A Review of Atrial Fibrillation and Current Therapeutic Strategies: Part 2
December 1, 2019
Reprints
AUTHORS
Hunter Mwansa, MD, St. Vincent Charity Medical Center, Case Western Reserve University, Cleveland, OH
Sula Mazimba, MD, MPH, Division of Cardiovascular Medicine, University of Virginia Health System, Charlottesville
PEER REVIEWER
Glen D. Solomon, MD, FACP, Professor and Chair, Department of Internal Medicine, Wright State University Boonshoft School of Medicine, Dayton, OH
EXECUTIVE SUMMARY
Atrial fibrillation (AF) is the most common arrhythmia, affecting nearly 34 million people globally in 2010. In the United States, it is estimated to reach 12.1 million by 2030. Associated with increased morbidity and mortality, AF is characterized by uncoordinated atrial electrical activity leading to compromised ventricular filling and hemodynamic instability.
- AF is commonly categorized based on the temporal nature of the disorder (paroxysmal, acute, or chronic), its response to cardioversion (persistent vs. permanent), and the presence or absence of structural heart disease (valvular vs. nonvalvular).
- Pathophysiologic mechanisms include genetic predisposition, and anatomical and electrophysiologic abnormalities, which may be primary or secondary to conditions such as valvular heart disease, heart failure, hypertension, diabetes mellitus, obstructive sleep apnea, and alcohol or tobacco use.
- Thrombogenesis is the most feared consequence of AF leading to cerebral vascular accidents.
- Acute management typically involves beta-blockers, calcium channel blockers, and/or digoxin to achieve rate control. Long-term mortality outcomes have been related mostly to appropriate and effective anticoagulation.
This two-part series presents a review of the current evidence on atrial fibrillation (AF). The first part included the definition of AF, its classification, risk factors, comorbidities, evaluation, and acute management of newly diagnosed patients. The second part focuses on long-term management, including risk factor modification, rate and rhythm control measures, stroke risk stratification, and anticoagulation management.
Long-Term Management of Atrial Fibrillation
Therapeutic considerations for AF include the long-term management of symptoms with either heart rate or rhythm control strategies, modification of risk factors, and management of comorbid medical conditions and anticoagulation. Ultimately, the preferred strategy needs to take into consideration informed patient choices after education on the available treatment options (rate control vs. rhythm control), including the potential harms and benefits of therapy.
Rate Control Strategy
Most patients with AF can be managed safely using measures aimed at addressing heart rate (HR) control and anticoagulation. Principles guiding long-term rate control of AF are not significantly different from those earlier advanced in the Acute Management of AF section in Part 1. Randomized clinical trials comparing outcomes of rate control vs. rhythm control strategies of AF management have demonstrated no major differences in mortality and stroke rates among patients randomized to either strategy. For example, the Atrial Fibrillation Follow-up Investigation of Rhythm Management (AFFIRM) trial showed that the rhythm strategy offered no survival advantage when compared to the rate control strategy among patients with AF.1
Additionally, stroke rates were not significantly different between the two treatment strategies.2 Furthermore, the rate control group had a lower incidence of adverse events and hospitalization than the rhythm control group.
Evidence has shown that oral beta-blocker therapy is the most effective single agent option among rate controlling medications and therefore should be attempted first in patients without contraindications. In the AFFIRM study, using approaches to control rate in AF, 70% of patients given a beta-blocker first (with or without digoxin) achieved overall rate control (defined as resting heart rate [HR] 60-80 bpm and a maximum HR of 110 bpm with exercise).3
Patients with contraindications to beta-blockers (e.g., severe bronchospastic asthma) may benefit from calcium channel blockers therapy, while those with hypotension and chronic systolic heart failure might be suitable candidates for digoxin. As shown by the AFFIRM study, most patients can achieve rate control with available rate-controlling drugs, but medication switches and combination therapy usually are necessary. However, there is no clear consensus on what constitutes adequate rate control. The RACE II (Rate Control Efficacy in Permanent Atrial Fibrillation) compared clinical outcomes between cohorts randomized to a lenient rate control strategy (resting HR < 110 bpm) and a strict rate control strategy (resting HR < 80 and HR during moderate exercise < 110 bpm); the primary outcome was a composite of cardiovascular death, hospitalization for HF, stroke, systemic embolism, bleeding, and life-threatening arrhythmias. At three years, the estimated cumulative incidence of the primary outcome was 12.9% in the lenient-control group and 14.9% in the strict-control group. Based on this landmark study, present guidelines advocate for more lenient rate control with a caveat that left ventricle (LV) function may deteriorate when tachycardia goes unabated for a long time. Interestingly, many patients with resting heart rates between 80-100 bpm can remain symptomatic, warranting further symptomatic management.4
Nonpharmacologic Rate Control
Rarely, patients with symptomatic medically refractory AF elect to undergo catheter atrioventricular (AV) node ablation and subsequent permanent pacemaker implantation (ablation and pacing therapy) as a rate control measure. This intervention has not been evaluated extensively by randomized controlled clinical trials, and evidence from multiple small uncontrolled trials demonstrated variable patient outcomes.5-8 However, meta-analysis of these findings demonstrated significant reduction in cardiac symptoms and healthcare utilization and improvement in variable measures, including quality of life, exercise endurance, and ejection fraction.9 Nevertheless, chronic right ventricular pacing post-AV ablation can cause a compromise in cardiac function, driven in part by LV dyssynchrony, that can provoke an exacerbation of HF. This has been observed even in patients with normal LV function prior to ablation and pacemaker placement.10,11 Notwithstanding these findings, a more recent study showed that the increases in hospitalization for congestive HF were observed predominantly among patients with HF with reduced ejection fraction (HFrEF) and in particular those with previous hospitalization for HF and/or pre-existing coronary artery disease (CAD) at the time of AV node ablation and pacing.12 Improvement in six-minute walk distance and LV ejection fraction at six months of follow-up was previously demonstrated with biventricular pacemaker implantation post-AV node ablation in patients with HF having EF ≤ 45% or New York Heart Association (NYHA) functional class II/III symptoms. However, the study did not show a significant difference in quality of life measures between patients randomized to biventricular pacing and right ventricular pacing.
