Blunt Pelvic Trauma
May 1, 2019
Reprints
AUTHORS
Benjamin Moran, MD, Trauma Fellow, University of Maryland School of Medicine, Baltimore
Jay Menaker, MD, FACEP, Professor, Department of Surgery/Emergency Medicine, University of Maryland School of Medicine, Baltimore
PEER REVIEWER
Andrew D. Perron, MD, Professor of Emergency Medicine, Maine Medical Center, Portland
EXECUTIVE SUMMARY
- Mechanisms, or force vectors, applied to the pelvis are used to describe the pelvic fracture patterns. These include vertical shear, anterior-posterior compression, and lateral compression fractures.
- Several classification systems exist for describing pelvic fractures. The most common system used is the Young-Burgess classification, which categorizes fractures based on force vectors applied to the pelvic ring.
- Patients with pelvic injuries often complain of hip, lower back, or abdominal pain.
- A pelvic X-ray and Focused Assessment with Sonography in Trauma exam can determine body compartments of hemorrhage in the polytrauma patient quickly. These tools are especially useful in patients with depressed Glasgow Coma Scale scores in whom a physical exam is unreliable. These tests can be performed quickly in the trauma bay, allowing rapid assessment and decision-making. Conversely, in patients who are awake, are hemodynamically stable, and have minimal to no pelvic pain, the pelvic X-ray is of little utility.
- Secondary to the high incidence of concomitant intra-abdominal injuries in pelvic trauma patients, obtaining abdominal and pelvic CT scans at the time of initial evaluation is recommended in stable patients.
- In patients with high suspicion of pelvic fractures or hemodynamically unstable patients with confirmed pelvic fractures, external compression of the pelvis should be performed. External compression reduces the amount of pelvic volume, thus limiting the amount of potential space for bleeding and allowing stabilization of the bony pelvic elements to provide a tamponade effect. External compression may reduce the pelvic volume by 10%.
- Both the Eastern Association for the Surgery of Trauma and the Western Trauma Association recommend use of angioembolization (AE) in patients with pelvic fractures, but they offer a few caveats. First, pelvic bleeding is 85% venous in origin; thus, not all patients will benefit from AE. Patients who remain hemodynamically unstable, who have undergone pelvic stabilization, or who have had preperitoneal packing may benefit from AE. Regardless of hemodynamic instability, patients who have contrast extravasation on CT or who are older than 60 years of age with major pelvic fractures (open-book, butterfly segment, or vertical shear) should be considered for AE.
The management of pelvic trauma has evolved significantly in the last 20 years, with advances in devices and procedures. The key to success is having a team of physicians, including specialists in emergency medicine, interventional radiology, and surgery, who can work together to provide each patient the best outcome possible.
— Ann M. Dietrich, MD, Editor
Introduction
Pelvic trauma that results in fractures occurs secondary to a significant force that typically causes multiple associated injuries. These associated injuries, as well as injuries in an aging population, associated patient comorbidities, and medications, may make pelvic trauma challenging to treat.
The management of pelvic trauma has evolved significantly during the past 20 years. New medical devices, procedures, and transfusion therapy continue to change algorithms and spark debate. Patients with blunt pelvic trauma require urgent recognition, prompt diagnosis, and a multidisciplinary approach to improve outcomes.1 A team including emergency medicine physicians, an interventional radiologist, trauma providers, and orthopedic surgeons is required for coordinated evaluation and rapid treatment.2 This article will focus on recognizing and managing trauma patients with pelvic injuries.
Epidemiology
Injury to the bony pelvis occurs in nearly one in 10 patients with blunt trauma.2 The severity of the damage ranges from stable pelvic ring fractures to unstable fractures with accompanying life-threatening exsanguination.2 The overall mortality rate of patients presenting with pelvic fractures is 6%; however, the mortality rate dramatically increases to 40% in patients who present with hemodynamic instability or who require multiple blood transfusions.2,3 Mortality within 24 hours most often is due to uncontrolled hemorrhage, accounting for 39% of early deaths, followed by head injury, which accounts for 31%.2 Because of the large amount of force required to break the pelvis, pelvic fractures are associated with concomitant injuries nearly 90% of the time.3 Rates of pelvic injuries combined with other injuries include: torso injuries 30-50%, abdominal injuries 30-50%, extremity injuries 60%, and head injuries 40-50%.3
Patients with pelvic fractures also have a higher in-hospital all-cause mortality. Even with simple pelvic ring fractures, patients have an increased risk of death.4 In older patients (> 65 years), decreased systolic blood pressure (< 90 mmHg), increased respiratory rate (> 30 breaths per minute), depressed Glasgow Coma Scale (GCS) score (< 8-10), or increased injury severity index (> 18) all are predictors of increased mortality.4,5
Mechanisms, or force vectors, applied to the pelvis are used to describe the pelvic fracture patterns. These include vertical shear (VS), anterior-posterior compression (APC), and lateral compression (LC) fractures. VS injuries occur most commonly in patients who fall from a great height and in motorcycle collisions when the cyclist with outstretched legs has a frontal impact. APC fracture patterns occur frequently in head-on motor vehicle collisions (MVCs) and pedestrians struck by vehicles. Finally, LC fractures typically result from forces directed to the patient’s side, which include side impact MVCs and pedestrians struck on the side. These will be discussed further in the classification of pelvic fractures.
Anatomy
Pelvis is a Latin-derived word for bucket or basin. The pelvis functions as support for ambulation, holds the abdominal contents within the abdomen, and expands during childbirth. The pelvis is ringed and is comprised of the sacrum with two innominate bones on either side. The innominate bone includes the ilium, the ischium, and the pubis. Both innominate bones join anteriorly at the pubic symphysis and posteriorly at the sacroiliac (SI) joint. The SI joints and the pubic symphysis are fused together by the body’s strongest ligaments. The anterior and posterior SI ligaments include short and long elements that extend from the sacrum to the iliac crests. These provide vertical stability across the SI joints.
The pelvic floor is bridged by the sacrospinous and sacrotuberous ligaments connecting the sacrum to the ischial spine and ischial tuberosity. In total, the anterior elements consist of the pubic rami and the pubic symphysis, which provide roughly 40% of pelvis stability. The posterior elements (sacrum, SI joints, ilium) provide the remainder of stability and are more important. Because of the pelvis’s ringed shape, a break in two places is required to cause instability.2 Compared with a fracture, ligamentous disruption is far more important when determining pelvic stability. Lastly, although the acetabulum is not a structural support of the pelvis, it provides an insertion site for the femoral head.
