A Review of Atrial Fibrillation and Current Therapeutic Strategies: Part 1
November 1, 2019
Reprints
Authors
Hunter Mwansa, MD,
St. Vincent Charity Medical Center, Case Western Reserve University, Cleveland, OH
Sula Mazimba, MD, MPH, Division of Cardiovascular Medicine, University of Virginia Health System, Charlottesville, VA
Peer Reviewer
Glen D. Solomon, MD, FACP, Professor and Chair, Department of Internal Medicine, Wright State University Boonshoft School of Medicine, Dayton, OH
EXECUTIVE SUMMARY
Atrial fibrillation (AF) is the most common arrhythmia, affecting nearly 34 million people globally in 2010. In the United States, it is estimated to reach 12.1 million by 2030. Associated with increased morbidity and mortality, AF is characterized by uncoordinated atrial electrical activity leading to compromised ventricular filling and hemodynamic instability.
• AF is commonly categorized based on the temporal nature of the disorder: paroxysmal, acute, or chronic; its response to cardioversion: persistent vs. permanent; and the presence or absence of structural heart disease: valvular vs. nonvalvular.
• Pathophysiologic mechanisms include genetic predisposition, and anatomical and electrophysiologic abnormalities, which may be primary or secondary to conditions such as valvular heart disease, heart failure, hypertension, diabetes mellitus, obstructive sleep apnea, alcohol, and tobacco.
• Thrombogenesis is the most feared consequence of AF leading to cerebral vascular accidents.
• Acute management typically involves beta-blockers, calcium channel blockers and/or digoxin to achieve rate control. Long-term mortality outcomes have been related mostly to appropriate and effective anticoagulation.
This two-part series presents a review of the current evidence on atrial fibrillation. The first part includes its definition, classification, risk factors, comorbidities, evaluation, and acute management of newly diagnosed patients. The second part will focus on long-term management, including risk factor modification, rate and rhythm control measures, stroke risk stratification, and anticoagulation management.
Introduction
Atrial fibrillation (AF) is one of the most common atrial arrhythmias, and it is associated with increased morbidity and mortality. It is characterized by uncoordinated atrial1 electrical activity that causes impaired atrial contraction and compromised ventricular filling and hemodynamic stability, especially in patients with pre-existing structural heart disease. These hemodynamic consequences of AF depend on the interplay of multiple factors, including atrioventricular dyssynchrony, suboptimal heart rate (either tachycardia or bradycardia), abnormal atrioventricular nodal conduction, presence or the absence of accessory conduction pathways, and sympathetic activation.2,3,4
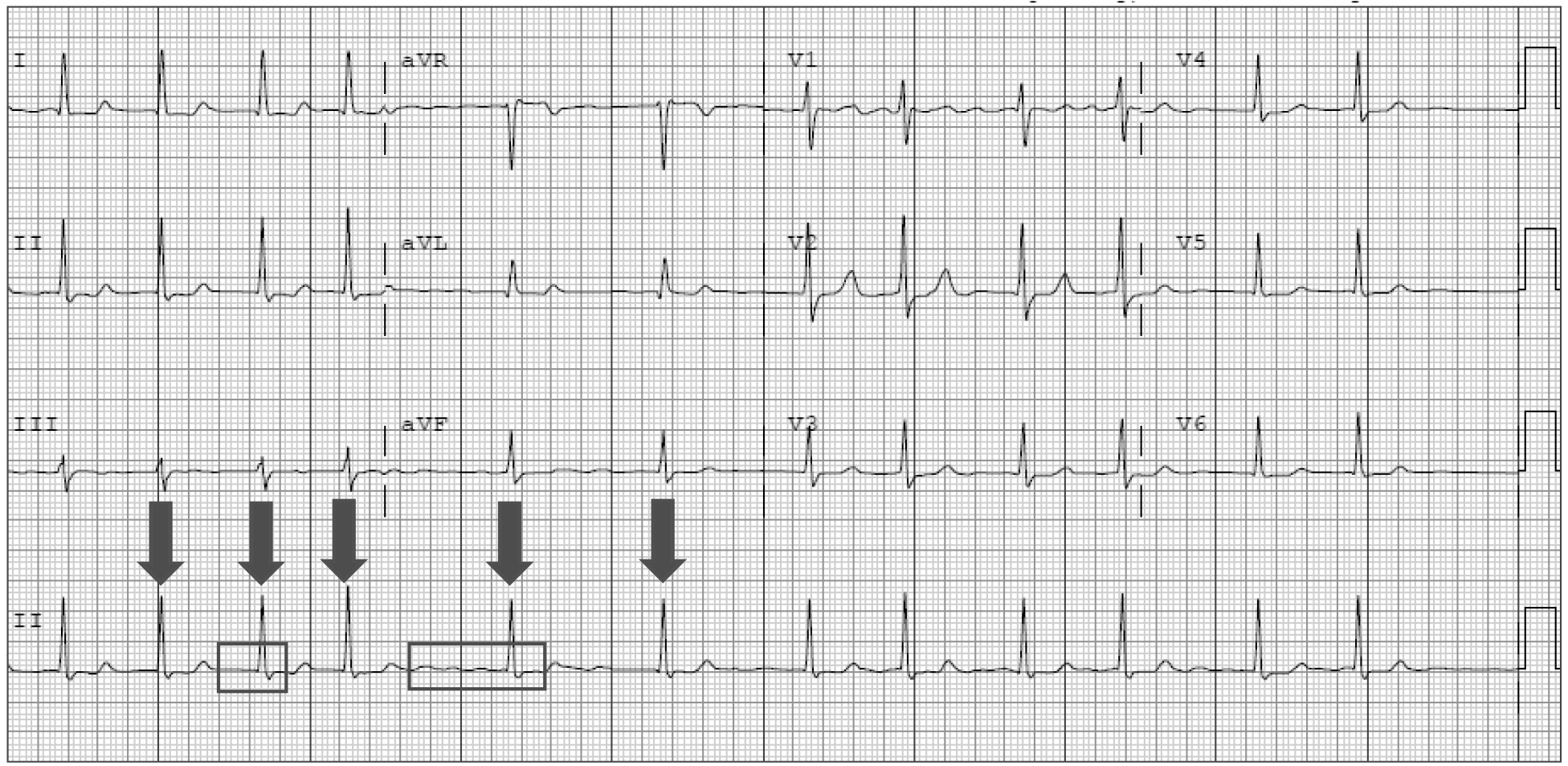
Electrocardiographically, AF is characterized by irregular R-R intervals and absence of distinct regular P waves (replaced by chaotic oscillatory or fibrillatory P waves) as shown in Figure 1.
