Update on Urinary Tract Infections in Children: What’s New in 2019?
March 1, 2019
Reprints
AUTHORS
Andrea Fang, MD, Clinical Assistant Professor of Emergency Medicine, Stanford School of Medicine, Stanford, CA
Jamie Everett, MD, Resident, Stanford/Kaiser Emergency Medicine Residency Program, Stanford, CA
N. Ewen Wang, MD, Professor of Emergency Medicine, Stanford School of Medicine, Stanford, CA
PEER REVIEWER
Katherine Baranowski, MD, FAAP, FACEP, Chief, Division of Pediatric Emergency Medicine, Department of Emergency Medicine, New Jersey Medical School, Rutgers, The State University of New Jersey, Newark
EXECUTIVE SUMMARY
- A urinary tract infection (UTI) is an infection involving the bladder or kidneys, known as cystitis and pyelonephritis, respectively. In infants and nonverbal children, UTIs generally refer to both cystitis and pyelonephritis, since differentiating between the two in the younger population can be difficult. In older children, this may be easier to differentiate, since pyelonephritis can be associated with flank pain or white blood cell clumps on urinalysis.
- In the pediatric population, females are at greater risk of developing UTIs after approximately 6 months of age. However, in neonates, males have a higher incidence of UTIs. This is especially true if the boy is uncircumcised, which poses a significant risk factor for UTIs in male infants.
- Clinicians should obtain a full septic workup in all febrile and hypothermic (temperature of < 36.0° C or 96.8° F) neonates. This includes urine studies, blood work including cultures, and cerebral spinal fluid studies to rule out meningitis.
- Per guidelines from the American Academy of Pediatrics, risk factors for UTIs in males aged 2-24 months include nonblack race, temperature ≥ 39° C, fever ≥ 24 hours (vs. 48 hours in females), and the absence of another source of infection. A patient with two or fewer risk factors (excluding circumcision) is deemed low risk, with < 1% chance of a UTI.
- The consensus is that cultures should be sent on any specimen received via catheterization or suprapubic aspiration. In addition, any positive findings on urine dipstick or urinalysis obtained via clean catch or bag specimen in patients younger than 24 months of age should have cultures sent via a catheterized specimen. The decision to treat can be based on provider discretion. However, of patients who screen positive, providers should treat those who are nitrite positive, have a high degree of pyuria, or are high-risk (e.g., a 50-day-old infant) and might have risk of decompensating before the cultures are returned.
Urinary tract infections (UTIs) are a potential cause of fever in the pediatric patient. Early testing and a timely diagnosis are critical to avert complications and potential scarring of the kidneys. Complication rates in children younger than 90 days of age include bacteremia, meningitis, and urosepsis. The authors present a concise review of the current status of the diagnosis and management of pediatric UTIs.
— Ann M. Dietrich, MD, FAAP, FACEP
Introduction
Urinary tract infections (UTIs) are a common diagnosis during pediatric emergency department (ED) visits. It is estimated that annually almost 400,000 pediatric visits to the ED involve a UTI diagnosis and that the annual nationwide economic burden of pediatric UTIs is more than $180 million. Although the number of visits with a UTI diagnosis is increasing, overall admissions for UTIs and their complications are decreasing.1
UTIs are the second most common cause of infection diagnosed in children in the ED after general viral illnesses.2 They are also the most common cause of serious bacterial infections in infants, especially as the incidence of bacteremia and meningitis has decreased with the introduction of the Streptococcus pneumoniae and Haemophilus influenzae type b vaccines.3,4 If untreated, UTIs can lead to urosepsis, and have been shown to cause renal scarring.5-7 While there are concerns that renal scarring may place patients at risk for hypertension or chronic kidney disease, some have argued that there are no actual long-term sequelae from renal scarring.8,9
ED providers may struggle to decide when to test for a UTI in a well-appearing pediatric patient. This is especially true in younger, nonverbal patients who are unable to express symptoms of discomfort and in children who are not yet toilet-trained and often require a urinary catheterization.
This article will discuss the pathophysiology and common causes of pediatric UTIs, as well as how UTIs may present differently in different aged pediatric populations through various symptoms, physical exam findings, and risk factors. In 2016, the American Academy of Pediatrics (AAP) published practice guidelines for the diagnosis and management of UTIs in infants 2-24 months old that reaffirmed many of their 2011 practice recommendations.3,10 This article also will review various ways to work up UTIs and interpret urine tests; discuss whether a bag specimen provides an adequate sample collection; identify which results on a urinalysis or urine dipstick warrant treatment without needing to wait for a culture to confirm the diagnosis; provide various treatment options; and recommend which pediatric patients with UTIs warrant admission.
Of note, complicated conditions such as the immunosuppressed, the ill-appearing septic patient, and in-depth discussion of neonatal fever are beyond the scope of this article.
