Assessing and Treating Exacerbations of Chronic Obstructive Pulmonary Disease
EXECUTIVE SUMMARY
Chronic obstructive pulmonary disease (COPD) affects 16 million people in the United States and 328 million people worldwide. It is associated with significant mortality and takes a heavy economic cost, projected at $49 billion in 2020.
- Exacerbations of COPD have multiple causes, including respiratory infection, air pollution, cold weather, gastroesophageal reflux, and poor adherence to home medications.
- Worsening of dyspnea and productive cough are the typical symptoms of exacerbation.
- Generally, treatment includes supplemental oxygen, bronchodilators, steroids, and antibiotics. Short-acting beta-2 agonists, such as albuterol, are recommended as first-line bronchodilators for COPD exacerbations. Short-acting anticholinergic agents, such as ipratropium bromide, are recommended in severe exacerbations, although are not shown to enhance bronchodilator effects or prevent hospitalization.
- Sputum cultures for patients treated outside the hospital do not appear to be helpful unless the patient has risk factors for P. aeruginosa infection.
- Testing for inflammatory markers, such as C-reactive protein and calcitonin, guides the need for antibiotic use without over-prescribing or compromising care.
- To prevent future exacerbations, long-acting beta-2 agonists and long-acting muscarinic antagonists, along with inhaled corticosteroids, commonly are used.
Chronic obstructive pulmonary disease (COPD) is a significant global health problem. People with COPD frequently seek care for acute exacerbations, which are associated with significant morbidity and mortality. Therefore, it is crucial for physicians to understand how to assess and treat these patients appropriately.
Epidemiology
COPD is a widespread illness, affecting approximately 16 million people in the United States and 328 million people around the world.1,2 The disease also is associated with significant mortality, contributing to 156,637 deaths in the United States in 2017 and about 2.9 million globally.2,3 COPD not only takes a toll on health, but it also imposes enormous economic costs. In 2010, COPD resulted in a total medical cost of about $32.1 billion in the United States, with the cost projected to rise substantially to $49 billion by 2020.4
In the United States, COPD is most prevalent among women ages 65 to 74 years and men ages 75 to 84 years.5 Symptoms such as chronic dyspnea and cough should prompt the clinician to consider a diagnosis of COPD. The diagnosis then is made definitively by spirometry demonstrating an airflow obstruction that does not fully respond to inhaled bronchodilators.6
Comorbidities are common among patients with COPD. Cardiovascular comorbidities are particularly common and are associated with poorer outcomes. Specifically, the prevalence of coronary artery disease among those with COPD is estimated to be as high as 33%, and approximately 24% have concomitant heart failure.6,7
COPD was responsible for an estimated 923,000 emergency department (ED) visits in 2017, the most recent year for which data are available.8 Emergency visits relating to COPD are commonly the result of acute exacerbations, characterized by a sudden worsening of symptoms. These are significant events in the progression of disease. A history of exacerbations predicts future exacerbations, with exacerbation frequency increasing in patients with more severe disease.9,10 Patients with a history of myocardial infarction and heart failure tend to have more frequent exacerbations.11 Even one exacerbation is associated with some deterioration in a patient’s respiratory status.12 Exacerbations also are associated with higher mortality.11
Pathophysiology
COPD, as its name suggests, is defined by a chronic impediment to airflow through the bronchial tree. This impediment is primarily the product of two processes. The first is obstruction of the small airways by fibrosis and mucus. The second is emphysema, which describes the destruction of the lung’s elastic structure by proteolytic enzymes. When the lung loses its supportive structure, the airways are prone to collapse with expiration. Both processes are triggered by chronic inflammation in the lungs that is most commonly the result of cigarette smoke but also can be influenced by environmental and occupational exposures.6,13,14
One notable complication of COPD is cor pulmonale. Because of the ventilation-perfusion (V/Q) mismatch that exists in the lungs, the pulmonary blood vessels undergo hypoxic vasoconstriction and remodeling over time. A decrease in nitric oxide production by endothelial cells also is theorized to contribute. Together, these processes increase the pressure in the pulmonary artery. This increased afterload induces the development of right ventricular hypertrophy, which can lead to right heart failure.15
Acute COPD exacerbations represent a sudden worsening of the chronic airway inflammation that defines this disease.16 Greater bronchial edema and bronchoconstriction worsen the obstruction that exists at baseline, trapping oxygen-depleted air in the lungs and resulting in impaired ventilation of gas-exchanging lung segments. Concurrently, an increase in resistance to blood flow through the pulmonary arteries limits the amount of blood that reaches the alveoli to participate in gas exchange. Together, these changes aggravate the existing V/Q mismatch and lead to worsening hypoxemia and hypercapnia.17
Etiology
There are many causes of COPD exacerbations, with respiratory infection being the most common. Various viruses account for about 30% of these infections.18 The viral pathogens most commonly implicated, in order of frequency, include rhinovirus, respiratory syncytial virus, coronavirus, and influenza.19 The prevalence of specific pathogens may vary geographically and seasonally. Bacterial infections also incite exacerbations. Causative bacteria tend to differ, depending on the severity of the underlying disease. Haemophilus influenzae and Moraxella catarrhalis predominate in patients with milder COPD, while Pseudomonas aeruginosa, Klebsiella pneumoniae, Staphylococcus aureus, and Streptococcus pneumoniae are found more commonly in patients with more severe underlying respiratory disease.18
Poor adherence to home medications is another notable factor that contributes to COPD exacerbations. One study that used data from a large COPD medication trial showed a clear correlation between adherence to COPD medications and clinical outcomes. The authors found that poor adherence was associated with both a higher rate of exacerbations necessitating hospitalization and greater mortality.20 Air pollution, cold weather, and gastroesophageal reflux disease are other factors postulated to play a role in triggering exacerbations, although data for some of these factors are inconclusive.18,21
Clinical Features
Symptoms commonly associated with COPD include dyspnea and productive cough. Sudden worsening of these symptoms may herald an acute exacerbation, for which patients may seek treatment from their primary care physician or in the ED.6
When assessing a patient with a suspected exacerbation, look first for signs of severe illness that require immediate intervention. For example, identify signs of respiratory distress, such as breathing through pursed lips, inability to speak in complete sentences, leaning forward, diaphoresis, and use of accessory muscles of respiration. Examine the patient’s chest and abdomen to determine the use of accessory muscles of respiration.22 Look also at the neck, because a deviated trachea could signify a tension pneumothorax. Take note of the vital signs, paying special attention to the respiratory rate and oxygen saturation to identify tachypnea and hypoxia, respectively. Tachycardia may signify a tachydysrhythmia or simply reflect the sympathetic response to a distressing situation. A fever may indicate an infectious process, such as pneumonia. A decrease of 10 mmHg or more in the patient’s systolic blood pressure associated with inspiration would be concerning for pulsus paradoxus.23 Speak with the patient to assess his or her mental status, since acute hypercarbic respiratory failure can cause patients to appear somnolent or even agitated and uncooperative.
If no threat to life requiring immediate action is identified on this initial evaluation, take the time to perform a thorough history and physical examination. In addition to the basic elements of the history of present illness, pertinent historical questions should be asked. If the patient has a standing diagnosis of COPD, find out what medications he or she uses at home and whether the patient requires supplemental oxygen. Ask about the rate of oxygen, usually reported as liters/minute, and whether that rate has increased during this acute exacerbation. Ask about the duration of oxygen use during the day, such as 24 hours or only at bedtime. Increased flow rate and hours of use indicate an exacerbation of underlying disease. Ask about oxygen source, either concentrator or pressurized tanks, and whether the oxygen supply has been lost because of delayed delivery or electrical power loss. Inquire about a history of exacerbations and if the patient has ever required noninvasive ventilation (NIV), intubation, or admission to the intensive care unit (ICU).
