B-Type Natriuretic Peptide: Clinical Utility in the Emergency Department Setting
Authors: Claudie H. Jimenez, MD, Clinical Instructor, Department of Emergency Medicine, University of Texas Southwestern Medical Center, Dallas; and Kurt Kleinschmidt, MD, Associate Professor of Surgery, Division of Emergency Medicine, University of Texas Southwestern Medical Center; Associate Director, Emergency Services Department, Parkland Memorial Hospital, Dallas.
Peer Reviewers: W. Frank Peacock IV, MD, FACEP, Director of Clinical Operations, Department of Emergency Medicine, Cleveland Clinic Foundation, Cleveland, OH; and Sean P. Collins, MD, Assistant Professor, Department of Emergency Medicine, University of Cincinnati/University Emergency Physicians, Cincinnati, OH.
The diagnosis and management of heart failure (HF) present unique challenges for the emergency medicine physician. While there is no ambiguity that HF is characterized by ventricular dysfunction (systolic or diastolic) and evidence of circulatory failure, including the presence of fluid retention and fatigue,1 confirming the diagnosis in a patient with multiple comorbid conditions can be problematic. In addition, mortality continues to increase from HF while death rates associated with coronary artery disease (CAD) and stroke are decreasing.1 HF is a common precipitant for acute hospitalization and is a major economic burden on developed countries.2 In the United States, almost 5 million people are afflicted with HF, and 400,000 new patients are diagnosed with HF each year.1 Of special concern is the fact that among all patients with this condition, about 35% will require at least one hospitalization each year, and the rate is increasing. Overall expenditure related to congestive HF (CHF) are $18.8 billion per year.1
Risk factors for HF include age, gender, hypertension, and CAD.1,3 The incidence of HF is 3 in 1000 in men age 50-59 years, but increases to 27 in 1000 in men by age 80-89 years. Patients with left ventricular hypertrophy on their resting electrocardiograms (ECGs) are at significantly greater risk of developing HF.1 Hypertension precedes the diagnosis of HF in 91% of patients,3 and approximately 85% of HF cases in the general population are associated with hypertension or CAD.2
As experienced physicians understand, HF can be a difficult diagnosis to establish in the emergency department (ED) setting, especially when there are factors that may complicate a patient’s presentation. In this regard, many HF symptoms are non-specific and co-exist with other diagnoses. Dyspnea on exertion has up to 100% sensitivity in HF but a specificity of only 17%. Orthopnea is 22% sensitive and 74% specific for the diagnosis of HF. Other classic symptoms of HF, such as lower extremity edema, rales upon pulmonary auscultation, or a presentation with diuretic use, are 47%, 77%, and 41% specific, respectively.4,5 A gallop rhythm is 99% specific,4,5 but this sign often is difficult to discern. Although the gold standard for confirming the diagnosis of CHF is echocardiography,4 this modality usually is unavailable in the ED.
Put simply, current diagnostic approaches for HF are less than optimal for establishing the diagnosis and guiding treatment decisions. As a result, an easy-to-use, non-invasive test available in the ED that can assist in confirming the diagnosis of HF, that yields prognostic information, and that may guide treatment decisions, including the need for DVT prophylaxis in patients with HF, would be a welcome addition to the ED assessment armamentarium. With these considerations in focus, this issue of Emergency Medicine Reports reviews the role and clinical utility of brain natriuretic peptide (BNP) in the management of patients suspected of having HF upon presentation to the ED.—The Editor
Pathophysiology: What is Brain Natriuretic Peptide?
The heart was found to have properties characteristic of an endocrine organ in 1956 when Kisch6 and others detected secretory granules in guinea pig atria. In 1984, the structure determined to be atrial natriuretic peptide (ANP) was identified.7
In 1988, B-type natriuretic peptide was identified from porcine brain. It causes natriuretic and diuretic effects similar to ANP.8 While this new peptide was called brain natriuretic peptide, it became clear that the primary site of synthesis was in the ventricles of the heart.9,10 Like ANP, BNP is stored as a prohormone within secretory granules and is secreted as an N-terminal fragment, N-proBNP, and a smaller active hormone, BNP. The circulating half-lives of BNP and N-proBNP do not differ to the same extent as ANP and N-proANP; therefore, there is no reason to select N-proBNP over BNP as a cardiac biomarker.11 The effects of BNP on the kidney, plasma renin activity, and aldosterone concentration are similar to those of ANP.12 BNP gene expression occurs more rapidly in response to stimulus than ANP.13 BNP is a vasodilator, has a natriuretic action, and antagonizes the effects of the renin-angiotensin-aldostereone system.2 It causes vasodilatation by increasing intracellular cyclic guanosine monophosphate (cGMP) via activation of guanylate cyclase. (See Table 1.)14
|
ANP and BNP do not cross the blood-brain barrier, but they do reach the hypothalamus and the subfornical organ. The effects of ANP and BNP in the brain reinforce their peripheral actions. They decrease sympathetic tone within the brainstem.15 BNP receptors particularly are concentrated in the hypothalamus where the peptide inhibits the secretion of arginine vasopressin and paradoxically stimulates sympathetic tone.15
BNP secretion directly correlates with severity of HF.16 Increased myocardial wall stretch is the main stimulus for cardiac natriuretic peptide secretion.2 One study compared BNP levels in patients with idiopathic dilated cardiomyopathy (IDC) to those with hypertrophic cardiomyopathy. Patients in both groups had similar left ventricular (LV) end diastolic pressure and similar LV mass; however, BNP levels were four times higher in the group with IDC. This correlated with the differences in LV end diastolic volume, suggesting that stretch rather than transmural pressure is the primary stimulus for BNP synthesis.16
The natriuretic peptides ANP and BNP are eliminated by two mechanisms. The peptides bind to the natriuretic peptide clearance receptor (NPR-C), resulting in their cellular uptake and degradation. The second mechanism of natriuretic peptide inactivation is cleavage by neutral endopeptidase, which is present on endothelial cells, smooth muscle cells, cardiac myocytes, renal epithelium, and fibroblasts.17 BNP has a lower affinity for the clearance receptor NPR-C than ANP, which explains its longer half-life.11 Because BNP fluctuates less than ANP in response to hemodynamic changes, it is a better predictor of CHF and prognosis. BNP correlates more closely with left ventricular ejection fraction (LVEF) than ANP.18,19 BNP assays are commercially available and approved by the U.S. Food and Drug Administration for clinical use.
