Pediatric Abdominal Emergencies
Pediatric Abdominal Emergencies
Author: Ademola Adewale, MD, Assistant Program Director, Florida Hospital Emergency Residency. Assistant Clinical Professor, University of Central Florida College of Medicine.
Peer Reviewer: Steven M. Winograd, MD, FACEP, Attending, Emergency Medicine, St. Joseph Medical Center, Yonkers, NY.
Abdominal pain in childhood is a common presentation to the emergency department (ED). While most cases are benign, they frequently create a diagnostic dilemma. The spectrum of disease varies widely from neonates to infants, children, and adolescents, and requires the clinician to rapidly differentiate between a benign medical diagnosis versus a potential surgical abdomen to minimize morbidity and mortality. (See Table 1.)
Classifying the disease process itself is essential, and can be simplified by detailed investigation of associated symptoms such as presence or absence of fever, obstructive versus non obstructive, local or systemic process, healthy versus sick-appearing, and abdominal or extra-abdominal in nature. (See Table 2.)
The author describes the common benign, potential surgical, and the life-threatening causes of abdominal pain in children. Emphasis is placed on the high-risk etiologies pertaining to neonates and infants.
– The Editor
Incarcerated Hernia
Congenital inguinal hernias occur in about 0.4% to 4.4% of children while the incidence in preterm infants is considerably higher.1 It develops in 13% of infants born before 32 weeks gestation, and in 30% of infants weighing less than 100 grams.2 The incidence is estimated to be up to 10 times more in boys.3 Indirect inguinal hernias account for about 90% of all hernias and are due to an arrest of embryonic development and incomplete closure of the processus vaginalis rather than an acquired weakness.4 The processus vaginalis is a peritoneal sac that passes through the inguinal canal with the testis as it descends into the scrotum during the seventh to ninth month of gestation. The greatest risk from a hernia is the development of intestinal incarceration and strangulation, which may also involve the gonads (testes and ovaries).
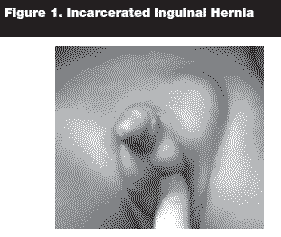
The clinical spectrum of incarcerated inguinal hernia in infants may mimic any intra-abdominal pathology described in Table 1. The presenting symptoms may include irritability, poor feeding, vomiting, listlessness, fever, lethargy, bloody stool, or an extremis state with necrotic bowel. The diagnosis depends on a detailed history, noting any bulging (including intermittent bulging) in the groin area, labia, or scrotum, and a physical examination with the child fully exposed. This should be confirmed by examining the child upright, applying pressure on the abdomen, or while straining or crying. For a non-crying child, gently holding the legs and arms against the examination table so that they cannot move may elicit crying that increases intra-abdominal pressure, resulting in visualization of the hernia. When no bulge is seen, palpable thickening of the spermatic cord (the "silk glove" sign) has over 90% sensitivity and specificity for presence of an inguinal hernia.4 (See Figure 1.) A recent prospective study from China involving 1,040 patients showed that the physical finding of the "silk glove or silk purse sign" has a sensitivity of 91% and a specificity of 97.3% for diagnosing inguinal hernia.5
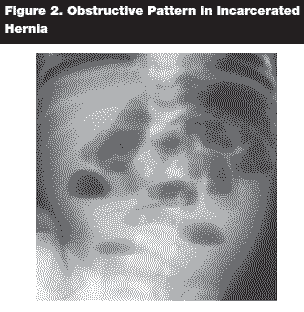
In addition to the diagnostic yield of the silk glove sign finding on physical examination, there are several radiologic diagnostic adjuncts, starting with a regular abdominal series. In an infant with abdominal fullness or distention on examination, an abdominal series may show an obstructive pattern with dilated bowel and air fluid levels. (See Figure 2.) This is a quick, inexpensive study that may facilitate the diagnosis.
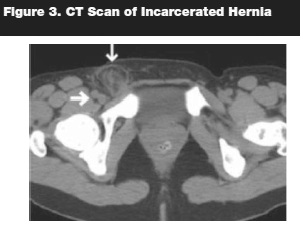
Furthermore, ultrasonographic evaluation has been shown to be very effective. In Asia, office ultrasound has been successfully used to differentiate between an inguinal hernia and a patent processus vaginalis.6 A study performed by Chen et al shows that office ultrasound increased diagnostic accuracy from 84% (physical exam alone) to 97.9%.7 In some rare instances, computed tomography (CT) of the abdomen and pelvis could be utilized to diagnose a hernia or identify other etiologies of bowel obstruction in a patient whose hernia could not be palpated. (See Figure 3.)
