Pulmonary Embolism: Recent Advances in Diagnosis and Treatment Modalities, Part II
Part II — Differential Diagnoses
Author: Marianne Haughey, MD, Assistant Professor of Emergency Medicine, Jacobi Hospital, Albert Einstein College of Medicine, Bronx, NY.
Peer Reviewers: Brian K. Snyder, MD, Assistant Clinical Professor of Medicine, Department of Emergency Medicine, University of California, San Diego; and Albert C. Weihl, MD, FACEP, Assistant Professor of Surgery- Emergency Medicine, Yale University School of Medicine, New Haven, CT.
The differential diagnosis of pulmonary embolism (PE) is broad and includes many life-threatening illnesses that must be identified promptly to intervene appropriately. These high-stakes alternatives include myocardial infarction, pneumonia, pneumothorax, and sepsis, among other illnesses. The physician has the difficult task of narrowing the differential list to the correct illness. The more quickly this is done, the more likely it is that definitive treatment can be initiated in time to save the patient.
The most difficult step in treating a patient who has PE is identifying the patient who requires treatment. Multiple options are available for treating a patient with known PE, some of which demonstrate significant changes from past practice. Treatment of PE has become more refined during the past few years, with the applicability of thrombolytics and the availability of low molecular weight heparins arising as exciting developments in the field. Other time-tested treatments, such as unfractionated heparin and surgically placed filters, still have significant roles.
The emergency physician is in the unique position of seeing many patients in whom PE can be prevented. As the commonly used portal to inpatient service, emergency physicians are able to address the treatments that can prevent this life-threatening illness in patients who will be hospitalized for other reasons. Preventing PE in a high-risk patient can be a lifesaving intervention.
Part one of this two-part series addressed the epidemiology, clinical presentation, and the appropriate use of diagnostic studies to identify those with PE. Unfortunately, no one exam is perfect in detecting PE. The exams assessing for PE, the clinical exam, and the exams that eliminate or identify other illnesses on the differential list can be used in combination. This is the most effective way to approach a patient with possible PE.
This second and final part of the series covers the topics of differential diagnosis that must be considered when a patient presents with symptoms consistent with PE, treatment, and considerations for prevention of this disease state.—The Editor
Differential Diagnosis
PE can present with a wide range of severity. Patients may present in a premorbid state of severe shock, with the differential diagnosis including all of the elements of the shock differential. The symptoms of PE in a stable patient with small emboli may be so protean and vague that they elicit a wide-ranging differential diagnosis for the presenting symptom, whether it is chest pain, respiratory difficulties, low-grade fever, or a feeling of anxiety. (See Table 1.)
Table 1. Differential Diagnosis of Patient with Symptoms of Pulmonary Embolism
Certainly, the symptoms alone of chest pain and shortness of breath immediately bring to mind a varied group of illnesses, many of which are life-threatening. The advantage of obtaining many of the tests that are not specific for PE is that they may identify an alternate diagnosis. It is important to distinguish diagnoses on the differential that contraindicate the use of anticoagulation as treatment for PE, such as pericarditis or aortic dissection.1
A patient in shock from a large embolus presents as a patient who is hypotensive with possible respiratory difficulties. Up to 8% of patients with PE present in extremis.2,3 The differential in this patient can include sepsis or shock from pump failure perhaps due to a myocardial infarction or severe congestive heart failure (CHF). Hemorrhagic shock also can present in this way. Neurogenic shock and anaphylactic shock are likely to present with other clinical scenarios. Treatment for these illnesses does have some similarities, such as airway control and blood pressure support, but there are some significant differences. As discussed in the first part of this series, the electrocardiogram (ECG) and chest x-ray are exams with low sensitivity and specificity for PE. Their usefulness in identifying and excluding other life-threatening illnesses, however, is essential to providing appropriate care. When a large myocardial infarction (MI) is discovered on the ECG of a patient with shock, definitive treatment can begin. If CHF or pneumonia is found on the chest x-ray, diuretics, antibiotics, or other appropriate, specific treatment can start. It is important to remember that the patient who presents in shock with a mostly normal ECG and normal or nearly normal chest x-ray may well have PE and needs further workup to evaluate the possibility.
The differential of the less acutely ill patient is broad. Symptoms such as dyspnea and/or chest pain can suggest illnesses that are primarily pulmonic in origin, such as asthma, pleurisy, pneumonia, or pneumothorax. Chest pain as the primary complaint can bring to mind cardiac ischemia, pericarditis, aortic dissection, pulmonary disease, or musculoskeletal pain. Patients presenting with tachypnea can have any of these above illnesses, but metabolic acidosis and anxiety also must be considered. Of course, thrombus is not the only possible source for emboli. Fat may embolize from a fracture, air may embolize during a procedure, bullets or other foreign bodies may embolize, and pregnancy allows for the possibility of amniotic fluid embolism. These emboli may present similarly to a thrombotic embolism, but the treatment will differ because anticoagulation will not cure any of these other types of emboli.
Because the differential diagnosis list is extensive and broad, it is imperative that the physician assess an individual patient’s risk for thromboembolic disease and consider it in the diagnostic workup of symptomatic patients, as described in part one of this article. It also is important to realize that it may be easy to dismiss a patient who appears anxious with complaints of dyspnea or chest pain once the other dangerous diagnoses are excluded, but the small PEs that may be causing these symptoms are a sign that a larger, life-threatening PE may be in the patient’s near future unless prevented by the astute physician.