The vascular supply to the pelvis is divided into inflow and outflow. The internal iliac arteries provide direct inflow perfusion to the pelvis. A large venous plexus surrounds the bony structure of the pelvis, collects venous blood, and drains into the large internal iliac venous system. These vessels lie just anterior to the SI joints, thus joint disruption makes these vessels vulnerable to injury. The most common and largest arterial vessel injured in trauma is the superior gluteal artery.2 Structures that lie in close proximity to the pelvic bones and ligaments are at risk of injury secondary to stretching that is associated with pelvic fractures. Nerves that may be injured include the extraperitoneal rectum, bladder, urethra, and sciatic, femoral, and obturator nerves.6
Classification
Several classification systems exist for describing pelvic fractures. The most common system used is the Young-Burgess classification. This system was first described in 1990 and developed to identify and classify pelvic fracture patterns according to force vectors applied to the pelvic ring.7 As noted before, these patterns are divided into LC, APC, and VS. (See Table 1.)
LC fractures are the most common type of pelvic fracture, occurring in roughly 60% of all pelvic fractures.8 MVCs in which a car is T-boned or situations in which a pedestrian is struck on the side by a vehicle cause a lateral force during impact. As a lateral impact occurs to the innominate bones, the pelvis on the side of impact rotates toward the midline. As the hemipelvis rotates, the anterior pelvic ligaments and the local vasculature shorten as the impact occurs. With the shortening, the vessels are not subject to shearing or tensile factors, thus they are more likely to remain intact. Severe hemorrhage is not typical of LC fractures, but in the older population (> 55 years of age), there is a higher likelihood of LC factures producing clinically significant hemorrhage.10 LC1 fractures usually involve a lower energy force, but are not benign; they result in an overall mortality rate of 8.2%.8
Table 1. Young-Burgess Classification7-9 |
|||
|
Type |
Common Characteristics |
Differentiation Characteristics |
Stability |
|
Lateral compression 1 |
Transverse pubic rami fracture |
Sacral compression on side of impact |
Stable |
|
Lateral compression 2 |
Transverse pubic rami fracture |
Crescent (iliac wing) fracture |
Stable or unstable |
|
Lateral compression 3 |
Transverse pubic rami fracture |
Contralateral open-book (anterior-posterior compression) injury |
Unstable |
|
Anterior-posterior compression 1 |
Symphyseal diastasis or vertical pubic rami fracture |
Widening of symphysis (< 2.5 cm) and/or sacroiliac joint, stretched but intact sacroiliac ligaments |
Stable |
|
Anterior-posterior compression 2 |
Symphyseal diastasis or vertical pubic rami fracture |
Complete hemipelvis separation but not vertical displacement, anterior sacroiliac ligaments ruptured more than posterior ones |
Stable or unstable |
|
Anterior-posterior compression 3 |
Symphyseal diastasis or vertical pubic rami fracture |
Complete hemipelvis separation but no vertical displacement, anterior and posterior sacroiliac joint ligaments ruptured |
Unstable |
|
Vertical shear |
Symphyseal diastasis or vertical pubic rami fracture |
Vertical hemipelvis displacement, usually through sacroiliac joint, occasionally through iliac wing or sacrum |
Unstable |
|
Combined mechanism |
Vertical or transverse pubic rami fractures |
Combination of patterns: Lateral compression + vertical shear or lateral compression + anterior-posterior compression |
Unstable |
In LC2 fractures, as the energy mechanism increases, the horizontal lateral impact continues to rotate and implode the hemipelvis inward, adding to the transverse pubic rami fracture seen in LC1. Additionally, the iliac wing is pushed inward, creating a crescent-shaped posterior iliac wing fracture. These fractures can be stable or unstable, depending on the amount of ligamentous disruption. In LC3 fractures, the force is increased, and the pelvis continues to implode and rotate inward, disrupting the pubic rami and SI joints and ligaments and causing a contralateral “open-book” APC injury. These are severely unstable fractures.
APC injuries, or open-book fractures, happen when force vectors occur in front-to-back impact. The front-to-back forces occur in front impact MVCs or in frontal pedestrian collisions. APC1 fractures are stable with only a slight widening (up to 2.5 cm) of the pubic symphysis. In APC2 injuries, the pubic symphysis widens greater than 2.5 cm. With widening, the SI ligaments are disrupted. These patients have rotational instability, and hemorrhage is more likely in this patient population.2 As the mechanism energy increases in APC3 fractures, the SI joint widens, and both anterior and posterior elements of the SI ligaments are disrupted. The result is full instability of the hemipelvis, with a high likelihood of bleeding, nerve damage, and abdominal injuries.2,9
VS injuries result from a vector force applied cephalad, which can occur from a fall from a height > 15 feet or in a motorcycle collision in which the rider has frontal impact with his or her foot outstretched. These fractures involve the anterior (pubic rami, pubic symphysis) and posterior (sacroiliac complex) bones, resulting in a hemipelvis that is pushed superiorly. On physical exam, the patient will have shortening of the ipsilateral leg.
Finally, in combined mechanism injuries, fractures cannot be classified by any specific category. Patients with these injuries have vertical or transverse pubic rami fractures with a combination of other elements. Typically, injuries in this pattern are unstable.
It is important to understand fracture classifications and force vector injuries because this may help predict the need for further intervention. APC and VS injuries are associated with a higher incidence of pelvic vascular injury and hemorrhage.11 Anything that disrupts the SI joint can result in tearing of the venous plexus and lead to hemorrhage.
In an American Association for the Surgery of Trauma prospective, observational, multicenter study, researchers attempted to characterize modern management and outcomes for patients with pelvic fractures.3 The researchers reviewed patients at 11 Level I trauma centers over two years, and included them if they presented with hypotension (systolic blood pressure [SBP] < 90 mmHg), tachycardia (> 120), and a base deficit (> 5). Investigators reviewed more than 46,000 admissions and found only 163 patients who met the criteria. The main outcome was identification of fracture pattern and the need for angioembolization. The most common fracture pattern associated with shock in these patients was LC1, followed by LC2 and VS. Fractures that required the most pelvic hemorrhage control included APC3 (83%) and VS. Although LC1 and LC2 fractures were most common in patients with shock, they required the least amount of hemorrhage control. Investigators found that patients who presented with a VS fracture pattern had a higher injury severity score, required blood transfusions, and had an associated a need for hemorrhage control.
Initial Assessment
The initial evaluation of the traumatically injured patient continues to be protocolized and standardized by the Advanced Trauma Life Support guidelines. This systematic approach rapidly identifies any life-threatening bleeding and ensures that all patients receive a thorough assessment. Patients can bleed into several body compartments (including the thoracic, abdominal, and pelvic/retroperitoneal cavities) and the thigh, as well as externally. First, the clinician should perform the primary survey in which the patient’s airway, breathing, circulation, disability, and exposure are assessed. Early establishment of intravenous access is essential, and initiation of resuscitation should begin immediately in unstable patients. The clinician also should perform a Focused Assessment with Sonography in Trauma (FAST) examination as an adjunct to the primary survey.
