% of patients with cyclic vomiting syndrome have abdominal pain, which broadens the differential diagnosis.108 In addition, symptoms of cyclic vomiting syndrome are highly variable between patients, although they usually are consistent within one individual.111 One study found that it takes a median of 15 ED visits and five to six years of symptoms to reach a diagnosis of cyclic vomiting syndrome.109
The Rome IV criteria were developed to assist in the diagnosis of cyclic vomiting syndrome. Patients must have each of the following for at least six months before meeting diagnostic criteria for the condition: stereotypical episodes of acute-onset vomiting that last less than one week; at least three discrete episodes in the previous year and two episodes in the past six months that occur at least one week apart; and absence of symptoms between episodes.109
Episodes of nausea and vomiting associated with cyclic vomiting syndrome are characterized by four distinct phases. In the pre-emetic period, a patient experiences pallor, diaphoresis, and nausea. In the intense emesis period, a patient can have up to 30 episodes of emesis daily, and also may develop abdominal pain or diarrhea. This is followed by the recovery phase, in which the nausea and vomiting gradually resolve, and the inter-episodic phase, in which the patient is symptom-free.109 Episodes of cyclic vomiting syndrome generally are more prolonged and frequent in adults than in children. In addition, some adults progress to a “coalescent stage,” characterized by subacute symptoms and near-continuous nausea.108
Diagnostic Studies
Emergency providers are unlikely to diagnose a patient with the condition in the ED. However, certain laboratory and imaging studies rule out more emergent pathologies. For example, a patient with multiple ED visits for nausea and vomiting, with prior laboratory (e.g., lipase, hepatic function tests) and imaging studies that are unremarkable, ultimately may be diagnosed with cyclic vomiting syndrome.108,120,121
Management
Many treatments for cyclic vomiting syndrome are based on pediatric studies or anecdotal evidence and lack robust evidence.122 In adults, sumatriptan has been shown to treat symptoms effectively in 56-83% of patients and is considered the first-line abortive treatment.108,111,115,116,121-123 (See Table 3.) It is approved only for patients older than 12 years of age.121 Side effects of sumatriptan include neck pain and vasospasm.121 Dextrose-containing solutions with potassium replacement (if the patient is hypokalemic from vomiting) are recommended for intravenous hydration.107,115,121 Diphenhydramine (an H1-histamine antagonist) and ondansetron (a 5-HT antagonist) are used routinely to treat nausea in adults.116 In children, diphenhydramine and prochlorperazine are used commonly to treat nausea.115 If antiemetics fail, an anxiolytic/sedative, such as lorazepam, can be used.108,111,121 Nonsteroidal anti-inflammatory drugs, such as ketorolac, typically are used to treat pain. Opioids should be used cautiously because of the risks of misuse and dependence as well as rebound symptoms.115
Table 3. Abortive Treatments for Cyclic Vomiting Syndrome |
|
|
Environmental |
Place patient in a dark, quiet room |
|
Abortive |
Sumatriptan (patients > 12 years of age)
|
|
Hydration |
|
|
Anti-nausea medications |
Ondansetron, prochlorperazine, chlorpromazine |
|
Sedatives |
Lorazepam, diphenhydramine |
|
Pain medications |
Nonsteroidal anti-inflammatory drugs, acetaminophen, opiates |
|
Experimental |
Haloperidol, topical capsaicin cream |
TCAs have been shown to be the most effective medications for long-term preventive therapy in both children and adults, with greater than 75% of patients achieving symptom resolution.116 (See Table 4.) In patients older than 5 years of age, amitriptyline is the first-line agent. In children younger than 5 years of age, cyproheptadine (a histamine-receptor antagonist) is preferred.111 Side effects of TCAs include behavioral changes, somnolence, constipation, and arrhythmias.121,123 Less commonly used medications that also are effective in preventing cyclic vomiting symptoms include propranolol, anticonvulsants (e.g., zonisamide, phenytoin), and vitamins (e.g., coenzyme Q10, L-carnitine, riboflavin). Long-term preventive medications usually are not initiated in the ED without first consulting a gastroenterologist.109,111,121,124,126
