Blunt Abdominal Trauma
May 1, 2021
Reprints
AUTHORS
Andrea L. Kaelin, MD, FAAEM, RDMS, Wright State University, Dayton, OH
Nathan James, MD, Wright State University, Dayton, OH
Andrew Gurtis, MD, Wright State University, Dayton, OH
Adam James, MD, Wright State University, Dayton, OH
PEER REVIEWER
Dennis Hanlon, MD, FAAEM, Vice Chairman, Academics, Department of Emergency Medicine, Allegheny General Hospital, Pittsburgh, PA
EXECUTIVE SUMMARY
- Blunt abdominal trauma accounts for the majority of abdominal injuries seen in the emergency department, with 75% of them occurring secondary to motor vehicle collisions, while fall mechanisms account for only 9%.
- Injuries to the hollow organs, the pancreas, or the kidneys occur less frequently than splenic or hepatic injuries; however, these presentations may be delayed and difficult to detect.
- A complete extended focused assessment with sonography for trauma (eFAST), indicated per American College of Emergency Physicians ultrasound guidelines in both blunt and penetrating trauma of the chest and abdomen, assesses for the presence of intra-abdominal free fluid, pericardial effusion, and appropriate pleural lung sliding bilaterally. The eFAST imaging sequence rapidly assesses hemodynamically unstable patients who could receive immediate life-saving interventions for pneumothorax, hemothorax, pericardial tamponade, or potentially direct the resuscitation to the operating suite if massive intra-abdominal hemorrhage is detected.
- Hemodynamically stable patients with a negative eFAST exam but with abdominal tenderness or evidence of trauma, including seat belt sign, should be evaluated further with a contrast computed tomography scan.
- The Advanced Trauma Life Support table for classifying signs and symptoms of shock now includes an assessment of base excess as an indicator of shock. A venous or arterial blood gas provides this crucial information in addition to an estimated hemoglobin.
- Of the injuries sustained from blunt abdominal trauma, liver injuries are the easiest to identify and manage with nonoperative management, while bowel or hollow organ injuries are the most difficult to identify and manage without surgical intervention.
- Trauma and Injury Severity Score (TRISS) is a scoring system that combines both the anatomical Injury Severity Score (ISS) and physiological Revised Trauma Score (RTS) into one score. It has been proven to be the most effective and validated scoring system for blunt abdominal trauma secondary to falls along with trauma scoring for elderly patients regardless of the type of trauma.
- Sepsis is a common and often debilitating outcome of blunt abdominal trauma. Several studies have identified sepsis as a leading cause for morbidity and mortality in operative and nonoperative abdominal trauma patients. The most common causes of sepsis in blunt abdominal trauma patients often stem from early onset transfusions, postoperative closure methods, damage control surgeries, age, mechanism of injury, and severity of injury pattern.
Blunt abdominal trauma is commonly encountered in any acute care center. Prompt recognition, assessment, diagnostic evaluation, and disposition are critical aspects that must be a part of every clinician’s expertise.
— Ann M. Dietrich, MD, Editor
Trauma patients present anytime, anywhere, to every emergency department, which must balance the duties of resuscitation, evaluation, management of traumatic injuries, and disposition or transfer to a specialized trauma center. In addition, underlying and contributory medical conditions should be considered. The process of emergent evaluation and management of blunt abdominal trauma continually evolves; it requires frequent review and integration of best practices by the emergency medicine physician. For developed countries, trauma leads as the most common cause of death for multiple age groups: pediatric, adolescent, and young and middle adulthood. Although the Advanced Trauma Life Support (ATLS) course provides a foundation for initial management of trauma, the emergency provider must further prepare for the expansive variety of injured patients.
Emergency physicians exude the ability to look beyond algorithms and apply a keen clinical assessment of the entirety of the patient, to include consideration of medical causes and sequelae of blunt abdominal traumatic injuries. The discussion that follows integrates a foundational approach to trauma, key considerations, injury severity scoring, imaging review, special populations, and upcoming modernizations to the process of the best practices for the care of the blunt abdominal trauma patient.
Epidemiology
Trauma is the leading cause of death in developed countries and the highest preventable mortality for productive-aged years of life.1 Motor vehicle collisions (MVCs) are a prominent public health concern and account for the most common trauma-related mortality; the rate of speed and type of impact directly correlate to the rate of mortality. Head-on collisions carry the greatest risk of mortality, while rear impacts carry the lowest risk. Speed also correlates directly with mortality. A speed of 25 mph or less has a mortality rate of 5%, but speeds ranging more than 55 mph carry an 85% risk of mortality.
Efforts to mitigate preventable death and minimize injuries have resulted in improved methods of restraint devices, crumple or impact zones built into vehicular frames, and additional safety through automated vehicular assist control mechanisms for driving at night and in inclement weather. Proper wearing of seat belts alone saves approximately 15,000 lives per year. However, despite additional safety mechanisms, MVCs remain a leading cause of blunt abdominal trauma in the United States. In fact, blunt abdominal trauma accounts for the majority of abdominal injuries seen in the emergency department, with 75% of them occurring secondary to MVCs, while fall mechanisms account for only 9%.2
The peak incidence of blunt abdominal trauma is between the ages of 14 and 30 years, but the highest mortality rate occurs in individuals 55 years of age or older. When blunt abdominal trauma is sustained, only 13% of cases are associated with intra-abdominal injuries, and of those, 25% require surgical intervention.2
Solid organs, primarily the spleen and liver, are the most commonly injured organs and are the most common causes of intra-abdominal hemorrhage. In a study, the combined mortality rate of splenic and liver injuries was lower when compared to penetrating abdominal trauma.3 Exsanguination from penetrating abdominal trauma or sequelae of projectiles serves as the most common cause of mortality; in blunt injury mechanisms, mortality results as a sequela of intra-abdominal abscess or wound infections.
Assessment of injury patterns in penetrating abdominal trauma may be easier than in blunt trauma; however, the skilled physician should identify subtle signs of blunt injuries and manage them while in the emergency department. Injuries to the hollow organs, the pancreas, or the kidneys occur less frequently than splenic or hepatic injuries. However, these presentations may be delayed and difficult to detect. Although less frequent, hollow organ rupture in blunt abdominal trauma may occur secondary to shear injuries of the duodenojejunal flexure and ileocecal junction sites.
Chest injury patterns accompany blunt abdominal trauma in multisystem traumas relative to the mechanism of injury and severity of abdominal insult. Given the extensive and lethal variability of thoracic injury possible in association with blunt abdominal trauma, evaluate for pneumothorax, hemothorax, aortic dissection, and pericardial effusions as well. No significant study to date has identified which lethal chest injury pattern associates greatest with blunt abdominal trauma. Therefore, all must be rapidly considered in the evaluation. Use initial bedside imaging studies in the resuscitation bay, including the plain film trauma single anteroposterior (AP) view of the chest along with point-of-care ultrasound, to quickly identify associated injuries. Consider both modalities complementary to, rather than in lieu of, advanced radiology studies such as computed tomography (CT).