Pathophysiology and Etiology
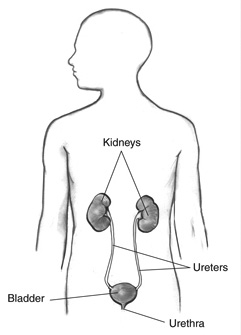
A UTI is an infection involving the bladder or kidneys, known as cystitis and pyelonephritis, respectively. While the urethra is part of the urinary tract, urethritis is associated more commonly with sexually transmitted diseases and is not considered a UTI in the traditional sense. In infants and nonverbal children, UTIs generally refer to both cystitis and pyelonephritis, since differentiating between the two in the younger population can be difficult. In older children, this may be easier to differentiate, as pyelonephritis can be associated with flank pain or white blood cell clumps on urinalysis. (See Figure 1.)
Figure 1. Urinary Tract Anatomy |
 |
|
Source: National Institute of Diabetes and Digestive and Kidney Diseases. The Urinary Tract & How It Works. Available at: https://www.niddk.nih.gov/health-information/urologic-diseases/urinary-tract-how-it-works. Accessed Jan. 6, 2019. |
The most common cause of UTIs is ascension of bacteria through the urethra. Given its presence in bowel flora, Escherichia coli is the most common pathogen, with some studies noting a prevalence of 83-93%. Other less common pathogens include Proteus mirabilis, Klebsiella, Enterobacter, Pseudomonas aeruginosa, Staphylococcus saprophyticus, Enterococcus, and Citrobacter.11-14
Given their shorter urethra, adult females are at greater risk than males for developing UTIs. In the pediatric population, females are at greater risk of developing UTIs after approximately 6 months of age.15 However, in neonates, males have a higher incidence of UTIs.16 This is especially true if the boy is uncircumcised, which poses a significant risk factor for UTIs in male infants.
Sexual intercourse also will increase a female’s likelihood of developing a UTI. Thus, an adolescent female with her first UTI should prompt a provider to inquire about sexual activity to ensure safe sex practices.
History, Physical Exam, and Risk Factors
For nonverbal infants, fever often is the primary symptom of a UTI. Patients also may have vomiting, increased irritability, hematuria, or a change in urinary output. It is helpful to ask parents if there has been a history of UTIs in the past. In male infants, it also is important to ask if there is a history of circumcision and examine the patient’s genitals. On physical exam, suprapubic tenderness also has been shown to correlate with a higher likelihood of having a UTI.12
In verbal children, the suspicion of a UTI may be guided by the history and physical exam. One study showed symptoms such as abdominal pain, dysuria, urinary frequency, or incontinence were associated with an increased chance of a UTI; the same study did not find an association with UTI and malodorous urine.12 Children also may note vomiting, back pain, or hematuria.
On the physical exam in older pediatric patients, costovertebral tenderness could be suggestive of pyelonephritis, and suprapubic tenderness also may increase the likelihood of a UTI by a factor of four.12 Of note, 50% of adult women with at least one UTI symptom will have a UTI. Thus, symptoms alone may give providers enough confidence to empirically treat an adult woman for a UTI without sending official urine studies.17 However, this is not the case for the pediatric population in which patients with urinary symptoms have only a 7.8% prevalence of a UTI.15 Thus, providers should send urine studies on pediatric patients and not just treat patients empirically based on symptoms.
Well-Appearing Febrile Infants and the Need for a UTI Workup
Since fever (defined as a temperature of at least 38.0° C or 100.4° F) often is the sole objective sign of a UTI in young infants, the question often arises as to when to order urine studies, especially if viruses are the number one cause of fevers. Approach these patients based on their age in conjunction with AAP guidelines. (See Table 1.)
Table 1. Age-Based Indications to Screen for a Urinary Tract Infection in Well-Appearing Patients and Treatment Options |
||
Age |
Signs and Symptoms |
Treatment Options* |
|
0 to 28 days |
Fever/hypothermia Unexplained jaundice |
Inpatient: Ampicillin and gentamicin |
|
29 to 59 days |
Fever |
Inpatient: Ceftriaxone Outpatient: Oral cephalosporin |
|
2 months to 2 years |
||
|
Female |
Fever with ≤ 1 risk factor Risk factors include:3
|
Oral cephalosporin |
|
Male, uncircumcised |
Febrile <12 months of age |
Oral cephalosporin |
|
Male, circumcised |
Febrile < 6 months of age OR Fever with ≤ 2 risk factors Risk factors include:3
|
Oral cephalosporin |
|
> 2 years |
Urinary symptoms, abdominal pain, or prolonged unexplained fever |
Oral cephalosporin |
|
* Please consult your local antibiogram for the appropriate first-line medications based on regional susceptibilities. |
||
Neonates
For neonates up to 28 days, the recommendations are straightforward. Clinicians should obtain a full septic workup in all febrile and hypothermic (defined as a temperature of < 36.0° C or 96.8° F) neonates. This includes urine studies, blood work including cultures, and cerebral spinal fluid (CSF) studies to rule out meningitis. The prevalence of UTIs in febrile neonates is 15.4%.18
Infants: 29-60 Days of Age
Given the prevalence of UTIs in young infants, we recommend obtaining urine samples in febrile infants younger than 60 days of age. The need for additional tests, such as blood and CSF studies, in well-appearing infants in this age range has been an ongoing debate within the pediatric community. However, at minimum, obtain a urinalysis and culture.