Table 4. Preventive Treatments for Cyclic Vomiting Syndrome |
|
|
Environmental |
Avoidance of triggers (e.g., cannabis) |
|
Medications |
Tricyclic antidepressants, cyproheptadine, anticonvulsants, vitamins |
In patients with cyclic vomiting syndrome secondary to cannabis use, abstinence from cannabis alleviates symptoms in up to 97% of patients.127 However, given the popular perception that cannabis relieves nausea, it often is difficult to convince patients to quit. There is anecdotal evidence that hot showers, benzodiazepines, haloperidol, and topical capsaicin cream also are effective treatments for symptoms associated with cyclic vomiting secondary to cannabis, although the exact mechanisms of action are unclear.127-129 Administration of haloperidol 5 mg intravenously or orally is effective in acute exacerbations of cyclic vomiting syndrome; however, it has the risk of inducing ventricular tachyarrhythmias in patients with prolonged QT intervals.128,130
Disposition
Patients with uncontrolled symptoms, dehydration, or electrolyte disturbances may require admission.119 Patients who are well enough to be discharged should be referred to a gastroenterologist for follow-up care. Generally, prophylactic medications are prescribed from the ED only after consultation with a gastroenterologist.
Conclusion
Patients with chronic abdominal pain disorders, including chronic pancreatitis and cyclic vomiting syndrome, commonly present to the ED with exacerbations of pain, nausea, and emesis. It is important for emergency providers to be aware of non-opiate treatments of pain, not only to avoid opiate side effects and misuse among patients, but also to prevent “central sensitization” and a maladaptive pain response from long-term opiate use. Rather than prescribing opiates, emergency providers should consider alternatives, such as pregabalin and gabapentin, in chronic pancreatitis, and sumatriptan in cyclic vomiting syndrome.
In addition to knowing how to control symptoms in patients with chronic abdominal pain disorders, providers also should be aware of emergent complications associated with the disorders. As described in this article, patients with chronic pancreatitis may present with episodes of either hyperglycemia (secondary to type 3c diabetes) or hypoglycemia. Finally, given the increased risk of pancreatic cancer, emergency providers should know how to counsel their patients on alcohol and drug cessation and to provide appropriate outpatient referrals.
REFERENCES
- Guantlett-Gilbert J, Rodham K, Jordan A, Brook P. Emergency department staff attitudes toward people presenting in chronic pain: A qualitative study. Pain Med 2015;16:2065-2074.
- Donovan E, Ranney ML, Reid AM, et al. Chronic pain in the emergency department. R I Med J 2013;100:25-29.
- Gee S. On fitful or recurrent vomiting. St Bartholomews Hosp Rep 1882;18:1-6.
- Ahmed AU, Issa Y, Hagenaars JC, et al. Risk of recurrent pancreatitis and progression to chronic pancreatitis after a first episode of acute pancreatitis. Clin Gastroenterol Heptaol 2016;14:738-746.
- Machicad JD, Yadav D. Epidemiology of recurrent acute and chronic pancreatitis: Similarities and differences. Dig Dis Sci 2017;62:1683-1691.
- Wilcox CM, Sandhu BS, Singh V, et al. Racial differences in the clinical profile, causes, and outcome of chronic pancreatitis. Am J Gastroenterol 2016;111:1488-1496.
- Majumder S, Chari ST. Chronic pancreatitis. Lancet 2016;387:1957-1966.
- Hall TC, Garcea G, Webb MA, et al. The socio-economic impact of chronic pancreatitis: A systematic review. J Eval Clin Pract 2014;20:203-207.
- Peery AF, Crockett SD, Barritt AS, et al. Burden of gastrointestinal, liver, and pancreatic diseases in the United States. Gastroenterology 2015;149:1731-1741.
- Dimastromatteo J, Brentnall T, Kelly KA. Imaging in pancreatic disease. Nat Rev Gastroenterol Hepatol 2017;14:97-109.
