Pain Management in the Emergency Department: Opioids and Alternative Pain Management Therapies
April 15, 2020
Reprints
AUTHORS
Catherine A. Marco, MD, FACEP, Professor of Emergency Medicine, Department of Emergency Medicine, Wright State University Boonshoft School of Medicine, Dayton, OH
Allison D. Ensign, MSN, APRN, FNP-C, Assistant Professor, Department of Nursing, Utah Valley University, Orem, UT
Jennifer M. Oakes, DNAP, MSNA, CRNA, Associate Program Director for Nurse Anesthesia, Texas Christian University, Fort Worth, TX
PEER REVIEWER
Steven M. Winograd, MD, FACEP, Attending Physician, Mt. Sinai Queens Hospital Center, Assistant Clinical Professor of Emergency Medicine, Mt. Sinai Medical School, Jamaica Queens, NY
EXECUTIVE SUMMARY
• Opioids are effective therapy for acute pain, but continuation on emergency department discharge is associated with the risk for tolerance and misuse.
• Acetaminophen and nonsteroidal anti-inflammatory drugs have a synergistic effect for treating acute musculoskeletal pain.
• Trigger point injections of lidocaine 1% are effective for acute headache and musculoskeletal pain.
• Sub-dissociative doses of ketamine can be effective for acute pain.
• Regional nerve blocks are useful for acute pain, thereby avoiding the mental status effects and respiratory depression sometimes seen with opioids.
• Topical analgesics, such as a lidocaine patch, are useful for musculoskeletal pain.
In my humble opinion, intravenous opioids remain the gold standard for emergency department (ED) management of acute severe pain. They have a fast onset, are reliable, and can be titrated to effect. But opioids have acute side effects, such as mental status effects or respiratory depression, that may be harmful to some patients. And, as we have seen, initiation of oral opioids for pain, whether in the outpatient clinic or on ED discharge, is associated with issues of tolerance and abuse. Thus, the concept that other agents can be used instead of (“opioid alternatives”) or in addition to (“opioid sparing”) opioids has evolved. Successful pain management in the ED should incorporate these alternatives as discussed in this issue.
— J. Stephan Stapczynski, MD, Editor
Historical Patterns of Opioid Use
Both the medicinal benefits and the alarming devastation related to the seemingly innocent poppy plant would have been difficult to predict more than 5,000 years ago, when the Sumerian culture first discovered the plant’s pain-relieving properties.1 The path to the current opioid crisis in the United States began when opium first became available in 1775, and led to the present day situation with more than 3 million U.S. citizens experiencing opioid use disorder. Trends in opioid-related deaths have shown a nearly fourfold increase between 1999 and 2008, with more than 64,000 deaths in 2017.2 That same year, the U.S. government increased awareness of the opioid epidemic by declaring it a public health emergency.
Pain control paradigms have attempted to address the moral obligations providers have to relieve pain and to minimize opioid misuse and addiction. The current epidemic is multifactorial, and it is evident that the problem will not be solved easily. An in-depth examination of the epidemiology, risk factors, pathophysiology, and management strategies is essential to curbing the problems of addiction and abuse.
Epidemiology
Opioid-related ED visits quadrupled between 1999 and 2010.3 Despite increased governmental and institutional prescribing regulations, opioid-related deaths have escalated dramatically over the past two decades. Reports estimate that 4 million people in the United States misused prescription pain medication in 2015. Almost half of those were diagnosed with an opioid use disorder or dependence in the prior year. Illicit drug use is closely related to prescription opioid abuse, with 5.1 million people admitting to having used heroin. In 2015, there were more than 80,000 ED visits for unintentional heroin-related poisonings in America, an estimated rate of approximately 26 per 100,000 people.4
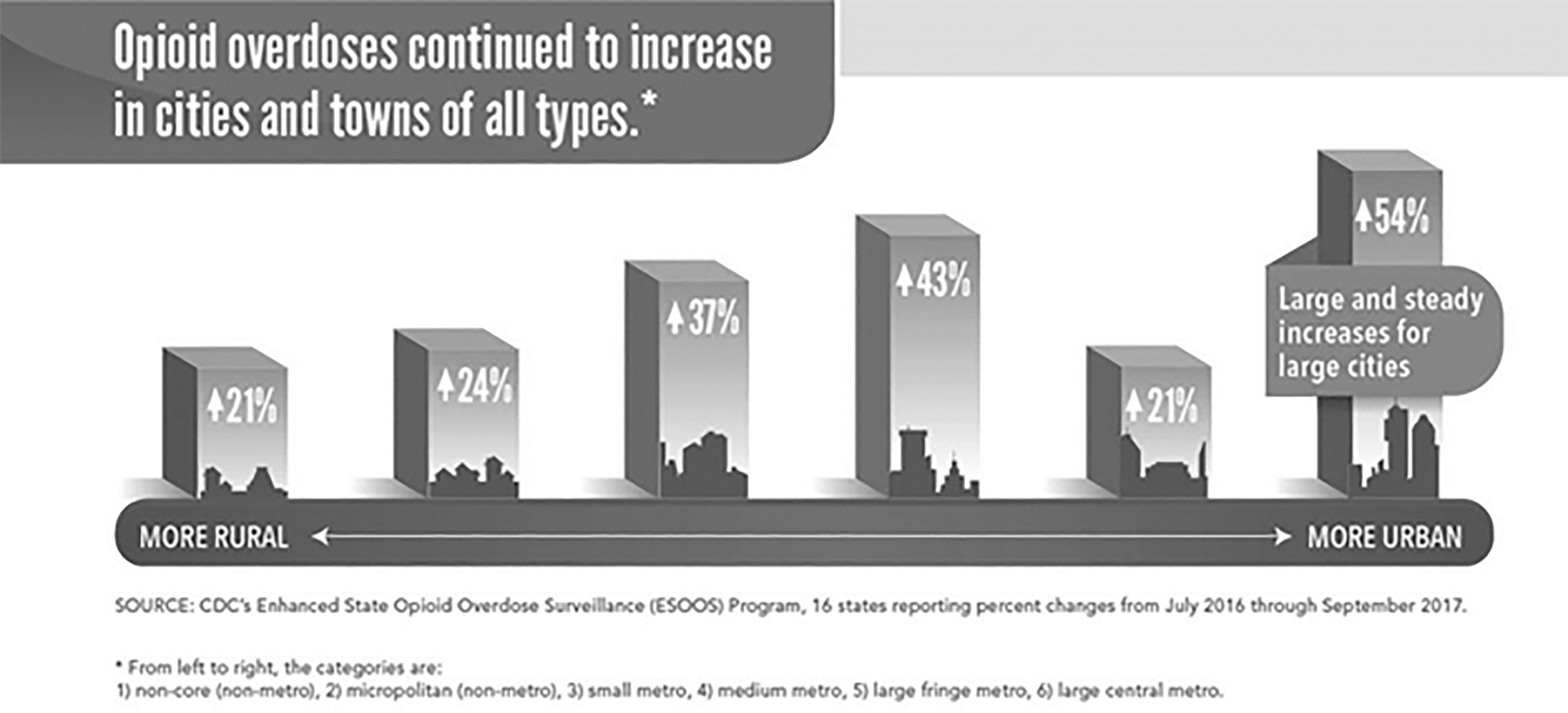
To put the epidemic into perspective, opioid-linked deaths now exceed deaths from motor vehicle accidents, guns, recent wars, and HIV infection. Further data show a significant increase in opioid overdose deaths, from 7.6 per 100,000 in 2013 to 21.7 per 100,000 in 2016. Opioid-related overdoses increased by a dramatic five times between 1999 and 2018.5 The largest areas of growth are large, central metropolitan areas. (See Figures 1 and 2.)
