Status Epilepticus
March 1, 2020
Reprints
AUTHORS
Guhan Rammohan, MD, FACEP, Emergency Medicine Faculty, Emergency Medicine Residency,
St. Luke’s Hospital, Bethlehem, PA
Jarrett Shugars, MD, Emergency Medicine Resident, St. Luke’s Hospital, Bethlehem, PA
PEER REVIEWER
Danuel K. Snelgrove, MD, Board Certified Neurologist, Medical University of South Carolina, Charleston
EXECUTIVE SUMMARY
• The operational definition for status epilepticus (SE) is a generalized tonic-clonic seizure lasting longer than five minutes or a complex partial seizure lasting longer than 10 minutes.
• Long-term consequences of sustained seizures, such as neuronal injury or alteration of neuronal networks, is believed to occur after 30 minutes of sustained tonic-clonic or 60 minutes of complex partial seizure activity.
• Most cases of SE are secondary to acute (within one week) insult to the brain.
• The most effective abortive treatment for SE in the prehospital setting is benzodiazepines, preferably through an intramuscular (IM) route, such a midazolam 0.2 mg/kg IM (maximum 10 mg).
• For SE refractory to benzodiazepines, intravenous anticonvulsants such as levetiracetam, fosphenytoin, or valproic acid are appropriate second-line treatments.
• Refractory SE is defined as a seizure that continues despite adequate treatment with a properly dosed benzodiazepine and a sufficient loading dose of an anticonvulsant medication, occurring in 23-48% of patients with SE.
Case Study
A 72-year-old woman presents to the emergency department (ED) with ongoing convulsions. The emergency medical services (EMS) crew reports that they were called to her home where her husband reported that the patient had the acute onset of jerking movements approximately 10 minutes before EMS arrival. EMS supplied the patient with supplemental O2 and administered 10 mg intramuscular diazepam en route with no resolution of symptoms.
Upon initial presentation to the ED, it is obvious the patient is undergoing a tonic-clonic seizure. She has been seizing for approximately 20 minutes according to EMS, and it is clear that the patient is in status epilepticus. The patient is not responsive to questioning or physical stimulus. The patient is placed on the monitor, and intravenous (IV) access is obtained. Initial vital signs are as follows: heart rate of 133 beats per minute, blood pressure
172/83 mmHg, O2 saturation 98% on 4 L nasal cannula, temperature 99.1°F rectally, point-of-care glucose is 81 mg/dL.
The patient is given an appropriate dose of lorazepam intravenously. Initial blood work is sent, including complete blood count (CBC), complete metabolic profile, magnesium level, phosphorus level, calcium level, ethanol level, acetaminophen/salicylate level, and lactate.
At this time, the patient’s husband arrives and provides further history information. He says that the patient has known stage IV breast cancer with metastasis to the spine and is currently receiving radiation, although she is not currently on active chemotherapy. The patient’s husband says that she has no known history of seizures and is not taking any antiepileptic medications. He does note that the patient has been complaining of some dull/achy generalized headaches upon awakening over the past week that seem to improve throughout the day. He denies any complaints of focal neurologic deficits or altered mental status associated with the headaches. No known history of head trauma is reported.
After initial laboratory work is sent and benzodiazepine is administered, the patient still appears to be seizing. The patient has been seizing continuously for approximately 30 minutes at this time.
Definition
Similar to other aspects of medicine, the definition of status epilepticus (SE) has evolved over time. Previously, SE was defined as a single seizure lasting more than 30 minutes or several seizures without return to baseline over a time period of 30 minutes.1 The current operational definition of SE, which is widely accepted and practiced by the healthcare community, is a seizure lasting longer than five minutes or more than one seizure without recovery to baseline between seizures. However, the most recent definition of SE was formed by the International League Against Epilepsy (ILAE): “Status epilepticus is a condition resulting either from the failure of the mechanisms responsible for seizure termination or from the initiation of mechanisms, which lead to abnormally, prolonged seizures (after time point t1). It is a condition, which can have long-term consequences (after time point t2), including neuronal death, neuronal injury, and alteration of neuronal networks, depending on the type and duration of seizures.”2
The two time points defined by the ILAE are t1, the time at which a seizure is considered to be abnormally prolonged, and t2, the time at which long-term consequences are likely to affect the patient. The reason that the ILAE uses these operational dimensions (t1/t2) is twofold. First, the time dimensions for SE vary based on the type of SE. For instance, for generalized convulsive SE, t1 is defined at five minutes and t2 is defined as 30 minutes. For complex partial SE, t1 is defined as 10 minutes and t2 at 60 minutes. The second reason that the ILAE gives variables in their definition instead of specific numbers is that research in SE is evolving. Current estimates for t1 and t2 are largely based on animal studies and clinical research. The authors state that, “these time points should be considered as the best estimates currently available.”2
Epidemiology
The incidence of SE has been reported from 7-41/100,000.3 The reason that the range is so broad is because of the varying definitions of SE, the unclear definitions of nonconvulsive status epilepticus (NCSE), and subsequent under-ascertainment of NCSE in population-based studies.4 As discussed earlier, the contemporary definition of convulsive SE is a seizure lasting more than five minutes. However, the majority of population-based studies have used a definition of a seizure lasting a minimum of 30 minutes.4 Therefore, the incidence reported in these studies could be considered a minimum level as compared to the true incidence.
There seems to be variation in the incidence of SE based on race. In several recent studies in European nations and in American Caucasians, the minimum incidence of SE is 10-20/100,000.4 However, the incidence found in an epidemiologic study in California among African Americans was substantially higher, with a reported relative risk as compared to Caucasians of 1.92.5 In the same study, relative risk of SE among Hispanics was 0.5 and among Asians was 0.4 compared to Caucasians.5 This same result was replicated in another epidemiologic study of SE in Richmond, VA, where the incidence of SE among African Americans was three times as high as in the Caucasian population.6
SE has been found to have a bimodal age distribution, typically affecting children and the elderly at higher rates.3 In a population-based study in California in the 1990s, the highest incidence was seen in children younger than 5 years of age (7.5/100,000) and the elderly (22.3/100,000).5 The incidence in the elderly is thought to be three to 10 times that of younger adults.4 It is likely the incidence of SE may rise in the future as the population continues to age.4
Etiologies
The etiology of SE can be defined within two broad categories. The majority of cases are symptomatic, with a small percentage found to be due to unknown etiology and classified as cryptogenic (or unprovoked). Symptomatic SE (or provoked SE) can be broken down further into acute symptomatic, remote symptomatic, and progressive symptomatic.