Because respiratory infections and environmental exposures are common causes of exacerbations, ask about infectious symptoms, such as fever and cough, as well as potential exposures. Ask whether the patient’s cough is productive, and if so, how the current sputum compares to the color and volume of sputum the patient usually has. Also include a pertinent review of systems, emphasizing symptoms like chest pain that, when endorsed, can suggest a serious complication of an exacerbation or an alternate diagnosis.
Examine the patient’s neck for jugular venous distention, which may suggest emergent conditions like acute heart failure or cardiac tamponade, but that also may be present in COPD patients who have cor pulmonale and other forms of chronic heart failure. The lung examination may reveal adventitious sounds, such as wheezes or rhonchi.24 Patients in extremis may have significantly decreased air movement and, thus, might not have appreciable wheezes or rhonchi on lung examination. Diminished or absent unilateral breath sounds may indicate a pneumothorax. Cardiac gallops and murmurs can indicate heart failure or valvular disease. However, because of the lung hyperinflation that often accompanies COPD, heart sounds may be difficult to auscultate.
Look at the patient’s fingers and perioral area for cyanosis, which may be present in severe hypoxia. Examine the lower extremities as well. Bilateral edema may be present in patients with heart failure, whereas unilateral swelling in a dyspneic patient would suggest venous thromboembolism.25
Diagnostic Studies
Basic laboratory studies, such as a complete blood count (CBC) or basic metabolic panel (BMP), often are ordered. The CBC may disclose anemia that could be contributing to the patient’s dyspnea, although anemia itself does not appear to affect exacerbation risk.26 The CBC also may show leukocytosis if there is an infectious process present, but this is a nonspecific finding. The BMP is useful for evaluating the patient’s bicarbonate, which may be low in the setting of respiratory acidosis.
Further evaluation of the patient’s acid-base state can be achieved with blood gas analyses. Guidelines from the Global Initiative for Chronic Obstructive Lung Disease (GOLD) recommend obtaining serial arterial blood gases (ABG) in patients with severe exacerbations, but this is probably unnecessary in patients with less severe symptoms.13,27 In fact, a combination of continuous pulse oximetry and venous blood gases (VBG) may be sufficient to monitor oxygenation status in milder exacerbations. One meta-analysis showed acceptable agreement between oxygen saturation readings from pulse oximeters and the arterial oxygen pressure as determined by ABG when the patient’s oxygen saturation was above 70%.28
Two other studies showed that a partial pressure of carbon dioxide (pCO2) greater than 45 mmHg on VBG identifies hypercarbia, which was defined as an arterial pCO2 greater than 50 mmHg, with perfect sensitivity, suggesting that the VBG can be used to screen for significant hypercarbia.
However, because arterial and venous pCO2 values differ substantially, an ABG would be required to evaluate the specific level of arterial pCO2 after a screening VBG identifies that some level of hypercarbia exists.29,30 The VBG also gives a pH and bicarbonate that are similar to those obtained from arterial blood.31
It is important to remember that laboratory values, while helpful, provide only part of the picture. Key management decisions, such as initiating NIV or intubating the patient, should take into account the patient’s overall clinical status and not be driven solely by the patient’s blood gases.27
If there is concern for a cardiac cause of the patient’s symptoms based on the medical history and clinical presentation, it may be reasonable to obtain an electrocardiogram (ECG), cardiac biomarkers, or brain natriuretic peptide. Guidelines from the National Institute for Health and Care Excellence (NICE) in the United Kingdom recommend an ECG for all patients.32 In addition to showing ischemic changes, the ECG will detect cardiac rhythm disturbances. Arrhythmias, including atrial fibrillation, atrial flutter, and ventricular tachycardia, are observed more commonly in patients with COPD. There is some suggestion that exacerbations may incite multifocal atrial tachycardia. The occurrence of arrhythmias during an exacerbation has been associated with higher mortality.33,34 Elevated cardiac biomarkers in the setting of a COPD exacerbation, even in the absence of an acute coronary syndrome, also are associated with a higher 30-day mortality.35
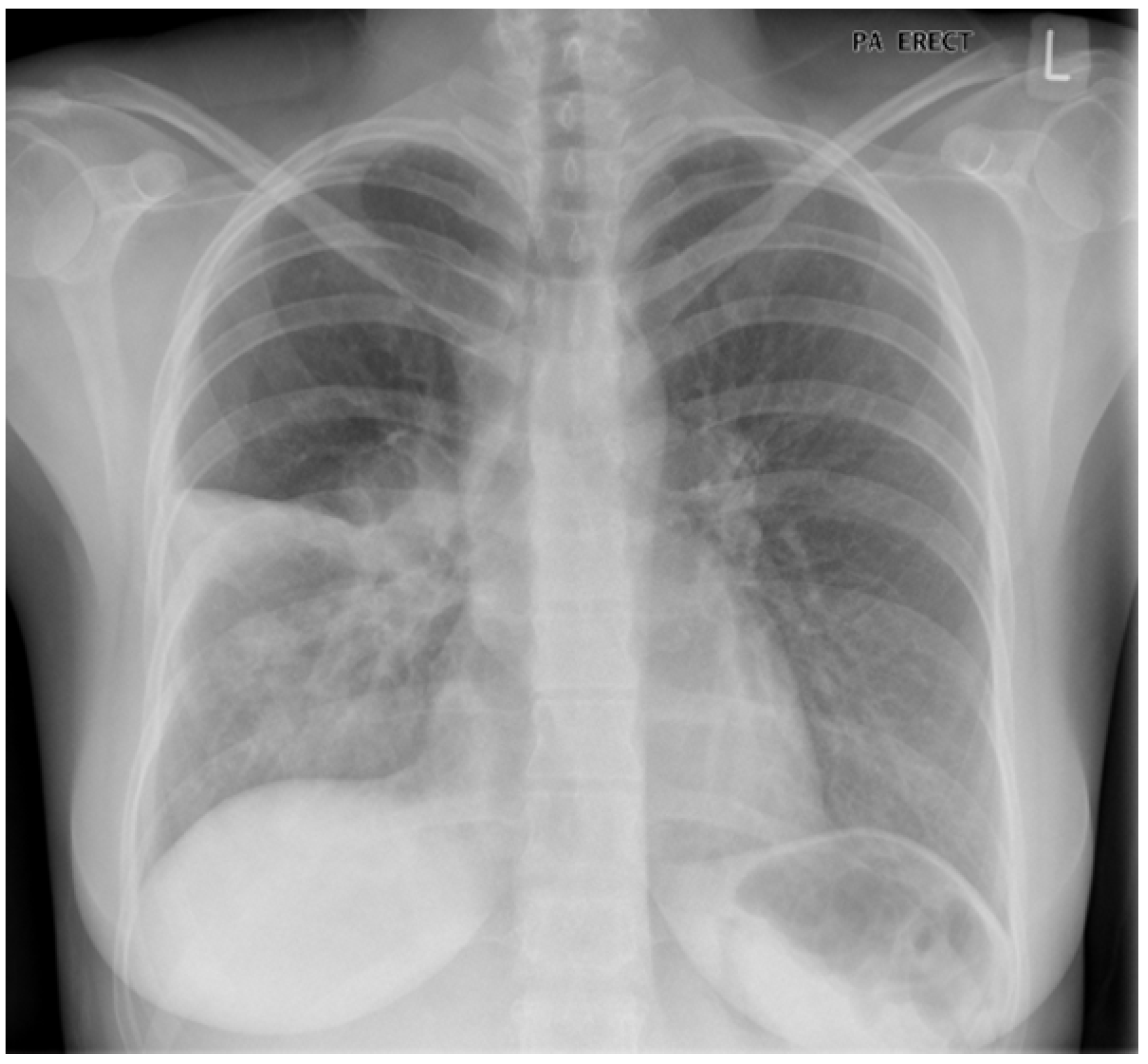
Chest radiographs often are ordered when a patient presents with symptoms suggestive of a COPD exacerbation.36 Radiographs are particularly useful in equivocal cases because they can provide strong evidence for alternate etiologies, such as pulmonary edema and pleural effusions in a patient with a heart failure exacerbation, or an air-filled pleural space in a patient with pneumothorax. As previously discussed, some COPD exacerbations are caused by bacteria, and the chest radiograph can provide evidence to support this diagnosis in the form of a lobar consolidation. Figure 1 demonstrates a chest radiograph from a COPD patient with a consolidation suggesting a lobar pneumonia.37 Further laboratory workup of a patient with a COPD exacerbation may include inflammatory markers to guide treatment. This will be discussed later.