Presentation and Initial Evaluation
Initial Assessment
Prior to emergency medical services (EMS) arrival, consider appropriate triage categorization of trauma to appropriately address a critical patient. A higher mechanism fall, such as a fall from a height of greater than 20 feet, high rate of vehicular speed, severe head injury, low Glasgow Coma Scale (GCS) score, or obvious deformity of more than two long bones, should involve coordination between trauma surgery and critical care specialties prior to arrival using system-based trauma criteria. Consider age as a risk factor in itself for increased mortality for similar traumas during triage. Female gender, fall mechanism, and age are the three greatest risk factors for undertriage.4
Prehospital EMS providers should be incorporated into the history process, providing critical information on the conditions and environment of the presentation, scene construct, and initial vital signs for the trauma patient. Field care and transport information applies immediately to the care of the trauma patient and provides critical clues on time, mechanism, and exposure hazards to the patient(s). Follow decontamination protocols for hazardous material contaminated patients.
Both traumatic work-up and medical work-up have the same starting point upon patient presentation to the ED. The patient’s history and physical exam during the primary and secondary surveys guide diagnostic studies needed to evaluate for a broad differential diagnosis in a cost-effective manner. Begin with the ATLS algorithm for the initial evaluation of a trauma patient to identify potential blunt abdominal injuries. Streamline the assessment for injuries requiring immediate intervention, i.e., tourniquets for severe pulsatile extremity hemorrhage, and simultaneously direct the rapid evaluation and resuscitation. ABCDE summarizes the initial steps to guide the primary survey.
Airway: Quickly evaluate the patency of the airway via an assessment of the patient’s speaking ability or visual inspection.
Stridor, changes in phonation, gurgling secretions, agitation, confusion, or decreased level of responsiveness signify impending airway compromise requiring action. Consider the need for additional expertise in the room with on-call anesthesia or otolaryngology colleagues. Assign the role for cervical spinal motion restriction during intubation, if appropriate to the mechanism of traumatic injury. Have the tools ready for airway management to include oxygen tubing, suction, gum elastic bougie, bag-valve mask with positive end expiratory pressure (PEEP) valve, naso-/oropharyngeal airways, multiple sizes of endotracheal tubes, rescue airway device, Magill forceps for debris removal, and an airway cart in the room with video laryngoscopy available. The cricothyroidotomy kit (at a minimum, a scalpel and size 6.0 endotracheal tube) should be readily accessible.
The updated ATLS now includes drug-assisted intubation as an alternative to rapid sequence intubation (RSI) for airway protection of patients with intact gag reflex.5
Breathing: Rapidly assess and verify adequate oxygenation and ventilation. Observe for symmetric chest rise, placing hands to the chest if necessary to gauge chest wall excursion, auscultate for breath sounds bilaterally, and establish pulse oximetry and end-tidal waveform capnography. Actively avoid hypoxemia through supplemental oxygenation. ATLS also promotes the use of ultrasound to investigate the presence or absence of lung sliding for early diagnosis of pneumothorax.5 If indicated, perform immediate needle decompression, now recommended to be performed at the fifth intercostal space in mid-axillary line rather than the second intercostal space midclavicular line location. As with all procedures, reassess after the intervention and ensure effectiveness. Plan to proceed later to subsequent placement of a tube thoracostomy after completion of the primary survey.
Circulation: Obtain a complete set of vital signs and be ready to intervene appropriately in states of shock as evident by hypotension and tachycardia. Palpate central and peripheral pulses bilaterally and assess pulse quality in addition to rate and symmetry. Immediately temporize active hemorrhage, and establish large-bore intravenous or intraosseous vascular access with the ultimate goal of volume infusion as appropriate of blood product. Avoid hemodilution with judicious crystalloids while initiating emergency release or awaiting type-specific blood products. Packed red blood cells (PRBCs) can be transfused in component therapy with platelets and fresh frozen plasma if the preferred fresh whole blood is unavailable. For an unstable patient, rapidly complete a focused assessment with sonography for trauma (FAST) examination with bedside ultrasonography to obtain critical information regarding resuscitation and a potential operative clinical course.6 (See Figures 1-3.)
Figure 1. Coronal View of the Right Upper Quadrant FAST Exam Without Free Fluid |
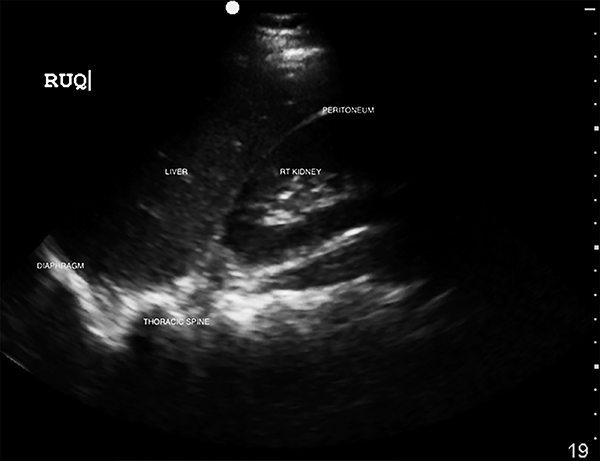
|
|
FAST: Focused assessment with sonography for trauma |
Figure 2. Free Fluid in Left Upper Quadrant View of the eFAST Exam |
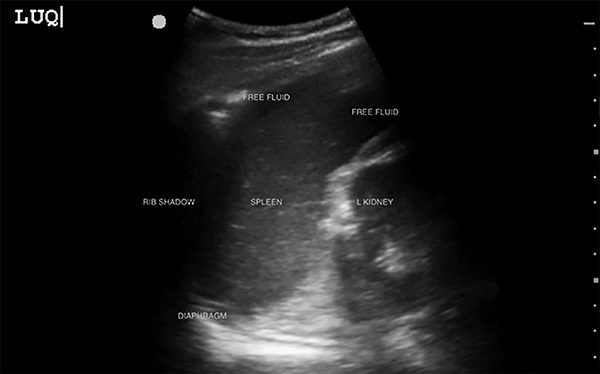 |
|
eFAST: Extended focused assessment with sonography for trauma |
Figure 3. Sagittal View of a Female Pelvis, Positive for Free Fluid |
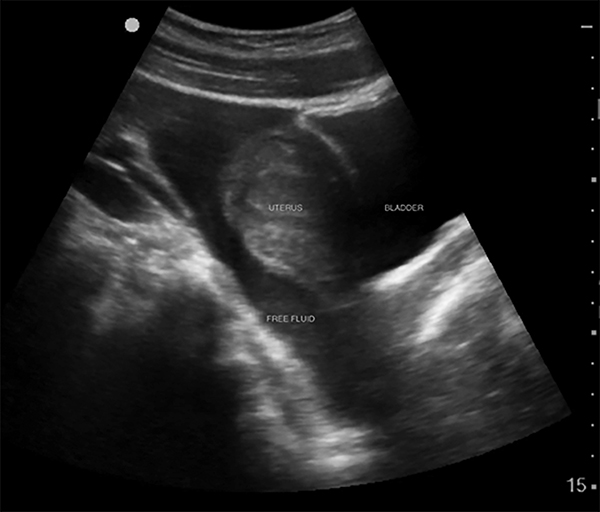 |
Disability: Perform a gross neurologic assessment to establish a baseline level of functionality and awareness. Obtain history as available to ascertain if a known loss of consciousness occurred, if any changes to mental status occurred en route, or if the patient has any known disability at baseline. Consider having special translator availability for non-English-speaking patients, hearing impaired, or other cultural and language barriers. Consider panic and agitation as potential signs of cerebral hypoperfusion.