Figure 1. Electrocardiogram of a Patient With Atrial Fibrillation |
 |
|
Bold arrows represent R waves; rectangular inserts arranged along the isoelectric line; R-R intervals = distance between consecutive bold arrows; note the variable R-R intervals and lack of meaningful P waves before every R wave. |
Classification of AF
AF is commonly categorized based on the temporal nature of the disease (paroxysmal, acute, or chronic), response to cardioversion (persistent vs. permanent), or whether there is the presence or absence of specific mitral valvular disease (valvular vs. nonvalvular).5,6 Other classification methods exist, but the above mentioned have therapeutic implications and affect outcomes. For example, catheter ablation therapy has better outcomes in patients with paroxysmal AF than in those with persistent AF, although this does not necessarily translate into improved mortality.7
Interestingly, both paroxysmal and persistent AF may occur in a single patient, and it is not exactly clear how long patients stay in sinus rhythm following successful cardioversion. Further, patients with non-paroxysmal AF (other patterns of AF) have demonstrated higher rates of thromboembolism compared to those with paroxysmal AF (hazard ratio [HR] = 1.38; 95% confidence interval [CI], 1.19-1.61;
P < 0.001) and death (HR = 1.22; 95% CI, 1.09-1.37; P < 0.001).8 Table 1 gives a descriptive summary of AF when characterized by its presentation, duration, and termination, either spontaneously or following cardioversion. AF can be categorized further as valvular and nonvalvular. Valvular AF occurs in the setting of moderate-to-severe mitral stenosis and/or the presence of a mechanical valve, as recently redefined by the 2019 American College of Cardiology (ACC)/American Heart Association (AHA) focused update of the 2014 guidelines on the management of AF. Nonvalvular AF does not signify implicit absence of valvular disease and includes patients with valvular abnormalities below the threshold required for inclusion under valvular AF.6 This classification of AF dictates whether a patient receives a vitamin K antagonist (VKA) or non-vitamin K oral anticoagulant (NOAC) to reduce the risk of stroke and systemic embolism. Except for dabigatran, which proved harmful, none of the NOACs have been evaluated for safety and efficacy in patients with valvular AF. The RE-ALIGN (Randomized, Phase II Study to Evaluate the Safety and Pharmacokinetics of Oral Dabigatran Etexilate in Patients After Heart Valve Replacement) trial was a prospective multicenter phase II dose-validation study of dabigatran vs. warfarin therapy in patients aged 18-75 years with either a mechanical mitral and/or aortic valve replacement within seven days of surgery (cohort A) or a mechanical mitral and/or mechanical aortic valve replacement more than three months before randomization (cohort B). The study closed prematurely because of prohibitively high thromboembolic and bleeding events in the dabigatran group.9,10
Table 1. Classification of Atrial Fibrillation |
|
|
Descriptive Term |
Comments |
|
Newly diagnosed AF |
First recognition of AF irrespective of duration and symptoms |
|
Paroxysmal AF* |
AF that spontaneously reverts to sinus rhythm, usually within 48 hours, although it may extend to seven days; includes AF episodes successfully cardioverted within seven days |
|
Persistent AF |
AF lasting more than seven days |
|
Longstanding persistent AF |
AF lasting one year or more when a rhythm control approach is chosen |
|
Permanent AF |
AF refractory to cardioversion, and which is accepted by both patient and physician and entails abandonment of rhythm control strategy with a focus on rate control. A change of therapeutic strategy because of a change in patient choices entails a switch back to longstanding persistent AF. |
|
*Paroxysmal and persistent AF usually are distinguished using long-term cardiac event monitors; both can coexist in an individual, and the predominant one should be adopted as the primary pattern.5 |
|
Only VKAs, specifically warfarin, have proven safety and efficacy in patients with valvular AF. Nevertheless, NOACs are effective at reducing stroke and systemic embolism among patients with valvular abnormalities other than moderate-to-severe mitral stenosis and mechanical valves, including those with valvuloplasties and bioprosthetic valves. They also compared well with warfarin in several clinical trials, except for differences in bleeding rates.11,12,13
Epidemiology of AF
AF is the most common arrhythmia, with estimates of global prevalence in 20.9 million men and 12.6 million women for the year 2010.14,15 In 2010, estimates of the prevalence of AF in the United States were 2.7 million to 6.1 million16,17 and is projected to reach 12.1 million in 2030.15 The rise in the prevalence of AF is attributable to a combination of factors, including an increasingly older U.S. population,18 improved disease detection made possible by widespread use of cardiac diagnostic and monitoring devices (Holter monitors, loop recorders, pacemakers, etc.), and increases in the prevalence of predisposing conditions for AF, such as cardiovascular disease (e.g. myocardial infarction [MI], valvular heart disease [VHD], heart failure [HF], hypertension [HTN]),19 obesity, obstructive sleep apnea (OSA), obesity hypoventilation syndrome (OHS), chronic obstructive pulmonary disease (COPD), diabetes mellitus (DM), and chronic kidney disease (CKD). AF prevalence increases with age and is highest among elderly patients older than 80 years of age, among whom it has previously been estimated at 8%.20
