Evaluation of Hair Loss in a Primary Care Setting
November 1, 2018
Reprints
AUTHORS
Molly Buckland, DO, Dermatology Resident, PGY3, St. Joseph Mercy Dermatology, Ann Arbor, MI
Ashley Jaglowicz, DO, Dermatology Resident, St. Joseph Mercy Dermatology, Ann Arbor, MI
Daniel Stewart, DO, FAAD, Program Director, St. Joseph Mercy Dermatology, Ann Arbor, MI
PEER REVIEWER
Karl Kellawan, MD, Forefront Dermatology, Centerville, OH
EXECUTIVE SUMMARY
Although hair loss is a common complaint by both male and female patients, establishing the cause and treatment of hair loss can be challenging for primary care physicians.
- A focused history consists of the nature of the hair loss, the patient’s medical history including medications and/or supplements, hair styling practices, and family history.
- A hair pull test and scalp biopsy may be helpful in narrowing the differential diagnosis.
- Non-scarring alopecias are classified as diffuse, focal, or patterned.
- Scarring alopecias often are difficult to manage and may warrant referral, regardless of the underlying cause.
Hair loss is a common complaint for both male and female patients, who often first present to their family physician for evaluation. However, the vast array of underlying etiologies can present a diagnostic challenge for physicians. Reviewing the main principles of hair biology will help provide a better understanding of the basic science behind hair loss. One of the most important steps in patient evaluation is conducting a thorough history and physical exam. This article will discuss key questions to ask, as well as how to interpret exam findings. In general, hair loss is categorized into scarring alopecia, non-scarring alopecia, and structural hair disorders. This article will focus on the most common causes of scarring and non-scarring alopecia that will be encountered in a primary care setting, as well as first-line treatments for each. Patients with non-scarring alopecia often can be managed by their family physician, while patients with scarring hair loss typically warrant referral to a dermatologist.
Introduction
There are approximately 100,000 hairs on the average scalp, with 10% more in blondes and 10% less in redheads.1 Each hair is a component of a pilosebaceous unit that includes the hair follicle, the arrector pili muscle, and the sebaceous gland. The follicle is subdivided into three portions: the infundibulum, the isthmus, and the bulb, from superficial to deep. The infundibulum is the portion of the follicle from the epidermis to the insertion of the sebaceous gland, while the isthmus is the portion between the insertion of the sebaceous gland and the insertion of the arrector pili muscle.2 Adjacent to the insertion of the arrector pili muscle is a region referred to as the bulge of the hair follicle. This is the location of hair follicle stem cells that are essential for hair growth.3 Everything beneath the insertion of the arrector pili muscle is referred to as the bulb. The infundibulum and isthmus are the permanent portions of the follicle, while the bulb is the transient portion that undergoes cyclical involution and regeneration.
The hair growth cycle is divided into anagen, catagen, and telogen growth stages. The anagen phase is the active phase of hair growth; 85-90% of all scalp hairs are in this phase, which lasts from three to five years.4 During the catagen phase, the cells undergo apoptosis and involution of the hair bulb occurs. Only 1% of scalp hairs are in this phase, which lasts for about three weeks. The telogen phase also is known as the resting or “shedding” phase, and 10-15% of hairs on the scalp are normally in this phase, which lasts for three to five months. The average person will lose 100 to 150 hairs daily. The percentage of hairs in these phases can be altered by outside factors, which will be discussed in later sections.
There are two main types of hair follicles on the body: terminal and vellus hairs. Terminal hairs are larger in diameter, with their bulb extending down to the subcutaneous fat, while vellus hairs are much smaller, with their bulb located in the mid dermis. Clinically, terminal hairs are coarse while vellus hairs are short, fine, and nonpigmented. At birth, terminal hairs are located on the scalp, eyebrows, and eyelashes, and vellus hairs populate the rest of the hair-bearing surfaces. During puberty, increasing androgen levels stimulate vellus hairs to become terminal hairs in several locations, such as the genital area and axilla.5
Hair loss is a common complaint for both male and female patients. It can cause significant psychological distress, and the diagnosis often is challenging. Hair loss can be a manifestation of a variety of disorders, including hormonal imbalance, nutritional deficiency, infections, medications, hair styling practices, autoimmune disease, inflammatory conditions, and inherited disorders. This article will discuss how to evaluate a patient presenting with a complaint of hair loss appropriately, with a focused history and physical exam. Furthermore, we will discuss the most common causes of hair loss with important key findings, including how to determine whether the hair loss is primarily a scarring (cicatricial) or non-scarring process. Laboratory workup and first-line treatment options that are appropriate for use in a primary care setting will be discussed. Further complex medication management will not be discussed, because this would indicate referral to a specialist.
History and Physical Exam
Because of the wide variety of underlying causes of hair loss, it is important to start the evaluation with a thorough history and physical exam. When evaluating a new patient with hair loss, the interview should focus on several key areas. The nature of hair loss, the patient’s medical history, current medications and/or supplements, hair care practices, and family history all should be discussed. It also is important to consider the patient’s age and race, as these can be useful clues to narrow the differential diagnosis. Since a complete interview can be time consuming, consider having a questionnaire for new patients to complete prior to being evaluated. Key questions and considerations are listed below. See Table 1 for an example of a patient questionnaire handout.