Patients with pelvic injuries often complain of hip, lower back, or abdominal pain. When evaluating the bony pelvis, place hands on the anterior superior iliac spine and direct gentle pressure inward. Additionally, palpate the pubic symphysis for widening. Only one person should perform this maneuver. It is essential to avoid rocking the pelvis during this maneuver because it may cause severe pain, widening, and clot disruption. Examining for pelvic ring instability has its limitations, as the presence of a pelvic deformity or an unstable pelvic ring on physical examination has poor sensitivity for the detection of mechanically unstable pelvic fractures.12
The Western Trauma Association has a well-defined algorithm for the management of patients with pelvic fractures.13 Initial trauma evaluation is completed and diagnosis of a pelvic fracture is made with an anteroposterior (AP) radiograph film. It is imperative to establish the diagnosis and begin early resuscitation. If the patient is not at a Level I trauma center, it is important to arrange transport early. In a review of 1,220 patients admitted to Level I and Level II trauma centers with matched pelvic fractures, patients treated at the Level I centers had decreased mortality rates (7.7% vs. 11.6%).14
With a pelvic fracture confirmed, the next decision point is determining hemodynamic instability, which is defined by a blood pressure ≤ 90 mmHg, a transfusion requirement of 4-6 units of packed red blood cells, or a base deficit ≤ -6. If the patient is unstable and has a negative FAST exam, then a pelvic binder should be applied for pelvic stabilization and an urgent orthopedic consultation should be obtained. Additionally, appropriate consultation should be obtained for emergent angiography with embolization if the patient does not become hemodynamically stable after pelvic stabilization. If these resources are not available, urgent transfer should be arranged.
If the patient is hemodynamically stable, then further diagnostic tests, including an abdominal/pelvic computed tomography (CT) scan, should be performed. The scan will identify the bony injuries and any pelvic blush. If there is a blush present, the patient should undergo angiography and embolization. It is important to know the available resources and limitations at each facility to provide appropriate care and transfer if needed.
Laboratory Workup
For patients with suspected pelvic trauma, the most important laboratory test is a type and cross. A blood chemistry and complete blood count also are useful, but often may misrepresent the state of shock. Other useful laboratory tests include coagulation profiles, prothrombin time, international normalized ratio, partial thromboplastin time, and thromboelastography, if available. These tests allow for early identification of coagulation abnormalities and facilitate appropriate reversal treatment. Serum lactate and arterial blood gas analysis will identify the presence and depth of acidosis, and serial levels can be drawn to determine the response of resuscitation. A urinalysis is useful to help identify any bladder injury or urethral injury. Five percent of patients with a bladder or urethral injury will present with only microscopic hematuria.6,11
Plain Films
A chest X-ray (CXR), pelvic X-ray (PXR), and FAST exam can determine body compartments of hemorrhage in the polytrauma patient quickly. These tools are especially useful in patients with depressed GCS scores in whom a physical exam is unreliable. Additionally, these tests can be performed quickly in the trauma bay, allowing rapid assessment and decision-making. Conversely, in patients who are awake, are hemodynamically stable, and have minimal to no pelvic pain, the PXR is of little utility. In a study about the utility of PXR for diagnosing fractures, patients who initially were screened with X-ray received the diagnosis of pelvic fracture in two minutes. In patients who waited for a CT scan, the fracture was diagnosed in 47 minutes.15 This delay in diagnosis was associated with increased time to angioembolization and further management.15 Any hemodynamically unstable polytrauma patient should receive an immediate CXR and PXR to help expedite a diagnosis and treatment.
When plain films are evaluated, acetabular and posterior pelvic fractures often are underestimated or overlooked.16 Nearly half of patients with pelvic fractures are underdiagnosed by pelvic plain films and roughly one-fifth of patients have a missed fracture.16 If the plain film is unremarkable or equivocal and the patient continues to have pelvic pain, further diagnostic imaging with CT scan should be obtained.
Each clinician should have a systematic way to review PXR films. First, focus on the soft tissues around the bony pelvis and evaluate for any soft tissue swelling or foreign objects. Next, look at the bony structure and evaluate the three circles of the pelvis. The bony cortex should be contiguous. Examine the large iliac bones, sacrum, and SI joints. If there is loss of continuance at the joint lines, a fracture likely is present even if no cortex abnormality is seen. Look at the pubic symphysis and evaluate for any diastasis. Finally, look at the femoral shaft, surgical neck, and head as it connects with the acetabulum.
Ultrasound
The FAST examination has become a critical tool in the trauma bay. It has replaced direct peritoneal lavage for identifying free intraperitoneal fluid. As stated earlier, this tool is used in conjunction with CXR and PXR to help determine the etiology of hypotension in unstable patients.
In the past, earlier studies stated that the FAST examination was unreliable in patients with blunt traumatic injuries, finding a 29% incidence of intra-abdominal injury without hemoperitoneum.17 However, with widespread use of ultrasound and more advanced training and experience, new reports have shown false-negative and false-positive rates for FAST in pelvic trauma patients to be 2% and 7%, respectively.18 As with any diagnostic test, ultrasound is just one component of the whole clinical picture. A negative FAST exam may turn positive with serial exams.
Computed Tomography Scanning
CT scanning has become the gold standard of imaging in the blunt poly-trauma patient because it allows for superior visualization of solid organ injuries and better characterization of the severity and displacement of pelvic fractures. A significant advantage of contrasted CT scanning is that it allows providers to visualize active contrast extravasation, otherwise known as a blush. The presence of intravascular contrast extravasation is considered a marker of active pelvic bleeding, which justifies immediate pelvic angiography and angioembolization.19,20 In addition to contrast extravasation, CT scanning can be used to assess for bladder injuries.
Because of the high incidence of concomitant intra-abdominal injuries in pelvic trauma patients, obtaining abdominal and pelvic CT scans at the time of initial evaluation is recommended. CT scanning comes with limitations. Most importantly, patients must be stable enough for transport to and from the CT scanner. Lastly, contrast administration involves risks, most notably allergic complications, kidney injury, and local tissue trauma if there is contrast extravasation.
Genitourinary Injuries
The urethra lies in close proximity to the pelvic bones, and urethral injuries occur in approximately 1% of patients with blunt pelvic injury.21 Findings suggestive of urethral injury include blood at the urethral meatus, perineal or scrotal hematoma, suprapubic tenderness, and a high-riding prostate on rectal exam. Bony fragments may cause lacerations in the perineum, vagina, or rectum, which increase suspicion of a urethral injury. Urethral injuries are twice as common in men as in women.21 Fracture patterns associated with urethral injury include bilateral superior and inferior pubic rami fractures (straddle fractures) or forces causing abrupt thigh abduction.6 Even if there are no physical exam findings concerning for a urethral injury, urinary catheters should be inserted very gently and should be stopped if any resistance occurs in patients with pelvic fractures. A retrograde urethrogram should be performed in patients unable to pass a Foley catheter and in patients with exam findings concerning for urethral injuries.
Bladder injuries also are common. In the setting of hematuria, the bladder should be evaluated with a CT cystogram with distended bladder and post-emptying views to establish the diagnosis. (See Figure 1.) Extraperitoneal bladder rupture can be managed with catheter drainage, while intraperitoneal rupture requires surgical repair.6 It is important to note that if angioembolization will be attempted, a cystogram should wait because extravasated contrast in a bladder injury will obscure the view of an arterial blush on imaging.
