Drug Criteria & Outcomes: Formulary Evaluation of Omalizumab (Xolair)®
Formulary Evaluation of Omalizumab (Xolair)®
By David McLellan, PharmD candidate Harrison School of Pharmacy, Auburn (AL) University Written while on clinical rotation at Huntsville (AL) Hospital
Asthma affects nearly 18 million individuals in the United States. Prevention and reduction of asthma exacerbations are important goals of asthma management. Despite improvements in treatment options and an increased understanding of the disease process, many patients continue to have serious exacerbations requiring emergency medical attention. This amounts to approximately 10 million outpatient visits to physician offices and ambulatory clinics, and almost two million visits to hospital emergency rooms annually.
Standard asthma treatment includes inhaled b-agonists, inhaled anticholinergics, and inhaled and systemic steroids. Although these medications are effective in controlling exacerbations of the disease, they are not without side effects. To reduce side effects and patient dependence on asthma treatments, research has moved in the direction of gene therapy to target the disease at its source. Omali-zumab (Xolair®) is the first medication of its kind to be approved for treatment of allergic asthma.
Mechanism of action
Omalizumab is a humanized anti-IgE (immuno-globulin E) monoclonal antibody that inhibits the binding of IgE to the high-affinity IgE receptor on mast cells and basophils. This action limits the release of mediators of the allergic response.
Indication
Omalizumab is indicated for adolescents and adults ages 12 years and older with moderate-to-severe, persistent allergic asthma:
- who have had positive skin test or in vitro reactivity to perennial aeroallergens;
- whose signs and symptoms are controlled inadequately by treatment with systemic and inhaled corticosteroids.
Dosing
Omalizumab is dosed in 150 to 375 mg doses and administered as a subcutaneous (SC) injection every two or four weeks (see Tables 1 and 2). Dosage strength and frequency are determined by serum IgE levels (IU/mL taken before treatment initiation) and weight (in kg). Administration is limited to 150 mg per single injection, per injection site.
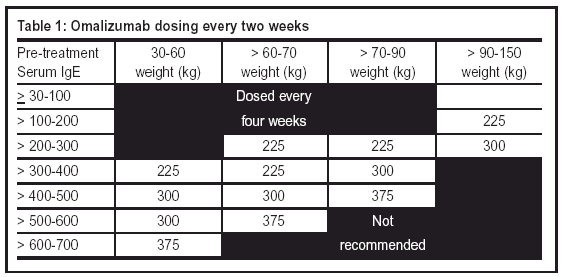
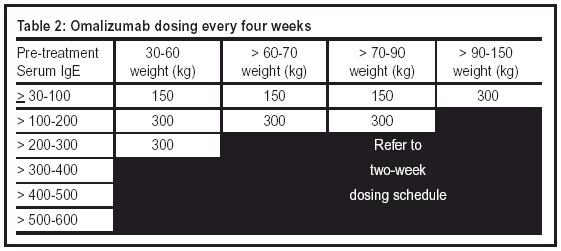
Dosing adjustments should be made when there are significant changes in body weight. Total serum IgE levels will be elevated during treatment and for up to one year after treatment discontinuation. Measured IgE levels during treatment and within one year following discontinuation should not be used in determining dosing adjustments.
Pharmacokinetics
Omalizumab is absorbed with an average bioavailability of 62% following a single SC injection. The medication exhibits linear kinetics at doses > 0.5 mg/kg, and takes an average of 7-8 days to reach peak serum concentrations. With multiple doses, the area under the serum concentration-time curve from day 0 to day 14 at steady state showed up to a sixfold increase vs. single injection. The volume of distribution in patients was a 78 ± 32 mL/kg.
The liver clears omalizumab in a method similar to IgG elimination. This process involves specific binding and complex formation of the drug with IgE. Omalizumab complexes are eliminated by the liver reticuloendothelial system and endothelial cells. The serum elimination half-life of the drug averaged 26 days with a daily clearance averaging 2.4 ± 1.1 mL/kg/day. Increases in body weight will increase clearance.
Contraindications
Omalizumab should not be administered to patients who have experienced a severe hypersensitivity reaction to the medication.
Warnings and precautions
Omalizumab is classified as pregnancy category B because animal studies have not illicited fetal toxicities; however, IgG (with which the omalizumab/
IgE complexes share elimination pathways) does cross the placenta. The medication should be used in pregnancy only if deemed medically necessary.
Anaphylaxis has occurred within two hours of the first or second dose of omalizumab in three patients without any other identifiable anaphylactic triggers. The patients presented with urticaria and throat and/or tongue edema. Patients who experience reactions such as these should be discontinued from omalizumab therapy.
Malignant neoplasms have occurred in 20 subjects treated with omalizumab. Breast, nonmela-noma skin, prostate, melanoma, and parotid neoplasms were most commonly observed.
Omalizumab is not intended for rescue therapy and should not be used in emergency situations. In patients who are dependent on high-dose steroids, abrupt discontinuation should not be performed. Instead, tapering under the close supervision of a physician is recommended.
