Sudden Acute Respiratory Syndrome (SARS)
Author: Michele Barry, MD, FACP, Professor of Medicine and Global Health, Yale University School of Medicine, New Haven, Conn.
Editor’s Note—An outbreak of an atypical pneumonia in Guangdong Province, Peoples Republic of China beginning in November 2002 and its initial spread to Hong Kong and Vietnam caused the World Health Organization (WHO) to take the unprecedented step of issuing a worldwide alert for atypical pneumonias on March 12, 2003.1 Since that alert, the rapid worldwide spread of a novel coronavirus that causes severe acute respiratory syndrome (SARS) has led to recognition and reporting of more than 7500 cases from 29 countries with more than 550 deaths in a little more than 6 months (case-fatality 7.7%).2 This review will provide an overview of SARS for the primary care physician (PCP), including epidemiology, etiology, review of the clinical and laboratory features as well as diagnosis, therapy, and prevention of SARS. Continuous daily updates on the epidemiologic investigation of SARS can be obtained from the following web sites: www.cdc.gov/ncidod/sars and www.who.int.
Epidemiology and History of SARS Outbreak
On Feb. 11, the Chinese Ministry of Health notified WHO that 305 cases of an acute respiratory syndrome of unknown etiology had occurred in 6 municipalities in Guangdong Province during the period Nov. 16, 2002, to Feb. 9, 2003.1 Guangdong Province is an agricultural area in southern China with a population of 75 million, thousands of farms with large and small animals, a subtropical climate, and a rainfall of 2 meters per year.3 The earliest cases in China included an unusually high proportion of food handlers, caterers, and chefs—constituting about 5% of the first patients compared with less than 1% of patients with other forms of pneumonia in Guangdong Province.4 In this part of the world, merchants commonly sit near stalls of exotic animals to be used as food—animals include the masked palm civet, a prime suspect for the reservoir of SARS.5 The disease rapidly spread outside of China and was characterized by surprisingly high transmission to health care workers and household contacts.6-9 Case clusters in health care settings, apartment complexes, and hotels have played an important role in the epidemic with transmission patterns suggesting droplet or close contact transmission.10,11
The term SARS was first used for a patient in Hanoi who became ill on Feb. 26, 2003, and evacuated back to Hong Kong. Dr. Carlo Ubani, the physician who raised the alarm in Hanoi, subsequently contracted SARS and died.12 The first case identified in Hanoi had stayed on the ninth floor of a hotel in Hong Kong at the same time as a 64-year-old doctor who had been treating atypical pneumonia cases in southern China. This physician was the first known case of SARS outside of China and appears to be the source of infection for most if not all cases in Hong Kong, Canada, Vietnam, Singapore, United States, Ireland, and subsequently Thailand and Germany.13 The mode of hotel dissemination by this index case has never been clearly identified although environmental persistence of the agent was postulated. Important factors in extensive dissemination at Prince of Wales Hospital (PWH) in Hong Kong by this index case was believed to be the use of a nebulizer bronchodilator, which increased droplet load surrounding the patient as well as overcrowding in the hospital ward and an outdated ventilation system.13
The second major epicenter cluster involving hundreds of cases occurred in an apartment complex in Hong Kong. The source has been attributed to a "super spreader" patient with renal failure on outpatient hemodialysis at PWH who had diarrhea.13 Infection has been speculated to have spread to other residents in this cluster by a leaking sewage drain in the apartment complex allowing an aerosol of virus-containing material to disseminate. Sewage also back flowed into bathroom floor drains of multiple apartments, likely enhancing epidemic transmission.13
The concept of "super spreaders," the cause of disease clusters in other infectious diseases, has been invoked for SARS.14 Super spreaders are index cases who have infected greater than 10 family members, HCWs, or visitors to a health care facility where the patient is hospitalized.15 It has been speculated that super spreaders may have been misdiagnosed and thus became prolonged hidden reservoirs of the virus. In reports, they have often been subjected to aerosolizing procedures such as bronchodilator nebulizers, BiPAP, endotracheal intubation, or bronchoscopy.14,15 They may have had a more vigorous cough reflex during high viral load.14,15 In addition, these individuals are postulated to have high viral loads perhaps because of defects in their immune system as could be the case in the cluster started by 2 documented patients with end-stage renal failure.13 Procedures such as nasopharyngeal aspiration, bronchoscopy, endotracheal intubations, airway suction, and cardiopulmonary resuscitation have caused high risk to health care workers (HCWs) who have composed an extraordinary percentage of the nosocomial cases.15 Cleaning the patient and bedding after fecal incontinence also appears to be a high-risk procedure.13 An astronomical 62% of 138 cases of secondary and tertiary spread in a Hong Kong cluster were HCWs, 51% of 144 secondary or tertiary cases in Toronto were HCWs.15