Use the GCS score to assess for potential brain injury involving three criteria: best of eye opening, verbal response, and motor ability. The classification of traumatic brain injury (TBI) severity can be made based on total score, which helps to guide management in the acute phase of resuscitation. Mild TBI is considered with GCS 13-15 (both with or without known loss of consciousness), moderate TBI involves a score of 9-12, and severe TBI is a score less than 8. ATLS updated recommendations for acute management of severe TBI include to avoid hyperventilation states (pCO2 < 25), maintain systolic blood pressure (SBP) > 100 mmHg in patients ages 50-69 years and > 110 mmHg in patients ages 15-49 years, use propofol for sedation, and reduce elevated intracranial pressure (ICP).
Exposure: Carefully remove any protective equipment and the patient’s clothing to visually inspect for all potential signs of trauma, such as ecchymosis, pattern wounds, abrasions, lacerations, or other signs of injury. Use a team to maintain cervical spinal motion restriction and to log-roll the patient, remove the long spine board if not already performed, and fully evaluate the back of the patient. Complete a rapid palpation of the cervical, thoracic, and lumbar spinous processes for any palpable tenderness or step offs, address any immediate life threats, replace the patient to supine positioning, and apply external warm blankets to reduce hypothermia. Actively work to protect the patient from the trauma “triad of death” and the contributions of the resuscitation toward the interconnected hypothermia, acidosis, and coagulopathy.7 In ATLS-10, the 2018 update, additional emphasis is placed on vital sign trends and base excess to assess hemorrhage class and transfusion needs, instead of using static values.5
If at any point the patient becomes unstable, advance the resuscitation efforts and rapidly reassess in sequence, once again starting with the airway. Endotracheal tubes can become dislodged with log-roll movement for posterior assessment or transfer for radiology studies. Pelvic fractures can shift with patient repositioning and cause sudden loss of blood pressure.
Abbreviated History
For the conscious patient without immediate life threat on primary survey, obtain a brief medical history; use the mnemonic SAMPLE to review symptoms, allergies, medications, past medical history, last oral intake and/or last menses, and events leading to the traumatic incident. Be especially aware of prodromal symptoms that may suggest an underlying medical contribution to the trauma incident, such as an injured driver who may have sustained a traumatic event as a result of hypoglycemia, acute stroke, or myocardial infarction. Be wary of a patient with chronic hypertension and a systolic blood pressure of 90 mmHg; reductions in SBP of 30 mmHg below baseline cause hypoperfusion to vital organs.8 Review medication lists for antihypertensives, cardiac rate or rhythm control, and anticoagulation to properly contextualize the patient in the setting of a trauma resuscitation.
Evaluate and closely monitor transient responders. While they may improve during resuscitation from initially unstable to stable, the duration may be brief in transient responders. Continue to manage the patient in the mindset of an unstable patient algorithm and carefully balance the benefits and risks of decompensation when considering to pursue definitive radiographic studies.
Point-of-Care Radiographic Studies
Portable X-rays of the chest may be completed between the primary and secondary surveys. Perform a trauma single-view chest and, if there is abdominopelvic pain or trauma, pelvis portable radiographs. Interpret these immediately to evaluate for aortic transection, large hemothorax, pneumothorax, and pelvic fractures.
Although diagnostic peritoneal lavage (DPL) historically was performed in evaluation for intraperitoneal bleeding, it has been replaced by bedside ultrasonography in the modern management of trauma. DPL still may be considered if no ultrasound is available for the unstable patient who cannot safely proceed for CT evaluation.
Rapidly rule in life-threatening traumatic injuries with an extended FAST (eFAST) ultrasound as part of the primary survey. A complete eFAST, indicated per American College of Emergency Physicians ultrasound guidelines in both blunt and penetrating trauma of the chest and abdomen, assesses for the presence of intra-abdominal free fluid, pericardial effusion, and appropriate pleural lung sliding bilaterally.6 The eFAST imaging sequence rapidly assesses hemodynamically unstable patients who could receive immediate life-saving interventions for pneumothorax, hemothorax, pericardial tamponade, or potentially direct the resuscitation to the operating suite if massive intra-abdominal hemorrhage is detected.
In the stable patient with a positive FAST exam, heightened awareness promotes a sense of urgency to complete the remainder of the patient’s assessment, continue resuscitation in earnest, and monitor carefully while performing CT scans of the chest, abdomen, and pelvis to further characterize the patient’s injuries for potential operative planning.
Hemodynamically stable patients with a negative eFAST exam but with abdominal tenderness or evidence of trauma, including seat belt sign, should be evaluated further with a contrast CT scan. Ultrasonographic evaluation of the dependent portions of the peritoneal layers of the right upper quadrant, left upper quadrant, and retrovesicular pelvic views are not diagnostically equivalent to a helical contrasted CT scan of the entire abdomen and pelvis. Do not rely on the eFAST to exclude all potential injuries. Patients who subsequently are found to have isolated free fluid should be evaluated and managed by surgery. However, a retrospective systematic review found that only 27% of patients in this category required laparotomy while the rest were managed conservatively.9 In patients with continued abdominal tenderness but a negative initial CT scan, consider occult injury and monitor closely with serial abdominal examinations and vital signs.
The eFAST ultrasound is repeatable for dynamic change and includes assessment of the pericardium and the chest for blood and pneumothoraces. Identification of these concomitant pathologies is crucial for the blunt abdominal trauma patient, since positive pressure ventilation may contribute to potentially lethal tension physiology of an unrecognized pneumothorax during advanced imaging, resuscitation, or operative phase of care. If the patient becomes unstable, repeat the eFAST as part of the reassessment of the ABCDE primary survey.
Secondary Survey
Proceed to a focused history and physical examination during the secondary survey, with questions about provocation, prodrome, duration of loss of consciousness, loss of bowel or bladder control, alcohol and substance use, comorbid conditions, and medication use. Verify the nature of the known injuries and that no others are present. Perform a detailed assessment for sequelae or further trauma. History taking is more time-consuming with altered or disabled patients; be sure to interview EMS and family members for collateral details. Additionally, chart reviews can divulge the patient’s comorbidities, medications, and baseline neurologic status. Finally, do not overlook a thorough review of systems, which adds clues in the search for potential causation of a trauma.
Polytrauma
In a polytrauma patient, the abdomen is the third most frequently injured body part overall.10 Approximately 31% of patients with polytrauma will have an abdominal injury. The likelihood of solid organ injury is highest in the liver, at around 16%, followed by the spleen in blunt abdominal trauma.11 Additional abdominal organ systems that may be injured in declining order of incidence include the serosa, diaphragm, large bowel, and blood vessels. Patients are less likely to experience any direct injury to the stomach, duodenum, and pancreas.12
The prevalence of liver injury in blunt multitrauma patients ranges from 1% to 8%. Based on one Level I trauma center study performed over the course of four years, these injuries range in order of occurrence from parenchymal contusion to capsular tear and frank liver fracture.11 In the setting of a primary liver injury, additional occurring injuries were noted most in the left lung, right lung, mesentery, and left kidney in order of incidence. For primary liver injuries, coexisting injuries were found to be highest with the spleen, right lung, mesentery, left lung, colon, and right kidney in declining order of incidence.11
Diagnostic Testing
During the initial intravenous placement of a large-bore vascular access, have labs drawn and sent for stat processing. Large trauma centers may standardize the trauma panel to include venous blood gas, lactate, type and screen, complete blood count, comprehensive metabolic panel, coagulation studies, cardiac troponin, blood alcohol level, urine or serum beta human chorionic gonadotropin (hCG), urinalysis, and urine drug screen.