Treatment Options
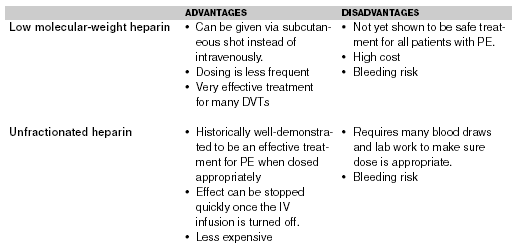
Once the difficult task of determining that a patient needs treatment for PE has been accomplished, different treatment options may be considered. The first task in treatment is to determine whether the current clot burden is such that immediate measures must be taken to remove the clot, either chemically through thrombolysis or mechanically via embolectomy. Once that decision is made, consideration must be given to anticoagulation, either as primary therapy or as prevention against future emboli. The choices among the anticoagulants include unfractionated heparin (UFH), low molecular weight heparin (LMWH), and oral anticoagulation with warfarin. Long-term prevention of future emboli may be achieved with anticoagulation or with filters that prevent the clot from traveling centrally. Each of these options will be discussed fully in the following sections. At each juncture, it is essential to consider the patient’s severity of illness as well as the indications and contraindications to each method of therapy. (See Table 2.)
Table 2. Low Molecular-Weight Heparin vs. Unfractionated Heparin
Heparin and Unfractionated Heparin. Since the 1930s, the treatment of thromboembolic disease has been based on anticoagulation.4 PE is a potentially fatal disease in which anticoagulant therapy has been shown to improve outcomes dramatically.5,6 In 1960, Barrit and Jordan established, with the first randomized trial, that those who were anticoagulated clearly had an improvement compared to those who received placebo.6
Standard treatment for patients with venous thromboembolism had been hospital admission, with UFH given by intravenous (IV) infusion for 5-10 days. Oral anticoagulation is begun in the hospital overlapping with the UFH, and then continues for at least three months.7-12 This has been the treatment plan for both DVT and PE. This treatment may not aid those with hemodynamic compromise on presentation quickly enough, but it was the only medical treatment available for a long time.
The usefulness of UFH in patients with PE is that it prevents further clot formation by accelerating the action of antithrombin III, allowing endogenous fibrinolysis to lyse the existing clot.1 The heparin can be administered subcutaneously, but most often is administered intravenously.1 Subcutaneous dosing can result in more erratic absorption of a medication that must be titrated carefully to be therapeutic. Achieving a therapeutic range quickly enough is one of the greatest concerns when deciding to use heparin for treatment of a patient with suspected PE. Patients who are anticoagulated adequately within the first 24 hours have better outcomes. It has been found that those who are anticoagulated adequately during that time period have a PE recurrence rate of only 4-6%, as opposed to a recurrence rate of 23% in those who are not anticoagulated in the first day.13
Dosing regimens have moved away from a standard dosing scheme with adjustments made over time according to laboratory exams used as a guidepost for titration. The suggested regimen now is one that is weight-based. First, a bolus is given with suggested doses ranging from 80 IU/kg to 150 IU/kg.1,4 The bolus is followed by a drip that is begun at 18 IU/ kg. Activated partial thromboplastin (PTT) levels then are monitored every 4-6 hours, with the drip adjusted appropriately. The goal is for the PTT to be 1.5 times the control value.4
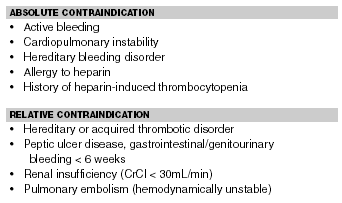
Anticoagulation is not an innocuous intervention, however. Disadvantages to using UFH include repeated lab work to check the level of anticoagulation frequently, required hospitalization of the patient, and a risk of bleeding. The frequency of major bleeding is 4% in those treated with continuous IV UFH with a weight-based dosage system for PE or DVT.14 Intracranial bleeding, retroperitoneal bleeds, and gastrointestinal bleeds are considered major bleeds. Those at higher risk for bleeding include patients who have had surgery within the past 14 days and patients with a history of peptic ulcer disease, gastrointestinal tract bleeding, genitourinary tract bleeding, disorders predisposing to bleeding, major thrombotic stroke within the previous 14 days, or severe thrombocytopenia.11,15 It is essential to identify patients who have diagnoses that would preclude the use of anticoagulation and to treat them with alternate means. (See Table 3.) The risk of bleeding also increases with age.16 The risk of recurrent VTE with standard UFH treatment is 6.8%.17 A particular advantage of heparin is that is safe for pregnant patients.
Table 3. Low Molecular-Weight Heparins
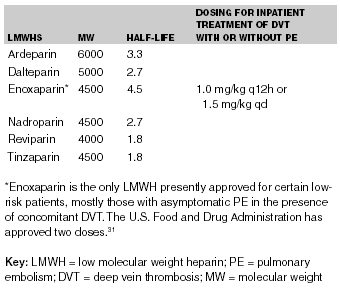
Low Molecular-Weight Heparins. LMWHs have been used in Europe for more than 10 years, although they are relatively new in the United States.18 LMWHs differ from standard UFH in that they have greater bioavailability, a longer half-life, a more predictable anticoagulant response when administered subcutaneously in fixed doses, and a higher ratio of anti-factor Xa to anti-factor IIa activity.19-22 These differences occur because the LMWHs bind less than UFH to plasma proteins, macrophages, and proteins released from activated platelets and endothelial cells.23
LMWHs, although often discussed as a group, are distinct compounds with different pharmacological profiles and dose regimens; it is uncertain whether the results obtained with one preparation can be extended to another.5 (See Table 4.) The U.S. Food and Drug Administration (FDA) regards LMWHs as individual drugs that cannot be used interchangeably.24 They are distinct entities that have the potential for clinically significant differences in safety and efficacy. The different LMWHs probably are equivalent for low-dose indications, such as venous thromboprophylaxism, but at higher doses the efficacy and safety differences cannot be excluded.24 Since November 2000, there have been two LMWHs approved by the FDA for use in treating VTE in the United States: enoxaparin and tinzaparin.1 In addition, dalteparin now is approved for treatment of DVT.25 The longer half-life of LMWHs and the more predictable anticoagulant response make them suitable for subcutaneous administration without laboratory monitoring.26-28 If it is determined that monitoring is needed in a particular patient, anti Xa levels can be measured, although few labs conduct this exam.