Seto and colleagues conducted a case-control study in 5 Hong Kong hospitals with 241 noninfected and 13 infected staff with documented exposures to 11 SARS patients during patient care.16 All participants were surveyed about use of mask, gloves, gowns, and hand washing. The 69 staff who reported use of all 4 measures were not infected whereas all infected staff had omitted at least 1 measure. Fewer staff who wore masks (P = 0.0001), gowns (P = 0.006), and washed their hands (P = 0.047) became infected compared with those who did not. Seto et al concluded that practice of droplet precaution and contact precaution is adequate to prevent nosocomial infection.16 Disturbingly, a recent review of 11 SARS-infected HCWs exposed to 1 index case who was intubated in Toronto revealed that all of these 11 HCWs had worn the recommended gown, gloves, PCM 2000 duckbill masks, and goggles.15 However, masks had not been fit tested nor were all NIOSH-approved N95 masks—although PCM 2000 duckbill masks were felt to be equivalent to N-95 masks by the Canadian Public Health Service.15 Still, the highly protective role of masks as compared to any of the other 3 protective measures in the Seto study suggests that in hospitals, infection is transmitted by droplets and not airborne as large droplets are generated at face level making the mask crucial for protection.16 Notably paper masks were not effective as N95 masks (as expected because of easy paper penetration by wet droplet transmission). Although large droplet transmission seems to be important in the spread of SARS, unusually rapid transmission suggest that airborne transmission can occasionally occur via smaller droplet nuclei (< 10 µm in diameter). Such droplet nuclei, in diseases like influenza, tuberculosis, and measles, allow organisms to directly reach the lungs of contacts.3
Case Definition
The classification of a case of SARS is based on clinical symptoms and epidemiologic linkage of travel to an area with suspected community transmission of SARS (see Table 1). CDC’s interim suspected SARS case definition (available at www.cdc.gov/ncidod/sars/casedefinition.htm) is based on clinical criteria and epidemiologic linkage to other patients with SARS.
Etiologic Agent
The isolation of a novel coronavirus from the respiratory secretions of patients with SARS was reported from both patients studied in Germany and overseas specimens sent to the Centers for Disease Prevention (CDC) in Atlanta.18,19 A team from Hong Kong concomitantly isolated this novel coronavirus as well in 45 of 50 patients with SARS, representing more than 5 separate epidemiologically linked transmission clusters.20 Cytopathological features were first noted microscopically in Vero E6 cells inoculated with a throat-swab specimen. Electron-microscopical examination revealed ultrastructural features characteristic of coronaviruses. Immunohistochemical and immunofluorescence staining revealed reactivity with group I coronavirus polyclonal antibodies. Reverse transcription-polymerase chain reaction (RT-PCR) were used to obtain a sequence that clearly identified the isolate as a unique coronavirus only distantly related to previously sequenced coronaviruses.18-20 Indirect fluorescent antibody tests and enzyme-linked immunoabsorbent assays made with the new isolate also demonstrated a virus-specific serologic response.18-19 The apparent lack of antibody in all serum specimens except those from patients with SARS suggests that this virus has not previously circulated in humans.19 High concentrations of coronavirus RNA is found in sputum and low concentrations in plasma and feces of patients.18 Inoculation of monkeys with SARS-associated coronavirus from cell cultures caused lower respiratory disease.22 Thus, tests done over the past months have shown that SARS-associated coronavirus fulfills Koch’s 6 postulates: isolation from a host, cultivation in host cells, filterability, production of a similar disease in a host or closely related species, re-isolation of the virus from infected test host, and detection of immune response to virus.22 Interestingly, other laboratories have found co-infection with human metapneumovirus in several cases of SARS yet antibody response to metapneumovirus has been absent in convalescent sera and Koch’s postulates have not been fulfilled; indicating coronavirus is the