The ATLS table for classifying signs and symptoms of shock now includes an assessment of base excess as an indicator of shock.5 A venous or arterial blood gas provides this crucial information in addition to an estimated hemoglobin.
If the patient is hemodynamically stabilized enough to transport to the radiology department with a provider, perform CT evaluation for trauma. For outlying rural or community hospitals without trauma specialists, transfer of a stabilized trauma patient to a definitive trauma center may be most prudent prior to advanced imaging. Work with the nearest trauma center to determine the preferred, most expedient transfer process so as not to delay definitive care. Depending on the mechanism of injury, a noncontrast CT of the head may be performed first, followed by intravenous (IV) contrast for imaging of the chest, abdomen, and pelvis. These studies identify active extravasation in addition to reconstruct the thoracic and lumbosacral spine. While extremity injuries are potentially dramatic and distracting, they should receive plain film X-ray trauma views for fracture and foreign body evaluation after the secondary survey unless vascular injury is suspected, in which case, a CT angiography should be pursued.
Management
The initial management of hypovolemic shock in trauma patients was shifted in the most recent ATLS guidelines toward limited and judicious crystalloid resuscitation and instead, early transfusion of blood products.5 Additional goals of preventing additional morbidity and mortality include avoidance of worsening coagulopathy, reversing known existing anticoagulation, and providing tranexamic acid (TXA) if within three hours of injury. TXA has been evaluated in patients with hemorrhage resulting from trauma, including those with reduced GCS, and a concern for potential intracranial hemorrhage. TXA has demonstrated potential benefit without harm for patients if administered as a 1-g TXA slow IV push within the initial three hours of injury, followed by 1 g IV over eight hours.13 Further research is ongoing regarding dosing and timing of TXA.
In cases requiring massive transfusion protocol (MTP), follow the local hospital guidance for initiation criteria, since this may vary from 6-10 PRBCs to prompt a more balanced 1:1:1 approach with platelets and fresh frozen plasma. Add cryoprecipitate as available. Because citrate from packed blood products binds intravascular calcium, ensure repletion of calcium using 3 grams of calcium gluconate IV. This is preferred over 1 gram of calcium chloride IV because of less tissue toxicity in cases of extravasation. Use thromboelastography, when available, for guiding product administration. Be expressly familiar with the blood bank capabilities and limitations for product delivery in the moment of need and do not delay to request emergency release units of type O Rh-negative blood for an unstable trauma patient.
Resuscitative endovascular balloon occlusion of the aorta (REBOA) is indicated for multidisciplinary resuscitation of severely unstable or peri-arrest adult patients with massive intra-abdominal hemorrhage in blunt and penetrating trauma, pelvic fracture, and noncompressible bleeding of the femoral vessels. Occlusion of the aorta is a temporization measure with a plan for near immediate surgical control of hemorrhage within the next 30 minutes. It is not a device for definitive hemorrhage control, unlike a resuscitative thoracotomy, which provides access from which to cross clamp the aorta.
Establish femoral arterial access with an 8 Fr or larger kit inserted into the common femoral artery under ultrasound guidance or via vascular cutdown technique under sterile technique. The arterial line functions for accurate blood pressures to guide resuscitation, independent of whether the catheter balloon is inflated once placed. Maintain sterile procedure, measure the REBOA catheter first externally to the patient from the common femoral artery to either the sternal notch (Zone I - arch) or the xiphoid process (Zone III - infrarenal). Check balloon integrity and fully deflate to place the catheter through the femoral arterial line, advance to the desired distance, gently inflate the balloon, and affix with sutures. Confirm placement with a portable chest X-ray. Alternatively, if placed and not required immediately in the resuscitation, the balloon may be left deflated until deemed necessary by the clinical situation of the patient. Transfer to the operative suite for definitive repair. Additional research is pending on the practice of partial inflation, timing of deflations, and further considerations of REBOA use.14
Nonoperative Management
With the advent of and improvement in CT scans, the decision for nonoperative management for blunt abdominal trauma has markedly changed the trajectory of management after the insult has occurred. In fact, a 10-year review study on nonoperative management (NOM) of patients with blunt abdominal trauma showed that, when implemented properly, a success rate of 89% was achieved.15 This study used trauma scores, hemodynamic status, response to resuscitation, and imaging studies to determine the likelihood a patient could forgo surgery and instead be admitted to the intensive care unit (ICU) for serial exams and vital sign monitoring. The parameters for which NOM was determined were a blood pressure > 110 mmHg and heart rate
< 90 beats/minute and responsiveness to resuscitative measures, i.e., fluids, transfusion, O2, etc. Point-of-care ultrasound and CT were performed in complementary fashion. Surprisingly, the FAST exam was positive in 85% of NOM patients, thus indicating that a positive FAST does not always require surgical intervention and, instead, should be used in conjunction with the hemodynamic status of the patient, mechanism of injury, and responsiveness to treatment measures to determine the appropriateness of surgical intervention.
Of the injuries sustained from blunt abdominal trauma, liver injuries were the easiest to identify and manage with NOM, while bowel or hollow organ injuries were the most difficult to identify and manage without surgical intervention. The most common cause for failed NOM was delayed bleeding, which was identified via hemodynamic status, serial laboratory tests, and recurring abdominal examinations. Overall, the decision to operate should be based on the mechanism of injury, clinical presentation, and response to resuscitative measures.
Trauma Scores
Trauma scores have been around for more than four decades and have proven to be effective at predicting morbidity and mortality in trauma patients. However, while often robust, they have somewhat fallen out of favor because of improved imaging techniques and resuscitative measures in the trauma bay, operating room, and ICU. Nevertheless, for austere or resource-limited environments, the concept of the components of trauma that increase mortality can provide critical input for triage and rationing of care. Several trauma scores have been devised over the years, but the most commonly used scoring systems include the Injury Severity Score (ISS), New Injury Severity Score (NISS), Revised Trauma Score (RTS), and Trauma and Injury Severity Score (TRISS). Each scoring system can be categorized into an anatomical scoring system, a physiological scoring system, or a combined anatomical and physiological scoring system. Trauma scores are valuable for quality assurance, research, and establishment of a common language for communication with trauma centers from outside facilities.16
Anatomical scoring systems describe all the injuries recorded by clinical examination, imaging, and surgery or during an autopsy.17 They use a scoring rubric based on the number of injuries to a specific body region along with the severity of the injuries themselves. Examples of anatomical scoring systems include the ISS and the NISS.
Physiological scoring systems describe changes due to trauma that then are translated to changes in vital signs and levels of consciousness. Examples of physiological scoring systems include the RTS, GCS score, and Acute Physiological and Chronic Health Evaluations (APACHE I-III). Combined trauma scores take into account anatomical changes along with physiological changes secondary to trauma. Only two combined trauma scores are used in practice: TRISS and A Severity Characterization of Trauma (ASCOT). However, only TRISS will be discussed later. As a whole, the combined scoring method has been far more consistent with predicting outcomes in blunt abdominal patients.
