Vaginal Bleeding in Pregnancy: Part I
Vaginal Bleeding in Pregnancy: Part I
Author:
Pamela Arsove, MD, FACEP, Associate Residency Director, Department of Emergency Medicine, Long Island Jewish Medical Center, New Hyde Park, NY; Assistant Professor of Emergency Medicine, Hofstra North Shore Long Island Jewish School of Medicine.
Peer Reviewer:
Richard S. Krause, MD, Department of Emergency Medicine, University of Buffalo, NY.
Remember the days before sensitive serum pregnancy tests and 24-hour ultrasound availability? Remember performing a culdocentesis to determine if the patient was stable for discharge? I do, and I don't miss them. But with the improved ancillary testing comes increased expectations for perfection, along with the uncertainty of dealing with non-diagnostic results. So, what do you do with a patient with pelvic pain and a serum beta hCG of 247 mIU/mL and a "normal" pelvic ultrasound? From my vantage point, we are getting better. We miss very few ectopic pregnancies, and it is rare for a patient to return to the ED after an initial visit and evaluation with a ruptured tube from an ectopic pregnancy.
The first part of this two-part series will discuss two important conditions that can cause vaginal bleeding in early pregnancy: the most common cause, spontaneous abortion and its subtypes, and the most potentially life-threatening cause, ectopic pregnancy. Part II of this series will discuss other causes of vaginal bleeding that typically present later in pregnancy.
J. Stephan Stapczynski, MD, FACEP,Editor
Introduction
Thirty percent of pregnant patients experience vaginal bleeding at some point during their pregnancy.1 These patients frequently seek medical care in the emergency department (ED), most commonly in the first trimester, when abortion and ectopic pregnancy are most prevalent. According to a study in the American Journal of Obstetrics and Gynecology, from 1993-2003 there were approximately 500,000 ED patient visits/year in the United States for bleeding early in pregnancy.2 The current number of ED visits for vaginal bleeding is likely much higher given the rising trend in ED visits.
Vaginal bleeding from non-obstetrical causes in the pregnant patient is usually apparent from history and physical exam, and will not be discussed in this article. Bleeding from obstetrical causes includes a range of disorders, some common and some rare, that differs significantly between the first and second half of pregnancy. Because most cases of spontaneous abortion occur before the end of the 20th week of gestation, it is convenient to consider any event before that time as the first half of pregnancy. The main causes of bleeding in the first half of pregnancy are due to spontaneous abortion or other problems with fetal implantation and development. In the second half of pregnancy, bleeding is usually from abnormalities of the placenta.3 (See Table 1.) In many tertiary care hospitals, patients in the second half of pregnancy with obstetrical complaints are seen primarily by the obstetrical service. As this is not universal, emergency physicians should understand the differential diagnosis, work-up, and treatment for the causes of obstetrical bleeding among all gestational ages. The primary challenge for the emergency physician is to diagnose potentially dangerous causes of bleeding.
Table 1: Important Obstetric Causes of Bleeding in the First Half and Second Half of Pregnancy
|
First Half of Pregnancy |
Second Half of Pregnancy |
|
Implantation bleeding |
Placenta previa |
|
Spontaneous abortion |
Placental abruption |
|
Anembryonic pregnancy |
Vasa previa |
|
Ectopic pregnancy |
Uterine rupture |
|
Gestational trophoblastic disease |
The purpose of this article is to familiarize the reader with the major causes of bleeding in pregnancy up until the time of labor.
General Considerations
The goal is to determine if the pregnancy is intrauterine or extrauterine, along with the gestational age and viability of the fetus. This assessment is typically done with a combination of serum beta hCG quantitative levels and pelvic ultrasound imaging. Later in pregnancy, when placental issues and fetal viability are the primary concern, ultrasound is the diagnostic test of choice.
Understanding the general principles of beta hCG and ultrasound testing is necessary to interpret the results, especially early in pregnancy. There are well-established parameters for both ancillary tests in early pregnancy. (See Tables 2 and 3.) Findings on transvaginal ultrasound (TVS) are detected about a week earlier than on abdominal ultrasound, and TVS is the preferred scanning technique early in pregnancy, especially if transabdominal ultrasound is inconclusive.