Table 1. Sample Patient Questionnaire for Evaluation of Hair Loss |
|
What is your specific hair complaint? ________Shedding _______Thinning ________Breaking ________Not growing _______Excess body hair ________Loss of body hair Length of time of the current episode of hair loss? ________Days __________Weeks ________Months ________Years Is the hair loss getting worse or is it stable? __________ How old were you when you first started losing hair? ______ Do you have a family history of hair loss? Yes / No Where is your hair loss located? _______Front scalp ______Back scalp _______Sides of scalp _______Everywhere _______Bald patches Symptoms _____Itching _____Burning ______Pain _____Flaking _____Pimples _____Bumps Hair care practices Natural hair color: __________________ Natural hair texture: ______Straight ______Wavy ______Curly Past hair care: _____Perm _____Straightening _____Coloring ____Blow dryer _____Hot iron _____Hot rollers ______Other Previous treatments for hair loss? _________________________________________________________________________ _________________________________________________________________________ Medication changes? (i.e., new medications, including supplements) _________________________________________________________________________ _________________________________________________________________________ Have you had any of the following in the last six months? ________ Serious illness or infection ________ Emotional stress ________ Hospitalization ________ Extreme weight loss ________ Surgery ________ Pregnancy ________ Initiation/discontinuation ________ Vaccinations |
The patient interview should begin with questions about the primary complaint. The two most common chief complaints are excessive shedding and progressive thinning of the hair. Other specific complaints may be that the hair is slow growing, not growing at all, breaking, or changing in texture, or the patient may have increased or decreased body hair. If a patient is complaining of hair shedding, differentiating between hair shaft breakage and hair shedding from the root is important. Hair breakage can be caused by tinea capitis, trichotillomania, damage from hair styling, structural shaft disorders, and anagen effluvium. Hair shedding from the root can be caused by telogen effluvium, alopecia areata, androgenetic alopecia, and medications. The duration and rate of hair loss should be evaluated, including when the episode of hair loss started and if it is worsening, improving, or stable. The pattern of loss also should be considered; common patterns are diffuse, localized, or patchy. Assessing which areas of the scalp are affected most also can help narrow down a cause. The extent of hair loss can be quantified by asking the patient what percentage of hair they have lost, as thinning usually is noticeable only after losing > 50% of the normal scalp hair.6 Some patients may notice a thinning ponytail, increased distance of parted hair, increased visibility of the scalp through the hair, or inability to wear their normal hairstyle.
The presence of associated symptoms may help differentiate between scarring and non-scarring alopecia. Itching, soreness, and burning sensations are more frequent in scarring alopecia. In addition, changes in scalp symptoms can help diagnose disease flares and help monitor response to treatment in types of scarring alopecia.
Thoroughly evaluating the patient’s medical and surgical history as well as their medication list may be of benefit. It is important to pay attention to specific changes during the six months prior to the onset of the hair loss. Nutritional status and psychosocial stressors should be considered. Any patient presenting with diffuse hair shedding should be evaluated for common triggers that may have occurred three to six months before the onset of hair loss. Often patients will have a positive family history, especially in androgenetic alopecia, alopecia areata, and congenital hair shaft disorders. Females should be assessed for menstrual irregularities, oral contraceptive use, issues with pregnancy, and menopause, since hormonal abnormalities or fluctuations may contribute to certain types of hair loss.
Many clinicians fail to consider the hair styling practices of the patient. Mechanical and chemical damage to the hair shaft may result from improper styling techniques. It is important to evaluate current and past styling techniques including hair color, bleaching, perms, chemical straightening, and styling with hot tools (blow dryer, flat iron, or hot rollers). Inquire about specific hair product use, the frequency of shampooing, and whether the patient has had prior treatment for hair loss. Repeated tension from tight ponytails, braids, sewn-in or clip-in hair extensions, or turbans may result in traction alopecia. It is imperative to gather hair-styling history for the previous 10 years because there may be a significant length of time between the period of repeated traction to the development of hair loss.7
The first step of the physical exam includes inspection of the entire scalp for findings such as erythema, scaling, papules, pustules, and scars. The next step involves examination of the follicular ostia, which are the pinpoint openings from which the hair grows out of the scalp. The loss of follicular ostia suggests a scarring alopecia. Next, the hair shaft should be examined carefully, considering the density, the features of the hair shaft, and whether there is evidence of breakage or fragility. The hair shafts should be evaluated for caliber, length, texture, and shape. Hair density is best evaluated by assessing the space between each part and comparing this in each area of the scalp. It is also important to examine other hair-bearing areas for abnormalities of hair growth, as this can further aid in the diagnosis.