- Kloppel G, Maillet B. The morphological basis for the evolution of acute pancreatitis into chronic pancreatitis. Virchows Arch A Pathol Anat Histopathol 1992;420:1-4.
- Gerasimenko JV, Gerasimenko OV, Petersen OH. The role of Ca2+ in the pathophysiology of pancreatitis. J Physiol 2014;592:269-280.
- Whitcomb DC. Hereditary pancreatitis: New insights into acute and chronic pancreatitis. Gut 1999;45:317-322.
- Jagannath S, Garg PK. Novel and experimental therapies in chronic pancreatitis. Dig Dis Sci 2017;62:1751-1761.
- Yadav D, Hawes RH, Brand RE, et al; North American Pancreatic Study Group. Alcohol consumption, cigarette smoking, and the risk of recurrent acute and chronic pancreatitis. Arch Intern Med 2009;169:1035-1045.
- Irving HM, Samokhvalov AV, Rehm J. Alcohol as a risk factor for pancreatitis. A systematic review and meta-analysis. JOP 2009;10:387-392.
- Shield KD, Parry C, Rehm J. Chronic diseases and conditions related to alcohol use. Alcohol Res 2013;35:155-173.
- Mumenthaler MS, Taylor JL, O’Hara R, Yesavage JA. Gender differences in moderate drinking effects. Alcohol Res Health 1999;23:55-64.
- Lin Y, Tamakoshi A, Hayakawa T, et al. Associations of alcohol drinking and nutrient intake with chronic pancreatitis: Findings from a case-control study in Japan. Am J Gastroenterol 2001;96:2622-2627.
- Takeyama Y. Long-term prognosis of acute pancreatitis in Japan. Clin Gastroenterol Hepatol 2009;7:S15-S17.
- Lankisch PG, Breuer N, Bruns A, et al. Natural history of acute pancreatitis: A long-term population-based study. Am J Gastroenterol 2009;104:2797-2805.
- Steer ML, Waxman I, Freedman S. Chronic pancreatitis. N Engl J Med 1995;332:1482-1490.
- Andriulli A, Botteri E, Almasio PL, et al. Smoking as a cofactor for causation of chronic pancreatitis: A meta-analysis. Pancreas 2010;39:1205-1210.
- Chowdhury P, Walker A. A cell-based approach to study changes in the pancreas following nicotine exposure in an animal model of injury. Langenbecks Arch Surg 2008;393:547-555.
- Frulloni L, Falconi M, Gabbrielli A, et al; Italian Association for the Study of the Pancreas (AISP). Italian consensus guidelines for chronic pancreatitis. Dig Liver Dis 2010;42(Suppl 6):S381-S406.
- Adler JM, Gardner TB. Endoscopic therapies for chronic pancreatitis. Dig Dis Sci 2017;62:1729-1737.
- Klöppel G, Maillet B. Pathology of acute and chronic pancreatitis. Pancreas 1993;8:659-670.
- Madsen P, Winkler K. The intraductal pancreatic pressure in chronic obstructive pancreatitis. Scand J Gastroenterol 1982;17:553-554.
- Whitcomb DC, Preston RA, Aston CE, et al. A gene for hereditary pancreatitis maps to chromosome 7q35. Gastroenterology 1996;110:1975-1980.
- Whitcomb DC, Gorry MC, Preston RA, et al. Hereditary pancreatitis is caused by a mutation in the cationic trypsinogen gene. Nat Genet 1996;14:141-145.
- Cohn JA, Friedman KJ, Noone PG, et al. Relation between mutations of the cystic fibrosis gene and idiopathic pancreatitis.
N Engl J Med 1998;339:653-658. - Sharer N, Schwarz M, Malone G, et al. Mutations of the cystic fibrosis gene in patients with chronic pancreatitis. N Engl J Med 1998;339:645-652.
- Witt H, Luck W, Hennies HC, et al. Mutations in the gene encoding the serine protease inhibitor, Kazal type 1 are associated with chronic pancreatitis. Nat Genet 2000;25:213-216.