Vaginal Bleeding in the First Half of Pregnancy
Twenty to thirty percent of women experience vaginal bleeding in the first half of pregnancy.3 Bleeding is most commonly caused by spontaneous abortion. With the exception of a ruptured ectopic pregnancy, most early pregnancy bleeding is not life-threatening, and mortality is rare. Routine work-up in the stable patient should include a complete blood count, type and crossmatch, serum beta-hCG, a pelvic exam, and ultrasound. In early pregnancy, urine hCG testing is used to confirm pregnancy. Urine testing is a qualitative test that varies in sensitivity from 5 to 50 mIU/mL, with 20 mIU/mL as the standard in over-the-counter tests.4 False-negative results are rare but do occur, most frequently in dilute urine samples very early in pregnancy when the hormone level is below the testing standard. A quantative serum beta hCG is the most definitive test to evaluate pregnancy in the ED. It should be obtained when the urine test is negative and pregnancy must be definitively ruled out. The quantitative serum hCG test can detect pregnancy within the first week after conception with levels less than 5 mIU/mL.5
In the stable patient with vaginal bleeding, treatment and consultation can await the results of ultrasound imaging and quantitative beta hCG testing. For the hemodynamically unstable patient, treatment is initiated immediately. A qualitative serum hCG test is done to confirm pregnancy along with a bedside ultrasound if available, and emergent consultation is obtained. The serum beta hCG does not alter management in this circumstance. In the unstable patient, two large-bore IVs should be placed for volume resuscitation with normal saline and, if available, a bedside pelvic or transabdominal ultrasound should be done to look for free fluid (e.g., blood) in the abdomen or pelvis. If free fluid is seen, an emergency obstetric consultation should be obtained.
Table 2: TVS Findings and Corresponding Gestational Age in Normal Pregnancy
|
TVS |
Gestational Age |
|
Double decidual sac sign |
5 weeks: uterine/placental lining interface |
|
Gestational sac |
5 weeks |
|
Yolk sac |
5.5 weeks |
|
Cardiac activity |
6 weeks, fetus is viable |
|
Yolk sac |
Sac diameter 10 mm or greater |
|
Fetal pole |
Sac diameter 1.8 cm or greater |
Implantation Bleeding. The patient's last menstrual period (LMP, calculated from first day of the most recent normal menstrual period) is an often unreliable estimate of gestational age.6 In a normal pregnancy, implantation of a fertilized egg occurs approximately 5–12 days after ovulation, or about 3 weeks after a patient's LMP. During implantation, many women may experience implantation bleeding as the trophoblastic tissue embeds into the myometrium. This bleeding is characteristically light, occurs earlier than expected, and is of shorter duration than a normal menstrual period. Patients should be questioned as to the timing and duration of their last menses, as they often confuse implantation bleeding as a lighter-than-normal cycle and may not realize they are pregnant. Implantation bleeding is a diagnosis of exclusion after an appropriate work-up for pathologic causes of bleeding.
Table 3: Characteristics of Serum Beta hCG in Normal Pregnancy
- The beta subunit is secreted by trophoblasts of embryonic placenta.
- It is detectable in serum 8-11 days after conception.
- The half life is 1.5 to 2 days.
- Levels increase more than 66% every two days in 85% of normal pregnancies.
- Levels peak at 10-12 weeks gestational age.
- Levels gradually decrease starting in the second trimester.
- After delivery, levels usually become undetectable by 3-4 weeks.
Spontaneous Abortion. Spontaneous abortion is a general term for the loss of a pregnancy prior to fetal viability outside the uterus. It is the most common cause of bleeding in the first half of pregnancy, with more than 80% occurring in the first 12 weeks.7 The true incidence of spontaneous abortion is unknown because the majority occur before pregnancy is noticed.8 Of the clinically recognized pregnancies, 15-20% end in spontaneous abortion.9 Major risk factors for clinically apparent abortion in early pregnancy include advanced maternal and paternal age, cigarette smoking, and previous miscarriages.10 The types of spontaneous abortion represent a spectrum of the same pathologic process and are distinguished from one another by the degree to which the products of conception are retained or expelled from the uterus. Fetal death occurs prior to abortion. (See Table 4.) The majority of deaths are due to chromosomal abnormalities in the fetus, with a smaller percentage caused by maternal disease such as diabetes and hypertension, or structural abnormalities of the uterus.11 Signs and symptoms of spontaneous abortion are bleeding; abdominal, pelvic, or back pain; a dilated cervix; and visualization of products of conception on exam. A single serum beta hCG rarely alters management in the ED, but is useful in serial testing at follow-up.
Table 4: Types of Spontaneous Abortion
- Threatened abortion
- Inevitable abortion
- Incomplete abortion
- Complete abortion
- Missed abortion
- Septic abortion
Types of Spontaneous Abortion and Corresponding Ultrasound Findings. Threatened abortion is defined as any bleeding with no evidence of fetal demise on ultrasound and a closed cervical os. It occurs in 20-25% of early pregnancies.12 Bleeding is typically mild, but may last from days to weeks. The risk of completed abortion in these patients is roughly 50%. If a fetal heartbeat is visualized at 5-6 weeks, the risk decreases to 4.5% in women younger than 36 years of age.13 In older patients, the risk is greater. In approximately 20% of patients, ultrasound shows a subchorionic hematoma, which is a blood clot between the placental and uterine membranes.14 (See Figure 1.)