Infants: 2-24 Months of Age
In febrile infants 2-24 months of age, the prevalence of a UTI is approximately 5-7%.3,15 The AAP guidelines note specific risk factors for UTIs based on the infant’s gender to help providers determine whether they should screen for a UTI. (See Table 2.)
Table 2. Probability of UTI in Febrile Infants 2-24 Months of Age Based on Gender and Risk Factors3 |
|||
Gender |
Risk Factors |
UTI Probability |
|
|
< 1% |
< 2% |
||
|
Female |
|
≤ 1 risk factor |
≤ 2 risk factors |
|
Circumcised Male |
|
≤ 2 risk factors |
≤ 3 risk factors |
|
Uncircumcised Male |
Uncircumcised febrile males’ probability of UTI is greater than 1% just from being uncircumcised, even without taking into account other risk factors. |
||
For female infants, risk factors included white race, age < 12 months, temperature ≥ 39° C, fever ≥ 2 days, and no other infectious source. If a female infant has only one risk factor, her probability of a UTI is less than 1% and watchful waiting is a reasonable plan.
For male infants, by far the greatest risk factor for a UTI is being uncircumcised.3 For febrile male infants younger than 3 months of age, the prevalence of a UTI in uncircumcised and circumcised males was 20.1% and 2.4%, respectively.15 This validates the importance of testing for a UTI in febrile male infants younger than 3 months of age, especially if they are uncircumcised. The rate of UTIs in febrile male infants declines after three months, with a prevalence of 7.3% and 0.3% at ages 6-12 months for uncircumcised and circumcised males, respectively.15 After 12 months of age, the risk of a UTI in a circumcised male is low enough that testing can be based on clinical judgment and review of other risk factors. Per AAP guidelines, risk factors for UTIs in males aged 2-24 months include nonblack race, temperature ≥ 39° C, fever ≥ 24 hours (vs. 48 hours in females), and absence of another source of infection. A patient with two or fewer risk factors (excluding circumcision) was deemed low risk, with < 1% chance of a UTI. (See Table 2.)
Unique Risk Factors
Neonates
Among neonates, jaundice has been associated with urinary tract infections. Ozdogan et al found that 12.2% of infants between 2 and 14 days of age with elevated indirect bilirubin above phototherapy limits had UTIs. Of those patients with UTIs, 6.2% of patients had bacteremia.19 The question then is when to test for a UTI when a neonate may have neonatal jaundice that simply warrants phototherapy. Although the topic is somewhat controversial, consider testing for UTIs in asymptomatic neonates with jaundice and elevated indirect bilirubin above phototherapy limits, patients with prolonged bilirubin not responding as expected to phototherapy, or patients with unexplained jaundice after 1 week of age.
Congenital Renal Abnormalities
Patients with congenital renal abnormalities are at increased risk of UTIs. Authors of a Danish study found that patients diagnosed prenatally with renal anomalies had a 19% risk of developing a UTI by 2 years of age compared to their normal cohort who had only a 1% risk. These patients also have the highest risk of sepsis (along with neonates).8 Thus, we recommend obtaining urine studies in febrile children with known renal abnormalities, especially in the absence of another source of fever.
Indwelling Foleys
For hospitalized patients, an indwelling Foley catheter or other instrumentation can put a child at risk for a UTI, most specifically from Candida, which has been shown to cause more than 50% of UTIs in a pediatric intensive care unit.20
Diagnostic Tests
Catheter vs. Clean Catch Urine vs. Bag Specimen
For children who are toilet-trained, a clean catch urine specimen is reasonable. However, parental education should emphasize the need for a mid-stream specimen to prevent contamination from genital skin flora. This contamination may be demonstrated by squamous cells in the urinalysis. However, it also may cause false-positive results on the urinalysis interpretation and subsequent urine culture.
For children who are not toilet-trained, many healthcare providers order a catheterized urine specimen because it provides a more accurate urine culture result, which is the gold standard of UTI diagnosis. However, urine via a bag or clean catch may be reasonable for a screening urinalysis or dipstick. If the results are negative, it can help rule out a UTI. However, urine from a bag or clean catch is less desirable for the urine culture and, thus, a positive urine screen often still will require a provider to obtain a catheterized specimen afterward.10
Although urethral catheterization is a low-risk procedure, it can be perceived as painful and upset infants and their families. To decrease catheterization rates, the ED at the Children’s Hospital of Philadelphia implemented a protocol in which urine bags were placed rather than obtaining catheterized specimens immediately in febrile infants 6-24 months of age. Patients with bags placed were encouraged to feed and were reassessed every 30 minutes. Patients with positive dipsticks subsequently were catheterized for the culture. In this busy, urban ED, this method saved 30% of children from experiencing a urine catheterization. This method did not change their length of stay or revisit rates, and the culture positivity rates were similar with no missed UTIs.21
More recently, suggestions also have been made to consider obtaining urine screen via unique clean catch method. In 2013, Herreros et al introduced a noninvasive method to obtain urine in neonates in the ED. This involved feeding the infant, cleaning the genitals after 25 minutes, holding the infant under the armpits, tapping the bladder 50 times in 30 seconds, and then massaging the lower paravertebral back for 30 seconds. This was repeated for up to five minutes and required two to three providers. Herreros et al showed that this clean catch method had a sensitivity of 86% and a specificity of 80% in diagnosing UTIs in patients younger than 90 days of age.22 Labrosse used this method in infants younger than 6 months of age and found it to be more successful in patients younger than 90 days of age. While the clean catch method specimens had a higher contamination rate compared to catheterized specimens (16% vs. 6%), this was not a statistically significant difference; however, this could be due to sample size and power.23
Special Consideration: Suprapubic Aspiration
Historically, suprapubic aspirations were performed as an alternative method to obtain urine that provided accurate urine culture results. However, this is not done in the ED routinely because of providers’ lack of experience and the fact that providers usually can obtain urethra catheterized specimens. The procedure is relatively safe if done correctly and sterilely. Indications for suprapubic aspirations primarily are for acute urinary retention or urgent need for a urine specimen after an unsuccessful urethra catheterization attempt, for example, as the result of a phimosis or labial adhesions.