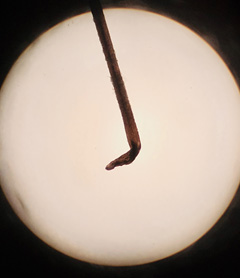
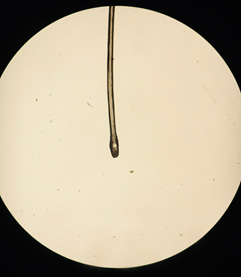
There are specific tests that may be helpful during hair examination. The hair pull test should be performed on all patients with the complaint of hair loss, as it is especially helpful in identifying active hair loss. To perform this test, grasp 50-60 strands of hair close to the scalp surface and gently apply traction by sliding fingers along the hair. The presence of more than 10% (six or more) of easily extractable hairs indicates a positive test.8 The hair pull test should be repeated on three separate areas of the scalp. The proximal ends of the extracted hairs should be examined visually or microscopically to determine the growth phase of the hair. Hairs in the anagen phase will have a pigmented bulb, while those in the telogen phase will have a white bulb. (See Figure 1.) This will help distinguish between breakage and hair shed from the scalp, as hairs that are shed will have the bulb on the proximal end. The hair shaft can be tested for fragility by grasping hair fibers in two places and pulling in opposite directions. Many clinicians visually inspect the hair under microscopy. This is especially helpful in patients with diffuse hair shedding to determine if the hair is in the anagen or telogen phase, or is broken. Trichoscopy is another technique that uses a handheld magnifier to visualize skin structures with more detail. Certain dermoscopic patterns suggestive of different types of hair loss are outside of the scope of this discussion.
Figure 1. Anagen and Telogen Hair |
|
|
Anagen hair with pigmented bulb |
Telogen hair with white bulb |
 |
 |
Laboratory Workup
Performing a scalp biopsy may be helpful to establish a diagnosis in cases that are not clear, especially in scarring alopecias. Ideally, two 4 mm punch biopsies should be taken from the periphery of the patch of hair loss. (See Figure 2.) The pathologist should examine one biopsy in the vertical section and the other in the transverse section to give a higher diagnostic yield. It is important to avoid performing a biopsy in completely bald areas, as results will be nonspecific and only show signs of scarring. The biopsy specimen should follow the direction of the hair growth and extend into the fat, where the bulbs of hair follicles usually reside.9 Laboratory studies will be guided based on the clinical presentation and suspected underlying etiology. See Table 2 for a list of the most commonly ordered labs.10,11
Figure 2. Laboratory Workup |
|
Arrows point to areas of highest diagnostic yield for a biopsy. |
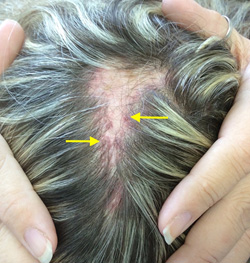 |
Table 2. Laboratory Workup in a Patient With Hair Loss |
|
Classification
In general, hair loss is categorized as scarring (cicatricial) alopecia, non-scarring alopecia, and structural hair disorders. (See Tables 3 and 4.) This article will focus on the most common causes of both scarring and non-scarring alopecia that will be encountered in a primary care setting. (See Table 5.)
Table 3. Clinical Features of the Most Common Non-scarring Alopecias |
||
Clinical Features |
Characteristics/Pearls |
|
|
Diffuse |
||
|
Telogen effluvium |
|
|
|
Anagen effluvium |
|
|
|
Patchy/Focal |
||
|
Alopecia areata |
|
|
|
Tinea capitis |
|
|
|
Trichotillomania |
|
|
|
Patterned |
||
|
Androgenetic alopecia |
|
|
|
Traction alopecia |
|
|
Table 4. Clinical Features of the Most Common Scarring Alopecias |
||
Clinical Features |
Characteristics/Pearls |
|
|
Central centrifugal cicatricial alopecia |
|
|
|
Lichen planopilaris |
|
|
|
Discoid lupus erythematosus |
|
|
|
Acne keloidalis nuchae |
|
|
|
Dissecting cellulitis of the scalp |
|
|
|
*Follicular occlusion tetrad: dissecting cellulitis of the scalp, hidradenitis suppurativa, acne conglobata, pilonidal cyst |
||
Table 5. Most Common Examples of Scarring and Non-scarring Alopecias Encountered in a Primary Care Setting |
|
Scarring alopecias |
Non-scarring alopecias |
|
|
Non-scarring Alopecias
Diffuse
Telogen effluvium. In a normal scalp, 10-15% of hairs will be in the telogen growth phase, during which approximately 100 to 150 hairs are shed each day. Telogen effluvium is defined as an increased shedding of telogen hairs that occurs in response to a variety of physiologic and pathologic conditions.12 Excessive hair shedding usually begins about three months after an inciting event. This delayed response is because of the period of time it takes for a hair follicle to finally be shed after entering the telogen phase, which lasts approximately three months. Any patient presenting with diffuse hair shedding should be evaluated for common triggers that may have occurred in the six months before the onset of hair loss. See Table 6 for a list of the most common causes. If the etiology of suspected telogen effluvium is unclear, it may be useful to evaluate for systemic causes. The workup should include thyroid and metabolic panels, iron studies, and sedimentation rate. A scalp biopsy should be considered in persistent cases lasting more than six months.
