Nonpenetrating Ocular Trauma Part II: Potential Vision Threats and Special Populations
May 1, 2021
Reprints
AUTHORS
Dustin B. Williams, MD, FACEP, Associate Professor, Program Director, Emergency Medicine Residency, University of Texas Southwestern, Dallas
Melissa Smith, MD, Assistant Professor, Assistant Program Director, Emergency Medicine Residency, University of Texas Southwestern, Dallas
Aman Pandey, MD, Senior Resident, Department of Emergency Medicine, University of Texas Southwestern, Dallas
Larissa Velez, MD, Professor of Emergency Medicine, Vice Chair for Education, University of Texas Southwestern, Dallas
PEER REVIEWER
Catherine Marco, MD, FACEP, Professor, Department of Emergency Medicine, Wright State University, Dayton, OH
EXECUTIVE SUMMARY
- Chemical injuries to the eye are ophthalmologic emergencies and must receive immediate irrigation. Conjunctival pH should only be measured after irrigation.
- Topical anesthetics, such as tetracaine, significantly reduce the pain of corneal abrasions or inflammation, but are not effective for inflammation of deeper structures, such as the iris.
- It is recommended that corneal abrasions be treated with prophylactic antibiotics. If patients are at risk for pseudomonal infection (i.e., contact lens wearers), fluoroquinolone drops are the optimal mono-drug therapy.
- Suspect retained foreign body underneath the upper eyelid if you see multiple vertically oriented linear corneal abrasions; examine the underside of the lid by everting it.
- Ocular ultrasound, also referred to as B-scan, can be useful in distinguishing between traumatic vitreous hemorrhage and retinal detachment.
Part I of this article reviewed nonpenetrating ocular trauma that presents a severe threat to vision. Part II will discuss potential vision threats of nonpenetrating ocular trauma, including burns, corneal abrasions, corneal foreign bodies, and hyphemas. Table 1 summarizes nonpenetrating ocular injuries that potentially threaten vision.
Ocular Burns
Ocular burns are a small but significant portion of eye injuries, totaling about 7.7% to 18% of all eye injuries, with a portion of them occurring bilaterally.1,2
Chemical Burns
Chemical burns include alkali (bases), acids, and detergents. The outcome depends on the chemical’s pH, concentration, volume, and the duration of exposure. In general, alkali exposures are more common than acids, and they tend to have worse clinical outcomes. The most common alkali agents associated with ocular burns include ammonia, sodium hydroxide, potassium hydroxide, and calcium hydroxide.3 These agents penetrate faster and deeper, since they result in saponification of the fatty acid components of the cell membrane.
Acids include sulfuric acid, sulfurous acid, hydrofluoric acid, and hydrochloric acid.3 They penetrate slower and cause coagulation necrosis (protein denaturation), which delays and somewhat prevents deeper penetration. However, acid burns can be devastating, so these characteristics should not result in a false sense of security. Clinically, any chemical eye exposure must be considered dangerous until decontamination and a full evaluation have been completed.
There are several classification systems for chemical eye burns. One of the most commonly used ones is the Roper-Hall classification (with four grades):
Grade I: conjunctival hyperemia, some conjunctival ecchymosis, chemosis, erosion of corneal epithelium, prognosis good;
Grade II: same as Grade I but worse, plus partial limbal ischemia, prognosis good;
Grade III: visible blood vessels are thrombosed and dark, extensive and deep tissue damage, corneal opacification, mydriasis, lens opacification, gray color to iris, prognosis guarded;
Grade IV: ischemic sclera, cloudy cornea, prognosis poor.
Another is the Dua classification, with six grades determined by the degree of limbal ischemia:
Grade 1: no clock hours of limbal ischemia, prognosis very good;
Grade 2: < 3 clock hours of limbal ischemia, prognosis good;
Grade 3: between 3 and 6 hours of limbal ischemia, prognosis good;
Grade 4: between 6 and 9 hours of limbal ischemia, prognosis good to guarded;
Grade 5: between 9 and 12 hours of limbal ischemia, prognosis guarded to poor;
Grade 6: total limbal ischemia, prognosis very poor.
The overall prognosis correlates with the presence of limbal ischemia (blanching of vessels at the junction of the cornea and sclera), damage to the conjunctival and episcleral tissues, severity of lid burns, and damage to the intraocular structures.3 Of these findings, limbal ischemia has the most subjectivity in its determination.4
Time to irrigation is the single most important factor affecting the outcomes in ocular chemical burns.5,6 Water is the most available irrigation fluid, but it is hypotonic relative to the corneal stroma, so amphoteric solutions (such as diphoterine), when readily available, are preferred.3 However, never delay irrigation and use whatever solution is readily available. Specially designed eye irrigation setups can be used.7 Alternatively, saline can be connected to a nasal cannula and the prongs placed on the nasal bridge as an inexpensive but effective irrigation setup.
Table 1. Summary of Nonpenetrating Ocular Trauma that Potentially Threatens Vision | |||
|
Condition |
Diagnostic |
ED Treatment |
Follow-up |
|
Ocular burns |
|
|
|
|
Corneal abrasions |
|
|
|
|
Corneal foreign bodies |
|
|
|
|
Hyphema |
|
|
|
|
Traumatic iritis and uveitis |
|
|
|
|
Traumatic mydriasis |
|
|
|
|
Iridodialysis |
|
|
|
|
Traumatic vitreous hemorrhage |
|
|
|
|
ED: emergency department; PO: orally; NSAIDs: nonsteroidal anti-inflammatory drugs; IOP: intraocular pressure; CNS: central nervous system | |||
When performing irrigation, make sure all areas of the cornea and conjunctiva are irrigated — have the patient look in all directions and passively open the lids. Use a topical anesthetic to control the pain and use repeat doses as needed. Irrigate for at least 15 minutes or to a volume between 500 mL and 1,000 mL. If a solid material was involved, evert the eyelids and make sure to flush everything and empty the cul de sac. After irrigation is completed, measure the conjunctival pH. The overall goal is to achieve a physiologic pH, between 7.0 and 7.2, as measured after a wait of at least five minutes after stopping irrigation.
Chemical injuries are an ophthalmologic emergency, and emergency clinicians should consult ophthalmology after initial management.
Riot Control Agents
The most commonly used riot control agents are chloroacetophenone (Mace), o-chlorobenzylidene malononitrile, and oleoresin capsicum (OC or pepper spray). They all result in conjunctival injection, blepharospasm, and lacrimation, but they also can result in permanent visual impairment.8 Exposures to these agents are treated the same way as other chemical exposures.