Figure 1: Subchorionic Hematoma
Transvaginal ultrasound of a subchorionic hematoma (arrow). The fetus is seen sitting above the hematoma. Image courtesy of Tripti Shah, MD.

TVS findings suggestive of an impending miscarriage include a large, irregular sac, a subchorionic hematoma greater than 66% of the sac size, a fetal heart rate less than 85 beats/minute, and an embryo small for dates.15 In this situation, patients should be counseled about the possibility of miscarriage, and close follow-up should be ensured. For patients who do not abort, there is an increase in both fetal and maternal morbidity and mortality, suggesting these patients are at high risk and should be followed closely.16,17
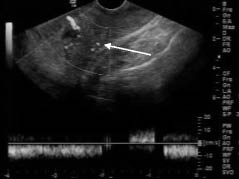
Inevitable abortion is one in which the cervical os has opened (defined as more than 2 cm or the ability to insert the examiner's finger tip into the cervical canal) but the products of conception have not yet been expelled. A fetal heartbeat may or may not be present. Almost all progress to complete abortion. TVS reveals products of conception in the lower uterine segment or cervical canal. (See Figure 2.)
Figure 2: Inevitable Abortion
Transvaginal ultrasound of fetus exiting the lower cervical segment of the uterus. Image courtesy of Tripti Shah, MD.
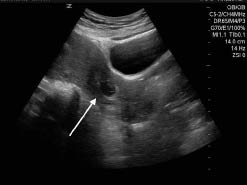
Incomplete abortion is one in which part of the products of conception are retained in the uterus. It is more common after the eighth week and typically presents with persistent bleeding and abdominal cramping.1 Patients can't reliably distinguish between the passage of clot or tissue, so the diagnosis is made on visualization of products of conception extruding from the cervical os, or by TVS. (See Figure 3.)
Figure 3: Retained Products of Conception
Transvaginal ultrasound with color Doppler showing the vascularity seen with retained products of conception (arrow). Image courtesy of Tripti Shah, MD.
A blood clot can be difficult to distinguish from retained products of conception on TVS. Recent studies showing predictable ultrasound findings for retained products of conception are the presence of hyperechoic material within the uterus, endometrial vascularity with color Doppler, and endometrial thickness greater than 15 mm.18,19
Complete abortion occurs when all products of conception are expelled from the uterus. Bleeding often is accompanied by abdominal pain, which resolves with passage of the products of conception. On pelvic exam, bleeding is usually minimal, the os is closed, and there is no tenderness. In approximately 30% of spontaneous abortions thought to be clinically complete (decreasing pain and bleeding, closed cervical os), the TVS will visualize retained products. This is more likely if the spontaneous abortion occurs after 12 weeks. Therefore, ultrasound should be considered in these patients.
Missed abortion is a condition where ultrasound demonstrates fetal demise, and the fetus is retained in the uterus. Characteristically, fetal age is less than gestational age if the patient's last menstrual period is reliable. These patients often are asymptomatic, but seek medical attention in the ED when bleeding occurs. On ultrasound, diagnosis is made when no cardiac activity is noted in an embryo of 5 mm or greater.
Septic abortion refers to any induced or spontaneous abortion that is complicated by a pelvic infection. Septic abortion characteristically presents with fever, malodorous discharge, and abdominal and pelvic tenderness.1 If untreated, systemic infection will result. This diagnosis is uncommon in the developed world where abortion is legal, but common in countries where nonsterile induced abortions are performed.20 Ultrasound findings depend on the type of abortion (see above). A hematometra or a collection of blood in the uterine cavity may be present.
Management of Spontaneous Abortion. Because fetal loss cannot be prevented in the first half of pregnancy, the goal in management of spontaneous abortion is to prevent significant blood loss and infection in the mother. Any patient greater than 13 weeks gestation or who has hemodynamic instability, significant ongoing blood loss, or evidence of septic abortion requires emergency obstetric consultation. In most of these cases, surgical evacuation of the uterus is the treatment of choice. In septic abortion, broad-spectrum antibiotics covering aerobic and anaerobic bacteria should be started as soon as the diagnosis is suspected.