Table 4. Contraindications to Heparin Use
Multiple studies now have demonstrated the safety and efficacy of LMWH treatment in properly selected patients with DVT,21,29-33 as well as patient satisfaction with this treatment.32 LMWHs have allowed the possibility of either abbreviated inpatient stays35 or outpatient treatment of DVT.29,32-34 Patients who are proper candidates for outpatient treatment of DVT should have normal vital signs, a low bleeding risk, and well-functioning kidneys. Their home situation must make administration of the medication practical. Monitoring of blood work and appropriate follow-up on an outpatient basis are required.33
A Canadian study compared costs of treatment with LMWH and UFA for DVT. The costs measured included hospitalization, travel, home care, medication, lab work, and lost productivity for the patient and caregiver. For patients with acute proximal DVT, the researchers found that treatment at home with LMWH was less costly than treatment in the hospital with UFH. They also found that this savings occurred without compromising clinical outcomes or the patients’ quality of life.30
Are LMWHs safe and appropriate treatments for those with PE? There is no large, randomized study that has demonstrated that patients with symptomatic PE safely can be treated with LMWH as a bridge to further anticoagulation. As has been stated previously, PE presents with significant variation in severity, presumably linked to the amount of clot and the ability of the patient to compensate. PE has been found in 50% or more of patients with DVT.17 Some studies have examined the treatment of PE with LMWH by evaluating patients with defined DVT and then looking for PE. Researchers then evaluated the resolution of the signs of PE on follow up V/Qs and found LMWH equal in efficacy to UFH.1,5,26 Most of these patients were asymptomatic for PE.
Tinzaparin and enoxaparin are FDA approved for inpatient treatment of patients with DVT as the primary presentation of their thromboembolic disease who also are found to have PE.1 Dalteparin is approved only for treatment of those with DVT.25 Treatment success of patients with DVT and PE that is found incidentally cannot be assumed to correlate with treatment of patients with symptomatic PE.
Two studies specifically evaluated the use of LMWH as a treatment for symptomatic PE as a bridge to warfarin treatment. Patients with diagnosed PE who were not candidates for thrombolysis (the criteria for thrombolysis were not described in the studies) and had no contraindications to anticoagulation were treated with LMWH or UFH. One study group used reviparin and the other study used tinzaparin.5,26 Both studies concluded that the LMWH was as safe and effective in treating PE as UFH when used as delineated in the study. The patients remained in the hospital for their treatment. No statement about outpatient treatment of PE was confirmed by either of these studies.5,26
Another study that using tinzaparin looked at 432 patients with symptomatic DVT.17 The patients were randomized to receive either tinzaparin or UFH. Patients were excluded from the study for the following reasons: acute bleeding or bleeding disorders; pregnancy; allergy to heparin, bisulfites, or fish; two or more previously documented episodes of PE or DVT; a history of protein C defiency; a history of heparin-associated thrombocytopenia; severe malignant hypertension (HTN); severe hepatic failure; or severe renal failure needing dialysis. All of the patients underwent baseline lung scanning, and 200 were found to have high-probability scans. Only 28 of those patients had any symptoms of PE. Of those 28, only 13 were randomized to receive LMWH.17,36 The patients in the LMWH group were hospitalized as long as the UFH group. The author concluded that tinzaparin once per day was no less effective—and probably more effective—than use of dose-adjusted intravenous UFH for preventing recurrent VTE in patients with DVT and associated PE. The heparin was not dosed in a weight-related fashion, whereas the LMWH was weight-dosed. This is not a result that can be generalized to symptomatic patients with PE.
Another study evaluated the safety of outpatient treatment of PE with LMWH.37 It is a small study, only 108 patients were followed for at least partial outpatient treatment. The 50 patients who had PE and were excluded had one of the following exclusion criteria:
- another medical condition requiring admission;
- active bleeding or a high risk of major bleeding;
- hemodynamic instability;
- pain requiring parenteral narcotics;
- requirement for O2 to keep O2 saturation above 90%;
- age younger than 18 years; or
- likelihood of poor compliance.
Researchers found a recurrent event rate of 5.6%, which is similar to other studies treating PE with LMWH.5,26,37 The truly ill patients with PE, however, were excluded from the study as they met the exclusion criteria.