Airbag Injuries
Airbag deployment results in a combination of both thermal and chemical burns. In a systematic review of airbag injuries, there were 21 total injuries: 38% chemical burns and 25% thermal burns. Twenty-five percent of cases had eye involvement and 71% of those had permanently impaired eye function.9
Ultraviolet Light Injuries
These injuries occur in solderers and arc welders and are termed keratoconjunctivitis photoelectrica, or “arc eye.” Excessive exposure to the sun without appropriate ultraviolet (UV) eye protection, such as at the beach, when skiing, or while mountaineering (termed “snow blindness”), also can result in these types of corneal injuries. These patients often have a slightly delayed presentation of photophobia, lacrimation, eye pain, and blepharospasm. On examination with fluorescein stain, there is a speckled pattern to the epithelial defects.10,11 These injuries are treated the same way as corneal abrasions.
Cyanoacrylate Tissue Adhesive
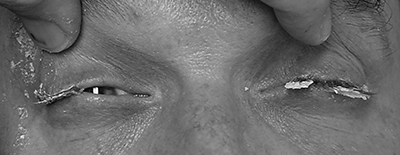
In one poison center study of cyanoacrylate exposures, 36.1% of reported exposures were ocular. (See Figure 1.) Most of the exposures were accidental and occurred in children. Although there is no specific consensus on how to manage these injuries, tissue adhesive can be removed using petroleum-based ointments, such as antibiotic ointment, mineral oil, or petroleum jelly.12
Figure 1. Eyelids Adherent Due to Cyanoacrylate Glue |
 |
|
Image courtesy of J. Stephan Stapczynski, MD |
Electrical and Lightning Injuries
Penetrating eye injuries due to impalement of the TASER barbs can result in catastrophic damage. High-voltage electrical injuries can result in ocular burns and traumatic cataracts.13 Lightning injuries, although rare, can result in a variety of injuries, from keratitis and cataracts to macular holes and maculopathy.14
Corneal Abrasions
Corneal abrasions are some of the most commonly encountered eye injuries in the emergency department (ED).15 The cornea is the clear, lens-like bulge at the anterior aspect of the eye and consists of several cellular layers that are densely innervated with pain fibers. The superficial corneal epithelium is fragile and can be damaged easily with minor trauma, most commonly a finger or fingernail. Damage to the sensitive corneal epithelium results in severe, acute eye pain.
If the abrasion is located in the visual axis, patients also may report decreased visual acuity. Patients also frequently present with complaints of tearing, foreign body sensation, a red eye, and photophobia.16
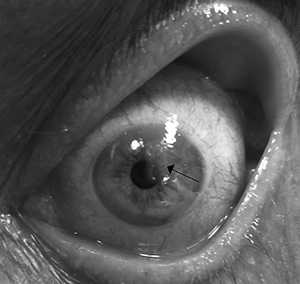
The diagnosis of a corneal abrasion is confirmed by direct visualization of the corneal epithelial surface defect, usually aided by fluorescein staining with the cobalt blue light on slit lamp microscopy. (See Figures 2 and 3.) The slit lamp is preferred because smaller abrasions may not be evident with the unaided eye using a Wood’s lamp.17 Fluorescein does not stain intact corneal epithelium but will pool in any areas of damaged corneal epithelium, allowing the examiner to visualize the defect. The defect appears as a fluorescent bright green pool of fluorescein under cobalt blue light.
Figure 2. Corneal Abrasion |
 |
|
Source: James Heilman, MD |
Figure 3. Corneal Abrasion Stained with Fluorescein Under Cobalt Blue Light |
 |
|
Image courtesy of J. Stephan Stapczynski, MD |
The eye examination can be limited because of pain, and the use of a topical anesthetic, such as tetracaine 0.5% or proparacaine 0.5%, can greatly increase patient comfort. Also, the immediate relief of pain upon instillation of these topical anesthetic drops can help with diagnosis; pain originating from the cornea is dramatically reduced by topical anesthetics, but pain from the iris is not.18
Patterns of several linear abrasions on the cornea, especially in a vertical orientation, should prompt the examiner to suspect a retained foreign body underneath the eyelid. Clinicians must remember to thoroughly inspect and evert the tarsal conjunctiva of the upper lid when assessing corneal abrasions.
The corneal epithelium heals rapidly, with most minor abrasions resolving in 24-72 hours. However, some patients can develop potentially sight-threatening complications, such as infectious keratitis, corneal scarring, and recurrent corneal erosions. While there are no current specific guidelines, the treatment for corneal abrasions is mainly directed at pain control, speeding healing, and preventing any subsequent infections (such as bacterial keratitis).
Given the potential devastating consequences of a corneal infection, prophylactic topical antibiotics are recommended for patients with corneal abrasions (i.e., erythromycin 0.5% ophthalmic ointment or sulfacetamide 10% ophthalmic ointment or solution), although there are no studies to support or refute their use.19 Patients who use contact lenses or who have abrasions associated with vegetal matter or from fingernails should have antipseudomonal antibiotic coverage (typically a fluoroquinolone, such as ciprofloxacin 0.3% or moxifloxacin 0.5%). Patients who wear contact lenses should discontinue their use until the abrasion has completely resolved.
Antibiotic treatment is aimed at preventing bacterial keratitis, most commonly caused by Staphylococcus and Streptococcus species, as well as by Pseudomonas. Several recent studies have shown growing Staphylococcus and Streptococcus resistance to ciprofloxacin and other early generation fluoroquinolones. Later generation quinolones (i.e., moxifloxacin and gatifloxacin) have shown improved gram-positive bacteria coverage.20-22 Additionally, the patient’s tetanus vaccine status should be assessed and updated as needed.