The value of anti-D immunoglobin (RhoGAM®) treatment of an Rh-negative patient who is having vaginal bleeding in early pregnancy (e.g., threatened spontaneous abortion or other subtypes) is unclear.21,22 However, it is commonly recommended by expert consensus opinion and has become standard in most settings, primarily because while the benefit is largely theoretical and implied, the risk of anti-D immunoglobin is essentially zero.23 The current recommendation is that non-sensitized Rh-negative women experiencing a threatened abortion, spontaneous abortion, or induced abortion should be treated with anti-D immunoglobin. The recommended dose has usually varied according to the potential amount of fetal blood to which the patient is exposed as determined by the estimated gestational age. For example, for an estimated gestational age of less than 12 weeks, the dose of anti-D immunoglobin is recommended to be 120 to 150 micrograms and for an estimated gestational age of greater than 12 weeks, the dose is 300 micrograms.23 As already noted, it is sometimes difficult to accurately estimate gestational age, and some physicians may chose to routinely use the 300 microgram dose regardless of estimated gestational age. The same expert opinion also recommends anti-D immunoglobin therapy in the non-sensitized Rh-negative patient with an ectopic pregnancy.23
Stable patients with spontaneous abortions may be treated expectantly, or with medical or surgical management. Successful outcomes are associated with each of these interventions.24-26 Expectant management involves no intervention. Medical management most commonly involves treatment with mifepristone, an antiprogestational steroid that acts as an abortifacient, with or without misoprostol, a medication that causes uterine contraction. Dilatation and curettage or evacuation is the traditional surgical approach for spontaneous abortion.
When the diagnosis of threatened or completed abortion is made, stable patients can be discharged from the ED with close follow-up and instructions to return if significant bleeding occurs. In threatened abortion, there is no scientific evidence to support bed rest or pelvic rest to prevent miscarriage.27
In inevitable, incomplete, and missed abortion, the ED physician's treatment plan should be made in conjunction with the patient and her obstetrician. The patient may also have cultural expectations for a particular approach. For example, patients from some Latin American countries may expect surgical management because that is the very common practice in their native country. If the physician does not recommend or perform surgical management, the patient may believe she is not receiving proper care and decide to go elsewhere rather than waiting for expectant management to work. Patients may not voice their expectations directly to the physician, so it is helpful to ask, particularly with an understanding translator if needed. Explanations that expectant management is more "natural" and less potentially damaging to the uterus may help persuade the patient that the physician is providing proper care.
If the pelvic exam shows products of conception extruding from the external os, they should be manually removed with ring forceps. This may be all that is needed to allow the uterus to contract and stop the bleeding.
Published literature supports expectant management alone as an initial treatment strategy in the first trimester, with the majority of women having spontaneous passage of tissue within 2-6 weeks.28 The success of medical management with prostaglandins varies with the dose and route of administration (oral, buccal, or vaginal).29,30 There is no standardized optimal dose, frequency, or route of administration, and the ED physician should use this medication only in consultation with an obstetrician. Both expectant and medical management have a higher risk of bleeding and incomplete abortion compared to surgical management.31 Surgical treatment offers the advantage of quicker resolution of symptoms. The risks of surgery include uterine perforation or adhesions, infection, and cervical trauma. There is no significant difference in success rates between medical and surgical management, however, and patient preference should be considered.32
Anembryonic Pregnancy. An anembryonic pregnancy, previously termed a "blighted ovum," is a nonviable intrauterine pregnancy. The fertilized egg implants into the myometrium, and the gestational sac grows, but there is no visible embryonic development. The etiology is unknown, but it is assumed to be secondary to chromosomal abnormalities.33 Presentation is indistinguishable from other causes of bleeding in early pregnancy. The diagnosis is made when ultrasound shows a mean sac diameter greater than 8 mm without a yolk sac, or a mean gestational sac greater than 16 mm with no embryo.34 Management follows the same guidelines as missed abortion. Emergency physicians should be cautious about providing a definite diagnosis of anembryonic pregnancy after a single ED evaluation. Ultrasounds in early pregnancy may miss a small fetal pole that will be seen on subsequent examinations. The patient may experience both undue worry and distrust of the physician if this diagnosis is in error.
Ectopic Pregnancy. Ectopic pregnancy is the most common cause of maternal mortality in the first trimester of pregnancy. Ectopic pregnancy occurs when an embryo implants outside of the uterine cavity. The most common site of implantation is the fallopian tube, but other sites include the ovary, the cervix, and the abdomen. (See Figure 4.) An ectopic pregnancy located in the interstitium of the tube, also known as a cornual ectopic, differs from other fallopian tube ectopics by its proximal location near the myometrium. The surrounding myometrial tissue may allow it to grow to a large size before becoming symptomatic. This type of ectopic pregnancy is noteworthy because it may appear to be intrauterine to the unskilled sonographer and can present with tubal or uterine rupture in the second trimester. A heterotopic pregnancy has a simultaneous intrauterine pregnancy (IUP) and ectopic gestation. The incidence of heterotopic pregnancy varies according to the clinical circumstance. The historical control rate was about 0.3 cases per 10,000 pregnancies, or about 1 in 30,000.35 In patients with a history of prior pelvic inflammatory disease, the rate increases to approximately 1 in 3000 pregnancies.36,37 In a patient currently undergoing ovarian induction treatment to facilitate conception, the rate is about 1 in 300 pregnancies.38,39 The highest rate is seen in patients undergoing assisted reproductive technology (e.g., in vitro fertilization), with a rate as high as 1 in 100 pregnancies. Patients undergoing assisted reproductive technology are at much higher risk for this complication than other patients.40-42
Figure 4: Location of Ectopic Pregnancy
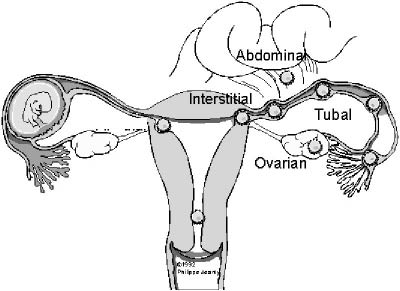
Figure reprinted with permission from TheFetus.net © 1992 Phillipe Jeanty.