After diagnosing an incarcerated hernia, the treatment approach to minimize morbidity is to obtain surgical consultation first while attempting a manual reduction. Most incarcerated hernias can usually be treated via manual reduction, which can be performed without sedation using the "unilateral frog-leg maneuver." This maneuver entails applying constant direct pressure on the hernia with the hip flexed and externally rotated, thus shortening the inguinal canal. Sedation should be utilized if necessary. However, after difficult manual reduction, the child should be admitted and surgery deferred for 24 to 48 hours to allow some of the edema to resolve. A recurrent episode of incarceration increases the risk of complications such as intestinal necrosis, infarction of the testes or ovaries, recurrent hernia, and wound infection.8 In one study, major complications occurred in 11% of incarcerated hernias compared to 0.6% in those having elective surgery.9 After diagnosis, a pediatric inguinal hernia should be repaired to avoid the risk of incarceration, since the risk is highest in young children.10
Hirschsprung's Disease (Aganglionic Colon)
Hirschsprung's disease is caused by the failed migration, during gestation, of colonic ganglion cells, which usually arrive in the proximal colon by eight weeks gestation and at the rectum by the 12th week. Consequently, varying lengths of the distal colon are unable to relax, causing functional constipation and colonic obstruction. It most commonly involves the recto-sigmoid region of the colon, but can affect the entire colon.11 The incidence of the disease is estimated to be 1 in 5,000 live births, and it is four times more prevalent in boys.12 The disease is further classified into short-segment (limited to recto-sigmoid) and long-segment (proximal to recto-sigmoid or can involve entire colon) diseases. It is estimated that 3% to 5% of male siblings and 1% of female siblings of children with short-segment disease also have the disease.13 For the sibling of an infant with the long-segment disease, the relative risk is significantly higher (12.4%–33%).14
Approximately 80% of patients present in the first few months of life with difficult bowel movements, poor feeding, and progressive abdominal distention.15 Some common presentations may include bilious vomiting, chronic constipation, progressive abdominal distention, poor feeding, poor weight gain, infrequent explosive bowel movements (which may be bloody in enterocolitis), jaundice, restlessness, fever, and failure to pass meconium within 48 hours. The physical examination of these patients, in addition to detailed history, may hold the key to early diagnosis. The palpation of dilated loops of bowel or tight anal sphincter in the absence of stool in the rectum, or explosive release of gas and stool after a rectal exam in a child with progressively distended bowel may aid diagnosis.

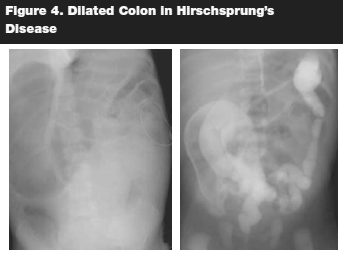
Diagnostic maging of these patients is useful. Plain abdominal radiographs may show massive distention of colon with gas and feces present, air fluid levels, or air in the bowel wall which may herald the presence of enterocolitis. (See Figure 4.) Non-prepped barium enema is also useful, although false negative findings are common. Dilated colon proximal to the aganglionic region or spastic transitional segment (irregular saw toothed outline) may be seen on lateral view. Barium enema is, however, contraindicated when enterocolitis is suspected. Other diagnostic adjuncts such as anal manometry or anal suction biopsy may be utilized.
Once diagnosis is made, management of this disease is mostly surgical. Owing to the significant morbidity (bowel rupture, enterocolitis, lifelong colostomy) that accompanies this disease, early diagnosis is extremely important.
Intussusception
Intussusception occurs when a portion of a bowel segment invaginates into an immediately adjacent segment of bowel. The majority of cases are due to hypertrophy of Peyer's patches of the terminal ileum. This is the lead point of the intussusceptum that is subsequently dragged through the ileo-cecal valve into the cecum and right colon. The mesentery of the intussusceptum becomes compressed, leading to edema and swelling of the bowel wall, which results in obstruction. Simultaneously, this compression also leads to venous engorgement and intestinal mucosal ischemia, which may result in bleeding and sloughing off of the mucosa. This is the cause of the classic "currant jelly stool." Studies have shown that ileo-colic intussusception is the most common cause of small bowel obstruction in children 3 months to 6 years of age, and occurs in at least 56 children per 100,000 per year in the United States.16 It rarely occurs before 3 months of age, and is less common after 3 years of age.
The clinical manifestations of the disease may be subtle, hence heightened suspicion is needed to make a diagnosis, and failure to make a prompt diagnosis and initiate appropriate treatment may lead to bowel ischemia, perforation, peritonitis, shock, and even death.17 The typical presentation of the disease is that of a previously healthy infant with sudden onset of abdominal pain and vomiting. The pain often occurs in paroxysms, lasting a few minutes, with subsequent return to normalcy. As the disease progresses, the infant may have a loose watery stool that may or may not be accompanied by mucus and blood. The classic triad of colicky abdominal pain, vomiting, and "currant jelly" stool are only present in about 21% of cases.3 In some instances, the infant may present in a lethargic state which may lead the clinician to pursue other causes of lethargy and delay the diagnosis. Understanding the evolution of the disease presentation and the age predilection will minimize the chances of a missed diagnosis.
The physical examination of these infants requires extreme vigilance. In most cases, the abdomen is soft and non-tender in the early stages of disease. The ideal time to evaluate the abdomen for intussusception is between attacks. The clinician might palpate a vertically oriented, "sausagelike" mass on the right side of the abdomen. The stool must be checked for occult blood. A positive stool guaiac is present in almost all cases. However, the classic "currant jelly" stool is only present in 50% of cases.18 A presumptive diagnosis could be ascertained from the history and physical alone.