The pain from corneal abrasions can be severe and debilitating. Although healing is rapid, short-term pain control is essential in these patients. The most optimal pharmacological management is debatable; however, current treatment options include oral or topical nonsteroidal anti-inflammatory drugs (NSAIDs), topical anesthetics, and cycloplegics. Oral opioid analgesics may need to be considered in severe cases. Topical NSAIDs are available; however, these medications are more expensive and offer little pain control benefit over oral NSAIDs.23,24
Cycloplegics (i.e., cyclopentolate 1% three times per day) are used occasionally for pain control in patients with large abrasions and associated ciliary spasm with photophobia. Most recent studies, however, failed to show significant improvement in pain with the use of cycloplegics.25
Outpatient topical anesthetics have been avoided historically, largely because of the concerns of increased risk of accidental trauma and delayed healing, as shown in early case reports and animal studies. More recent studies using low concentrations of topical anesthetics for pain control in simple corneal abrasions for short durations of time did not show any increase in side effects. However, when not limited to simple abrasions, topical anesthetic use was associated with increased ED return rate in large abrasions.26 Although recent studies have shown short-duration, low-concentration topical anesthetics to be safe and effective, caution should be used, since the medications have the potential for patient abuse/misuse — and failure to ensure close follow-up can result in severe complications.27-29 Larger, more robust studies still are needed to determine the overall safety profile and rate of adverse events.30
Eye patching is no longer recommended for small abrasions because it has been shown to prolong healing times and was not shown to provide effective pain control.31 Patching should be avoided in the ED unless in consultation with ophthalmology.
All patients should be instructed to follow up with a primary care physician or ocular specialist in 24-48 hours to evaluate for improvement. Patients at risk for pseudomonal infection should be started on topical quinolone antibiotics and followed up closely, ideally within 24 hours.32 If pain and symptoms are not improving or resolved in 48 hours, patients should be referred to ophthalmology for further evaluation and treatment.
Corneal Foreign Bodies
Foreign bodies (FBs) can easily become lodged in the superficial layers of the cornea, especially when dealing with high velocity, projectile foreign bodies, such as when grinding or striking metal and when using power tools. Patients may or may not recall a traumatic event and usually present with symptoms that include acute eye pain, foreign body sensation, eye redness, lacrimation, blurred vision, and photophobia.
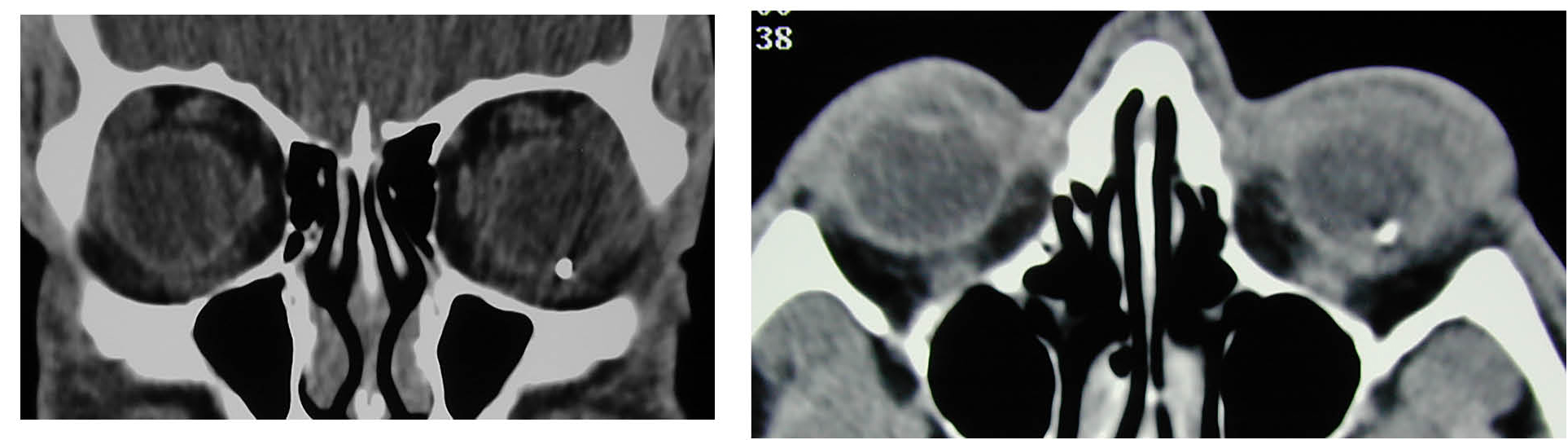
Corneal FBs usually are superficial, but globe penetration must always be excluded, especially with a concerning mechanism, as mentioned earlier. A thorough history of symptoms, mechanism of injury, and suspected type of material involved — including size, weight, and velocity — must be elicited. All patients with suspected corneal FB should have a comprehensive eye examination, including the use of slit lamp microscopy. (See Figure 4.) Slit lamp exam is particularly helpful in assessing the location, size, and depth of the FB. Removal must be deferred to an ophthalmologist for full thickness corneal penetration.33 If there is any suspicion of ocular penetration, computed tomography (CT) or ultrasonography must be obtained to exclude an intraocular FB. (See Figure 5.)
Figure 4. Corneal Foreign Body |
 |
|
Source: E van Herk |
Figure 5. CT Images of Intraocular Metallic Foreign Body |
 |
|
Image courtesy of J. Stephan Stapczynski, MD |
Corneal Foreign Body Removal
Ideally, patients should be seated upright at eye level and positioned in a slit lamp for corneal FB removal. Adequate pain control with topical anesthetics (i.e., tetracaine 0.5% or proparacaine 0.5%) is essential for successful evaluation and removal.34 Superficial FBs often can be removed by gently swiping the anterior cornea with a saline or anesthesia-soaked cotton-tipped applicator. The applicator is rotated over the surface of the cornea, and often that sweeping motion dislodges the FB.
For more adherent FBs, a foreign body spud or 25-gauge needle can be used to dislodge and remove. It is helpful to use a tangential approach until contact with the cornea when presenting instruments close to the eye, to prevent the patient from blinking. The spud or needle with bevel edge up then can be used to lift the foreign body with a sweeping motion. Care should be taken to avoid additional damage to the corneal epithelium, but corneal abrasions are common during FB removal. A Seidel test should be performed after corneal foreign body removal with a sharp instrument to ensure there is no corneal perforation.16
Management is similar to that of patients with corneal abrasions and includes ensuring an updated tetanus vaccine status, pain control, and the use of topical antibiotics.
Ferrous-containing, metallic FBs can result in deposits of iron oxide into the cornea, commonly referred to as “rust rings.” Rust can be toxic to the corneal stroma and should be removed, although this does not have to be performed emergently. Rust rings can be removed at a 24-hour follow-up in the ophthalmologist’s office. Delaying removal also can allow rust to migrate to the corneal surface, making removal easier.
After adequate corneal topical anesthesia, superficial rust rings may be removed in the ED under slit lamp guidance using a 25-gauge needle, an ophthalmic spud, or an Alger brush fitted with an ophthalmic burr. If the rust ring is located in the central visual axis, removal should be deferred to an ophthalmologist because of the risk of permanent corneal scarring.35 Any deep or prolonged burring also must be avoided in the ED.