As tempting as it may be to pursue the thought of outpatient treatment of PE with the LMWHs, the data do not yet demonstrate that it is a safe way to treat those with symptomatic PE. The FDA has approved two LMWHs for outpatient treatment of acute DVT without PE as a bridge to wafarin.36 Enoxaparin is approved for the treatment of DVT with or without PE on an inpatient basis.38 The strategies of either an abbreviated hospital stay or treatment with LMWHs for symptomatic PE, however, remain without FDA approval.36
Thrombolysis in PE. Thrombolysis is a powerful tool in the treatment of patients with PE. (See Table 5.) If successfully used, it reverses right heart failure rapidly and safely in the appropriate patient.36 Unfortunately, due to the lack of large randomized trials, the effectiveness of this treatment in increasing survival in patients with PE is not nearly as certain as thrombolysis in MI. It usually is far more difficult to diagnose with certainty that a patient in shock has PE. The indications for giving thrombolysis and the odds of successful outcome are much better studied in MI because it is an easier diagnosis to make and, therefore, to study in the acute setting. Yet, our premortem diagnosis and treatment of those with PE remains lower than that demonstrated at autopsy.39-42 In fact, only about one-third of patients who die of PE have the correct premortem diagnosis.40
Table 5. Thrombolytics Dosing

Thrombolytics have the ability not merely to limit further clot propagation, as do the heparins, but also to dissolve clots. Twelve hours after treatment with thrombolytics, there is a large decrease in right ventricular strain as compared to treatment with heparin. The difference seen on echo between the two treatment groups disappears after a week.43 This clearly appears to be an advantage when the clot is in a position where timely removal can create differences in outcome. Use of thrombolytics in patients with PE demonstrates rapid improvement in hemodynamic measurements and pulmonary perfusion.44 Right ventricular function recovers quickly after thrombolysis.45 There is a more rapid decrease in pulmonary hypertension.45 A difference in the long-term mortality rate or the recurrence of PE has not been demonstrated, although it may exist.44
Of course, for every treatment there is risk as well as benefit. (See Tables 6 and 7.) The increase in the speed of resolution of the clot lysis is offset by the increased risk of bleeding among those who receive thrombolytics. In the MAPPET trial, it was found that the patients who received thrombolytics had a 21.9% rate of major bleeding complications as opposed to 7.8% in the group who received heparin.46 In other trials, the risk of major bleeding in those treated with thrombolytics was found to be 5-10%, with the risk of an intracranial bleeding found to be about 1-2%.44,47-49 The risk of major bleeding with heparin treatment generally is about 4%.14
Table 6. Absolute Contraindications to Thrombolytics

Table 7. Relative Contraindications to Thrombolytics

Another risk of using thrombolytics is that as the large clot lyses, it can move on to occlude other areas. This has been noted in those who have mobile right atrial thrombi. After thrombolysis, the atrial thrombosis is resolved, but new perfusion defects are noted on ventilation/perfusion scans.50
In thinking of those who have PE, it is useful to consider them in three categories of illness.51 The first group of patients presents in shock. The second group of patients is symptomatic but has normal hemodynamic parameters and normal right ventricular pressures. Finally, the third group is composed of those who have normal clinical hemodynamic parameters but, when studied, are found to have signs of right ventricular overload.
The treatment of the first two groups generally is agreed upon. A 1995 study of a small group of patients who presented in shock due to PE demonstrated that none of the patients receiving thrombolysis died but all who received heparin died.52 Only eight patients were studied because the trial was terminated when the results of the first eight were found to be so dramatic. For patients who present in shock, and therefore, demonstrate a higher risk of mortality, the risk of bleeding is offset by the benefit of rapid clot lysis. Patients this ill cannot tolerate the workup often required to diagnose PE. A bedside echocardiogram often can be helpful in determining whether there is right heart strain or failure, and therefore, a high likelihood that PE is the cause of shock. In the appropriate clinical scenario, echo may be the best exam available to guide the practioner in choosing to use thrombolytics for the treatment of suspected massive PE.53,54 It must be remembered that transthoracic echo fails to identify 50% of patients with angiographically proven PE; echo is merely a tool, not a gold standard.55 Treatment with thrombolytics is most effective when there is a sudden onset of symptoms, although treatment with thrombolytics can be effective even after a delayed presentation.56
The second group of patients, those who are hemodynamically stable with no signs of right heart strain noted on study, is treated best with heparin. The increased risk of major bleeding that occurs with thrombolysis generally is felt to be unwarranted among this group. Heparin provides resolution of the clot in an adequate time frame.
There is controversy regarding thrombolytics in the decisions to treat the third group of patients who occupy the clinical middle ground.44 They are not so sick as to be in shock, yet they have right ventricular (RV) strain that can be detected by echocardiogram. This group comprises about 40% of normotensive patients with acute PE.57,58 It had been presumed that the RV strain demonstrated increases clot load, but another study found that RV dysfunction did not correlate with perfusion abnormalities on ventilation/perfusion scan.60 Nonetheless, mortality among this group is high. In the MAPPET study, 8% of those who were normotensive but demonstrated RV strain on echo died.53,59 The members of this group have been described as having "impending hemodynamic instability."23 Significant studies have led some researchers to conclude that thrombolysis in this group is of enough benefit to warrant the extra bleeding risk.53,61,62 Yet a study from 2001 has concluded that the patients in this group do not fare better clinically after treatment and they suffer from a higher complication rate due to bleeding than the patients treated with anticoagulation alone.61 All of these studies were registry studies and have the limitations imposed by such a structure. As yet, there is no randomized controlled study available to answer this question.
In summary, thrombolytics have been shown to be beneficial in patients with PE who are in shock. In PE patients who are normotensive and have normal right ventricular pressures, the benefit of treatment with thrombolytics instead of anticoagulation is not enough to outweigh the risk of complications from thrombolytics. Anticoagulation should be used in these patients. The treatment of patients who are normotensive yet demonstrate right ventricular strain is controversial, with some advocating treatment with thrombolytics and some stating that there is no added benefit of thrombolytics over anticoagulation.
Embolectomy. Embolectomy can be performed either as an open surgical procedure or by a fragmentation and suction method in a closed percutaneous procedure using special apparatus. Patients who do not become hypotensive uniformly fare better without surgery, as long as further embolization can be prevented.4 Patients who meet the current indications for embolectomy include patients who fail thrombolysis, patients who are too ill to attempt thrombolysis, and patients with contraindications to thrombolysis.1
Surgical embolectomy first was performed more than 75 years ago.63 At that time, bypass was not available. Some still advocate a similar procedure done without bypass. Clarke and Abrams reported on their 25 years of experience, having done 55 embolectomies during that time. They utilize a short period of normothermic cardiac standstill after clamping both vena cava.64 Those patients who had a cardiac arrest prior to the procedure predictably did worse than those who did not. Eighty percent of 19 patients who had arrested prior to the procedure died. The mortality rate for the 36 patients who had not arrested, however, was only 20%.64 This demonstrates that even in institutions without bypass facilities, a surgical remedy is possible for those who are in dire straits.