key player.23 The 3 serologically unrelated known groups of coronavirus are associated with a variety of diseases in humans and domestic animals. They are enveloped single-stranded RNA viruses that have a characteristic halo or crown-like (corona) appearance under electron microscopy. Most coronaviruses cause disease in only 1 host species. The known human coronaviruses (types 229E and OC43) are a common cause of mild-to-moderate upper respiratory tract infections and are responsible for one-third of common colds.24 They can occasionally cause pneumonia in older patients, immunocompromised patients, neonates, or military recruits.25 Several animal coronaviruses cause respiratory, gastrointestinal, neurologic, and hepatic disease in their respective hosts (eg, porcine transmissible gastrointestinal virus, murine hepatitis virus, avian infectious bronchitis virus).19,24 The human-associated SARS-coronavirus’ genome has been entirely sequenced and appears to be neither a mutant of any known coronavirus nor a recombinant of a known coronavirus indicating a novel coronavirus.23,25 Recent data have revealed an almost identical coronavirus in the saliva and feces of the masked palm civet, an animal considered a food delicacy in Guangdong Province thus offering a clue to the original host.5 Already genetic signature mutations of the SARS corona-virus have linked geographic and temporal clusters of patients suggesting immunological pressures have started to influence the evolution of the SARS virus in human populations.26
Clinical Features: Symptoms
Clinical features of SARS have been described in several retrospective cohorts in Canada, Vietnam, Hong Kong, and Singapore.10,11,20,27-30 Incubation period in these series ranged from a low of 2 days to a maximum of 16 days. Median incubation in all series was approximately 6 days. The one prospective study in Hong Kong involved a major outbreak involving 321 patients at a high-rise housing complex in Hong Kong.31 Epidemiological investigations revealed a point source outbreak involving a faulty sewage system initially contaminated by excreta of an index case. Investigators prospectively followed a cohort of 75 patients who eventually contracted SARS and they monitored viral load and symptoms. They noted a triphasic pattern to clinical disease. Stage 1 was characterized by fever, myalgia, and other systemic symptoms. Increasing viral load during this phase suggested the symptoms were due to replication and cytolysis. Defervescence occurred, but in week 2 patients had recurrence of fever indicating stage 2. Onset of diarrhea (in an unusually high frequency [73%] in this cohort compared to other reports) and O2 desaturation with shifting radiographic shadows were seen in stage 2. IgG seroconversion to coronavirus was seen on day 10 with severe clinical worsening of respiratory status correlating with falls in viral load. This pattern suggests that lung damage may be related to immunopathologic damage and not viral replication. The third phase was characterized by acute respiratory distress syndrome (ARDS), nosocomial sepsis, and multi-organ failure. Others have confirmed the clinical scenario of a break in the fever and worsening of respiratory symptoms, which may indicate a second phase of lung inflammation and damage due to the host’s immune response.26,32
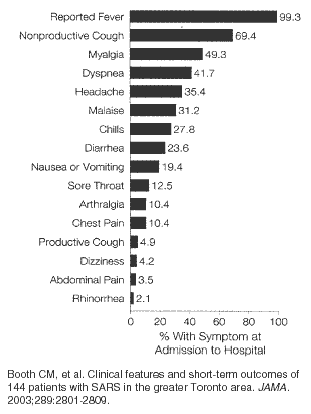
The Canadian series of 144 patients was retrospective and comprised 111 patients who contracted SARS in the hospital setting as a health care worker (51%) or as a visitor or fellow patient.27 The majority of patients reported fever often with rigors and chills as the earliest symptoms—often accompanied by nonproductive dry cough and myalgias (see Table 2). Less than 2% had rhinorrhea and only 12% sore throat confirming the lack of initial upper respiratory symptoms. Diarrhea was not a prominent early feature in this cohort occurring in only 25% of patients. After 3-7 days, a second lower respiratory phase began with worsening of dyspnea and hypoxemia.
|
A milder clinical presentation is seen in children. The lower number of children involved in the SARS epidemic suggests that children develop a milder form of the disease with a less aggressive clinical course than adults.33 Two distinct patterns were noted in 10 children studied in Hong Kong. Teenage patients presented with symptoms of fever, malaise, myalgias, chills and rigors similar to adults whereas younger children presented with fever, cough, and rhinorrhea without rigors or myalgias. The younger children had milder clinical courses with more rapid improvement, but all children did well. All ages were treated with ribavirin and steroids.33 Very few children have been described with disease in other clustered settings. Documentation of asymptomatic seroconversion of children as well as adults would be of interest in these epidemic settings.