Diagnostic Implications of BNP
The most intriguing aspect of BNP for emergency medicine providers is its potential role in enhancing diagnostic accuracy for HF. Cited specificities (74%-92%)20,21 and sensitivities (77%-98%)2,20 vary considerably. Factors that affect sensitivity and specificity of BNP for the diagnosis of HF include patients’ age, pre-test probability for HF, severity of HF, cut-off value used, and type of ventricular dysfunction. For example, researchers assessed 1252 non-selected patients ages 25-74 and found BNP levels to have a sensitivity of 77% and a specificity of 87% for identifying left ventricular systolic dysfunction (LVSD). However, in those older than 55 years of age, the sensitivity was increased to 92%.22 One study reflected the inverse relationship between sensitivity and specificity based upon different cut-off values for "abnormal" BNP. When the researchers chose 46 pg/mL to be the cut off, the sensitivity was 93% and the specificity 80%. But when they chose 75 pg/mL as the cut-off, the sensitivity dropped to 86% while the specificity rose to 98%.23
The predictive values of BNP for HF can be high. Very high BNP levels (greater than 600-800 pg/mL), for example, have a high positive predictive value (PPV) for CHF. Unfortunately, mildly elevated BNP levels have low PPVs, as low as 50%.2 However, the greatest power for BNP may lie in its very high negative predictive value (NPV). Very low BNP levels have an NPV, which can be as high as 98%.2,21,23 One researcher found a 93% NPV with a BNP cut-off of 38.5 pg/mL while another found a 98% NPV with a cut-off of 80 pg/mL.20,21 This means a normal test virtually excludes significant cardiac function impairment.
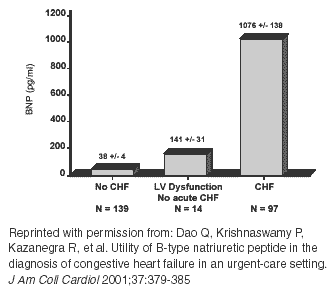
One of the first prospective evaluations of BNP was conducted by researchers who examined different diagnostic modalities in the assessment of 250 patients who presented to a Veterans Administration (VA) urgent care area with shortness of breath.20 Data included many aspects of history, signs, chest radiographs, and ECGs. Final diagnoses were established by two blinded cardiologists who had access to patient evaluations but not to BNP levels. The mean BNP level in the patients diagnosed with HF was 1076 ± 138 pg/mL while the non-CHF group mean level was 38 ± 4 pg/mL. BNP levels correlated with the clinical severity of CHF and the best clinical predictor was a past history of CHF (81% accuracy). However, elevated BNP levels had the best accuracy: 93-95%, with the cut-off of 80 pg/mL being most optimal. On multivariate analysis, the addition of the BNP level substantially enhanced the power of the model, suggesting that BNP levels added meaningful diagnostic information. However, it must be noted that the study was conducted in a cohort of predominantly male VA patients and it did not assess how BNP levels actually changed the clinical practice of the providers.