Table 6. Common Causes of Telogen Effluvium |
|
The hair loss in telogen effluvium is diffuse and involves the entire scalp. It also may involve the pubic and axillary hair. In pure telogen effluvium, there should be no evidence of inflammation both clinically and histologically. A hair pull test likely will be positive, and extracted hairs will have the characteristic white telogen bulb on the proximal end. The prognosis is favorable, and complete hair regrowth is expected; therefore, treatment is usually unnecessary. Proper counseling of expectations is extremely important for the patient’s peace of mind in these cases.
Anagen effluvium. Anagen effluvium is characterized by extensive hair loss as a result of an acute interruption of the anagen growth phase. Hair loss occurs two weeks following an inciting event, with the most common cause being initiation of chemotherapy. Anagen effluvium caused by chemotherapy usually is reversible except in some cases after treatment with busulfan or docetaxel. Other causes of anagen effluvium include radiation, mercury ingestion, thallium exposure, and severe protein malnutrition.8
Medication-induced hair loss. Medication-induced hair loss usually presents as a diffuse, non-scarring, reversible alopecia.8 It typically only affects the scalp, lacks inflammation, and is more common in women. Discontinuation of the inciting medication leads to recovery of hair in almost all cases. Most medications are associated with telogen hair loss. Common classes of medications with telogen hair loss or hair thinning as an adverse effect include psychotropics, anticoagulants, oral contraceptives containing progesterone, retinoids, antimicrobials, antithyroid medications, beta-adrenergic blocking agents, antiretrovirals, and androgen hormones.7,8 (See Table 7.)
Table 7. Medications Associated With Telogen Hair Loss |
|
Focal
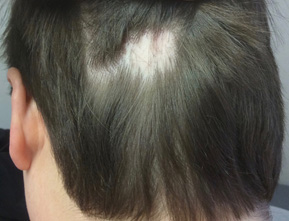
Alopecia areata. Alopecia areata is a non-scarring hair loss that is thought to be an autoimmune disease involving T lymphocytes. It most commonly presents as an abrupt onset of circular patches of alopecia without evidence of scarring. (See Figure 3.) The eyebrows and beard hair often are involved. Patients are typically young and may have a personal or family history of autoimmune disease. A characteristic feature of active disease is short hairs that are broader at the distal end than the proximal end; these are called “exclamation point” hairs. There are several other rare presentations of alopecia areata. The loss of all scalp hair is called alopecia totalis and the loss of all scalp and body hair is called alopecia universalis. The ophiasis pattern is characterized by a band-like loss of hair along the temporal and occipital scalp.13 In all cases, the hairs initially may be white or grey with regrowth but usually re-pigment with time. The nails also may be involved, with nail pitting as the most common finding.
Figure 3. Alopecia Areata |
|
Alopecia areata demonstrated by a single bald patch with normal follicular ostia. |
 |
Alopecia areata often has a chronic, relapsing nature, and disease course is difficult to predict. Treatment is largely symptomatic and does not prevent relapse. There are numerous therapies available. First-line therapy for localized disease usually involves potent topical and intralesional corticosteroids. Single patches have a good prognosis even without treatment, as 80% of patients with a single patch present for less than one year will enter remission spontaneously.14 Poor prognostic factors include early onset, persistence for more than one year, family history, associated nail changes, and the ophiasis pattern.
Infectious. Two notable infectious causes of hair loss should be considered: tinea capitis and syphilis. Dermatophyte infection of the scalp or tinea capitis occurs more commonly in children, particularly black children, and is relatively rare in adults. The clinical picture varies depending on the causative organism; however, focal or multifocal patches of alopecia with or without scale is the most common presentation. Often there are broken hairs that resemble “black dots” on exam. (See Figure 4.) Severe cases may result in formation of a kerion, which is an edematous, purulent plaque with overlying alopecia. The hair usually returns following appropriate treatment. However, the longer an infection persists, the more likely it is to cause a scarring alopecia, which is why it is important to consider and exclude a fungal infection in a patient with hair loss. The presence of posterior cervical lymphadenopathy in tinea capitis is a helpful way to differentiate it from less inflammatory causes of patchy alopecia, such as alopecia areata.7
Figure 4. Tinea Capitis |
|
Tinea capitis demonstrated by a patch of alopecia with prominent scale and black dots representing broken hairs. |
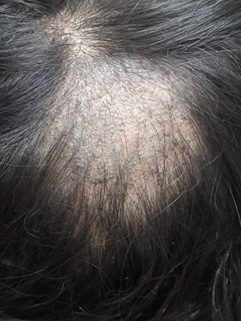 |
A rare percentage of patients with secondary syphilis present with localized, patchy, non-scarring alopecia. The characteristic clinical pattern is described as a “moth-eaten” appearance in which there are numerous, small patches of hair loss scattered over the entire scalp. Secondary syphilis also may present with diffuse hair loss. Other diseases that could present with a similar “moth-eaten” appearance include alopecia areata, tinea capitis, trichotillomania, and lupus. Although a rare finding in secondary syphilis, all sexually active patients with otherwise unexplained hair loss and a high clinical suspicion should be tested for syphilis. Hair regrowth usually occurs within three months with appropriate treatment with penicillin G.15
Trichotillomania. Trichotillomania is a self-induced disorder in which the hair is repeatedly plucked or pulled resulting in hair breakage and hair loss. The onset of this condition is common in childhood and adolescence and often is associated with underlying psychological disorders. The hair loss is usually in irregular patches with bizarre or geometric borders that contain hairs of varying lengths. Plucking often results in fractures of the hair shaft, which causes the emerging hairs to feel rough on physical exam. There are limited randomized, controlled studies for treatment of trichotillomania. Referral to a mental health professional and cognitive behavioral therapy is recommended over pharmacologic therapy. However, multiple studies have shown the benefit of clomipramine as a pharmacologic agent.16 Selective serotonin reuptake inhibitors have been tried but with low success rates. N-acetylcysteine may be effective in reducing hair pulling symptoms.17 Occasionally this condition will resolve without treatment if the hair pulling occurs in infancy or in young children.