Physical Examination of SARS Patients
All clinical descriptions of patients with SARS have noted normal physical exams outside of the respiratory system. Often crackles, bronchial breath sounds, and dullness on percussion were noted. As expected in atypical pneumonias, both respiratory symptoms and positive auscultatory findings could often be disproportionally mild compared with chest radiographs or chest CT findings. Similarly although less frequently, radiographs could be normal with prominent respiratory symptoms.27-32
Laboratory Features of SARS
Consistent laboratory features in SARS patients include lymphopenia, elevated lactate dehydrogenase (LDH), elevated hepatic transaminase levels (2-6 times upper limits of normal) and creatinine kinase (CPK) levels in the low thousand range.6-11,29,30 Thrombocytopenia, leukopenia, abnormal PTT, and elevated D-dimers mimicking hemorrhagic fever patients have been described.29,32 In the majority of patients, renal function has remained normal but electrolyte abnormalities such as mild hyponatremia, hypokalemia, hypocalcemia, hypomagnesemia and hypophosphatemia have been seen early in the disease before ribavirin administration or multiorgan failure, which can exacerbate these electrolyte abnormalities.27,30,6-9 Hypoxemia and poor outcome often correlated with high LDH values indicating diffuse lung damage.29 Hallmark laboratory features of SARS appear to be lymphopenia and elevated LDH.27-32
Radiographs
Typically, patients with SARS present with unilateral peripheral areas of consolidation progressing to patchy consolidation correlating to deterioration in respiratory function.10,11,28 The primary radiologic appearance of SARS is air-space shadowing determined on CT to be subpleural, focal consolidation with air bronchograms and ground glass opacities predominately affecting the lower lobes.10,11,29 Chest radiographs and CT scans offer important diagnostic clues. When plain radiographs appear normal in an early febrile period associated with lymphopenia, a CT of the thorax often reveals bilateral peripheral air-space consolidation mimicking that in bronchiolitis obliterans with organizing pneumonia, a finding that heightens suspicion of SARS as a diagnosis. Spontaneous pneumothoraces have been described in many patients unrelated to intubation or positive-pressure ventilation.27,31
Diagnosis of SARS (see Table 3 and Table 4)
Routine laboratory testing for a suspected case of SARS should include:34
- CBC with WBC differential, platelets, and clotting profile
- Biochemical testing (electrolytes, liver, renal function, calcium, phosphorus, magnesium, CPK, and LDH
- Arterial blood gas/O2 oximetry
- Microbiological tests to exclude other causative agents
- Bacterial cultures of blood, sputum, and urine
- Serologies by microagglutination for mycoplasma
- Legionella urinary antigen
- Serologies by direct immunofluorescence for chlamydia, influenza, parainfluenza,
- RSV, adenoviruses
- Nasopharyngeal aspirates for viral cell cultures, and direct sputum smear for pneumocystic carinii by silver stain if indicated
- A chest x-ray and/or CT scan should be obtained
- Specific SARS specimens (see Table 5 and Table 6)
Laboratory diagnostic tests used at CDC to test clinical specimens for evidence of this novel coronavirus are still in development and are not available outside a research setting as of this publication. Laboratory criteria and collection of specimens for culture and serologic testing for coronavirus are described in Tables 5 and 6. Serologic antibody testing consists of indirect fluorescent antibody testing and enzyme-linked immunosorbent assays that are specific for antibody produced after infection can be used. Although some patients have detectable IgM antibody within 14 days of illness onset, definitive interpretation of negative coronavirus antibody tests is possible only for specimens obtained > 21 days after onset of fever.35
A reverse transcriptase-polymerase chain reaction RT-PCR test specific for the RNA of the SARS coronavirus has been developed. This has been positive within the first 10 days after fever onset but RT-PCR tests on samples during convalescence may be negative.35
Lastly, cell culture of virus in specimens of respiratory secretions, blood, and feces from SARS patients can be inoculated into cell cultures such as VERO E6 cells and coronavirus identified by characteristic electron microscopy findings.35
Pathology