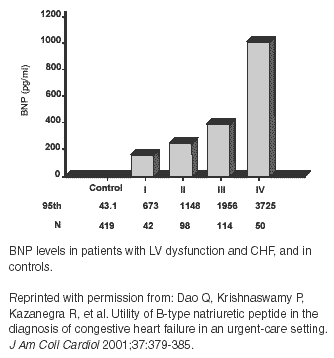
Emergency medicine providers should be familiar with the diagnostic information from two recent trials. The Dao study was confirmed with the most recent and largest study to date, the BNP (Breathing Not Properly) trial, which was presented at the American College of Cardiology’s 2002 meeting, and only preliminary data are available.21 It was a prospective, blinded evaluation of the use of BNP levels in 1586 patients with dyspnea presenting to the ED. HF was diagnosed in 744 patients. The gold standard of HF diagnosis was two cardiologists blinded to the BNP results. A BNP level of less than 100 pg/mL had a high NPV for HF. The sensitivity of a BNP level greater than 100 pg/mL was 90% and specificity was 74%. The BNP results provided an accurate diagnosis 81.1% of the time, while clinical judgment provided an accurate diagnosis 74% of the time. Clinical indecision was reduced from 43% to 11% with the use of BNP. BNP correctly provided the diagnosis of HF in 83% of cases compared to 67%-73% when using subjective scales such as the Framingham criteria.21 BNP levels also correlated with New York Heart Association (NYHA) functional classes. The mean BNP level was 150 pg/mL for class I, 250 pg/mL for class II, 550 pg/mL for class III, and 900 pg/mL for class IV.21 This correlation with NYHA class has been found in other trials. (See Figure 1.)24,25
|
Comparisons of BNP with other markers for the diagnosis of CHF have been done. (See Table 2.) Kelly and Struthers summarized some of this information.15 They found BNP to be superior to signs and symptoms for the diagnosis of CHF.15 In the setting of post-myocardial infarction (MI) patients, BNP levels were superior to all clinical indices of left ventricular systolic dysfunction, including signs and symptoms and modified clinical scoring tests (Peel Index), with a sensitivity of 84% (vs 46-64%) in post-MI patients.15
BNP also may be useful in identifying patients at risk for developing HF or those with mild LV dysfunction but still without symptoms. BNP increases in patients receiving anthracycline chemotherapeutic agents before they develop clinical HF.26 Patients with known LV dysfunction but without acute symptoms have BNP levels 3-4 times higher than those with normal LV function.20 (See Figure 2.) Thus, mild elevations in BNP are less specific for diagnosing acutely decompensated CHF. Conversely, mild elevations may be used as a screening tool for diagnosing early, asymptomatic CHF.
|
A relationship also has been established between BNP and hemodynamics in HF patients at rest and with exercise.27 Sixteen patients with HF who underwent right heart catheterization had natriuretic peptide levels drawn and hemodynamic parameters assessed at rest and immediately after exercise. BNP correlated with LVEF, mean pulmonary arterial pressure (MPAP), pulmonary capillary wedge pressure (PCWP), and cardiac index (CI). When compared to normal controls, BNP was elevated at rest. During exercise, both BNP levels increased and correlated with MPAP and pulmonary artery wedge pressure after exercise. Levels inversely correlated with CI.27
BNP correlates with LV function in recent MI patients with signs and symptoms of CHF. One study assessed BNP in 200 recent MI patients with acute dyspnea. The mean BNP was 30 pg/mL in patients with normal LV function and 489 pg/mL in those with abnormal LV function.23 BNP has its highest sensitivity, specificity, and positive predictive value in post-MI patients who have developed HF.28
BNP levels are increased in patients with isolated diastolic dysfunction. Therefore, conditions that cause diastolic dysfunction, such as aortic stenosis and hypertrophic cardiomyopathy, are associated with an increased BNP levels.16,29,30 Patients with left ventricular hypertrophy (LVH) also may have elevated BNP levels even in the presence of normal systolic function.31-33 Increases in BNP levels in the setting of isolated diastolic dysfunction are not as great as in patients with systolic dysfunction. The highest levels are found in patients who have both systolic and diastolic dysfunction.23
Other conditions are associated with increased BNP levels. Restrictive cardiomyopathies, such as those due to amyloidosis, have caused elevation in BNP. Patients with right ventricular (RV) dysfunction, such as that due to cor pulmonale or massive pulmonary embolism, may result in modest elevations of BNP.34 In primary pulmonary hypertension, BNP increases in proportion to the degree of RV dysfunction.35 The elevations of BNP in these conditions are less than with decompensated HF. Thus, the specificity of mild BNP elevations is lower because cardiac conditions other than HF can cause these elevations.15
Troponin levels have correlated directly with BNP levels in patients with LV wall strain. This suggests that cardiac myolysis in HF may be related to increased wall strain independent of coronary artery disease.36 Patients with decompensated HF have elevated troponin-I levels that correlate with increased BNP levels.36 In one study, 71 patients with nonischemic NYHA class II-IV CHF with normal coronary angiograms were compared to a control group of nine healthy patients and 15 patients with noncardiac dyspnea. Troponin levels were increased in 19 of the non-ischemic HF patients who were determined to have severe HF (either class IV or severe pulmonary edema) while they were normal in the controls.36
BNP compares favorable to other readily available ED tests. Radiographic cardiomegaly is only 51% sensitive and 79% specific for the diagnosis of HF.15,33 When comparing BNP measurements with radiographic cardiomegaly, Cowie and others found that the area under the receiver operating curve for BNP was 0.96 while for radiographic cardiomegaly it was 0.76. This suggests that BNP resulted in a greater combined sensitivity and specificity than did cardiothoracic ratios. In a logistic regression analysis, including both cardiothoracic ratio and BNP, only the BNP level was independently predictive of the presence of heart failure.15