Adverse reactions
In placebo-controlled trials, adverse reaction frequency was similar in both the omalizumab-treated and placebo-treated subjects. Some of the adverse reactions reported were: upper respiratory tract infections, viral infections, headache, pharyngitis, back pain, rhinitis, cough, myalgia, urticaria, nausea, dyspepsia, diarrhea, insomnia, and sinusitis.
Drug interactions
To date, no formal, published studies have evaluated omalizumab and potential drug-drug interactions.
Product information
Omalizumab (Xolair) is supplied as a lyophilized, sterile powder in a single-use, 5 mL vial, designed to deliver 150 mg of medication upon reconstitution with 1.4 mL of sterile water for injection. Unreconstituted vials should be stored at controlled temperatures between 2° and 8° C. Because the product contains no preservatives, upon reconstitution it should be used within eight hours if refrigerated or within four hours if stored at room temperature. The reconstituted product also should be protected from direct sunlight.
Cost
The hospital cost for one 5 mL vial of Xolair is $420.88. The suggested average wholesale price is: $541.25 per vial. This price does not include the cost of sterile water or other injection supplies. For the average patient, the monthly cost of Xolair may run from $600 to more than $3,000 dollars a month, including physician visits and supplies.
Evidence of efficacy and safety
Trial 1: Solér M, Matz J, Townley R, et al. The anti-IgE antibody omalizumab reduces exacerbations and steroid requirement in allergic asthmatics. Eur Respir J 2001;18:254-261.
Objective: To assess the clinical benefit and steroid-sparing effect of treatment with the anti-immunoglobulin-E antibody, omalizumab, in patients with moderate-to-severe allergic asthma.
Study design: In this multicenter, parallel-group, double-blind study, 546 allergic asthmatics that were symptomatic despite inhaled corticosteroids were randomized to receive placebo or omalizumab every two or four weeks subcutaneously for seven months.
Inclusion criteria
- Male or female, 12-75 years of age.
- Asthma diagnosis of one year or more based on the American Thoracic Society criteria.
- Positive skin prick test to a common household allergen.
- Total serum IgE level ³ 30 and £ 700 IU/mL.
- Total body weight £ 150 kg.
- Baseline FEV1 ³ 40% and £ 80% while not on bronchodilator treatment.
- A mean total symptom score of 3 or more for 14 days prior to randomization.
- Treatment with inhaled corticosteroids with a dose of 500-1,200 micrograms of beclomethasone dipropionate daily for three months or more prior to randomization.
- Use of b2 agonists on an as-needed basis.
- Stable asthma with no significant changes in regular medication and no acute exacerbations for one month or more.
Exclusion criteria
- Regularly taking systemic corticosteroids.
Treatment regimen: Five hundred forty-six subjects were randomized to receive either SC injection of omalizumab or matching placebo after a run-in phase of four to six weeks on beclomethasone dipropionate. During the first 16 weeks of the study, subjects were maintained on the inhaled cortico-steroid therapy while receiving omalizumab or placebo injections (steroid-stable phase). Over the last 12 weeks of the study, the steroid dose was reduced by 25% of baseline dose every two weeks for eight weeks (steroid-reduction phase). During the final four weeks, subjects were maintained on the lowest possible dose of inhaled corticosteroid.
Endpoints: The primary endpoint was the number of asthma exacerbations experienced per patient during the steroid-stable and steroid-reduction phases. The secondary endpoints were the number of patients experiencing asthma exacerbations during either phase, the percent reduction of inhaled corticosteroids, use of rescue inhaler, asthma symptom scores, morning peak expiratory flow, and FEV1 as percent predicted.
Results: Compared to the placebo group, the omalizumab group showed 58% fewer exacerbations per patient during the stable-steroid phase (P < 0.001). During the steroid-reduction phase, there were 52% fewer exacerbations in the omalizumab group vs. the placebo group (P < 0.001). Treatment with omalizumab was well-tolerated; the incidence of adverse events was similar in both groups.
Study conclusion: In this study, omalizumab therapy safely improved asthma control in allergic asthmatics that remain symptomatic despite regular treatment regimens, and showed marked reduction in dependence on inhaled corticosteroids.
Study strengths
- Randomized, placebo controlled, double-blind.
- Objective clearly stated.
- Endpoints clearly defined.
- Intent-to-treat analysis performed.
- Baseline demographics evenly matched between treatment and control group.
- Study design reproducible.
Study weaknesses
- Funded by Novartis.
- Did not follow the National Asthma Education and Prevention Program (NAEPP) guidelines for long-term asthma management.
Trial 2: Busse W, Corren J, Lanier BQ, et al. Omalizumab, anti-IgE recombinant humanized monoclonal antibody, for the treatment for severe allergic asthma. J Allergy Clin Immunol 2001;108:
184-190.
Objective: To evaluate the efficacy and safety of omalizumab in the treatment of inhaled cortico-steroid-dependent asthma.
Study design: In this Phase III, multicenter, parallel-group, double-blind study, 525 inhaled corticosteroid-dependent asthmatics were randomized to receive placebo or omalizumab every two or four weeks subcutaneously for seven months.
Inclusion criteria
- Male or female, 12-75 years of age.
- Asthma diagnosis one year or more.