Figure 1. Opioid Overdoses Continued to Increase in Cities and Towns of All Types |
|
* From left to right, the categories are: non-core (non-metro): 21% increase; micropolitan (non-metro): 24% increase; small metro: 37% increase; medium metro: 43% increase; large fringe metro: 21% increase; large central metro: 54% increase |
|
Source: CDC’s Enhanced State Opioid Overdose Surveillance (ESOOS) Program, 16 states reporting percent changes from July 2016 through September 2017. Centers for Disease Control and Prevention. Vital Signs: Opioid overdoses treated in emergency departments: Identify opportunities for action. https://www.cdc.gov/vitalsigns/opioid-overdoses/infographic.html#graphic |
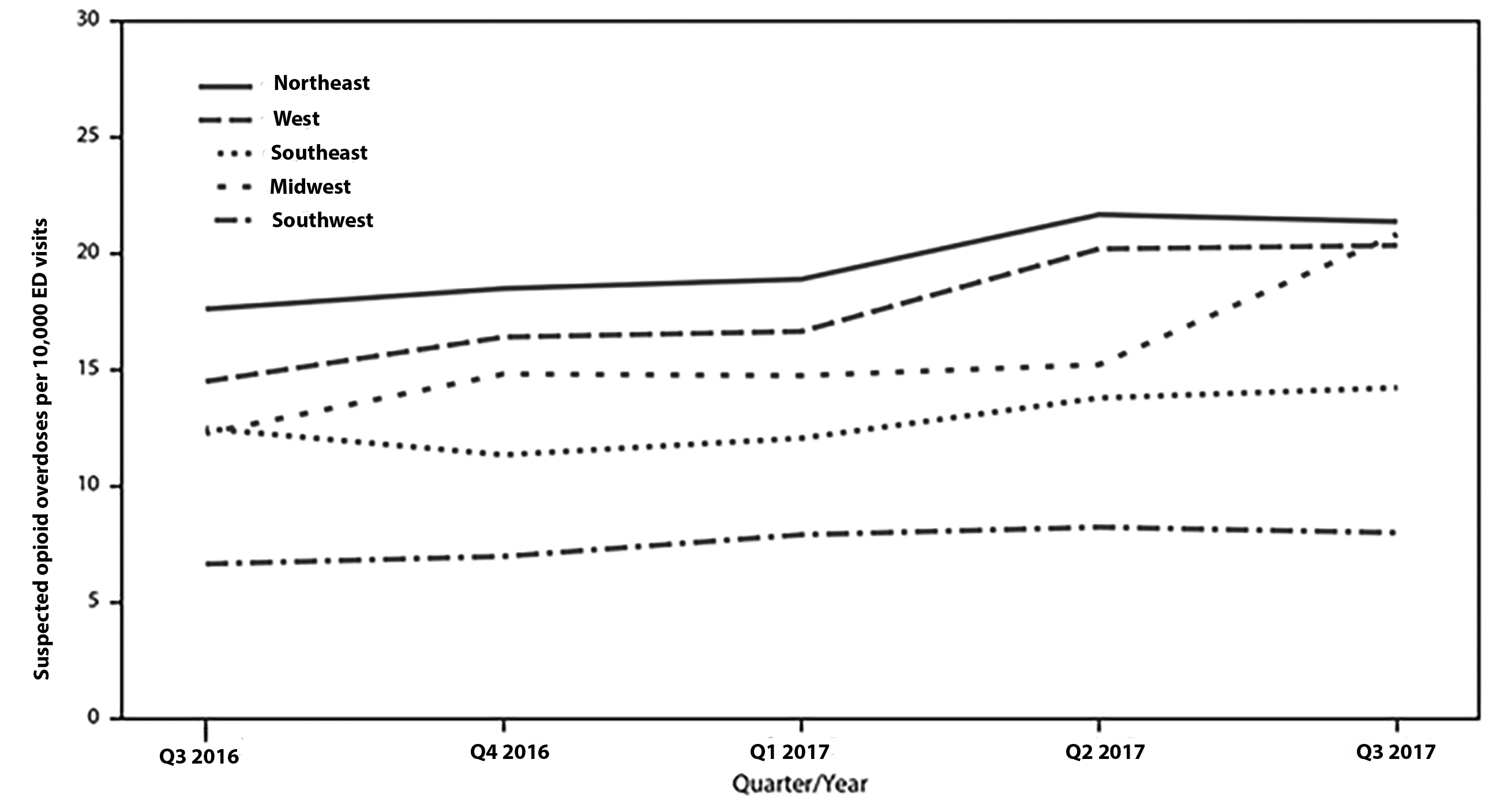
Figure 2. Quarterly Rate of Suspected Opioid Overdose, by U.S. Region — 52 Jurisdictions in 45 States, National Syndromic Surveillance Program, July 2016-September 2017 |
 |
|
Source: Vivolo AM, Seth P, Gladden RM, et al. Vital Signs: Trends in emergency department visits for suspected opioid overdoses — United States, July 2016–September 2017. MMWR Morb Mortal Wkly Rep 2018;67:279-285. https://www.cdc.gov/mmwr/volumes/67/wr/mm6709e1.htm#F1_down |
Opioid misuse also results in significant morbidity. Patients may present with local or systemic infection related to unsterile injection techniques, increased risk of HIV, hepatitis B, pneumonia, tuberculosis, opioid-induced bowel syndrome, opioid-induced hyperalgesia, increased incidence of motor vehicle collisions, and amnestic syndrome.