Acute symptomatic SE is when a seizure is found to be associated within one week of some insult, including brain trauma, central nervous system (CNS) infection, cerebrovascular accident, acute diffuse encephalopathy including anoxia, and metabolic insults including drug or alcohol withdrawal.7 The majority of SE cases have been found to be due to an acute symptomatic etiology.7,8 Remote symptomatic is defined as SE in the presence of a remote history (greater than one week) of CNS insult, such as stroke, head trauma, or CNS infection, thought to lead to a static lesion.7 Progressive symptomatic is when the etiology of SE is found to be due to nonstatic CNS pathology, including growing tumors and degenerative neurologic disease.7 SE is considered unprovoked if it did not meet the earlier definition of provoked SE.
In one epidemiologic study, researchers found that 63% of SE cases were acute symptomatic, 28% were remote symptomatic, and 9% were cryptogenic.8 SE occurs in patients who often have never had a seizure in the past. In one study, only 42.4% of people with SE had a history of seizures.8 In fact, SE can be the presentation of those with epilepsy.7
The causes of SE are slightly different in children as compared with adults. Again, the most common etiology is acute symptomatic, with about half of the cases found to be associated with an acute precipitant, including febrile seizures, CNS infection, electrolyte imbalance, trauma, vasculitis or vascular malformations, and toxins.9 Febrile seizures are much more common in children younger than 4 years of age. Remote symptomatic causes made up a smaller proportion of cases in children, at about 10%. Remote symptomatic causes included cerebral dysgenesis, mesial temporal sclerosis, remote infection, and remote vascular events. The remainder of cases were determined to be either cryptogenic or idiopathic.9
Classification
Historical Classification
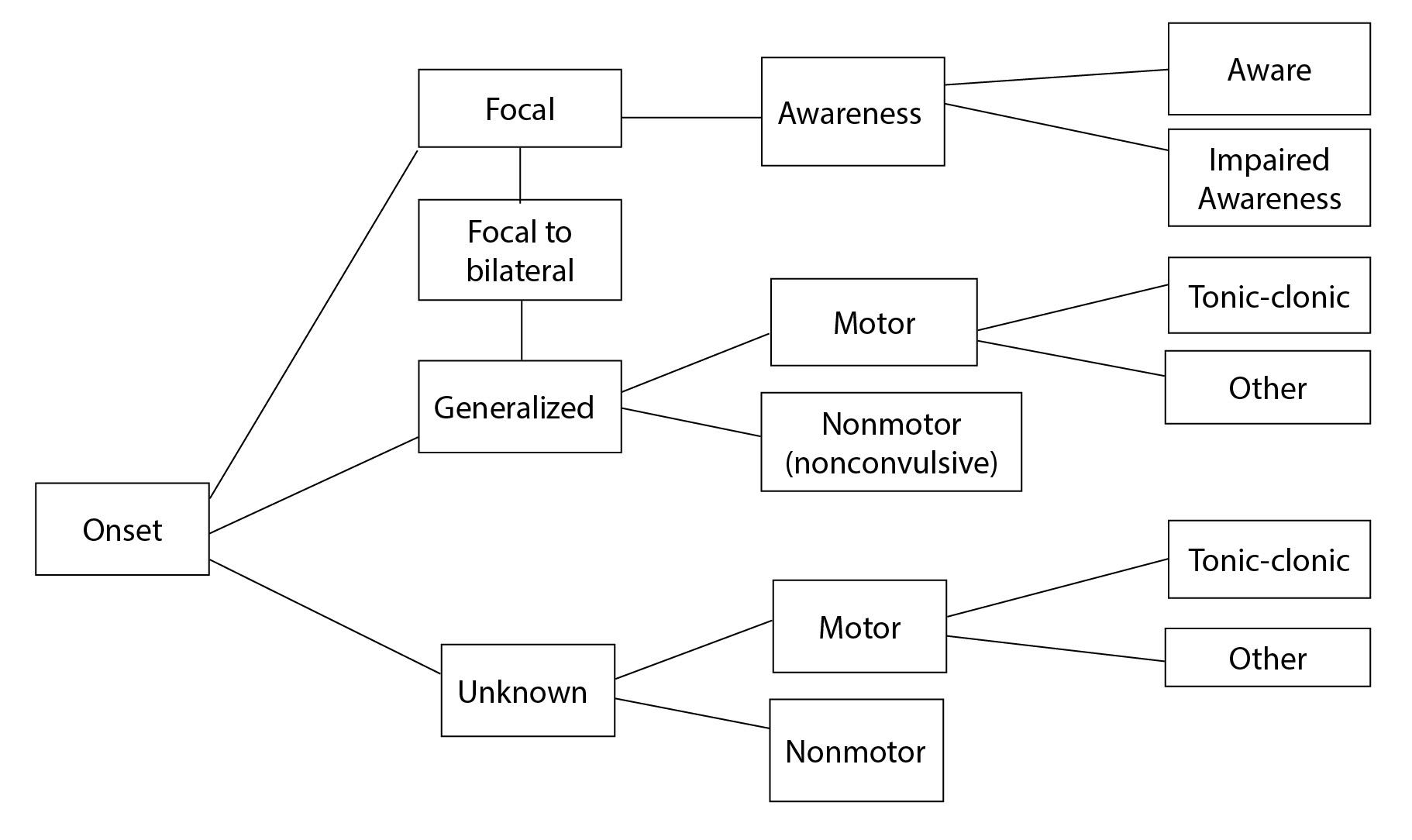
For many years, the terms “grand mal” and “petit mal” sufficed to dichotomize seizures into two basic types. Subsequently, seizures were differentiated by labeling them either “generalized” or “focal.” Focal seizures were defined as initiating in one part of the brain, whereas generalized seizures were defined as starting in bilateral cerebral hemispheres.10 Focal seizures were differentiated further based on the level of consciousness during the seizure, with simple focal seizures displaying intact consciousness and complex focal seizures involving an impaired level of consciousness by definition.10 With evolving knowledge, including improved imaging modalities, genomic technologies, and molecular biology, in 2010 the ILAE made substantial changes to previous definitions and further revised the classification in 2017. (See Figure 1.)
Figure 1. ILAE 2017 Classification of Seizure Types |
 |
|
Adapted from: Trinka E, Cock H, Hesdorffer D, et al. A definition and classification of status epilepticus — Report of the ILAE Task Force on Classification of Status Epilepticus. Epilepsia 2015;56:1515-1523. |
Structure of Classification
The new structure of classification is based on three key features:
• Location of seizure origin;
• Level of awareness throughout the seizure;
• Other features.10,11
A discussion of the basis on which seizures are classified by these three features follows.