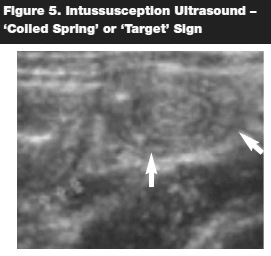
Once a presumptive diagnosis is made, it should be confirmed with radiographic studies. A plain abdominal radiograph may show a filling defect or a mass in the right upper quadrant or, it could be normal. Ultrasonography is an adjunctive diagnostic tool. A series by Navarro and colleagues demonstrated that ultrasound identified the pathologic lead point in 66% of cases.19 (See Figure 5.) The use of barium enema, which demonstrates the classic "coiled spring" appearance, may lead to diagnosis while simultaneously treating the disease. (See Figures 6 and 7.)
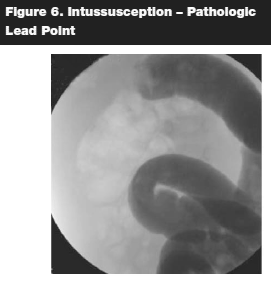
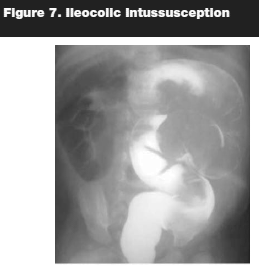
The safety of barium enema, with the potential risk of perforation, has been questioned. Presently, air contrast enema has been frequently utilized as an alternative to barium enema. A study by Saenz et al concluded that "air enema treatment is easy to perform, is lower in cost and presents less morbidity, and that it should be considered as the treatment of choice in the initial management of intussusceptions."19 Recurrent rate after reduction is about 5%–10% and it often occurs within 24 to 48 hours. Cases that cannot be reduced using barium or hydrostatic air insufflations require emergent surgical intervention.
Malrotation and Midgut Volvulus
Intestinal malrotation is a congenital condition with a potential for catastrophic or life threatening sequelae. Volvulus is a major life-threatening complication of malrotation.
Failure of normal embryologic processes to occur due to abnormal rotation results in the cecum riding high in the abdomen, the peritoneal attachment crossing the duodenum in an obstructing fashion, the mesentery failing to fan out, and the midgut and its entire vascular supply becoming suspended as it travels through a narrow pedicle.20,21
The estimated incidence of the disease is 1 in every 500 live births, with male to female ratio of 2:1.22 It is usually diagnosed in newborns and young infants; up to 75% of symptomatic cases occur in newborns, and up to 90% of symptomatic cases occur within the first year of life. Mortality in affected newborns was approximately 30% in the 1950s and 1960s, but since then has markedly decreased, to 3%–5%.23
The potential for catastrophic consequences of missed or delayed diagnoses mandates a high index of suspicion to make prompt diagnosis and treatment to prevent morbidity and mortality. The clinical presentation is often heralded by poor feeding, bilious vomiting, irritability, and bloody stool.24 However, these symptoms could mimic other medical or intra-abdominal pathologies. Physical examination alone is often not sufficient to make the diagnosis. A detailed history with a high index of suspicion will facilitate diagnosis. Once the diagnosis is suspected, immediate consultation with a pediatric surgeon should be obtained while official diagnostic studies are being performed.
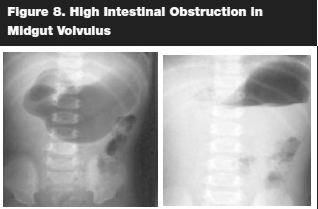
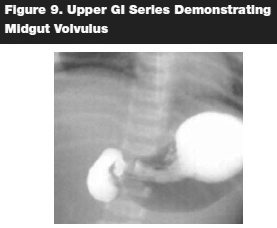
An important diagnostic aide is an upright abdominal series radiograph. This often demonstrates a high intestinal obstruction pattern. (See Figure 8.) Upper gastrointestinal (GI) series remains the fastest and most accurate diagnostic modality of choice.25 (See Figure 9.) It demonstrates duodenal obstruction, the position of the ligament of Treitz, and, if the contrast agent is followed distally, cecal malposition.25 The pathognomonic finding of the "corkscrew sign" is often present on the upper GI fluoroscopy. A series by Sizemore et al showed a sensitivity of 96% with an upper GI series.26
Understanding the catastrophic sequelae of a missed or delayed diagnosis makes it imperative for any clinician involved in the care of neonates and infants to always consider the diagnosis of malrotation with volvulus in any neonate or infant that presents with bilious vomiting, lethargy, irritability, poor feeding, or bloody stool. Since treatment is mostly surgical, prompt surgical consultation on patients with a suspicious presentation will minimize morbidity and mortality.