In the United States, official reporting of ectopic pregnancies stopped in 1992, so accurate prevalence and mortality data are not available. Data prior to 1992 showed an increase in the incidence of ectopic pregnancies, but a decrease in mortality.43 Sensitive serum beta hCG tests and TVS have given the physician a better understanding of the management of this disease and allow for earlier diagnosis. Risk factors associated with ectopic pregnancy should increase the physician's suspicion for the diagnosis, but do not alter the work-up of the patient as their absence does not exclude ectopic pregnancy. (See Table 5.)
Table 5: Risk Factors for Ectopic Pregnancy
|
Risk Factor |
Odd Ratio (Approximate Confidence Intervals) |
|
Previous tubal sugery |
21 (9-47) |
|
Previous tubal ligation |
9 (5-18) |
|
Previous ectopic pregnancy |
8 (6-11) |
|
Current IUD use |
5 (1-28) |
|
Current infertility treatment |
5 (1-28) |
|
History of pelvic inflammatory disease |
2.5 (2-3) |
|
Data from: Mol BW, Ankum WM, Bossuyt PM, et al. Contraception and the risk of ectopic pregnancy: A meta-analysis. Contraception 1995;52:337-341. Ankum WM, Mol BW, Van der Veen F, et al. Risk factors for ectopic pregnancy; A meta-analysis. Fertil Steril 1996;65:1093-1099. Dart RG, Kaplan B, Varaklis K. Predictive value of history and physical examination in patients with suspected ectopic. Ann Emerg Med 1999;33:283-290. |
|
History and physical exam findings are unreliable in the diagnosis of ectopic pregnancy in the stable patient.44,45 Common presentations include some combination of missed menses and abdominal pain with or without vaginal bleeding. The differential diagnosis includes other more common causes of first-trimester bleeding, such as threatened abortion. A palpable mass on bimanual exam is not a reliable or consistent finding on physical examination.46 Patients with ruptured ectopic pregnancies are easier to diagnose. They typically present with sudden onset of severe, localizing abdominal pain, a positive pregnancy test, and rebound or guarding on abdominal examination. It is important to note that rare cases of ruptured ectopic pregnancies with negative pregnancy tests have been reported.47,48
The diagnosis of ruptured ectopic pregnancy can be done by performing a bedside ultrasound. The finding of free pelvic fluid or fluid in Morrison's pouch is all the ED physician needs to make a presumptive diagnosis. If an intrauterine pregnancy is visualized, the differential diagnosis narrows to a heterotopic pregnancy with rupture, or rupture of a hemorrhagic corpus luteal cyst with a normal gestation. In either situation, the definitive diagnosis will be made in the operating room.
The diagnostic work up to exclude ectopic pregnancy in the stable patient includes ultrasound and quantitative serum beta hCG. Visualizing a gestational sac is reassuring if a double ring sign is present. The pseudogestational sac of an ectopic pregnancy lacks this distinguishing double membrane and may be confused with an early IUP. A definitive diagnosis of ectopic pregnancy can only be made when a fetal heartbeat is visualized outside the uterus. Other ultrasound findings suggestive of an ectopic pregnancy are: a pseudogestational sac, an extrauterine sac with a yolk sac or fetal pole (see Figure 5), a complex adnexal mass, a cystic structure in the adnexa separate from the uterus and ovary, or moderate amount of free pelvic fluid.49 A recent study evaluating ED physicians' learning curve performing bedside ultrasound for first-trimester pregnancies found suboptimal results for detecting ectopic pregnancy or adnexal masses.50 Caution must be taken in interpreting results from the less experienced sonographer, whether in the ED or radiology suite.
Figure 5: Ectopic Pregnancy
Extrauterine pregnancy in the interstitial portion of the fallopian tube. The embryo is seen to the left in an ectopic gestational sac (white arrow). Figure reprinted with permission from TheFetus.net. © 2009 Andrey Volkov.
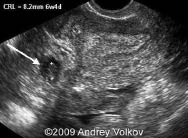
If an IUP is not visualized on transabdominal ultrasound or TVS, serum beta hCG results are compared against the discriminatory zone (DZ). The DZ is a cutoff serum hCG level above which, in a normal IUP, products of conception should be visualized on TVS. The level varies among different institutions but is usually about 1,500 mIU/mL. Values are lower for TVS and, thus, this is the preferred technique, especially when transbdominal ultrasound is nondiagnostic.