Prognostic Implications of BNP
BNP provides important prognostic value in the treatment of HF. BNP is one of the best independent predictors of patient outcome identifying high mortality, morbidity, and successful treatment.37-39 Higher BNP levels are associated with increased mortality independent of age, NYHA class, prior MI, or LVEF.28,40,41 Patients who died within 12 months of a hospital admission for HF had significantly higher BNP levels than survivors: 325.6 pg/mL ± 37.4 vs. 169.9 pg/mL ± 14.7.34 The patients with elevated BNPs had a one-year mortality rate of 42% compared to 11.6% for those with a lower BNP.38 All patients who died within one month had a BNP greater than 165 pg/mL.38
BNP is the strongest predictor of death.25 In a small retrospective study of admitted patients with HF, those who die or are readmitted have an increasing BNP level during admission stay vs. patients in whom treatment resulted in a decreasing BNP level.39 Patients with a discharge BNP of less than 1220 pg/mL after treatment are less likely to be readmitted within 30 days.39 A BNP level of less than 430 pg/mL has a strong negative predictive value for readmission.39 In a trial of 112 patients with chronic severe HF (NYHA class III or IV) who were hospitalized with exacerbations, measurements of plasma levels of interleukin-6, cytokines, and BNP were obtained.40 Plasma levels of these proteins and LVEF before and at three months after additional treatment for HF were obtained. High levels of BNP and interleukin-6 are significant independent predictors of morbidity and mortality after optimal treatment. The sensitivity and specificity for mortality and morbidity of patients with a plasma level of BNP greater than 240 pg/mL were 73.1% and 89.1%, respectively. Thus, an elevated BNP level three months after optimized treatment is an independent prognostic indicator in patients with HF despite improvements of LVEF and symptoms.40
Increased BNP levels are associated with increased mortality over 24 months, independent of clinical and hemodynamic variables, including pulmonary artery pressure and cardiac output.42 Elevated BNP in patients with cor pulmonale indicate an increased risk of mortality.43 In a study involving 541 patients older than 85 years in Sweden, BNP levels predicted a five-year mortality among patients with or without known cardiovascular disease.44 In the Glasgow MONICA survey involving 1242 patients, mortality correlated with both decreased LVEF and increased BNP.45 Those patients with a BNP greater than 17.9 pg/mL had a mortality rate three times higher at four years.45
BNP is a prognostic indicator in patients with acute myocardial infarction (AMI).46-49 Levels reflect changes in gene expression and rise much more than ANP levels in early AMI.48,50,51 BNP rises rapidly over the first 24 hours after an AMI and then levels off. A large infarct may result in a second peak of BNP level five days after the AMI.2,49,50 Elevated BNP levels on days 1 and 7 after AMI reflect risk for adverse left ventricular remodeling,35 left ventricular dysfunction,2,48,51 clinical CHF,47 and death.46-50 The prognostic value of BNP following AMI is greater than that of LVEF and is independent of age or prior history of HF.47,49
BNP also has prognostic value in non-MI acute coronary syndromes (ACS). One study found that BNP was higher in patients with unstable angina than in those with stable angina or in healthy control subjects.2,52 In another study of patients with ACS, N-proBNP levels were higher in those who died than in survivors. Higher baseline levels of BNP were associated with older age; female sex; or a history of hypertension, CHF, vascular disease, or renal insufficiency in 2525 patients with ACS enrolled in the OPUS-TIMI 16 study. Patients with higher BNPs are more likely to present in a Killip Class of HF greater than 1, have ST changes on their baseline ECGs, or have elevated cardiac markers. BNP also correlates with the extent of angiographically determined CAD.53 The rates of mortality and HF are increased in ACS patients who have elevated baseline BNP levels.53 BNP is predictive of mortality among patients with normal troponin levels at presentation with AMI regardless of the presence of HF. An increasing BNP is associated with a higher 10-month mortality even after adjusting for age, renal function, HF, ST deviation, or troponin I.53 A BNP level may be an index of the size or severity of the ischemic insult. Even transient ischemia increases wall stress and induces BNP synthesis and release in proportion to the degree of injury. One study also showed BNP to be a predictor of mortality in 204 patients with ST-elevation MI, 220 patients with non-ST segment elevation MI, and 185 patients with unstable angina in the subacute phase. The median BNP levels were 442 pmol/L, significantly lower in long-term survivors, vs. 1306 pmol/L in those patients who died. Adjustment for peak troponin T levels did not markedly alter the relationship between BNP and mortality, and BNP levels remained a significant predictor of mortality after adjustment for age and ejection fraction (EF).54
Another study showed that BNP level was a predictor of future cardiac events. One study group showed that BNP levels of patients presenting with dyspnea to the ED were highly predictive of cardiac events over the next six months.55
Therapeutic Implications of BNP
Angiotensin-converting enzyme (ACE) inhibitors and beta-blockers are routine therapy in the treatment of HF because they reduce mortality. Because these agents antagonize the neurohormonal system, use of ACE inhibitors results in early, dose-dependent, sustained reductions in natriuretic peptide levels.19,56-58 Following MI, ACE inhibitors may prevent the second peak in BNP levels that occurs with ventricular remodeling.59 Addition of spirinolactone to standard HF regimens further reduces BNP levels. The reduction in BNP observed with spironolactone correlates with favorable changes in left ventricular remodeling.60