Hypertrophic Pyloric Stenosis
This is a common congenital condition affecting the proper development of the pylorus. Although a common entity, the exact pathophysiology is unknown. The pathognomonic feature of pyloric stenosis is gastric outlet obstruction with an abnormal pyloric antrum. The prevalence of the disease is about 1–5 per 1,000 live births and it is about 4–5 times more common in boys. It is purported to be less common in Black and Asian children. Also, the incidence is higher in first-born infants and it has been shown to reoccur in families, prompting the considerations for both environmental and hereditary factors.27 A study performed by Schechter et al shows that 95% of cases present between ages 3 weeks and 12 weeks.28
An understanding of the condition and the effective treatment has undergone a remarkable evolution in the 20th century, with a mortality rate reduction from over 50% to nearly 0%.29 The initial presentation of pyloric stenosis is often characterized by nonbilious, projectile vomiting that gradually worsens over several days to weeks. These infants eventually become wasted and dehydrated. The presentation of an emaciated infant with persistent nonbilious emesis with a voracious appetite should alert the clinician to the possibility of this disease. On physical examination, the palpation of thickened pylorus, or the so called "olive," in the epigastrium or right upper quadrant on abdominal examination could make the diagnosis certain. The common laboratory findings due to persistent vomiting are hyperchloremic hypokalemic metabolic alkalosis. Some may even have signs of renal insufficiency due to severe dehydration.
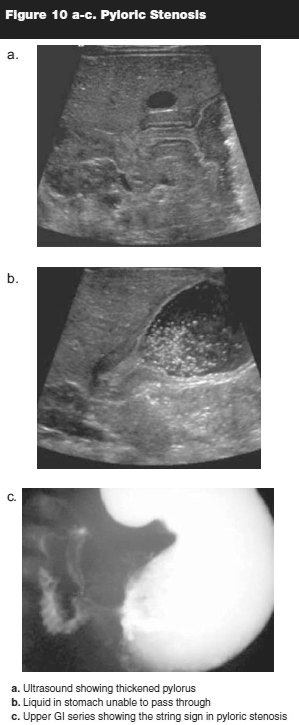
As an adjunct to physical examination, several imaging modalities are available to assist in diagnosis. Sonography in particular is noninvasive and accurate. Barium upper GI series, which will show the "string sign" (elongation of the pylorus canal) or the "double tract sign"(linear tract of contrast material separated by mucosa), is also an effective diagnostic modality.30 (See Figure 10 a-c.)
The definitive treatment of pyloric stenosis is mostly surgical. Currently, laparoscopic pyloromyotomy has become the standard treatment for hypertrophic pyloric stenosis. However, there are some growing proponents of medical management. A study by Kawahara et al showed the success of medical management of pyloric stenosis. They utilized intravenous atropine 6 times a day before feeding. They noted that once vomiting ceased, the infants were able to ingest 150 mL/kg of formula per day after stepwise increase in volume. This atropine regimen was later converted to oral dosing 6 times a day. When compared to surgical intervention, weight gains were the same after one year, with lower morbidity and readmission rates.31 Despite promising results, more research is needed to evaluate the effect of long-term atropine therapy.
Meckel's Diverticulum
Remnants of the prenatal yolk sac present the most common congenital anomaly that affects the small intestine. It is a true diverticulum because it is an outpouching of the intestine that contains all the layers of the intestinal wall. It is important to note that the lining of the vitelline duct or the diverticulum is made of undifferentiated cells that evolve into any mucosal layer. The most common is heterotropic gastric (50%) mucosa, followed by pancreatic, endometrial, and hepatobiliary tissues.32 It is usually found in the ileum within two feet of the ileocecal valve and receives blood supply from the superior mesenteric artery.33 It is estimated to occur in about 2% of the population and affects both males and females equally.33,34 In most instances the disease is silent, but an infant with the disease has a 4%–6% lifetime risk of complications.33,34
The most common presentation of the disease is that of painless rectal bleeding in a child younger than 2 years of age. More dramatic cases may manifest as profuse bleeding, intestinal obstruction, perforation, or diverticulitis. The diagnosis should be considered early in a child who presents with rectal bleeding to minimize complications. The child might be well-appearing or might present with bilious vomiting and abdominal distention with rectal bleeding.
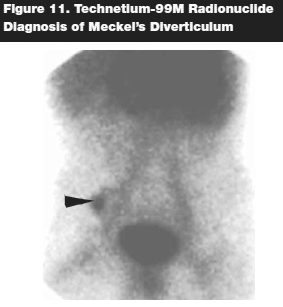
The management of the disease depends on the clinical manifestations. In a child with painless rectal bleeding without abdominal distention or vomiting, radioisotope technetium-99 scintigraphy is the diagnostic modality of choice. (See Figure 11.) The idea behind 99m-Tc pertechnetate scanning is that the pertechnetate anion is selectively taken up by the surface mucous-secreting cells that line the gastric mucosa. This modality has been shown to have a sensitivity of 85%, and a specificity of 95%.35 The definitive treatment of the disease is surgical, with the resection of the involved segment of the ileum. Obtain a pediatric surgical consultation early when the diagnosis is suspected to prevent morbidity.
Acute Appendicitis
This inflammatory process can occur in any age group, and in pediatrics carries significant morbidity owing to delayed diagnosis. Perforation, peritonitis, and sepsis are common presentations in children younger than 2 years of age. For children old enough to complain of abdominal pain, especially older than 2 years old, appendicitis should be considered in the differential diagnosis. It is the most common cause of acute surgical abdomen and the lifetime risk for the disease in the population is approximately 7%.36 The appendix is mainly located intraperitoneally either anterior or retrocecal. However, in some cases, it may be hidden in the pelvis, retrocolic, or retroileal location. The location of the appendix plays a significant role in findings during physical examination.