Postmortem examination of SARS victims have revealed diffuse alveolar damage especially if long-standing disease was present.19 The early phase has been characterized by pulmonary edema with hyaline membrane formation suggestive of ARDS. In patients with disease duration of less than 10 days, bronchial epithelial denudation, loss of cilia, and squamous metaplasia were early features. Diffuse alveolar damage was common in cases of longer duration.36 In contrast to typical alveolar damage in which neutrophils and fibroplasts are the main cellular agents, in patients with fatal SARS, macrophages are the prominent leucocyte in the alveoli. Hemophagocytosis, attributed to cytokine dysregulation has been seen.36 There has been scanty lymphocyte infiltration and only rare virus identified in lung tissue indicating that immune response, perhaps mediated by cytokines released by SARS-infected macrophages, mediates more lung damage than viral infection.19,36
Management of a Patient with SARS
Algorithms have been developed for management of definite contact with a SARS patient as well as for patients with no definite contact but potential social contact.37 (www.cdc.gov/ncidod/sars/clinicians.htm.) Triage and isolation of patients suspected of having SARS with the use of negative pressure rooms is recommended along with infection control measures that include the use of fitted N95 masks, gloves, disposable gowns, and eye protection (see Table 7). Careful attention to hand washing or disinfection after removal of gloves is necessary. Limiting and grouping of health care workers involved with SARS should be attempted. Visitors to suspected or known case should be limited or prevented. After the appropriate laboratory specimens are sent off, chest x-ray and CT scanning of the chest should be considered.
Therapy of SARS (see Table 8)
|
No antiviral has been demonstrated to be effective by randomized, controlled trials as yet.
Many protocols include early ribavirin therapy (oral formulation with only 50% bioavailability, 1200 mg every 8 hrs, intravenous form 8 mg/kg of body weight every 8 hours (available through CDC).3,34 No controlled trials of ribavirin have been described and preliminary reports from in vitro testing indicate that ribavirin concentrations that inhibit ribavirin-sensitive viruses do not inhibit replication or cell spread of the novel coronavirus.36 Although CDC does not officially recommend ribavirin therapy, the Hong Kong group caring for patients have expressed their opinion that early ribavirin administration makes a difference in outcome.20,34 Ribavirin is a ribonucleosidase analogue that induces lethal mutagenesis of RNA viral genomes and has broad-spectrum activity against RNA viruses including respiratory syncytial virus and other coronaviruses. Oseltamivir has been used for treatment of presumptive influenza in early cases before influenza is ruled out and diagnosis is confirmed.3 Most providers have used high-dose pulsed steroids to prevent the immunologic response felt to be the etiology of the diffuse alveolar damage that occurs with the drop of viremia and defervescence.3,20,34 One regimen suggested includes methylprednisolone 1 mg/kg every 8 hr (3 mg/kg/d) intravenously for 5 days with a tapering dose: 1 mg/kg q 12 hrs × 5 days, prednisolone 0.5 mg/kg twice daily orally × 5 days, prednisolone 0.5 mg/kg once daily orally for 5 days, then prednisolone 0.25 mg/kg once daily orally for 3 days.34 Before SARS confirmation patients have been treated with appropriate antibiotics for community-acquired atypical pneumonia.
SARS Quarantine and Travel
On April 4, 2003, the president of the United States signed an executive order adding SARS to the list of quarantinable communicable diseases. This act provides the CDC, through its Division of Global Migrations and Quarantine, the legal authority to implement isolation and quarantine measures as part of transmissible disease-control measures. Isolation refers to the practice of keeping a patient with a known communicable disease separate; quarantine refers to keeping a person or group of persons who have been exposed and potentially infected apart from others. Certain countries, such as Singapore, have invoked infectious disease acts to call for mandatory home quarantine of all contacts exposed to SARS.14 Electronic cameras are placed in the home of each contact quarantined and electronic tagging or forced detention has been permitted for violators of quarantine in Singapore. In Singapore, all inpatients who are discharged from a hospital with previous SARS cases are under telephone surveillance for 21 days; all probable or suspected SARS patients are discharged to home quarantine for 14 days. A dedicated private ambulance service is sanctioned to transport all suspects or probable cases to dedicated hospitals.