ISS is an anatomical scoring system that provides an overall score for patients with multiple injuries. The body is divided into six regions, with each injury in a body region given a score. The most severe injury is given a higher score. The six body regions include the thorax, abdomen and visceral pelvis, head and neck, face, bony pelvis and extremities, and external structures, such as the skin. The highest score in a region is taken, and the three most severely injured body regions have their scores squared and then added together to yield the ISS score. Values < 9 are considered mild, 9-15 are moderate, and > 16 are severe.
There are several drawbacks to this scoring system, which include laborious calculations and the fact that the scoring system only uses one injury per body region and only three body regions total. This leads to undervaluing other injuries or even a body region altogether, thus not giving the full extent of the injuries sustained. Additionally, it minimizes the significance of head injuries and weighs each body region equally. Despite the flaws of this scoring system, the ISS has a 91% sensitivity and 89% specificity for predicting mortality.
The NISS is a simple and modified version of the ISS developed to combat the concern of overlooking injuries within the same body regions. Instead, it takes the sum of the squares of the three most severe injuries regardless of the body region. This recategorization places more emphasis on the head and torso. The scoring rubric is the same as the ISS, with < 9 considered mild and > 16 considered severe. It has 91% sensitivity and 93% specificity for predicting mortality. The NISS has outperformed ISS in predicting outcomes, but because of the complexity of computation, it has failed to be adopted.
The RTS is a physiological scoring tool that uses vital signs and the GCS to predict outcomes. However, the scoring system uses a computation not consistent with practical use in the emergency room. To yield an RTS score, the clinician will sum 0.9368(GCS) + 0.7326(SBP) + 0.2908(respiratory rate [RR]). The applicability of this score occurs when there is an electronic version for computation, where a provider can just place the GCS, SBP, and RR into a given computerized algorithm and have it generate the value instead of performing the calculation by hand. Notably, the RTS system places greater emphasis on the GCS to compensate for head injuries. The scoring system for RTS is markedly different than any anatomical scoring system in the sense that the lower the score, the worse the predictive measures. A score < 4 indicates significant severity and is comparable to an ISS score of > 16. RTS has a sensitivity of 97% and a specificity of 80% for predicting mortality. RTS has limitations because of the GCS estimation, especially in ventilated or intoxicated patients, likely indicating a worse level of severity than actually exists. Additionally, it does not account for the length of time for physiological derangement.
TRISS is a scoring system that combines both the anatomical ISS and physiological RTS into one score. It has been proven to be the most effective and validated scoring system for blunt abdominal trauma secondary to falls, along with trauma scoring for elderly patients regardless of the type of trauma. The calculation is markedly robust and has been incorporated into MDCalc, thus making the values easily obtainable. For completion’s sake, the equation will be provided.
Ps = 1/(1 + e –b), where “b” is obtained from b = b0 + b1 (RTS) + b2 (ISS) + b3 (age index). The coefficients b0 and b3 are derived from multiple regression analysis of the Major Trauma Outcome Study (MTOS) database. The age index is 0 if the patient is younger than 54 years of age or 1 if the patient is 55 years of age or older. Notably, the values and coefficients provided are specific to blunt abdominal trauma and are different for patients who have sustained penetrating trauma. The TRISS scoring range has had significant variability, but what has been accepted through several studies is that a value < 91 is considered severe and indicates poor outcomes. It has a sensitivity of 97% and a specificity of 88% for predicting mortality.17 Similar to RTS, it does not account for intubated or intoxicated patients and, thus, can lead to more severe scoring despite marginal insults.
Despite the somewhat convoluted series of equations and descriptions regarding different modalities for calculating trauma scores, they can be helpful for triaging patients in the field or in a rural tertiary emergency room. Several studies have validated TRISS as the most predictive measure for mortality in blunt trauma secondary to falls and trauma to the elderly. Its use, in conjunction with clinical gestalt, can aid an emergency medicine clinician in determining the need for immediate intervention or transport out of the rural setting, or it can predict future outcomes of patients who present to the emergency department. It should be considered an additive measure to clinicians’ practice.
Special Populations
Investigation of blunt abdominal trauma merits special consideration in elderly, pediatric, and pregnant patients. Each of these populations presents with unique pitfalls.
Elderly
In the elderly population, clinicians should give special consideration to the unreliability of the physical exam and maintain a high suspicion for intra-abdominal injury following blunt abdominal trauma. TRISS assesses the risk of preventable vs. unpreventable deaths following trauma and should highlight the increased mortality associated with abdominal trauma in the elderly. Patients older than 55 years of age are at a significantly increased risk of unpreventable deaths compared to those younger than 55 years. The elderly population should be evaluated with CT following abdominal trauma, and physicians should not rely on physical exams to exclude pathology as one might in the younger population. The risk for possible contrast-induced or worsened nephropathy can be mitigated with assessment of the glomerular filtration rate of the patient, a review of the patient’s medication list for nephrotoxic medications, and consideration of the gentle use of isotonic crystalloid for volume expansion prior to the use of contrast, if the patient’s comorbidities allow.18 In addition, monitor and support renal function; coordinate with pharmacy for renally dosed medication regimens.
Treat severe pain appropriately, since uncontrolled pain increases the incidence of delirium in the geriatric patient population.19 Achieve pain control through a combination of nonopiate medications with cautiously reduced doses of opiates. Geriatric patients have increased sensitivity and adverse effects with opiates, which include hypotension, respiratory depression, and delirium.19 In the absence of liver injury or failure, acetaminophen 1,000 mg IV every six hours is a safe option for pain control. Avoid nonsteroidal anti-inflammatory drugs because of the potential for medication interactions, renal impairment, and gastric mucosal damage.19
Pediatric
Unlike the elderly population, which has the typical injuries similar to the general population, the pediatric population has specific injuries that are not typically seen or are not seen in isolation throughout the general population. Additionally, these injuries often have a unique pattern.18 Complicating these diagnoses is the hesitancy to expose pediatric patients to high-dose radiation, which is typical of CT scans. Ultrasonography has been applied in pediatric populations; however, it has a variable and lower specificity (as low as 28%) for detection of intra-abdominal free fluid.20
To make an accurate diagnosis without unnecessary risk, different clinical decision rules have been studied. One such rule, which was validated by a prospective observational cohort study, consists of seven patient history and physical exam findings and has a negative predictive value (NPV) of 99.9%. The factors comprised in this decision rule include no evidence of abdominal wall trauma or seat belt sign, Glasgow Coma Scale score greater than 13, no abdominal tenderness, no evidence of thoracic wall trauma, no complaints of abdominal pain, no decreased breath sounds, and no vomiting. Of note, this study did not use imaging including a FAST or laboratory values, which allows for a wide use of the rule throughout many different settings.21