- Aoun E, Muddana V, Papachristou GI, Whitcomb DC. SPINK1 N34S is strongly associated with recurrent acute pancreatitis but is not a risk factor for the first or sentinel acute pancreatitis event. Am J Gastroenterol 2010;105:446-451.
- Yadav D, Lowenfels AB. Trends in the epidemiology of the first attack of acute pancreatitis: A systematic review. Pancreas 2006;33:323-330.
- Hart PA, Kamisawa T, Brugge WR, et al. Long-term outcomes of autoimmune pancreatitis: A multicentre, international analysis. Gut 2013;62:1771-1776.
- Dite P, Uvirova M, Bojkova M, et al. Differentiating autoimmune pancreatitis from pancreatic cancer. Minerva Gastroenterol Dietol 2014;60:247-253.
- Etemad B, Whitcomb DC. Chronic pancreatitis: Diagnosis, classification, and new genetic developments. Gastroenterology 2001;120:682-707.
- Wilcox CM, Yadav D, Ye T, et al. Chronic pancreatitis pain pattern and severity are independent of abdominal imaging findings. Clin Gastroenterol Hepatol 2015;13:552-560.
- Hoffmeister A, Mayerle J, Beglinger C, et al. English language version of the S3-consensus guidelines on chronic pancreatitis: Definition, aetiology, diagnostic examinations, medical, endoscopic and surgical management of chronic pancreatitis. Z Gastroenterol 2015;53:1447-1495.
- Ammann RW, Akovbiantz A, Largiader F, Schueler G. Course and outcome of chronic pancreatitis. Longitudinal study of a mixed medical-surgical series of 245 patients. Gastroenterology 1984;86:820-828.
- Hart PA, Bellin M, Andersen DK, et al. Type 3c (pancreatogenic) diabetes mellitus secondary to chronic pancreatitis and pancreatic cancer. Lancet Gastroenterol Hepatol 2016;1:226-237.
- DiMagno EP, Go VL, Summerskill WH. Relations between pancreatic enzyme outputs and malabsorption in severe pancreatic insufficiency. N Engl J Med 1973;288:813-815.
- Harris S, Nadkarni NA, Naina HV, Vege SS. Splanchnic vein thrombosis in acute pancreatitis: A single-center experience. Pancreas 2013;42:1251-1254.
- Andren-Sandberg A, Dervenis C. Pancreatic pseudocysts in the 21st century. Part I: Classification, pathophysiology, anatomic considerations and treatment. JOP 2004;5:8-24.
- Cheruvu CV, Clarke MG, Prentice M, Eyre-Brook IA. Conservative treatment as an option in the management of pancreatic pseudocyst. Ann R Coll Surg Engl 2003;85:313-316.
- Lerch MM, Stier A, Wahnschaffe U, Mayerle J. Pancreatic pseudocysts: Observation, endoscopic drainage, or resection? Dtsch Arztebl Int 2009;106:614-621.
- Gurusamy KS, Pallari E, Hawkins N, et al. Management strategies for pancreatic pseudocysts. Cochrane Database Syst Rev 2016;4:CD011392.
- Keane MG, Sze SF, Cieplik N, et al. Endoscopic versus percutaneous drainage of symptomatic pancreatic fluid collections: A 14-year experience from a tertiary hepatobiliary centre. Surg Endosc 2016;30:3730-3740.
- Varadarajulu S, Bang JY, Sutton BS, et al. Equal efficacy of endoscopic and surgical cystogastrostomy for pancreatic pseudocyst drainage in a randomized trial. Gastroenterology 2013;145:583.
- Rosch T, Daniel S, Scholz M, et al. Endoscopic treatment of chronic pancreatitis: A multicenter study of 1000 patients with long-term follow-up. Endoscopy 2002;34:765-771.
- Vijungco JD, Prinz RA. Management of biliary and duodenal complications of chronic pancreatitis. World J Surg 2003;27:1258-1270.