Figure 1. COPD Chest X-Ray |
 |
|
Image used with permission from: Dr. John Hurst, University College London, United Kingdom |
Differential Diagnosis
A history of COPD exacerbations is a key indication that the patient may be experiencing a recurrent acute exacerbation. However, other causes of dyspnea that present similarly should always be considered. The differential diagnosis for a patient presenting with dyspnea is extensive and includes some critical diagnoses that can be fatal if not promptly identified and treated. Myocardial infarction and acute heart failure are two important considerations because, as previously discussed, coronary artery disease and heart failure are frequent comorbidities among COPD patients.7,38 Pneumothorax also is a notable cause of acute dyspnea in patients with COPD.39 Another consideration in the differential diagnosis is pulmonary embolism (PE). Patients with severe COPD are at higher risk for developing PE, which can present similarly to an acute COPD exacerbation.40 This means that correctly diagnosing PE may be more challenging in this cohort.41 It may be appropriate to strongly consider PE in a patient with pleuritic chest pain, no clear infectious symptoms, or failure to improve with typical COPD therapy.27,42 A high-sensitivity D-dimer is recommended as a screening test, and those with a value above the threshold established by the test manufacturer should undergo imaging with computed tomography (CT) pulmonary angiography to detect pulmonary emboli or duplex ultrasound to detect thrombi in the leg veins.
Management
Treatment for COPD exacerbations generally includes supplemental oxygen, bronchodilators, systemic steroids, and antibiotics. Key treatment points discussed in the following sections are summarized in Table 1.
Table 1. Key Treatment Points |
||
Treatment |
Mechanism of Action |
Key Points |
|
Supplemental oxygen |
Improves hypoxemia |
|
|
Bronchodilators |
Produce bronchodilation by relaxing smooth muscle in bronchi, effect lasts four to six hours67 |
|
|
Steroids |
Reduce airway inflammation and enhance bronchodilation74 |
|
|
Antibiotics |
Treat underlying bacterial infections |
|
|
SpO2 = oxygen saturation; NIV = noninvasive ventilation; SABAs = short-acting beta-2 agonists; MDI = metered dose inhaler; PO = by mouth; IV = intravenous |
||
Oxygenation
Assessment of the patient’s airway patency and oxygenation status is the most important first step. Patients who are hypoxic should be given supplemental oxygen. Usually, this is accomplished with a nasal cannula or non-rebreather mask connected to an external oxygen source. Evidence shows that both hypoxia and hyperoxia are harmful in COPD, and, therefore, the literature currently recommends targeting an oxygen saturation of 88% to 92%.43
Traditional teaching cautioned against the use of high supplemental oxygen concentrations because of the belief that this would suppress the hypoxic respiratory drive in the COPD patient and potentially lead to apnea. Excessive oxygen administration can lead to hypercapnia and, therefore, should be avoided. However, the primary driving force behind this effect is not thought to be suppression of the respiratory drive. Instead, reversal of hypoxic vasoconstriction and increased delivery of oxygenated air to dead-space areas of the lung are thought to be the driving forces.44
For patients who require greater respiratory support but not endotracheal intubation, NIV with continuous positive airway pressure (CPAP) or bilevel positive airway pressure (BiPAP) is indicated. Specifically, guidelines recommend NIV for patients who are acidemic, who are in respiratory distress, or who exhibit hypoxia refractory to supplemental oxygen.13 Using NIV in these patients may help to avoid intubation, and is associated with shorter lengths of stay and improved mortality.45
Guidelines from the British Thoracic Society recommend initiating BiPAP with an inspiratory positive airway pressure (IPAP) of about 15 cm H2O and an expiratory pressure of about 3 cm H2O. They also recommend gradually increasing the IPAP to 20 to 30 cm H2O, depending on the patient’s clinical status, respiratory rate, and blood gas values.46 However, note that caution is advised when raising the IPAP above 20 cm H2O because of the risk of gastric distention at these elevated pressures.47
Avoidance of intubation is a notable benefit of NIV associated with lower mortality, shorter length of stay, and less cost compared to invasive mechanical ventilation.48 Some novel therapies are under investigation to assist patients failing NIV in an attempt to improve their respiratory status and prevent intubation. One large, prospective study examined the use of a gaseous mixture of helium and oxygen. This mixture, commonly known as heliox, did not improve the NIV failure rate compared to oxygen alone, although it did show beneficial effects on respiratory rate and pCO2.49 Another rescue strategy under investigation is veno-venous extracorporeal carbon dioxide removal. There is some evidence to suggest that initiating this therapy in patients who are failing BiPAP may reduce the degree of hypercarbia and help to avoid intubation. However, extracorporeal carbon dioxide removal is associated with serious complications, including life-threatening bleeding.50 Guidelines from the British Thoracic Society caution that this therapy is experimental and requires specialized training to implement.46 It is not included in the GOLD guidelines.13
Despite its clear benefits in COPD exacerbations, disadvantages to NIV include the discomfort of the tight-fitting mask and the limitations on oral intake and speaking while undergoing treatment. High-flow nasal cannula (HFNC), which provides humidified air at up to 60 liters per minute through nasal prongs, is a proposed alternative to NIV.51 Compared to NIV modalities such as BiPAP, HFNC is associated with similar intubation rates and short-term mortality in COPD exacerbations.52-54 It also has been shown to be superior to standard nasal cannula and approximately equal to NIV in improvement of hypercarbia, although there are conflicting findings in the literature.52,54-57 The ability of HFNC to flush air forcefully from the anatomical dead space of the upper airway in a flow rate-dependent manner appears to be the primary mechanism behind this reduction in the arterial carbon dioxide pressure.58,59 Studies also indicate that HFNC is better tolerated than BiPAP and even more comfortable than the standard nasal cannula.52,56,57 The comparable efficacy and superior tolerability suggest that HFNC could be an acceptable alternative if the patient has difficulty tolerating NIV.