A separate clinical decision rule that incorporates imaging and laboratory values also has been studied and has been shown to have an NPV of 97%. The criteria in this rule include hypotension for age, abnormal abdominal exam, aspartate aminotransferase > 200 U/L, amylase > 100 U/L, microhematuria > 5 erythrocytes/high power field, hematocrit < 30%, and abnormal chest radiograph. Of note, the Holmes et al study stated that the use of FAST and laboratory values in conjunction with their decision rule would increase the NPV. However, both studies point out that FAST is not an adequate screening tool, and a negative bedside FAST exam does not rule out intra-abdominal injury in the pediatric population. Current practice is to reserve CT scans for patients who meet the clinical decision rules outlined earlier.22
Abdominal injuries secondary to blunt trauma in the pediatric population most commonly cause injury to spleen, liver, kidney, hollow viscus, pancreas, and adrenal gland in decreasing order.18 Over the past 50 years, the standard of care for spleen and liver injuries has shifted to nonoperative management. Overall, nonoperative management has been successful in more than 90% of patients, with higher splenic salvage rates, a decrease in the need for blood transfusions, and a lower rate of complications. Even successful nonoperative management of high-grade injuries has been demonstrated recently. Patients presenting with hemodynamic instability who are unresponsive to resuscitative management are reserved for operative management.22
Pancreatic injuries in children typically occur after blunt trauma. Bicycle handlebar injuries are the most common mechanism, followed by a pedestrian struck by a motor vehicle, motor vehicle collisions, and falls. The pediatric population typically has lower-grade injuries than adults, although they are more likely to have isolated pancreatic injuries. Much like splenic and liver injuries, low-grade pancreatic injuries are treated nonoperatively with conservative management, including bowel rest, pain management, and repeated abdominal exams. However, patients with known ductal disruptions are treated with operative exploration and intervention. Although hospital admissions due to intra-abdominal injuries typically are less than 10%, pancreatic trauma can be associated with significant morbidity.23
Isolated adrenal injury among the pediatric population after blunt abdominal trauma is rare. Typically, injury to the adrenal gland is diagnosed as adrenal hemorrhage via CT. In children, it appears to be an incidental finding that is followed up by repeat imaging. In most cases of suspected injury, follow-up imaging shows complete resolution. However, it is important to consider a wide differential in a patient with adrenal hemorrhage, especially in the absence of other traumatic findings on CT. Adrenal neoplasm within the pediatric population must be considered. However, the morbidity and mortality of true isolated adrenal injury following blunt trauma in the pediatric population appears to be minimal.24
Although abdominal injuries associated with nonaccidental trauma (NAT) are less common than other injuries due to abuse, consider NAT as a mechanism in the evaluation of pediatric trauma. Complete a thorough and complete skin examination, evaluate injuries in the context of the reported mechanism, interview the child and witnesses, and maintain a high index of suspicion for abusive patterns of bruising and fractures. Ecchymosis found on the torso, neck, and ears, as well as on soft parts of the body, should heighten awareness and trigger further investigation, such as a skeletal survey series of plain films and reporting of concern through appropriate facility and local procedures. Fractures are the second most common presentation of NAT. Although accidental fractures are a common presentation in the pediatric population, certain fractures should prompt further investigation. These fractures include posterior rib fractures without severe trauma, metaphyseal fractures, and femur fractures.18
Pregnancy
Rapid identification of a pregnancy in a reproductive-age female is paramount during the initial evaluation and trauma resuscitation. The pregnant population presenting with blunt abdominal trauma has many of the same pitfalls as the pediatric population. The use of CT is not the first-line approach, and the anatomy of the pregnant patient presents with its own pattern of injuries. These injuries include placental abruption, uterine rupture, direct fetal injury, and fetomaternal hemorrhage.
Evaluation of the stable pregnant population begins with bedside ultrasound, which is preferred because of the lack of ionizing radiation to the patient and developing fetus. However, do not withhold CT in unstable patients for whom appropriate imaging is necessary to identify acute life-threatening pathology. CT remains the imaging modality of choice for organ injury, including placental abruption, and should not be delayed over concern for fetal radiation exposure. The highest risk for fetal viability after exposure to radiation is within the first two weeks for malformations, two to eight weeks for central nervous system teratogenesis, and the first eight to 15 weeks of gestation.18 Once the patient has been stabilized, fetal tocographic monitoring should be initiated as quickly as possible to assess for uterine irritability, which is defined as greater than three contractions per hour.
Uterine irritability on tocographic monitoring is the most sensitive finding for placental abruption. With the exception of maternal death, placental abruption is the most common cause of fetal death. Placental abruption can cause fetomaternal hemorrhage, which occurs when fetal red blood cells enter the maternal bloodstream after trauma. This may stimulate disseminated intravascular coagulation or amniotic fluid embolism. It is important to administer Rho(D) immunoglobulin to pregnant patients who are Rh-negative.18
Uterine rupture typically occurs as the result of the most violent accidents and, in most cases, results in fetal death and severe maternal hemorrhage or significant maternal morbidity/mortality.25 The clinical presentation for uterine rupture is quite nonspecific, but loss of palpable uterine contour and palpation of fetal parts are suggestive of the diagnosis.18 Unfortunately, uterine rupture results in a high fetal mortality rate. The case reports addressing uterine injury following maternal trauma show that the majority result in fetal demise.25
The pregnant population presents a unique complication when maternal cardiac arrest occurs. This can occur because of a medical emergency, but it also may be the result of traumatic injury. In the setting of maternal cardiac arrest with a fetus of viable age, a perimortem cesarean delivery should be performed. Fetal survival of a perimortem cesarean delivery significantly diminishes if the procedure is not performed within the first four minutes of cardiac arrest. Delivery of the fetus not only improves fetal outcome, but it enhances resuscitation efforts for the mother as well.18
As with pediatric trauma patients, pregnant trauma patients also should prompt consideration of domestic partner violence. Complete a thorough skin examination, interview the patient privately from visitors, and be mindful of patient safety in disposition planning.
Sequelae and Disposition
Occult Injuries
Blunt abdominal trauma is a high-risk mechanism for occult or missed injury. Although often rare, these injuries can represent a significant mortality and morbidity and, thus, they require a high index of suspicion. Such injuries include small and large bowel hollow viscus injuries, occult pancreatic contusion or hemorrhage, diaphragm rupture, biliary tree injury, and subcapsular liver and spleen injuries. Injuries from blunt abdominal trauma may evolve over time, which poses a question in terms of management, including observation with conservative management vs. possible diagnostic laparoscopy or laparotomy. One study compared the efficacy of diagnostic laparoscopy as an adjunct to select patients for conservative management of solid organ injuries following blunt abdominal trauma. In this study, 15 patients were prospectively evaluated after CT scans, which identified 17 distinct solid organ injuries that were subsequently visualized well on diagnostic laparoscopy in 15 of the 17 identified injuries; there were two occult hollow viscus injuries, including one colonic injury and one small bowel injury that required subsequent laparotomy for management of both. In contrast, diagnostic laparoscopy was followed by conservative management in eight patients that revealed only minor injury or adequate hemostasis that did not require further laparotomy.9 Although rare, the small study identified two separate occult injuries that otherwise may have been missed had a laparoscopy not been performed.