- Frey CF, Suzuki M, Isaji S. Treatment of chronic pancreatitis complicated by obstruction of the common bile duct or duodenum. World J Surg 1990;14:59-69.
- Kolodecik T, Shugrue C, Ashat M, Thrower EC. Risk factors for pancreatic cancer: Underlying mechanisms and potential targets. Front Physiol 2014;4:415.
- Maire F, Micard S, Hammel P, et al. Differential diagnosis between chronic pancreatitis and pancreatic cancer: Value of the detection of KRAS2 mutations in circulating DNA. Br J Cancer 2002;87:551-554.
- Lowenfels AB, Maisonneuve P, Cavallini G, et al. Pancreatitis and the risk of pancreatic cancer. International Pancreatitis Study Group. N Engl J Med 1993;328:1433-1437.
- Kirkegard J, Mortensen FV, Cronin-Fenton D. Chronic pancreatitis and pancreatic cancer risk: A systematic review and meta-analysis. Am J Gastroenterol 2017;112:1366-1372.
- Raimondi S, Lowenfels AB, Morselli-Labate AM, et al. Pancreatic cancer in chronic pancreatitis; aetiology, incidence, and early detection. Best Pract Res Clin Gastroenterol 2010;24:349-358.
- Maisonneuve P, Lowenfels AB. Risk factors for pancreatic cancer: A summary review of meta-analytical studies. Int J Epidemiol 2015;44:186-198.
- Gardner TB, Kennedy AT, Gelrud A, et al. Chronic pancreatitis and its effect on employment and health care experience: Results of a prospective American multicenter study. Pancreas 2010;39:498-501.
- Yadav D, Timmons L, Benson JT, et al. Incidence, prevalence, and survival of chronic pancreatitis: A population-based study. Am J Gastroenterol 2011;106:2192-2199.
- Nojgaard C, Bendtsen F, Becker U, et al. Danish patients with chronic pancreatitis have a four-fold higher mortality rate than the Danish population. Clin Gastroenterol Hepatol 2010;8:384-390.
- Bang UC, Benfield T, Hyldstrup L, et al. Mortality, cancer, and comorbidities associated with chronic pancreatitis: A Danish nationwide matched-cohort study. Gastroenterology 2014;146:989-994.
- Brugge WR. Role of endoscopic ultrasound in the diagnosis of cystic lesions of the pancreas. Pancreatology 2001;1:637-640.
- Zuccaro G Jr, Sivak MV Jr. Endoscopic ultrasonography in the diagnosis of chronic pancreatitis. Endoscopy 1992;24:347-349.
- Siddiqi AJ, Miller F. Chronic pancreatitis: Ultrasound, computed tomography, and magnetic resonance imaging features. Semin Ultrasound CT MRI 2007;28:384-394.
- Shalimar, Midha S, Hasan A, et al. Long-term pain relief with optimized medical treatment including antioxidants and step-up interventional therapy in patients with chronic pancreatitis. J Gastroenterol Hepatol 2017;32:270-277.
- Ponchon T, Bory RM, Hedelius F, et al. Endoscopic stenting for pain relief in chronic pancreatitis: Results of a standardized protocol. Gastrointest Endosc 1995;42:452-456.
- Smits ME, Badiga SM, Rauws EA, et al. Long-term results of pancreatic stents in chronic pancreatitis. Gastrointest Endosc 1995;42:461-467.
- Binmoeller KF, Jue P, Seifert H, et al. Endoscopic pancreatic stent drainage in chronic pancreatitis and a dominant stricture: Long-term results. Endoscopy 1995;27:638-644.
- Smits ME, Rauws EA, Tytgat GN, Huibregtse K. Endoscopic treatment of pancreatic stones in patients with chronic pancreatitis. Gastrointest Endosc 1996;43:556-560.
- Sherman S, Lehman GA, Hawes RH, et al. Pancreatic ductal stones: Frequency of successful endoscopic removal and improvement in symptoms. Gastrointest Endosc 1991;37:511-517.