Figure 1. Cystogram |
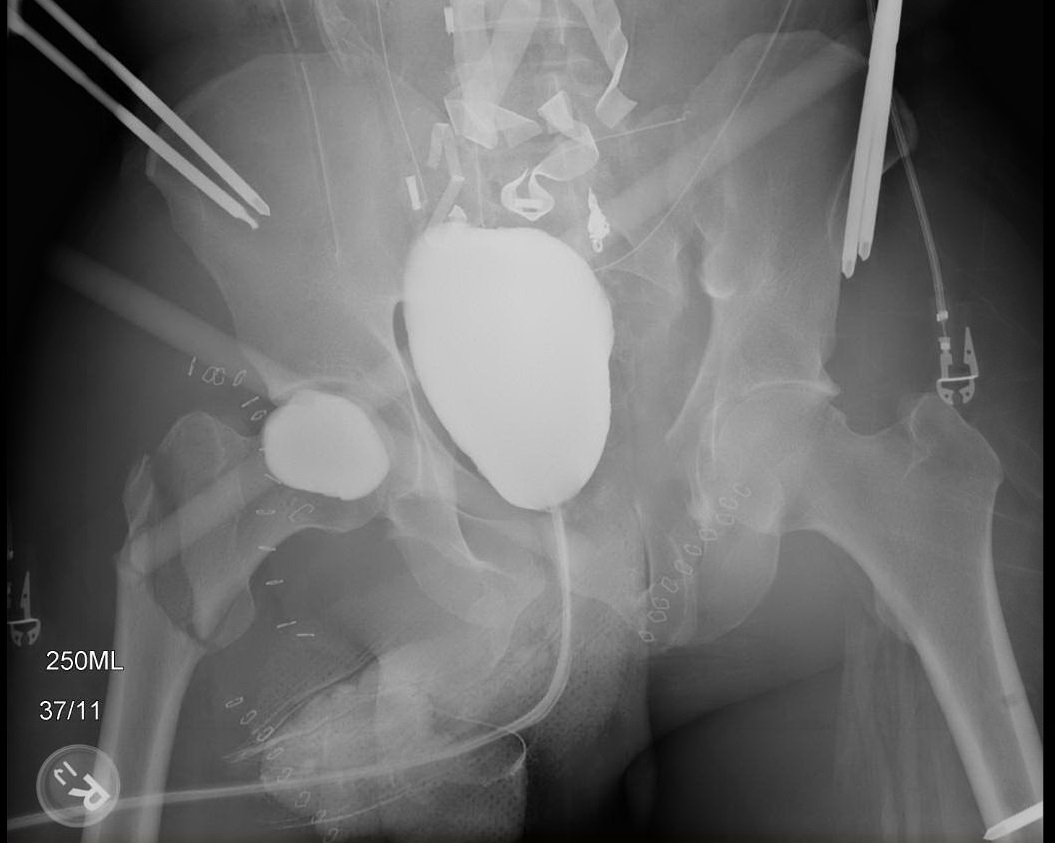 |
|
No active bladder extravasation. Note the patient’s multiple bony injuries, previous external pelvis fixation, and abdominal packing. |
Pelvic Stabilization
In patients with high suspicion of pelvic fractures or hemodynamically unstable patients with confirmed pelvic fractures, external compression of the pelvis should be performed. External compression reduces the amount of pelvic volume, thus limiting the potential space for bleeding and allowing stabilization of the bony pelvic elements to provide a tamponade effect. External compression may reduce the pelvic volume by 10%.22 (See Figure 2.)
Figure 2. APC3 Fracture |
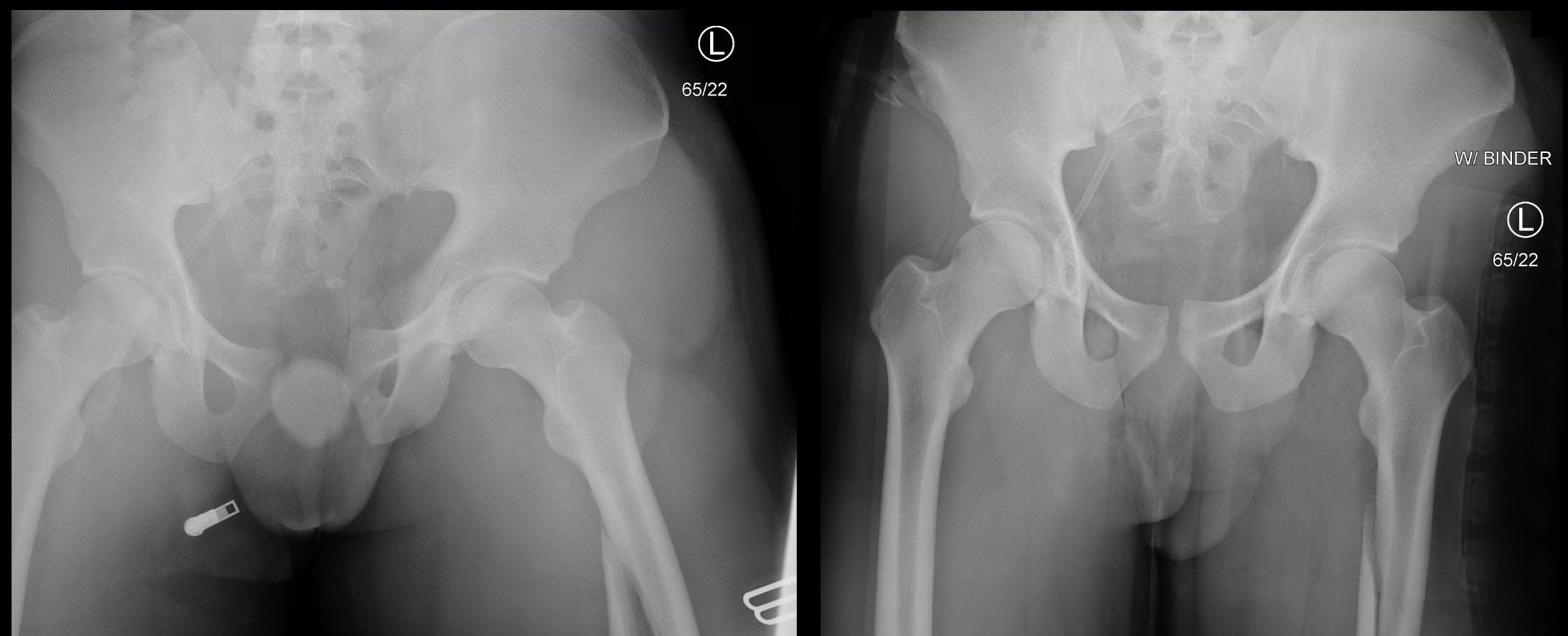 |
|
APC3 fracture before (left) and after (right) application of an external pelvic binder. Note pubic diastasis and disruption of the left sacroiliac joint. Pelvic binder application with good reduction of pelvis. |
Noninvasive external compression can be achieved with a commercial device or with a bed sheet. A bed sheet should be kept wide (20 cm), crisscrossed around the patient, and secured with large Kelley clamps or chest tube clamps. Typically, two people are needed to tighten and secure the wrap properly. Commercially available devices generally consist of a pelvic wrap with a set of Velcro pulleys to reduce the pelvic volume. To ensure proper placement, center the device over the greater trochanters of the femur. If the device is placed too high, it can act as a fulcrum to open the pelvis further. In addition to closing down pelvic volume, the patient’s toes/feet can be taped together. AP pelvic films should be obtained after placement. It is imperative to monitor for over-reduction of fractures. Pelvic binding devices should not be left in place for more than 24 hours because they can precipitate skin necrosis over injured areas and bony prominences.23
Placement of an external skeletal fixator is another approach to stabilize unstable pelvic fractures. Much like a binder, an external fixator can decrease the diameter and the volume of the pelvis and stabilize the bony elements to provide a tamponade effect to decrease the total amount of bleeding.24 Early placement of external fixation has been shown to reduce mortality.24 Patients with unstable pelvic fractures undergoing laparotomy for hemorrhage control may benefit from the application of external fixation, either with a C-clamp or another external fixator for hemorrhage control.13,25,26 Additionally, in some centers, pelvic clamps can be applied in the trauma bay, allowing for rigid pelvic fixation prior to laparotomy. These resources are limited and are available only in certain centers. (See Figure 3.)