One case review in particular outlines the caution that must be taken, as well as the high degree of suspicion one must have, simply based on mechanism, when evaluating an otherwise well-appearing patient with abdominal injury. In this case, a 32-year-old male suffered blunt abdominal trauma from a high-speed MVC. He was admitted and observed for 24 hours before being discharged in stable condition. However, he then re-presented 10 days after his initial trauma with abdominal pain and vomiting that a CT scan then confirmed as mesenteric inflammation and small bowel obstruction. The patient then was managed conservatively with bowel rest and decompression. However, he presented once again at day 23 following his initial injury and subsequently had a laparotomy that revealed a mesenteric hematoma leading to recurrent small bowel obstruction. This case outlines several factors, including younger age and an initial reassuring exam, which, although in the setting of significant mechanism, led to a failure of conservative management and significant morbidity with delayed definitive intervention.26
One pitfall from this particular case is the overreliance on CT scan that creates a sense of false reassurance. Several studies have estimated the false-negative rate of CT for traumatic small bowel perforation to be as high as 13% to 15%.8,27 Commonly associated signs or injuries that increase the risk for traumatic small bowel perforation include a seat belt sign with associated tenderness and a Chance fracture of the lumbar spine.27 Even with a high degree of suspicion and proper management, sequelae of occult small bowel injuries can present in the weeks or months following injury as a small bowel obstruction that is thought to be a result of localized or transient ischemia that does not cause frank necrosis but more inflammation and fibrosis.28
Another rare but significant occult injury that has significant pitfalls in diagnosis is blunt diaphragmatic injury. One retrospective review of hospital CT scans found that seven of 57 blunt diaphragmatic injuries were missed on initial CT evaluation, and recognition was delayed anywhere between two days and three months.29 Two patients had left-sided injuries that presented with normal CT scans, and five patients had right-sided diaphragm ruptures that were identified on CT at initial presentation, although they were attributed to other chest trauma.29 These numbers are significant, since, according to the National Trauma Databank in the year 2012 in particular, 3,873 of 833,309 patients were found to have traumatic diaphragm injuries. Patients tended to be older and have a high injury severity score and mortality rate.12
The issue of occult pancreatic injury represents yet another difficult to diagnose although significant injury that must be considered in the setting of blunt abdominal trauma. In all abdominal trauma presentations, pancreatic injury occurs in between 3% and 12% of cases and has a wide mortality rate, ranging from 3% to 70%.30-32 Factors that increase the rate of pancreatic injury include compulsory seat belts and deceleration injuries. The retroperitoneal location of the pancreas predisposes to these injuries with compression of the spine. A CT scan often is unreliable for the diagnosis of blunt pancreatic injury and, therefore, one study evaluated the use of endoscopic retrograde cholangiopancreatography (ERCP) to augment pancreatic injury diagnosis. In the study, 26 patients were evaluated by ERCP for suspected pancreatic ductal injury at a Level I trauma center over the span of 13 years. Twenty-six patients were selected for elevations in amylase and lipase, CT scan results, or findings at the time of laparotomy for other injuries. Of the 26 patients, eight patients had an intact biliary tree, one patient had a gallbladder injury with otherwise intact biliary tree, and 18/26 had partial or complete transection of the main pancreatic duct branches.33 This article advocates for the use of ERCP in cases of suspected pancreatic injury, since it may provide more definitive diagnosis and the option for intervention that may reduce the extent of open surgery as relates to the repair of pancreatic injuries.
Sepsis
Sepsis is a common and often debilitating outcome of blunt abdominal trauma. Several studies have identified sepsis as a leading cause for morbidity and mortality in operative and nonoperative abdominal trauma patients. The most common causes of sepsis in blunt abdominal trauma patients often stem from early onset transfusions, postoperative closure methods, damage control surgeries, age, mechanism of injury, and severity of injury pattern. Regarding transfusions, it has been documented that more than three transfusions were closely correlated with increased rates of infections and, thus, lends itself to the recommendation of minimizing transfusions only to patients in either critical condition or not responsive to fluid administration.34 Postabdominal wound closures, specifically leaving the abdomen open, were closely linked to increased rates of infection and are often the closure method of choice for damage control surgeries, thus linking the two. In the context of the latter, it is still unclear on the exact causality, but it is more likely related to the severity of this initial insult and not the actual operative management, since the most commonly isolated microbes from these types of infections were Escherichia coli and Staphylococcus aureus.34 Notably, drains and organ removal were not associated with increased rates of infection, nor were hours of operative time. The length of time spent in the ICU did have a correlation, but the infections were more commonly nosocomial rather than innate.
Several measures, including trauma scoring and the use of biomarkers, have been attempted to predict infection rates. Trauma scoring has been a cornerstone for predicting the likelihood of mortality in abdominal trauma patients, but it has fallen out of practice because of the laborious calculation methods, improved methods for early identification of injuries, and enhanced treatment methods. However, there still is utility for using trauma scores to gauge the severity of injury and predict outcomes, including the development of sepsis in the blunt abdominal trauma patient. ISS scores > 20 and Abdominal Trauma Index (ATI) scores > 24 have shown a strong correlation with poor outcomes in the blunt abdominal trauma patient and have been scores used to determine and often prepare for the development of sepsis in this patient population.
The use of biomarkers has been a controversial one but still warrants consideration. The most commonly used markers include: procalcitonin, C-reactive protein (CRP), interleukin-6 (IL-6), and, most recently, interleukin-10 (IL-10). However, of the serum markers available, procalcitonin has been the only one to show a correlation to infection, but the studies supporting this are lacking.34 CRP has shown no association to levels of infection, and IL-6 and IL-10 are tests not likely to result while the patient is in the emergency department, but can be reserved for the ICU or inpatient settings. As a result, interleukins could be reliable serum markers for measurement if studied in the proper setting and context.
A clinician should initiate standard resuscitation measures including fluid therapy when appropriate, blood cultures, and initiate broad-spectrum antibiotics, but the difference in management stems from the identification of a source. Although source control is the primary objective regardless of the circumstance, the offending microbes and likely source of infection differ greatly. Blunt abdominal trauma patients most commonly develop sepsis secondary to either resuscitative measures or in-hospital acquisition. As a result, consider and ensure treatment coverage against methicillin-resistant S. aureus, carbapenem-resistant Klebsiella pneumoniae (CRKP), and E. coli. Notably, CRKP rates of infection have increased dramatically in recent years, likely from overuse of carbapenem in the inpatient setting, thus making antimicrobial selection more difficult.
Antimicrobial selection typically involves broad-spectrum antibiotics, with tigecycline used more frequently as resistant microbes continue to plague management plans. Similar to nontrauma patients with sepsis, the use of steroids is controversial and has not shown concise efficacy. Thus, steroids should be used sparingly, since their use likely could blunt recovery and resuscitative efforts in operative and nonoperative patients.
Summary
The key updates and aspects for the management of blunt abdominal trauma have been discussed, the application of which will vary greatly from a remote critical access center to a Level I trauma center for single vs. multiple vs. mass casualty incidents. Regardless of the setting, no trauma runs without room for improvement. It is imperative to debrief the department team to appropriately learn and improve from each encounter.
Upgrade the practice of trauma through addressing best practice methods for the management of blunt abdominal trauma from prehospital to disposition. Be especially aware of updates in ATLS: stabilization, airway management, blood product resuscitation, and point-of-care ultrasound. Work rapidly and simultaneously as a team to complete the primary survey, initiate stabilization, apply point-of-care testing, perform a thorough secondary survey, and decide for advanced imaging studies or operative management of blunt abdominal trauma. Know the protocols of the nearest specialty trauma center for expedient transfer.
Avoid the pitfalls of undertraining, undertriage, and underresuscitation of the trauma patient. Do not use point-of-care testing to rule out life-threatening diagnoses. Instead, incorporate these valuable studies to rapidly rule in and guide resuscitation of serious traumatic blunt abdominal injuries. Serial examinations and admission for observation still may be required in the setting of negative CT imaging. For positive findings, the decision for nonoperative management requires close supervision. Be familiar with injury severity scoring systems and components of which to ensure accurate triage, trauma training, and trauma transfers. Prepare to apply core trauma concepts to special populations, such as pediatric, pregnant, and elderly patients, while considering the potential for nonaccidental trauma in vulnerable populations.