- Kozarek RA, Brandabur JJ, Ball TJ, et al. Clinical outcomes in patients who undergo extracorporeal shock wave lithotripsy for chronic calcific pancreatitis. Gastrointest Endosc 2002;56:496-500.
- Seven G, Schreiner MA, Ross AS, et al. Long-term outcomes associated with pancreatic extracorporeal shock wave lithotripsy for chronic calcific pancreatitis. Gastrointest Endosc 2012;75:997.
- Ahmed Ali U, Pahlplatz JM, Nealon WH, et al. Endoscopic or surgical intervention for painful obstructive chronic pancreatitis. Cochrane Database Syst Rev 2015;3:CD007884.
- Yang CJ, Bliss LA, Schapira EF, et al. Systematic review of early surgery for chronic pancreatitis: Impact on pain, pancreatic function, and re-intervention. J Gastrointest Surg 2014;18:1863-1869.
- Yang CJ, Bliss LA, Freedman SD, et al. Surgery for chronic pancreatitis: The role of early surgery in pain management. Pancreas 2015;44:819-823.
- Zhang L, Kline RH 4th, McNearney TA, et al. Cannabinoid receptor 2 agonist attenuates pain related behavior in rats with chronic alcohol/high fat diet induced pancreatitis. Mol Pain 2014;10:66.
- Baumert JT, Sparmann G, Emmrich J, et al. Inhibitory effect of interferons on pancreatic stellate cell activation. World J Gastroenterol 2006;12:896-901.
- Zhao HF, Ito T, Gibo J, et al. Anti-monocyte chemoattractant protein 1 gene therapy attenuates experimental chronic pancreatitis induced by dibutyltin dichloride in rats. Gut 2005;54:1759-1767.
- Kapural L, Cywinski JB, Sparks DA. Spinal cord stimulation for visceral pain from chronic pancreatitis. Neuromodulation 2011;14:423-427.
- Talukdar R, Reddy DN. Pain in chronic pancreatitis: Managing beyond the pancreatic duct. World J Gastroenterol 2013;19:6319-6328.
- Drewes AM, Krarup AL, Detlefsen S, et al. Pain in chronic pancreatitis: The role of neuropathic pain mechanisms. Gut 2008;57:1616-1627.
- Demir IE, Tieftrunk E, Maak M, et al. Pain mechanisms in chronic pancreatitis: Of a master and his fire. Langenbecks Arch Surg 2011;396:151-160.
- Frøkjær JB, Olesen SS, Drewes AM. Fibrosis, atrophy, and ductal pathology in chronic pancreatitis are associated with pancreatic function but independent of symptoms. Pancreas 2013;42:1182-1187.
- Jadad AR, Browman GP. The WHO analgesic ladder for cancer pain management. Stepping up the quality of its evaluation. JAMA 1995;20;274:1870-1873.
- Olesen SS, Bouwense SA, Wilder-Smith OH, et al. Pregabalin reduces pain in patients with chronic pancreatitis in a randomized, controlled trial. Gastroenterology 2011;141:536-543.
- Mannion S, O’Brien T. Ketamine in the management of chronic pancreatic pain. J Pain Symptom Manage 2003;26:1071-1072.
- Bouwense SA, Buscher HC, van Goor H, Wilder-Smith OH. S-ketamine modulates hyperalgesia in patients with chronic pancreatitis pain. Reg Anesth Pain Med 2011;36:303-307.
- Sheehy KA, Lippold C, Rice AL, et al. Subanesthetic ketamine for pain management in hospitalized children, adolescents, and young adults: A single-center cohort study. J Pain Res 2017;10:787-795.
- Zhou D, Wang W, Cheng X, et al. Antioxidant therapy for patients with chronic pancreatitis: A systematic review and meta-analysis. Clin Nutr 2015;34:627-634.
- Cai GH, Huang J, Zhao Y, et al. Antioxidant therapy for pain relief in patients with chronic pancreatitis: Systematic review and meta-analysis. Pain Physician 2013;16:521-532.