The relationship between beta-blockers and the natriuretic peptides is more complex. There appears to be a paradoxical relationship between the beta-blockers and natriuretic peptides. Although the natriuretic peptides inhibit sympathetic activity, beta-blockers increase BNP levels.61 BNP levels may reflect their efficacy. In one trial involving 415 patients with CHF who received carvedilol, mortality was significantly reduced in those with a BNP level above 82 pg/mL, but not in those with a BNP lower than 82 pg/mL.28 However, in patients with idiopathic dilated cardiomyopathy, the addition of carvedilol to standard therapy with ACE inhibitors reduced BNP levels over six months.62 The reduction of BNP levels mirrors improvements in LV dimensions and function.62
Among patients with HF, the reduction in BNP reflects improvement in hemodynamics and LV function, which overcomes the intrinsic BNP-raising effects of beta-blockers.62
BNP can be used in the titration of ACE inhibitors and other medications in the treatment of HF with a goal of BNP less than 200.2 BNP-guided CHF treatment with ACE inhibitors, diuretics, and digoxin reduces cardiovascular events compared with clinically guided treatment.62 BNP significantly decreases after treatment as LVEF improves in HF patients.62
BNP may be useful to monitor the efficacy of therapy because it is a noninvasive marker that correlates closely with filling pressures in patients with symptomatic HF. One study involving 69 patients with HF compared therapy guided by symptoms vs. therapy guided by N-proBNP levels. The N-proBNP group received higher doses of ACE inhibitors and diuretics than the patients treated based on symptoms. During 10 months of follow-up, fewer CHF exacerbations occurred in the N-proBNP group than in the symptom-guided group.63 In another study involving 20 patients, the group who had ACE inhibitor doses titrated to BNP levels had greater inhibition of the renin-angiotensin-aldosterone system and reduction of heart rate than the group managed with empiric therapy.58
In patients with ACS, therapy decisions based on BNP levels have not been well defined. One study, which compared invasive vs. noninvasive strategies for patients with non-ST elevation acute coronary syndromes, showed that patients with a BNP greater than 80 pg/mL were at increased risk for mortality independent of other variables. However, an early invasive strategy did not reduce mortality in the group with BNP elevation and there was no relation between BNP elevation and the observed benefit of invasive therapy.53 This highlights the limitation of BNP testing in patients with ACS. While a high-risk group may be identified, it is not yet clear how this affects management.
Prevention of Venous Thromboembolism (VTE) in HF: Possible Role for BNP
Using BNP levels to guide acute and, in some cases, long-term pharmacotherapeutic intervention with such agents as nesiritide, diuretics, ACEIs, beta-blockers, angiotension receptor blockers (ARBs), aldosterone antagonists, and other agents in patients with suspected HF is only one of the potentially important clinical roles for this marker. Recent investigations and epidemiological data suggest this marker may have additional utility in guiding patient management. In this regard, one of the other potential roles for BNP is its possible usefulness in identifying high-risk HF patients who require prophylaxis against venous thromboembolism (VTE).
Studies confirm that patients with HF have a hypercoagulable state and are at increased risk for acquiring VTE during acute hospitalization.64 Moreover, the potential complications, costs, and morbidity associated with VTE in the HF patient population can be significant, and may include pulmonary embolism (PE), prolonged hospitalization, post-phlebitic syndrome, and in some cases, sudden death. Accordingly, the threshold for routine, risk stratification-driven prophylaxis of VTE in this patient population must be balanced against the relatively low risk of serious complications associated with anticoagulation-facilitated prophylaxis.
In a recent study (The PRINCE TRIAL), patients were stratified and enrolled according to their underlying disease: severe respiratory disease or HF, two conditions that sometimes are difficult to distinguish from each other. The primary efficacy parameter was a thromboembolic event up to one day after the treatment period. Of the 665 patients enrolled, 451 patients were able to be evaluated in the primary efficacy analysis. Patients who were enrolled were 18 years of age or older and hospitalized for severe respiratory disease or HF. To be eligible for enrollment, the severity of HF had to be class III or IV according to the NYHA classification.64
In this controlled, randomized, open study, patients received either enoxaparin (40 mg once daily) or unfractionated heparin (UFH) (5000 IU three times daily) for 10 ± 2 days in 64 medical departments in Germany. Patients were stratified and enrolled according to their underlying disease: severe respiratory disease or HF.
The overall incidence of thromboembolic events was higher in patients with HF (12.6%) than in patients with respiratory disease (6.8%) In addition, patients with HF were more likely than patients with respiratory disease to be older than 70 years of age, (70% vs 40%, respectively) and had a higher prevalence of chronic venous disease (47% vs 33%). Of special note is that among patients with HF, 9.7% of patients on enoxaparin and 16.1% of patients on UFH developed thromboembolic events (i.e., DVT, PE, or DVT and PE). More than twice the number of patients (44/333; or 13.2%) receiving UFH discontinued the study because of adverse events or serious adverse events, including death, as compared to those on enoxaparin (18/332).