REFERENCES
- Arumugam S, Al-Hassani A, El-Menyar A, et al. Frequency, causes and pattern of abdominal trauma: A 4-year descriptive analysis. J Emerg Trauma Shock 2015;8:193-198.
- O’Rourke MC, Landis R, Burns B. Blunt abdominal trauma. In: StatPearls. StatPearls Publishing; 2019.
- Gad MA, Saber A, Farrag S, et al. Incidence, patterns, and factors predicting mortality of abdominal injuries in trauma patients. N Am J Med Sci 2012;4:129-134.
- Jeong JH, Park YJ, Kim DH, et al. The new trauma score (NTS): A modification of the revised trauma score for better trauma mortality prediction. BMC Surg 2017;17:77.
- ATLS Subcommittee; American College of Surgeons’ Committee on Trauma; International ATLS working group. Advanced trauma life support (ATLS): The ninth edition. J Trauma Acute Care Surg 2013;74:1363-1366.
- American College of Emergency Physicians. Ultrasound guidelines: Emergency, point-of-care, and clinical ultrasound guidelines in medicine. Ann Emerg Med 2017;69:E27-E54.
- Murphy P, Colwell C, Pineda G. Understand the trauma triad of death. EMS World February 2012. https://www.emsworld.com/article/10565011/understand-trauma-triad-death
- Banz VM, Butt MU, Zimmermann H, et al. Free abdominal fluid without obvious solid organ injury upon CT imaging: An actual problem or simply over-diagnosing. J Trauma Manag Outcomes 2009;3:10.
- Townsend MC, Flancbaum L, Choban PS, Coutier CT. Diagnostic laparoscopy as an adjunct to selective conservative management of solid organ injuries after blunt abdominal trauma. J Trauma 1993;35:647-651.
- Cox EF. Blunt abdominal trauma. A 5-year analysis of 870 patients requiring celiotomy. Ann Surg 1984;199:467-474.
- Abdelrahman H, Ajaj A, Atique S, et al. Conservative management of major liver necrosis after angioembolization in a patient with blunt trauma. Case Rep Surg 2013;2013:954050.
- Leenen LP. Abdominal trauma: From operative to nonoperative management. Injury 2009;40(Suppl 4):S62-S68.
- Roberts I, Shakur H, Coats T, et al. The CRASH-2 trial: A randomised controlled trial and economic evaluation of the effects of tranexamic acid on death, vascular occlusive events and transfusion requirement in bleeding trauma patients. Health Technol Assess 2013;17:1-79.
- Bulger EM, Perina DG, Qasim Z, et al. Clinical use of resuscitative endovascular balloon occlusion of the aorta (REBOA) in civilian trauma systems in the USA, 2019: A joint statement from the American College of Surgeons Committee on Trauma, the American College of Emergency Physicians, the National Association of Emergency Medical Services Physicians and the National Association of Emergency Medical Technicians. Trauma Surg Acute Care Open 2019;4:e000376.
- Radjou AN, Paramsivam J, Mohan Kumar S The efficacy and safety of non-operative management in abdominal injury in a high volume but resource stretched setting. Int Surg J 2015;2:527-533.
- Javali RH, Krishnamoorthy, Patil A, et al. Comparison of Injury Severity Score, New Injury Severity Score, Revised Trauma Score and Trauma and Injury Severity Score for mortality prediction in elderly trauma patients. Indian J Crit Care Med 2019;23:73-77.
- Orhon R, Eren SH, Karadayı S, et al. Comparison of trauma scores for predicting mortality and morbidity on trauma patients. Ulus Travma Acil Cerrahi Derg (Turkish J Trauma Emerg Surg) 2014;20:258-264.
- Tintinalli JE, Stapczynski JS, Ma OJ, et al. Tintinalli’s Emergency Medicine: A Comprehensive Study Guide. 8th ed. McGraw-Hill Education; 2016.
- Tsang KS, Page F, Mackenney P. Can intravenous paracetamol reduce opioid use in preoperative hip fracture patients? Orthopedics 2013;36(Suppl 2):20-24.
- Riera A, Hayward H, Silva CT, Chen L. Reevaluation of FAST sensitivity in pediatric blunt abdominal trauma patients: Should we redefine the qualitative threshold for significant hemoperitoneum? Pediatr Emerg Care 2019; Jul 25. doi: 10.1097/pec.0000000000001877
- Holmes JF, Lillis K, Monroe D, et al. Identifying children at very low risk of clinically important blunt abdominal injuries. Ann Emerg Med 2013;62:107-116.e2.
- Dalton BGA, Dehmer JJ, Gonzalez KW, Shah SR. Blunt spleen and liver trauma. J Pediatr Intensive Care 2015;4:10-15.
- Lesher A, Williams R. Pancreatic and duodenal trauma in children. J Pediatr Intensive Care 2015;4:21-26.
- Roupakias S, Papoutsakis M, Mitsakou P. Blunt adrenal gland trauma in the pediatric population. Asian J Surg 2011;34:103-110.
- Suchecki G, Tilden H, Roloff K, et al. Management of traumatic uterine rupture in blunt abdominal trauma: A case report and literature review. Cureus 2020;12:e8396.
- McGuigan A, Brown R. Early and delayed presentation of traumatic small bowel injury. BMJ Case Rep 2016;2016:bcr2016214586.
- Fakhry SM, Watts DD, Luchette FA; EAST Multi-Institutional Hollow Viscus Injury Research Group. Current diagnostic approaches lack sensitivity in the diagnosis of perforated blunt small bowel injury: Analysis from 275,557 trauma admissions from the EAST Multi-Institutional HVI Trial. J Trauma 2003;54:295-306.
- Tugnoli G, Di Saverio S, Casadei GP, et al. Houdini’s last deception (or, Aristotle’s bowel). Lancet 2008;372:862.
- Fair KA, Gordon NT, Barbosa RR, al. Traumatic diaphragmatic injury in the American College of Surgeons National Trauma Data Bank: A new examination of a rare diagnosis. Am J Surg 2015;209:864-869.
- Recinos G, DuBose JJ, Teixeira PGR, et al. Local complications following pancreatic trauma. Injury 2009;40:516-520.
- Seamon MJ, Kim PK, Stawicki SP, et al. Pancreatic injury in damage control laparotomies: Is pancreatic resection safe during the initial laparotomy? Injury 2009;40:61-65.
- Al-Ahmadi K, Ahmed N. Outcomes after pancreatic trauma: Experience at a single institution. Can J Surg 2008;51:118-124.
- Rogers SJ, Cello JP, Schecter WP. Endoscopic retrograde cholangiopancreatography in patients with pancreatic trauma. J Trauma 2010;68:538-544.
- Jin H, Liu Z, Xiao Y, et al. Prediction of sepsis in trauma patients. Burns Trauma 2014;2:106-113.
Blunt abdominal trauma is commonly encountered in any acute care center. Prompt recognition, assessment, diagnostic evaluation, and disposition are critical aspects that must be a part of every clinician’s expertise.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