Mortality rates among those utilizing bypass during the embolectomy range from 20-40%.66-69 The patients who entered the studies were considered to be too ill for a trial of thrombolysis, failed thrombolysis, or had contraindications to thrombolysis.65 Partial cardiopulmonary bypass (extracorporeal membrane oxygenation, or ECMO) also is an option to consider for open embolectomy.70
Percutaneous routes also are available to remove an embolus. Catheters can be placed that mechanically break up the clot and then suction the pieces.71,72 Another option is to place a catheter percutaneously and then use it as a vehicle to directly introduce thrombolytics in close proximity to the pulmonary artery.73 These are reasonable options in those found to have large, central clot load who are not candidates for systemic thrombolytic therapy.
It is important to consider ahead of time what route your institution is able to follow in patients who are not candidates for thrombolysis but who have severe disease due to PE. It is hoped that these patients are a rare occurrence, but when they do come to the ED, it is very important to mobilize as quickly as possible. A prepared protocol may save essential time.
Warfarin. After initial anticoagulation is achieved with heparin, long-term anticoagulation with warfarin should begin.4 Warfarin should be started within three days of initial heparin therapy, as delayed schedules are associated with increases in hospital stay and slightly higher rates of recurrence.10,74 The first 24 hours of warfarin therapy are associated with a transient hypercoagulable state, and heparin should not be discontinued until the international normalized ratio (INR) has been in the therapeutic range for at least two days.11 Treatment with heparin and warfarin typically overlaps for five days to successfully bypass the transient rise in hypercoagulability.
The optimal duration of therapy with oral anticoagulation is uncertain.76 The American College of Chest Physicians in 1998 called for at least three months of oral anticoagulant therapy after PE or DVT unless contraindicated.11,12 Others have shown that six months of therapy are better than six weeks.78-80 A study of 897 patients (107 with PE) randomized to six weeks or six months of therapy found that the former group had twice as many episodes of recurrence with no difference in rates of major hemorrhage.78 For patients with a low risk of recurrence and temporary risk factors, three months of treatment seems to be sufficient. For patients with idiopathic venous thromboembolism or permanent risk factors who have a high risk of recurrence, other trials are necessary to assess prolonged therapy beyond six months.81
The research committee of the British Thoracic Society studied 712 patients with venous thromboembolism randomized to four weeks or three months of therapy. They found that patients treated for three months had significantly fewer recurrence episodes and failures of disease resolution than those treated for a shorter period of time.82 However, they also found that the subgroup of patients with postoperative DVT and PE had a low rate of treatment failures and recurrence rates (3%), with no difference between the four-week and three-month therapy groups. They recommended only four weeks of anticoagulation (AC) for these patients.
Once the time period of treatment is determined, it is essential to consider the other obstacles to ongoing treatment. Among those patients age 65 and older who were hospitalized in New Jersey between January 1991 and June 30, 1994, and who actually began treatment for DVT or PE, 23% received therapy for less than the recommended length of time after hospital discharge. African-Americans were more likely to have a shorter than recommended length of treatment.77 It is unclear why they had a higher rate of failure to complete treatment.
Inferior Vena Caval Filters. Inferior vena caval interruption filters are not a replacement for adequate anticoagulation.4 The filters are inserted via the internal jugular vein or the femoral vein and positioned just below the renal vessels. The filters are supposed to catch large clots originating in the legs before they get to the pulmonic vessels. Unfortunately, there are various ways that they can fail. The filters can be positioned inadequately so that clots pass by them. Over time, collateral circulation can develop, permitting another route for the passage of clots. Rarely, clots may occlude the filter, causing caval thrombosis. Clots also may originate above the filter. Of course, a caval filter does nothing to treat the initial PE, so a method of emergent treatment must be considered as well.
There are two indications for the use of the caval filters.1 One indication is that the patient has a contraindication to anticoagulation, and the second is that treatment with anticoagulation has failed.4 Absolute contraindications to anticoagulation include active bleeding, neurological trauma, or surgery within the past week.1 There are many other lesser contraindications, but they can be weighed against the need for anticoagulation vs. a filter, and often will be found to be less of a risk.
In patients who have an apparent failure of anticoagulation, it is imperative to determine whether the entire period of treatment has been at therapeutic levels. It is understandable that although a medication that must be taken religiously to maintain appropriate blood levels, patients occasionally may miss a dose. If this is the case, then anticoagulation has not failed; what has failed is adherence to the treatment regimen.
DVT does not occur only in the lower extremity. Patients with central venous lines or patients at high risk for clotting easily can have upper extremity DVT. For the patients with an upper extremity DVT, it is possible to place a superior vena caval filter.83
Prevention
It has long been known that those who are bed-bound or immobilized for a period of time are at higher risk for thromboembolism. Patients who undergo major orthopedic procedures are at particularly high risk of developing venous thromboembolism. Without heparin prophylaxis, 48% of those patients will develop DVT.82,84 PE is the third most common cause of death in trauma patients who survive more than 24 hours.86 Medical patients who are limited in their movements also are prone to venous thromboembolic disease.87 Others at high risk include patients with disseminated malignancy, CHF, recent stroke, and previous venous thromboembolism.88
There are multiple ways to address prophylaxis for venous thromboembolism in the hospitalized patient. Traditional choices for prevention include the use of low-dose UFH given subcutaneously two or three times per day at a dose of 5000 units, intermittent pneumatic compression, elastic stockings, warfarin, or a combination of these options. (See Table 8.) LMWHs also are a choice in prevention.85 It has been shown that prophylactic treatment with the LMWH enoxaparin at 40 mg per day subcutaneously safely reduces the risk of venous thromboembolism in patients with acute medical illness.87 One researcher used reviparin once daily to effectively decrease DVT in casted and, therefore, immobilized patients. No patients taking the LMWH developed PE, but 2/188 patients in the placebo group developed PE.89 LMWHs also have been shown to be more effective than UFH in preventing VTE in patients at especially high risk.91
Table 8. Prevention of DVT and PE

Aspirin at a dose of 160 mg a day reduces the risk of PE and DVT by at least one-third during a period of increased risk (orthopedic surgery), and there now is good evidence for considering aspirin routinely in a wide range of surgical and medical patients at high risk of venous thromboembolism,92 although other methods may be more effective.85
The decision regarding venous thromboembolic prophylaxis may be left to the admitting physician, but a reminder from the emergency physician about the necessity of prophylaxis might be helpful. Indeed, in these days of increased ED congestion as patients wait for inpatient beds, it behooves the ED physician to at least consider instituting prophylaxis for appropriate patients in the ED. Thromboembolic disease is a high stakes illness, and prevention can begin in the ED.