The classic presentation of periumbilical abdominal pain migrating to the right lower quadrant only occurs in about 50% of cases and may not apply to children unable to communicate their symptoms to clinicians.37 Young children may not be able to relate their history, leading to an estimated perforation rate of 50%.37 Furthermore, in children, palpation of the abdomen may be nonspecific. An effective methodology when examining the pediatric abdomen is to first distract the child, then watch the facial expression closely while palpating the abdomen. In most cases, this facilitates the physician in localizing the pain. Facial expression of a child during abdominal examination is an important clinical vignette.
Clinical presentation of the disease varies significantly. It may include fever, nausea, vomiting, diarrhea, loss of appetite, nonspecific abdominal pain, irritability, lethargy, or a shock-like state. These diverse presentations make the diagnosis difficult despite the fact that appendicitis is the most common surgical abdominal emergency. Several laboratory parameters such as leukocytosis (WBC > 10,000), neutrophilia (left shift), and elevated C-reactive protein often aid in the diagnosis in addition to physical examination. Some studies have shown that in the presence of normal WBC and C-reactive protein, the diagnosis of appendicitis is highly unlikely.38 To assist in diagnosing this disease, several appendicitis scoring systems have been utilized over the years.39-41 Additionally, several studies have been performed to validate the clinical applications of these appendicitis scores.42,43 The validation studies show that scoring systems were good adjuvant methods to help in the early diagnosis of appendicitis.
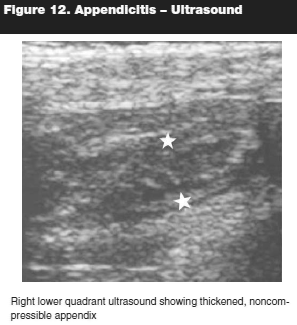
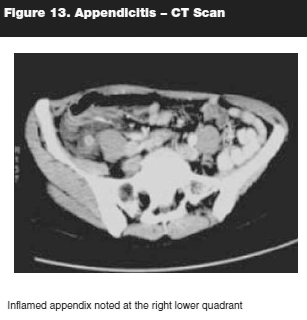
Despite a detailed history and physical examination, laboratory studies, and application of clinical scoring rules, diagnosing appendicitis is still a dilemma in the extremely young pediatric population. Radiographic studies are an important adjunct in the diagnostic evaluation. The modality utilized is dependent on the institution. Graded compression ultrasonography is the preferred initial imaging modality in the evaluation of pediatric acute appendicitis. (See Figure 12.) This modality is inexpensive, noninvasive, and minimizes radiation exposure. However, with the increase in infantile and childhood obesity, the diagnostic yield could be limited because body habitus is one of the limiting factors in obtaining accurate ultrasonography. The shortcoming of ultrasonography is that the examination is operator dependent. However, in the hands of experienced sonographer, the sensitivity ranges between 55% and 98% and specificity 78%–100%.44,45 Also, failure to visualize the appendix may require further imaging, typically CT. Presently, CT scan is the modality of choice for diagnosing appendicitis in children. (See Figure 13.) Although radiation exposure is a concern, CT scans have been shown to have an accuracy of 97%. Doria et al show that CT-scan has a sensitivity of 94% and specificity of 95%.46 Advantages of CT include the ability to evaluate the entire abdomen; locate perforation, abscesses, and phlegmon; and make alternative diagnosis. The disadvantages include the exposure to radiation and need for oral and intravenous contrast.
Regardless of the diagnostic modality utilized, most institutions have a diagnostic pathway in place to minimize radiation exposure.47 The definitive treatment when diagnosed is surgical intervention.
Testicular Torsion
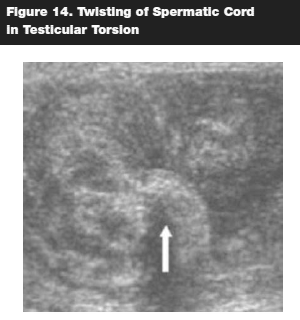
Testicular torsion is the most common pediatric urologic emergency and demands timely attention. Torsion occurs when the testicle is twisted on the spermatic cord, which holds the testicle in place within the scrotum. (See Figure 14.) This could be spontaneous or as a result of trauma. This twisting cuts off the blood supply to the affected testicle, with subsequent risk of testicular infarction. The common presenting symptoms include sudden, severe pain in one testicle, which later evolves into scrotal swelling due to edema and reactive hydrocele. In addition, the pain may radiate to the abdomen, flank, and groin, with accompanying nausea, vomiting, and difficult urination. Testicular torsion should be treated as an emergency; the testicle could be lost if blood flow is not restored within six hours.48,49
The presence of reactive hydrocele may make testicular examination difficult. However, the history presented should alert clinicians of the diagnosis. On physical examination, the involved testis will most often be in a transverse lie and high-riding when compared to the contralateral testis. The presence of this classic finding on exam should be pathagnomonic without requiring any other evaluation. Another finding on exam that may assist in the diagnosis is the absence of the cremasteric reflex (elicited by gently stroking the inner thigh with subsequent ascent of the testis) on the involved side. Attempts should then be made to immediately reduce (detorse) the torsion with concomitant consultation to the surgeon / urologist.