Generalized vs. Focal. A seizure is first classified by its type of onset. The onset of a seizure can be classified as generalized, focal, focal to bilateral, or unknown onset.11 Generalized seizures are described as “originating at some point within, and rapidly engaging, bilaterally distributed networks.”11 Conversely, focal seizures are defined as those that start in an area or network of cells on one side of the brain.10 Previously, seizures that initially were focal and progressed to become generalized were described as secondary generalized. Now, these seizures are described as focal to bilateral.10 Finally, if the location of the seizure’s onset is not clear or known at the time, it is defined as unknown onset.
Awareness During the Seizure. The second feature by which a seizure can be classified is based on the patient’s awareness. By definition, generalized seizures have some type of impaired awareness/consciousness since they involve both cerebral hemispheres.11 Focal seizures can be classified further as aware, impaired awareness, or awareness unknown. Focal seizures are considered to be aware if the patient remains aware throughout the seizure, even if they lose the ability to talk or respond. The patient must fully recall the event for a seizure to be considered aware.10 In contrast, focal seizures involve impaired awareness if the patient’s awareness is impaired or affected at any time. Even if the patient has a vague idea of what happened, the seizure would still be considered to have impaired awareness. Finally, if awareness is unknown, then the seizure is considered to have unknown awareness.10
Other Features. Finally, seizures are classified by other features, including motor activity during the seizure and the presence of an aura. For instance, generalized seizures can be defined as motor or nonmotor. Typical forms of generalized motor seizures include tonic-clonic, clonic, tonic, myoclonic, myoclonic-tonic-clonic, myoclonic-atonic, atonic, and epileptic spasms.10
Focal seizures also can be defined as motor and nonmotor. Focal motor seizures display some type of movement, including twitching, jerking, or stiffening movements of a body part or automatisms (lip smacking, eye twitching, etc.).10 Focal nonmotor seizures involve some manifestation other than motor symptoms, including possible changes in sensation, emotion, thinking, or experiences.10
Briefly, a note on “nonconvulsive” seizures. Based on the classification system that has been laid out, nonconvulsive is an antiquated term to describe a seizure. However, because it has been widely accepted in the past, it is a part of much of the previous literature on SE. Since the majority of seizures involve some form of motor component, nonconvulsive seizures give the physician a diagnostic challenge. No single clinical symptom is specific for nonconvulsive SE; however, the clinician should have a heightened suspicion for nonconvulsive SE if there are certain subtle motor findings present. These can include ocular movement abnormalities (nystagmus, eye deviation), automatisms (lip smacking, twitching of the face or extremities), and negative symptoms (aphasia, mutism, catatonia).
Nonconvulsive seizures have been found to be a common cause of SE, with nonconvulsive types of SE making up about 25-50% of all SE cases, including some 1-6% of cases with absence SE and 44% with complex partial SE.4 Furthermore, most cases of SE are overt, but as the duration of SE lengthens, it can become more subtle.12 This is all to say that dramatic motor symptoms are not always seen in SE. It is the clinician’s duty to include SE as a part of any differential for altered mental status.
Workup and Diagnosis
Physical Exam/Neurologic Exam
As with all patients in extremis, a rapid physical exam should be performed to assess the cardiovascular and respiratory status of the patient. If there is evidence of a compromised airway, then a definitive airway should be sought. Providers should aim to prevent any secondary injuries from seizures by placing patients on stretchers with padding on them.
A comprehensive neurologic exam can be infeasible to obtain in a patient who is actively seizing. One aspect of the neurologic exam that is essential to ascertain is the patient’s level of consciousness because this can help differentiate the type of seizure the patient is experiencing. It also is important to characterize the type of physical movement that the patient is displaying during the seizure. Seizures can be described as tonic, tonic-clonic, or myoclonic. Tonic seizures are marked by prolonged muscle contraction.13 Clonus is defined as a rapid succession of alternating contractions and relaxations of muscles that is rhythmic in nature.13 Finally, myoclonus is defined as irregular involuntary contractions of a muscle.13 Accurately describing the type of seizure the patient experienced is helpful for those who take care of the patient down the road.
Once the patient has stopped seizing, a comprehensive neurologic exam should be completed. Important factors of the neurologic exam are the level of consciousness, orientation, cranial nerve exam, strength, sensation, and coordination, noting any asymmetric findings because these may be a clue to the underlying etiology of the seizure.
Laboratory Tests
Laboratory work for a patient with SE is undertaken to help elicit the etiology of the underlying seizure. A point-of-care glucose should be measured as soon as possible in patients with SE; hypoglycemia is a potential cause for seizures, and detecting this condition and treating it can stop seizures and prevent neuronal injury. If the seizure is found to be caused by a reversible derangement, then this information can be used to help end the seizures and prevent new ones from occurring. As discussed previously, the majority of cases of SE are acute symptomatic and include cases of metabolic encephalopathy, CNS infection, cerebrovascular accident, or alcohol or drug withdrawal.7 The laboratory workup should include electrolytes (including magnesium, phosphorus, and calcium), glucose, liver function tests (LFT), CBC with differential, toxicology, and antiepileptic drug levels. If there is any suspicion for encephalitis or meningitis, then a lumbar puncture and cerebrospinal fluid studies should be performed.
Electroencephalography
Electroencephalography (EEG) is used as a measure of the brain’s electrical activity. It can be used at the bedside and recorded continuously to measure alterations in brain structure and function. EEG can be used to detect subclinical neurologic impairment, identify locations of dysfunction, and indicate the presence or absence of continuing epileptic activity.14
The initial diagnosis of SE often is a clinical one made by the stereotypical movements detailed in the physical exam section. EEG has little role in the acute setting of generalized tonic-clonic SE, as it will be impossible to read secondary to the artifact created by the physical motion of the patient.