Although avoiding intubation is preferred, sometimes intubation and mechanical ventilation are necessary in patients experiencing severe COPD exacerbations. The alterations in respiratory physiology that characterize COPD patients at baseline have significant implications for ventilator management. The central issue is dynamic hyperinflation, whereby inspired air becomes trapped in the lungs because of the great resistance to air flow in the constricted and inflamed bronchioles. This process causes the end-expiratory volume to increase with ventilator-delivered breaths. The result is the development of intrinsic positive end-expiratory pressure, which is reflected by increasing plateau pressures. The higher lung volumes created by this dynamic hyperinflation make it harder for the patient to draw spontaneous breaths, decrease cardiac output, and increase the risk of pneumothorax.
There are two primary strategies to mitigate dynamic hyperinflation. The first strategy involves the use of steroids and bronchodilators to open the airways and reduce the expiratory obstruction, allowing more air to exit the lungs. The second way to reduce dynamic hyperinflation is to increase the amount of time spent in expiration, which can be done in the mechanically ventilated patient either by decreasing the respiratory rate or adjusting the inspiratory to expiratory (I:E) ratio. Some degree of hypercapnia and acidosis is acceptable in patients who maintain a respiratory acidosis at baseline, as attempting to normalize the patient’s blood gas values may make successfully extubating the patient more challenging.60
Prone positioning has been studied as an intervention in patients who require mechanical ventilation as a result of a COPD exacerbation. This method has been shown to improve mortality in patients with acute respiratory distress syndrome.61 Although there is no evidence of a similar mortality benefit in COPD exacerbations, prone positioning does appear to enhance oxygenation and reduce dynamic hyperinflation.62-64 However, the few studies examining this intervention were small, so prone positioning in COPD exacerbations likely will require further study to clarify its potential benefits.
Bronchodilators
Short-acting beta-2 agonists (SABAs), such as albuterol, are recommended as first-line bronchodilators for COPD exacerbations.13 Regarding the choice between nebulizers and metered dose inhalers (MDI) for bronchodilator delivery, there is no high-quality evidence that favors one mode over the other.65 However, for practical purposes, the nebulizer system may be more efficient because it can be worn as a mask and does not require patient operation. Doses commonly used are 2.5 mg and 5 mg. One study that compared these two doses of nebulized albuterol for treatment of mild-to-moderate exacerbations in inpatients found no difference in length of stay or time until respiratory improvement between the two groups, suggesting that the higher dose does not offer additional benefit.66
Be aware of the adverse effects of SABAs, which include tachycardia, palpitations, hyperglycemia, hypokalemia, and tremor. Nebulization of SABAs is associated with a higher likelihood of adverse effects than drug delivery by MDI.67 Adding a short-acting anticholinergic agent, such as ipratropium bromide, has not been shown to enhance the bronchodilator effect or prevent hospitalization.68,69 However, GOLD guidelines still recommend combining SABAs and anticholinergic drugs in severe exacerbations.13
Magnesium
Sometimes magnesium is used along with bronchodilators in COPD exacerbations, and it can be administered either intravenously or by nebulization. There is some evidence that intravenous magnesium sulfate modestly enhances air flow in some patients when used adjunctively with SABAs. However, the addition of magnesium has not been shown to affect patient-centered outcomes, such as need for admission, length of stay, and mortality.70-72 Nebulized magnesium has not been shown to be effective in improving lung function or avoiding hospital admission.73 Magnesium is not included as a recommended adjuvant treatment in either the GOLD or NICE guidelines.13,32
Corticosteroids
Systemic corticosteroids act to both reduce airway inflammation and enhance bronchodilation.74 They have been shown to reduce both the risk of treatment failure and length of hospitalization during a COPD exacerbation, and they are associated with accelerated improvement in lung function.75,76
Patients who can tolerate oral intake, including those who require hospital admission, can be given steroids by mouth because there is evidence that oral steroids are equally effective to those given parenterally,76,77 and in fact are associated with a shorter length of stay for inpatients.78 Guidelines from the European Respiratory Society and American Thoracic Society (ERS/ATS) affirm this evidence, recommending oral over intravenous steroids when oral intake is tolerated.79 The typical dose of daily oral prednisone ranges from 20 mg to 60 mg.78 Evidence suggests that prednisone doses greater than 80 mg per day do not provide more benefit than lower doses, including in inpatients.80 GOLD guidelines recommend a five-day course of prednisone 40 mg.13
Longer steroid courses only expose the patient to more potential harms without providing any observable benefit.81,82 One of the most clinically relevant short-term adverse effects associated with steroids is hyperglycemia.76 Although the effect of this transient hyperglycemia may be negligible for many patients, it can be problematic in those with diabetes,83 which is estimated to be present in up to 23% of those with COPD.7
When parenteral steroids are provided, either methylprednisolone or dexamethasone often is chosen. In one head-to-head study of ED patients who were admitted to the hospital, methylprednisolone appeared more effective in treating cough, while dexamethasone was more effective for dyspnea and hypoxia. However, these differences appear small, and there is no compelling evidence to recommend one over the other.84
One cohort study examined dosing strategies for patients requiring admission to the ICU as the result of a COPD exacerbation. The researchers found that an initial daily dose of 240 mg or less of methylprednisolone, which is equivalent to 60 mg given every six hours, was associated with a shorter length of stay and less time requiring invasive mechanical ventilation compared to higher doses. This suggests that lower doses are preferred even in critically ill COPD patients, although specific dosing recommendations for this population require further study.85
Antibiotics
Sometimes antibiotics are prescribed in COPD exacerbations. GOLD guidelines recommend antibiotics in three clinical scenarios. These are outlined in Table 2, along with recommended classes of empiric antibiotics.13 In addition, patients with signs of bacterial pneumonia, such as a lobar consolidation on chest X-ray, should receive antibiotics.
Table 2. Antibiotic Recommendations13 |
|
Patients with the indications below can be treated with any one of the three recommended agents. |
|
|
Indications |
Recommended Agents* |
|
|
|
* Other antibiotic classes may be appropriate depending on local and patient-specific resistance factors. |
|
Research shows that antimicrobial therapy is associated with a lower risk of treatment failure in outpatients and patients requiring ICU care. Improved mortality and shorter lengths of stay are additional benefits associated with antibiotic use in ICU patients.86 Sputum cultures for patients treated outside the hospital do not appear to be helpful and are not recommended routinely, unless the patient has risk factors for P. aeruginosa infection, including repeated exacerbations.13,32,87
Although antibiotics are shown to provide some benefit in COPD exacerbations, the indiscriminate and excessive use of antibiotics is a widely recognized problem with significant, deleterious downstream effects. Therefore, there is great incentive to prescribe antibiotics appropriately.88 To this end, there has been interest recently in determining whether inflammatory biomarkers can predict a need for antibiotics in COPD exacerbations. Most investigation to date has focused on C-reactive protein (CRP) and procalcitonin.