Figure 6: The Discriminatory Zone (DZ)

The area under the curve represents the discriminatory zone. In a normal pregnancy, TVS may or may not reveal an IUP when the serum hCG is within the discriminatory zone. Once the serum hCG is above the cutoff for the DZ (thick arrow), an IUP should be seen.
Ultrasound findings suspicious for ectopic pregnancy are present when no IUP is visualized and the serum hCG level is above the discriminatory zone. In this situation, ectopic pregnancy should be suspected, and obstetrical consultation obtained. When no IUP is visualized on TVS and the serum beta hCG is below the DZ, an early IUP cannot be distinguished from an ectopic pregnancy.
Patients with these findings who are stable and have reliable follow-up may be discharged from the ED. In these patients, serial quantitative beta hCG determinations must be performed every 48 hours until a diagnosis is made. Failure of beta hCG values to approximately double every two days suggests an ectopic pregnancy or other pathologic pregnancy.
Despite the availability of sensitive beta hCG and ultrasound testing, pitfalls in the diagnosis of ectopic pregnancy remain. (See Table 6.)
Table 6: Pitfalls to Be Avoided in the Diagnosis of Ectopic Pregnancy
- Assuming a low serum hCG rules out an EP
- Relying on an inexperienced sonographer
- Failure to assure adequate follow-up if no IUP seen
- Failure to review pathology for products of conception if patient presents post dilation and curettage
- Failure to consider heterotopic pregnancy if unstable, and IUP seen
- Relying on ultrasound results done at another facility
- Misdiagnosing corneal ectopic as IUP
- Assuming patient had complete abortion when no products of conception seen
The management of ectopic pregnancy depends on whether it has ruptured. Ruptured ectopic pregnancies are considered unstable and treated surgically. Stabilization with aggressive fluid resuscitation and pre-op labs including type and cross match must be instituted early. Emergent obstetrical consultation is mandatory as soon as the diagnosis is suspected.
Unruptured ectopic pregnancies may be treated medically, surgically, or expectantly. Treatment decisions must be made in consultation with the patient and her obstetrician. Decisions on treatment options take into account patient compliance, cost, risk of treatment failure, and side effects. Medical management with methotrexate (MTX) has gained popularity in recent years since its introduction in the 1980s and has the advantage of being a non-invasive outpatient treatment option. The ideal candidate for medical management is one who will comply with follow-up and is diagnosed early. Emergency physicians should have obstetric consultation (preferably in person, not by phone) before initiating MTX treatment for ectopic pregnancy in the ED.
Patients with a serum beta hCG greater than 5000 mIU/mL have a higher incidence of treatment failure.51 MTX is usually given IM in single- or multi-dose regimens.52 Abdominal pain is a common side effect of MTX, which may occur 6–7 days after treatment.53 If a patient returns to the ED with abdominal pain after treatment with MTX, rupture of the ectopic is the primary concern. A repeat ultrasound must be performed looking for signs of hemoperitoneum. Adnexal size on TVS and serum hCG measurements may actually increase after MTX, and are therefore unreliable indicators of MTX failure.54
Surgery is performed when a patient has a contraindication to MTX, hemodynamic instability, heterotopic pregnancy, impending rupture of ectopic pregnancy, ongoing abdominal pain, greater than 300 cc free fluid, is not available for follow up, or has failed medical therapy.55 Laparoscopy is the surgical treatment of choice. Expectant management of ectopic pregnancy is based on the premise that many ectopic pregnancies spontaneously regress over time. It may be considered in women with low serum beta hCG levels or levels that are known to be decreasing, but is not the treatment of choice for most physicians. Studies are currently underway to evaluate expectant management as a viable treatment option in select patient populations.56
References
1. Keeling J, Khong T. Spontaneous abortion and the pathology of early pregnancy. Fetal and Neonatal Pathology 2007:102-122.
2. Wittels KA, Pelletier AJ, Brown DFM, et al. United States emergency department visits for vaginal bleeding during early pregnancy, 1993-2003. Am J Obstet Gynecol 2008;198:523.e1-523.e6.
3. Yang J, Hartmann K, Savitz D, et al. Vaginal bleeding during pregnancy and preterm birth. Am J Epidemiol 2004;160: 118-125.
4. Cole L, Ladner D. Background hCG in non-pregnant individuals: Need for more sensitive point-of-care and over-the-counter pregnancy tests. Clinical Biochemistry 2009;42:168-175.
5. Omori S, Suehiro M, Fukuchi M. [Fundamental evaluation of ELSA.F-beta HCG kit as an immunoradiometric assay specific for serum beta HCG]. Kaku Igaku 1992;29:709-16.