Patterned
Androgenetic alopecia (AGA). Also known as male or female pattern baldness, AGA is caused by a hereditary trait resulting in abnormal conversion of terminal hairs to vellus hairs. It is androgen-dependent and results from elevated dihydrotestosterone levels in both men and women. By 70 years of age, at least 80% of men and 50% of women will have AGA.18 Patients usually complain of thinning hair and do not experience significant shedding. In men, the hair loss typically is symmetric thinning of the vertex and frontotemporal scalp. (See Figure 5.) In women, the most common pattern is symmetric thinning of the crown, which creates a characteristic “Christmas tree” pattern with more loss in the frontal area. (See Figure 6.) In addition, women will have preservation of the frontal hairline. Less commonly, they also may have thinning in the more typical male patterns. Minoxidil 2% and 5% is FDA approved for male pattern hair loss, with preference given to the 5% strength. Minoxidil also is approved for female pattern hair loss, and there is no difference between the male and female versions of the solution. Oral finasteride (1 mg daily) is FDA approved for use in men and also is used off-label in postmenopausal women at doses of 2.5-5 mg daily.19 Oral spironolactone and combined oral contraceptive pills also have been shown to be effective in premenopausal women because of the antiandrogen effects. It is important to remember that appropriate contraception should be used in those of childbearing potential while taking spironolactone.7,11
Figure 5. Male Pattern Hair Loss |
|
Male pattern hair loss with bitemporal recession and thinning of the vertex scalp |
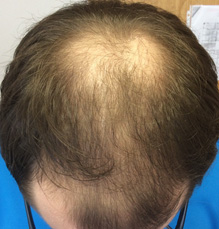 |
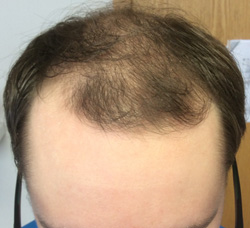 |
Figure 6. Female Pattern Hair Loss |
|
Female pattern hair loss with widening of the part in the characteristic “Christmas tree” pattern |
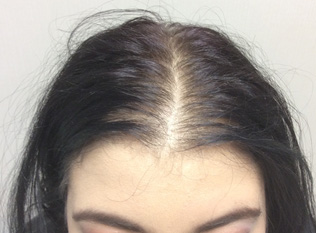 |
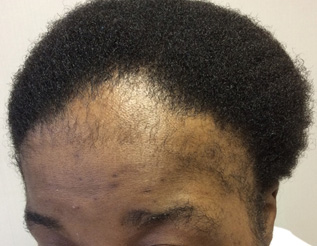
Traction alopecia. Traction alopecia results in repeated tension on the hair, usually from hair styling practices. The most common presentation is in young black females with bitemporal or frontal hair loss. (See Figure 7.) Initially, the hair loss is non-scarring, and regrowth can occur if the hair styling practices are modified. If the traction continues repeatedly for years, there is the potential for permanent loss, also called “end-stage” or “burnt-out” traction alopecia.2 Other less common causes of non-scarring alopecia include loose anagen syndrome, temporal triangular alopecia, and pressure-induced alopecia.