EEG can be useful for a number of reasons with regard to non-tonic-clonic presentations of SE. First, it can be used to detect nonconvulsive seizures/SE. It is a common occurrence that patients who are treated for generalized convulsive SE display a persistently abnormal mental status. In these patients, it is recommended that the patient be placed on continuous EEG monitoring to determine if there is ongoing nonconvulsive SE.15 One study attempted to determine the etiology of persistently altered mental status following an episode of generalized convulsive SE. The researchers found that of these patients, 48% were found to have had nonconvulsive seizures and 14% were found to have had nonconvulsive SE.16 It is recommended to initiate continuous EEG monitoring as soon as possible and for at least 24 hours for patients in whom nonconvulsive seizures are suspected.15
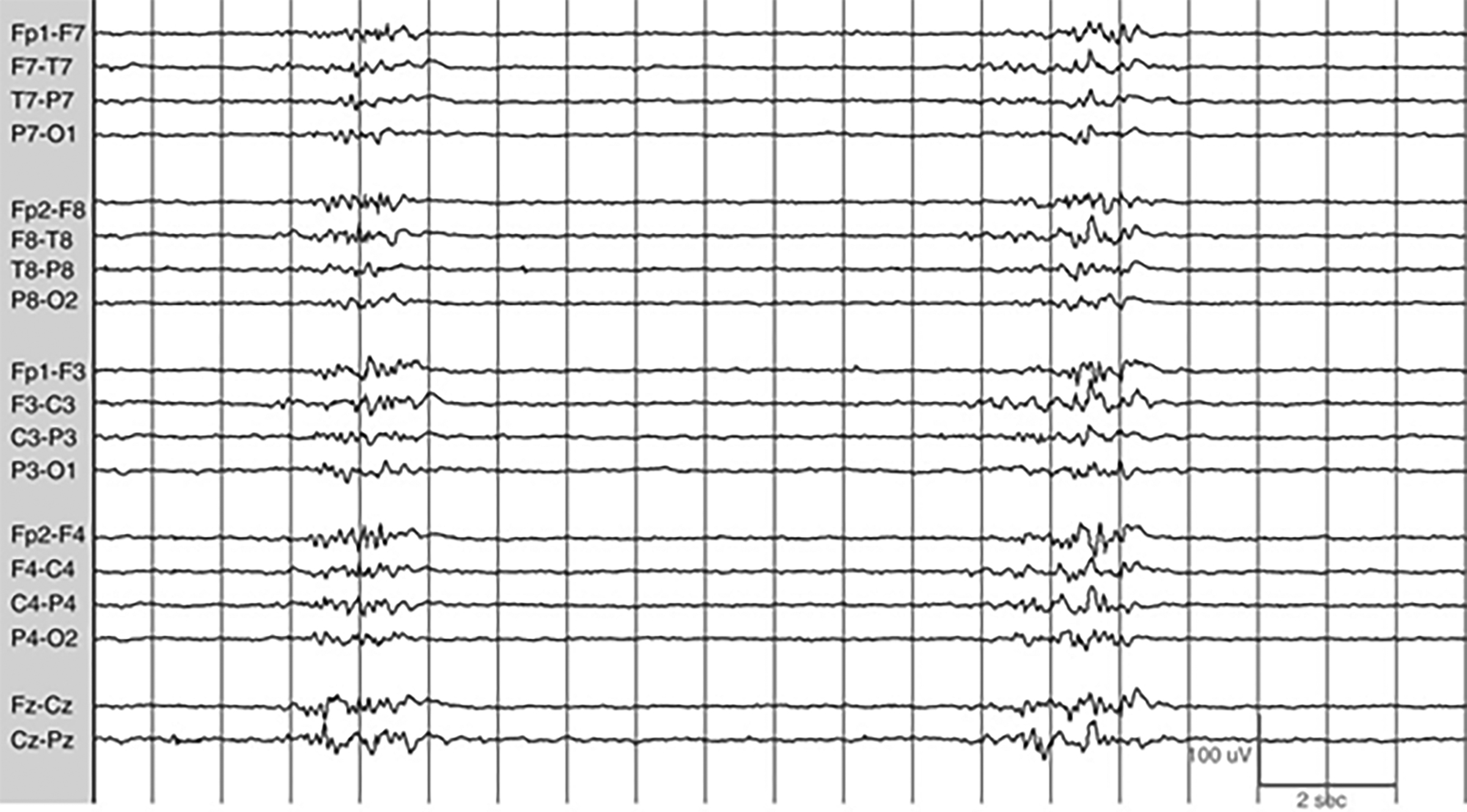
The second purpose for continuous EEG monitoring in patients with SE is to assess the efficacy of therapy.15 For patients with refractory SE, continuous EEG monitoring should be used in concordance with continuous IV infusions (midazolam, propofol, pentobarbital) for seizure suppression, burst suppression, or complete EEG suppression. (See Figure 2.) After the use of continuous IV infusions of antiepileptic drugs, seizures often will be subclinical, and EEG is needed to determine their presence.17
Figure 2. Example of Burst Suppression EEG |
 |
|
Reprinted from: Marcuse LV, Fields MC, Yoo J. Rowan’s Primer of EEG. 2nd ed. Elsevier; 2016: fig. 6-6, with permission from Elsevier. |
Finally, EEG can be used to determine the depth of sedation in patients with IV sedation or pharmacologically induced coma.15 Using these drugs can make it difficult to determine the level of sedation on clinical grounds alone. EEG can help differentiate seizure suppression, burst suppression, or complete suppression.
Of note, during focal seizures without impaired awareness, the EEG often can appear as normal. During these seizures, patients were conversant and recalled ictal events. In one study, an epileptologist was able to identify epileptic activity on the EEG in only 33% of motor focal seizures and in only 15% of nonmotor focal seizures.18
Neuroimaging
Similar to the laboratory workup obtained on a patient in SE, imaging can be obtained to better delineate the etiology of seizure. Imaging studies are particularly adept at finding structural lesions within the brain that cause seizures.