CRP is an acute phase protein that is thought to play a part in the inflammatory response to microbial infection by triggering the complement cascade.89 The use of CRP testing to guide antibiotic prescribing for outpatients has been shown to decrease antibiotic use without compromising care.90,91
The other biomarker of interest, procalcitonin, is the precursor to the hormone calcitonin. Although the primary purpose of this pathway is to maintain calcium homeostasis, microbial lipopolysaccharides and inflammatory interleukins can upregulate the production of procalcitonin and, therefore, make it a potential marker for bacterial infection. In clinical practice, procalcitonin often is preferred over CRP for identifying infections.92
Regarding procalcitonin use in COPD exacerbations, some studies have shown an association of procalcitonin with fewer antibiotic prescriptions without adversely affecting outcomes.93-95 However, others have found that procalcitonin does not reduce overall antibiotic exposure,96 and still others have challenged the ability of these biomarkers to differentiate bacterial from viral infections in COPD exacerbations.97,98 Because of these conflicting findings, GOLD neither endorses nor discourages the use of biomarkers in prescribing decisions.13 The role of these biomarkers in guiding treatment decisions in COPD exacerbations likely will require more study and clarification.
Exacerbation Prevention
GOLD guidelines recommend optimizing outpatient therapy after an exacerbation in an effort to prevent subsequent exacerbations.13 Whether this is done in the ED, on hospital discharge, or by the patient’s primary care physician may vary by provider preference and practice location. Combination inhalers have become common and generally are more effective than single-medication inhalers. Specifically, the inhaler most effective at reducing the risk of future exacerbations appears to be a long-acting beta-2 agonist (LABA) combined with a long-acting muscarinic antagonist (LAMA).99 Inhaled corticosteroids (ICS) are used frequently in combination with LABAs. Although this combination improves quality of life,100 the ICS component appears to increase the risk for developing pneumonia.101,102 Regarding monotherapy, ERS/ATS guidelines indicate a preference for LAMAs over LABAs.103
Disposition
Disposition decisions are relatively straightforward regarding patients on either end of the severity spectrum. Addressing those who reside between the extremes becomes more challenging. To inform those decisions, it is important to note which clinical features are associated with worse outcomes. Patients with comorbidities, such as heart failure and diabetes, tend to have higher readmission and mortality rates.104,105 A visible consolidation on the chest radiograph during a COPD exacerbation is associated with more frequent administration of supplemental oxygen, greater degree of acidemia, longer hospitalization, and higher mortality. Not surprisingly, these patients frequently also have purulent sputum and receive antibiotics.106 Selected indications for admission from GOLD and NICE guidelines are combined in Table 3. They include some of the high-risk factors discussed previously.
Table 3. Selected Indications for Hospitalization13,32 |
|
Symptoms |
|
|
Signs |
|
|
Diagnostic studies |
|
|
Medical history |
|
|
Social factors |
|
|
Emergency department course |
|
Several COPD-specific clinical decision rules in the literature are intended to help this decision-making process. One rule, called DECAF, combines dyspnea severity, eosinopenia, radiographic consolidation, acidemia, and atrial fibrillation to estimate both in-hospital and 30-day mortality. Dyspnea is graded based on the extended Medical Research Council dyspnea score.107 It was shown to be a better predictor of in-hospital and 30-day mortality than prior decision rules, such as CURB-65, BAP-65, and CAPS,108 although this conclusion has been challenged recently.109
One caveat to DECAF is that it was studied only in the inpatient population and is meant to help identify patients who may be safely discharged early from an inpatient unit. Therefore, it is unclear how this decision rule applies to patients in the ED.
To address this gap, the Ottawa COPD Risk Scale (OCRS) was designed specifically for aiding disposition decisions in the ED. OCRS uses 10 factors to predict the risk of short-term adverse events, and is meant to help determine which patients are sufficiently low-risk that they can be discharged safely from the ED with outpatient follow-up.110,111 The authors declined to recommend a cutoff value that would differentiate patients who should be admitted from those who are safe for discharge, deferring to clinician judgment. Remember that decision rules are meant to be factored into the milieu of clinical data that inform decision-making and are not intended to serve as stand-alone deciding factors.
Summary
Acute exacerbations frequently prompt patients with COPD to present to their provider, so it is crucial for physicians to understand how to assess and treat these patients effectively. Viral and bacterial respiratory infections are the most common causes of exacerbations, but nonadherence to home medications also is a significant contributing factor. Because comorbidities are common in those with COPD, consider other serious causes for the patient’s symptoms, such as myocardial infarction, acute heart failure, and pulmonary embolism.
In the evaluation of patients with milder symptoms, continuous pulse oximetry and venous blood gases may be sufficient to monitor oxygenation status without an arterial blood gas. Even though venous and arterial carbon dioxide pressures differ, evidence suggests that a venous blood gas can be used to screen for hypercarbia.
Treatment of COPD exacerbations often involves supplemental oxygen, bronchodilators, and systemic corticosteroids. Titrate the supplemental oxygen to achieve an SpO2 of 88% to 92%. For patients who require more respiratory support than a nasal cannula or non-rebreather mask, NIV has been shown to reduce intubation rates and mortality. Patients who do not improve on NIV or cannot tolerate it should be intubated, although there is some evidence to suggest that high-flow nasal cannula is tolerated better and can improve hypercarbia and help to avoid intubation.
Ventilator management of COPD patients is focused on reducing intrinsic positive airway pressure and dynamic hyperinflation. This can be accomplished by administering medications to reduce airway inflammation and bronchoconstriction, reducing the respiratory rate, and adjusting the I:E ratio on the ventilator to allow for greater expiratory time.
Short-acting bronchodilators, such as albuterol, can be delivered effectively by either nebulizer or MDI. An outpatient steroid course of about five days is appropriate for patients who are discharged. Use steroids with caution, especially in patients with a history of diabetes, because they can cause hyperglycemia.
Antibiotics generally are recommended when there are signs of bacterial infection, such as purulent sputum, and in patients requiring mechanical ventilation. Inflammatory markers, such as CRP and procalcitonin, are being investigated as indicators for antimicrobial therapy in COPD exacerbations, but there is not yet enough evidence to recommend their routine use for this purpose.
Guidelines recommend assessing the patient’s long-term regimen after an exacerbation and making adjustments as needed to reduce the risk of subsequent exacerbations. Patients with severe symptoms, those who do not respond appropriately to treatment in the ED, and those with significant comorbidities should be admitted, while it may be reasonable to allow those with milder symptoms and good social support to go home with close follow-up. Clinical decision rules can be used to assist disposition decisions.
References
- Sullivan J, Pravosud V, Mannino DM, et al. National and state estimates of COPD morbidity and mortality – United States, 2014-2015. Chronic Obstr Pulm Dis 2018;5:324-333.
- López-Campos JL, Tan W, Soriano JB. Global burden of COPD. Respirology 2016;21:14-23.
- Kochanek KD, Murphy SL, Xu J, Arias E. Deaths: Final data for 2017. Nat Vital Stat Rep 2019;68:1-77.
- Ford ES, Murphy LB, Khavjou O, et al. Total and state-specific medical and absenteeism costs of COPD among adults aged ≥ 18 years in the United States for 2010 and projections through 2020. Chest 2015;147:31-45.
- Akinbami LJ, Liu X. Chronic obstructive pulmonary disease among adults aged 18 and over in the United States, 1998-2009. NCHS Data Brief 2011;63:1-8.
- Riley CM, Sciurba FC. Diagnosis and outpatient management of chronic obstructive pulmonary disease: A review. JAMA 2019;321:786-797.
- Putcha N, Drummond MB, Wise RA, Hansel NN. Comorbidities and chronic obstructive pulmonary disease: Prevalence, influence on outcomes, and management. Semin Respir Crit Care Med 2015;36:575-591.