Patients with centrally located corneal FBs, large corneal abrasions, or associated rust rings should have close, next-day ophthalmology follow-up. Other patients may follow up as needed, usually if symptoms persist longer than 48 hours.32
Hyphema
Hyphema is the accumulation of blood in the anterior chamber, most commonly from tears in the anterior face of the ciliary body.
The incidence of hyphemas is approximately 1 per 100,000 population, which is about 16,222 cases per year in the United States. Specifically, 43.8% of trauma-related hyphemas resulted after a blow to the eye from another person; 34.3% were sports-related injuries; and 15.6% were a result of a motor vehicle collision. The incidence of hyphemas increases in the summer months, likely secondary to the increased amount of time people are outdoors and engaging in recreational and sports-related activities.36
Patients typically present to the ED after a blunt eye injury with complaints of pain and decreased vision. Intraocular pressures often are elevated. The remainder of the examination may be normal initially as the blood is still freely floating. As the blood settles, the hyphema becomes more evident, and the patient’s vision may clear unless the pupil is obscured by the layering blood.
The grading scale used to characterize hyphemas serves as a diagnostic tool to communicate effectively with ophthalmologists and helps guide management and prognostication. Higher-grade hyphemas require more aggressive, acute interventions and have worse outcomes. The scale also is useful for monitoring progression and resolution of the hyphema; a sudden jump in the grade signifies a rebleed or new trauma. The grades of hyphema are:
- microhyphema: involves floating red blood cells that never settle;
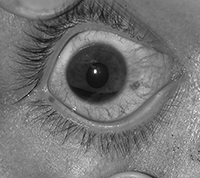
- grade I: blood that occupies less than one-third of the anterior chamber (see Figure 6);
- grade II: hyphema occupies one-third to one-half of the anterior chamber;
- grade III: hyphema occupies more than one-half of the anterior chamber;
- grade IV: hyphema occupies the whole anterior chamber and usually is referred to as an “eight-ball” hyphema.37,38 It is called an eight-ball hyphema because, traditionally, the blood in the anterior chamber takes on a dark color because of the poor circulation, making it look like the eight ball of billiards. However, in clinical practice, the blood is rarely black but rather retains a dark red color. When present, however, a true eight-ball hyphema has a much worse prognosis than a red ball hyphema.38
A Cochrane review failed to show standardized guidelines for the management of hyphemas.37 The current management largely is targeted at prevention of complications, such as rebleeds, corneal staining, and secondary glaucoma. Bed rest with head of bed elevation can help keep the layer of blood at the bottom of the anterior chamber and, thus, help keep visual acuity clear. Head elevation also will help keep blood away from the trabecular meshwork and corneal endothelium, thus helping to prevent two common complications: corneal staining and traumatic glaucoma. Eye shielding is recommended to prevent further trauma to the eye.38
Figure 6. Grade I Hyphema |
 |
|
Image courtesy of J. Stephan Stapczynski, MD |
Cycloplegics are used commonly in the treatment of hyphemas. Atropine might decrease the risk of posterior synechiae, might provide comfort to patients who present with concurrent traumatic iritis, and might decrease the risk of rebleeding. Current recommendations are to use one drop of atropine sulphate 1% solution three times a day for two weeks.38
Antifibrinolytics also are used in the treatment of hyphemas to prevent rebleeding. These work by inhibiting plasmin, which disrupts fibrinolysis. The use of oral aminocaproic acid (ACA) at a dose of 50 mg/kg every four hours for five days (maximum dose of 50 g/day) results in a very small decrease in rates of rebleeding. One drop of topical ACA 30% solution every four hours for five days has similar efficacy. Topical ACA is not commercially available in the United States.37,38 Additionally, ACA also demonstrated a longer time-to-resolution of the hyphema, caused significant nausea and emesis, and is contraindicated in pregnancy. Tranexamic acid at a dose of 25 mg/kg orally three times a day (maximum dose of 1.5 g/day) shows similar efficacy without the gastrointestinal effects of ACA.39
Corticosteroids, either topical or systemic, reduce inflammation, which may help prevent posterior synechiae and likely may stabilize the blood-ocular barrier. In one review, topical dexamethasone 0.1% four times a day was used for patients with hyphemas and showed a decrease in secondary hemorrhage.40 Importantly, the use of any of these agents described earlier must be discussed with ophthalmology before prescribing them.
Aspirin and other NSAIDs should be avoided, given their antiplatelet properties. The use of carbonic anhydrase inhibitors in settings of increased intraocular pressure (IOP) must be discussed with ophthalmology. The acidic environment that results from their use will lead to more erythrocyte dysfunction in the setting of sickle cell disease.38
High-grade or eight-ball hyphemas, patients with persistently elevated IOPs, and those with corneal staining might benefit from surgical evacuation of the hyphema.38,40 Therefore, an emergent ophthalmology consult should be obtained in these cases.
Although most patients will be monitored and managed in the outpatient setting, consider admission for those patients with sickle cell disease, taking antiplatelet or anticoagulation medications, with persistently elevated IOPs, with high-grade hyphemas, and/or with significant comorbidities. This decision should always be made in consultation with an ophthalmologist.38
Traumatic Iritis/Uveitis
Anterior uveitis refers to inflammation of the iris and ciliary body, whereas the term iritis describes inflammation isolated to the iris. Trauma is a common cause of both anterior uveitis and iritis, although the terms traumatic (anterior) uveitis and traumatic iritis often are used interchangeably. This section will focus on anterior uveitis and iritis secondary to blunt trauma. However, it is worth noting that uveitis has multiple systemic and infectious causes that will not be discussed here.