Figure 7. Traction Alopecia |
|
Traction alopecia demonstrated by loss of the frontal and parietal hairline. Notice that follicular ostia are still present. |
 |
Scarring Alopecias
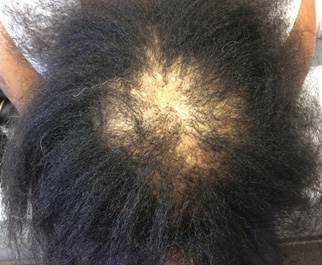
Central centrifugal cicatricial alopecia (CCCA). Formerly known as “hot comb alopecia,” this type of hair loss is the most common form of scarring alopecia in black patients, especially females.20 Although the cause is not known completely, the current hypothesis is that it results from an anatomic abnormality that predisposes these patients to follicular damage.21 It often presents as a patch of hair loss on the crown or vertex scalp that expands in a roughly symmetric, centrifugal pattern. (See Figure 8.) The greatest disease activity occurs in the peripheral zone that surrounds the patch of alopecia.22 Patients may complain of mild burning or tenderness in the involved areas. Caustic hair products, such as hot combs or chemical relaxers, may aggravate the disease or hasten its progression. Thus, it is important to counsel patients to stop all traumatic hair practices.23 Treatment with a high-potency topical steroid (clobetasol or fluocinonide) plus a long-acting oral tetracycline (doxycycline or minocycline) usually is sufficient to halt disease progression.24 It is important to counsel patients that prolonged treatment is necessary, often for several years. For highly inflammatory cases that present with pustules, referral to a specialist is recommended.
Figure 8. Central Centrifugal Cicatricial Alopecia (CCCA) |
|
CCCA demonstrated by a round patch of alopecia with loss of follicular ostia on the vertex scalp in a black female |
 |
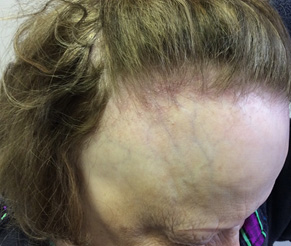
Lichen planopilaris (classic, frontal fibrosing alopecia, Graham-Little syndrome). Lichen planopilaris most commonly affects middle-aged Caucasian women between 40-60 years of age.25 Although the etiology is unknown, up to 50% of patients will have lesions of lichen planus at other sites, including nails and mucous membranes.26 The clinical presentation is widely variable, but it often presents as several foci of partial hair loss with prominent perifollicular erythema and follicular spines. Anagen hairs can be pulled out easily during the hair pull test in active areas of alopecia.27 The hair loss usually starts at the parietal scalp, and patients often complain of pruritus, a burning sensation, and/or tenderness of the affected areas.9 Frontal fibrosing alopecia (FFA) is a distinctive clinical subset that results in progressive hair loss from the anterior hairline of the frontal scalp. (See Figure 9.) A characteristic finding in FFA is a few isolated hairs within the band of alopecia, which is referred to as the “lonely hair sign.”28 It is important to examine the eyebrows of these patients, as the lateral eyebrow also frequently is affected.29 If a biopsy is performed, it should be taken from areas with perifollicular erythema, because this clinical finding represents active disease. Often, treatment is difficult and disappointing. First-line therapy includes topical or intralesional steroids, but patients often require additional systemic medications such as hydroxychloroquine.30
Figure 9. Frontal Fibrosing Alopecia (FFA) |
|
FFA demonstrated by loss of the anterior hairline with prominent perifollicular erythema. Notice loss of the eyebrow hair in this patient as well. |
 |
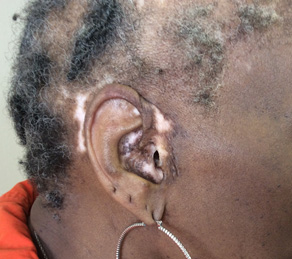
Discoid lupus erythematosus. Discoid lupus erythematosus (DLE) is classified as a type of chronic cutaneous lupus erythematosus. Most often, this type occurs independent of systemic lupus erythematosus (SLE), but patients with localized DLE above the neck have a 5-10% risk of progression to SLE.31 Early evidence of DLE is nonspecific, with patches of hair loss and variable perifollicular erythema. Eventually, these patches progress to discrete, erythematous plaques covered by an adherent scale that extends into hair follicles, resulting in follicular plugging. If this scale is removed, “carpet tack spines” may be visible underneath, representing the plugging. As the plaques continue to enlarge, there is active inflammation at the periphery, resulting in hyperpigmentation, with healing of the central portion, leaving behind atrophic, hypopigmented scars.32 The areas of alopecia vary in size, but often are round in shape, hence the name “discoid.” In addition to the scalp, the conchal bowl also frequently is involved and should be examined to help aid with diagnosis.33 (See Figure 10.) A punch biopsy taken from the active edge of the lesion is usually diagnostic. Biopsies of the central portion should be avoided because results will be nonspecific and only show evidence of scarring.
Figure 10. Discoid Lupus Erythematosus (DLE) |
|
DLE demonstrated by areas of scarring hair loss on the scalp. Notice the prominent hypopigmented patches affecting the scalp and conchal bowl of the ear. |
 |
Further screening labs to evaluate for SLE should be considered based on a full review of systems, and should include an ANA, CBC, CMP, and possible complement studies. Treatment includes photoprotection, with broad spectrum sunscreen and protective hats and clothing. For localized cases, a topical potent steroid can be applied twice daily. If there is no improvement, intralesional triamcinolone can be injected (5-10 mg/mL). Hydroxychloroquine is the most commonly used oral therapy at a dose of
200 mg twice daily.9 However, it is important to note that patients typically require two to three months of oral therapy before efficacy can be assessed.