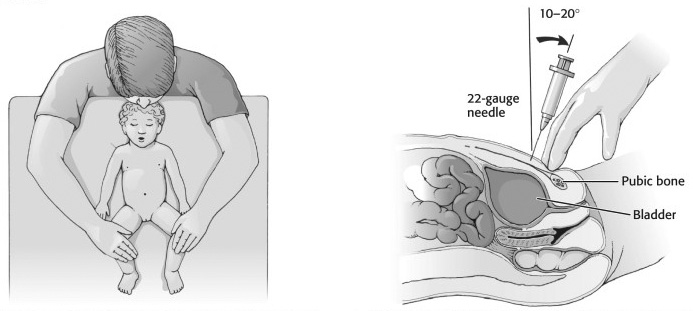
Suprapubic aspiration is performed by placing the patient in a supine frog-leg position and palpating the bladder. Visualization of the bladder with an ultrasound can help confirm the location. If desired, local anesthesia can be administered topically or subcutaneously with a small wheal to decrease patient discomfort. Attach a syringe to a 22-gauge needle at least 3.5 cm long. Then, using a sterile technique, place the needle 1 to 2 cm above the pubic symphysis at a 10- to 20-degree angle cephalad. (See Figure 2.) Apply negative pressure to the syringe as the needle advances until the urine is aspirated. Complications from suprapubic aspirations are reported minimally, and the use of ultrasound has been shown to increase the success rate.24,25
Figure 2. Suprapubic Aspiration Technique |
|
 |
|
|
1. Restrain the infant and place her in a frog-leg position. Prepare and drape the skin, and raise a skin wheal with local anesthetic to reduce discomfort. Despite the safety of this procedure, it may be disconcerting for worried parents, and they may wish to leave the room during the aspiration. |
2. Puncture the abdominal wall with a 22-gauge needle in the midline approximately 1 to 2 cm cephalad to the superior border of the pubic bone. Keep the syringe perpendicular to the plane of the abdominal wall (usually 10 to 20 degrees from the true vertical). The bladder is an abdominal organ in infants, and placing the needle too close to the pubic bone or angling toward the feet might cause the needle to miss the bladder. Localizing the bladder with bedside ultrasound facilitates this procedure. |
|
Reprinted with permission. This image was published in Roberts and Hedges' Clinical Procedures in Emergency Medicine and Acute Care. 7th ed. Davis J, Silverman M. Urologic Procedures. Copyright Elsevier 2019. |
|
Urine Studies
Urine Dipstick
Screening tests are highly sensitive so as not to miss patients with a possible infection. For urine dipsticks, the leukocyte esterase and nitrite results are of primary interest. Leukocyte esterase is a product of neutrophils and a marker of white blood cells in the urine. Nitrites are a product of bacteria that convert urinary nitrates to nitrites. Literature suggests that urine dipsticks that are positive for leukocyte esterase have a sensitivity up to 95%.26,27 Additionally, nitrites are highly specific for infection.3 Thus, patients with nitrite-positive urine dipsticks should be treated. Since urine dipsticks do not provide information on pyuria or bacteriuria, if given the option, urinalysis should be obtained rather than a urine dipstick when possible.
Urinalysis
A positive urinalysis is considered to have any leukocyte esterase, nitrites, pyuria (≥ 5 white blood cells [WBCs] per high power field), or bacteriuria. For febrile infants younger than 60 days of age, a positive urinalysis is 94% sensitive and 91% specific.28 Per the AAP guidelines, a urinalysis with any evidence of leukocyte esterase, nitrates, pyruria, or bacteriuria is almost 100% sensitive.3 Confirmation of a UTI is based on a suggestive urine analysis and a urine culture of > 50,000 colony-forming units (cfu) per milliliter of bacteria obtained from a catheterization.10 Therefore, urinalysis is an excellent screening tool, and urine cultures should be sent if there are any positive findings. Nitrites also have a very high specificity (98%), so patients with a nitrite-positive urinalysis should be treated without waiting for the culture results.