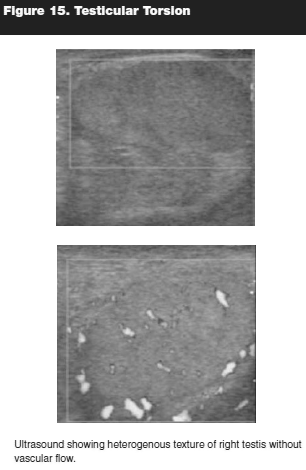
Testicular salvage in acute torsion is directly proportional to the duration of symptoms to time of manual detorsion or surgical exploration. A study by Davenport shows the following testicular salvage rates; if detorsed in less than 6 hours from onset, 90%; 50% after 12 hours; and less than 10% after 24 hours.50 In instances where the time of onset has lagged several hours prior to presentation, nuclear testicular blood flow scan or emergent Doppler ultrasound with color flow should be obtained to determine the viability of the involved testis. The presence of normal testicular echo texture (homogenous) despite lack of blood flow on ultrasound is indicative of a salvageable testis.51 Hence, ultrasound should be performed in consultation with the surgeon / urologist to minimize delay in immediate surgical exploration. (See Figure 15.)
Although torsion is more common in older children, cases in neonates and infants have been reported. These cases may accompany a congenital hydrocele. One such case was that of a 6-week-old male infant with congenital bilateral hydroceles that presented with a reddish, painful, and swollen left scrotum of two days' duration. Doppler studies revealed obstruction of blood flow to the left testicle. These findings confirmed the diagnosis of left-sided testicular torsion. Surgical exploration revealed the left testicle to be necrotic; unilateral orchidectomy was performed.52
Another mimicker of testicular torsion is the torsion of the testicular appendix. The testicular appendix is the remnant of the paramesonephric duct and it normally lies on the superior pole of the testis or epididymis. Torsion of the testicular appendix, though a benign condition, might present in a similar fashion as torsion except for the absence of nausea, vomiting, and abdominal pain. On physical examination, effort should be made to palpate the upper pole of the testes, which often is tender. A finding on the scrotum of a blue spot (the "blue-dot sign") is indicative of this diagnosis. Despite the presence of this sign, ultrasound should still be obtained to exclude other testicular pathology. Once diagnosed, the treatment is usually supportive because it is a self-limited condition. However, in cases of persistent pain, surgical exploration may be warranted to excise the appendage.
Ovarian Torsion
Acute ovarian torsion in infants and young children is rare and very difficult to diagnose when present because the diagnosis is often not suspected. Ovarian torsion is caused by rotation of the ovary or adnexa with the vascular pedicle on its axis. This rotation results in arterial, venous, and lymphatic obstruction. The twisted vascular pedicle includes the broad ligament, fallopian tube, and adnexal and ovarian branches of the uterine artery and vein. It is one of the most frequent gynecologic emergencies, but the incidence is not well defined because it is such a rare entity in young children. Kokoska et al, in a 16-year study, found the mean age to be 12.5 years, with ranges from 8–16 years of age, and most likely to occur at or prior to reproductive age.53
The clinical presentation of the disease is often intermittent colicky abdominal pain. The patterns of presentation, however, vary significantly (from dysuria, vomiting, fever, and abdominal mass) and only 44% of patients present with the classic symptoms.54 In a patient with right lower quadrant pain, it may mimic appendicitis. The author's first pediatric ovarian torsion case was in a 7-year-old girl with right lower quadrant pain. Initially, suspicion of appendicitis and kidney stone were high on the differential diagnosis list. However, the history of severe, sudden onset of right lower abdominal pain, lasting a few minutes with subsequent complete resolution, clinched the diagnosis. In this case, the patient was spontaneously detorsing. An emergent sonogram confirmed the diagnosis. The patient was taken to the operating room with laparoscopic detorsion and spontaneous return of perfusion.
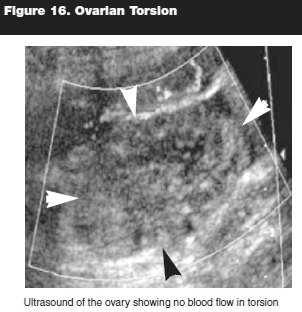
Although the diagnosis of ovarian torsion is challenging, detailed attention to the pattern of pain may assist in diagnosis since missed diagnosis carries significant morbidity. The exact duration from onset of symptom to potential for ovarian salvage has not been delineated. Anders et al, in a series of 22 patients over 15 years, reported that 18 of the patients presented after eight hours of symptom onset.55 Of these patients, 28% were detorsed in the operating room with evidence of reperfusion. Should eight hours from time of inset be the golden rule? When ovarian torsion is suspected, ultrasonography with Doppler flow is the diagnostic modality of choice. (See Figure 16.) The sonographic findings vary widely, from ipsilateral echogenic adnexal mass to the "whirlpool sign" that depicts a twisted pedicle. On grayscale sonography, the whirlpool sign is seen as a hypoechoic band representing the vessels wrapping around the central axis. In a series, the presence of positive whirlpool sign in the twisted vascular pedicle is the most definitive sign of ovarian torsion.56
The definitive treatment is mostly surgical and involves attempting to salvage the ovary. Most cases are performed laparoscopically. Ovarian torsion should be considered as part of the differential diagnosis of pediatric acute abdominal pain.
