Teriparatide (rDNA) origin (Forteo) formulary evaluation
Teriparatide (rDNA) origin (Forteo) formulary evaluation
By Brian Lee, PharmD candidate Harrison School of Pharmacy Auburn (AL) University Richard Cramer, PharmD Drug Information Coordinator Huntsville (AL) Hospital
Osteoporosis treatment medication
Teriparatide (rDNA) origin (Forteo) — Eli Lilly & Co.
Alendronate sodium (Fosamax) — Merck & Co. [To be used as standard therapy for comparative purposes in this evaluation.]
Mechanism of action
Teriparatide is the first synthetic, recombinant human parathyroid hormone (1-34) [rhPTH(1-34)] to stimulate new bone growth and increase bone mineral density (BMD) of the hip and lumbar spine by accelerating osteoblastic activity and restore bone integrity and architecture. The rhPTH has the same biological properties as the endogenous PTH synthesized by the parathyroid gland. The PTH is an endogenous 84-amino acid peptide that is secreted due to a low plasma Ca (calcium) concentration in the extracellular fluid. The released PTH rapidly binds to the specific high-affinity PTH-1 receptors and initiates a cascade of events: adenylyl cyclase activation, increased cAMP production, and phospholipase C activity induction.
PTH regulates Ca and phosphate metabolism at these three target sites: bone, kidney, and gastrointestinal (GI) tract. PTH increases ionic Ca and phosphate resorption at the bone and increases bone formation. The kidney responds to PTH by 1) increasing the reabsorption of both Ca and Mg (magnesium) at the distal convoluted tubule and the ascending Loop of Henle; 2) accelerating the transport of phosphate, amino acids, Na (sodium), Cl (chlorine), SO42- (sulfate ion), and HCO3- (bicarbonate ions) into the urine; and 3) increasing the production of the active form 1a,25-dihydroxy-vitamin D (Calcitriol). PTH stimulates intestinal Ca and phosphate absorption across the small intestine. A negative feedback system inhibits hormone release when ionic Ca levels in the plasma achieve homeostasis.
Indications
• Teriparatide (rDNA) origin
• Postmenopausal osteoporotic women at high risk for fracture.
— History of osteoporotic fracture.
— Multiple risk factors for fracture.
— Intolerant of bisphosphonates, selective estrogen-receptor modulators, or hormone replacement therapy/estrogen replacement therapy.
• Primary or hypogonadal osteoporosis in men at high risk for fracture
— History of osteoporotic fracture.
— Multiple risk factors for fracture.
— Intolerant of bisphosphonates, selective estrogen-receptor modulator, or hormone replacement therapy/estrogen replacement therapy.
• Alendronate is used to prevent and treat post- menopausal osteoporosis, glucocorticoid- induced osteoporosis, and Paget’s disease, and to increase bone mass in men with osteoporosis.
One of the most significant clinical differences between teriparatide and other treatments for osteoporosis is the subcutaneous route of administration. Table 1 summarizes the pharmacokinetic parameters associated with teriparatide. The pharmacokinetic data for teriparatide in pediatric patients are not currently available. There are no age-related pharmacokinetic differences among patients between 31 and 85 years of age. There are also no pharmacokinetic differences in patients with mild or moderate renal insufficiency, but limited data demonstrate patients with severe renal insufficiency may have a 73% increase in area under the curve (AUC) and a 77% increase in T½. There are no specifically recommended dosage reductions in these patients at this time. No pharmacokinetic data are available for patients with chronic renal failure undergoing dialysis, heart failure, hepatic impairment, and pediatrics.
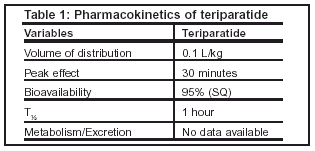
The pharmacokinetics of alendronate differ significantly from teriparatide. Alendronate may remain in the bone tissue for many years and is not hepatically metabolized. Alendronate is not recommended in patients with a creatinine clearance of less than 30 mL/min.
Dosing
The Forteo pen is supplied in a glass cartridge, disposable pen injector (750 mcg/3.3 mL). The device contains 3.3 mL of drug (a 28-day supply). The recommended dosage is 20 mcg/day for currently approved indications.
For treatment and prevention of osteoporosis, 70 mg/week and 35 mg/week are recommended, respectively. Patients diagnosed with Paget’s disease should take 40 mg daily for six months or 5 mg daily for glucocorticoid-induced osteoporosis.
Administration
Teriparatide is given as a subcutaneous injection in the thigh or abdominal wall. When administering an initial injection, the patient should be in an area where they can quickly sit or lie down should they experience orthostatic hypotension. RhPTh should be refrigerated (36-46° F) and not frozen. The safety and efficacy of this drug has not been evaluated beyond two years; a trial funded by Eli Lilly currently is testing the efficacy of Forteo for a 10-year period.
Alendronate should be taken one-half hour before food with plain water. Patients should not lie down for 30 minutes and until after their first meal of the day.