The PRINCE study is the first prospective, head-to-head investigation demonstrating that: a) enoxaparin is at least as effective as UFH (UFH, 5000 IU 3 times daily) for the prevention of venous thromboembolic disease (VTED) in patients with HF or respiratory disease; and b) that, in the PRINCE patient population, enoxaparin is associated with fewer deaths, less bleeding, and significantly fewer adverse events (45.8% vs 53.8%, P= 044 [adverse event analysis]). The role of BNP as a VTE prophylaxis trigger for patients suspected of having HF requires further investigation, but because it has been shown to be a specific marker for the presence of HF, it is likely to be useful in identifying hospitalized patients who are appropriate candidates for VTE prophylaxis.
Conclusions: Use of BNP in the ED
There is clear evidence that BNP is a diagnostic marker in determining whether a patient’s symptoms may be caused by HF. However, it has not yet been clearly shown to provide diagnostic help when compared to the entire usual evaluation. It may be useful in the ED for initial treatment decisions. BNP may be used to rule out HF in a patient with dyspnea. However, it seems that clinical judgment by the physician ultimately will determine patient disposition. In high-risk populations, such as patients with a recent MI or those receiving cardiotoxic drugs, BNP may be a useful screening tool to detect LV dysfunction in the ED. In patients with dual diagnoses, such as CHF plus pneumonia or chronic obstructive pulmonary disease (COPD), BNP may not be particularly useful.
BNP is a useful prognostic tool in predicting risk of death and HF in a wide variety of patients. This includes patients with chronic CHF, recent MI, and patients with ACS without myocardial necrosis. It also may be a useful screening tool in the general population for risk-stratification. The predictive value of BNP is independent of clinical variables, the severity of HF, and LVEF. BNP also may be used in-house after initiation of therapy to help assess the response to treatment. The major limitation of BNP testing is that the management implications are not clear. While BNP levels can identify patients who are at high risk for morbidity and mortality, it is not clear how to treat these patients. However, it may be wise to admit these patients to initiate aggressive workup and treatment in the hospital.
References
1. Adams KF. New epidemiologic perspectives concerning mild-to-moderate heart failure. Am J Med 2001;110:6S-13S.
2. Talwar S, Downie PF, Ng LL, et al. Towards a blood test for heart failure: The potential use of circulating natriuretic peptides. Br J Clin Pharmacol 2000;50:15-20.
3. Levy DB, Larson MG, Vasan RS. The progression from hypertension to congestive heart failure. JAMA 1996;275:1557-1562.
4. Struthers AD. Heart failure: The diagnosis of heart failure. Heart 2000;84:334-338.
5. Davie AP, Francis CM, Caruana L, et al. Assessing diagnosis in heart failure: Which features are any use? Q J Med 1997;90: 335-339.
6. Kisch B. Electron microscopy of the atrium of the heart: I Guinea pig. Exp Med Surg 1956;14:99-112.
7. Kangawa K, Fukuda A, Minamino N, et al. Purification and complete amino acid sequence of beta-rat atrial natriuretic polypeptide (beta-rANP) of 5000 daltons. Biochem Biophys Res Commun 1984;119:933-940.
8. Sudoh T, Kangawa K, Minamino N, et al. A new natriuretic peptide in porcine brain. Nature 1988;332:78-81.
9. Hosada K, Nakao K, Mukoyama M, et al. Expression of brain natriuretic peptide gene in human heart. Hypertension 1991;17: 1152-1155.
10. Yasue H, Yoshimura M, Sumida H, et al. Localization and mechanism of secretion of B-type natriuretic peptide in comparison with with those of A-type natriuretic peptide in normal subjects and patients with heart failure. Circulation 1994;90:195-203.
11. Mukoyama M, Nakao K, Hosada K, et al. Brain natriuretic peptide as a novel cardiac hormone in humans. Evidence for an exquisite dual natriuretic peptide system, atrial natriuretic peptide and brain natriuretic peptide. J Clin Invest 1991;87:1402-1412.
12. Schrier RW, Abraham WT. Hormones and hemodynamics in heart failure. N Engl J Med 1999;341:577-585.
13. Hama N, Itoh H, Shirakami G, et al. Rapid ventricular induction of brain natriuretic peptide gene expression in experimental acute myocardial infarction. Circulation 2002;92:1558-1564.
14. Matsumoto T, Wada A, Tsutamoto T, et al. Vasorelaxing effects of atrial and brain natriuretc peptides on coronary circulation in heart failure. The American Physiological Society 1999;H1935-H1942.
15. Kelly R, Struthers AD. Are natriuretic peptides clinically useful as markers of heart failure? Ann Clin Biochem 2001;38:94-102.
16. Mizuno Y, Yoshimura M, Harada E, et al. Plasma levels of A- and B-type natriuretic peptides in patients with hypertrophic cardiomyopathy or idiopathic dilated cardiomyopathy. Am J Cardiol 2000; 86:1036-1040.
17. Sonnenberg JL, Sakane Y, Jeng AY, et al. Identification of protease 3.4.24.11 as a major artrial natriuretic factor degrading enzyme in the rat kidney. Peptides 1988;9:173-180.
18. Davidson NC, Naas AA, Hanson JK, et al. Comparison of atrial natriuretic peptide, B-type natriuretic peptide, and N-terminal proatrial natriuretic peptide as indicators of left ventricular systolic dysfunction. Am J Cardiol 1996;77:828-831.