Leukocyte esterase-positive urinalysis results are not highly specific (78%), so providers should consider other factors (i.e., high-risk patient, degree of pyuria, bacteriuria) when deciding whether to treat prior to culture results. Likewise, pyuria alone has poor sensitivity (73%) and specificity (81%). Other causes of pyuria include chlamydia, appendicitis, Kawasaki’s disease, nephrolithiasis, balanitis, epididymitis, urethritis, and even exercise and viral infections.29 (See Table 3.) However, a high degree of pyuria (e.g., > 50 WBCs per high power field) in the urine in the right clinical setting should increase the suspicion of a UTI.
Table 3. Non-UTI Causes of Pyuria |
|
One benefit of urinalysis is that a completely negative study means there is < 1% chance of UTI in nonverbal patients younger than 24 months of age.3,23 We would recommend not treating the patient. If the culture is positive, treatment can be based on provider discretion, since a positive urine culture with a negative urinalysis has a reasonable chance of being a contaminant or asymptomatic bacteriuria.3
Of note, urine should be tested within one hour of obtaining the sample (or within four hours if the sample is refrigerated) to maintain the accuracy of the results and prevent bacterial growth.10
Urine Culture
A urine culture always should be sent when obtaining urine from catheterized specimens from all pediatric patients. This is because of the false-negative rates in which 9-10% of pediatric patients without pyuria later still are confirmed to have a UTI.30 This may be due to pediatric patients having less of an inflammatory response to an early UTI or not concentrating their urine as much as adults. In addition, per the 2016 AAP guidelines, a definitive diagnosis of a UTI requires a urine culture with more than 50,000 cfu/mL of bacteria obtained from a catheterization or suprapubic aspiration, along with a supporting urinalysis showing white blood cells (pyuria) or bacteria (bacteriuria).10
Although a urine culture from a catheterized sample has a high sensitivity and specificity (95% and 99%, respectively), it often takes one to two days to grow. Therefore, it is important for healthcare providers to know which urine screening results (i.e., the urine dipstick or urinalysis) should reassure them that a pediatric patient does not need treatment or what results should drive them to empirically treat for a UTI before waiting for the culture results.
The consensus is that cultures should be sent on any specimen received via catheterization or suprapubic aspiration. In addition, any positive findings on urine dipstick or urinalysis obtained via clean catch or bag specimen in patients younger than 24 months of age should have cultures sent via a catheterized specimen. The decision to treat can be based on provider discretion, although of patients who screen positive, providers should treat those who are nitrite-positive, have a high degree of pyuria, or are high-risk (e.g., a 50-day-old infant) and might have risk of decompensating before the cultures are returned.
Additional Workup
When is obtaining urine enough in evaluating a pediatric patient? Any toxic-appearing child should be worked up accordingly and is beyond the scope of this article. However, the following discussions address when to consider obtaining additional studies.
When Should I Check Blood?
All febrile neonates should have blood drawn, including blood cultures. In infants 29-60 days of age or even 29-90 days of age, the threshold to obtain blood is less clear, and providers should follow their institutional guidelines. After 60 days, well-appearing patients with confirmed UTIs do not warrant blood studies. However, patients older than 60 days of age with signs of sepsis may need blood drawn for assessment, fluid resuscitation, and/or intravenous antibiotics.
When Should I Perform a Lumbar Puncture?
Some ED providers naturally may wonder if they need to perform a lumbar puncture (LP) on a febrile neonate with a floridly positive urinalysis for a UTI. In one recent study of 236 febrile neonates 30 days of age or younger with UTIs, none had bacterial meningitis.31 In a larger recent study, Thomson et al noted that seven of 803 (0.9%) infants 28 days of age or younger with UTIs had associated bacterial meningitis (all with associated bacteremia).32 In a 2011 study, two out of 163 infants 28 days of age or younger (1.2%) had UTIs with meningitis.33 Although 1% may be considered low risk, the devastating effects of meningitis in neonates and their high-risk nature make us continue to recommend a full septic workup in febrile neonates, including performing an LP.
In febrile infants 29-60 days of age with a confirmed or suspected UTI, the decision to perform an LP may be less obvious, since there are no clear guidelines for this age group. These patients have not started their vaccination series yet, so there is concern that they are at higher risk. Studies show the incidence of meningitis in these patients to be 0-0.26%,32-34 and those who do have meningitis often are toxic appearing.34 Thus, a documented UTI alone is not an indication to obtain an LP in an infant 29-60 days of age. In fact, UTI has been associated with sterile CSF pleocytosis (18% of UTIs) without clinically significant adverse effects.35
Should I Order a Renal and Bladder Ultrasound?
The AAP recommends performing a renal and bladder ultrasound to assess for renal scarring and vesicoureteral reflux in patients 2-24 months of age with UTIs.10 However, this does not need to be done emergently in the ED and can be deferred to the patient’s primary care provider. In a pediatric patient with a confirmed UTI and a persistent fever or highly suggestive urinalysis despite several days of treatment, providers should obtain a renal ultrasound to rule out a renal abscess.
Should I Send Any Other Studies?
Although it is easy to focus on the infant with a UTI, it is important to remember that a first-time UTI in an adolescent patient often is a sign of the start of sexual activity. Thus, it is important to discuss safe sex practices and investigate whether studies to assess for pregnancy or sexually transmitted diseases are warranted.