In addition to morbidity and mortality, there is a financial burden of opioid misuse. The Centers for Disease Control and Prevention (CDC) estimates that the economic cost of opioid misuse is a staggering $78.5 billion annually.6
Opioid addiction does not discriminate; it is prevalent in individuals from all educational and socioeconomic groups. The people at highest risk for prescription opioid and heroin abuse are male, 18-25 years of age, non-Hispanic whites, with an income of less than $20,000. However, it is important to recognize that recent trends indicate the gaps between gender, age, ethnicity, and socioeconomic groups are closing; therefore, any demographic should be considered at risk.1
Risk Factors
Risk factors for opioid misuse include age, mental health disorders, prolonged prescription opioid use, and chronic pain. Age extremes have been implicated as a contributing risk factor, with pediatric and geriatric surgical patients at greatest risk for developing an opioid disorder.7 Psychiatric conditions, such as depression and anxiety, also are associated with increased long-term opioid use following surgery. The risk of abuse increases in connection with the length of the prescription; in a study of more than 1 million opioid-naïve patients, each prescription refill of an opioid was associated with a 44% increased incidence of opioid misuse.8 Furthermore, patients experiencing chronic pain from conditions such as osteoarthritis and rheumatoid arthritis are at higher risk for opioid abuse.
Pathophysiology
By binding to opioid receptors in the central and peripheral nervous systems, opioids relieve pain, diarrhea, and cough. When acted upon, these delta, kappa, and mu receptors also may create physiologic effects, such as intense euphoria, antidepressant activity, sedation, miosis, dysphoria, respiratory depression, vasodilation, and reduced gastrointestinal (GI) motility. Tolerance also may occur as chronic use alters the sensitivity of receptors. These alterations have the potential to change pain perception, leading to hyperalgesia. If opioids are discontinued abruptly, withdrawal symptoms, such as vomiting, insomnia, anxiety, sweating, and dehydration, may occur.
Management Strategies
Employing effective prevention strategies for opioid prescribing is an important component of reducing opioid misuse and addiction. The approach must be mindful of the entire disease continuum, including the three basic levels of prevention: primary, secondary, and tertiary.9 (See Table 1.) Primary prevention focuses on deterring the initial development of opioid misuse. Secondary strategies aim to improve early detection and reduce severity. Tertiary strategies concentrate on decreasing complications and improving support and treatment once the disease has been established.
Table 1. Strategies for Safe Prescribing of Opioid Agents |
|
Primary Strategies |
|
|
Secondary Strategies |
|
|
Tertiary Strategies |
|
Opioid Therapy in the Emergency Department
Opioids are commonly prescribed in the ED setting for acute pain. Opioid therapy in the ED setting doubled between 2000 and 2010.10 It has been estimated that one in six ED patients received an opioid prescription.11 Opioid prescribing patterns vary significantly among ED providers.12,13
Opioids are effective therapy for acute pain. Nonopioid therapy may be considered, including such agents as nonsteroidal anti-inflammatory drugs (NSAIDs), acetaminophen, a lidocaine patch, methocarbamol, gabapentin, or topical NSAID agents. If an opioid is deemed the best agent, the potential benefits and harms should be considered. The risks of acute opioid therapy include dysphoria, confusion, somnolence, falls, and motor vehicle collisions. Physical dependence can develop within days for some individuals. Tolerance results in the reduced effectiveness of opioid therapy. Some patients may experience opioid hyperalgesia, where pain and distress are experienced out of proportion to the painful stimulus. Even short-term prescriptions may result in long-term opioid dependence, tolerance, and abuse.14-16 All opioids have the potential for addiction and abuse, regardless of the perceived reputation of “weak opioids” (i.e., codeine, dihydrocodeine, or tramadol).17
For acute pain, the lowest reasonable effective dose should be prescribed for the expected duration of the painful condition.18 Shared decision making is an effective strategy for pain management in the ED. Patients may be counseled regarding the goal of pain therapy to reduce (not necessarily eliminate) pain.19
Some EDs have initiated protocols to reduce the ED use of opioid therapy.20,21 Protocols often suggest the use of nonpharmacologic interventions, nonnarcotic analgesics, or local or regional anesthesia.
Prescription drug monitoring programs (PDMP) may provide helpful information regarding recent opioid therapy for individual patients. However, limitations of these programs may include missing or inaccurate data. The use and impact of PDMP data in EDs vary significantly.22-24 Clinical history and screening for opioid use disorder also are important to identify patients at risk.25
When an opioid is indicated, the choice and route of agent is based on the individual patient, clinical condition, age, previous history of opioid therapy, comorbidities, allergies, and intravenous access. The dose should be titrated to reach the minimal effective dose.26-28 Many textbooks and other resources suggest equianalgesic dosing of comparable opioids. However, opioid conversion tables are an oversimplification of pain management and may not be evidence-based.29
Pediatrics
Pain in pediatric patients requires a cautious approach. Pediatric patients may be vulnerable to addictive behavior. A recent study of the U.S. National Poison Data System (2005-2018) demonstrated that 25% of opioid overdoses occur among patients younger than 18 years of age.30 Between 2006 and 2015, ED prescriptions for opioids in pediatric patients declined.31 An approach to pain in the pediatric patient should include nonpharmacologic methods, such as immobilization and wound care, treatment with nonnarcotic analgesics (acetaminophen, NSAIDs), or treatment with opioids for severe pain.32 Intranasal fentanyl or ketamine may be effective alternatives to parenteral opioids.33-35
Chronic Pain
Some patients with chronic pain may come to the ED requesting opioid therapy.36 In most cases, treatment of chronic pain should be undertaken by the patient’s primary care physician or other treating physician. ED prescriptions for opioids for chronic pain are fraught with risk, including the risk of addiction or diversion. Patients who are taking long-acting opioids at baseline are at high risk of poor analgesic control following ED administration of IV opioids.37
Nonnarcotic options should be used whenever appropriate for the ED treatment of chronic pain. Patients may be referred to addiction treatment if appropriate.