In acute situations, computed tomography (CT) imaging is the modality of choice secondary to its ready availability and quick turnaround times.19 CT can accurately detect intracranial hemorrhage/infarcts, gross malformations, lesions with underlying calcification, and large tumors.20 Although CT can be useful as an initial screening imaging modality, it will miss up to 50% of more discrete lesions, including small tumors and malformations.20
In the non-acute setting, magnetic resonance imaging (MRI) is the imaging modality of choice for patients who present with seizures. MRI has both better sensitivity and specificity to detect small lesions and abnormalities of the cerebral cortex.19 Imaging should include T1 and T2 weighted images at a minimum, although gadolinium contrast enhanced images are not typically necessary other than for specific cases in which initial noncontrast MRI was nondiagnostic.19 Specific indications for MRI in the setting of SE include focal neurological deficit, difficulty in control of seizures with first-line agents, change of pattern in chronic seizures, and evidence of partial onset seizure on history or EEG.20
Treatment
Rapid Assessment
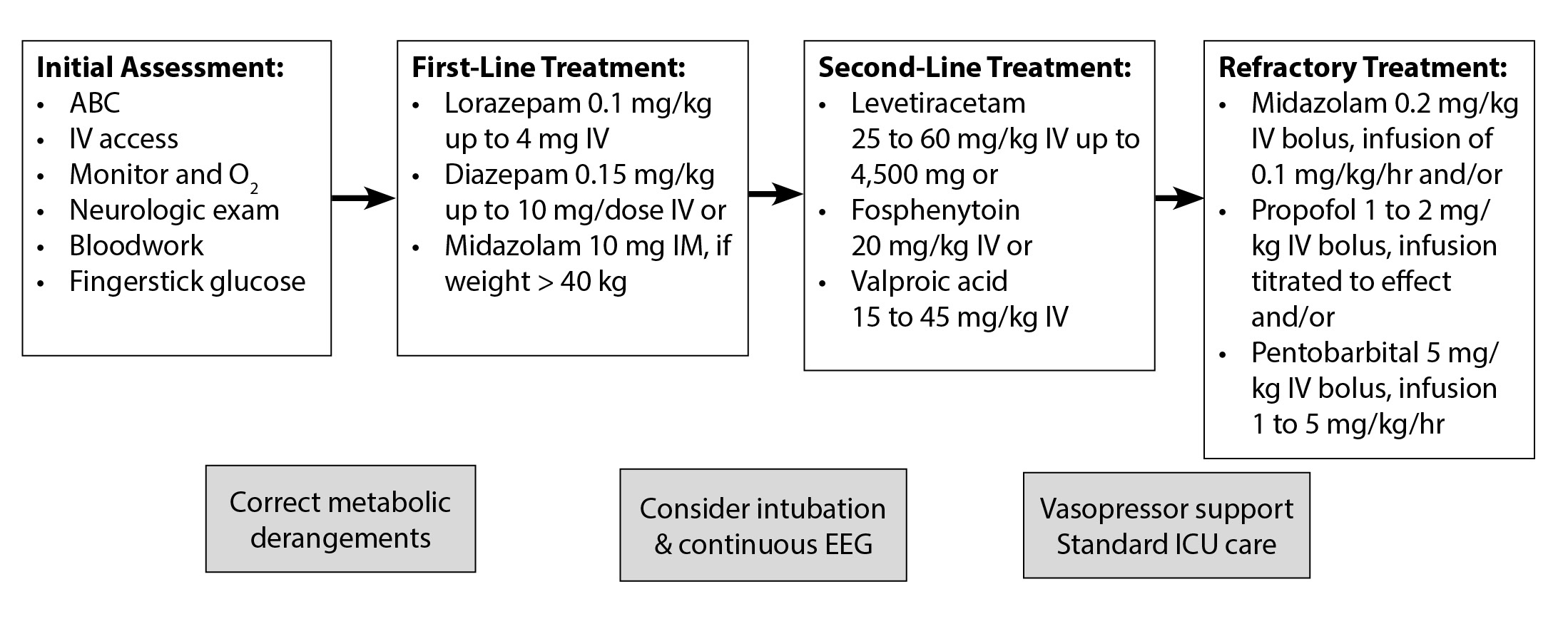
As with all patients in the emergent setting, there must be a rapid assessment of the patient’s airway, breathing, and circulation. Treatment of generalized tonic-clonic SE should begin with basic life support measures and monitoring.12 (See Figure 3.) If a patient is currently seizing, a cautious inspection of the airway should be undertaken to be sure that the patient is not at risk for aspiration.
Figure 3. Treatment |
 |
|
ABC = airway, breathing, circulation; IV = intravenous; IM = intramuscular; EEG = electroencephalogram; ICU = intensive care unit Adapted from: Up To Date |
Simultaneously, with the help of others, the next goal should be to place the patient on oxygen, a cardiac monitor, a pulse oximeter, and end-tidal capnography. Establishment of IV access is of utmost importance. The initial laboratory test to be drawn should be a point-of-care blood glucose. Other lab tests, including basic metabolic profile, LFTs, calcium, phosphorus, magnesium, CBC, anticonvulsant levels, pregnancy test, and toxicology studies, can be drawn.
If there are family members or prehospital personnel, perform a focused history, including any known history of epilepsy/SE, recent illnesses, recent trauma, and medication compliance that could further direct treatment.
Prehospital Care
The prehospital care of SE is of utmost importance since many cases can be aborted prior to arrival in the ED. Benzodiazepines are the initial treatment of out-of-hospital SE/seizures. In a double-blinded randomized trial between IV lorazepam, IV diazepam, and placebo, researchers found that there was a significant difference in termination of SE between the benzodiazepines and the placebo (59.1% vs. 42.6% vs. 21.1%, respectively). In this study, treatment with benzodiazepines reduced the rates of respiratory or circulatory complications (10.6%, 10.3%, and 22.5%, respectively).21 The odds ratio for the termination of SE in the lorazepam group vs. the placebo group was 4.8 (95% confidence interval [CI], 1.8-13.).21 Benzodiazepines are first-line out-of-hospital therapy because of their effectiveness and the low rates of complications.
Of note, there has been research to indicate that intramuscular (IM) midazolam can be even more effective than IV lorazepam. In a double-blind, randomized, noninferiority trial, investigators found that IM midazolam was not only noninferior, but superior at SE cessation on ED arrival (73.4% vs. 63.4%) with similar rates of endotracheal intubation and repeat seizure activity.22 It is thought that the time it takes to get an IV is likely the reason why the IM midazolam is more effective.
Finally, there has been evidence that adding an anticonvulsant (levetiracetam) to a benzodiazepine (clonazepam) was no more effective than the benzodiazepine alone.23
Based on the current evidence, the most effective abortive treatment for SE in the prehospital setting is benzodiazepines, preferably through an IM route.
Initial Therapy
Benzodiazepines. Benzodiazepines are first-line treatment in the cessation of SE. They work quickly and are effective. In the late 1990s, a five-year, prospective, double-blind, randomized trial compared four treatments for generalized convulsive SE. The four treatments compared were IV lorazepam, IV phenytoin, IV diazepam followed by phenytoin, and IV phenobarbital. IV lorazepam was found to be the most effective in terminating SE, with a success rate of 64.9%, compared to diazepam/phenytoin at 55.8%, phenobarbital at 58.2%, and phenytoin alone at 43.6%. The only significant difference was found between lorazepam and phenytoin alone.24
Therapy with specific benzodiazepines:
• Lorazepam: Lorazepam is a first-line medication in the treatment of SE. The generally accepted dosage is 0.1 mg/kg IV.24 In children, lorazepam also has been found to be effective in abortion of convulsive SE.25
• Diazepam: Diazepam also is a first-line treatment of SE. It has proven to be advantageous in the prehospital setting secondary to its IM preparations.21 Diazepam is successful in seizure termination in 50-80% of patients.24 The generally accepted dosage of diazepam is 0.15 mg/kg, with maximum 10 mg per dose.24 Diazepam rectal gel also has been established as an appropriate treatment in adults with epilepsy for breakthrough seizures with good efficacy and tolerability.26
• Midazolam: Midazolam is another effective treatment in the termination of SE. It can be given IM, buccally, and intranasally with a dose of 0.2 mg/kg, with maximum 10 mg/dose.