- Rui P, Kang K. National hospital ambulatory medical care survey: 2017 emergency department summary tables. National Center for Health Statistics. https://www.cdc.gov/nchs/data/nhamcs/web_tables/2017_ed_web_tables-508.pdf
- Hurst JR, Vestbo J, Anzueto A, et al. Susceptibility to exacerbation in chronic obstructive pulmonary disease. N Engl J Med 2010;363:1128-1138.
- Han MK, Quibrera PM, Carretta EE, et al. Frequency of exacerbations in patients with chronic obstructive pulmonary disease: An analysis of the SPIROMICS cohort. Lancet Respir Med 2017;5:619-626.
- Rothnie KJ, Müllerová H, Smeeth L, Quint JK. Natural history of chronic obstructive pulmonary disease exacerbations in a general practice-based population with chronic obstructive pulmonary disease. Am J Respir Crit Care Med 2018;198:464-471.
- Halpin DMG, Decramer M, Celli BR, et al. Effect of a single exacerbation on decline in lung function in COPD. Respir Med 2017;128:85-91.
- Global Initiative for Chronic Obstructive Lung Disease, Inc. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: 2020 report. https://goldcopd.org/wp-content/uploads/2019/12/GOLD-2020-FINAL-ver1.2-03Dec19_WMV.pdf
- Reilly JJ, Silverman EK, Shapiro SD. Chronic obstructive pulmonary disease. In: Jameson JL, Fauci A, Kasper DL, et al, eds. Harrison’s Principles of Internal Medicine. 19th ed. McGraw-Hill Education; 2015:1700-1707.
- Shujaat A, Minkin R, Eden E. Pulmonary hypertension and chronic cor pulmonale in COPD. Int J Chron Obstruct Pulmon Dis 2007;2:273-282.
- Wedzicha JA, Seemungal TAR. COPD exacerbations: Defining their cause and prevention. Lancet 2007;370:786-796.
- Brill SE, Wedzicha JA. Oxygen therapy in acute exacerbations of chronic obstructive pulmonary disease. Int J Chron Obstruct Pulmon Dis 2014;9:1241-1252.
- Hogea SP, Tudorache E, Fildan AP, et al. Risk factors of chronic obstructive pulmonary disease exacerbations. Clin Respir J 2020;14:183-197.
- Kwak HJ, Park DW, Kim JE, et al. Prevalence and risk factors of respiratory viral infections in exacerbations of chronic obstructive pulmonary disease. Tohoku J Exp Med 2016;240:131-139.
- Vestbo J, Anderson JA, Calverley PMA, et al. Adherence to inhaled therapy, mortality and hospital admission in COPD. Thorax 2009;64:939-943.
- Liang B, Wang M, Yi Q, Feng Y. Association of gastroesophageal reflux disease risk with exacerbations of chronic obstructive pulmonary disease. Dis Esophagus 2013;26:557-560.
- Bates CG. Chronic obstructive pulmonary disease. In: Tintinalli JE, Ma OJ, Yealy D, et al, eds. Emergency Medicine: A Comprehensive Study Guide. 9th ed. McGraw-Hill; 2020:467-471.
- Hamzaoui O, Monnet X, Teboul JL. Pulsus paradoxus. Eur Respir J 2013;42:1696-1705.
- Kelly AM, Holdgate A, Keijzers G, et al. Epidemiology, treatment, disposition and outcome of patients with acute exacerbation of COPD presenting to emergency departments in Australia and South East Asia: An AANZDEM study. Respirology 2018;23:681-686.
- Zoorob RJ, Campbell JS. Acute dyspnea in the office. Am Fam Physician 2003;68:1803-1811.
- Putcha N, Fawzy A, Paul GG, et al. Anemia and adverse outcomes in a chronic obstructive pulmonary disease population with a high burden of comorbidities. An analysis from SPIROMICS. Ann Am Thorac Soc 2018;15:710-717.
- Tabatabai RR, Gruber PF. Chronic obstructive pulmonary disease. In: Walls RM, Hockberger R, Gausche-Hill M, et al, eds. Rosen’s Emergency Medicine: Concepts and Clinical Practice. 9th ed. Elsevier; 2018:848-856.
- Jensen LA, Onyskiw JE, Prasad NG. Meta-analysis of arterial oxygen saturation monitoring by pulse oximetry in adults. Heart Lung 1998;27:387-408.
- Kelly A, Kyle E, McAlpine R. Venous pCO(2) and pH can be used to screen for significant hypercarbia in emergency patients with acute respiratory disease. J Emerg Med 2002;22:15-19.
- McCanny P, Bennett K, Staunton P, McMahon G. Venous vs arterial blood gases in the assessment of patients presenting with an exacerbation of chronic obstructive pulmonary disease. Am J Emerg Med 2012;30:896-900.
- McKeever TM, Hearson G, Housley G, et al. Using venous blood gas analysis in the assessment of COPD exacerbations: A prospective cohort study. Thorax 2016;71:210-215.
- National Institute for Health and Care Excellence. Chronic obstructive pulmonary disease in over 16s: Diagnosis and management. Published Dec. 5, 2018. Updated July 26, 2019. https://www.nice.org.uk/guidance/ng115
- Goudis CA, Konstantinidis AK, Ntalas IV, Korantzopoulos P. Electrocardiographic abnormalities and cardiac arrhythmias in chronic obstructive pulmonary disease. Int J Cardiol 2015;199:264-273.
- Konecny T, Park JY, Somers KR, et al. Relation of chronic obstructive pulmonary disease to atrial and ventricular arrhythmias. Am J Cardiol 2014;114:272-277.
- Chang CL, Robinson SC, Mills GD, et al. Biochemical markers of cardiac dysfunction predict mortality in acute exacerbations of COPD. Thorax 2011;66:764-768.
- Spece LJ, Donovan LM, Griffith MF, et al. Quality of care delivered to veterans with COPD exacerbation and the association with 30-day readmission and death. COPD 2018;15:489-495.
- Hurst JR. Consolidation and exacerbation of COPD. Med Sci 2018;6:44.
- Canepa M, Straburzynska-Migaj E, Drozdz J, et al. Characteristics, treatments and 1-year prognosis of hospitalized and ambulatory heart failure patients with chronic obstructive pulmonary disease in the European Society of Cardiology Heart Failure Long-Term Registry. Eur J Heart Fail 2018;20:100-110.
- Sahn SA, Heffner JE. Spontaneous pneumothorax. N Engl J Med 2000;342:868-874.
- BØrvik T, Brækkan SK, Enga K, et al. COPD and risk of venous thromboembolism and mortality in a general population. Eur Respir J 2016;47:473-481.
- Torres-Macho J, Mancebo-Plaza AB, Crespo-Giménez A, et al. Clinical features of patients inappropriately undiagnosed of pulmonary embolism. Am J Emerg Med 2013;31:1646-1650.
- Aleva FE, Voets LWLM, Simons SO, et al. Prevalence and localization of pulmonary embolism in unexplained acute exacerbations of COPD: A systematic review and meta-analysis. Chest 2017;151:544-554.
- Austin MA, Wills KE, Blizzard L, et al. Effect of high flow oxygen on mortality in chronic obstructive pulmonary disease patients in prehospital setting: Randomised controlled trial. BMJ 2010;341:c5462.
- Abdo WF, Heunks LMA. Oxygen-induced hypercapnia in COPD: Myths and facts. Crit Care 2012;16:323.