End-of-Life Care
Treatment of pain near the end of life is a unique clinical scenario. Effective pain management outweighs the potential for addiction or misuse, and opioid therapy should be prescribed to provide effective analgesia.38 The World Health Organization (WHO) suggests an analgesic stepladder, which categorizes pain intensity according to severity, and recommends analgesic agents based on their strength. Analgesics should be prescribed at regular intervals, not as needed to treat exacerbations of pain. Step 1 includes mild pain, and appropriate treatment should include nonopioid analgesia, such as acetaminophen or an NSAID. Step 2 refers to patients with mild to moderate pain that is poorly controlled. Treatments may include acetaminophen-containing products, such as hydrocodone, oxycodone, codeine, and tramadol. Step 3 refers to patients with moderate to severe pain. Appropriate analgesic therapy includes morphine, hydromorphone, fentanyl, levorphanol, methadone, and oxycodone.39-41 High doses may be required to treat severe pain effectively.42
Alternatives to Opioid Use in the ED
In response to the problems related to opioid use, several institutions have developed protocols for the initial use of nonopioid therapy to address common pain ED presentations.43 Conditions such as musculoskeletal pain, extremity fracture, joint dislocation, renal colic, headache, and exacerbations of chronic abdominal pain can be managed with opioid alternatives without a decrease in patient satisfaction scores focused on pain treatment.44 (See Figure 3.)
Figure 3. Pain Pathways by Indication |

|
|
Figure used with permission from: Duncan RW, Smith KL, Maguire M, Stadler DE III. Alternatives to opioids for pain management in the emergency department decreases opioid usage and maintains patient satisfaction. Am J Emerg Med 2019;37:42. |
Systemic Nonopioid Pain Management
Acetaminophen
The exact mechanism of pain management properties of acetaminophen is unknown, but it is believed to act as a cyclooxygenase (COX) inhibitor. Acetaminophen is metabolized primarily in the liver through first-order kinetics. Because of this, there is a risk of hepatotoxicity in patients with preexisting liver impairment.
Acetaminophen may provide analgesia for a variety of painful conditions, including musculoskeletal pain, headache, infection, and numerous others. Acetaminophen administered with an NSAID has a synergic analgesic effect.45,46 Intravenous acetaminophen is a safe and effective option for patients unable to tolerate oral pain medications.47
Nonsteroidal Anti-Inflammatory Drugs
NSAIDs can be grouped in two categories: selective and nonselective. Nonselective NSAIDs (such as ibuprofen and ketorolac) inhibit the synthesis of cyclooxygenase isoenzyme 1 (COX-1) and cyclooxygenase isoenzyme 2 (COX-2), leading to decreased production of prostaglandins. All nonselective NSAIDs inhibit platelet aggregation, and are capable of adverse effects on the GI system, such as gastritis or gastric ulcers, interference with autoregulation of renal blood flow, hepatotoxicity, and hypertension. Therefore, a nonselective NSAID should be considered for short-term use for acute painful conditions.
The newer COX-2 inhibitors demonstrate similar efficacy to their traditional NSAID counterparts with several advantages. These compounds inhibit COX-2, which prevents an increase in prostaglandin E2, which leads to increased sensitization of peripheral nociceptors and central sensitization. The lack of COX-1 inhibition decreases the adverse effects and, therefore, constitutes an important part of multimodal analgesia. COX-2 inhibitors lack the GI effects of traditional NSAIDs and also decrease the risk of bleeding, renal dysfunction, and bronchospasm in sensitive patients.48,49
NSAIDs play an important role in analgesia for a variety of conditions, including musculoskeletal pain, trauma, nephrolithiasis, headache, and other painful conditions. NSAIDs should be avoided in patients who are pregnant, as well as those with a history of GI bleeding or aspirin allergy. Relative contraindications include renal disease, heart disease, or age older than 65 years.
Topical diclofenac is an effective treatment for musculoskeletal pain and has a lower risk of systemic adverse effects.50,51
Alpha-2 Adrenergic Agonists
Alpha-2 agonists have been used since the 1960s for high blood pressure treatment, sedation, and drug detoxification. Recently, alpha-2 agonists have been cited for their use in modifying the pain response (examples include tizanidine, clonidine, and dexmedetomidine). The use of this class in the management of pain perception seems to take place both centrally and peripherally. Pre- and postsynaptic alpha-2 adrenergic receptors are found on primary afferent nerves, dorsal horn neurons, and brainstem nuclei.
Anticonvulsants
Pregabalin and gabapentin are used commonly to treat seizures and neuropathic pain. Both drugs bind to the α2δ subunit of voltage-gated calcium channels in the spinal cord and brain. This results in a post-synaptic inhibition of calcium influx, reducing the presynaptic excitatory neurotransmitter release.
Pregabalin and gabapentin generally are safe, with mostly mild to moderate side effects. The side effects reported most commonly are dizziness, somnolence, and peripheral edema. The reported adverse reactions include angioedema and hypersensitivity reactions. Dosing should be reduced in patients with renal insufficiency. Elderly patients may have a higher frequency of the side effects mentioned earlier.52
Ketamine
Ketamine binds to the N-methyl-d-aspartate (NMDA) receptors, modulating central sensory processing and reducing pain. Ketamine in low doses has demonstrated analgesic efficacy without causing the psychomimetic effects that typically are associated with larger doses. Low-dose ketamine has not been associated with adverse effects on respirations, cardiovascular function, or GI function. Ketamine is thought to potentiate the effects of opioid analgesic effects. Although it is not an opioid, ketamine does have the potential for abuse.49,52,53
Ketamine has demonstrated efficacy for a variety of clinical indications, including agitation, depression, acute pain, procedural sedation, and rapid sequence intubation.54-56 (See Table 2.) Ketamine should be avoided in patients with cardiovascular disease, pregnancy, hepatic dysfunction, elevated intracranial pressure, or elevated intraocular pressure.