In the case of ocular blunt trauma, the mechanism of anterior uveitis and iritis is attributed to inflammation. Necrotic debris then triggers a cytokine-mediated response, which causes increased permeability of the blood vessels in the anterior chamber.41 This, in turn, leads to accumulation of white blood cells and proteinaceous material in the anterior chamber. This anterior chamber reaction causes the hallmark “cells and flare” finding commonly mentioned on eye examinations.41
Traumatic uveitis/iritis should be suspected in any patient who presents to the ED with a history of blunt ocular trauma. The most common presenting symptoms are unilateral eye pain, photophobia, eye redness, and decreased or blurred vision. Consensual photophobia and pain are from ciliary nerve irritation and spasm. Inflammatory cells and proteinaceous debris in the anterior chamber typically account for decreased visual acuity.42 Inflammatory material in the anterior chamber also can deposit on the posterior endothelium layer of the cornea, characterized as keratic precipitates, which also can lead to a decrease in visual acuity.41
The eye examination should focus on the finding of conjunctival injection with heavier concentration around the limbus. This is known as perilimbal injection or ciliary flush. Patients commonly also will have a miotic pupil as a reflexive mechanism to photophobia. A slit lamp exam is required to visualize cells and flare in the anterior chamber as well as to look for keratic precipitates (KPs). In rare circumstances, the traumatic force may be so great that pigment from the iris epithelial cells gets transferred onto the anterior lens, a finding known as a Vossius ring.43
IOP measurements can be elevated or decreased. IOP elevations can be caused by trabecular congestion from cellular debris and inflammatory products, while decreases in IOP are from an impaired aqueous humor production by ciliary damage.41,44
The management goals include reducing inflammation, preventing complications, and preserving visual acuity, all while managing pain. Topical ophthalmic corticosteroids, such as prednisolone acetate 1% solution, one to two drops every two to six hours, aid in reducing inflammation. Cycloplegics, like cyclopentolate 1% solution, one drop every eight hours, or homatropine 5% solution, one to two drops two to three times per day, relieve pain by immobilization of the iris sphincter and ciliary muscles. This immobilization provides both pain relief and stabilization to prevent further exudative leakage across into the anterior chamber. Cycloplegic agents also reduce the chance of formation of posterior synechia, a complication that occurs when adhesions form between the iris and the anterior lens. Synechiae can lead to increased IOP. In the case of significantly elevated IOPs, a topical beta-blocker, such as timolol maleate 0.5% given every 12 hours, can be used. Oral NSAIDs can be used for adjunctive pain control.
All patients with traumatic uveitis or iritis require prompt ophthalmology follow-up. Any decrease in visual acuity, abnormalities in IOPs, presence of cells and flare, or evidence of corneal keratic precipitates on slit lamp require follow-up within one to two days. Less severe cases can be followed up in four to seven days. In all cases of traumatic uveitis or iritis, ophthalmology follow-up is necessary for repeat slit lamp examination and repeat visual acuity and IOP testing.
Secondary glaucoma with permanent visual loss can develop as a result prolonged elevations in IOPs.42 Depending on repeat examination findings, the ophthalmologist will adjust the medication duration. Patients should be given strict discharge precautions not to abruptly discontinue steroid drop use until seen for follow-up by ophthalmology because sudden discontinuation can lead to a rebound inflammatory effect and delayed healing. Medication duration and taper regimen should be left to the discretion of the ophthalmologist at the time of follow-up.
Traumatic Mydriasis/Stunned Iris
In rare circumstances, blunt ocular trauma can lead to an isolated pupillary defect. Injury to the pupillary sphincter muscle can result in an isolated finding of mydriasis, a condition referred to as traumatic mydriasis or stunned iris. If the muscle is only contused, the effects will be transient, while complete tears can result in permanent mydriasis.45
In addition to patients noticing a change in their pupillary appearance and size, other presenting symptoms include blurred vision and photophobia. Prolonged mydriasis also can result in adjacent ciliary muscle strain, a condition known as accommodative asthenopia. Ciliary muscle fatigue ensues as the ciliary muscles attempt constant lens accommodation for mydriasis. This can result in eye strain, fatigue, and headaches for the patient.46
On eye examination, in addition to anisocoria, other findings include little or no reactivity in both direct and consensual light exposure of the affected eye. A decrease in visual acuity also can be present.
Although there is no emergent management of traumatic mydriasis itself, it is vital in the ED to exclude other potential causes of anisocoria. While there are medicinal and drug-related causes for mydriasis, in the event of trauma, intracranial injury should be excluded first. Traumatic mydriasis is a diagnosis of exclusion.
All patients should have ophthalmology follow-up within a week. Patients with partial and complete tears can be offered surgical and laser therapy options. Temporary corrective lenses also often are needed to relieve ciliary muscle strain.
Iridodialysis
Iridodialysis is defined as the iris detaching from the ciliary body. This defect is a common finding after ocular blunt trauma, but exact data on its incidence are lacking.47
Patients usually will present after ocular injury with complaints of pain, diplopia, blurry vision, photophobia, or even vision loss.48,49 Some also may complain of having a “second pupil,” referring to the defect created by the iridodialysis, which may appear like an eccentric pupil.
On examination, patients typically will have decreased visual acuity. An irregularly shaped iris is seen where the iridodialysis is present. This may only be evident during a slit-lamp examination for small defects. The slit-lamp examination also is important to assess for secondary injuries when an iridodialysis is present. Hyphemas are seen commonly because of the damage to the ciliary body’s vessels when the iris pulls away from its insertion site.
Ultrasonography can help assess for concurrent damage to posterior segment structures. CT scans can help assess for foreign body involvement in cases of penetrating trauma and for concomitant facial injuries.48
Patients will need an emergent consult to ophthalmology, since large defects often will require iridoplasty.47
Traumatic Vitreous Hemorrhage
Posterior eye structures, such as the retina and the posterior vitreous, are susceptible to injury from blunt trauma.
The vitreous humor is a jelly-like, avascular structure in the posterior pole of the eye with attachments at the ora serrata anteriorly and the optic disc posteriorly. Traction at vascular areas due to trauma or neovascularization results in tears of the superficial retinal vessels and hemorrhage into the vitreous. Vitreous hemorrhage (VH) also can be associated with posterior vitreous detachments and with retinal detachments (RDs).
The presentation of a traumatic VH is that of sudden onset of painless vision loss or decreased visual acuity. Patients often report the appearance of black or red spots or cobweb-like floaters in the vision. On fundoscopic examination, the red reflex may be absent in severe cases, and blood in the vitreous can be seen obstructing the view of the retina. Bedside ultrasonography is a useful tool for evaluating the posterior pole, especially when blood may be obscuring the view of the fundus.
Ocular ultrasound, also known as B-scan, can help distinguish between RD and VH. On ultrasound, VH appears as bright, freely moving, echogenic material in the posterior chamber within the vitreous. (See Figure 7.) VH will have a semi-mobile, wave-like appearance with eye movements that has been likened to swaying seaweed or laundry in a washing machine.32 In RD, the retina can be traced back to its insertion, as it remains tethered to the optic disc.