- Osadnik CR, Tee VS, Carson-Chahhoud KV, et al. Non-invasive ventilation for the management of acute hypercapnic respiratory failure due to exacerbation of chronic obstructive pulmonary disease. Cochrane Database Syst Rev 2017;7:CD004104.
- Davidson AC, Banham S, Elliott M, et al. BTS/ICS guideline for the ventilatory management of acute hypercapnic respiratory failure in adults. Thorax 2016;71(Suppl 2):ii1-35.
- Seigel TA. Mechanical ventilation and noninvasive ventilatory support. In: Walls RM, Hockberger R, Gausche-Hill M, et al, eds. Rosen’s Emergency Medicine: Concepts and Clinical Practice. 9th ed. Elsevier; 2018:25-33.
- Tsai CL, Lee WY, Delclos GL, et al. Comparative effectiveness of noninvasive ventilation vs invasive mechanical ventilation in chronic obstructive pulmonary disease patients with acute respiratory failure. J Hosp Med 2013;8:165-172.
- Jolliet P, Ouanes-Besbes L, Abroug F, et al. A multicenter randomized trial assessing the efficacy of helium/oxygen in severe exacerbations of chronic obstructive pulmonary disease. Am J Respir Crit Care Med 2017;195:871-880.
- Morales-Quinteros L, Del Sorbo L, Artigas A. Extracorporeal carbon dioxide removal for acute hypercapnic respiratory failure. Ann Intensive Care 2019;9:79.
- Pisani L, Astuto M, Prediletto I, Longhini F. High flow through nasal cannula in exacerbated COPD patients: A systematic review. Pulmonology 2019;25:348-354.
- Sun J, Li Y, Ling B, et al. High flow nasal cannula oxygen therapy versus non-invasive ventilation for chronic obstructive pulmonary disease with acute-moderate hypercapnic respiratory failure: An observational cohort study. Int J Chron Obstruct Pulmon Dis 2019;14:1229-1237.
- Kyu Lee M, Choi J, Park B, et al. High flow nasal cannulae oxygen therapy in acute-moderate hypercapnic respiratory failure. Clin Respir J 2018;12:2046-2056.
- Doshi P, Whittle JS, Bublewicz M, et al. High-velocity nasal insufflation in the treatment of respiratory failure: A randomized clinical trial. Ann Emerg Med 2018;72:73-83.e5.
- Pilcher J, Eastlake L, Richards M, et al. Physiological effects of titrated oxygen via nasal high-flow cannulae in COPD exacerbations: A randomized controlled cross-over trial. Respirology 2017;22:1149-1155.
- Longhini F, Pisani L, Lungu R, et al. High-flow oxygen therapy after noninvasive ventilation interruption in patients recovering from hypercapnic acute respiratory failure: A physiological crossover trial. Crit Care Med 2019;47:e506-e511.
- McKinstry S, Singer J, Baarsma JP, et al. Nasal high-flow therapy compared with non-invasive ventilation in COPD patients with chronic respiratory failure: A randomized controlled cross-over trial. Respirology 2019;24:1081-1087.
- Möller W, Celik G, Feng S, et al. Nasal high flow clears anatomical dead space in upper airway models. J Appl Physiol (1985) 2015;118:1525-1532.
- Drake MG. High-flow nasal cannula oxygen in adults: An evidence-based assessment. Ann Am Thorac Soc 2018;15:145-155.
- Ward NS, Dushay KM. Clinical concise review: Mechanical ventilation of patients with chronic obstructive pulmonary disease. Crit Care Med 2008;36:1614-1619.
- Guérin C, Reignier J, Richard J, et al. Prone positioning in severe acute respiratory distress syndrome. N Engl J Med 2013;368:2159-2168.
- Reignier J, Lejeune O, Renard B, et al. Short-term effects of prone position in chronic obstructive pulmonary disease patients with severe acute hypoxemic and hypercapnic respiratory failure. Intensive Care Med 2005;31:1128-1131.
- Mentzelopoulos SD, Zakynthinos SG, Roussos C, et al. Prone position improves lung mechanical behavior and enhances gas exchange efficiency in mechanically ventilated chronic obstructive pulmonary disease patients. Anesth Analg 2003;96:1756-1767.
- Mentzelopoulos SD, Roussos C, Zakynthinos SG. Prone position improves expiratory airway mechanics in severe chronic bronchitis. Eur Respir J 2005;25:259-268.
- van Geffen WH, Douma WR, Slebos DJ, Kerstjens HA. Bronchodilators delivered by nebuliser versus pMDI with spacer or DPI for exacerbations of COPD. Cochrane Database Syst Rev 2016;(8):CD011826.
- Nair S, Thomas E, Pearson SB, Henry MT. A randomized controlled trial to assess the optimal dose and effect of nebulized albuterol in acute exacerbations of COPD. Chest 2005;128:48-54.
- Cazzola M, Page CP, Rogliani P, Matera MG. β2-agonist therapy in lung disease. Am J Respir Crit Care Med 2013;187:690-696.
- McCrory DC, Brown CD. Anti-cholinergic bronchodilators versus beta2-sympathomimetic agents for acute exacerbations of chronic obstructive pulmonary disease. Cochrane Database Syst Rev 2002;(4):CD003900.
- Beltaief K, Msolli MA, Zorgati A, et al. Nebulized terbutaline and ipratropium bromide versus terbutaline alone in acute exacerbation of chronic obstructive pulmonary disease requiring noninvasive ventilation: A randomized double-blind controlled trial. Acad Emerg Med 2019;26:434-442.
- Shivanthan MC, Rajapakse S. Magnesium for acute exacerbation of chronic obstructive pulmonary disease: A systematic review of randomised trials. Ann Thorac Med 2014;9:77-80.
- Mukerji S, Shahpuri B, Clayton-Smith B, et al. Intravenous magnesium sulphate as an adjuvant therapy in acute exacerbations of chronic obstructive pulmonary disease: A single centre, randomised, double-blinded, parallel group, placebo-controlled trial: A pilot study. N Z Med J 2015;128:34-42.
- Solooki M, Miri M, Mokhtari M, et al. Magnesium sulfate in exacerbations of COPD in patients admitted to internal medicine ward. Iran J Pharm Res 2014;13:1235-1239.
- Edwards L, Shirtcliffe P, Wadsworth K, et al. Use of nebulised magnesium sulphate as an adjuvant in the treatment of acute exacerbations of COPD in adults: A randomised double-blind placebo-controlled trial. Thorax 2013;68:338-343.
- MacIntyre NR. Corticosteroid therapy and chronic obstructive pulmonary disease. Respir Care 2006;51:289-296.
- Niewoehner DE, Erbland ML, Deupree RH, et al. Effect of systemic glucocorticoids on exacerbations of chronic obstructive pulmonary disease: Department of Veterans Affairs Cooperative Study Group. N Engl J Med 1999;340:1941-1947.
- Walters JA, Tan DJ, White CJ, et al. Systemic corticosteroids for acute exacerbations of chronic obstructive pulmonary disease. Cochrane Database Syst Rev 2014;(9):CD001288.
- de Jong YP, Uil SM, Grotjohan HP, et al. Oral or IV prednisolone in the treatment of COPD exacerbations: A randomized, controlled, double-blind study. Chest 2007;132:1741-1747.
- Lindenauer PK, Pekow PS, Lahti MC, et al. Association of corticosteroid dose and route of administration with risk of treatment failure in acute exacerbation of chronic obstructive pulmonary disease. JAMA 2010;303:2359-2367.