Notwithstanding the higher prevalence of predisposing factors for AF among black U.S. residents, the risk of incident AF was highest among whites when compared to blacks (HR = 0.84; 95% CI, 0.82-0.85; P < 0.001), Hispanics (HR = 0.78; 95% CI, 0.77-0.79; P < 0.001), and Asians (HR = 0.78; 95% CI, 0.77-0.79; P < 0.001) in a cohort of hospitalized patients followed over a median period of 3.2 years.21 This race-risk factor AF paradox has been investigated before, and current evidence suggests that Caucasian ancestry and genetic factors might play a bigger role in the pathogenesis of AF given the predominantly higher AF risk among these cohorts, even in the absence of cardiovascular comorbidities.21 Furthermore, blacks with implanted cardiac devices did not demonstrate a higher burden of AF compared to Caucasians.22 The lifetime risk of AF in adults of European descent also has increased, from about one in four adults to one in three adults.23 The increased risk of AF among Caucasians also was observed in the recent ARIC (Atherosclerosis Risk in Communities) study that evaluated ethnic differences in the lifetime risk of AF.24
AF is associated with an increased burden of morbidity, including HF, MI, stroke, gastrointestinal (GI) bleeding, and death. Rates of these complications increase with age (e.g. rates of death, HF, MI, stroke, and GI bleeding for populations aged 67-69 years and 85-89 years were 28.8%, 11.0%, 3.3%, 5.0%, and 4.4%, and 67.0%, 15.8%, 4.4%, 8.9%, and 6.6%, respectively) until age ≥ 90 years.25
AF incurs a huge financial burden and was estimated to cost $26 billion in 2005 alone.25 Its association with reversible lifestyle risk factors calls for attention to address these potential precursors and effective treatment of the disease to prevent and/or reduce complications associated with the disease.
Pathophysiology and Mechanisms of AF
The pathophysiology and mechanisms responsible for initiation, propagation, and perpetuation of AF are not completely understood. Currently proposed mechanisms suggest an interplay of factors that include genetic susceptibility to developing AF and anatomic and electrophysiologic abnormalities that may be primary (rarely) and/or secondary effects of prolonged exposure to the known risk factors (e.g. VHD, chronic HF, HTN, DM, OSA, alcohol, tobacco, etc.).
Genetic Factors
There is evidence to suggest a strong heritable component to AF. Relatively early onset AF, apparently independent of cardiovascular disease, has been observed in young patients with a strong family history of AF.26,27 Early onset AF in young patients with heritable channelopathies and cardiomyopathies with underlying monogenic mutations also has been described. For example, a mutation in the gene encoding the pore-forming α-subunit (KCNQ1) of the cardiac IKs channel on chromosome 11 that causes a shortened refractory period and increased function of the channel has been implicated in persistent AF observed in these patients.28
Familial AF is most likely polygenic, mediated by an interplay of multiple gene mutations, and is currently a subject of ongoing research.29,30,31 It is likely a phenotypic manifestation of the atrial action potential changes and atrial arrhythmogenic remodeling may occur in patients with these gene polymorphisms.32,33 Patients with these gene variants also have been reported to have a heightened risk for cardioembolic or ischemic strokes, possibly from silent AF.34
Arrhythmogenic Atrial Remodeling and Alteration of Cardiac Ion Channel Function
Several risk factors for AF, as well as AF itself, can cause progressive cardiac damage with accompanying fibrosis. Physiologic stress arising from pressure overload acting alone or in concert with an activated renin-angiotensin aldosterone system (RAAS) in the setting of VHD, chronic HF, and hypertension may lead to structural changes that culminate in atrial myocardial damage and ensuing repair via activation of fibroblasts, connective tissue deposition, and ultimately cardiomyocyte and interstitial fibrosis.35-38 This remodeling may result in the creation of atrial ectopy and re-entry circuits, alteration of conduction pathways, and electrical dissociation related to the juxtaposition of normal myocytes with fibrotic tissue that act together to propagate and perpetuate AF.39,40 Evidence also exists of atrial fatty infiltration and cardiomyocyte inflammation, necrosis, and amyloid deposition in AF patients with concomitant risk factors, such as obesity and advanced age.41,42
Electrophysiologic Basis of AF
Etiologically, two principle scientific constructs have been advanced to explain AF. First, AF is thought to involve an interplay of ectopic foci that cause rapid firing, which may be sustained by local re-entry. Commonly, these ectopic foci are located in muscular sleeves extending from the left atrium into the distal pulmonary veins, but they also are found less frequently in the distal superior vena cava as it enters the right atrium, ligament of Marshall, and other parts of both atria.43,44,45 These foci of ectopic atrial electrical activity have been implicated in the initiation of AF, and this has been demonstrated in individuals with paroxysmal AF without evidence of underlying structural heart disease44 and patients who revert to AF after successful cardioversion.46 The exact mechanisms underpinning the initiation of ectopic and rapid electrical activity have not been fully elucidated. There is some evidence to suggest that the alteration of autonomic tone characterized by either sympathetic or parasympathetic (vagal) hyperactivity, as observed during intense physical exercise and during sleep, respectively, may precede paroxysms of AF.47 Whereas sympathetic overdrive leads to increased intracellular calcium, which possibly creates triggers and increased automaticity, parasympathetic overdrive causes increased susceptibility to re-entry because of activation of acetylcholine-mediated potassium ion channels that cause heterogeneous shortening of the atrial action potential and refractory period.48 Sympathetic activation may play a role in AF that occurs in postoperative settings, although the major drivers behind this form of AF are possibly inflammation and oxidative stress.49,50