19. Motwani JG, McAlpine H, Kennedy N, et al. Plasma brain natriuretic peptide as an indicator for angiotensin-converting-enzyme inhibition after myocardial infarction. Lancet 1993;341:1109-1113.
20. Dao Q, Krishnaswamy P, Kazanegra R, et al. Utility of B-type natriuretic peptide in the diagnosis of congestive heart failure in an urgent-care setting. J Am Coll Cardiol 2001;37:379-385.
21. Maisel A, Krishnaswamy P, Nowak R, et al. Rapid measurement of B-type natriuretic peptide in the emergency diagnosis of heart failure. N Engl J Med 2002;347:161-167.
22. McDonagh TA, Robb SD, Murdoch DR, et al. Biochemical detection of left-ventricular systolic dysfunction. Lancet 1998;351:9-13.
23. Maisel A, Koon J, Krishnaswamy P, et al. Utility of B-natriuretic peptide as a rapid, point-of-care test for screening patients undergoing echocardiography to determine left ventricular dysfunction. Am Heart J 2001;141:367-374.
24. Yoshimura M, Yasue H, Okumura K, et al. Different secretion patterns of atrial natriuretc peptide and brain natriuretic peptide in patients with congestive heart failure. Circulation 1993;87:464-469.
25. Lerman A, Gibbons RJ, Rodeheffer RJ, et al. Circulating N-terminal atrial natriuretic peptide as a marker for symptomless left-ventricular dysfunction. Lancet 1993;341:1105-1109.
26. Okumura H, Iuchi K, Yoshida T, et al. Brain-natriuretic peptide is a predictor of anthrancycline-induced cardiotoxicity. Acta Haemotol 2000;104:158-63.
27. Friedl W, Mair J, Thomas S, et al. Relationship between natriuretic peptides and hemodynamics in patients with heart failure at rest and after ergometric exercise. Clin Chim Acta 1999;281:121-126.
28. Richards AM, Nicholls MG, Yandle TG, et al. Neuroendocrine prediction of left ventricular function and heart failure after acute myocardial infarction. Heart 1999;81:114-120.
29. Qi W, Mathisen P, Kjekshus J, et al. Natriuretic peptides in patients with aortic stenosis. Am Heart J 2001;142:725-732.
30. Yamazaki H, Senju Y, Kinoshita N, et al. Plasma brain natriuretic peptide in athletes. Am J Cardiol 2000;85:1393-1394.
31. Luchner A, Burnett JC, Jougasaki M, et al. Evaluation of brain natriuretic peptide as marker of left ventricular dysfunction and hypertrophy in the population. J Hypertension 2000;18:1121-1128.
32. Schirmer H, Omland T. Circulating N-terminal pro-atrial natriuretic peptide is an idependent predictor of left ventricular hypertrophy in general population. Eur Heart J 1999;20:755-763.
33. Yamamoto K, Burnett JC Jr, Jougasaki M, et al. Superiority of brain natriuretic peptide as a hormonal marker of ventricular systolic and diastolic dysfunction and ventricular hypertrophy. Hypertension 1996;28:988-994.
34. Takemura G, Takatsu Y, Doyama K, et al. Expression of atrial and brain natriuretic peptides and there genes in hearts of patients with cardiac amyloidosis. J Am Coll Cardiol 1998;31:254-265.
35. Nagaya N, Nishikimi T, Goto Y, et al. Clinical investigations: Acute ischemic heart disease. Am Heart J 1998;135:21-28.
36. Loegart D, Beyne P, Cusson C, et al. Evidence of cardiac myolysis in severe nonischemic heart failure and the potential role of increased wall strain. Am Heart J 2001;141:247-253.
37. Selvais PL, Donckier JE, Robert A, et al. Cardiac natriuretic peptides for diagnosis and risk stratification in heart failure: Influences of left ventricular dysfunction and coronary artery disease on cardiac hormonal activation. Eur J Clin Invest 1998;28:636-642.
38. Yu CM, Sanderson JE. Plasma brain natriuretic peptide-an independent predictor of cardiovascular mortality in acute heart failure. Eur J Heart Fail 1999;59-65.
39. Cheng V, Kazanegra R, Garcia A, et al. A rapid bedside test for B-type peptide predicts treatment outcomes in patients admitted for decompensated heart failure: A pilot study. J Am Coll Cardiol 2001;37:386-391.
40. Maeda K, Tsutamoto T, Wada A, et al. High levels of plasma brain natriuretic peptide and interleukin-6 after optimizrd treatment for heart failure are independent risk factors for morbidity and mortality in patients with congestive heart failure. J Am Coll Cardiol 2000;36: 1587-1593.
41. Stanek B, Frey B, Hulsmann M, et al. Prognostic evaluation of neurohumoral plasma levels before and during beta-blocker therapy in advanced left ventricular dysfunction. J Am Coll Cardiol 2001;38: 436-442.
42. Nagaya N, Nishikimi T, Okano Y, et al. Plasma brain natriuretic peptide levels increase in proportion to the extent of right ventricular dysfunction in pulmonary hypertension. J Am Coll Cardiol 1998;31:202-208.