Acne keloidalis nuchae. Acne keloidalis nuchae classically affects young black patients, particularly men.34 While the pathogenesis is unknown, it is commonly found in association with CCCA, suggesting a related underlying cause that has not been determined. It often presents as multiple small, smooth, firm papules with scattered pustules in the occipital scalp and posterior neck. (See Figure 11.) With time, these papules tend to coalesce into hairless, keloid-like, protuberant plaques. Treatment is most efficacious if initiated during the papular phase. The standard therapy includes a strong topical steroid such as clobetasol, and a long-acting oral antibiotic such as doxycycline.35 Patients also should avoid close shaving or trimming of the posterior hairline, as well as avoid irritation from tight-fitting hats or helmets.
Figure 11. Acne Keloidalis Nuchae |
|
Acne keloidalis nuchae demonstrated by multiple smooth, firm papules on the occipital scalp. |
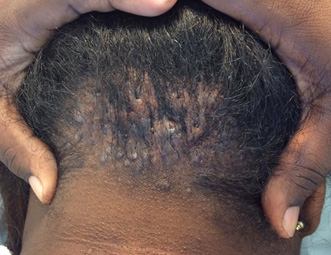 |
Dissecting cellulitis of the scalp. While uncommon, dissecting cellulitis is a distinct clinical entity most commonly seen in young adult black men between 20-40 years of age.35 The pathogenesis is secondary to follicular hyperkeratosis that results in occlusion of the follicle with accumulation of sebaceous and keratinous material. Eventually this material is released into the dermis, which triggers an inflammatory reaction followed by sinus tract formation and often bacterial superinfections.35 Dissecting cellulitis is part of the “follicular occlusion tetrad,” which also includes hidradenitis suppurativa, acne conglobate, and pilonidal cysts. Lesions present as firm nodules found on the mid vertex and upper occipital scalp. The nodules rapidly develop into interconnecting, boggy, fluctuant ridges that eventually discharge purulent material. The lesions typically are asymptomatic, and patients present complaining of hair loss and a foul-smelling discharge. However, some patients have significant pain. Systemic medications frequently are required for treatment, and in some cases a biologic medication may be required. Patients should be referred to a specialist for management of this condition.
Other less common causes of scarring alopecia that will not be discussed include erosive pustular dermatosis of the scalp, alopecia mucinosa, pseudopelade of Brocq, end-stage alopecia (burnt out), folliculitis decalvans, and end-stage traction alopecia.
Acquired/Inherited
Acquired and inherited structural disorders can result in brittle or fragile hair that easily breaks or has the appearance of failure to grow. Two of the most common acquired structural hair abnormalities are acquired trichorrhexis nodosa and trichoptilosis (commonly referred to as “split ends”), both of which may occur as a result of harmful hair care practices, such as chemical processing, excessive brushing or teasing of hair, or the application of excessive heat.36
Examples of genetic conditions that cause hair fragility and excessive hair breakage include Menkes disease, monilethrix, trichothiodystrophy, trichorrhexis invaginata, and numerous others. Because of the rarity of these disorders, further discussion is outside the scope of this article.
When to Refer
Ultimately, if the underlying cause of hair loss is unclear, patients should be referred to a dermatologist. In addition, patients with extensive alopecia areata may benefit from referral to a dermatologist for more intensive therapies than were discussed in this article. Patients with scarring hair loss often are difficult to manage and may warrant referral regardless of the underlying cause.
REFERENCES
- Farvolden D, Mahalingham M. The structure of the human hair follicle. J Am Acad Dermatol 2007;56:717.
- Childs JM, Sperling LC. Histopathology of scarring and nonscarring hair loss. Dermatol Clin 2013;31:43-56.
- Myung P, Ito M. Dissecting the bulge in hair regeneration. J Clin Invest 2012;122:448-454.
- Price VH. Treatment of hair loss. N Engl J Med 1999;341:964-973.
- Hiort O. Androgens and puberty. Best Pract Res Clin Endocrinol Metab 2002;16:31-41.
- Mubki T, Rudnicka L, Olszewska M, Shapiro J. Evaluation and diagnosis of the hair loss patient: Part I. History and clinical examination. J Am Acad Dermatol 2014;71:415.e1-415.e15.
- Sperling L, Sinclair R, el Shabrawi-Caelen L. Alopecias. In: Bolognia JL, Schaffer JV, Cerroni L, eds. Dermatology. Vol. 1, 3rd ed. Philadelphia: Elsevier Saunders; 2012: 1093-1109.
- Patel M, Harrison S, Sinclair R. Drugs and hair loss. Dermatol Clin 2013;31:67-73.
- Ross EK, Tan E, Shapiro J. Update on primary cicatricial alopecias. J Am Acad Dermatol 2005;53:1-37; quiz 38-40.
- Gordon KA, Tosti A. Alopecia: Evaluation and treatment. Clin Cosmet Investig Dermatol 2011;4:101-106.