References
1. Edlow JA. Emergency department management of pulmonary embolism. Emerg Med Clin North Am 2001;4:995-1011.
2. Tapson VF, Hull RD. Management of venous thromboembolic disease. The impact of low molecular weight heparin. Clin Chest Med 1995;16:281-294.
3. Stein PD. Acute pulmonary embolism. Dis Month 1994;40:467-523.
4. Fried C. Pulmonary embolism. In Rosen P, Barkin et al, eds. Emergency Medicine, 4th ed St. Louis: Mosby; 1998:1770-1805.
5. Simonneau G, Sors H, Charbonnier B, et al. A comparison of low-molecular-weight heparin with unfractionated heparin for acute pulmonary embolism. N Engl J Med 1997;337:663-669.
6. Barritt DW, Jordan SC. Anticoagulant drugs in the treatment of pulmonary embolism: A controlled trial. Lancet 1960;1: 1309-1312.
7. Glazier RL, Crowell E. Randomized prospective trial of continuous vs. intermittent heparin therapy. JAMA 1976;236:1365-1367.
8. Mant MJ, O’Brien ED, Thong KL, et al. Haemorrhagic complications of heparin therapy. Lancet 1977;1:1133-1135.
9. Hull RD, Raskob GE, Hirsh J, et al. Continuous intravenous heparin compared with intermittent subcutaneous heparin in the initial treatment of proximal vein thrombosis. N Engl J Med 1986;315: 1109-1114.
10. Gallus AS, Jackaman J, Tillet J, et al. Safety and efficacy of wafarin started early after submassive venous thrombosis or pulmonary embolism. Lancet 1986;2:1293-1296.
11. Hull RD, Raskob GE, Rosenbloom D, et al. Heparin for 5 days as compared with 10 days in the initial treatment of proximal vein thrombosis. N Engl J Med 1990;322:1260-1264.
12. Hyers TM, Hull RD, Weg JG. Antithrombotic therapy for venous thromboembolic disease. Chest 1998;114(suppl):561S-578S.
13. Hull RD, Raskob GE, Brant RF, et al. Relation between the time to achieve the lower limit of apt therapeutic range and recurrent venous thromboembolism during heparin treatment for deep venous thrombosis. Arch Intern Med 1997;157:2562-2568.
14. Zidane M, Schram MT, Planken EW, et al. Frequency of major hemorrage in patients treated with unfractionated intravenous heparin for deep venous thrombosis or pulmonary embolism. Arch Intern Med 2000;160:2369-2373.
15. Stein PD, Hull RD, Raskob GE. Withholding treatment in patients with acute pulmonary embolism who have a high risk of bleeding and negative serial noninvasive leg tests. Am J Med 2000:109: 301-306.
16. Berman AR. Pulmonary embolism in the elderly. Clin Geriatr Med 2001;17:107-130.
17. Hull RD, Raskob GE, Brant RF, et al. Low-molecular-weight heparin vs heparin in the treatment of patients with pulmonary embolism. Arch Intern Med 2000;160:229-236.
18. Kleinschmidt K, Charles R. Pharmacology of low molecular weight heparins. Emerg Med Clin 2001;19:1025-1049.
19. Hirsh J, Levine MN. Low molecular weight heparin. Blood 1992; 79:1-17.
20. Ginsberg JS. Management of venous thromboembolism. N Engl J Med 1996;335:1816-1828.
21. Brewer D. Should low-molecular-weight heparins replace unfractionated heparin as the agent of choice for adults with deep venous thrombosis? J Fam Pract 1998;47:185-192.
22. Dolovich LR, Ginsberg JS, Douketis JD, et al. A meta-analysis comparing low-molecular-weight-heparins with unfractionated heparin in the treatment of venous thromboembolism: Examining some unanswered questions regarding location of treatment, product type, and dosing frequency. Arch Intern Med 2000;160: 181-188.
23. Goldhaber SZ. Unsolved issues in the treatment of pulmonary embolism. Thrombosis Research 2001;103:V245-V255.
24. Mammen EF, Arcelus J, Messmore H, et al. Clinical differentiation of low molecular weight heparins. Semin Thromb Hemost 1999; 25:135-140.
25. Bauld DL, Kovacs MJ. Dalteparin in emergency patients to prevent admission prior to investigation for venous thromboembolism. Am J Emerg Med 1999;17:11-14.
26. The Columbus Investigators. Low molecular weight heparin in the treatment of patients with venous thromboembolism. N Engl J Med 1997;337:657-662.
27. Clark S. Current issues in management of thrombosis. Lancet 1995; 346:113-114.
28. Hirsh J. Heparin. N Engl J Med 1991;324:1565-1574.
29. Wells PS, Kovacs MJ, Bormanis J, et al. Expanding eligibility for outpatient treatment of deep venous thrombosis and pulmonary embolism with low molecular weight heparin. Arch Intern Med 1998:158:1809-1812.
30. O’Brien B, Levine M, Willan A, et al. Economic evaluation of outpatient treatment with low-molecular-weight heparin for proximal vein thrombosis. Arch Intern Med 1999;159:2298-2304.