Figure 3. Vertical Shear Fracture |
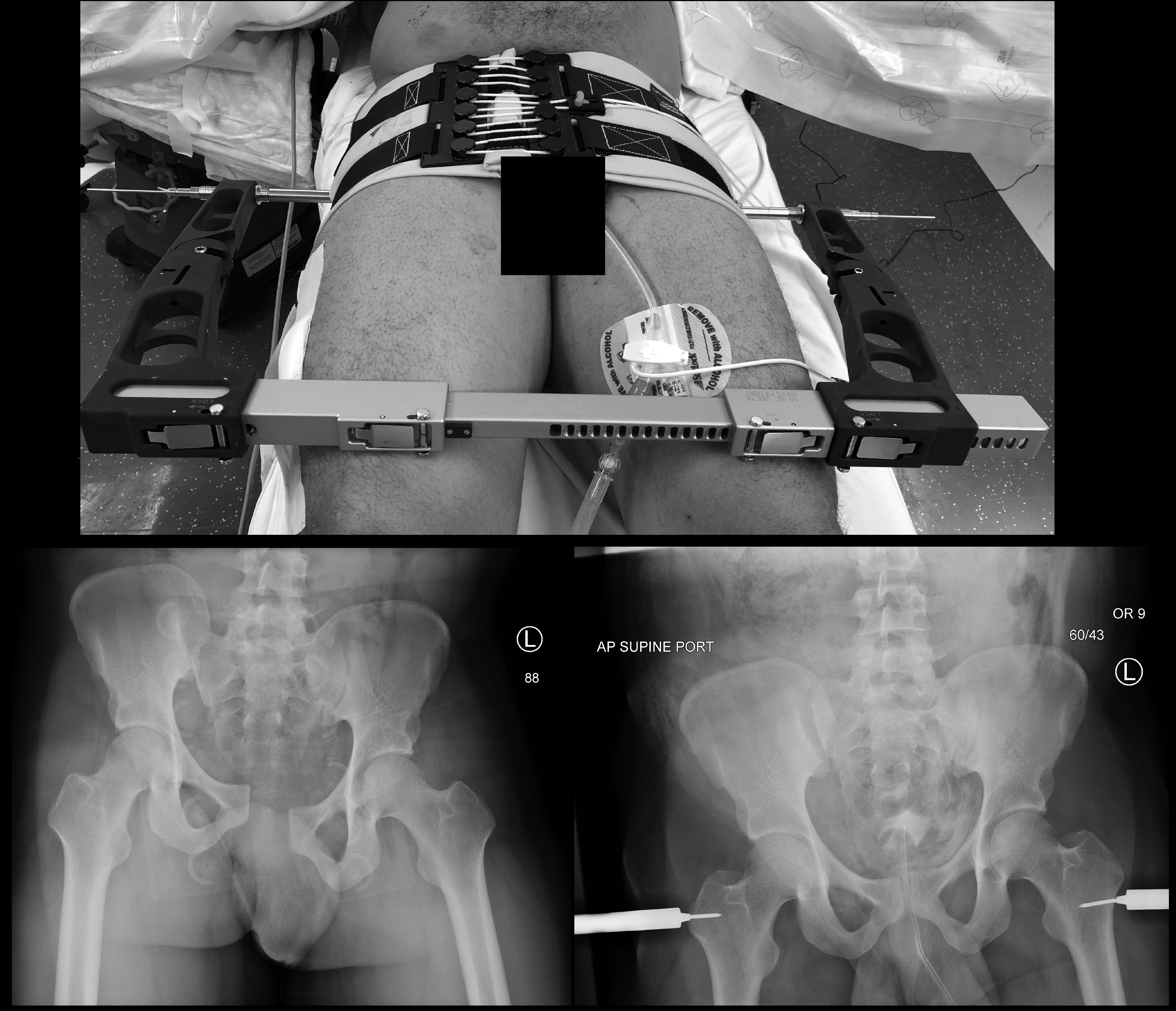 |
|
Vertical shear fracture prior to stabilization (bottom left). Initially stabilized with a pelvic binder, then placement of an emergent C-clamp prior to laparotomy for pelvic stabilization (top). Post-reduction films (bottom right) |
External fixation may provide definitive fixation in certain patients. However, many patients will need further internal fixation, which is accomplished best when the patient has achieved normal physiology and can withstand a prolonged surgery. The optimal timing of definitive fixation after damage-control orthopedic surgery has been debated, but usually it is between post-trauma days 5 and 10. In one study, definitive fixation between days 2 and 4 was associated with more frequent development of multiple organ failure compared to fixation between days 6 and 8.27 If possible, definitive fixation should be accomplished within two weeks because external fixator pin site infections increase after this time.28
Angiography and Embolization
Angiography with embolization is a useful tool for the diagnosis and therapeutic management of hemorrhage associated with pelvic fractures. Following arterial access, typically obtained in the common femoral artery with retrograde placement of a wire, injection of contrast is used to identify sources of bleeding. The bleeding is stopped either by a selective (identifying exact site of extravasation) or nonselective means. Gel foam or prothrombotic coils are injected into the source vessel of bleeding to stop arterial inflow. These procedures require an interventional radiologist and angiography suite or a combined hybrid operating room in some centers. The intervention may be performed by a vascular surgeon in conjunction with an ongoing laparotomy. These approaches are selected based on the institutional resources.