6. Dietz PM, England LJ, Callaghan WM, et al. A comparison of LMP-based and ultrasound-based estimates of gestational age using linked California livebirth and prenatal screening records. Paediatr Perinat Epidemio 2007; 21: 62-71.
7. Coppola P, Coppola M. Vaginal bleeding in the first 20 weeks of pregnancy. Emerg Med Clin N Am 2003;21:667–677.
8. Wilcox A, Weinberg C, O'Conno J, et al. Incidence of early loss of pregnancy. N Engl J Med 1988;319:189-194.
9. Griebel C, Halvorsen J, Golemon T, et al. Management of spontaneous abortion. Am Fam Physician 2005;72:1243-1250.
10. Arck P, Rucke M, Rose M. Early risk factors for miscarriage: A prospective cohort study in pregnant women. Reprod Biomed Online 2008;17:101-13.
11. Simpson, J. Genetic causes are responsible for most first trimester pregnancy losses. International Journal of Gynecology & Obstetrics 2000;70:D15.
12. Sotiriadis A, Papatheodorou S, Makrydimas G. Threatened miscarriage: evaluation and management. BMJ 2004;329:152.
13. Deaton JL, Honore GM, Huffman CS, et al. Earrly transvaginal ultrasound following an dated pregnancy: The importance of finding a yolk sac or fetal heart motion. Hum Reprod 1997;12:2820.
14. Maso, G; D'Ottavio, G, De Seta, F. First-trimester intrauterine hematoma and outcome of pregnancy. Obstetrics & Gynecology 2005;105:339-344.
15. Dighe M, MD, Cuevas C, Moshiri M, et al. Sonography in first trimester bleeding. J Clin Ultrasound 2008;36:352–366.
16. Dadkhah F, Kashanian M, Eliasi G. A comparison between the pregnancy outcome in women both with or without threatened abortion. Early Human Development 2010;86:193-196.
17. Saraswat L, Bhattacharya S, Maheshwari A,et al. Maternal and perinatal outcome in women with threatened miscarriage in the first trimester: A systematic review. BJOG 2010;11:245-57.
18. Abbasi, S, Jamal A, Eslamian L, et al. Role of clinical and ultrasound findings in the diagnosis of retained products of conception. Ultrasound Obstet Gynecol 2008;32:704–707.
19. Kamaya A, Petrovitch I, Chen B, et al. Retained Products of Conception: Spectrum of Color Doppler Findings. J Ultrasound Med 2009;28:1031-1041.
20. Rahangdale L. Infectious complications of pregnancy termination. Clin Obstet Gynecol 2009;52:198-204.
21. Jabara S, Barnhart KT. Is Rh immune globulin needed in early first-trimester abortion? A review. Am J Obstet Gynecol 2003;188:623-627.
22. Hannafin B, Lovecchio F, Blackburn P. Do Rh-negative women with first trimester spontaneous abortions need Rh immune globulin? Am J Emerg Med 2006;24:487-489.
23. Fung Kee Fung K, Eason E, et al; Maternal-Fetal Medicine Committee, Genetics Committee. Prevention of Rh alloimmunization. J Obstet Gynaecol Can 2003;25:765-773.
24. Neilson JP, Gyte GML, Hickey,M, et al. Medical treatments for incomplete miscarriage (less than 24 weeks). Cochrane Database Syst Rev 2010; 1: CD007223.
25. Gronlund L, Gronlund AL, Clevin, et al. Spontaneous abortion: expectant management, medical treatment or surgical evacuation. Acta Obstet Gynecol Scand 2002;81:781–2.
26. Casikar T, Bignardi J, Riemke J, et al. Expectant management of spontaneous first-trimester miscarriage: Prospective validation of the "2-week rule." Ultrasound Obstet Gynecol 2010;35:223–227.
27. Aleman A, Althabe F, Belizán J, et al. Bed rest during pregnancy for preventing miscarriage. Cochrane Database Syst Rev 2005;18;CD003576.
28. Casikar I, Bignardi T, Riemke J, et al. Expectant management of spontaneous first-trimester miscarriage: prospective validation of the "2-week rule." Ultrasound in Obstetrics and Gynecology 2010;35:223–227.
29. Tang OS, Gemzell-Danielssson K, Ho PC. Misoprostol: Pharmokinetic profiles, effects on the uterus and side effects. International Journal of Gynecology and Obstetrics 2007;99:160-167.
30. Allen R, O'Brien B. Uses of misoprostol in obstetrics and gynecology. Rev Obstet Gynecol 2009;2:159–168.
31. Moodliar S, Bagratee JS, Moodley J. Medical vs. surgical evacuation of first-trimester spontaneous abortion. International Journal of Gynecology and Obstetrics 2005;91:21-26.