Postoperative AF commonly complicates cardiac surgery, with incidence estimates of about 15-30% after coronary bypass surgery and 30-60% after cardiac valve replacement.51,52 Catheter-directed radiofrequency ablation can be an effective intervention in subjects with these ectopic foci, particularly for those that are localized to the distal pulmonary veins.45
Second, the hypothesis of multiple wavelets and rotors suggests that AF is perpetuated by the occurrence of multiple wavelets that propagate within larger abnormal atrial tissue with refractory and conduction heterogeneity that render them highly arrhythmogenic and self-sustaining.53 Propagation of these wavelets is sustained so long as their number remains above a certain threshold.39
It is often said that “AF begets AF.” This is because AF leads to other electrophysiological changes within the atrial substrate. For example, AF induces shortening of the atrial action potential refractory period and AF cycle length, which, if persistent, is associated with atrial remodeling that can further perpetuate AF.54,55,56
The duration of antecedent persistent AF is inversely associated with the time a patient stays in sinus rhythm post cardioversion. In light of these pathophysiological insights, avoiding irreversible atrial remodeling through early therapy initiation may be an important therapeutic consideration. However, there is no evidence that a universal rhythm control approach over a rate control strategy has mortality benefit.57
Thrombogenesis
One of the most dreaded consequences of AF is cerebral vascular accident (CVA) due to thromboembolism. Atrial remodeling, including dilatation, and functional abnormalities associated with AF promote a suitable milieu for the formation of thrombi with potential for systemic propagation. Stasis of blood (due to atrial hypocontractility), atrial endothelial damage, and an inflammatory milieu all converge to produce a procoagulant state that may account for thromboembolism seen in AF.58,59
Atrial hypocontractility is probably the most important factor in AF-related thrombogenesis because there is a heightened risk of thromboembolism post cardioversion due to delayed return of atrial contractility in the setting of atrial myocardial stunning.
Risk Factors and Associated Comorbidities
Table 2 shows some risk factors and clinical conditions associated with AF. It is important to note that some of these risks and clinical comorbidities are potentially reversible. Interventions aimed at addressing them may, therefore, prevent incident AF and potentially cure concomitant AF. For example, catheter ablation may effectively treat AF associated with Wolff-Parkinson-White (WPW) syndrome, atrial nodal (AV) reentrant tachycardia, and atrial ectopic tachycardia.60
Table 2. Cardiac and Noncardiac Risk Factors for Atrial Fibrillation
- Cardiovascular comorbidities61
- Heart failure
- Myocardial infarction
- Valvular heart disease
- Hypertension
- Heritable channelopathies and cardiomyopathies
- Left ventricular hypertrophy and atrial dilation
- Infiltrative cardiac and inflammatory conditions, e.g. amyloidosis, sarcoidosis, pericarditis, and myocarditis
- Family history and genetic variants5
- Advanced age and male gender62
- Metabolic and endocrine abnormalities61,63
- Diabetes mellitus
- Hypothyroidism
- Hyperthyroidism
- Obesity61,64
- Obstructive sleep apnea and obesity hypoventilation syndrome64
- Chronic obstructive pulmonary disease65
- Chronic kidney disease66
- Alcoholism67
- Tobacco use68
- Habitual exercise69
- Surgery52,70
- Cardiothoracic surgery
- Coronary artery bypass
- Valve replacement
- Lung surgeries, e.g. pneumonectomy and lobectomy
Clinical Evaluation of AF
The diagnosis of AF requires evidence of AF on ECG (see Figure 1), noninvasive ambulatory rhythm monitors (e.g. Holter monitors, event recorders), pacemakers, defibrillators, and electrophysiologic studies. Patients may present either asymptomatically or with non-specific symptoms, including palpitations, lightheadedness, fatigue, dyspnea, and chest pain. It is not uncommon for patients, especially those with pre-existing cardiac disease, to present with worsening symptoms of heart failure, angina, and hypotension if the ventricular response is rapid enough to cause hemodynamic instability.
The initial evaluation of patients should focus on obtaining a thorough history of symptoms, risk factors (see Table 2), family history, and a detailed physical examination looking for an irregularly irregular pulse, an irregular jugular venous pulse, and a variation in loudness of the first heart sound (S1). A physical exam may further show a pulse deficit when the heart rate is compared to the peripheral venous pulse as well as murmurs in the presence of underlying cardiac structural disease.
Extracardiac manifestations may reflect either complications of the disease, such as focal neurological deficits in those with AF complicated by cerebrovascular disease, or manifestations of comorbid non-cardiac diseases.
The diagnosis of AF may pose a challenge in some patients and this requires a high index of suspicion, particularly in those with risk factors for the disease and/or those presenting with possible complications of AF, commonly cardioembolic stroke. Long-term cardiac rhythm monitoring devices, e.g. loop recorders, may be warranted in patients suspected of having paroxysmal AF, such as patients with intermittent palpitations and survivors of cryptogenic stroke (stroke of unknown origin) in whom the diagnosis of AF via external cardiac event monitors is inconclusive.71 These devices are effective in diagnosing silent AF in patients with cryptogenic stroke and may help decrease the chances of recurrent stroke.
Once a diagnosis of AF is confirmed, a further workup aimed at identifying possible causes, including laboratory tests and transthoracic echocardiography to identify associated structural heart disease, atrial enlargement, and left ventricular function, must be obtained. Important investigations required in patients diagnosed with AF are shown in Table 3.