Figure 7. Ultrasound Vitreous Hemorrhage |
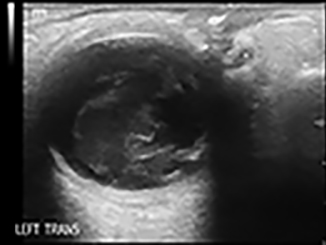 |
|
Image courtesy of Steven M. Field, DO. |
For isolated traumatic VH, intravitreal blood usually absorbs within a few weeks to a month. The treatment is directed at reducing any risk for rebleeding (i.e., bed rest, elevated head of bed, elimination of any anticlotting drugs).32 Patients usually are monitored closely as outpatients for the first two to three days with an urgent ophthalmology referral to ensure there is no progression of bleeding. Ophthalmology can be consulted emergently if there is associated RD or with elevated IOP.
Delayed Presentations and Complications
There are various disease entities that can present days to years after an initial blunt ocular injury. Glaucoma is a common delayed complication. Trauma leads to inflammation and adhesions of the trabecular meshwork, causing chronic elevations in the IOP. Optic nerve atrophy can result either from direct trauma or chronic traumatic glaucoma.38 Traumatic cataracts result from inflammatory cells damaging the lens and causing its opacification.50 Hyphemas can result in rebleeding, since the vessels are damaged and fragile. They also can cause corneal staining leading to permanent vision changes.38 Foreign bodies and burns can cause corneal scarring that might be permanent. Retinal detachments and vitreous detachments also are common after trauma to the eye, due to retinal breaks and adhesions. Sympathetic ophthalmoplegia is a type of panuveitis that presents in the opposite eye from the one that suffered trauma. It is a rare entity that can result years after a trauma.51 All of these delayed presentations can present as photophobia, pain, and vision changes. Inquiring about patients’ prior history of trauma will help the emergency clinicians in reaching the correct diagnosis.
Special Populations
Pediatric Patients
Assessing ocular trauma in pediatric patients is complicated by the challenge in obtaining patient cooperation required for a complete eye examination.
Visual acuity testing should be attempted prior to the remainder of the eye exam, when the patient likely is most calm. A modified Snellen chart containing pictures of objects familiar to children is available for preschool-age patients or those patients who are not yet able to recognize letters. The patient can be given a toy or electronic device for distraction.52 Having a patient sit with the parent during the exam and explaining to the patient what you are about to do can be helpful sometimes.
Crying children are more likely to open their eyes in a darkened room. The light intensity of the ophthalmoscope should be kept to a minimum.53 Physicians should take care to minimize unnecessary distress because crying can increase ocular pressures and lead to extrusion of intraocular contents in patients with ruptured globes. If the patient is uncooperative after all of the described techniques, manual restraint with sedation can be used, especially if the patient needs emergent therapeutic interventions or if there is reasonable suspicion of a sight-threatening injury.
Most guidelines for ophthalmology follow-up and consultation parallel those used for adults. However, there are a few exceptions worth noting. Children have a more cartilaginous bone structure, and the increased elasticity makes the pediatric population more prone to orbital fractures that can lead to entrapment.53 Post-traumatic motility dysfunction places these patients at high risk for amblyopia and lifelong diplopia.53 Emergent ophthalmology consultation should be sought in these cases, even if this involves transfer to another facility.
One final consideration for pediatrics is blunt eye injury secondary to nonaccidental trauma. Studies have suggested that 4% to 6% of abused children will present with a primary eye complaint.53 While retinal hemorrhages are most common in children younger than 2 years of age and can present in up to 80% of infants with abusive head trauma (previously known as shaken baby syndrome), retinal hemorrhages are present in up to 23% of cases of physically abused children across all age groups.53 For this reason, nonaccidental trauma should be on the differential for any pediatric eye injury. If other physical examination findings, such as rib fractures or long bone injuries, are found, ophthalmology should be consulted from the ED to exclude associated ocular injuries.
Communication Barriers or Developmental Delays
The approach to patients with communication barriers or developmental delays is much the same as it is for pediatric patients. Elderly patients with cognitive impairment or hearing difficulties and patients of any age with existing developmental delays or prior history of traumatic brain injury might not be able to provide a detailed history or have insight to their current condition. Since the history by the patient often will be limited, caretakers, family members, and social workers should be involved early.
Elderly patients frequently will have baseline visual impairment, such as glaucoma or cataracts, or they may require corrective lenses. Seeking out collateral when available can assist in determining new from old visual impairment. Minimizing external stimulation in the environment and having someone the patient knows at the bedside can be helpful in keeping patients calm. When a sight-threatening injury is suspected, an anxiolytic or sedative agent can be considered to complete the eye exam. In some cases, procedural sedation may be necessary.
This group of patients will require more detailed discharge planning. Understanding the patient’s living environment and who will be in charge of home care and assisting with follow-up is essential to minimizing potential complications.53 Of note, there also should be a high index of suspicion for nonaccidental trauma in this population.
Anticoagulated Patients, Sickle Cell Disease
Patients with sickle cell trait and disease and patients on anticoagulation can present unique challenges, especially related to the treatment of hyphemas.
Any African American patient presenting to the ED with a traumatic hyphema should be asked if they have sickle cell disease or trait. Both carriers of the trait and patients with the disease are predisposed to sickling of red blood cells in the anterior chamber. These cells obstruct the trabecular meshwork and lead to significant increases in IOP. As IOP increases, hypoperfusion, hypoxia, and hypercapnia ensue, further exacerbating the sickling of cells.53 As mentioned in the discussion of hyphemas, both carbonic anhydrase inhibitors and hyperosmolar agents should be avoided because they exacerbate sickling by lowering pH and inducing hemoconcentration.54
Patients with hyphemas who are on anticoagulation or are taking aspirin have an increased risk of rebleeding. While the reported rate of rebleeding varies depending on different studies, two to five days after the initial injury is suggested to be the time of highest risk for rebleed.53 Aspirin and anticoagulation should be held until the hyphema resolves or surgical intervention is possible. In those patients with larger hyphemas who are not surgical candidates, resolution sometimes can take weeks to months. In these cases, ophthalmology and other specialists must weigh the risk versus benefits of withholding anticoagulation and the timeline for restarting anticoagulation.
Intubated Patients
In trauma patients who arrive intubated or who are intubated shortly after arrival, it becomes imperative to ensure there is no oversight to potential ocular injuries. The mechanism of injury often will be limited to what is provided by the accompanying prehospital providers or family. Any history of head or facial trauma should heighten the concern for associated ocular injury. However, a lack of history should not prevent emergency clinicians from performing a basic eye examination.