The CDC have issued their own guidelines to prevent spread of SARS or import into the United States. (www.cdc.gov/ncidod/sars/qa/travel.htm.) Quarantine inspectors are meeting all aircraft and ships reporting ill passengers or crew. Health alert notices are being distributed to passengers arriving from a SARS epidemic setting. In response to the SARS epidemic, CDC has issued 2 types of notices to travelers: advisories and alerts. A travel advisory recommends that nonessential travel be deferred; a travel alert does not advise against travel but informs travelers of a health concern. Most recently, CDC-amended travel advisories as of May 23, 2003, have suggested travel advisories (ie, elimination of all nonessential travel) to mainland China, Hong Kong, Singapore, and Taiwan. Recently, a travel alert was reissued for Toronto, Canada. Vietnam has been taken off the travel alert or advisory list.
Future Directions for Prevention and Therapy of SARS
The fact that SARS is a respiratory illness reduces the chance of its eradication. Finding SARS virus in nature has taken scientists to the most logical place in the environment, the food market in Guangdong for an animal reservoir. The masked palm civet appears most likely the origin of human-associated SARS.5 The civet is a cat-sized animal related to the mongoose and eaten in the autumn and winter in China to help people withstand the cold. Prevention of human contact with this reservoir will include a cultural change in eating habits as well as a banning of sales.5
Although there are no approved drugs with proven efficacy against SARS-coronavirus, future efforts potentially include protease inhibitors, which could prevent the processing of RNA polymerase or cleavage of the viral S glycoprotein.21 Inhibitors of coronavirus acetylesterase activity might limit viral replication as neuraminidase inhibitors do against influenza viruses. Inhibitors of membrane fusion might block viral entry as do several of the new antiretroviral HIV drugs. Immunoglobulin from recovered SARS patients might contain protective antibodies against the viral S glycoprotein or as yet an unidentified entry receptor for the SARS-coronavirus.21
Vaccines are effective for some animal coronavirus. Vaccination with live, attenuated virus is effective against porcine epidemic diarrhea virus and avian infectious bronchitis virus, both of which are caused by animal coronaviruses.21 However, using live, attenuated coronavirus vaccines in humans may risk recombination of genomes of vaccine strains with wild type coronavirus. Killed or subunit vaccines containing the spike glycoprotein may be effective although some vaccines against feline coronaviruses actually enhanced disease when vaccinated animals were exposed to wild type virus again raising the specter of immune response playing a major role in disease morbidity.21
Conclusion
Clearly, development of drugs and vaccines for SARS as well as interruption of contact between animal reservoirs and human hosts will all need to be addressed to provide a multipronged strategy for prevention and treatment of SARS. Global cooperation of laboratories and internet communication of surveillance have played a remarkable role in containing this epidemic and one hopes for such global cooperation in future prevention and development of drugs and vaccines directed against SARS coronavirus as well as in surveillance and eradication of this epidemic disease.
References
1. World Health Organization. WHO issues global alert about cases of severe respiratory illness may spread to hospital staff. Geneva: WHO Mar 12, 2003. Available at: www.who.int/mediacentre/releases/2003/pr22/en/. Accessed 5/31/03.
2. World Health Organization. Cumulative number of SARS. Available at: www.who.int/csr/sars/country/2003_05_14/en. Accessed 5/14/03.
3. Wenzel R, Edmond M. Managing SARS amidst uncertainty. N Engl J Med. 2003;348(20):1947-1948.
4. Altman L. Search for SARS’ past may yield glimpse of its future. NY Times, May 20, 2003:F4.
5. Bradsher K, Altman L. Strain of SARS is found in 3 animal species in Asia. NY Times, May 24, 2003:1.
6. MMWR Morb Mortal Wkly Rep. CDC Update: Severe acute respiratory syndrome—United States 2003. 2003;52:226-228.
7. MMWR Morb Mortal Wkly Rep. CDC Update: Severe acute respiratory syndrome—United States 2003. 2003;52:357-360.
8. MMWR Morb Mortal Wkly Rep. CDC Update: Severe acute respiratory syndrome—United States 2003. 2003;52:411-413.
9. MMWR Morb Mortal Wkly Rep. CDC Update: Severe acute respiratory syndrome—United States 2003. 2003;52:436-437.
10. Poutanen SM. Identification of severe acute respiratory syndrome in Canada. N Engl J Med. 2003;348:1995-2005.
11. Tsang KW, et al. A cluster of severe acute respiratory syndrome in Hong Kong. N Engl J Med. 2003;348(20):1977-1985.
12. Reilley B, et al. SARS and Carlo Urbani. N Engl J Med. 2003;348(20):1951-1952.
13. Tomlinson B, Cockram C. SARS: Experience at Prince of Wales Hospital, Hong Kong. Lancet. 2003;361:1486-1487.
14. MMWR Morb Mortal Wkly Rep. Severe acute respiratory syndrome—Singapore 2003. 2003;52:405-411.
15. MMWR Morb Mortal Wkly Rep. Cluster of severe acute respiratory syndrome among protected health-care workers—Toronto, Canada. 2003;52(19):433-436.