Table 3. Investigations in Patients Diagnosed With Atrial Fibrillation |
|
|
Laboratory Workup |
Comments |
|
CBC |
|
|
CMP |
|
|
Thyroid function (TSH) |
|
|
BNP and NT-proBNP |
|
|
Echocardiography |
|
|
Exercise stress testing |
|
|
Chest X-ray |
|
|
Electrophysiologic studies |
|
|
BNP = B-type natriuretic peptide; CBC = complete blood count; CMP = complete metabolic panel; ECG = electrocardiogram; HF = heart failure; LV = left ventricle; MCV = macrocytic anemia; NT-proBNP = N-terminal pro-B-type natriuretic peptide; OHS = obesity hypoventilation syndrome; TSH = thyroid stimulating hormone |
|
Approach to Management of Newly Diagnosed AF
Acute Management
It often is challenging to distinguish between new-onset AF and recurrent AF, as it may go unrecognized as asymptomatic disease. Acutely, assessing for hemodynamic instability is a critical first step in dealing with newly diagnosed AF. Clinical evidence of hemodynamic instability, including hypotension, acute coronary syndrome, pulmonary edema, and/or congestive HF, calls for urgent electrical cardioversion. Hemodynamically stable patients presenting with rapid ventricular response (heart rate [HR] > 100 beats/minute) usually require a rate control strategy that employs measures such as the use of intravenous beta-blockers (e.g. esmolol, metoprolol), calcium channel blockers (e.g. diltiazem), and/or digoxin before a patient can be transitioned to oral heart rate control measures for symptomatic treatment and preservation of left ventricular (LV) function. It is important to recognize possible precipitants for the AF and/or tachycardia, such as thyrotoxicosis, anemia, and intercurrent infections, as addressing these might be key to spontaneous restoration of sinus rhythm and/or HR control. In appropriate candidates for the rate control strategy, LV systolic function and concurrent medical comorbidities (e.g. heart failure and chronic pulmonary diseases like COPD and asthma) dictate the therapeutic choice of a beta-blocker, a calcium channel blocker, and/or digoxin. Beta-blockers should be used in patients with chronic HF with reduced ejection fraction (HFrEF) (EF < 40%), as calcium channel blockers have significant inotropic effects that may worsen LV dysfunction and lead to deterioration of HF.73,74,75
Calcium channel blockers are preferred agents in those with chronic reactive airway diseases such as asthma. Acutely, digoxin usually is not a preferred initial choice because it has a slow onset of action and appears less efficacious in achieving rate control than beta-blockers and calcium channel blockers in this setting.76,77,78 Nevertheless, it is an important agent in patients with hypotension and can be a useful adjunct in patients requiring combination therapy for effective rate control. Acutely stabilized patients typically are transitioned to either oral beta-blockers, calcium channel blockers, digoxin, or combination therapy for long-term rate control. These rate controlling medications are listed in Table 4.
Table 4. Commonly Used Rate Control Drugs in Atrial Fibrillation |
||
|
Drug |
Dosage Range and Route of Administration |
Comments |
|
Beta-Blockers |
||
|
Bisoprolol |
1.25-20 mg qd or divided bid orally |
|
|
Carvedilol |
3.125-50 mg bid orally |
|
|
Esmolol |
0.5 mg/kg IV bolus over 1 minute followed by 0.05-0.25 mg/kg/min |
|
|
Metoprolol tartrate |
2.5-10 mg IV repeated as necessary |
|
|
12.5-100 mg bid orally |
||
|
Nebivolol |
2.5-10 mg qd or divided bid orally |
|
|
Non-Dihydropyridine Calcium Channel Blockers |
||
|
Diltiazem |
15-25 mg bolus, repeated as necessary |
|
|
60 mg tid to qid up to 360 mg total daily dose; modified release available for once daily dosing |
||
|
Verapamil |
2.5-10 mg bolus, repeated as necessary |
|
|
80-120 mg tid to qid |
||
|
Cardiac Glycosides |
||
|
Digoxin |
0.5 mg IV bolus followed by 0.75-1.5 mg in divided doses over 24 hours |
|
|
0.0625-0.25 mg qd |
||
|
qd = once daily; bid = twice a day; qid = four times a day; IV = intravenous |
||
Patients who present acutely with pre-excitation should not be managed with AV nodal blocking agents because AV nodal blockade can precipitate rapid conduction through the accessory pathway, leading to degeneration of AF into ventricular fibrillation, sudden cardiac arrest, and death. If they are hemodynamically stable, these patients can be managed with intravenous procainamide or propafenone acutely to control ventricular rates.79,80 These patients, particularly those with an antegrade accessory pathway, may benefit from catheter-directed AF ablation because of its effectiveness in this setting.5,81 Urgent catheter ablation of the accessory pathway is warranted in those who survive a sudden cardiac death. Patients with AF in the setting of pre-existing Ebstein’s anomaly, symptomatic pre-excitation, a short R-R interval (< 250 ms), and the presence of multiple accessory pathways on electrophysiologic studies have an increased risk for death and require catheter-directed ablation of the accessory pathway.
Whatever the choice of therapy, it is important to realize that long-term mortality outcomes in AF mostly have been tied to appropriate and effective anticoagulation. This important aspect of AF management must be addressed alongside the instituted rate and rhythm control measures.
References
- [No authors listed]. Definition of terms related to cardiac rhythm. Am Heart J 1978;95:796-806.
- Ikeda T, Murai H, Kaneko S, et al. Augmented single-unit muscle sympathetic nerve activity in heart failure with chronic atrial fibrillation. J Physiol 2012;590:509-518.
- Segerson NM, Sharma N, Smith ML, et al. The effects of rate and irregularity on sympathetic nerve activity in human subjects. Heart Rhythm 2007;4:20-26.
