New Targets For Staphylococcal Antibiotics
New Targets For Staphylococcal Antibiotics
Abstract & Commentary
Synopsis: Drug discovery activities have yielded agents effective against a novel target in staphylococci.
Source: Choudhry AE, at al. Inhibitors of pantothenate kinase: Novel antibiotics for staphylococcal infections. Antimicrob Agents Chemother. 2003;47:2051-2055.
The search for new targets for antibacterials goes on. Now from the microbial group at GlaxoSmithKline and the Department of Chemistry and Chemical Biology at Cornell University in Ithaca, NY, comes an exciting target. Ironically, it is an enzyme that most of us learned in college chemistry during memorization of the Krebs Cycle. Yes, good old coenzyme A (CoA) is as crucial for bacterial metabolism as for eukaryotes and better yet that several studies suggested that there was little sequence homology between prokaryotic and eukaryotic CoA biosynthetic enzymes.
Choudhry and associates wanted to find a step in CoA biosynthesis that was targetable in bacteria. One of the first enzymes involved in CoA biosynthesis in Escherichia coli, CoaA, is not present in the published sequences for the Staphylococcus aureus strains Mu50 and N15. So the study group cloned and purified CoaA from S aureus and characterized the enzyme. The phylogenetic tree analysis showed CoaA from S aureus was very similar to that in S haemolyticus and S epidermidis and was related to CoaA of Bacillus anthracis and B cereus but distantly related to fruit fly and mammalian CoaA. The enzyme was actually absent in most others like E coli. So this looked liked CoaA may be a good target for S aureus inhibition.
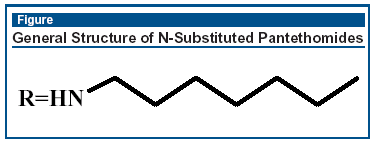
The next step was to find novel inhibitors of CoaA in S aureus. It turns out that N-substituted pantothenamides did the trick, some better than others (best MICs around 1-4 mg/mL). It is thought that the mechanism of inhibition is due to a CoA derivative (ethyldethia-CoA) to form acyl-CoA esters.1 These inhibitory compounds have the general structure shown in the Figure.
And the least active is 1e, containing a short chain terminating in a benzyl group probably acting as a substrate rather than an inhibitor of CoA; it had no antibacterial activity. The most active compounds, 1c and 1d, showed little toxicity against human hepatocytes.
Comment by Joseph F. John, Jr., MD
It is very early in the development of these inhibitors of staphylococcal pantothenate kinase. Nevertheless, since there are so few alternatives presently available that any new nontoxic antistaphylococcal compounds have to be taken very seriously. As we have seen with the recent upsurge of work with lysostaphin, new compounds may be attractive simply if they act as topical antibacterials, since decolonization of nasal staphylococci is taking on new dimensions.
This article demonstrates how complex the early pathways are that bring new antibiotics to market. This current article demonstrates how molecular biology plus enzyme chemistry are needed to evaluate compounds. In the case of the pantothenate kinase inhibitors, there is good reason to think that we may see them in some kind of clinical trials involving staphylococcal infection or colonization.
Dr. John is Chief Medical Subspecialty Services, Ralph H. Johnson Veterans Administration Medical Center; Professor of Medicine, Medical University of South Carolina, Charleston, SC.
Reference
1. Strauss E, Begley TP. The antibiotic activity of N-pentylpantothenamide results from its conversion to ethyldethia-coenzyme a, a coenzyme a antimetabolite. J Biol Chem. 2002;277:48205-48209.
Drug discovery activities have yielded agents effective against a novel target in staphylococci.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