Both the Eastern Association for the Surgery of Trauma and the Western Trauma Association recommend the use of angioembolization (AE) in patients with pelvic fractures, but they offer a few caveats. First, pelvic bleeding is 85% venous in origin; thus, not all patients will benefit from AE. AE may be beneficial for patients who remain hemodynamically unstable, who have undergone pelvic stabilization, or who have had preperitoneal packing.13 Regardless of hemodynamic instability, patients who have contrast extravasation on CT or who are older than 60 years of age with major pelvic fractures (open-book, butterfly segment, or VS) should be considered for AE.19 Lastly, if a patient who has had AE continues to be unstable with other sources of bleeding excluded, repeat AE should be considered.19
The authors of a multi-institutional cohort study examined the use of nonselective vs. selective AE.29 First, the study investigators examined the use of nonselective AE in patients with negative angiograms. The theory behind nonselective AE is to decrease the arterial pressure head on the venous system, thereby decreasing bleeding. In this study, embolization did not change the need for transfusion; however, patients who were embolized had a decreased amount of blood products within the first 24 hours (7.5 vs. 4 units of packed red blood cells). Patients in this group also experienced more frequent inpatient complications (66.6% vs. 34.1%). With a positive angiogram, there was no consensus about whether nonselective or selective angiography proved to have the best risk-benefit ratio. There were no differences in transfusions between the two groups, and total procedure times did not differ. However, complications were increased in the nonselective group (51.8% vs. 27.1%). If needed, all attempts should be made at selective embolization to help minimize the amount of complications.
AE comes with inherent risks. There is a risk of improper placement of catheters, resulting in non-target embolization. This is rare, since pelvic embolization has high technical success rates reported from 85-100%. After embolization, clinical success has been reported from 90-95%. Rebleeding after embolization occurs in 6-8% of cases, and clinical success rates reach 97% with repeat embolization.30,31
Ischemic complications can arise after embolization. Bilateral internal iliac embolization in association with significant tissue trauma can lead to gluteal necrosis, wound complications, claudication, neuropathy, and poor fracture healing.29 Additionally, 2% of patients with pelvic trauma experience impotence. Although these complications are noted in the literature in patients who have undergone pelvic AE, it is unknown if these complications are the result of embolization or the initial injury. Despite these risks, AE is beneficial in treating these patients.19 (See Figure 4.)
Figure 4. Angiography of Pelvis |
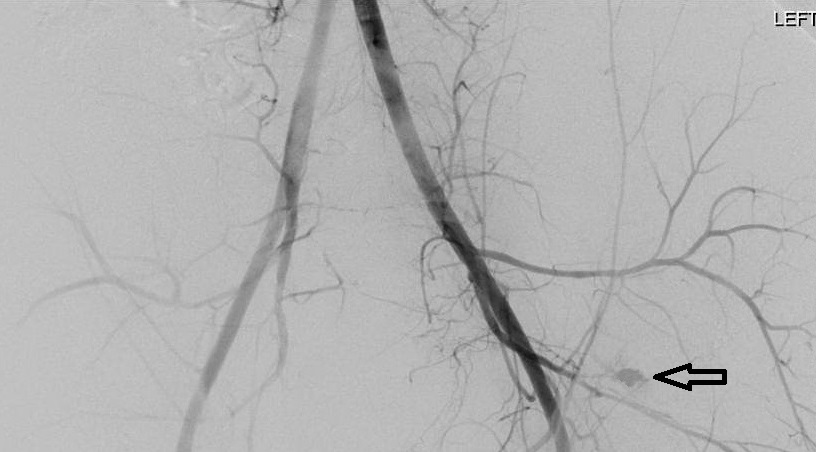 |
|
The arrow shows active arterial extravasation. |
Endovascular Technology: REBOA
Another tool in the physician’s armamentarium for hemorrhage control is endovascular therapy. Endovascular technology is continually advancing into smaller, more easily deployable devices that aid in rapid hemorrhage control in the trauma setting. The resuscitative endovascular balloon occlusion of the aorta (REBOA) catheter is the device most studied. Since its inception, this device has shrunk from a 14 French device that required a femoral cutdown, guidewire, and X-ray confirmation to a 7 French device that is centimeter hash-marked, with internal stiffness and p-tip for a wireless insertion. The REBOA catheter is introduced through a 7 French sheath and inserted based on external measurement, sternal notch for zone 1 and umbilicus for zone 3, then confirmed by X-ray. When used for hemorrhage control in the setting of pelvic fracture, the balloon is inflated in zone 3, which is the level of the infrarenal aorta before the aortic bifurcation. This serves as a temporizing measure to allow for further resuscitation, induction of anesthesia, and operative preparation or transfer to the angiography suite.
Multiple animal studies have demonstrated the benefit of REBOA in controlling pelvic hemorrhage.32 Later case studies continued to prove its feasibility and effectiveness as a method of internal aortic control in patients in end-stage shock with truncal and pelvic trauma.33,34 These procedures are effective for gaining critical time needed to stabilize the patient and obtain more definitive treatment.
One last important consideration when deciding to deploy REBOA is the initial act of obtaining common femoral arterial (CFA) access. For patients with pelvic fractures that have “borderline” hemodynamic stability or instability, a useful adjunct is early insertion of a femoral arterial line. When looking at total time to aortic occlusion with REBOA deployment in patients with and without femoral arterial access, total mean time to occlusion was 158 seconds if CFA access had been obtained vs. a total procedure time (CFA access and REBOA deployment) of 366 seconds in patients without CFA access.35 Thus, if a patient is unstable and may need REBOA deployment, obtain early femoral arterial access. (See Figure 5.)