Rhythm Control Strategy
Some patients may not be candidates for rate control strategy because of contraindications (e.g., medical comorbidities), intolerance to medications, or a personal preference for the rhythm control strategy. These patients make ideal candidates for the rhythm control strategy. Most commonly, the rhythm control strategy involves the use of either synchronized direct current (DC) cardioversion or pharmacologic (antiarrhythmic drugs [AADs]) cardioversion alone, or in combination, depending on patient characteristics and overall risk for recurrent AF. Rhythm control can further be achieved using invasive strategies like catheter ablation and AF surgery. Except for scenarios discussed in Part 1 (under Acute Management of AF), cardioversion generally is done on an elective basis. Both pharmacologic and electrical cardioversion are associated with an elevated risk for stroke in non-anticoagulated patients.13 This risk is reduced significantly with the use of anticoagulation before cardioversion.14
Historically, the duration of AF (AF duration < 48 hours [acute] vs. AF duration ≥ 48 hours [chronic]) has been an important consideration in deciding the need for anticoagulation before cardioversion. Patients with AF duration < 48 hours traditionally would undergo immediate cardioversion, provided a transesophageal echocardiogram (TEE) excluded left atrial thrombi, while those with AF duration ≥ 48 hours underwent at least three weeks of anticoagulation before cardioversion. While a negative TEE can exclude the majority of left atrial thrombi, the safety of this practice has been thrown into question because of recent evidence that showed an increased risk of stroke in patients with new onset AF (duration < 48 hours) having a CHA2DS2-VASc (Congestive HF, HTN, Age [doubled], Diabetes mellitus, Stroke/TIA/thromboembolism [doubled], Vascular disease [coronary artery disease, peripheral artery disease, and aortic atheroma]) score ≥ 2, who were not adequately anticoagulated before undergoing cardioversion (incidence of neurological events 1.06% compared to 0.22% for patients on anticoagulation [odds ratio, 4.8; P = 0.03]).13-15 Therefore, all AF patients with significant risk for thromboembolism must be adequately anticoagulated before undergoing elective cardioversion, and this must be continued for about four weeks post-cardioversion to mitigate stroke risk.16
Table 1. Antiarrhythmic Drugs (AADs) Commonly Used for Rhythm Management of AF |
||
|
Drug |
Class |
Contraindications and Potential Adverse Effects |
|
Amiodarone* |
IB, III |
|
|
Dronedarone |
IB |
|
|
Flecainide* |
IC |
|
|
Propafenone |
IC |
|
|
Ibutilide* |
III |
|
|
Procainamide* |
IA |
|
|
d, I Sotalol |
II, III |
|
|
*AADs also used for pharmacologic cardioversion AV = atrioventricular; TCAs = tricyclic antidepressants; SA = sinoatrial; VKAs = vitamin K antagonists; NYHA = New York Heart Association; HF = heart failure; SSS = sick sinus syndrome; QTc = corrected QT interval; TdP = torsades de pointes; msec = milliseconds; IHD = ischemic heart disease; MI = myocardial infarction; LV = left ventricular; CrCl = creatinine clearance; SSRIs = selective serotonin reuptake inhibitors; TdP = torsades de pointes; LVH = left ventricular hypertrophy; SLE = systemic lupus erythematosus; mEq = milliequivalents |
||
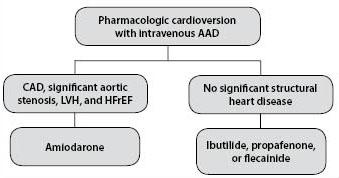
Restoration of sinus rhythm through either pharmacologic or electrical cardioversion might be particularly helpful in ameliorating AF-related symptoms. This may be especially helpful in AF patients with symptoms refractory to medical therapy. AAD therapy can be effective in restoring sinus rhythm in 50% of patients with AF.19 However, electrical cardioversion is more effective at restoring sinus rhythm than pharmacologic cardioversion,20 and also is associated with shorter hospitalization in the short term.21,22 It also has fewer adverse effects compared to antiarrhythmics, which may be associated with cardiac and extracardiac adverse effects as illustrated in Table 1. The choice between electrical and pharmacologic cardioversion ultimately depends on cardiac and medical comorbidities and patient preferences. Common AADs for achievement of pharmacologic cardioversion include flecainide, propafenone, ibutilide, and amiodarone. The presence of coronary artery disease, heart failure with reduced ejection fraction (HFrEF), or structural or valvular heart disease (VHD) dictates the choice of AAD for pharmacologic cardioversion. (See Figure 1.)
Figure 1. Pharmacologic Conversion |
 |
|
AAD = antiarrhythmic drug ; CAD = coronary artery disease; LVH = left ventricular hypertrophy; HFrEF = heart failure with reduced ejection fraction |
Not uncommonly, AADs also are used in combination with electrical cardioversion to enhance rates of successful electrical cardioversion in the short term (pre- and post-cardioversion for a limited period) or as an adjunct to electrical cardioversion for long term maintenance of sinus rhythm. Pretreatment with flecainide,23 propafenone,24 sotalol,25 or amiodarone26 (typically longer duration for amiodarone) is associated with improved efficacy of cardioversion. Post-cardioversion AAD therapy, either short-term (less than four weeks) or long-term (greater than or equal to four weeks), improves maintenance of sinus rhythm, although long-term AAD therapy has proven more effective.23
AADs are associated with significant potential for toxicity and adverse effects. Short-term AAD therapy, particularly in those perceived to be at low risk for recurrent AF, may prove reasonably effective and safer than long-term therapy. For example, four weeks of flecainide post-cardioversion proved effective (prevented 80% of AF recurrences) and more tolerable than long-term AAD therapy.23
Ultimately, safety rather than efficacy should inform the choice of AAD for long-term rhythm management. Decisions on whether to combine electrical cardioversion with either short- or long-term AAD therapy should focus on individual patient characteristics, including risk for recurrent AF and potential for AAD toxicity.