Once all life-threatening injuries have been addressed and managed, details should be noted to any signs of potential orbital injury. Any facial bony instability suggestive of fracture should further increase this suspicion, since facial fractures have an increased association with ocular injury.55 Although visual acuity cannot be tested, a careful inspection should take place to look for pupillary defects or irregularities, proptosis, periorbital ecchymosis, swelling or crepitus, conjunctival injection, foreign bodies, lid lacerations or contusions, and signs of entrapment. The forced duction test, which has been described previously, can be used to assess for entrapment. However, some studies suggest that succinylcholine interferes with test results for up to 20 minutes after intubation due to sustained ocular muscle contraction.56
Fluorescein staining should be performed on all intubated trauma patients to ensure there are no corneal abrasions, corneal lacerations, or foreign bodies. Fluorescein staining is especially important in those patients with thermal burns or chemical injuries. Once any potential open globe injury has been ruled out, IOPs can be obtained. It also is worth noting that some interventions, such as the use of ketamine or succinylcholine, pressure from bag valve mask, inadequate ventilation with elevated end tidal CO2, and positive pressure ventilation, can cause transient elevations in IOPs.32 Ultrasound is a helpful adjunct in identifying any signs of retinal detachment, lens injury, or vitreous hemorrhage.
Any physical examination or CT findings suggestive of retrobulbar hematoma should warrant an examination to exclude orbital compartment syndrome. As mentioned before, failure to act promptly can result in permanent sight loss for the patient.
Prevention
Using eye protection at work and during high-risk sports and other recreational activities has been shown to prevent more than 90% of blunt ocular injuries.57 Recreational activities, such as paintball and other toy gun fights, the use of bungee and other elastic cords and bands, and home remodeling and gardening, also are considered high-risk activities.
Athletes should undergo basic visual examination to ensure that they have adequate visual acuity before engaging in sports. Both the American Academy of Pediatrics and the American Academy of Ophthalmology have recommendations for use of protective eyewear during sports activities.58 The protective eyewear must prevent ocular damage without causing secondary facial or intracranial damage. Typically, these protective glasses are made of polycarbonate, which is a stronger form of plastic and can be made with prescription or nonprescription lenses.
The American National Standards Institute (ANSI) has minimum acceptable standards for eyewear protection from occupational hazards and should also be used when engaging in home improvement projects. Protective eyewear with this certification can be found at most hardware stores. Contact lenses and regular eyeglasses do not provide adequate protection from ocular injuries.59
REFERENCES
- Zagelbaum BM. Sports-related eye trauma. Phys Sportsmed 1993;21:25-42.
- Zagelbaum BM, Tostanoski JR, Kerner DJ, Hersh PS. Urban eye trauma. A one-year prospective study. Ophthalmology 1993;100:851-856.
- Kuckelkorn R, Schrage N, Keller G, Redbrake C. Emergency treatment of chemical and thermal eye burns. Acta Ophthalmol Scand 2002;80:4-10.
- Kam KW, Patel CN, Nikpoor N, et al. Limbal ischemia: Reliability of clinical assessment and implications in the management of ocular burns. Indian J Ophthalmol 2019;67:32-36.
- Lubeck D, Greene JS. Corneal injuries. Emerg Med Clin North Am 1988;6:73-94.
- Kuckelkorn R, Kottek A, Schrage N, Reim M. Poor prognosis of severe chemical and thermal eye burns: The need for adequate emergency care and primary prevention. Int Arch Occup Environ Health 1995;67:281-284.
- Morgan LB. A new drug delivery system for the eye. IMS Ind Med Surg 1971;40:11-13.
- Kim YJ, Payal AR, Daly MK. Effects of tear gases on the eye. Surv Ophthalmol 2016;61:434-442.
- Erpenbeck SP, Roy E, Ziembicki JA, Ergo FM. A systematic review on airbag-induced burns. J Burn Care Res 2020; Oct 22;iraa186. [Online ahead of print].
- Korzeniewski K. Eye diseases in travelers. Int Marit Health 2020;71:78-84.
- McIntosh SE, Buercio B, Tabin GC, et al. Ultraviolet keratitis among mountaineers and outdoor recreationalists. Wilderness Environ Med 2011;22:144-147.
- Carstairs SD, Koh C, Qian L, et al. Sticky situations: cyanoacrylate exposures reported to a poison control system. Clin Toxicol (Phila) 2017;55:1001-1003.
- Bae EJ, Hong IH, Park SP, et al. Overview of ocular complications in patients with electrical burns: An analysis of 102 cases across a 7-year period. Burns 2013;39:1380-1385.
- Pradhan E, Khatri A, Ahmed AA, et al. Lightning injury to eye: Brief review of the literature and case series. Clin Ophthalmol 2020;14:597-607.
- Channa R, Zafar SN, Canner JK, et al. Epidemiology of eye-related emergency department visits. JAMA Ophthalmol 2016;134:312-319.
- Ahmed F, House RJ, Feldman BH. Corneal abrasions and corneal foreign bodies. Prim Care 2015;42:363-375.
- Hooker EA, Faulkner WJ, Kelly LD, Whitford RC. Prospective study of the sensitivity of the Wood’s lamp for common eye abnormalities. Emerg Med J 2019;36:159-162.
- Mihora LD, Enzenauer RW. Traumatic corneal abrasions: A review of the current literature. Curr Surg 2003;60:487-492.
- Fusco N, Stead TG, Lebowitz D, Ganti L. Traumatic corneal abrasion. Cureus 2019;11:e4396.
- Mah FS. Fourth-generation fluoroquinolones: New topical agents in the war on ocular bacterial infections. Curr Opin Ophthalmol 2004;15:316-320.
- Donnenfeld E, Perry HD, Chruscicki DA, et al. A comparison of the fourth-generation fluoroquinolones gatifloxacin 0.3% and moxifloxacin 0.5% in terms of ocular tolerability. Curr Med Res Opin 2004;20:1753-1758.
- Kowalski RP, Dhaliwal DK, Karenchak LM, et al. Gatifloxacin and moxifloxacin: An in vitro susceptibility comparison to levofloxacin, ciprofloxacin, and ofloxacin using bacterial keratitis isolates. Am J Ophthalmol 2003;136:500-505.
- Wakai A, et al. Topical non-steroidal anti-inflammatory drugs for analgesia in traumatic corneal abrasions. Cochrane Database Syst Rev 2017;5:CD009781.