31. U.S. Food and Drug Administration website. www.FDA.gov. Accessed 05/24/2004.
32. Harrison L, McGinnis J, Crowther M, et al. Assessment of outpatient treatment of deep vein thrombosis with low-molecular-weight heparin. Arch Intern Med 1998;158:2001-2003.
33. Yusen RD, Haraden BM, Gage BF, et al. Criteria for outpatient management of proximal lower extremity deep venous thrombosis. Chest 1999;115:972-979.
34. Dunn AS, Schecter C, Gotlin A, et al. Outpatient treatment of deep venous thrombosis in diverse inner-city patients. Am J Med 2001; 110:458-462.
35. Goldhaber SZ, Morrison RB, Diran LL, et al. Abbreviated hospitalization for deep venous thrombosis with the use of Ardeparin. Arch Intern Med 1998;158:2325-2328.
36. Goldhaber SZ. Unsolved issues in the treatment of pulmonary embolism. Thromb Res 2001;103:V245-V255.
37. Kovacs MJ, Anderson D, Morrow B, et al. Outpatient treatment of pulmonary embolism with Dalteparin. Thromb Haemost 2000; 3:209-211.
38. Goldhaber SZ. Optimizing anticoagulant therapy in the management of pulmonary embolism. Semin Thromb Hemost 1999;25: 129-133.
39. Janke RM, McGovern PG, Folsom AR. Mortality, hospital discharges, and case fatality for pulmonary embolism in the Twin Cities: 1980-1995. J Clin Epidemiol 2000;53:103-109.
40. Pineda LA, Hathwar VS, Grant BJ. Clinical suspicion of fatal pulmonary embolism. Chest 2001;120:791-795.
41. Morpurgo M, Schmid C, Mandelli V. Factors influencing the clinical diagnosis of pulmonary embolism: Analysis of 229 postmortem cases. Int J Cardiol 1998; 65(suppl.1):S79-S82.
42. Kurkciyan I, Meron G, Sterz F, et al. Pulmonary embolism as cause of cardiac arrest. Arch Intern Med 2000;160:1529-1535.
43. Konstantinides S, Tiede N, Geibel A, et al. Comparison of Alteplase versus heparin for resolution of major pulmonary embolism. Am J Cardiol 1998;82:966-970.
44. Arcasoy SM, Kreit JW. Thrombolytic therapy of pulmonary embolism. Chest 1999;115:1695-1706.
45. Nass N, McConnell MV, Goldhaber SZ, et al. Recovery of regional right ventricular function after thrombolysis for pulmonary embolism. Am J Cardiol 1999;83:804-806.
46. Konstantinides S, Geibel A, Olschewski M, et al. Association between thrombolytic treatment and the prognosis of hemodynamically stable patients with major pulmonary embolism: Results of a multicenter registry. [comments]. Circulation 1997;96: 2498-2500.
47. Levine MN. Thrombolytic therapy for venous thromboembolism: Complications and contraindications. Clin Chest Med 1995;16: 321-328.
48. Dalen JE., Alpert JS, Hirsch J. Thrombolytic therapy for pulmonary embolism: Is it effective? Is it safe? When is it indicated? [Comments]. Arch Intern Med 1997;157:2550-2556.
49. Kanter DS, Mikkola KM, Patel SR, et al. Thrombolytic therapy for pulmonary embolism: Frequency of intracranial hemorrhage and associated risk factors [Comment]. Chest 1997;111:1241-1245.
50. Cracowski JL, Tremel F, Baguet JP, et al. Thrombolysis of mobile right atrial thrombi following severe pulmonary embolism. Clin Cardiol 1999;22:151-154.
51. Goldhaber SZ. Pulmonary embolism thrombosis: Broadening the paradigm for its administration. Circulation 1997;96:716-718.
52. Jerjes-Sanchez C, Ramirez-Rivera A, Garcia M de L, et al. Streptokinase and heparin versus heparin alone in massive pulmonary embolism: A randomized controlled trial. J Thromb Thrombolysis 1995;2:227-229.
53. Kasper W, Konstantinides S, Geibel A, et al. Management strategies and determinants of outcome in acute major pulmonary embolism: Results of a multicenter registry. J Am Coll Cardiol 1997;30: 1165-1171.
54. Krivec B, Voga G, Zuran I, et al. Diagnosis and treatment of shock due to massive pulmonary embolism. Chest 1997;112:1310-1316.
55. Miniati M, Monti S, Pratali L, et al. Value of transthoracic echocardiography in the diagnosis of pulmonary embolism: Results of a prospective study in unselected patients. Am J Med 2001;110: 528-535.
56. Daniels LB, Parker A, Patel SR, et al. Relation of duration of symptoms with response to thrombolytic therapy in pulmonary embolism. Am J Cardiol 1977;80:184-188.
57. Ribiero A, Lindmarker P, Juhlin-Danfelt A, et al. Echocardiography Doppler in pulmonary embolism: Right ventricular dysfunction as a predictor of mortality rates. Am Heart J 1997;134:479-487.
58. Goldhaber SZ, Haire WD, Feldstein ML, et al. Alteplase versus heparin in acute pulmonary embolism: Randomized trial assessing right ventricular function and pulmonary perfusion. Lancet 1993; 341:507-511.
59. Goldhaber SZ. Pulmonary embolism for cardiologists. J Am Coll Cardiol 1997;30:1172-1173.
60. Miller RL, Das S, Anandarangam T, et al. Association between right ventricular function and perfusion abnormalities in hemodynamically stable patients with acute pulmonary embolism. Chest 1998;113: 665-670.
61. Hamel E, Pacouret G, Vincentelli D, et al. Thrombolysis or heparin therapy in massive pulmonary embolism with right ventricular dilation. Chest 2001;120:120-125.
62. Miller GH, Sutton GC, Kerr IH, et al. Comparison of Streptokinase and heparin in treatment of isolated acute massive pulmonary embolism. BMJ 1971;2:681-684.