Patients on AAD for rhythm control require close monitoring. A baseline ECG (paying attention to the PR interval, QRS complex, and QT interval, and the presence of conduction abnormalities and TU wave abnormalities), and liver and renal function test can help inform the choice of AAD. Follow-up ECGs and laboratory tests, including electrolytes, can reveal ominous signs and avert potentially serious and life-threatening adverse effects. Long-term AAD therapy should be routinely scrutinized to ascertain the need for ongoing therapy, emergence of contraindications, and potential drug-to-drug interactions, and adjusted accordingly or discontinued while working with the patient’s cardiologist to mitigate harm. Further, it is important to understand that most clinical trials have demonstrated no appreciable mortality or morbidity benefit from AAD therapy, and some have suggested an increase in AF-related hospitalizations.27,28
Catheter Ablation for AF
Catheter ablation is a common form of rhythm control strategy for the treatment of recurrent AF. Catheter ablation primarily involves isolation of the pulmonary veins, and when performed in experienced centers, is more effective than AAD therapy at restoring and maintaining sinus rhythm and has similar complication rates.29
This effectiveness has been demonstrated in patients with paroxysmal and persistent AF who required ablation therapy after failure of or intolerance to AAD therapy.29-32
Randomized trials demonstrated similar complications, but with modest improvement in rhythm outcomes compared to AAD therapy when catheter ablation was done as initial therapy, making it a viable first-line therapeutic option for patients opposed to AAD therapy.33-35 There’s a paucity of data to support catheter ablation as a first-line treatment option in patients with persistent and longstanding persistent AF, but the current evidence base argues for the superiority of catheter ablation to AAD therapy in the maintenance of sinus rhythm in patients with symptomatic recurrences of AF while on AAD therapy.36,37
Catheter ablation has evolved over the years and several techniques exist, ranging from primary pulmonary vein isolation (PVI) to extended ablation procedures. Nevertheless, complete PVI achievable through targeted point-to-point or creation of circumferential linear lesions around the pulmonary vein via radiofrequency ablation, or cryoballoon ablation, has proven to be superior to incomplete PVI38 and noninferior to more extensive ablative procedures in patients with paroxysmal and permanent AF.39,40
It is difficult to predict outcomes following AF ablation, but as much as 70% of patients with paroxysmal AF and 50% of patients with persistent AF achieve maintenance of sinus rhythm with few incidences of severe symptomatic episodes of AF.16 Generally, rhythm and safety outcomes are better in young patients without significant structural heart disease who have a short history of AF and whose AF paroxysms are frequent but short-lived.41
AF ablation is not without complications. Given the recent popularity of catheter ablation, clinicians need to be aware of its potential complications and how to recognize them to mitigate possible death and long-term morbidity. About 5-7% of patients undergoing AF ablation will experience severe complications, while 2-3% suffer life-threatening complications.36,42,43 Life-threatening complications include peri-procedural death (< 0.2%); esophageal injury, including atrio-esophageal fistula development (characterized by fever without clear focus of infection, retrosternal chest pain, and stroke/transient ischemic attack [TIA] symptoms) (< 0.5%); periprocedural stroke (< 1%); and cardiac tamponade (1-2%).16 Severe complications include vascular complications, such as arteriovenous malformations and femoral pseudoaneurysms (2-4%), persistent phrenic nerve palsy (1-2%), and pulmonary vein stenosis (< 1%). Silent strokes, likely representing cerebral embolism, have been reported to occur in 5-20% of patients, but their clinical significance has yet to be known.16
Catheter ablation is associated with elevated stroke risk, and all patients undergoing this intervention require therapeutic anticoagulation and must continue on anticoagulation for a minimum of eight weeks even if they have no indication for lifelong anticoagulation.16
Ablation of AF in HF Patients
Catheter ablation has been shown to be more effective than amiodarone therapy in reducing recurrent AF in patients with HFrEF. Multiple small studies have suggested improvement in hospitalization and LV function following catheter ablation for patients with HFrEF, particularly those without prior myocardial infarction.44-46
Most recently, the authors of the CASTLE (Catheter Ablation versus Standard Conventional Treatment in patients with LV Dysfunction and AF) study evaluated patients with concomitant AF and HFrEF who had implantable cardioverter defibrillator or cardiac resynchronization defibrillator device, whose AF was refractory to or who were intolerant of AAD therapy, by randomizing them to either ablation therapy or medical therapy (rate or rhythm control).47
The study revealed that patients who were randomized to catheter ablation had decreased overall mortality and rates of hospitalization for worsening HF, improved LV function, and higher numbers of patients in sinus rhythm. However, the CABANA (Catheter Ablation versus AAD Therapy for AF) trial showed different results when it compared the outcomes of catheter ablation and AAD therapy in patients with new-onset or untreated AF requiring medical therapy after a five-year follow-up.48 Findings from the CABANA study demonstrated that catheter ablation was not superior to AAD therapy for the primary outcome of mortality, thromboembolism, and bleeding events. Clearly, larger RCTs are needed to help establish the long-term efficacy and safety of catheter ablation in several subgroups of AF patients.
AF Surgery
The Cox maze surgery is a surgical ablation technique that is conducted to isolate atrial tissue thought to be involved in the creation of triggers, ectopy, and micro-reentry implicated in propagation and maintenance of AF. Incisions in the atrial wall are made strategically with a view to creating an electrical maze or labyrinth of passages that allow for conduction of electrical impulses to the atrioventricular node while preventing propagation of fibrillatory waves. The procedure also achieves surgical exclusion or closure of the left atrial appendage, thereby mitigating stroke risk. The procedure is limited to patients undergoing open-heart surgery (coronary artery bypass and valve surgery). A Cochrane review of the evidence base for concomitant Cox maze surgery for AF patients undergoing open-heart surgery revealed an increase in freedom from AF, atrial flutter, and atrial tachycardia as compared to no concomitant AF surgery.49 Similar findings confirming the efficacy and safety of the maze procedure have been documented, with a major downside being an incremental risk for pacemaker implantation post-maze procedure (adjusted odds ratio 1.26; 95% confidence interval [CI], 1.07-1.49).50
Appropriate patient selection that focuses on avoiding factors that increase chances of AF recurrence post-maze, including older age, AF duration > 10 years, and existence of non-paroxysmal AF, may help optimize outcomes.51-55