- West JR. Are topical nonsteroidal anti-inflammatory drugs useful for analgesia in patients with traumatic corneal abrasions? Ann Emerg Med 2019;73:157-159.
- Meek R, Sullivan A, Favilla M, et al. Is homatropine 5% effective in reducing pain associated with corneal abrasion when compared with placebo? A randomized controlled trial. Emerg Med Australas 2010;22:507-513.
- Shipman S, Painter K, Keuchel M, Bogie C. Short-term topical tetracaine is highly efficacious for the treatment of pain caused by corneal abrasions: A double-blind, randomized clinical trial. Ann Emerg Med 2020;77:338-334.
- Puls HA, Cabrera D, Murad MH, et al. Safety and effectiveness of topical anesthetics in corneal abrasions: Systematic review and meta-analysis. J Emerg Med 2015;49:816-824.
- Swaminathan A, Otterness K, Milne K, Rezaie S. The safety of topical anesthetics in the treatment of corneal abrasions: A review. J Emerg Med 2015;49:810-815.
- Waldman N, Winrow B, Densie I, et al. An observational study to determine whether routinely sending patients home with a 24-hour supply of topical tetracaine from the emergency department for simple corneal abrasion pain is potentially safe. Ann Emerg Med 2018;71:767-778.
- Yu CW, Kirubarajan A, Yau M, et al.Topical pain control for corneal abrasions: A systematic review and meta-analysis. Acad Emerg Med 2021; Jan 28. doi: 10.1111/acem.14222. [Online ahead of print].
- Lim CH, Turner A, Lim BX. Patching for corneal abrasion. Cochrane Database Syst Rev 2016;7:CD004764.
- Ehlers JP, Chirag P. In: Bagheri N, Wajda B, Calvo C, Durrani A, eds. The Wills Eye Manual: Office and Emergency Room Diagnosis and Treatment of Eye Disease. 7th ed. Lippincott Williams and Wilkins; 2016.
- Walker RA, Adkhikari S. Eye emergencies. In: Tintinalli JE, Yealy DM, Meckler GD, et al, eds. Tintinalli’s Emergency Medicine: A Comprehensive Study Guide. McGraw-Hill; 2021: 1523.
- Babineau MR, Sanchez LD. Ophthalmologic procedures in the emergency department. Emerg Med Clin North Am 2008;26:17-34, v-vi.
- Fraenkel A, Lee LR, Lee GA. Managing corneal foreign bodies in office-based general practice. Aust Fam Physician 2017;46:89-93.
- Zafar S, Canner JK, Mir T, et al. Epidemiology of hyphema-related emergency department visits in the United States between 2006 and 2015. Ophthalmic Epidemiol 2019;26:208-215.
- Gharaibeh A, Savage HI, Scherer RW, et al. Medical interventions for traumatic hyphema. Cochrane Database Syst Rev 2019;1:CD005431.
- Bansal S, Gunasekeran DV, Ang B, et al. Controversies in the pathophysiology and management of hyphema. Surv Ophthalmol 2016;61:297-308.
- Deans R, Noël LP, Clarke WN. Oral administration of tranexamic acid in the management of traumatic hyphema in children. Can J Ophthalmol 1992;27:181-183.
- Walton W, Von Hagen S, Grigorian R, Zarbin M. Management of traumatic hyphema. Surv Ophthalmol 2002;47:297-334.
- Pinch CM. Evaluation of traumatic uveitis in the emergency department. Adv Emerg Nurs J 2019;41:122-128.
- Mahabadi N, Kim J, Edens MA. Iritis. StatPearls 2021; StatPearls Publishing.
- Seth NG, Thattaruthody F, Pandav SS. Vossius ring after blunt ophthalmic trauma. Ophthalmol Glaucoma 2019;2:54.
- Gutteridge IF, Hall AJ. Acute anterior uveitis in primary care. Clin Exp Optom 2007;90:390; author reply 390.
- Serrano F, Stack LB, Thurman RJ, et al. Traumatic eye injuries: Management principles for the prehospital setting. JEMS 2013;38:56-62.
- Young CC, Simons KB, Jacobs B. Traumatic mydriasis: “Cat’s eye” in a football player. Clin J Sport Med 2006;16:179-180.
- Silva JL, Póvoa J, Lobo C, Murta J. New technique for iridodialysis correction: Single-knot sewing-machine suture. J Cataract Refract Surg 2016;42:520-523.
- Hsiao CC, Chen WL. Traumatic tridodialysis. N Engl J Med 2019;380:1463.
- Knoop KJ, Palma JK. Iridodialysis. In: Koop KJ, Stack LB, Storrow AB, Thurman RJ, eds. The Atlas of Emergency Medicine. 5th ed. McGraw-Hill; 2020.
- Kuhn F. Traumatic cataract: What, when, how. Graefes Arch Clin Exp Ophthalmol 2010;248:1221-1223.
- Chang GC, Young LH. Sympathetic ophthalmia. Semin Ophthalmol 2011;26:316-320.
- Silverstein E. How to conquer the pediatric eye exam. June 14, 2018. American Academy of Ophthalmology. https://www.aao.org/young-ophthalmologists/yo-info/article/how-to-conquer-the-pediatric-eye-exam
- Kuhn F, Pieramici DJ. Ocular Trauma: Principles and Practice. Thieme; 2002.
- Leffler CT, Bharucha G, Donaldson J. Management of hyphema in patients with sickle cell disease or trait. Surv Ophthalmol 2016;61:689-690.
- Johnson NR, Singh NR, Oztel M, et al. Ophthalmological injuries associated with fractures of the orbitozygomaticomaxillary complex. Br J Oral Maxillofac Surg 2018;56:221-226.
- France NK, France TD, Woodburn JD Jr, Burbank DP. Succinylcholine alteration of the forced duction test. Ophthalmology 1980;87:1282-1287.
- Mishra A, Verma AK. Sports related ocular injuries. Med J Armed Forces India 2012;68:260-266.
- American Academy of Pediatrics, Committee on Sports Medicine and Fitness; American Academy of Ophthalmology, Eye Health and Public Information Task Force. Protective eyewear for young athletes. Ophthalmology 2004;111:600-603.
- Rodriguez JO, Lavina AM, Agarwal A. Prevention and treatment of common eye injuries in sports. Am Fam Physician 2003;67:1481-1488.
Part I of this article reviewed nonpenetrating ocular trauma that presents a severe threat to vision. Part II will discuss potential vision threats of nonpenetrating ocular trauma, including burns, corneal abrasions, corneal foreign bodies, and hyphemas.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