Table 2. Ketamine Indications, Dosages, and Routes of Administration* |
|
|
Indication |
Dose and Route |
|
Acute pain |
0.2 to 0.3 mg/kg IV bolus or 0.1 to 0.2 mg/kg/hour infusion |
|
Agitation |
0.5 to 1 mg/kg IV 1 to 3 mg/kg IM |
|
Rapid sequence intubation |
1 to 1.5 mg/kg IV |
|
Procedural sedation |
0.5 to 1 mg/kg IV 1 to 3 mg/kg IM 5 to 10 mg/kg IN (pediatrics) |
|
IV = intravenous; IM = intramuscular; IN = intranasal *Dosages provided as an estimate. Appropriate dosing should be based on individual patient characteristics and risk factors. |
|
Corticosteroids
Corticosteroids are used to reduce pain and inflammation. These drugs act by inhibiting phospholipase, resulting in decreased production of prostaglandins, leukotrienes, and cytokines, which play a role in inflammatory pain. Despite the long history of use and the evidence for analgesic actions, few studies have been conducted and no widespread use has been seen. This may be because of the adverse effects associated with repeated corticosteroid administration.50 Steroids have been used with variable success to treat painful conditions, such as neck pain, back pain, dental pain, gout, and headache.57-59
Local and Regional Anesthesia for Acute Pain Management
Regional anesthesia can be an effective analgesic technique for acute musculoskeletal pain, including traumatic pain.60 Regional anesthesia is performed by a nerve block of the affected area with local anesthesia, such as lidocaine or bupivacaine. Ultrasound-guided regional anesthesia provides the relevant view of anatomic structures to ensure a successful nerve block and reduce complications. Complications of regional anesthesia may include accidental intravascular injection, intraneural injection, bleeding, and infection. Examples of relevant regional anesthetic nerve blocks are discussed in the following sections. Authoritative resources may be consulted for specific techniques for a wide variety of nerve blocks.
Occipital Nerve Block
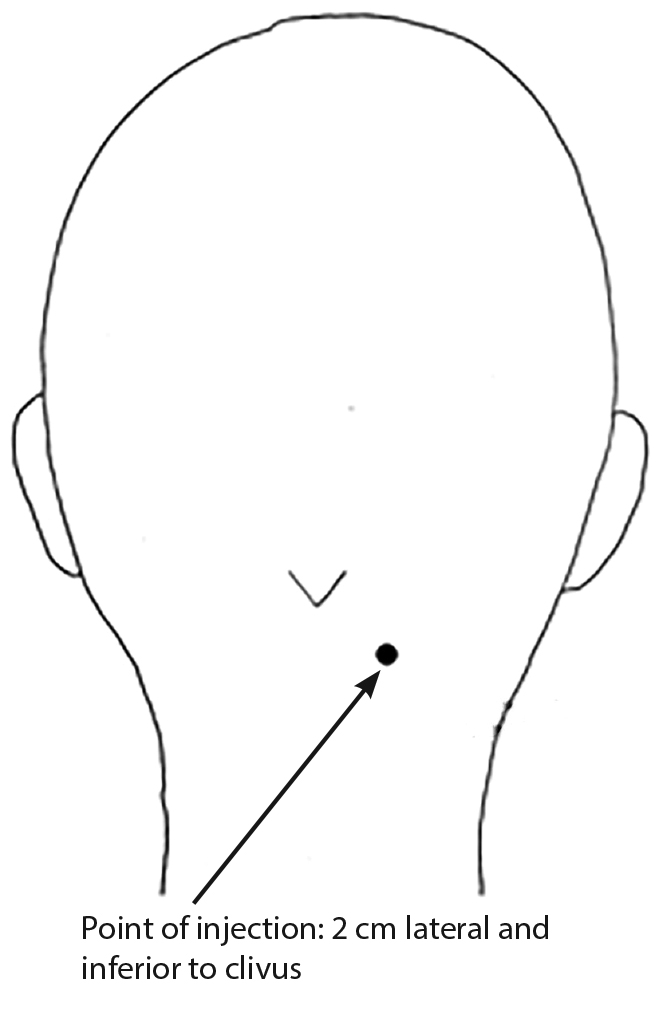
Headaches are listed consistently among the top 20 primary diagnoses in EDs.61 An occipital nerve blockade can be an effective treatment for symptoms associated with migraine headaches with cervico-occipital spreading. Several anatomic approaches to the point of injection are possible. A simple technique to the occipital nerve block is injecting 2 mL to 3 mL of local anesthetic to the greater occipital nerve, located 2 cm lateral and inferior to the occipital clivus (or one-third of the distance from the clivus to the mastoid process). (See Figure 4.)
Figure 4. Greater Occipital Nerve Block |
 |
Sphenopalatine Ganglion Block
The sphenopalatine ganglion is the largest collection of neurons in the calvarium outside of the brain.62 As such, the sphenopalatine ganglion block has the potential to resolve the symptoms of several types of headache, including trigeminal neuralgia, migraine, nasal/sinus headache, and post-dural puncture headaches. A sphenopalatine ganglion block may be achieved by injection or direct application. The injection technique involves the injection of 0.5 mL of viscous lidocaine via a needleless syringe in each nostril. An atomizer can be used as an alternative. Alternatively, local anesthetic can be applied directly with cotton tip applicators along the superior border of the middle turbinate of each nostril.