Conclusion
Abdominal pain in children is a common presentation to the ED. Missed diagnoses in neonates, infants, and children who cannot communicate carry significant morbidity, and in some cases mortality. Understanding the spectrum of the disease process in addition to rapid assessment with surgical consultation early in the evaluation will minimize morbidity.
References
1. Manoharan S, Samarakkody U, Kulkarni M, et al. Evidence based change of practice in the management of unilateral inguinal hernia. J Pediatrr Surg 2005;40:1163-1166.
2. Kurkchubasche A, Tracy T. Unique features of groin hernia repair in infants and children. In: Fitzgibbons R, Greenburg A, eds. Nyus and Condon's Hernia. Philadelphia:Lippincott Williams and Wilkins;2002: 435-451.
3. Lloyd D. Inguinal and femoral hernia. In: Ziegler M, Azizkhan R, Weber T, eds. Operative Pediatric Surgery. New York: McGraw-Hill;2003: 543-554.
4. Brandt ML. Pediatric hernias. Surg Clin North Am 2008;88:27-43.
5. Luo CC, Chao HC. Prevention of unnecessary contralateral exploration using the silk glove sign (SGS) in pediatric patients with unilateral inguinal hernia. Eur J Pediatr 2007;166:667-669.
6. Toki A, Watanabe Y, Sasaki K, et al. Ultrasonographic diagnosis for potential contralateral inguinal hernia in children. J Pediatr Surg 2003; 38:224-226.
7. Chen KC, Chu CC, Chou TY, et al. Ultrasonography for inguinal hernia in boys. J Pediatr Surg 1998;33:1784-1787. Abstract.
8. Niedzielski J, Kr l R, Gawlowska A. Could incarceration of inguinal hernia in children be prevented? Med Sci Monit 2003;9:CR16-18.
9. Stephens BJ, Rice WT, Koucky CJ, et al. Optimal timing of elective indirect inguinal hernia repair in healthy children: Clinical considerations for improved outcome. World J Surg 1992;16:952-956.
10. Zamakhshary M, To T, Guan J, et al. Risk of incarceration of inguinal hernia among infants and young children awaiting elective surgery. CMAJ 2008;179:1001-1005.
11. Kessmann J. Hirschsprung's Disease: Diagnosis and management. Am Fam Physician 2006;74:1319-1322, 1327-1328.
12. Amiel J, Lyonnet S. Hirschsprung disease, associated syndromes, and genetics: A review. J Med Genet 2001;38:729-739.
13. Holschneider AM, Puri P. Hirschsprung's Disease and Allied Disorders. 2nd ed. Amsterdam: Harwood Academic Publishers; 2000.
14. Coran AG, Teitelbaum DH. Recent advances in the management of Hirschsprung's disease. Am J Surg 2000;180:382-387.
15. Parashar UD, Holman RC, Cummings KC, et al. Trends in intussusception-associated hospitalizations and deaths among U.S. infants. Pediatrics 2000;106 :1413-1421.
16. Daneman A, Navarro O. Intussusception. Part 1: A review of diagnostic approaches. Pediatr Radiol 2003;33:79-85. Epub 2002 Nov 19.
17. Yamamoto LG, Morita SY, Boychuk RB, et al. Stool appearance in intussusceptions: Assessing the value of the term "currant jelly." Am J Emerg Med 1997;15:292.
18. Navarro O, Dugougeat F, Kornecki A. The impact of imaging in the management of intussusception owing to pathologic lead points in children. A review of 43 cases. Pediatr Radiol 2000;30:594-603.
19. Sanz N, Sánchez M, García Aroca J, et al. Intussusception: Barium vs. pneumatic reduction. Cir Pediatr 1996;9:21-24.
20. Schafermeyer RW. Pediatric abdominal emergencies. In: Tintinalli J, et al. Emergency Medicine: A Comprehensive Study Guide, 5th ed. New York: McGraw-Hill Company Inc.; 2000:844-852.
21. Williams H. Green for danger! Intestinal malrotation and volvulus. Arch Dis Child Educ Pract Ed 2007;92:ep87-ep91.
22. Torres AM, Ziegler MM. Malrotation of the intestine. World J Surg 1993; 17:326-331.
23. Applegate KE, Anderson JM, Klatte EC. Intestinal malrotation in children: A problem-solving approach to the upper gastrointestinal series. Radiographics 2006;26:1485-1500.
24. Palmas G, Maxia L, Fanos V. Volvulus and intestinal malrotation in the newborn. Pediatr Med Chir 2005;27:62-66.
25. Lampl B, Levin TL, Berdon WE, et al. Malrotation and midgut volvulus: A historical review and current controversies in diagnosis and management. Pediatr Radiol 2009;39:359-366. Epub 2/25/2009.
26. Sizemore AW, Rabbani KZ, Ladd A, et al. Diagnostic performance of the upper gastrointestinal series in the evaluation of children with clinically suspected malrotation. Pediatr Radiol 2008;38:518-528. Epub 2/12/2008.