- Olsen EA, Messenger AG, Shapiro J, et al. Evaluation and treatment of male and female pattern hair loss. J Am Acad Dermatol 2005;52:301-311.
- Kligman AM. Pathologic dynamics of human hair loss. I. Telogen effuvium. Arch Dermatol 1961;83:175-198.
- Gilhar A, Etzioni A, Paus R. Alopecia areata. N Engl J Med 2012;366:1515-1525.
- Brzezinska-Wcislo L, Bergler-Czop B, Wcisto-Dziadecka D, Lis-Swiety A. New aspects of the treatment of alopecia areata. Postepy Dermatol Alergol 2014;31:262-265.
- Qiao J, Fang H. Moth-eaten alopecia: A sign of secondary syphilis. CMAJ 2013;185:61.
- McGuire JF, Ung D, Selles RR, et al. Treating trichotillomania: A meta-analysis of treatment effects and moderators for behavior therapy and serotonin reuptake inhibitors. J Psychiatr Res 2014;58:76-83.
- Grant JE, Odlaug BL, Kim SW. N-acetylcysteine, a glutamate modulator, in the treatment of trichotillomania: A double-blind, placebo-controlled study. Arch Gen Psychiatry 2009;66:756-763.
- Gan DC, Sinclair RD. Prevalence of male and female pattern hair loss in Maryborough. J Investig Dermatol Symp Proc 2005;10:184-189.
- Brough KR, Torgerson RR. Hormonal therapy in female pattern hair loss. Int J Womens Dermatol 2017;3:53-57.
- Herskovitz I, Miteva M. Central centrifugal cicatricial alopecia: Challenges and solutions. Clin Cosmet Investig Dermatol 2016;9:175-181.
- Sperling LC, Sau P. The follicular degeneration syndrome in black patients. ‘Hot comb alopecia’ revisited and revised. Arch Dermatol 1992;128:68-74.
- Suchonwanit P, Hector CE, Bin Saif GA, McMichael AJ. Factors affecting the severity of central centrifugal cicatricial alopecia. Int J Dermatol 2016;55:e338-343.
- Gathers RC, Lim HW. Central centrifugal cicatricial alopecia: Past, present, and future. J Am Acad Dermatol 2009;60:660-668.
- Fu JM, Price VH. Approach to hair loss in women of color. Semin Cutan Med Surg 2009;28:109-114.
- Assouly P, Reygagne P. Lichen planopilaris: Update on diagnosis and treatment. Semin Cutan Med Surg 2009;28:3-10.
- Tang L, Sundberg JP, Lui H, Shapiro J. Old wine in new bottles: Reviving old therapies for alopecia areata using rodent models. J Investig Dermatol Symp Proc 2003;8:212-216.
- Kang H, Alzolibani AA, Otberg N, Shapiro J. Lichen planopilaris. Dermatol Ther 2008;21:249-256.
- Tosti A, Miteva M, Torres F. Lonely hair: A clue to the diagnosis of frontal fibrosing alopecia. Arch Dermatol 2011;147:1240.
- MacDonald A, Clark C, Holmes S. Frontal fibrosing alopecia: A review of 60 cases. J Am Acad Dermatol 2012;67:955-961.
- Chiang C, Sah D, Cho BK, et al. Hydroxychloroquine and lichen planopilaris: Efficacy and introduction of Lichen Planopilaris Activity Index scoring system. J Am Acad Dermatol 2010;62:387-392.
- Chong BF, Song J, Olsen NJ. Determining risk factors for developing systemic lupus erythematosus in patients with discoid lupus erythematosus. Br J Dermatol 2012;166:29-35.
- Rowell NR. Laboratory abnormalities in the diagnosis and management of lupus erythematosus. Br J Dermatol 1971;84:210-216.
- Elman SA, Joyce C, Nyberg F, et al. Development of classification criteria for discoid lupus erythematosus: Results of a Delphi exercise. J Am Acad Dermatol 2017;77:261-267.
- Kelly AP. Pseudofolliculitis barbae and acne keloidalis nuchae. Dermatol Clin 2003;21:645-653.
- Shapero J, Shapero H. Acne keloidalis nuchae is scar and keloid formation secondary to mechanically induced folliculitis. J Cutan Med Surg 2011;15:238-240.
- Sisto T, Bussoletti C, D’Amore A, et al. Inability to grow long hair: A presentation of trichorrhexis nodosa. Cutis 2015;95:E15-16.
Hair loss is a common complaint for both male and female patients, who often first present to their family physician for evaluation. However, the vast array of underlying etiologies can present a diagnostic challenge for physicians. Reviewing the main principles of hair biology will help provide a better understanding of the basic science behind hair loss. One of the most important steps in patient evaluation is conducting a thorough history and physical exam. This article will discuss key questions to ask, as well as how to interpret exam findings. In general, hair loss is categorized into scarring alopecia, non-scarring alopecia, and structural hair disorders. This article will focus on the most common causes of scarring and non-scarring alopecia that will be encountered in a primary care setting, as well as first-line treatments for each.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