Fascia Iliaca Compartment Block
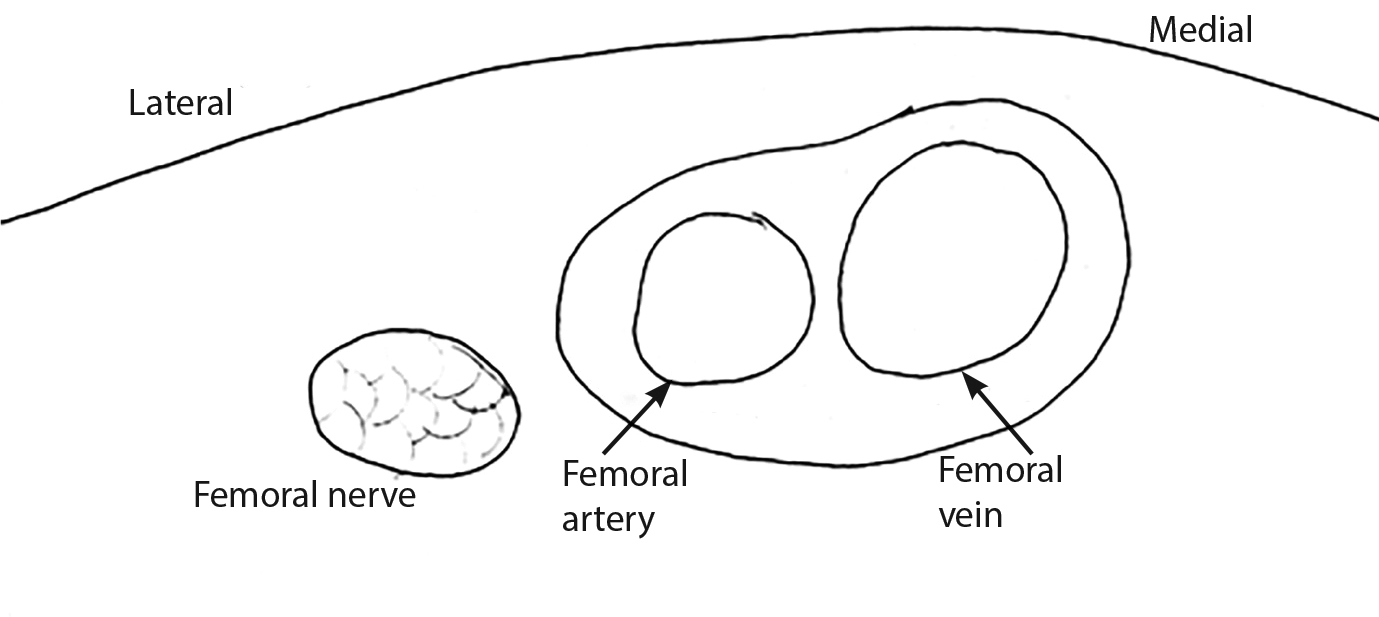
A fascia iliaca compartment block is an effective technique to achieve regional anesthesia of the femoral nerve. This procedure has demonstrated effective pain control following hip fracture or dislocation.63-66 The procedure should be performed under ultrasound guidance, with injection of 20 mL of dilute anesthetic surrounding the femoral nerve, located lateral and deep to the femoral artery. Pain relief usually can be accomplished within 30 minutes of the placement of the block. Relevant anatomy is shown in Figure 5. (For more information on ultrasound-guided nerve blocks, see the March 1, 2018 issue of Emergency Medicine Reports.)
Figure 5. Anatomy of Fascia Iliaca Block |
 |
Lidocaine Patch
A lidocaine transdermal patch is an option to treat localized pain. The lidocaine patch is composed of an adhesive material that contains 5% lidocaine applied to a polyester felt backing. Four percent patches are available over-the-counter, and 5% patches are available by prescription. Lidocaine patches are effective in the treatment of musculoskeletal pain, neuropathic pain, and sickle cell crisis pain.67-74 The use of a lidocaine patch also may be helpful in reducing pain for procedures such as incision and drainage, or intravenous access in the ED.75,76 A lidocaine patch is effective for up to 12 hours and then removed, because the patch is designed to be applied for no longer than 12 hours per day. Potential adverse effects typically are local skin reactions, such as a burning sensation, erythema, papules, pruritus, or vesicles. Although the use of a lidocaine patch does not treat the underlying condition, its analgesic efficacy makes it a valuable nonopioid alternative.
Conclusions
Opioid therapy can be an effective form of pain management in the ED for acute painful conditions. The risk of addiction and abuse should be considered in every case. Alternatives to opioid therapy include systemic agents, such as acetaminophen, NSAIDs, lidocaine, alpha agonists, anticonvulsants, ketamine, corticosteroids, and local and regional anesthesia.
REFERENCES
- Green J. Epidemiology of opioid abuse and addiction. J Emerg Nurs 2017;43:106-113.
- Jones MR, Viswanath O, Peck J, et al. A brief history of the opioid epidemic and strategies for pain medicine. Pain Ther 2018;7:13-21.
- Tadros A, Layman SM, Davis SM, et al. Emergency visits for prescription opioid poisonings. J Emerg Med 2015;49:871-877.
- Centers for Disease Control and Prevention. 2018 Annual Surveillance Report of Drug-Related Risks and Outcomes — United States. Surveillance Special Report. Centers for Disease Control and Prevention, U.S. Department of Health and Human Services. Published Aug. 31, 2018. https://www.cdc.gov/drugoverdose/pdf/pubs/2018-cdc-drug-surveillance-report.pdf
- Neville K, Foley M. The economic impact of the opioid use disorder epidemic in America: Nurses’ call to action. Nursing Economic$ 2020;38:7-51.
- National Institute on Drug Abuse. Opioid overdose crisis. February 2020. www.drugabuse.gov/drugs-abuse/opioids/opioid-overdose-crisis
- Zhao S, Chen F, Feng A, et al. Risk factors and prevention strategies for postoperative opioid abuse. Pain Res Manag 2019;2019:7490801.
- Brat GA, Agniel D, Beam A, et al. Postsurgical prescriptions for opioid naive patients and association with overdose and misuse: Retrospective cohort study. BMJ 2018;17;360:j5790.
- Mathis SM, Hagemeier N, Hagaman A, et al. A dissemination and implementation science approach to the epidemic of opioid use disorder in the United States. Current HIV/AIDS Rep 2018;15:359-370.
- Chang HY, Daubresse M, Kruszewski SP, Alexander GC. Prevalence and treatment of pain in EDs in the United States, 2000 to 2010. Am J Emerg Med 2014;32:421-431.
- Hoppe JA, Nelson LS, Perrone J, Weiner SG. Opioid prescribing in a cross section of US emergency departments. Ann Emerg Med 2015;66:253-259.
- Smulowitz PB, Cary C, Boyle KL, et al. Variation in opioid prescribing patterns between ED providers. Intern Emerg Med 2016;11:1121-1124.
- Barnett ML, Olenski AR, Jena AB. Opioid-prescribing patterns of emergency physicians and risk of long-term use. N Engl J Med 2017;376:663-673.
- Hoppe JA, Kim H, Heard K. Association of emergency department opioid initiation with recurrent opioid use. Ann Emerg Med 2015;65:493-499.
- Hooten WM, St Sauver JL, McGree ME, et al. Incidence and risk factors for progression from short-term to episodic or long-term opioid prescribing: A population-based study. Mayo Clin Proc 2015;90:850-856.