Now, the Cox maze procedure can be accomplished as a standalone surgery thoracoscopically. This approach has proven to be effective in preventing recurrent AF and maintaining sinus rhythm in patients with recurrent AF post-catheter ablation but it is associated with significantly higher rates of serious complications, among them conversion to sternotomy, pneumothorax, pericardial effusion, TIA, and pacemaker implantation.56
Prevention of Stroke and Systemic Thromboembolism in AF
Stroke Risk Stratification Tools. Both symptomatic and silent AF, irrespective of their pattern or classification, confer an elevated risk for ischemic thromboembolic stroke.57-59 Compared to patients in sinus rhythm, patients with valvular AF and nonvalvular AF have an increased stroke risk by more than 20 times and five times, respectively.60 Unfortunately, AF-related stroke has a high recurrence rate and incurs more disability and death.61 Oral anticoagulant (OAC) therapy can mitigate stroke risk and improve mortality in AF patients.62-64
In AF patients with elevated stroke risk, OAC is superior to either aspirin or placebo in preventing stroke.65,66 Despite the robust evidence of benefit in stroke prevention, OAC therapy is underused and very often discontinued because of bleeding concerns in those who might have been on OAC therapy.67-70
Although OAC therapy elevates bleeding risk, the overall benefit of OAC in decreasing stroke risk and its attendant complications - such as death and disability, even among the elderly and cognitively impaired, with or without frailty and/or significant fall risk - warrant its use.71,72
In fact, OACs have similar bleeding risk to aspirin, but the latter has no demonstrated efficacy in AF-related stroke prevention except when other compelling indications for its use exist.73,74
AF-Related Thromboembolism and Bleeding Risk Prediction Tools. AF registry data have been crucial in developing, validating, and refining stroke risk stratification tools among patients with nonvalvular AF. The CHADS2 score was derived from European registry data, and study findings showing an increase in stroke rate of about 2.0% for a corresponding one-point rise in the CHADS2 score led to its validation and widespread clinical use.75
However, because of its significant limitations in identifying patients at lower risk of stroke (lowest score of 1 on the CHADS2 was defined as “intermediate risk”), inclusion of more clinical variables to refine the tool led to the development of the CHA2DS2-VASc score. (See Table 2.) The CHA2DS2-VASc score proved to better discriminate stroke risk among patients with a baseline CHADS2 score of 0-1 and more clearly defined and aided decisions on anticoagulation than the CHADS2 score.76,77 These studies demonstrated that older women had a significantly higher risk of stroke compared to men, but women younger than 65 years of age who had no other AF risk factors had a low risk of stroke.78 Despite the superiority of the CHA2DS2-VASc, both tools underestimate the risk of stroke in those with prior ischemic events.79
Table 2. CHA2DS2-VASc Score for Stratification of Thromboembolism Risk in Nonvalvular AF |
|
|
CHA2DS2-VASc risk factor79 |
Score |
|
Congestive heart failure Symptoms/signs of congestive HF or objective evidence of reduced LVEF |
1 |
|
Hypertension (confirmed diagnosis or presence of antihypertensive drug therapy) |
1 |
|
Age ≥ 75 years |
2 |
|
Diabetes mellitus (confirmed diagnosis or active pharmacotherapy for the disease) |
1 |
|
Prior stroke/TIA/Thromboembolism |
2 |
|
Vascular disease (prior MI, PAD, or aortic plaque) |
1 |
|
Age 65-74 years |
1 |
|
Sex category (female) |
1 |
|
CHA2DS2-VASc = Congestive HF, HTN, Age ≥ 75 (doubled), Diabetes, Stroke/TIA/Thromboembolism (doubled), Vascular disease (CAD, PAD, and aortic plaque), Age 65–74, and Sex (female); LVEF = left ventricular ejection fraction |
|
Therefore, the CHA2DS2-VASc has been recommended for stroke risk estimation and can aid decision-making on initial OAC therapy in patients with nonvalvular AF. Evidence from controlled trials indicates an elevated stroke risk for men and women with a CHA2DS2-VASc score of ≥ 2 and ≥ 3, respectively.64,79,81,82 These patients require OAC for stroke prevention. In general, patients with nonvalvular AF with CHA2DS2-VASc scores below this threshold can forego anticoagulation.
A variable international normalized ratio (INR) and short time in the therapeutic range (TTR) in patients treated with vitamin K antagonists (VKAs) and elevated troponin and N-terminal pro-B-type natriuretic peptide also have been shown to have predictive and prognostic value in AF patients, but their clinical role is undefined at present.83,84
Bleeding Risk Stratification Tools. Concerns for bleeding complications while on anticoagulation led to the development of several bleeding risk scores. Some of these scores include HAS-BLED (Hypertension [HTN] [systolic blood pressure > 160 mmHg], Abnormal renal/liver function, Stroke, Bleeding history or predisposition, Labile INR, Elderly [age > 65 years], Drugs/alcohol concomitantly),85 ABC (Age, Biomarkers, and Clinical history),86 ATRIA (Anticoagulation and Risk Factors in Atrial Fibrillation),87 and a composite of bleeding risk factors from other scoring schemes called HEMORR2HAGES (Hepatic or renal disease, Ethanol abuse, Malignancy, Older age [> 75 years], Reduced platelet count or function, Hypertension [uncontrolled], Anemia, Genetic factors, Excessive fall risk, and Stroke), which assigns two points for a prior bleed and one point for each of the other clinical variables.
A HAS-BLED score ≥ 3 translates to a potentially high risk of bleeding, which warrants close monitoring, redress of modifiable bleeding risk factors, and possible dose adjustments to OAC therapy. The ATRIA score incorporates anemia, severe renal disease, age > 75 years, prior hemorrhage, and hypertension and assigns them scores of 3, 3, 2, 1, and 1, respectively. While these scores may inform bleeding risk in patients with AF, their clinical utility is less clear. The HAS-BLED discriminates bleeding risk better than either the ATRIA or HEMORR2HAGES, but they all appear to have poor predictive accuracy.85 Older age is a risk factor for both stroke and bleeding in AF patients, and this overlap can complicate OAC treatment decisions.86,88 Treatment decisions generally should be based on a balance of benefits and potential harms. A high bleeding risk should prompt attention to modifiable bleeding risk factors (uncontrolled hypertension, labile INR, medications [e.g., nonsteroidal anti-inflammatory drugs and antiplatelet agents like aspirin, clopidogrel]) and should not necessarily preclude OAC therapy. Informed patient choices should ultimately direct decisions on anticoagulation.
Thromboembolism Prevention in AF
As alluded to earlier, thromboembolism and stroke are the most important complications of AF. Appropriate use of an OAC, using either a VKA or non-vitamin K oral anticoagulant (NOAC), is proven to lower stroke risk,89 despite reports of residual stroke risk (estimated at 1.7% for warfarin and 1.4% per year for NOAC at 2.2 years follow-up).89
Comorbidity control (e.g., addressing modifiable and potentially modifiable AF risk factors, including effective treatment of cardiovascular conditions, diabetes, and weight loss) and stroke prevention are arguably the most important aspects of AF management because of their prognostic value. The type of AF (valvular or nonvalvular) dictates the choice of oral anticoagulation, since no NOAC has proven efficacy and safety in the anticoagulation of patients with valvular AF.