Treatment and Disposition
Table 1 addresses treatment options by age in well-appearing patients with suspected UTIs. Needless to say, all febrile neonates should be admitted regardless of whether they have a UTI. As previously discussed, they should have a full septic workup and should be started on ampicillin and gentamicin.36
There are no clear guidelines about whether a febrile infant 29-60 days of age with a confirmed UTI should be admitted. In 2014, Schnadower et al investigated this question in a study involving 19 EDs; they found that the vast majority (92.5%) of febrile patients between 29-60 days of age with UTIs were admitted.37 However, of those who initially were discharged, none had adverse outcomes, although 22% did come back for admission after culture results returned. The primary factor that determined likelihood for discharge was institutional practice, with five sites having a 15-20% discharge rate and two sites admitting all the patients. It is possible that certain institutions have specific pathways or guidelines that recommend admission at that age.38 Patients admitted for parenteral antibiotics should receive a third-generation cephalosporin or aminoglycoside. For patients deemed safe for outpatient oral antibiotics, a cephalosporin is considered a reasonable first-line agent.3 However, we recommend reviewing local antibiograms for the most appropriate therapy given variation in susceptibilities based on region.
The AAP notes that a well-appearing infant 2-24 months of age with a UTI and good follow-up can be managed as an outpatient with oral antibiotics for seven to 14 days.3 A cephalosporin, such as first-generation cephalexin, is a reasonable first-line agent. Providers also could consider amoxicillin-clavulanate or trimethoprim-sulfamethoxazole. However, it is best to know local susceptibility patterns.
In patients older than 2 years of age with simple UTIs, a cephalosporin is still a reasonable first-line agent, but providers should review local antibiograms. Although adult women with uncomplicated UTIs may be prescribed an antibiotic regimen of three days or less, the standard duration of the antibiotic regimen in pediatric patients remains seven to 14 days.3,39,40
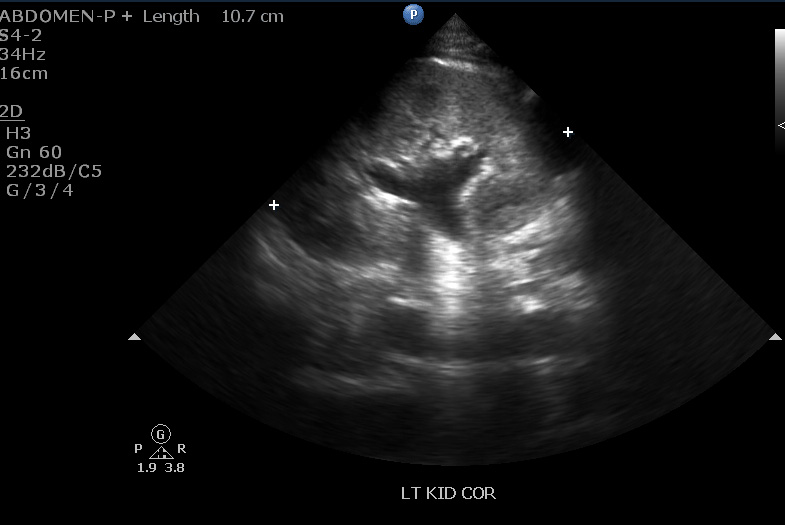
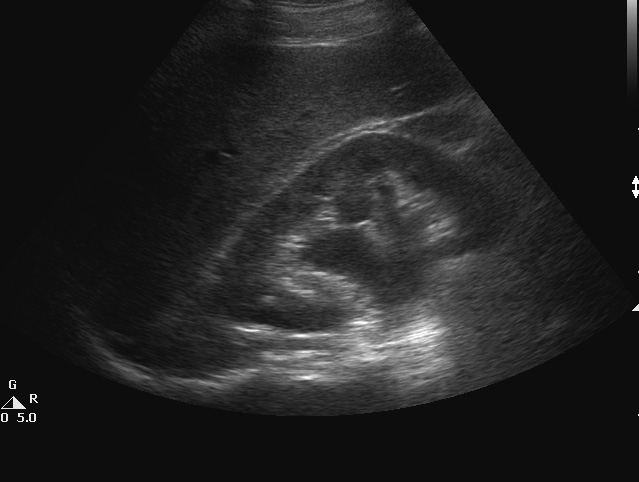
For admitted patients older than 60 days of age who warrant intravenous antibiotics, a third-generation cephalosporin, such as ceftriaxone, is a good first-line agent while awaiting the culture results. Predictors of admission include male gender, higher comorbidity status (such as from obesity or diabetes), pyelonephritis, concurrent hydronephrosis, kidney stones, or urosepsis.1 See Figures 3 and 4 for ultrasounds showing hydronephrosis. Patients who are immunosuppressed (e.g., renal transplant, immunosuppressed cancer patients) with UTIs should be admitted, although specifics about treatment are out of the scope of this article. Providers should discuss these cases with the appropriate subspecialty services.