Second-Line Therapy
Phenytoin/Fosphenytoin. Phenytoin has been used to treat seizures for the past 50 years and it has been shown to be effective. The dosage of phenytoin is 20 mg/kg, and
15 mg/kg in the elderly (> 65 years). However, there are drawbacks to phenytoin that often make its use cumbersome. It has a slow rate of infusion (max 50 mg/min) and delayed onset of action. It has been found that the overall success rate with phenytoin to stop seizures has ranged from 44% in a randomized, controlled study to 90% in the uncontrolled studies.27 Side effects of phenytoin include hypotension and cardiac arrhythmias.27
Fosphenytoin is a pro-drug that is hydrolyzed by serum phosphatases to phenytoin. The advantages of fosphenytoin include the rapid rate of administration as compared to phenytoin (150 mg/min) and the decreased chance of local irritation at the site of the infusion. However, it is thought that it takes about 15 minutes to be converted from fosphenytoin to phenytoin in the serum, so there is a delayed onset of action similar to phenytoin.
Levetiracetam. Levetiracetam is a well-tolerated antiepileptic drug with a wide therapeutic range and relatively minor side effects. It has been proven to be effective as a second-line agent to control SE, with reported control rates from 44-94%.28 There are differing opinions on the dosage of levetiracetam for SE, ranging from 25 mg/kg up to 60 mg/kg, with a maximum of 4,500 mg.29,30
Adverse events when using levetiracetam to treat SE occurred in 7.1% of cases, and most adverse events were transient and minor.31 The most commonly reported side effects included mild sedation, dizziness, headache, nausea, and transient thrombocytopenia.31
Valproic Acid. Valproic acid has a broad range of effectiveness against all seizure types and a well-understood side effect profile. The onset of action is typically within minutes, with maximum plasma concentrations reached in that time.27 Valproic acid is primarily metabolized by the liver via glucuronidation and beta-oxidation.27
In a large systematic review on valproic acid, investigators found that it was effective in terminating SE 70.9% of the time.31 The most commonly reported effective doses were between 15 mg/kg and 45 mg/kg in a bolus given at a rate of 6 mg/kg/min, followed by 1 to 3 mg/kg/hour infusion.31
Adverse events generally are seen in less than 10% of cases, with the most common reactions including dizziness, thrombocytopenia, and mild hypotension independent of rate of administration.31 In rare cases, acute encephalopathy can occur, which most likely is related to hyperammonemia and/or hepatic metabolism abnormalities.27
Phenobarbital. Phenobarbital has been found to be effective in seizure cessation. One study found that using phenobarbital as compared to a benzodiazepine plus phenytoin led to shorter seizures, quicker onset of action, and similar rates of adverse events.32 However, the adverse events associated with phenobarbital, especially in conjunction with benzodiazepines, include hypotension, respiratory depression, and sedation, which are significant and somewhat limit its clinical utility initially in SE treatment.27 The typical recommended loading dose is 10 mg/kg with a maximum of 700 mg infused at a rate of 100 mg/min.27 Patients undergoing phenobarbital loading should be under continuous cardiovascular/respiratory observation.
Second-Line Treatment Summary
Benzodiazepines should be the first-line treatment for any patient who presents with seizure or SE. There has been some debate regarding which medications are most effective in the second line. One recent meta-analysis published in 2013 evaluated five different treatment options in benzodiazepine-resistant SE. Interventions included IV lacosamide, levetiracetam, phenobarbital, phenytoin, and valproate. Efficacy of levetiracetam was 68.5%, phenobarbital was 73.6%, phenytoin was 50.2%, and valproate was 75.7% (95% CI, 63.7-84.8%).33 Lacosamide studies were excluded from the meta-analysis because of insufficient data.28 The authors of this study concluded that levetiracetam, phenobarbital, and valproate all were appropriate second-line agents.33
Recently, results from the long-awaited Established Status Epilepticus Treatment Trial were released. In patients who had benzodiazepine-refractory SE, the investigators found that fosphenytoin, valproate, and levetiracetam each achieved seizure cessation within one hour in approximately 50% of the patients.34 Seizure recurrence was observed in about 10% of patients in all groups.34
In the acute setting, levetiracetam, valproate, and fosphenytoin all seem to offer the best chance of seizure cessation with the best side effect profile.
Refractory Status Epilepticus
Definition
Refractory SE is defined as a seizure that continues despite adequate treatment with a properly dosed benzodiazepine and a sufficient loading dose of an anticonvulsant medication.35 Although some researchers have defined the minimum length of a seizure to be considered refractory SE (60 minutes), it is generally held that the length of the seizure is not a parameter of refractory SE. Refractory SE occurs in 23-48% of patients who present with SE.35
Monitoring and Intensive Care Unit
Because of the high mortality of patients with refractory SE and the necessity for intubation, patients with refractory SE require intensive care unit (ICU) level care. EEG is an indispensable tool to determine if seizures are being treated appropriately. EEG can guide the use of antiseizure medications and to determine the level of neurologic suppression achieved. There has been some debate over the appropriate depth of neurologic suppression that is appropriate in refractory SE. There are two thoughts on this, the first being to achieve a burst-suppression pattern on EEG and the second being an achievement of a “flat” EEG with complete suppression of background activity.36 In one study, researchers found that those who were maintained at the greatest depth of sedation with a flat EEG remained seizure free in 17 of 20 cases, while only six of 12 remained seizure free when titrated to burst suppression.36 While these data are by no means conclusive, it does support a deeper neurologic sedation on EEG.
Drugs and Management
The three drugs that are used commonly in the setting of refractory SE include midazolam, propofol, and pentobarbital. There is some debate regarding which of these agents should be preferred over the others. One systematic review comparing the three agents for use in refractory SE found that there was no significant difference in mortality between those receiving the three drugs.17 Pentobarbital was associated with a lower frequency of short-term treatment failure and breakthrough seizures, although there also was a higher frequency of hypotension.17 Overall mortality was found to be 48% in this study.17 Based on the current evidence, all three agents can be used in refractory SE.