Stroke prevention is attainable pharmacologically, using either a VKA or a NOAC, and non-pharmacologically.
Vitamin K Oral Anticoagulants. Warfarin and other oral VKAs (commonly acenocoumarol and phenprocoumon in most European countries) are the oldest anticoagulants used in AF. VKAs exert their anticoagulant effect through inhibition of vitamin K epoxide reductase, which in essence prevents vitamin K-dependent γ-carboxylation of vitamin K-dependent coagulation factors II, VII, IX, and X. Compared to placebo, including aspirin therapy, VKA therapy has been documented to decrease stroke risk by two-thirds and overall mortality by one-fourth in nonvalvular AF patients.62 VKAs have inherent limitations, among them a narrow therapeutic window and multiple drug-drug and drug-food interactions, which necessitate close monitoring and frequent dosage adjustments in patients on VKA therapy. These limitations and the emergence of ease to administer NOACs requiring less frequent laboratory monitoring have led to a decline in VKA popularity in clinical practice. Universal predictability of efficacy for stroke prevention while on VKAs also has been questioned. However, when delivered with adequate time in the therapeutic range (TTR), VKA therapy is efficacious and safe.
SAMe-TT2R2, a score that incorporates patient-related clinical parameters to help determine the likelihood of effective anticoagulation through attainment of good INR control while on VKA therapy, has potential to improve the predictability of VKA outcomes on stroke prevention.90 Patients with SAMe-TT2R2 scores 0-1 (associated with higher average TTR) do well on VKA therapy, while those with scores ≥ 2 may need additional intervention to attain effective anticoagulation.91,92 Nonetheless, VKA therapy remains the only effective and safe oral anticoagulation option for patients with valvular AF. The RE-ALIGN (Randomized, Phase II Study to Evaluate the Safety and Pharmacokinetics of Oral Dabigatran Etexilate in Patients After Heart Valve Replacement) trial for the dose-validation of dabigatran vs. warfarin in patients with valvular AF undergoing mechanical valve replacement in the mitral and/or aortic position had to be stopped prematurely because of a prohibitively high rate of thromboembolism and bleeding events in the dabigatran arm. Until evidence of thromboembolism prevention efficacy and safety is validated, all NOACs, by extension, are contraindicated in patients with valvular AF.93
The goal INR for patients with nonvalvular AF is 2.0-3.0, as this level of anticoagulation has been shown to offer the optimal balance of effective anticoagulation for the improvement of both stroke risk and its attendant 30-day mortality and safety outcomes.94
Thromboembolism Prevention in Valvular AF
The INR goals for those with valvular AF depend on the type of valve (mechanical prosthesis vs. bioprosthetic), the location of the valve, and the surgical intervention (repair vs. replacement). Lifelong anticoagulation with a VKA is recommended to achieve a therapeutic INR (2.5-3.5) in AF patients with a mechanical mitral valve replacement (MVR) and patients with a mechanical aortic valve replacement (AVR).95,96 Additionally, studies have demonstrated a further 65% reduction in the risk of death or thromboembolism with concomitant VKA and aspirin therapy as compared to VKA therapy alone.97,98 Therefore, 75 mg to 100 mg of aspirin daily is recommended for these patients, although the benefits must be weighed against the risks of major bleeding.96 Patients with AF and native valvular heart disease (excluding those with bioprosthetic valves) who do not meet the criteria for valvular AF (presence of a mechanical prosthetic valve and/or moderate to severe mitral valve stenosis) should be assessed for stroke/thromboembolic risk with the CHA2DS2-VASc score, and those with a CHA2DS2-VASc score ≥ 2 should receive anticoagulation with either a NOAC or a VKA.96
Patients with bioprosthetic valves have a higher risk of stroke and thromboembolism, and this risk is significantly increased in the first 90 to 180 days of the postoperative period.99 These patients require anticoagulation with either a NOAC or a VKA with an INR goal of 2.0-3.0, regardless of their CHA2DS2-VASc score.
Non-Vitamin K Oral Anticoagulants. NOACs, also commonly referred to as direct acting oral anticoagulants (DOACs), include a direct thrombin inhibitor (dabigatran) and factor Xa inhibitors (rivaroxaban, apixaban, and edoxaban). Unlike VKAs, these drugs have predictable effects and require no close laboratory monitoring. Most clinical trials comparing NOACs to warfarin revealed their non-inferiority100,101 or superiority102,103 to warfarin in decreasing stroke or thromboembolism in AF patients. Compared to warfarin, NOACs also are associated with decreased intracranial hemorrhage.100-104 Further, the results of a meta-analysis of the major NOAC trials showed that the mortality and stroke benefit derived from NOAC therapy is driven mostly by a significant decrease in hemorrhagic stroke (relative risk [RR] 0.49; 95% CI, 0.38-0.64; P = 0.0001) compared to warfarin.89
Recently, NOACs have been recommended as first-line OAC therapy in nonvalvular AF given the current evidence for their noninferiority to warfarin in AF-related stroke prevention and superior safety profile. However, there are no head-to-head randomized clinical trial data to compare the efficacy and safety of NOACs relative to each other, and the choice of therapy should be dictated by patient comorbidities, drug cost, and patient preferences.
There is a need to evaluate patients for hepatic and renal disease before initiation of NOAC therapy. Ongoing monitoring of hepatic and renal function (including creatinine clearance) is equally beneficial because changes in function might warrant discontinuation or dose adjustments. There is no strong evidence to suggest a correlation between NOAC serum drug levels and clinical outcomes, including bleeding. However, recent guidelines from the American Heart Association/American College of Cardiology (ACC) suggest the possible role for the determination of serum drug levels in patients undergoing urgent surgical procedures, chronic kidney disease patients, and in cases of suspected drug-drug interactions to inform dose modification, confirm patient adherence to OAC therapy, and to ascertain therapeutic drug levels in patients with body mass index > 35 or body weight > 120 kg.