63. Meyer J. Freidrich Trendelenberg and the surgical approach to massive pulmonary embolism. Arch Surg 1990;125:1202-1205.
64. Clark DB, Abrams LD. Pulmonary embolectomy: A 25-year experience. J Thorac Cardiovasc Surg 1986;92:442-445.
65. Tschirkov A, Krause F, Elert O, et al. Surgical management of massive pulmonary embolism. J Thorac Cardiovasc Surg 1978;75: 730-733.
66. Gray HH, Morgan JM, Paneth M, et al. Pulmonary embolectomy for acute massive pulmonary embolism: An analysis of 71 cases. Br Heart J 1988;60:196-200.
67. Gulba DC, Schmid C, Borst HG, et al. Medical compared with surgical treatment for massive pulmonary embolism. Lancet 1994;343: 576-577.
68. Lund O, Nielsen TT, Schifter S, et al. Treatment of pulmonary embolism with full dose heparin, streptokinase or embolectomy- results and indications. Thorac Cardiovasc Surg 1986;34:240-246.
69. Meyer G, Tamisier D, Sors H, et al. Pulmonary embolectomy: A twenty year experience at one center [Aomments]. Ann Thorac Surg 1991;51:232-236.
70. Hsieh PC, Wang SS, Ko WJ, et al. Successful resuscitation of acute massive pulmonary embolism with extracorporeal membrane oxygenation and open embolectomy. Ann Thorac Surg 2001;72: 266-267.
71. Schmitz-Rode T, Janssens U, Duda SH, et al. Massive Pulmonary embolism: Percutaneous emergency treatment by pigtail rotation catheter. J Am Coll Cardiol 2000;36:375-380.
72. Koning R, Cribier A, Gerber L, et al. A new treatment for severe pulmonary embolism: Percutaneous rheolytic therapy. Circulation 1997;96:2498-2500.
73. Cotter CJ, Chang KS, Fearrington EI. Intrapulmonary artery infusion of urokinase for treatment of massive pulmonary embolism: A review of 26 patients with and without contraindications to systemic thrombolytic therapy. Clin Cardiol 1999;22:661-664.
74. Tai NMR, Atwal AS, Hamilton G. Modern management of pulmonary embolism. Br J Surg 1999;86:853-868.
76. Goldhaber SZ. Pulmonary embolism. N Engl J Med 1998;339: 93-104.
77. Ganz DA, Glynn RJ, Mogun H, et al. Adherence to guidelines for oral anticoagulation after venous thromboembolism and pulmonary embolism. J Gen Intern Med 2000;15:776-781.
78. Schulman S, Rhendin A-S, Lindmarker P, et al. A comparison of six weeks with six months of oral anticoagulant therapy after a first episode of venous thromboembolism. N Engl J Med 1995;332: 1661-1665.
79. Schulman S, Grandqvist S, Holmstrom M, et al. The duration of anticoagulation therapy after a second episode of venous thromboembolism. N Engl J Med 1997;336:393-398.
80. Kearon C, Gent M, Hirsh J, et al. A comparison of three months of anticoagulation with extended anticoagulation for a first episode of idiopathic venous thromboembolism. N Engl J Med 1999;340: 901-907.
81. Pinede L, Ninet J, Duhaut P, et al. Comparison of 3 and 6 months of oral anticoagulation therapy after a first episode of proximal deep vein thrombosis or pulmonary embolism and comparison of 6 and 12 weeks of therapy after isolated calf deep vein thrombosis. Circulation 2001;103:2453-2460.
82. Research Committee of the British Thoracic Society. Optimum duration of anticoagulation for deep-vein thrombosis and pulmonary embolism. Lancet 1992;341:507-511.
83. Ascher E, Hingorani A, Mazzariol F, et al. Clinical experience with superior vena caval Greenfield filters. J Endovasc Surg 1999;6: 365-369.
84. Kennedy D, Setnick G, Li J. Physical exam findings in deep venous thrombosis. Emerg Med Clin 2001;19:869-876.
85. Collins R, Scrimgeour A, Yusuf S, et al. Reduction in fatal pulmonary embolism and venous thrombosis by perioperative administration of subcutaneous heparin. Overview of results of randomized trials in general, orthopedic, and urologic surgery. N Engl J Med 1988;318:1162-1173.
86. Dhami MS, Bona RD. Thrombosis in patients with cancer. Postgrad Med 1993;92:131-140.
87. Samama MM, Cohen AT, Darmon,JY, et al. A comparison of enoxaparin with placebo for the prevention of venous thromboembolism in acutely ill medical patients. N Engl J Med 1999;341:793-800.
88. Agnelli G, and Solaglia F. Anticoagulant agents in the management of pulmonary embolism. Int J Cardiol 1998;65 (Suppl 1):S95-S98.
89. Lassen MR, Borris LC, Nakov RL. Use of the low-molecular-weight heparin reviparin to prevent deep-vein thrombosis after leg injury requiring immobilization. N Engl J Med 2002;347:726-730.
90. Ginsberg JS, Crowther MA. Deep venous thrombosis. In: Wachter RW, Goldnar L, Hollander H, et al, eds. Hospital Medicine. Philadelphia: Lippincaott, Williams and Wilkins; 2000:347-355.
91. Geerts WH, Jay R, Code KI, et al. A comparison of low dose heparin with LMWH as prophylaxis against venous thromboembolism after major trauma. N Engl J Med1996;335:701-707.
92. Pulmonary Embolism Prevention (PEP) Trial Collaborative Group. Prevention of pulmonary embolism and deep vein thrombosis with low dose aspirin: Pulmonary Embolism Prevention (PEP) trial. Lancet 2000;355:1295-1302.
This second and final part of a series covers the topics of differential diagnosis that must be considered when a patient presents with symptoms consistent with PE, treatment, and considerations for prevention of this disease state.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