Midazolam can be given as a bolus followed by an infusion for rapid control of refractory SE. The typical dosage is an initial bolus of 0.2 mg/kg given at a rate of 2 mg/minute followed by an infusion of 1 mcg/kg/min that can be titrated up to seizure freedom.37
Propofol also is given as a bolus followed by an infusion. The typical dosage is 1 mg/kg bolus given over five minutes followed by an infusion rate of 2 to 4 mg/kg/hour. The infusion rate can be titrated to clinical seizure cessation and EEG findings up to 15 mg/kg/hour.38
Finally, pentobarbital can be used in refractory SE. The typical dosage of pentobarbital is a bolus of 10 to 30 mg/kg followed by an infusion of 50 mg/min until there is no EEG evidence of seizures. This initial treatment is followed by an infusion of 1 to 10 mg/kg/hour for 24 hours and then subsequently tapered over the next 24 hours.38
Complications
Disease-Related Complications
Acute complications of SE are a function of the physiologic strain that seizing puts on the human body. The primary acute complication that can be fatal in patients is cardiac arrhythmias.39 One study found that 58% of patients developed potentially fatal arrhythmias during SE.40 Fever is a common initial complication in generalized convulsive SE, occurring in 80% of patients.39 Fever is thought to develop secondary to the intense and ongoing muscle contractions associated with generalized convulsive SE and may lead to neuronal cell death.41 A second effect of the extreme muscle contractions of generalized tonic-clonic SE is lactic acidosis as the patient’s skeletal muscle transitions to anaerobic metabolism.39
The chronic complications attributed to SE are difficult to differentiate from the underlying etiology of SE. Epilepsy is a serious complication that can develop after SE. It has been found that 40% of patients will develop epilepsy after SE, as compared to 10% of patients who experience a single symptomatic seizure.42 Specifically, temporal lobe epilepsy often is seen after SE.39 Chronic encephalopathy also can be seen in 6-15% of cases following SE.39
Treatment-Related Complications
Benzodiazepines are used commonly in EDs throughout the country for a myriad of indications. Typical adverse effects that benzodiazepines can cause in the acute setting include respiratory depression, hypotension, and excessive salivation.43
Levetiracetam is a well-tolerated second-line antiepileptic drug with a wide therapeutic range. The most common side effects in the acute setting are mild sedation, dizziness, headache, nausea, and transient thrombocytopenia.31 Phenytoin and fosphenytoin have serious adverse events to consider, including cardiac depression, cardiac arrhythmias, hypotension, and paresthesias.43 Valproic acid adverse effects include dizziness, thrombocytopenia, and mild hypotension,31 and, in rare cases, acute encephalopathy.27
Agents used for refractory SE including midazolam, propofol, and pentobarbital have similar adverse effects, such as hypotension and respiratory depression.43 Patients typically will be intubated at this point. Of note, propofol also can cause propofol infusion syndrome characterized by severe metabolic acidosis, hyperlipidemia, refractory bradycardia, rhabdomyolysis, and cardiovascular/renal collapse.44 This is thought to occur is less than 1% of patients on propofol infusions.
Outcomes and Prognosis
SE is a condition with strikingly high mortality and poor outcomes. Short-term, in-hospital mortality has been reported from 7.6-22%3 in non-refractory SE, whereas mortality rates as high as 38% have been reported in refractory SE.45
Unsurprisingly, the age of the patient who presents with SE contributes to mortality rates.6 It has been found that those older than 65 years of age are more likely to die as a result of SE compared to those 20-64 years of age, while those younger than 20 years have the lowest mortality rate.4
The etiology of the seizure that leads to SE also has a strong bearing on outcomes for patients. Hypoxia, stroke, CNS infections, and metabolic disorders tend to be associated with the highest case fatalities of up to 80%, whereas low antiepileptic drug levels, fever, and alcohol‐related and
traumatic‐related etiologies were associated with lower mortalities.4 In one study, hypoxic injuries and brain tumors were independently associated with death.45 It should be noted that in many cases, the patients die not because of SE, but because of the underlying etiology that initially caused the seizure. In one study, 89% of deaths were attributed to the underlying etiology as opposed to SE itself.46
Unfortunately, even if patients survive to hospital discharge, they are still at risk for increased mortality. In one study, at 10 years, cumulative mortality among 30-day survivors was 43%. The standardized mortality ratio at 10 years was 2.8.47 It is unclear if the increased rate of mortality in this study is secondary to a history of SE or if it is caused by the underlying etiology of the seizure. However, there is no denying that SE is a very serious medical condition that has a high mortality in the short term and also significantly decreases the life expectancy of those with the condition.
Case Study Conclusion
The patient is loaded with the appropriate dose of levetiracetam. At this time, the initial blood work returns. The CBC is unremarkable. The comprehensive metabolic panel shows a metabolic acidosis, but electrolytes including magnesium, phosphorus, and calcium are otherwise within normal limits. The blood glucose is 77 mg/dL. The lactate is elevated at 4.3 mg/dL.
The patient still is seizing after levetiracetam administration. The decision is made to intubate the patient. The patient is sedated with propofol and paralyzed with succinylcholine. The intubation goes well, and the patient is sedated with propofol. She is transferred to the ICU and is immediately placed on EEG monitoring. The propofol infusion is titrated up to the point of burst suppression on EEG. The patient is kept on a propofol drip for 48 hours, at which time she is weaned off. The patient begins to have epileptiform activity on EEG during the propofol wean. The patient then is transitioned to a pentobarbital drip. However, the patient continues to have epileptiform activity after pentobarbital is weaned. The decision is made to provide the patient comfort care, and the patient dies several days later.
REFERENCES
- [No authors listed]. Guidelines for epidemiologic studies on epilepsy. Commission on Epidemiology and Prognosis, International League Against Epilepsy. Epilepsia 1993;34:592-596.
- Trinka E, Cock H, Hesdorffer D, et al. A definition and classification of status epilepticus — Report of the ILAE Task Force on Classification of Status Epilepticus. Epilepsia 2015;56:1515-1523.
- Chin RF, Neville BG, Scott RC. A systematic review of the epidemiology of status epilepticus. Eur J Neurol 2004;11:800-810.
- Rosenow F, Hamer HM, Knake S. The epidemiology of convulsive and nonconvulsive status epilepticus. Epilepsia 2007;48 (Suppl 8):82-84.
- Wu YW, Shek DW, Garcia PA, et al. Incidence and mortality of generalized convulsive status epilepticus in California. Neurology 2002;58:1070-1076.
- DeLorenzo RJ, Pellock JM, Towne AR, Boggs JG. Epidemiology of status epilepticus. J Clin Neurophysiol 1995;12:316-325.
- Hesdorffer DC, Logroscino G, Cascino G, et al. Incidence of status epilepticus in Rochester, Minnesota, 1965-1984. Neurology 1998;50:735-741.
- Coeytaux A, Jallon P, Galobardes B, Morabia A. Incidence of status epilepticus in French-speaking Switzerland: (EPISTAR). Neurology 2000;55:693-697.
- Singh RK, Stephens S, Berl MM, et al. Prospective study of new-onset seizures presenting as status epilepticus in childhood. Neurology 2010;74:636-642.
- Fisher RS, Shafer PO, D’Souza C. 2017 revised classification of seizures. Epilepsy Foundation. December 2016. Available at: www.epilepsy.com/article/2016/12/2017-revised-classification-seizures. Accessed Feb. 17, 2020.
- Berg AT, Millichap JJ. The 2010 revised classification of seizures and epilepsy. Continuum (Minneap Minn.) 2013;19:571-597.
- Treiman DM. Treatment of convulsive status epilepticus. Int Rev Neurobiol 2007;81:273-285.
- “Tonic,” “clonus,” “myoclonus.” Dictionary by Merriam-Webster. Merriam-Webster; 2020. Available at: www.merriam-webster.com/.