Figure 5. Resuscitative Endovascular Balloon Occlusion of the Aorta (REBOA) Placement, Zone III |
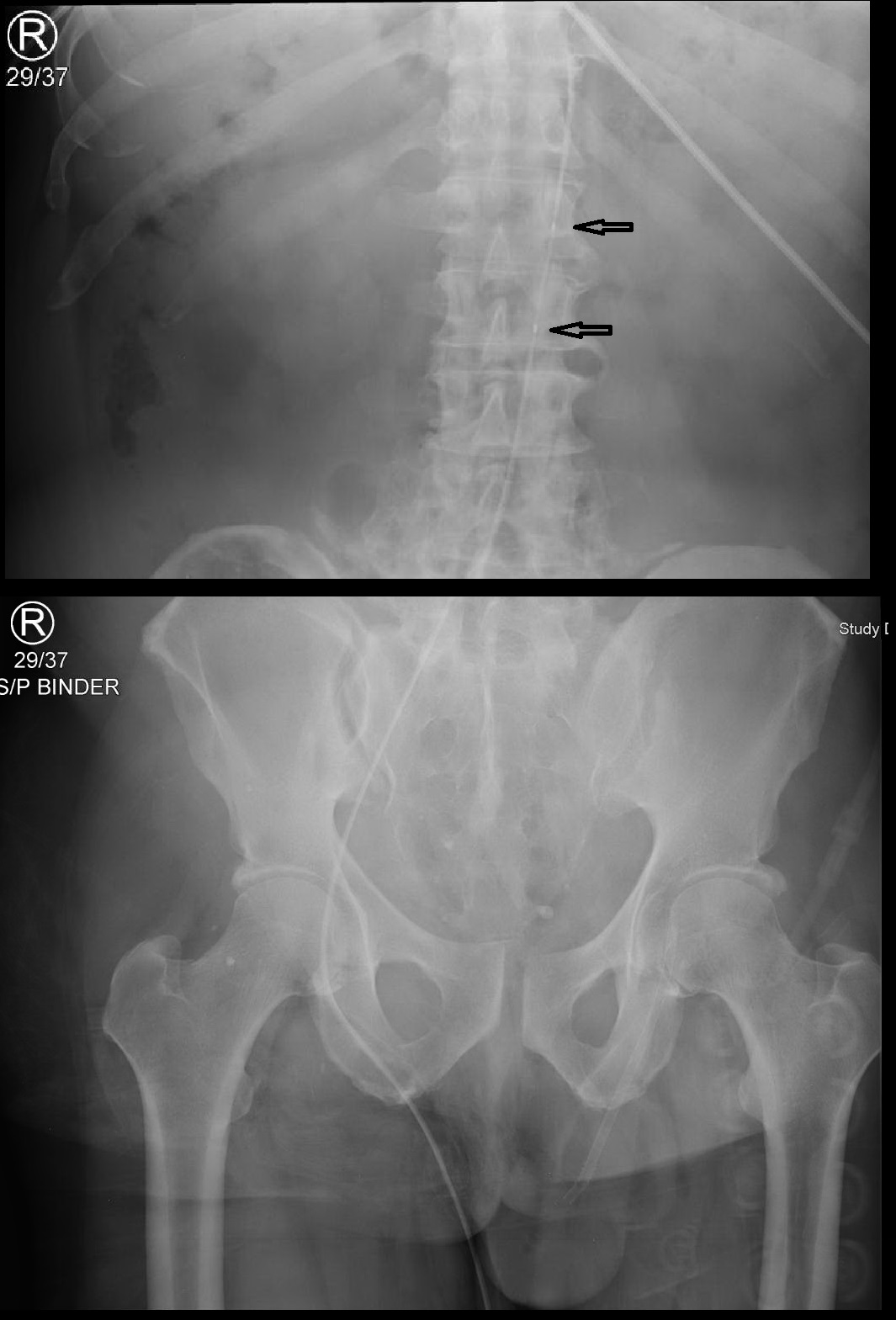 |
|
Arrows denote radiopaque markers of the superior and inferior aspects of REBOA balloon. |
Operative Techniques: Preperitoneal Packing, Internal Iliac Ligation
In patients who are hemodynamically unstable or when transport times are prolonged and/or angioembolization is unavailable, preperitoneal packing is a viable option. The concept behind preperitoneal packing is that 85% of pelvic bleeding is venous, coming from the rich plexus that lies anterior to the pelvic bony structure in the preperitoneal and retroperitoneal planes. Entering this disrupted field and directly placing laparotomy pads in this potential space provide tamponade to prevent bleeding.
To pack the pelvis, a horizontal Pfannenstiel incision is made above the pubic symphysis.36 Dissection is carried down to the level of the anterior fascia. This is opened, and care must be taken to avoid entering the peritoneum. In this preperitoneal plane, the pelvic hematoma will be visualized and should be evacuated. The bladder should be retracted laterally with a malleable retractor as the pelvic brim is palpated and manually dissected. Avoid disrupting any vascular connections between the iliac and obturator vessels. Three rolled laparotomy packs are placed sequentially deep to the pelvic brim. The first is placed posterior, just below the sacroiliac joint; the second sponge is placed anteriorly in the middle of the pelvic brim; and the third sponge is placed in the retropubic space, deep and lateral to the bladder. When one side is completed, the other side then is packed. The fascia should be closed to help tamponade the bleeding. Bright red hemorrhage observed while packing the pelvis is an emergent indication for internal iliac AE. Once packing is complete, the patient should be transported to the intensive care unit for ongoing resuscitation. Packs should be removed or exchanged in 24-48 hours.
Bilateral internal iliac ligation is a second operative approach that is used less commonly. This technique consists of intraperitoneal dissection of the internal iliac artery with direct ligation using clips, Rummel tourniquets, or suture ligation. This technically challenging operation is performed only when patients are in extremis. In a series from Los Angeles, only 28 patients required internal iliac ligation out of 201 severe pelvic fractures.37 The patients were critically ill, unresponsive to fluid and external pelvic binding, and required operative intervention. These patients had an overall mortality of 64%, mainly from brain injury and cardiac arrest, but underwent internal iliac ligation as described earlier. Of the patients who survived (18/28), no complications of gluteal necrosis or perineal skin changes were seen. Study investigators concluded that this is a viable option in patients who are in extremis.
Conclusion
Blunt pelvic trauma represents a common entity in the injured patient. Injuries range from minor to life-threatening hemorrhage. It is important for the clinician to have a high index of suspicion for pelvic fractures and any accompanying or associated injury in the polytrauma patient. Patients should be approached systematically with the aim of identifying potential sources of bleeding. A thorough physical exam followed by pertinent imaging is essential. Early transfer arrangements should be made in institutions with limited resources.
In patients with unstable pelvic fracture as a source of hemorrhage, all efforts should be made to stabilize the fracture. This can be accomplished using a pelvic binder or early external stabilization. A CT scan of the pelvis is the best imaging modality to identify fracture patterns and the presence of a pelvic blush. With active contrast extravasation, arrangements should be made for immediate AE.
In patients with unstable pelvic fractures who require emergent laparotomy, consideration of external stabilization and preperitoneal packing at the time of laparotomy should take place. In the setting of unstable patients without the need for urgent laparotomy, the decision for using AE vs. external stabilization often is institutionally dependent. Lastly, early arterial access and placement of REBOA should be considered as a measure to control hemorrhage until the patient can receive definitive therapy.
Pelvic fracture management will continue to evolve with new treatment modalities. Early recognition and identification of pelvic fractures with prompt stabilization and treatment or expeditious transfer will help improve patient outcomes.
REFERENCES
- Tran TL, Brasel KJ, Karmy-Jones R, et al. Western Trauma Association Critical Decisions in Trauma: Management of pelvic fracture with hemodynamic instability—2016 updates. J Trauma Acute Care Surg 2016;81:1171-1174.
- Moore EE, Feliciano DV , Mattox KL. Trauma. 8th ed. New York: McGraw-Hill Education/Medical; 2017.
- Costantini TW, Coimbra R, Holcomb JB, et al; AAST Pelvic Fracture Study Group. Pelvic fracture pattern predicts the need for hemorrhage control intervention — Results of an AAST multi-institutional study. J Trauma Acute Care Surg 2017;82:1030-1038.