- Hsu LF, Jais P, Sanders P, et al. Catheter ablation for atrial fibrillation in congestive heart failure. N Engl J Med 2004;351:2373-2383.
- Kirchhof P, Benussi S, Kotecha D, et al. 2016 ESC Guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Eur Heart J 2016;37:2893-2962.
- January CT, Wann LS, Calkins H, et al. 2019 AHA/ACC/HRS Focused Update of the 2014 AHA/ACC/HRS Guidelines for the Management of Patients with Atrial Fibrillation. J Am Coll Cardiol 2019;74:104-132.
- Calkins H, Kuck KH, Cappato R, et al. 2012 HRS/EHRA/ECAS expert consensus statement on catheter and surgical ablation of atrial fibrillation: Recommendations for patient selection, procedural techniques, patient management and follow-up, definitions, endpoints, and research trial design: A report of the Heart Rhythm Society (HRS) Task Force on Catheter and Surgical Ablation of Atrial Fibrillation. Developed in partnership with the European Heart Rhythm Association (EHRA), a registered branch of the European Society of Cardiology (ESC) and the European Cardiac Arrhythmia Society (ECAS); and in collaboration with the American College of Cardiology (ACC), American Heart Association (AHA), the Asia Pacific Heart Rhythm Society (APHRS), and the Society of Thoracic Surgeons (STS). Heart Rhythm 2012;9:632-696.
- Ganesan AN, Chew DP, Hartshorne T, et al. The impact of atrial fibrillation type on the risk of thromboembolism, mortality, and bleeding: A systematic review and meta-analysis. Eur Heart J 2016;37:1591-1602.
- Van de Werf F, Brueckmann M, Connolly SJ, et al. A comparison of dabigatran etexilate with warfarin in patients with mechanical heart valves: The randomized, phase II study to evaluate the safety and pharmacokinetics of oral dabigatran etexilate in patients after heart valve replacement (RE-ALIGN). Am Heart J 2012;163:931-937.
- Eikelboom JW, Connolly SJ, Brueckmann M, et al. Dabigatran versus warfarin in patients with mechanical heart valves. N Engl J Med 2013; 369:1206-1214.
- Pan KL, Singer DE, Ovbiagele B, et al. Effects of non-vitamin K antagonist oral anticoagulants versus warfarin in patients with atrial fibrillation and valvular heart disease: A systematic review and meta-analysis. J Am Heart Assoc 2017;6:e005835.
- Di Biase L. Use of direct oral anticoagulants in patients with atrial fibrillation and valvular heart lesions. J Am Heart Assoc 2016;5:e002776.
- Ezekowitz MD, Nagarakanti R, Noack H, et al. Comparison of dabigatran and warfarin in patients with atrial fibrillation and valvular heart disease: The RE-LY Trial (Randomized Evaluation of Long-Term Anticoagulant Therapy). Circulation 2016;134:589-598.
- Chugh SS, Havmoeller R, Narayanan K, et al. Worldwide epidemiology of atrial fibrillation: A Global Burden of Disease 2010 Study. Circulation 2014;129:837-847.
- Colilla S, Crow A, Petkun W, et al. Estimates of current and future incidence and prevalence of atrial fibrillation in the U.S. adult population. Am J Cardiol 2013;112:1142-1147.
- Go AS, Hylek EM, Phillips KA, et al. Prevalence of diagnosed atrial fibrillation in adults: National implications for rhythm management and stroke prevention: The AnTicoagulation and Risk Factors in Atrial Fibrillation (ATRIA) Study. JAMA 2001;285:2370-2375.
- Miyasaka Y, Barnes ME, Gersh BJ, et al. Secular trends in incidence of atrial fibrillation in Olmsted County, Minnesota, 1980 to 2000, and implications on the projections for future prevalence [published correction appears in Circulation 2006;114:e498]. Circulation 2006;114:119-125.
- Piccini JP, Hammill BG, Sinner MF, et al. Incidence and prevalence of atrial fibrillation and associated mortality among Medicare beneficiaries, 1993-2007. Circ Cardiovasc Qual Outcomes 2012;5:85-93.
- Wang TJ, Larson MG, Levy D, et al. Temporal relations of atrial fibrillation and congestive heart failure and their joint influence on mortality: The Framingham Heart Study. Circulation 2003;107:2920-2925.
- Furberg CD, Psaty BM, Manolio TA, et al. Prevalence of atrial fibrillation in elderly subjects (the Cardiovascular Health Study). Am J Cardiol 1994;74:236-241.
- Dewland TA, Olgin JE, Vittinghoff E, et al. Incident atrial fibrillation among Asians, Hispanics, Blacks, and Whites. Circulation 2013;128:2470-2477.
- Lau CP, Gbadebo TD, Connolly SJ, et al. Ethnic differences in atrial fibrillation identified using implanted cardiac devices. J Cardiovasc Electrophysiol 2013;24:381-387.
- Magnussen C, Niiranen TJ, Ojeda FM, et al. Sex differences and similarities in atrial fibrillation epidemiology, risk factors, and mortality in community cohorts: Results from the BiomarCaRE Consortium (Biomarker for Cardiovascular Risk Assessment in Europe). Circulation 2017;136:1588-1597.
- Mou L, Norby FL, Chen LY, et al. Lifetime risk of atrial fibrillation by race and socioeconomic status: ARIC study (Atherosclerosis Risk in Communities). Circ Arrhythm Electrophysiol 2018;11: e006350.
- Benjamin EJ, Muntner P, Alonso A, et al. Heart disease and stroke statistics-2019 update: A report from the American Heart Association. Circulation 2019;139:e56-e528. doi: 10.1161/CIR. 0000000000000659.