43. Ishii J, Nomura M, Ito M, et al. Plasma concentration of brain natriuretic peptide as a biochemical marker for the evaluation of right ventricular overload and mortality in chronic respiratory disease. Clin Chim Acta 2000;301:19-30.
44. Wallen T, Landahl S, Hedner T, et al. Brain natriuretic peptide predicts mortality in the elderly. Heart 1997;77:264-267.
45. McDonagh TA, Cunningham AD, Morrison CE, et al. Left ventricular dysfunction, natriuretic peptide, and mortality in an urban population. Heart 2001;86:21-26.
46. Omland T, Aakvaag A, Vik-Mo H. Plasma cardiac natriuretic peptide determination as a screening test for the detection of patints with mild left ventricular impairment. Heart 1996;76:232.
47. Darbar D, Davidson NC, Gillespie N, et al. Dianostic value of B-type natriuretic peptide concentrations in patients with acute myovardial infarction. Am J Cardiol 1996;78:284-287.
48. Richards AM, Nicholls MG, Yandle TG, et al. Plasma N-terminal pro-brain natriuretic peptide and adrenomedullin. Circulation 1998; 97:1921-1929.
49. Arakawa N, Nakamura M, Aoki H, et al. Plasma brain natriuretc peptide concentrations predict survival after acute myocardial infarction. J Am Coll Cardiol 1996;27:1656-1661.
50. Morita E, Yasue H, Yoshimura M, et al. Increased plasma levels of brain natriuretic peptide in patients with acute mycardial infarction. Circulation 1993;88:82-91.
51. Horio T, Shimada K, Kohno M, et al. Serial changes in atrial and brain natriuretic peptides in patients with acute mycardial infarction treated with early coronary angioplasty. Am Heart J 1993;126: 293-299.
52. Kikuta K, Yasue H, Yoshimura M, et al. Acute coronary disease. Am Heart J 1996;132:101-107.
53. De Lemos JA, Morrow DA, Bentley JH, et al. The prognostic value of B-type natriuretic peptide in patients with acute coronary syndromes. N Engl J Med 2001;345:1014-1021.
54. Omland T, de Lemos JA, Morrow DA, et al. Prognostic value of N-terminal pro-atrial and pro-brain natriuretic peptide in patients with acute coronary syndromes. Am J Cardiol 2002:89;463-465.
55. Harrison A, Morrison LK, Krishnaswamy P, et al. B-natriuretic peptide predicts future cardiac events in patients presenting to the emergency department with dyspnea. Ann Emerg Med 2002;39:131-138.
56. Kohno M, Horio T, Yokokawa K, et al. Brain natriuretic peptide as a marker for hypertensive left ventricular hypertrophy: Changes during 1-year antihypertensive therapy with angiotensin-converting enzyme inhibitor. Am J Med 1995;98:257-265.
57. van Veldhuisen DJ, Genth-Zotz S, Brouwer J, et al. High- versus low-dose ACE inhibition in chronic heart failure: A double-blind, placebo-controlled study of imidapril. J Am Coll Cardiol 2002;39: 1623-1629.
58. Murdoch DR, McDonagh TA, Byrne J, et al. Titration of vasodilator therapy in chronic heart failure according to plasma brain natriuretic peptide concentration: Randomized comparison of the hemodynamic and neuroendocrine effects of tailored versus empirical therapy. Am Heart J 1999;138:1126-1132.
59. Mizuno Y, Yasue H, Oshima S, et al. Effects of angiotensin-converting-enzyme inhibitor on plasma B-type natriuretic peptide levels in patients with acute myocardial infarction. J Card Fail 1997;3: 287-293.
60. Tsutamoto T, Wada A, Maeda K, et al. Effect of spironolactone on plasma brain natriuretic peptide and left ventricular remodeling in patients with congestive heart failure. J Am Coll Cardiol 2001;37: 1228-1233.
61. Luchner A, Burnett JC, Jougasaki M, et al. Augmentation of the cardiac natriuretic peptides by beta-receptor antagonism: Evidence from a population-based study. J Am Coll Cardiol 1998; 32: 1839-1844.
62. Kawai K, Hata K, Takaoka H, et al. Plasma brain natriuretc peptide as a novel therapeutic indicator in idiopathic dilated acrdiomyopathy during beta-blocker therapy: A potential of hormone-guided treatment. Am Heart J 2001;141:925-932.
63. Troughton RW, Frampton C, Yandle TG, et al. Treatment of heart failure guided by plasma aminoterminal brain natriuretic peptide (N-BNP) concentrations. Lancet 2000;355:1126-1130.
64. Kleber FX, Witt C, Vogel G, et al, for THE-PRINCE Study Group Berlin, Erfurt, and Cologne, Germany. Randomized comparison of enoxaparin with unfractionated heparin for the prevention of venous thromboembolism in medical patients with heart failure or severe respiratory disease. Am Heart J 2003;145:614-621.
As experienced physicians understand, heart failure can be a difficult diagnosis to establish in the emergency department setting, especially when there are factors that may complicate a patients presentation. This issue of Emergency Medicine Reports reviews the role and clinical utility of brain natriuretic peptide in the management of patients suspected of having heart failure upon presentation to the ED
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.