Two antidotes now exist for both the direct thrombin inhibitor dabigatran and factor Xa inhibitors. Idarucizumab, a monoclonal antibody that binds dabigatran, was approved for urgent reversal of dabigatran anticoagulation by the U.S. Food and Drug Administration (FDA) in 2015. The RE-VERSE AD (Reversal Effects of Idarucizumab on Active Dabigatran) trial demonstrated that idarucizumab rapidly normalized hemostasis and reduced circulating levels of dabigatran in patients who had serious bleeding or required urgent procedures.105
Recently, andexanet alfa (recombinant factor Xa), was reported to reverse the anticoagulant effects of factor Xa inhibitors (rivaroxaban and apixaban) in healthy volunteers and is FDA-approved for urgent reversal of anticoagulation in patients with life-threatening bleeding.106
Table 3 shows a summary of the currently available NOACs and their contraindications.
Table 3. Current FDA-approved NOACs |
||||
|
Dabigatran |
Rivaroxaban |
Apixaban |
Edoxaban |
|
|
Peak time (hours) |
1-3 |
1-4 |
1-3 |
1-2 |
|
Protein binding |
35% |
92-95% |
87% |
55% |
|
Half-life (hours) |
12-17 |
5-13 |
9-14 |
10-14 |
|
Clearance |
80% renal |
66% hepatic; 33% renal |
25% renal |
50% renal |
|
Drug interactions |
Renal dysfunction: CrCl > 30 mL/min: 150 mg bid CrCl 15-30 mL/min: 75 mg bid Consider dose adjustment with concomitant P-gp inducer (e.g. rifampin) and inhibitor (e.g. dronedarone & ketoconazole) use |
Renal dysfunction: CrCl > 50 mL/min: 20 mg qd with evening meal CrCl 15-50 mL/min: Caution with moderate to severe hepatic impairment Avoid concomitant use with dual P-gp and strong CYP3A4 inhibitors (e.g. azole antifungals) and dual P-gp and strong CYP3A4 inducers (e.g. carbamazepine) |
Usually 5 mg bid Presence of ≥ 2 of the following: age 80 years, body weight 60 kg, or serum creatinine 1.5 mg/dL, the recommended dose is 2.5 mg bid Strong dual inhibitors of CYP3A4 and P-gp increase serum levels of apixaban: give 2.5 mg bid if not possible to avoid concomitant use Strong inducers of CYP3A4 and P-gp reduce serum levels of apixaban; avoid use |
60 mg qd if CrCl 30 mg qd if CrCl 15-50mL/min Contraindicated if CrCl > 95 mL/min |
|
Antidote |
Idarucizumab |
Andexanet alfa |
Andexanet alfa |
|
|
FDA = U.S. Food and Drug Administration; CrCl = creatinine clearance; qd = once daily; bid = twice daily |
||||
Apixaban. In the ARISTOTLE (Apixaban for Reduction in Stroke and Other Thromboembolic Events in AF) trial,103 compared to warfarin, apixaban 5 mg twice daily demonstrated a 21% reduction in stroke or thromboembolism risk, 31% reduction in major bleeding, and 11% reduction in all-cause mortality. The differences in major bleeds were driven by a higher rate of hemorrhagic stroke and intracranial bleeds in the warfarin group. The two anticoagulants demonstrated similar rates of ischemic stroke and gastrointestinal bleeding.107 The efficacy and safety of apixaban in AF patients also has been compared to aspirin in a study that showed the superiority of apixaban to aspirin in stroke prevention (55% decrease in risk compared to aspirin), while also demonstrating comparable rates of major or intracranial bleeding.66,108
Dabigatran. The RE-LY (Randomized Evaluation of Long-term Anticoagulation Therapy) study evaluated the efficacy and safety of dabigatran (150 mg twice daily and dabigatran 110 mg twice daily) compared to warfarin therapy. Dabigatran 150 mg twice daily decreased incident stroke and thromboembolism by 34% compared to warfarin, while the lower dosage (dabigatran 110 mg twice daily) showed noninferiority to warfarin for the same primary outcome (RR with dabigatran, 0.91; 95% CI, 0.74-1.11; P < 0.001 for noninferiority).102 Whereas dabigatran 150 mg twice daily showed similar rates of major bleeding events to warfarin (3.11% and 3.36% per year; P = 0.31, respectively), dabigatran 110 mg twice daily showed about 19% fewer major bleeds than warfarin (2.71% per year; P = 0.003).102,109 Compared with warfarin, both doses of dabigatran demonstrated a significant decrease in hemorrhagic stroke and intracranial hemorrhage. While the higher dosage of dabigatran showed additional benefits in terms of ischemic stroke (about 24% decrease as compared to warfarin) and vascular mortality (about 12% decrease compared to warfarin), the risk for gastrointestinal bleeding was markedly increased (about 50% compared to warfarin).
Edoxaban. Edoxaban (30 mg or 60 mg once daily) was compared to warfarin in the ENGAGE AF-TIMI 48 (Effective Anticoagulation with Factor Xa Next Generation in Atrial Fibrillation-Thrombolysis in Myocardial Infarction 48) trial. This large, randomized, prospective AF trial demonstrated that either dose of edoxaban was noninferior to warfarin in the prevention of stroke and thromboembolism (1.2% with 60 mg of edoxaban; HR = 0.79; 97.5% CI, 0.63-0.99; P < 0.001 for noninferiority and 1.6% with 30 mg of edoxaban, HR = 1.07; 97.5% CI, 0.87-1.31; P = 0.005 for noninferiority, as compared with 1.5% with warfarin).101 Edoxaban was also associated with significantly lower rates of bleeding (about 18% and 53% decrease with edoxaban 60 mg and edoxaban 30 mg respectively, compared to warfarin) and death from cardiovascular causes.101
Rivaroxaban. In the ROCKET AF (Rivaroxaban Once Daily Oral Direct Factor Xa Inhibition Compared with Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation) trial, rivaroxaban was noninferior to warfarin in the primary outcome of stroke and thromboembolism prevention (1.7% per year compared to 2.2% per year in the warfarin group [rivaroxaban group HR = 0.79; 95% CI, 0.66-0.96;
P < 0.001 for noninferiority]).100 There was a significant reduction in rates of hemorrhagic stroke and intracranial hemorrhage and an increase in rates of gastrointestinal bleeding with rivaroxaban compared to warfarin.