- Fox CS, Parise H, D’Agostino RB Sr, et al. Parental atrial fibrillation as a risk factor for atrial fibrillation in offspring. JAMA 2004;291:2851-2855.
- Oyen N, Ranthe MF, Carstensen L, et al. Familial aggregation of lone atrial fibrillation in young persons. J Am Coll Cardiol 2012;60:917-921.
- Chen YH, Xu SJ, Bendahhou S, et al. KCNQ1 gain-of-function mutation in familial atrial fibrillation. Science 2003;299:251-254.
- Olesen MS, Nielsen MW, Haunso S, et al. Atrial fibrillation: The role of common and rare genetic variants. Eur J Hum Genet 2014;22:297-306.
- Sinner MF, Tucker NR, Lunetta KL, et al. Integrating genetic, transcriptional, and functional analyses to identify 5 novel genes for atrial fibrillation. Circulation 2014;130:1225-1235.
- Benjamin SM, Muhammad R, Parvez B, et al. Common atrial fibrillation risk alleles at 4q25 predict recurrence after catheter-based atrial fibrillation ablation. Heart Rhythm 2013;10:394-400.
- Franco D, Chinchilla A, Daimi H, et al. Modulation of conductive elements by Pitx2 and their impact on atrial arrhythmogenesis. Cardiovasc Res 2011;91:223-231.
- Wang J, Bai Y, Li N, et al. Pitx2-microRNA pathway that delimits sinoatrial node development and inhibits predisposition to atrial fibrillation. Proc Natl Acad Sci U S A 2014;111:9181-9186.
- Tada H, Shiffman D, Smith JG, et al. Twelve-single nucleotide polymorphism genetic risk score identifies individuals at increased risk for future atrial fibrillation and stroke. Stroke 2014;45:2856-2862.
- Chimenti C, Russo MA, Carpi A, et al. Histological substrate of human atrial fibrillation. Biomed Pharmacother 2010;64:177-183.
- Nguyen BL, Fishbein MC, Chen LS, et al. Histopathological substrate for chronic atrial fibrillation in humans. Heart Rhythm 2009;6:454-460.
- Xu J, Cui G, Esmailian F, et al. Atrial extracellular matrix remodeling and the maintenance of atrial fibrillation. Circulation 2004;109:363-368.
- Gramley F, Lorenzen J, Plisiene J, et al. Decreased plasminogen activator inhibitor and tissue metalloproteinase inhibitor expression may promote increased metalloproteinase activity with increasing duration of human atrial fibrillation. J Cardiovasc Electrophysiol 2007;18:1076-1082
- Allessie MA, de Groot NM, Houben RP, et al. Electropathological substrate of long-standing persistent atrial fibrillation in patients with structural heart disease: Longitudinal dissociation. Circ Arrhythm Electrophysiol 2010;3:606-615.
- Spach MS, Josephson ME. Initiating reentry: The role of nonuniform anisotropy in small circuits. J Cardiovasc Electrophysiol 1994;5:182-209.
- Venteclef N, Guglielmi V, Balse E, et al. Human epicardial adipose tissue induces fibrosis of the atrial myocardium through the secretion of adipo-fibrokines. Eur Heart J 2013;36:795-805a.
- Rocken C, Peters B, Juenemann G, et al. Atrial amyloidosis: An arrhythmogenic substrate for persistent atrial fibrillation. Circulation 2002;106:2091-2097.
- Schmitt C, Ndrepepa G, Weber S, et al. Biatrial multisite mapping of atrial premature complexes triggering onset of atrial fibrillation. Am J Cardiol 2002;89:1381-1387.
- Lin WS, Ching-Tai T, Ming-Hsiung H, et al. Catheter ablation of paroxysmal atrial fibrillation initiated by non-pulmonary vein ectopy. Circulation 2003;107:3176-3183.
- Haïssaguerre M, Jaïs P, Shah DC, et al. Spontaneous initiation of atrial fibrillation by ectopic beats originating in the pulmonary veins. N Engl J Med 1998;339:659-666.
- Lau CP, Tse HF, Ayers GM. Defibrillation-guided radiofrequency ablation of atrial fibrillation secondary to an atrial focus. J Am Coll Cardiol 1999;33:1217-1226.
- Bettoni M, Zimmermann M. Autonomic tone variations before the onset of paroxysmal atrial fibrillation. Circulation 2002;105:2753-2759.
- Shen MJ, Choi EK, Tan AY, et al. Neural mechanisms of atrial arrhythmias. Nat Rev Cardiol 2011;9:30-39.
- Ishii Y, Schuessler RB, Gaynor SL, et al. Inflammation of atrium after cardiac surgery is associated with inhomogeneity of atrial conduction and atrial fibrillation. Circulation 2005;111:2881-2888.
- Goldstein RN, Ryu K, Khrestian C, et al. Prednisone prevents inducible atrial flutter in the canine sterile pericarditis model. J Cardiovasc Electrophysiol 2008;19:74-81.
- Creswell LL, Schuessler RB, Rosenbloom M, Cox JL. Hazards of postoperative atrial arrhythmias. Ann Thorac Surg 1993;56:539-549.
- Almassi GH, Schowalter T, Nicolosi AC, et al. Atrial fibrillation after cardiac surgery: A major morbid event? Ann Surg 1997;226:501-511.
- Moe GK, Abildskov JA. Atrial fibrillation as a self-sustaining arrhythmia independent of focal discharge. Am Heart J 1959;58:59-70.
- Van Wagoner DR, Pond AL, Lamorgese M, et al. Atrial L-type Ca2+ currents and human atrial fibrillation. Circ Res 1999;85:428-436.