- Positive skin prick test to a common household allergen.
- Total serum IgE level ³ 30 and £ 700 IU/mL.
- Baseline FEV1 ³ 40% and £ 80% while not on bronchodilator treatment.
- Treatment with inhaled corticosteroids with a dose of 420-840 micrograms of beclomethasone dipropionate or equivalent daily.
- Provided informed consent.
Exclusion criteria
- Prior exposure or sensitivity to omalizumab.
- Acute upper respiratory tract infection within one month.
- Less than three months of stable immunotherapy.
- Elevated IgE level for reasons other than atopy.
- Regular treatment with b-adrenergic antagonists.
- 750 mg or more omalizumab required per four weeks on the basis of serum IgE and body weight
Treatment regimen: Five hundred twenty-five subjects were randomized to receive either SC injection of omalizumab or matching placebo after a run-in phase of four to six weeks on beclomethasone dipropionate, if they were not already on it. During the first 16 weeks of the study, patients were maintained on the inhaled corticosteroid while receiving omalizumab or placebo injections (steroid-stable phase). During the last 12 weeks of the study, subjects’ steroid dose was reduced by 25% of baseline dose every two weeks for eight weeks (steroid-reduction phase). Subjects were maintained on the lowest possible dose of inhaled corticosteroid in the final four weeks.
Endpoints: The primary and secondary endpoints were the same as those used in Trial 1.
Results: Omalizumab treatment resulted in significantly fewer asthma exacerbations during the steroid-stable phase (P = 0.006), and fewer exacerbations during the steroid-reduction phase (P = 0.003). Inhaled corticosteroid use was significantly reduced in the omalizumab treatment group vs. placebo (P < 0.001), and steroid discontinuation was more likely with omalizumab (P < 0.001). Improvements in symptoms and pulmonary function occurred along with a reduction in rescue b-agonist use. Omalizumab was well-tolerated, with an adverse event profile similar to placebo.
Study conclusion: This study showed that addition of omalizumab to standard asthma therapy safely improves asthma control, and showed marked reduction in inhaled corticosteroid and rescue medication use.
Study strengths
- Randomized, placebo-controlled, double-blinded, parallel group.
- Reproducible.
- Objective clearly stated.
- Endpoints clearly defined.
- Intent-to-treat analysis performed.
- Baseline demographics evenly matched between treatment and control groups.
Study weaknesses
- Funded by Novartis.
- Did not follow NAEPP guidelines.
Trial 3: Finn A, Gross G, van Bavel J, et al. Omalizumab improves asthma-related quality of life in patients with severe allergic asthma. J Allergy Clin Immunol 2003;111:278-284.
Objective: To evaluate the effects of omalizumab on asthma-related quality of life.
Study summary: The data presented in this article were collected from the previous study (Trial 2) and followed the same criteria from start to finish with the addition of a 24-week double-blind extension phase during which subjects continued on omalizumab or placebo.
Results: The omalizumab and placebo groups were comparable in terms of baseline quality-of-life scores, as determined by the Juniper Asthma Quality of Life Questionnaire (AQLQ). At weeks 16, 28, and 52 (the extension phase), omalizumab-treated subjects demonstrated statistically significant improvements across all AQLQ domains with P values ranging from < 0.05 to < 0.001.
Study conclusion: In patients requiring moderate-to-high inhaled corticosteroid doses for severe allergic asthma, the improvement in disease control added by omalizumab therapy was paralleled by clinically meaningful and statistically significant improvements in asthma-related quality of life.
Additional studies have been performed to show that omalizumab has a positive benefit on asthma control in pediatric subjects. However, the product manufacturer did not seek FDA approval for this indication and does not support its use in this population. Ongoing studies are evaluating omalizumab use for seasonal allergy symptoms. The manufacturer and FDA do not yet support omalizumab’s use in this instance and further research is needed to ensure efficacy and safety.
Conclusion
Clinical studies prove that this medication is safe, efficacious, and has a positive benefit on patient quality of life, in populations with moderate-to-severe asthma. Omalizumab is not a substitute for current treatment regimens; the product should be a second-line option for patients whose asthma cannot be controlled by standard treatment regimens.
Because of the expense of the medication and chronic nature of the disease, a yearly financial plan would need to be developed to cover costs of treatment. In addition to the expense of the medication itself, there is increased cost associated with the laboratory work required to obtain initial doses of the drug. Omalizumab should therefore be considered nonformulary. If a patient already receiving omalizumab is hospitalized for a prolonged period of time, the patient should provide his/her home medication when time for administration of the next injection occurs.
Additional resources
- American Lung Association. Asthma in Adults Fact Sheet, March 2003. Web site available at: www.lungusa.org/asthma/aduasthmfac99.html. Accessed Sept. 15, 2003.
- Genentech. Xolair® (omalizumab) [product information]. South San Francisco, CA; 2003.
- Milgrom H, Fick RB, Su J, et al. Treatment of allergic asthma with monoclonal anti-IgE antibody. N Engl J Med 1999;341:1966-1973.
Asthma affects nearly 18 million individuals in the United States. Prevention and reduction of asthma exacerbations are important goals of asthma management.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