27. MacMahon B. The continuing enigma of pyloric stenosis of infancy: A review. Epidemiology 2006;17:195-201.
28. Schechter R, Torfs CP, Bateson TF. The epidemiology of infantile hypertrophic pyloric stenosis. Paediatr Perinat Epidemiol 1997;11:407-427.
29. Hernanz-Schulman M. Infantile hypertrophic pyloric stenosis. Radiology 2003;227:319-331. Epub 3/13/2003.
30. Roldán-Valadez E, Solórzano-Morales S, Osorio-Peralta S. Imaging diagnosis of infantile hypertrophic pyloric stenosis: Report of a case and review of the literature. Rev Gastroenterol Mex 2007;72:126-132.
31. Kawahara H, Takama Y, Yoshida H, et al. Medical treatment of infantile hypertrophic pyloric stenosis: Should we always slice the "olive"? J Pediatr Surg 2005;40:1848-1851.
32. Evers BM. Small intestine. In: Townsend CM, ed. Sabiston Textbook of Surgery, 17th ed. Philadelphia:Elsevier;2004;1159-1160.
33. Brunicardi C, et al, eds. Schwartz's Principle of Surgery. 4th ed. New York:McGraw-Hill; 2005:27.
34. Sharma RK, Jain VK. Emergency surgery for Meckel's diverticulum. World J Emerg Surg 2008;3:27.
35. Horn F, Trnka J, Simicková M, et al. Symptomatic Meckel's diverticulum in children. Rozhl Chir 2007;86:480-482.
36. Addiss DG, Shaffer N, Fowler BS, et al. The epidemiology of appendicitis and appendectomy in the United States. Am J Epidemiol 1990;132: 910-925.
37. Liu CD, McFadden DW. Acute abdomen and appendix. In: Greenfield LJ, et al., eds. Surgery: Scientific Principles and Practice, 2nd ed. Philadelphia: Lippincott-Raven; 1997:1246-1261.
38. Grönroos JM; Grönroos P. Leucocyte count and C-reactive protein in the diagnosis of acute appendicitis. Br J Surg 1999;86:501-504.
39. Alvarado A. A practical score for the early diagnosis of acute appendicitis. Ann Emerg Med 1986;15:557-564.
40. Samuel M. Pediatric appendicitis score. J Pediatr Surg 2002;37:877-881.
41. Bhatt M, Joseph L, Ducharme FM, et al. Prospective validation of the pediatric appendicitis score in a Canadian pediatric emergency department. Acad Emerg Med 2009;16:591-596.
42. Hsiao KH, Lin LH, Chen DF. Application of the MANTRELS scoring system in the diagnosis of acute appendicitis in children. Acta Paediatr Taiwan 2005;46:128-131.
43. Schneider C, Kharbanda A, Bachur R. Evaluating appendicitis scoring systems using a prospective pediatric cohort. Ann Emerg Med 2007;49: 778-784.
44. Binnebösel M, Otto J, Stumpf M, et al. Acute appendicitis. Modern diagnostics–Surgical ultrasound. Chirurg 2009;80:579-587.
45. Doria AS, Moineddin R, Kellenberger CJ, et al. US or CT for diagnosis of appendicitis in children and adults? A meta-analysis. Radiology 2006; 241:83-94.
46. Ramarajan N, Krishnamoorthi R, Barth R, et al. An interdisciplinary initiative to reduce radiation exposure: Evaluation of appendicitis in a pediatric emergency department with clinical assessment supported by a staged ultrasound and computed tomography pathway. Acad Emerg Med 2009;16:1258-1265.
47. Garcia Peña BM, Cook EF, Mandl KD. Selective imaging strategies for the diagnosis of appendicitis in children. Pediatrics 2004;113:24-28.
48. Leslie JA, Cain MP. Pediatric urologic emergencies and urgencies. Pediatr Clin North Am 2006;53:3.
49. Kapoor S. Testicular torsion: A race against time. Int J Clin Pract 2008; 62:821-827.
50. Davenport M. ABC of general surgery in children. Acute problem of the scrotum. BMJ 1996;312:435-437.
51. Chmelnik M, Schenk JP, Hinz U, et al. Testicular torsion: Sonomorphological appearance as a predictor for testicular viability and outcome in neonates and children. Pediatr Surg Int 2010;26:281-286.
52. Okatan E, Keijzer R. Images in clinical medicine. Testicular torsion in a hydrocele. N Engl J Med 2009;361:698.
53. Kokoska ER, Keller MS, Weber TR. Acute ovarian torsion in children. Am J Surg 2000;180:462-465.
54. Houry D, Abbot JT. Ovarian torsion: A fifteen-year review. Ann Emerg Med 2001;38:156-159.
55. Anders JF, Powell EC. Urgency of evaluation and outcome of acute ovarian torsion in pediatric patients. Arch Pediatr Adolesc Med 2005;159: 532-535.
56. Vijayaraghavan SB. Sonographic whirlpool sign in ovarian torsion. J Ultrasound Med 2004;23:1643-1649.
Abdominal pain in childhood is a common presentation to the emergency department (ED). While most cases are benign, they frequently create a diagnostic dilemma. The spectrum of disease varies widely from neonates to infants, children, and adolescents, and requires the clinician to rapidly differentiate between a benign medical diagnosis versus a potential surgical abdomen to minimize morbidity and mortality.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