Nonpharmacological Thromboembolism Prevention in AF
Percutaneous Left Atrial Appendage Occlusion. There is growing interest in nonpharmacologic approaches to stroke prevention because some AF patients may not be suitable candidates for long-term anticoagulation. Recently, two randomized studies, the PROTECT AF (WATCHMAN Left Atrial Appendage System for Embolic Protection in Patients With Atrial Fibrillation) and the PREVAIL (Evaluation of the WATCHMAN LAA Closure Device in Patients With Atrial Fibrillation Versus Long Term Warfarin Therapy) evaluated the efficacy and safety of percutaneous left atrial appendage (LAA) closure with the Watchman device vs. warfarin therapy in patients without a contraindication to short-term VKA therapy. A pooled meta-analysis of the two trials showed that the Watchman device was noninferior to warfarin for the primary composite outcome of cardiovascular death, stroke, or systemic embolism (event rate 2.8% in the device group vs. 3.4% in the warfarin group;
P = 0.23). Instances of non-procedure-related and major bleeding were also fewer in the device group (1.7% vs. 3.6%; P = 0.0003).110 The primary efficacy endpoint was driven mainly by the lower rates of hemorrhagic stroke in the device group compared to the warfarin group. Higher rates of ischemic strokes attributable to periprocedural device-related thrombosis were observed in the device group.110 Periprocedural pericardial effusions were also more common in the device group.
It is important to pay attention to these studies’ inclusion and exclusion criterion. First, patients included were mostly those with a moderate risk for stroke and/or thromboembolism. Second, contraindication to long-term warfarin therapy was an exclusion criteria, and patients in the device group required 45 days of warfarin therapy and six months of dual aspirin and clopidogrel therapy followed by long-term aspirin therapy. These observations have generated questions about the applicability of this procedure to patients who have absolute contraindications to anticoagulation and those with a particularly high stroke risk. A more recent observational study from a European registry sought to answer these questions and reported a high implantation rate of 98% and 4% rate of procedure-related complications at 30 days.111
Clinical outcomes of surgical LAA occlusion for AF-related stroke prevention are less clear. Studies have yielded mixed results, with one study demonstrating no benefit in those who received prophylactic LAA occlusion in patients who underwent cardiac surgery,112 while another study demonstrated potential benefits, including lower unadjusted thromboembolism readmissions and all-cause mortality at 2.6 years of follow-up in older patients with AF who had concomitant LAA occlusion during cardiac surgery.113
Perioperative Anticoagulation and the Role of Bridging Therapy in Nonvalvular AF. Bridging therapy refers to substitution of an OAC with a parenteral anticoagulant before and/or after an invasive or surgical procedure. This is necessitated by a need to mitigate bleeding risk while minimizing the potential for periprocedural thromboembolism. Clinical practice guidelines on this subject differ, mainly because of a lack of large randomized controlled trials to guide current clinical practice.
While a comprehensive review of bridging anticoagulation is beyond the scope of this article, a summary of guiding principles is offered. With a view to promoting informed and more consistent clinical practice, the American College of Chest Physicians and American College of Cardiology developed guidelines based on the available evidence and expert consensus where evidence is currently lacking. Recently, the BRIDGE (Bridging Anticoagulation in Patients who Require Temporary Interruption of Warfarin Therapy for an Elective Invasive Procedure or Surgery) study compared clinical outcomes of bridging vs. no bridging among nonvalvular AF patients requiring perioperative interruption of warfarin therapy.114 Results from this study demonstrated no significant difference in rates of arterial thromboembolism between AF patients who received bridging anticoagulation (low molecular weight heparin) and those who did not before undergoing an elective surgical procedure. However, caution should be exercised in the interpretation of these study findings. Of note, most patients in this study had a low to moderate CHA2DS2-VASc score (mean 2.3) and patients undergoing surgical procedures with higher risks for bleeding, such as vascular surgery, were not included in the study. Therefore, these results may not be applicable to patients with higher stroke risk; that is, patients with CHA2DS2-VASc score ≥ 5. Another study demonstrated a similar thrombotic risk of 0.6% when bridging vs. no bridging was evaluated among AF patients,115 but rates of adverse cardiovascular outcomes and major bleeds were higher among those who underwent bridging.115 Therefore, decisions on bridging anticoagulation must be individualized and must weigh the risks of thromboembolism and major bleeding (incorporate both patient and procedure-related bleeding risks). Generally, patients with a high annual thromboembolism or stroke risk > 10% (CHA2DS2-VASc score 7-9), including patients with recent thromboembolism (ischemic stroke, TIA, or systemic embolism within the last three months), require bridging except in patients with a high bleeding risk in whom the decision to bridge is based on clinical judgement.116
Deferring surgery is an option for those within three months of a thromboembolic event. By the same token, clinicians should consider no bridging in AF patients with an intracranial hemorrhage within three months of planned surgery if deferring the surgery is not feasible. Bridging decisions in patients with a moderate annual risk of thromboembolism of 5-10% (CHA2DS2-VASc score 5-6) or prior embolic ischemic stroke, TIA, or systemic embolism ≥ three months should be based on patient bleed risk.116
Interruption of OAC therapy without bridging is reasonable in patients with an elevated bleeding risk. Among these patients, those with prior stroke or thromboembolism and a low bleeding risk can be bridged safely, and no bridging is required for those without stroke or thromboembolism history.116 Based on the results of the BRIDGE trial, for patients with a low risk of stroke or thromboembolism (CHA2DS2-VASc score ≤ 4), anticoagulation can be interrupted safely without bridging.114
Unfortunately, differences exist in the assessment of procedure-related bleeding risk. Several professional societies have published consensus statements to harmonize bleed risk evaluation for commonly performed invasive procedures.117-119 Surgeons should be actively engaged when making decisions on periprocedural anticoagulation, and this may be important particularly when determining the appropriate time to resume OAC.
Bridging with a parenteral anticoagulant for patients on a VKA usually is initiated when INR is < 2 and allowed to run until four to six hours before the procedure and can be restarted four to six hours post-procedure if hemostasis is achieved. VKA therapy usually is resumed 24-48 hours post-procedure and runs concurrently with the parenteral agent until a desired INR is achieved.
Counterintuitively, for some procedures, uninterrupted anticoagulation with VKA is associated with lower bleeding risk than bridging therapy. In the BRUISE CONTROL (Bridge or Continue Coumadin for Device Surgery Randomized Controlled) trial, uninterrupted OAC (with INR 2-3) with warfarin in patients undergoing implantation of pacemakers and implantable cardioverter defibrillators was associated with significantly less risk of bleeding.120
NOACs have different pharmacokinetics than VKAs, and the perioperative management of patients on NOACs is managed differently. Bridging is hardly ever required in patients taking NOACs because of the drugs’ short half-lives. Generally, NOACs can be interrupted before elective procedures but their pharmacokinetics, including peak and trough levels, half-life, and hepatic and renal clearance, must be taken into consideration. Patient-related bleeding risks and procedural bleeding risk require special attention in making decisions on periprocedural interruption of NOAC therapy.