- Drewes AM, Bouwense SAW, Campbell CM, et al. Guidelines for the understanding and management of pain in chronic pancreatitis. Pancreatology 2017;17:720-731.
- Wilder-Smith CH, Hill L, Osler W, O’Keefe S. Effect of tramadol and morphine on pain and gastrointestinal motor function in patients with chronic pancreatitis. Dig Dis Sci 1999;44:1107-1116.
- Gress F, Schmitt C, Sherman S, et al. A prospective randomized comparison of endoscopic ultrasound- and computed tomography-guided celiac plexus block for managing chronic pancreatitis pain. Am J Gastroenterol 1999;94:900-905.
- Gress F, Schmitt C, Sherman S, et al. Endoscopic ultrasound-guided celiac plexus block for managing abdominal pain associated with chronic pancreatitis: A prospective single center experience. Am J Gastroenterol 2001;96:409-416.
- Kaufman M, Singh G, Das S, et al. Efficacy of endoscopic ultrasound-guided celiac plexus block and celiac plexus neurolysis for managing abdominal pain associated with chronic pancreatitis and pancreatic cancer. J Clin Gastroenterol 2010;44:127-134.
- Puli SR, Reddy JB, Bechtold ML, et al. EUS-guided celiac plexus neurolysis for pain due to chronic pancreatitis or pancreatic cancer pain: A meta-analysis and systematic review. Dig Dis Sci 2009;54:2330-2337.
- Stevens T, Costanzo A, Lopez R, et al. Adding triamcinolone to endoscopic ultrasound-guided celiac plexus blockade does not reduce pain in patients with chronic pancreatitis. Clin Gastroenterol Hepatol 2012;10:186-191.
- Dumonceau JM. Endoscopic therapy for chronic pancreatitis. Gastrointest Endosc Clin N Am 2013;23:821-832.
- Uc A, Fishman DS. Pancreatic disorders. Pediatr Clin North Am 2017;64:685-706.
- Morinville VD, Husain SZ, Bai H, et al. Definitions of pediatric pancreatitis and survey of present clinical practices.
J Pediatr Gastroenterol Nutr 2012;55:261-265. - Villatoro E, Mulla M, Larvin M. Antibiotic therapy for prophylaxis against infection of pancreatic necrosis in acute pancreatitis. Cochrane Database Syst Rev 2010;5:CD002941
- Niederau C, Niederau M, Luthen R, et al. Pancreatic exocrine secretion in acute experimental pancreatitis. Gastroenterology 1990;99:1120-1127.
- Petrov MS, Kukosh MV, Emelyanov NV. A randomized controlled trial of enteral versus parenteral feeding in patients with predicted severe acute pancreatitis shows a significant reduction in mortality and in infected pancreatic complications with total enteral nutrition. Dig Surg 2006;23:336-344.
- Van Driessche A, Sermijn E, Paemeleire K, et al. Cyclic vomiting syndrome: Case report and short review of the literature. Acta Clin Belg 2012 67:123-126.
- Hejazi RA, McCallum RW. Review article: Cyclic vomiting syndrome in adults – rediscovering and redefining an old entity. Aliment Pharmacol Ther 2011;34:263-273.
- Evans RW, Whyte C. Cyclic vomiting syndrome and abdominal migraine in adults and children. Headache 2013;53:984-993.
- Stanghellini V, Chan FK, Hasler WL, et al. Gastroduodenal disorders. Gastroenterology 2016;150:1380-1392.
- Bhandari S, Venkatesan T. Clinical characteristics, comorbidities and hospital outcomes in hospitalizations with cyclic vomiting syndrome: A nationwide analysis. Dig Dis Sci 2017;62:2035–2044.
- Irwin S, Barmherzig R, Gelfand A. Recurrent gastrointestinal disturbance: Abdominal migraine and cyclic vomiting syndrome. Curr Neurol Neurosci Rep 2017;17:21.