16. Seto W, et al. Effectiveness of precautions against droplets and contact in prevention of nosocomial transmission of severe acute respiratory syndrome (SARS). Lancet. 2003;361:1519-1520.
17. CDC. SARS website. Updated interim U.S. case definition for severe acute respiratory syndrome (SARS). Available at: www.cdc.gov/ncidod/sars/pdf/sars-casedefinition.pdf. Accessed 5/30/03.
18. Drosten C, et al. Identification of a novel coronavirus in patients with severe acute respiratory syndrome. N Engl J Med. 2003; 348(20):1967-1976.
19. Ksiazek T, et al. A novel coronavirus associated with severe acute respiratory syndrome. N Engl J Med. 2003;348(20): 1953-1966.
20. Peiris J, et al. Coronavirus as a possible cause of severe acute respiratory syndrome. Lancet. 2003;361:1319-1325.
21. Holmes K. SARS-associated coronavirus. N Engl J Med. 2003; 348(20):1948-1951.
22. Fouchier RA, et al. Aetiology: Koch’s postulates fulfilled for SARS virus. Nature. 2003;423:240.
23. MMWR Morb Mortal Wkly Rep. Update: Outbreak of severe acute respiratory syndrome—worldwide, 2003. 2003;52:269-272.
24. Holmes KV. Coronaviruses. In: Knipe DM, Howley PM, eds. Fields Virology. 4th ed. Philadelphia, Penn: Lippincott, Williams Wilkins; 2001:1187.
25. Wenzel R, et al. Coronavirus infections in military recruits: Three year study with coronavirus strains OC43 and 229E. Am Rev Respir Dis. 1974;109:621-624.
26. YiJun Ruan, et al. Comparative full-length genome sequence analysis of 14 SARS coronavirus isolates and common mutations associated with putative origins of infection. Lancet. 2003; 361:1779-1785.
27. Booth C, et al. Clinical features and short-term outcomes of 144 patients with SARS in the greater Toronto area. JAMA. 2003;289(DOI 10.1001/jama 289.21.JOC30885).
28. Vu T, et al. SARS in Northern Vietnam. Letter in N Engl J Med. 2003;348(20):2035.
29. Lee N, et al. A major outbreak of severe acute respiratory syndrome in Hong Kong. N Engl J Med. 2003;348:1986-1994.
30. Hsu LY, et al. Severe acute respiratory syndrome (SARS) in Singapore. Clinical features of index patient and initial contacts. Emerg Infect Dis. 2003;9(6):713-717. Available at: www.cdc.gov/ncidod/EID/vol9no6/03-0264.htm. Accessed 5/30/03.
31. Peiris J, et al. Clinical progression and viral load in a community outbreak of coronavirus-associated SARS pneumona: A prospective study. Lancet. 2003;361:1767-1778.
32. Wu E, Sung J. Haemorrhagic-fever-like changes and normal chest radiograph in a doctor with SARS. Lancet. 2003;361: 1520-1521.
33. Hon K, et al. Clinical presentations and outcome of severe acute respiratory syndrome in children. Lancet. 2003;361:1701-1703.
34. So L, et al. Development of a standard treatment protocol for severe acute respiratory syndrome. Lancet. 2003;361:1615-1617.
35. MMWR Morb Mortal Wkly Rep. Severe Acute Respiratory Syndrome (SARS) and coronavirus testing. United States—2003. 2003;52(14):297-302.
36. Nicholls J, et al. Lung pathology of fatal severe acute respiratory syndrome. Lancet. 2003;361:1773-1778.
37. Ho W. Guideline on management of SARS. Lancet. 2003; 361(Apr 19):1313-1315.
This review will provide an overview of SARS for the primary care physician, including epidemiology, etiology, review of the clinical and laboratory features as well as diagnosis, therapy, and prevention of SARS.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.