- Wedzicha JA, Miravitlles M, Hurst JR, et al. Management of COPD exacerbations: A European Respiratory Society/American Thoracic Society guideline. Eur Respir J 2017;49:1600791.
- Cheng T, Gong Y, Guo Y, et al. Systemic corticosteroid for COPD exacerbations, whether the higher dose is better? A meta-analysis of randomized controlled trials. Clin Respir J 2013;7:305-318.
- Leuppi JD, Schuetz P, Bingisser R, et al. Short-term vs conventional glucocorticoid therapy in acute exacerbations of chronic obstructive pulmonary disease: The REDUCE randomized clinical trial. JAMA 2013;309:2223-2231.
- Walters JA, Tan DJ, White CJ, Wood-Baker R. Different durations of corticosteroid therapy for exacerbations of chronic obstructive pulmonary disease. Cochrane Database Syst Rev 2018;3:CD006897.
- Aldibbiat AM, Al-Sharefi A. Do benefits outweigh risks for corticosteroid therapy in acute exacerbation of chronic obstructive pulmonary disease in people with diabetes mellitus? Int J Chron Obstruct Pulmon Dis 2020;15:567-574.
- Ardestani ME, Kalantary E, Samaiy V, Taherian K. Methyl prednisolone vs dexamethasone in management of COPD exacerbation; A randomized clinical trial. Emerg (Tehran) 2017;5:e35.
- Kiser TH, Allen RR, Valuck RJ, et al. Outcomes associated with corticosteroid dosage in critically ill patients with acute exacerbations of chronic obstructive pulmonary disease. Am J Respir Crit Care Med 2014;189:1052-1064.
- Vollenweider DJ, Frei A, Steurer-Stey CA, et al. Antibiotics for exacerbations of chronic obstructive pulmonary disease. Cochrane Database Syst Rev 2018;10:CD010257.
- Ra SW, Kwon YS, Yoon SH, et al. Sputum bacteriology and clinical response to antibiotics in moderate exacerbation of chronic obstructive pulmonary disease. Clin Respir J 2018;12:1424-1432.
- Centers for Disease Control and Prevention. Antibiotic Use in the United States, 2018 Update: Progress and Opportunities. Atlanta, GA: US Department of Health and Human Services, CDC; 2019.
- Sproston NR, Ashworth JJ. Role of C-reactive protein at sites of inflammation and infection. Front Immunol 2018;9:754.
- Butler CC, Gillespie D, White P, et al. C-reactive protein testing to guide antibiotic prescribing for COPD exacerbations. N Engl J Med 2019;381:111-120.
- Strykowski DF, Nielsen ABS, Llor C, et al. An intervention with access to C-reactive protein rapid test reduces antibiotic overprescribing in acute exacerbations of chronic bronchitis and COPD. Fam Pract 2015;32:395-400.
- Vijayan AL, Vanimaya, Ravindran S, et al. Procalcitonin: A promising diagnostic marker for sepsis and antibiotic therapy. J Intensive Care 2017;5:51.
- Mathioudakis AG, Chatzimavridou-Grigoriadou V, Corlateanu A, Vestbo J. Procalcitonin to guide antibiotic administration in COPD exacerbations: A meta-analysis. Eur Respir Rev 2017;26:160073.
- Li Z, Yuan X, Yu L, et al. Procalcitonin-guided antibiotic therapy in acute exacerbation of chronic obstructive pulmonary disease: An updated meta-analysis. Medicine (Baltimore) 2019;98:e16775.
- Lin C, Pang Q. Meta-analysis and systematic review of procalcitonin-guided treatment in acute exacerbation of chronic obstructive pulmonary disease. Clin Respir J 2018;12:10-15.
- Daubin C, Valette X, Thiollière F, et al. Procalcitonin algorithm to guide initial antibiotic therapy in acute exacerbations of COPD admitted to the ICU: A randomized multicenter study. Intensive Care Med 2018;44:428-437.
- Kawamatawong T, Apiwattanaporn A, Siricharoonwong W. Serum inflammatory biomarkers and clinical outcomes of COPD exacerbation caused by different pathogens. Int J Chron Obstruct Pulmon Dis 2017;12:1625-1630.
- Chang CH, Tsao KC, Hu HC, et al. Procalcitonin and C-reactive protein cannot differentiate bacterial or viral infection in COPD exacerbation requiring emergency department visits. Int J Chron Obstruct Pulmon Dis 2015;10:767-774.
- Oba Y, Keeney E, Ghatehorde N, Dias S. Dual combination therapy versus long-acting bronchodilators alone for chronic obstructive pulmonary disease (COPD): A systematic review and network meta-analysis. Cochrane Database Syst Rev 2018;12:CD012620.
- Kew KM, Dias S, Cates CJ. Long-acting inhaled therapy (beta-agonists, anticholinergics and steroids) for COPD: A network meta-analysis. Cochrane Database Syst Rev 2014;(3):CD010844.
- Nannini LJ, Lasserson TJ, Poole P. Combined corticosteroid and long-acting beta(2)-agonist in one inhaler versus long-acting beta(2)-agonists for chronic obstructive pulmonary disease. Cochrane Database Syst Rev 2012;(9):CD006829.
- Kew KM, Seniukovich A. Inhaled steroids and risk of pneumonia for chronic obstructive pulmonary disease. Cochrane Database Syst Rev 2014;(3):CD010115.
- Wedzicha JA, Calverley PMA, Albert RK, et al. Prevention of COPD exacerbations: A European Respiratory Society/American Thoracic Society guideline. Eur Respir J 2017;50:1602265.
- Singh JA, Yu S. Utilization due to chronic obstructive pulmonary disease and its predictors: A study using the U.S. National Emergency Department Sample (NEDS). Respir Res 2016;17:1.
- Spece LJ, Epler EM, Donovan LM, et al. Role of comorbidities in treatment and outcomes after chronic obstructive pulmonary disease exacerbations. Ann Am Thorac Soc 2018;15:1033-1038.
- Saleh A, LÓpez-Campos JL, Hartl S, et al. The effect of incidental consolidation on management and outcomes in COPD exacerbations: Data from the European COPD Audit. PLoS One 2015;10:e0134004.
- Steer J, Gibson J, Bourke SC. The DECAF score: Predicting hospital mortality in exacerbations of chronic obstructive pulmonary disease. Thorax 2012;67:970-976.
- Echevarria C, Steer J, Heslop-Marshall K, et al. Validation of the DECAF score to predict hospital mortality in acute exacerbations of COPD. Thorax 2016;71:133-140.
- Shafuddin E, Chang CL, Hancox RJ. Comparing severity scores in exacerbations of chronic obstructive pulmonary disease. Clin Respir J 2018;12:2668-2675.
- Stiell IG, Clement CM, Aaron SD, et al. Clinical characteristics associated with adverse events in patients with exacerbation of chronic obstructive pulmonary disease: A prospective cohort study. CMAJ 2014;186:E193-E204.
- Stiell IG, Perry JJ, Clement CM, et al. Clinical validation of a risk scale for serious outcomes among patients with chronic obstructive pulmonary disease managed in the emergency department. CMAJ 2018;190:E1406-E1413.
People with chronic obstructive pulmonary disease frequently seek care for acute exacerbations, which are associated with significant morbidity and mortality. Therefore, it is crucial for physicians to understand how to assess and treat these patients appropriately.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