Figure 3. Ultrasound of Kidney Showing Hydronephrosis |
 |
|
Image courtesy of Sandra L. Werner, MD, FACEP |
Figure 4. Ultrasound of Hydronephrosis |
 |
|
Image courtesy of Sandra L. Werner, MD, FACEP |
Follow-Up
The AAP recommends primary care follow-up within 48 hours for febrile infants.10 It also recommends infants younger than 24 months of age receive an ultrasound to assess for risk for renal scarring and vesicoureteral reflux, although the significance of renal scarring seems to be less certain at this point.8-10 Workup with a voiding cystourethrogram also can be deferred to the primary care provider and is no longer recommended routinely.10
CONCLUSION
Pediatric UTIs are a common cause of ED visits. Providers should obtain urine specimens in all febrile infants younger than 2 months of age. Febrile or hypothermic neonates with a UTI still warrant a full neonatal sepsis workup. In febrile infants 2-24 months of age, providers should consider the patient’s risk factors and screen for a UTI if the risk is more than 1%, per AAP guidelines. Urinalyses are preferable to urine dipsticks for screening. Initially, urine samples can be obtained via a urine bag or clean catch methods in infants. However, urine cultures are considered the gold standard necessary for diagnosis and should be obtained via a catheterized specimen in infants not yet toilet-trained. In verbal patients with symptoms associated with UTIs, urine samples should be obtained and patients should not be treated purely based on symptoms. In an adolescent female with a first-time UTI, providers should inquire about sexual activity and discuss associated risks. Treatment should be based on local susceptibility patterns, although cephalosporins often are first-line agents. Finally, patients with UTIs who are younger than 24 months of age warrant outpatient follow-up.
REFERENCES
- Sood A, Penna FJ, Eleswarapu S, et al. Incidence, admission rates, and economic burden of pediatric emergency department visits for urinary tract infection: Data from the nationwide emergency department sample, 2006 to 2011. J Pediatr Urol 2015;11:246.e1-8.
- American College of Emergency Physicians Clinical Policies Subcommittee on Pediatric Fever; Mace SE, Gemme SR, Valente JH, et al. Clinical Policy for Well-Appearing Infants and Children Younger Than 2 Years of Age Presenting to the Emergency Department With Fever. Ann Emerg Med 2016;67:625-639.e613.
- Subcommittee on Urinary Tract Infection, Steering Committee on Quality Improvement and Management, Roberts KB. Urinary tract infection: Clinical practice guideline for the diagnosis and management of the initial UTI in febrile infants and children 2 to 24 months. Pediatrics 2011;128:595-610.
- Greenhow TL, Hung YY, Herz AM, et al. The changing epidemiology of serious bacterial infections in young infants. Pediatr Infect Dis J 2014;33:595-599.
- Karavanaki KA, Soldatou A, Koufadaki AM, et al. Delayed treatment of the first febrile urinary tract infection in early childhood increased the risk of renal scarring. Acta Paediatr 2017;106:149-154.
- Shaikh N, Craig JC, Rovers MM, et al. Identification of children and adolescents at risk for renal scarring after a first urinary tract infection: A meta-analysis with individual patient data. JAMA Pediatr 2014;168:893-900.
- Shaikh N, Mattoo TK, Keren R, et al. Early antibiotic treatment for pediatric febrile urinary tract infection and renal scarring. JAMA Pediatr 2016;170:848-854.
- Newman DH, Shreves AE, Runde DP. Pediatric urinary tract infection: Does the evidence support aggressively pursuing the diagnosis? Ann Emerg Med 2013;61:559-565.
- Craig JC, Williams GJ. Denominators do matter: It’s a myth—urinary tract infection does not cause chronic kidney disease. Pediatrics 2011;128:984-985.
- Subcommittee on Urinary Tract Infection. Reaffirmation of AAP Clinical Practice Guideline: The Diagnosis and Management of the Initial Urinary Tract Infection in Febrile Infants and Young Children 2-24 Months of Age. Pediatrics 2016;138(6). pii: e20163026.
- Shaikh N, Wald ER, Keren R, et al. Predictors of non-Escherichia coli urinary tract infection. Pediatr Infect Dis J 2016;35:1266-1268.
- Shaikh N, Morone NE, Lopez J, et al. Does this child have a urinary tract infection? JAMA 2007;298:2895-2904.
- Shaw KN, Gorelick M, McGowan KL, et al. Prevalence of urinary tract infection in febrile young children in the emergency department. Pediatrics 1998;102:e16.
- Edlin RS, Shapiro DJ, Hersh AL, Copp HL. Antibiotic resistance patterns of outpatient pediatric urinary tract infections. J Urol 2013;190:222-227.
- Shaikh N, Morone NE, Bost JE, Farrell MH. Prevalence of urinary tract infection in childhood: A meta-analysis. Pediatr Infect Dis J 2008;27:302-308.
- Arshad M, Seed PC. Urinary tract infections in the infant. Clin Perinatol 2015;42:17-28, vii.
- Bent S, Nallamothu BK, Simel DL, et al. Does this woman have an acute uncomplicated urinary tract infection? JAMA 2002;287:2701-2710.
- Bonadio W, Maida G. Urinary tract infection in outpatient febrile infants younger than 30 days of age: A 10-year evaluation. Pediatr Infect Dis J 2014;33:342-344.