Generally, for a patient with CrCl ≥ 80 scheduled to undergo a low bleed-risk procedure, a factor Xa inhibitor (apixaban, edoxaban, and rivaroxaban) can be held for ≥ 24 hours and safely resumed within 24 hours of the procedure if post-procedure hemostasis is achieved.116
Patients undergoing intermediate to high bleed-risk procedures require interruption of NOAC OAC for ≥ 48 hours and OAC can be restarted in 48-72 hours. With dabigatran, patients with CrCl ≥ 80 can have interruption of NOAC OAC at ≥ 24 hours for procedures with low and intermediate bleeding risk and at ≥ 48 hours for procedures with high bleeding risk.116 Patients with CrCl 50-79, 30-49, and 15-29 can have their NOAC held for ≥ 36 hours, ≥ 48 hours, and ≥ 72 hours for low bleed-risk procedures and ≥ 72 hours, ≥ 96 hours, and ≥ 120 hours for intermediate to high bleed-risk procedures, respectively. Held NOAC therapy can be resumed safely from 48-72 hours in patients undergoing intermediate to high bleed-risk procedures, provided post-procedure hemostasis is achieved. Data are less clear for patients with CrCl
< 15 in whom NOAC-specific laboratory testing to ascertain anticoagulation can help inform decisions.
Anticoagulation in Patients With Concurrent AF and Coronary Artery Disease (CAD). An estimated 5-15% of AF patients require a stent during their lifetime.121 Incident AF occurs in about 10-21% of acute coronary syndromes (ACS), and rises particularly with advancing age and MI severity.122 Concurrent AF is a poor predictor of long-term outcomes and is associated with increased stroke and mortality in patients with ACS.123,124
The co-existence of AF and CAD complicates OAC management, and patients treated for ACS may require dual antiplatelet therapy (DAPT) with aspirin and a platelet P2Y12 inhibitor in addition to a VKA or a NOAC if elevated stroke risk exists. However, concomitant OAC and antiplatelet therapy, especially triple therapy, is associated with increased risk of major bleeding.125
Evidence from a meta-analysis study revealed that the addition of a NOAC to antiplatelet therapy in patients with a recent ACS was associated with a modest decrease in cardiovascular events (all-cause mortality, MI, and stroke) but a substantial increase in bleeding events (major and non-major bleeding requiring medical attention, but not necessarily hospitalization); the risk for bleeding was higher among those on triple therapy (aspirin, clopidogrel, and NOAC). Compared with aspirin alone and dual antiplatelet therapy with aspirin and clopidogrel, the addition of a NOAC to either aspirin or DAPT reduced the composite cardiovascular endpoint (HR = 0.70; 95% CI, 0.56-0.84 and HR = 0.87; 95% CI, 0.80-0.95, respectively), but increased bleeding events (HR = 1.79; 1.54-2.09 and HR = 2.34; 2.06-2.66, respectively). A recent trial, RE-DUAL PCI,126 evaluated the safety (clinically relevant bleeding events) and efficacy (risk of thromboembolism, MI, death, and unplanned revascularization) of dual therapy in AF patients after percutaneous coronary intervention (PCI) as compared to triple therapy of warfarin and DAPT. This trial randomized patients to a group receiving dual therapy (dabigatran 110 mg or 150 mg twice daily) plus a P2Y12 inhibitor (clopidogrel or ticagrelor) and no aspirin or to a group receiving triple therapy with warfarin plus a P2Y12 inhibitor (clopidogrel or ticagrelor) and aspirin. The study demonstrated that among AF patients who underwent PCI, dual therapy with dabigatran and a P2Y12 inhibitor was associated with fewer clinically relevant bleeding events than triple therapy (warfarin, aspirin, and a P2Y12 inhibitor) and that dual therapy with dabigatran and a P2Y12 inhibitor was noninferior to triple therapy in preventing thromboembolism.
Because of this demonstrated benefit in bleeding events and a lack of evidence for harm as related to stent thrombosis among AF patients with concomitant ACS, recent guidelines recommend the use of dual antiplatelet therapy with an OAC plus a P2Y12 inhibitor without aspirin. In patients in whom the risk for stent thrombosis is a major concern, such as patients with ST-elevation-MI, triple therapy with an OAC, aspirin, and a P2Y12 inhibitor can be given for 4-6 weeks following PCI with stenting followed by a transition to dual therapy without aspirin.127
AF patients with concurrent ACS but without significant stroke or thromboembolism risk, as determined by the CHA2DS2-VASc score of 0-1, can be managed with DAPT only; those with stable CAD can be managed with OAC alone.127
Postoperative AF Management
As discussed earlier, AF is common after cardiac surgery and can occur with non-cardiac surgery. It is associated with an increased risk of death.128 Based on evidence from studies of post-cardiac surgery patients, postoperative administration of beta-blockers, particularly propranolol, is effective in the prevention of postoperative AF (16.3% incident AF in treatment group vs. 31.7% in control group).129,130 Similarly, evidence suggests that amiodarone shortens the length of hospital stay and lowers the risk of postoperative AF compared to beta-blocker therapy.129,131
Just as in patients with other forms of AF, patients with postoperative AF derive benefit from OAC. For example, a study found that OAC at discharge decreased long-term mortality in patients with postoperative AF.132 Nonetheless, there appears to be a need for more data from large RCTs to establish the long-term benefit of OAC in patients with postoperative AF.
Summary
AF is a highly prevalent and costly disease that usually complicates medical care and is associated with a high burden of morbidity and mortality. Risk reduction and/or modification of known AF risks and appropriate treatment of common medical comorbidities through lifestyle measures and medical therapy can prove helpful. Anticoagulation of AF patients with substantial stroke risk improves morbidity and mortality.
REFERENCES
A complete list of references is available online: http://bit.ly/2rFd1GZ.
This two-part series presents a review of the current evidence on atrial fibrillation (AF). The first part included the definition of AF, its classification, risk factors, comorbidities, evaluation, and acute management of newly diagnosed patients. The second part focuses on long-term management, including risk factor modification, rate and rhythm control measures, stroke risk stratification, and anticoagulation management.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