32. Hajenius PJ, Mol F, Mol BWJ, Bossuyt PMM, et al. Interventions for tubal ectopic pregnancy. Cochrane Database Syst Rev 2007, Issue 1. Art. No.: CD000324.
33. Munoz M, Arigita M, Bennasar M, et al. Chromosomal anomaly spectrum in early pregnancy loss in relation to presence or absence of an embryonic pole. Fertility and Sterility 2010; doi:10.1016/j.fertnstert.2010.04.011.
34. Dighe M, MD, Cuevas C, Moshiri M, et al. Sonography in first trimester bleeding. J Clin Ultrasound 2008;36:352–366.
35. DeVoe RW, Pratt JH. Simultaneous intrauterine and extrauterine pregnancy. Am J Obstet Gynecol 1948;56:1119-26.
36. Richards SR, Stempel LE, Carlton BD. Heterotopic pregnancy: Reappraisal of incidence. Am J Obstet Gynecol 1982;142:928-30.
37. Bello GV, Schonholz D, Moshirpur J, et al. Combined pregnancy: the Mount Sinai experience. Obstet Gynecol Surv 1986;41:603-13
38. Schenker JG, Yarkoni S, Granat M. Multiple pregnancies following induction of ovulation. Fertil Steril 1981;35:105-23.
39. Glassner MJ, Aron E, Eskin BA. Ovulation induction with clomiphene and the rise in heterotopic pregnancies. A report of two cases. J Reprod Med 1990;35:175-8.
40. Yovich JL, McColm SC, Turner SR, et al. Heterotopic pregnancy from in vitro fertilization. J In Vitro Fert Embryo Transf 1985;2:143-50.
41. Dor J, Seidman DS, Levran D, et al. The incidence of combined intrauterine and extrauterine pregnancy after in vitro fertilization and embryo transfer. Fertil Steril 1991;55:833-4.
42. Goldman GA, Fisch B, Ovadia J, et al. Heterotopic pregnancy after assisted reproductive technologies. Obstet Gynecol Surv 1992;47:217-21.
43. Centers for Disease Control and Prevention. Current trends ectopic pregnancyUnited States, 1990-1992. MMWR Morb Mortal Wkly Rep 1995;44;46-48.
44. Brown T, Herbert M. Medical myth: Bimanual pelvic examination is a reliable decision aid in the investigation of acute abdominal pain or vaginal bleeding. CJEM 2003;5:120-122.
45. Dart R, Kaplan B, Varaklis K. Predictive value of history and physical examination in patients with suspected ectopic pregnancy. Ann Emerg Med 1999;33:283-290.
46. Brown T, Herbert ME. Medical myth: Bimanual pelvic examination is a reliable decision aid in the investigation of acute abdominal pain or vaginal bleeding. CJEM 2003;5:120-2.
47. Lee JKS, Lamaro VP. Ruptured tubal ectopic pregnancy with negative serum beta hCGa case for ongoing vigilance? J New Zealand Medical Association 2009;122:1288.
48. Grynberg M. Rupture of ectopic pregnancy with negative serum ß-hCG leading to hemorrhagic shock. Obstetrics & Gynecology 2009;113:537-539.
49. Lin E, Bhatt S, Dogra V. Diagnostic clues to ectopic pregnancy. RadioGraphics 2008;28:1661-1671.
50. Jang T, Ruggeri W, Dyne P, et al. Learning curve of emergency physicians using emergency bedside sonography for symptomatic first-trimester pregnancy. J Ultrasound Med 2010;29:1423-1428.
51. Menon S, Collins J, Barnhart K. Establishing a human chorionic gonadotropin cutoff to guide methotrexate treatment of ectopic pregnancy: A systematic review. Fertility and Sterility 2007;87:481-484.
52. Barnhart, KT, Gosman G, Asnby R, et al. The medical management of ectopic pregnancy: A meta-analysis comparing single dose and multidose regimens. Obstet Gynecol 2003;101:778.
53. Dilbaz S, Guvendag Guven ES, Aykan Yildirim B, et al. Is it necessary to operate on all women with an acute abdomen following medical treatment of tubal ectopic pregnancy? J Obstet & Gynaecol 2010;30:496-500.
54. Thurman A, Cornelius M, Korte J, et al. An alternative monitoring protocol for single-dose methotrexate therapy in ectopic pregnancy. Am J Obstet & Gynecol 2010;202:139.e1-139.e6.
55. Al-Sunaidi M, Tulandi T. Surgical treatment of ectopic pregnancy. Semin Reprod Med 2007;25:117-122.
56. van Mello N, Mol F, Adriaanse A, et al. The METEX study: Methotrexate versus expectant management in women with ectopic pregnancy: A randomised controlled trial. BMC Women's Health 2008;8:10.
Remember the days before sensitive serum pregnancy tests and 24-hour ultrasound availability? Remember performing a culdocentesis to determine if the patient was stable for discharge? I do, and I don't miss them.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