- Schulman JE, O’Toole RV, Castillo RC, et al. Pelvic ring fractures are an independent risk factor for death after blunt trauma. J Trauma 2010;68:930-934.
- Jung K, Matsumoto S, Smith A, et al. Analyses of clinical outcomes after severe pelvic fractures: An international study. Trauma Surg Acute Care Open 2018;3:e000238.
- Flint L, Cryer HG. Pelvic fracture: The last 50 years. J Trauma 2010;69:483-488.
- Burgess AR, Eastridge BJ, Young JW, et al. Pelvic ring disruptions: Effective classification system and treatment protocols. J Trauma 1990;30:848-856.
- Manson T, O’Toole RV, Whitney A, et al. Young-Burgess classification of pelvic ring fractures: Does it predict mortality, transfusion requirements, and non-orthopaedic injuries? J Orthop Trauma 2010;24:603-609.
- Manson TT, Nascone JW, Sciadini MF, O’Toole RV. Does fracture pattern predict death with lateral compression type 1 pelvic fractures? J Trauma 2010;69:876-879.
- Henry SM, Pollak AN, Jones AL, et al. Pelvic fracture in geriatric patients: A distinct clinical entity. J Trauma 2002;53:15.
- Mattox KL, Moore EE, Feliciano DV. Trauma. 7th ed. New York: McGraw-Hill Education/Medical; 2012: 1248.
- Shlamovitz GZ, Mower WR, Bergman J, et al. How (un)useful is the pelvic ring stability examination in diagnosing mechanically unstable pelvic fractures in blunt trauma patients? J Trauma 2009;66:815-820.
- Western Trauma Association. Management of pelvic fracture with hemodynamic instability algorithm. Available at: http://www.westerntrauma.org/algorithms/PelvicFractureNotes/Summary.html. Accessed Dec. 31, 2018.
- Oliphant BW, Tignanelli CJ, Napolitano LM, et al. American College of Surgeons Committee on Trauma verification level affects trauma center management of pelvic ring injuries and patient mortality. J Trauma Acute Care Surg 2019;86:1-10.
- Verbeek DO, Burgess AR. Importance of pelvic radiography for initial trauma assessment: An orthopedic perspective. J Emerg Med 2016;50:852-858.
- Barleben A, Jafari F, Rose J Jr, et al. Implementation of a cost-saving algorithm for pelvic radiographs in blunt trauma patients. J Trauma 2011;71:582-584.
- Chiu WC, Cushing BM, Rodriguez A, et al. Abdominal injuries without hemoperitoneum: A potential limitation of focused abdominal sonography for trauma (FAST). J Trauma 1997;42:617-623; discussion 623-625.
- Christian NT, Burlew CC, Moore EE, et al. The focused abdominal sonography for trauma examination can reliably identify patients with significant intra-abdominal hemorrhage in life-threatening pelvic fractures. J Trauma Acute Care Surg 2018;84:924-928.
- Cullinane DC, Schiller HJ, Zielinski MD, et al. Eastern Association for the Surgery of Trauma practice management guidelines for hemorrhage in pelvice fracture — update and systematic review. J Trauma 2011;71:1850-1868.
- Ramin S, Hermida M, Millet I, et al. Limits of intravascular contrast extravasation on computed tomography scan to define the need for pelvic angioembolization in pelvic blunt trauma: A specific assessment on the risk of false positives. J Trauma Acute Care Surg 2018;85:527-535.
- Bjurlin MA, Fantus RJ, Mellett MM, Goble SM. Genitourinary injuries in pelvic fracture morbidity and mortality using the National Trauma Data Bank. J Trauma 2009;67:1033-1039.
- Krieg JC, Mohr M, Ellis TJ, et al. Emergent stabilization of pelvic ring injuries by controlled circumferential compression: A clinical trial. J Trauma 2005;59:659-664.
- Jowett AJL, Bowyer GW. Pressure characteristics of pelvic binders. Injury 2007;38:118-121.
- Ben-Menachem Y, Coldwell DM, Young JW, Burgess AR. Hemorrhage associated with pelvic fractures: Causes, diagnosis, and emergent management. AJR Am J Roentgenol 1991;157:1005-1014.
- Archdeacon MT, Hiratzka J. The trochanteric C-clamp for provisional pelvic stability. J Orthop Trauma 2006;20:47-51.
- Ertel W, Keel M, Eid K, et al. Control of severe hemorrhage using C-clamp and pelvic packing in multiply injured patients with pelvic ring disruption. J Orthop Trauma 2001;15:468-474.
- Pape H, Stalp M, V Griensven M, et al. [Optimal timing for secondary surgery in polytrauma patients: An evaluation of 4,314 serious-injury cases]. [Article in German] Chirurg 1999;70:1287-1293.
- Harwood PJ, Giannoudis PV, Probst C, et al. The risk of local infective complications after damage control procedures for femoral shaft fracture. J Orthop Trauma 2006;20:181.
- Hymel A, Asturias S, Zhao F, et al. Selective versus nonselective embolization versus no embolization in pelvic trauma: A multicenter retrospective cohort study. J Trauma Acute Care Surg 2017;83:361-367.
- Martin JG, Kassin M, Park P, et al. Evaluation and treatment of blunt pelvic trauma. Tech Vasc Interv Radiol 2017;20:237-242.
- Velmahos GC, Toutouzas KG, Vassiliu P, et al. A prospective study on the safety and efficacy of angiographic embolization for pelvic and visceral injuries. J Trauma 2002;53:303-308; discussion 308.
- Morrison JJ, Percival TJ, Markov NP, et al. Aortic balloon occlusion is effective in controlling pelvic hemorrhage. J Surg Res 2012;177:341-347.
- Brenner ML, Moore LJ, DuBose JJ, et al. A clinical series of resuscitative endovascular balloon occlusion of the aorta for hemorrhage control and resuscitation. J Trauma Acute Care Surg 2013;75:506-511.
- Moore LJ, Martin CD, Harvin JA, et al. Resuscitative endovascular balloon occlusion of the aorta for control of noncompressible truncal hemorrhage in the abdomen and pelvis. Am J Surg 2016;212:1222-1230.
- Romagnoli AN, Teeter W, Wasicek P, et al. No wire? No problem: Resuscitative endovascular balloon occlusion of the aorta can be performed effectively and more rapidly with a wire-free device. J Trauma Acute Care Surg 2018;85:894.
- Cothren CC, Osborn PM, Moore EE, et al. Preperitonal pelvic packing for hemodynamically unstable pelvic fractures: A paradigm shift. J Trauma 2007;62:834-839; discussion 839-842.
- DuBose J, Inaba K, Barmparas G, et al. Bilateral internal iliac artery ligation as a damage control approach in massive retroperitoneal bleeding after pelvic fracture. J Trauma 2010;69:1507-1514.
The management of pelvic trauma has evolved significantly in the last 20 years, with advances in devices and procedures. The key to success is having a team of physicians, including specialists in emergency medicine, interventional radiology, and surgery, who can work together to provide each patient the best outcome possible.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