- Calcaterra SL, Yamashita TE, Min SJ, et al. Opioid prescribing at hospital discharge contributes to chronic opioid use. J Gen Intern Med 2016;31:478-485.
- [No authors listed]. “Weak” opioid analgesics. Codeine, dihydrocodeine and tramadol: No less risky than morphine. Prescrire Int 2016;25:45-50.
- Strayer RJ, Motov SM, Nelson LS. Something for pain: Responsible opioid use in emergency medicine. Amer J Emerg Med 2017;35:337-341.
- Lee TH. Zero pain is not the goal. JAMA 2016;315:1575-1577.
- Motov S, Drapkin J, Butt M, et al. Analgesic administration for patients with renal colic in the emergency department before and after implementation of an opioid reduction initiative. West J Emerg Med 2018;19:1028-1035.
- Chacko J, Greenstein J, Ardolic B, Berwald N. Effect of an emergency department opioid prescription policy on prescribing patterns. Am J Emerg Med 2017;35:1327-1329.
- Pomerleau AC, Nelson LS, Hoppe JA, et al. The impact of prescription drug monitoring programs and prescribing guidelines on emergency department opioid prescribing: A multi-center survey. Pain Med 2017;18:889-897.
- Elder JW, DePalma G, Pines JM. Optimal implementation of prescription drug monitoring programs in the emergency department. West J Emerg Med 2018;19:387-391.
- Martello J, Cassidy B, Mitchell A. Evaluating emergency department opioid prescribing behaviors after education about mandated use of the Pennsylvania Prescription Drug Monitoring Program. J Addict Nurs 2018;29:196-202.
- Hawk K, D’Onofrio G, Fiellin DA, et al. Past-year Prescription Drug Monitoring Program opioid prescriptions and self-reported opioid use in an emergency department population with opioid use disorder. Acad Emerg Med 2018;25:508-516.
- Geppetti P, Benemei S. Pain treatment with opioids: Achieving the minimal effective and the minimal interacting dose. Clin Drug Investig 2009;29(Suppl 1):3-16.
- Griggs CA, Schulz CJ. A changing landscape of opioid prescribing in emergency medicine. Am J Emerg Med 2019;37:327-328.
- American College of Emergency Physicians. Critical issues in the prescribing of opioids for adult patients in the emergency department (June 2012). https://www.acep.org/patient-care/clinical-policies/opioids/
- Patanwala AE, Duby J, Waters D, Erstad BL. Opioid conversions in acute care. Ann Pharmacother 2007;41:255-266.
- Boyles S. Severity of kids’ opioid poisoning on the rise. Medpage Today. Feb. 17, 2020. https://www.medpagetoday.com/meetingcoverage/sccm/84925
- Tomaszewski DM, Arbuckle C, Yang S, Linstead E. Trends in opioid use in pediatric patients in US emergency departments from 2006 to 2015. JAMA Netw Open 2018;1:e186161.
- Bailey B, Trottier ED. Managing pediatric pain in the emergency department. Paediatr Drugs 2016;18:287-301.
- Borland M, Jacobs I, King B, O’Brien D. A randomized controlled trial comparing intranasal fentanyl to intravenous morphine for managing acute pain in children in the emergency department. Ann Emerg Med 2007;49:335-340.
- Graudins A, Meek R, Egerton-Warburton D, et al. The PICHFORK (Pain in Children Fentanyl or Ketamine) trial: A randomized controlled trial comparing intranasal ketamine and fentanyl for the relief of moderate to severe pain in children with limb injuries. Ann Emerg Med 2015;65:248-254.e1.
- Frey TM, Florin TA, Caruso M, et al. Effect of intranasal ketamine vs fentanyl on pain reduction for extremity injuries in children: The PRIME randomized clinical trial. JAMA Pediatr 2019;173:140-146.
- Olsen JC, Ogarek JL, Goldenberg EJ, Sulo S. Impact of a chronic pain protocol on emergency department utilization. Acad Emerg Med 2016;23:424-432.
- O’Connor AB, Zwemer FL, Hays DP, Feng C. Outcomes after intravenous opioids in emergency patients: A prospective cohort analysis. Acad Emerg Med 2009;16:477-487.
- Reyes-Gibby CC, Anderson KO, Todd KH. Risk for opioid misuse among emergency department cancer patients. Acad Emerg Med 2016;23:151-158.
- Prommer E, Ficek B. Management of pain in the elderly at the end of life. Drugs Aging 2012;29:285-305.
- Schug SA, Chandrasena C. Pain management of the cancer patient. Expert Opin Pharmacother 2015;16:5-15.
- World Health Organization: WHO’s cancer pain ladder for adults. https://www.who.int/cancer/palliative/painladder/en/
- Groninger H, Vijayan J. Pharmacologic management of pain at the end of life. Am Fam Physician 2014;90:26-32.
- George Washington University. Urgent Matters. Alternatives to opioids for pain management in the ED. https://smhs.gwu.edu/urgentmatters/content/alternatives-opioids-pain-management-ed
- Duncan RW, Smith KL, Maguire M, Stader DE 3rd. Alternatives to opioids for pain management in the emergency department decreases opioid usage and maintains patient satisfaction. Am J Emerg Med 2019;37:38-44.
- Hyllested M, Jones S, Pedersen JL, Kehlet H. Comparative effect of paracetamol, NSAIDs or their combination in postoperative pain management: A qualitative review. Br J Anaesth 2002;88:199-214.
- Ong CK, Seymour RA, Lirk P, Merry AF. Combining paracetamol (acetaminophen) with nonsteroidal antiinflammatory drugs: A qualitative systematic review of analgesic efficacy for acute postoperative pain. Anesth Analg 2010;110:1170-1179.
- Sin B, Wai M, Tatunchak T, Motov SM. The use of intravenous acetaminophen for acute pain in the emergency department. Acad Emerg Med 2016;23:543-553.
- Dahlen L, Oakes JM. A review of physiology and pharmacology related to acute perioperative pain management. AANA J 2017;85:300-308.
- Sullivan D, Lyons M, Montgomery R, Quinlan-Colwell A. Exploring opioid-sparing multimodal analgesia options in trauma: A nursing perspective. J Trauma Nurs 2016;23:361-375.