- Venkatesan T, Tarbell S, Adams K, et al. A survey of emergency department use in patients with cyclic vomiting syndrome. BMC Emerg Med 2010;10:4.
- Hikita T, Kodama H, Ogita K, et al. Cyclic vomiting syndrome in infants and children: A clinical follow-up study. Pediatr Neurol 2016;57:29-33.
- Brown Tonore T, Spree DC, Abell T. Cyclic vomiting syndrome: A common, underrecognized disorder. J Am Assoc Nurse Pract 2014;26:340-347.
- Shearer J, Luthra P, Ford AC. Cyclic vomiting syndrome: A case series and review of the literature. Frontline Gastroenterol 2018;9:2-9.
- Lee LY, Abbott L, Mahlangu B, et al. The management of cyclic vomiting syndrome: A systematic review. Eur J Gastroenterol Hepatol 2012;24:1001-1006.
- Tarbell SE, Li BU. Anxiety measures predict health-related quality of life in children and adolescents with cyclic vomiting syndrome. J Pediatr 2015;167:633-638.
- Lagman-Bartolome AM, Lay C. Pediatric migraine variants: A review of epidemiology, diagnosis, treatment, and outcome. Curr Neurol Neurosci Rep 2015;15:34.
- McAninch, S, Smithson III, CC. Gastrointestinal Emergencies. In: Stone CK, Humphries RL. CURRENT Diagnosis and Treatment Emergency Medicine, 8th ed. New York: McGraw-Hill; 2017.
- Hang BS, Bork S, Ditkoff J, et al. Nausea and Vomiting. In: Tintinalli JE, et al. Tintinalli’s Emergency Medicine: A Comprehensive Study Guide, 8th ed. New York: McGraw-Hill; 2016.
- Li BU, Lefevre F, Chelimsky GG, et al. North American Society for Pediatric Gastroenterology, Hepatology, and Nutrition Consensus Statement on the Diagnosis and Management of Cyclic Vomiting Syndrome. J Pediatr Gastroenterol Nutr 2008;47:379-393.
- Ozdemir HH, Bulut S, Berilgen MS, et al. Resistant cyclic vomiting syndrome successfully responding to chlorpromazine. Acta Medica (Hradec Kralove) 2014;57:28-29.
- Okumura T, Ohhira M, Kumei S, Nozu T. An adult patient with cyclic vomiting syndrome successfully treated with oral sumatriptan. Am J Gastroenterol 2014;109:292-293.
- Kumar N, Bashar Q, Reddy N, et al. Cyclic vomiting syndrome (CVS): Is there a difference based on onset of symptoms - pediatric versus adult? BMC Gastroenterol 2012;12:52.
- Nakazato Y, Tamura N, Shimazu K. An adult case of cyclic vomiting syndrome successfully responding to valproic acid. J Neurol 2008;255:934-935.
- Martinez-Esteve Melnikova A, Schappi MG, Korff C. Riboflavin in cyclic vomiting syndrome: Efficacy in three children. Eur J Pediatr 2016;175:131-135.
- Sorensen CJ, DeSanto K, Borgelt L, et al. Cannabinoid hyperemesis syndrome: Diagnosis, pathophysiology, and treatment — a systematic review. J Med Toxicol 2017;13:71-87.
- Hickey JL, Witsil JC, Mycyk MB. Haloperidol for treatment of cannabinoid hyperemesis syndrome. Am J Emerg Med 2013;31:1003.e5-1003.e6
- Richards JR, Gordon BK, Danielson AR, Moulin AK. Pharmacologic treatment of cannabinoid hyperemesis syndrome: A systematic review. Pharmacotherapy 2017;37:725-734.
- Jones JL, Abernathy KE. Successful treatment of suspected cannabinoid hyperemesis syndrome using haloperidol in the outpatient setting. Case Rep Psychiatry 2016;2016:3614053.
It is important for emergency providers to understand the pathophysiology and management of chronic abdominal pain disorders. This article will review two common chronic abdominal pain disorders encountered in the ED: chronic pancreatitis and cyclic vomiting syndrome.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