- Nuwer MR. Electroencephalograms and evoked potentials. Monitoring cerebral function in the neurosurgical intensive care unit. Neurosurg Clin N Am 1994;5:647-659.
- Herman ST, Abend NS, Bleck TP, et al; Critical Care Continuous EEG Task Force of the American Clinical Neurophysiology Society. Consensus statement on continuous EEG in critically ill adults and children, part I: Indications. J Clin Neurophysiol 2105;32:87-95.
- DeLorenzo RJ, Waterhouse EJ, Towne AR, et al. Persistent nonconvulsive status epilepticus after the control of convulsive status epilepticus. Epilepsia 1998;39:833-840.
- Claassen J, Hirsch LJ, Emerson RG, Mayer SA. Treatment of refractory status epilepticus with pentobarbital, propofol, or midazolam: A systematic review. Epilepsia 2002;43:146-153.
- Devinsky O, Kelley K, Porter RJ, Theodore WH. Clinical and electroencephalographic features of simple partial seizures. Neurology 1988;38:1347-1352.
- Commission on Neuroimaging of the International League Against Epilepsy. ILAE Commission Report. Recommendations for neuroimaging of patients with epilepsy. Epilepsia 1997;38:1255-1256.
- Roy T, Pandit A. Neuroimaging in epilepsy. Ann Indian Acad Neurol 2011;14:78-80.
- Alldredge BK, Gelb AM, Isaacs SM, et al. A comparison of lorazepam, diazepam, and placebo for the treatment of out-of-hospital status epilepticus. N Engl J Med 2001;345:631-637.
- Silbergleit R, Durkalski V, Lowenstein D, et al. Intramuscular versus intravenous therapy for prehospital status epilepticus. N Engl J Med 2012;366:591-600.
- Navarro V, Dagron C, Elie C, et al. Prehospital treatment with levetiracetam plus clonazepam or placebo plus clonazepam in status epilepticus (SAMUKeppra): A randomised, double-blind, phase 3 trial. Lancet Neurol 2016;15:47-55.
- Treiman DM, Meyers PD, Walton NY, et al. A comparison of four treatments for generalized convulsive status epilepticus. Veterans Affairs Status Epilepticus Cooperative Study Group. N Engl J Med 1998;339:792-798.
- Sreenath TG, Gupta P, Sharma KK, Krishnamurthy S. Lorazepam versus diazepam-phenytoin combination in the treatment of convulsive status epilepticus in children: A randomized controlled trial. Eur J Paediatr Neurol 2010;14:162-168.
- Fakhoury T, Chumley A, Bensalem-Owen M. Effectiveness of diazepam rectal gel in adults with acute repetitive seizures and prolonged seizures: A single-center experience. Epilepsy Behav 2007;11:357-360.
- Trinka E, HÖfler J, Leitinger M, Brigo F. Pharmacotherapy for status epilepticus. Drugs 2015;75:1499-1521.
- Zelano J, Kumlien E. Levetiracetam as alternative stage two antiepileptic drug in status epilepticus: A systematic review. Seizure 2012;21:233-236.
- Glauser T, Shinnar S, Gloss D, et al. Evidence-based guideline: Treatment of convulsive status epilepticus in children and adults: Report of the Guideline Committee of the American Epilepsy Society. Epilepsy Curr 2016;16:48-61.
- Mundlamuri RC, Sinha S, Subbakrishna DK, et al. Management of generalised convulsive status epilepticus (SE): A prospective randomised controlled study of combined treatment with intravenous lorazepam with either phenytoin, sodium valproate or levetiracetam — Pilot study. Epilepsy Res 2015;114:52-58.
- Trinka E, Dobesberger J. New treatment options in status epilepticus: A critical review on intravenous levetiracetam. Ther Adv Neurol Disord 2009;2:79-91.
- Shaner DM, McCurdy SA, Herring MO, Gabor AJ. Treatment of status epilepticus: A prospective comparison of diazepam and phenytoin versus phenobarbital and optional phenytoin. Neurology 1988;38:202-207.
- Yasiry Z, Shorvon SD. The relative effectiveness of five antiepileptic drugs in treatment of benzodiazepine-resistant convulsive status epilepticus: A meta-analysis of published studies. Seizure 2014;23:167-174.
- Kapur J, Elm J, Chamberlain JM, et al. Randomized trial of three anticonvulsant medications for status epilepticus. N Engl J Med 2019;381:2103-2113.
- Mayer SA, Claassen J, Lokin J, et al. Refractory status epilepticus: Frequency, risk factors, and impact on outcome. Arch Neurol 2002;59:205-210.
- Krishnamurthy KB, Drislane FW. Depth of EEG suppression and outcome in barbiturate anesthetic treatment for refractory status epilepticus. Epilepsia 1999;40:759-762.
- Ulvi H, Yoldas T, Müngen B, Yigiter R. Continuous infusion of midazolam in the yreatment of refractory generalized convulsive status epilepticus. Neurol Sci 2002;23:177-182.
- Stecker MM, Kramer TH, Raps EC, et al. Treatment of refractory status epilepticus with propofol: Clinical and pharmacokinetic findings. Epilepsia 1998;39:18-26.
- Fountain NB. Status epilepticus: Risk factors and complications. Epilepsia 2000;41(Suppl 2):S23-S30.
- Boggs JG, Painter JA, DeLorenzo RJ. Analysis of electrocardiographic changes in status epilepticus. Epilepsy Res 1993;14:87-94.
- Blennow G, Brierley JB, Meldrum BS, Siesjo BK. Epileptic brain damage: The role of systemic factors that modify cerebral energy metabolism. Brain 1978;101:687-700.
- Hesdorffer DC, Logroscino G, Cascino G, et al. Risk of unprovoked seizure after acute symptomatic seizure: Effect of status epilepticus. Ann Neurol 1998;44:908-912.
- Sirven JI, Waterhouse E. Management of status epilepticus. Am Fam Physician 2003;68:469-476.
- Kam PC, Cardone D. Propofol infusion syndrome. Anaesthesia 2007;62:690-701.
- Sutter R, Marsch S, Fuhr P, Rüegg S. Mortality and recovery from refractory status epilepticus in the intensive care unit: A 7-year observational study. Epilepsia 2013;54:502-511.
- Shorvon S. Prognosis and outcome of status epilepticus. In: Shorvon S, ed. Status Epilepticus: Its Clinical Features and Treatment in Children and Adults. Cambridge University Press; 1999:293.
- Logroscino G, Hesdorffer DC, Cascino GD, et al. Long-term mortality after a first episode of status epilepticus. Neurology 2002;58:537-541.
Status epilepticus is a serious medical condition that is defined as a seizure lasting longer than five minutes or more than one seizure without recovery to baseline between seizures.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