- Schotten U, Verheule S, Kirchhof P, et al. Pathophysiological mechanisms of atrial fibrillation: A translational appraisal. Physiol Rev 2011;91:265-325.
- Wijffels MC, Kirchhof CJ, Dorland R, Allessie MA. Atrial fibrillation begets atrial fibrillation. A study in awake chronically instrumented goats. Circulation 1995;92:1954-1968.
- Rodriguez LM, Timmermans C, Wellens HJ. Are electrophysiological changes induced by longer lasting atrial fibrillation reversible?: Observations using the atrial defibrillator. Circulation 1999;100:113-116.
- Nattel S, Opie LH. Controversies in atrial fibrillation. Lancet 2006;367:262-272.
- Watson T, Shantsila E, Lip GY. Mechanisms of thrombogenesis in atrial fibrillation: Virchow's triad revisited. Lancet 2009;373:155-166.
- Waldo AL, Feld GK. Inter-relationships of atrial fibrillation and atrial flutter mechanisms and clinical implications. J Am Coll Cardiol 2008;51:779-786.
- Schnabel RB, Yin X, Gona P, et al. 50 year trends in atrial fibrillation prevalence, incidence, risk factors, and mortality in the Framingham Heart Study: A cohort study. Lancet 2015;386:154-162.
- Psaty BM, Manolio TA, Kuller LH, et al. Incidence of and risk factors for atrial fibrillation in older adults. Circulation 1997;96:2455-2461.
- Selmer C, Olesen JB, Hansen ML, et al. The spectrum of thyroid disease and risk of new onset atrial fibrillation: A large population cohort study. BMJ 2012;345:e7895.
- Gami AS, Hodge DO, Herges RM, et al. Obstructive sleep apnea, obesity, and the risk of incident atrial fibrillation. J Am Coll Cardiol 2007;49:565-571.
- Buch P, Friberg J, Scharling H, et al. Reduced lung function and risk of atrial fibrillation in the Copenhagen City Heart Study. Eur Respir J 2003;21:1012-1016.
- Baber U, Howard VJ, Halperin JL, et al. Association of chronic kidney disease with atrial fibrillation among adults in the United States: REasons for Geographic and Racial Differences in Stroke (REGARDS) Study. Circ Arrhythm Electrophysiol 2011;4:26-32.
- Larsson SC, Drca N,Wolk A. Alcohol consumption and risk of atrial fibrillation: A prospective study and dose-response meta-analysis. J Am Coll Cardiol 2014;64:281-289.
- Chamberlain AM, Agarwal SK, Folsom AR, et al. Smoking and incidence of atrial fibrillation: Results from the Atherosclerosis Risk in Communities (ARIC) study. Heart Rhythm 2011;8:1160-1166.
- Aizer A, Gaziano JM, Cook NR, et al. Relation of vigorous exercise to risk of atrial fibrillation. Am J Cardiol 2009;103:1572-1577.
- De Decker K, Jorens PG, Van Schil P. Cardiac complications after noncardiac thoracic surgery: An evidence-based current review. Ann Thorac Surg 2003;75:1340-1348.
- Sanna T, Diener H-C, Passman RS, et al. Cryptogenic stroke and underlying atrial fibrillation. N Engl J Med 2014;370:2478-2486.
- January CT, Wann LS, Alpert JS, et al. 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation. J Am Coll Cardiol 2014;64:e1-e76. doi: 10.1016/j.jacc.2014.03.022.
- Kotecha D, Piccini JP. Atrial fibrillation in heart failure: What should we do? Eur Heart J 2015;36:3250-3257.
- Olsson LG, Swedberg K, Ducharme A, et al. Atrial fibrillation and risk of clinical events in chronic heart failure with and without left ventricular systolic dysfunction: Results from the Candesartan in Heart failure-Assessment of Reduction in Mortality and morbidity (CHARM) program. J Am Coll Cardiol 2006;47:1997-2004.
- Kotecha D, Chudasama R, Lane DA, et al. Atrial fibrillation and heart failure due to reduced versus preserved ejection fraction: A systematic review and meta-analysis of death and adverse outcomes. Int J Cardiol 2016;203:660-666.
- Segal JB, McNamara RL, Miller MR, et al. The evidence regarding the drugs used for ventricular rate control. J Fam Pract 2000;49:47-59.
- Schreck DM, Rivera AR, Tricarico VJ. Emergency management of atrial fibrillation and flutter: Intravenous diltiazem versus intravenous digoxin. Ann Emerg Med 1997;29:135-140.
- Siu CW, Lau CP, Lee WL, et al. Intravenous diltiazem is superior to intravenous amiodarone or digoxin for achieving ventricular rate control in patients with acute uncomplicated atrial fibrillation. Crit Care Med 2009;37:2174-2179.
- Boahene KA, Klein GJ, Yee R, et al. Termination of acute atrial fibrillation in the Wolff-Parkinson-White syndrome by procainamide and propafenone: Importance of atrial fibrillatory cycle length. J Am Coll Cardiol 1990;16:1408-1414.
- O’Nunain S, Garratt CJ, Linker NJ, et al. A comparison of intravenous propafenone and flecainide in the treatment of tachycardias associated with the Wolff-Parkinson-White syndrome. Pacing Clin Electrophysiol 1991;14:2028-2034.
- Tischenko A, Fox DJ, Yee R, et al. When should we recommend catheter ablation for patients with the Wolff-Parkinson-White syndrome? Curr Opin Cardiol 2008;23:32-37.
This two-part series presents a review of the current evidence on atrial fibrillation. The first part includes its definition, classification, risk factors, comorbidities, evaluation, and acute management of newly diagnosed patients. The second part will focus on long-term management, including risk factor modification, rate and rhythm control measures, stroke risk stratification, and anticoagulation management.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