- Bahat Ozdogan E, Mutlu M, Camlar SA, et al. Urinary tract infections in neonates with unexplained pathological indirect hyperbilirubinemia: Prevalence and significance. Pediatr Neonatol 2018;59:305-309.
- Brindha SM, Jayashree M, Singhi S, Taneja N. Study of nosocomial urinary tract infections in a pediatric intensive care unit. J Trop Pediatr 2011;57:357-362.
- Lavelle JM, Blackstone MM, Funari MK, et al. Two-step process for ED UTI screening in febrile young children: Reducing catheterization rates. Pediatrics 2016;138(1). pii: e20153023. doi: 10.1542/peds.2015-3023. Epub 2016 Jun 2.
- Herreros ML, Tagarro A, Garcia-Pose A, et al. Performing a urine dipstick test with a clean-catch urine sample is an accurate screening method for urinary tract infections in young infants. Acta Paediatr 2018;107:145-150.
- Labrosse M, Levy A, Autmizguine J, Gravel J. Evaluation of a new strategy for clean-catch urine in infants. Pediatrics 2016;138(3). pii: e20160573. doi: 10.1542/peds.2016-0573. Epub 2016 Aug 19.
- Bozicnik S, Diez Recinos A, Moreno Canto MC, et al. [Ultrasound-guided suprapubic bladder aspiration increases the success of the technique in infants less than 4 months-old]. [Article in Spanish]. An Pediatr (Barc) 2013;78:321-325.
- Munir V, Barnett P, South M. Does the use of volumetric bladder ultrasound improve the success rate of suprapubic aspiration of urine? Pediatr Emerg Care 2002;18:346-349.
- Kanegaye JT, Jacob JM, Malicki D. Automated urinalysis and urine dipstick in the emergency evaluation of young febrile children. Pediatrics 2014;134:523-529.
- Chaudhari PP, Monuteaux MC, Bachur RG. Urine concentration and pyuria for identifying UTI in infants. Pediatrics 2016;138(5). pii: e20162370. Epub 2016 Oct 18.
- Tzimenatos L, Mahajan P, Dayan PS, et al. Accuracy of the urinalysis for urinary tract infections in febrile infants 60 days and younger. Pediatrics 2018;141(2). pii: e20173068. doi: 10.1542/peds.2017-3068. Epub 2018 Jan 16.
- Simerville JA, Maxted WC, Pahira JJ. Urinalysis: A comprehensive review. Am Fam Physician 2005;71:1153-1162.
- Yamasaki Y, Uemura O, Nagai T, et al. Pitfalls of diagnosing urinary tract infection in infants and young children. Pediatr Int 2017;59:786-792.
- Wallace SS, Brown DN, Cruz AT. Prevalence of concomitant acute bacterial meningitis in neonates with febrile urinary tract infection: A retrospective cross-sectional study. J Pediatr 2017;184:199-203.
- Thomson J, Cruz AT, Nigrovic LE, et al. Concomitant bacterial meningitis in infants with urinary tract infection. Pediatr Infect Dis J 2017;36:908-910.
- Tebruegge M, Pantazidou A, Clifford V, et al. The age-related risk of co-existing meningitis in children with urinary tract infection. PLoS One 2011;6:e26576.
- Paquette K, Cheng MP, McGillivray D, et al. Is a lumbar puncture necessary when evaluating febrile infants (30 to 90 days of age) with an abnormal urinalysis? Pediatr Emerg Care 2011;27:1057-1061.
- Schnadower D, Kuppermann N, Macias CG, et al. Sterile cerebrospinal fluid pleocytosis in young febrile infants with urinary tract infections. Arch Pediatr Adolesc Med 2011;165:635-641.
- Polin RA, Committee on Fetus and Newborn. Management of neonates with suspected or proven early-onset bacterial sepsis. Pediatrics 2012;129:1006-1015.
- Schnadower D, Kuppermann N, Macias CG, et al; American Academy of Pediatrics Pediatric Emergency Medicine Collaborative Research Committee UTI Study Group. Outpatient management of young febrile infants with urinary tract infections. Pediatr Emerg Care 2014;30:591-597.
- Poole NM, Kronman MP, Rutman L, et al. Improving antibiotic prescribing for children with urinary tract infection in emergency and urgent care settings. Pediatr Emerg Care 2018; Jan 2. doi: 10.1097/PEC.0000000000001342. [Epub ahead of print].
- Keren R, Chan E. A meta-analysis of randomized, controlled trials comparing short- and long-course antibiotic therapy for urinary tract infections in children. Pediatrics 2002;109:E70-70.
- Fitzgerald A, Mori R, Lakhanpaul M, Tullus K. Antibiotics for treating lower urinary tract infection in children. Cochrane Database Syst Rev 2012;(8):CD006857.
Urinary tract infections (UTIs) are a potential cause of fever in the pediatric patient. Early testing and a timely diagnosis are critical to avert complications and potential scarring of the kidneys. Complication rates in children younger than 90 days of age include bacteremia, meningitis, and urosepsis. The authors present a concise review of the current status of the diagnosis and management of pediatric UTIs.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