- Tieppo Francio V, Davani S, Towery C, Brown TL.Oral versus topical diclofenac sodium in the treatment of osteoarthritis. J Pain Palliat Care Pharmacother 2017;31:113-120.
- Baraf HS, Gloth FM, Barthel HR, et al. Safety and efficacy of topical diclofenac sodium gel for knee osteoarthritis in elderly and younger patients: Pooled data from three randomized, double-blind, parallel-group, placebo-controlled, multicentre trials. Drugs Aging 2011;28:27-40.
- Nicol AL, Hurley RW, Benzon HT. Alternatives to opioids in the pharmacologic management of chronic pain syndromes: A narrative review of randomized, controlled, and blinded clinical trials. Anesth Analg 2017;125:1682-1703.
- Knezevic NN, Yekkirala A, Yaksh TL. Basic/translational development of forthcoming opioid- and nonopioid-targeted pain therapeutics. Anesth Analg 2017;125:1714-1732.
- Schwenk ES, Viscusi ER, Buvanendran A, et al. Consensus guidelines on the use of intravenous ketamine infusions for acute pain management from the American Society of Regional Anesthesia and Pain Medicine, the American Academy of Pain Medicine, and the American Society of Anesthesiologists. Reg Anesth Pain Med 2018;43:456-466.
- Riddell J, Tran A, Bengiamin R, et al. Ketamine as a first-line treatment for severely agitated emergency department patients. Am J Emerg Med 2017;35:1000-1004.
- Mahshidfar B, Mofidi M, Fattahi M, et al. Acute pain management in the emergency department, low dose ketamine versus morphine: A randomized clinical trial. Anesth Pain Med 2017;7:e60561.
- Elkhadem A, Ezzat K, Ramadan M, et al. The effect of preoperative oral administration of prednisolone on postoperative pain in patients with symptomatic irreversible pulpitis: A single-centre randomized controlled trial. In Endod J 2018;51(Suppl 3):e189-e196.
- Rainer TH, Cheng CH, Janssens HJ, et al. Oral prednisolone in the treatment of acute gout: A pragmatic, multicenter, double-blind, randomized trial. Ann Intern Med 2016;164:464-471.
- Wei J, Robbins MS. Greater occipital nerve injection versus oral steroids for short term prophylaxis of cluster headache: A retrospective comparative study. Headache 2018;58:852-858.
- Slade IR, Samet RE. Regional anesthesia and analgesia for acute trauma patients. Anesthesiol Clin 2018;36:431-454.
- Yanuck J, Shah S, Jen M, Dayal R. Occipital nerve blocks in the emergency department for initial medication-refractory acute occipital migraines. Clin Pract Cases Emerg Med 2019;3:6-10.
- Ho KWD, Przkora R, Kumar S. Sphenopalatine ganglion: Block, radiofrequency ablation and neurostimulation - a systematic review. J Headache Pain 2017;18:118.
- Groot L, Dijksman LM, Simons MP, et al. Single fascia iliaca compartment block is safe and effective for emergency pain relief in hip-fracture patients. West J Emerg Med 2015;16:1188-1193.
- Eyi YE, Arziman I Kaldirim U, Tuncer SK. Fascia iliaca compartment block in the reduction of dislocation of total hip arthroplasty. Am J Emerg Med 2014;32:1139.
- Ritcey B, Pageau P, Woo MY, Perry JJ. Regional nerve blocks for hip and femoral neck fractures in the emergency department: A systematic review. CJEM 2016;18:37-47.
- Chesters A, Atkinson P. Fascia iliaca block for pain relief from proximal femoral fracture in the emergency department: A review of the literature. Emerg Med J 2014;31:e84-e87.
- Binder A, Bruxelle J, Rogers P, et al. Topical 5% lidocaine (lignocaine) medicated plaster treatment for post-herpetic neuralgia: Results of a double-blind, placebo-controlled, multinational efficacy and safety trial. Clin Drug Investig 2009;29:393-408.
- Galer BS, Gammaitoni AR, Oleka N, et al. Use of the lidocaine patch 5% in reducing intensity of various pain qualities reported by patients with low-back pain. Curr Med Res Opin 2004;20 (Suppl 2):S5-S12.
- Sommer C, Cruccu G. Topical treatment of peripheral neuropathic pain: Applying the evidence. J Pain Symptom Manage 2017;53:614-629.
- Nalamachu S, Crockett RS, Gammaitoni AR, Gould EM. A comparison of the lidocaine patch 5% vs naproxen 500 mg twice daily for the relief of pain associated with carpal tunnel syndrome: A 6-week, randomized, parallel-group study. MedGenMed 2006;8:33.
- Nalamachu S, Nalamasu R, Jenkins J, Marriott T. An open-label pilot study evaluating the effectiveness of the heated lidocaine/tetracaine patch for the treatment of pain associated with carpal tunnel syndrome. Pain Pract 2014;14:607-612.
- Rauck R, Busch M, Marriott T. Effectiveness of a heated lidocaine/tetracaine topical patch for pain associated with myofascial trigger points: Results of an open-label pilot study. Pain Pract 2013;13:533-538.
- Jensen MP, Trudeau JJ, Radnovich R, et al. The pain quality response profile of a corticosteroid injections and heated lidocaine/tetracaine patch in the treatment of shoulder impingement syndrome. Clin J Pain 2015;31:342-348.
- Rousseau V, Morelle M, Arriuberge C, et al. Efficacy and tolerance of lidocaine 5% patches in neuropathic pain and pain related to vaso-occlusive sickle cell crises in children: A prospective multicenter clinical study. Pain Pract 2018;18:788-797.
- Bourne CL, Brewer KL, House J. Injectable lidocaine provides similar analgesia compared to transdermal lidocaine/tetracaine patch for the incision and drainage of skin abscesses: A randomized, controlled trial. J Emerg Med 2014;47:367-371.
- Singer AJ, Taira BR, Chisena EN, et al. Warm lidocaine/tetracaine patch versus placebo before pediatric intravenous cannulation: A randomized controlled trial. Ann Emerg Med 2008;52:41-47.
Opioid therapy can be an effective form of pain management in the ED for acute painful conditions. The risk of addiction and abuse should be considered in every case. Alternatives to opioid therapy include systemic agents, such as acetaminophen, NSAIDs, lidocaine, alpha agonists, anticonvulsants, ketamine, corticosteroids, and local and regional anesthesia.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.