Contraindications
Forteo should not be administered to patients with hypersensitivity to teriparatide or any excipients used in the formulation.
Warning/precautions
The only significant black box warning for teriparatide is the increased potential risk of developing osteosarcoma. This incidence was observed in laboratory rats exposed to three to 60 times the recommended dosage of 20 mcg. Table 2 lists the warnings/precautions of teriparatide.
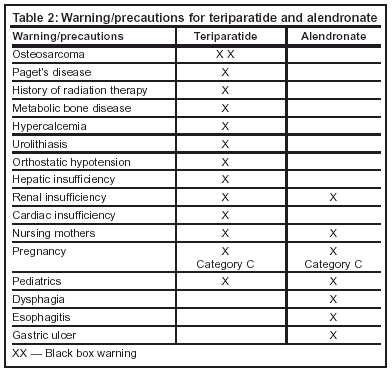
Because no clinical trials have been conducted on the safety in patients with active urolithiasis or hypercalcemia using teriparatide, it should be used with caution. Usage of teriparatide in these patients may exacerbate their condition; therefore, close monitoring of urine calcium is highly recommended. A transient orthostatic hypotension may occur after the first several doses of teriparatide. However, the adverse reaction should resolve within a few minutes to hours. Placing a patient in a reclining position in a chair relieves the dizziness, lightheadedness, or palpitations associated with the hypotension.
Some of the precautions in using alendronate include renal insufficiency, pregnancy, pediatrics, and patients with gastric ulcers. Nursing mothers should take precautions before taking alendronate.
Drug interactions
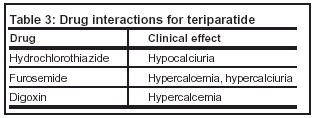
Serum calcium levels should be assessed 16 hours post-injection of teriparatide. If persistent hypercalcemia occurs, the medication should be discontinued promptly pending further evaluation. Table 3 lists potential drug interactions with teriparatide.
Adverse effects
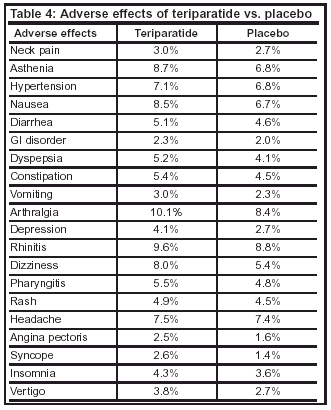
Most adverse effects associated with teriparatide involve the nervous and respiratory systems. Table 4 summarizes the adverse effects of teriparatide.
The adverse effects of alendronate are mild and include abdominal pain, constipation, and nausea.
Monitoring parameters
No clinical studies have been conducted on overdose with teriparatide. Treatment of overdose should include discontinuing the drug, implementing supportive treatment, and monitoring serum concentrations of phosphate and calcium. Serum uric acid levels should be assessed since a clinical trial has demonstrated increased uric acid concentrations in 2.8% of patients treated with teriparatide, although the hyperuricemia did not cause the development of arthralgia, urolithiasis, or gout in these patients.
Patients on alendronate should have their alkaline phosphatase and serum potassium monitored periodically.
Costs
A month supply of alendronate costs $53.10 — much less than a month supply of teriparatide, approximately $450 per month. Due to Forteo pen injector’s availability only as a 28-day drug supply, the hospital would incur significant outpatient therapy costs by supplying Forteo for inpatients.
Clinical trials
Trial one: Neer RM, Arnaud JR, Zanchetta RP, et al. Effect of parathyroid hormone (1-34) on fractures and bone mineral density in postmenopausal women with osteoporosis (N Engl J Med 2001;344: 1434-1441).
Objective: To determine the efficacy of teriparatide and a safe dose in the treatment of postmenopausal women diagnosed with osteoporosis who had two or more vertebral fractures.
Study design: A double-blind, randomized, placebo-controlled Phase III study up to 24 months (median of 21 months), including 1,637 postmenopausal women with at least two vertebral fractures.
Intervention: All women received 1,000 mg calcium and 400-1,200 IU of vitamin D for one to six months. All women gave themselves daily injections of a placebo for the first two weeks and then were randomly chosen to receive a placebo (n = 544, 18 months), 20 mcg teriparatide (n = 541, 18 months), or 40 mcg teriparatide (n = 552, 17 months).
Patient population
• Inclusion criteria
• Postmenopausal women (at least five years) with at least one moderate or two mild vertebral fractures. If the women had fewer than two moderate fractures, they had to have a bone mineral density of the hip or lumbar spine of one or more standard deviations below normal premenopausal women (20-35 years).
• Exclusion criteria
• Bone or calcium metabolism.
• Drug abuse.
• Urolithiasis within five years.
• Corticosteroid treatment within six months.
• Renal insufficiency (Scr > 2 mg/deciliter).
• Abnormal urine calcium levels.
• Hepatic insufficiency.
• Abnormal serum calcium levels.
• Alcohol abuse.
Statistical analysis: Pearson’s chi-square test and evaluation by analysis of variance.
Outcomes measured:
• Serum calcium measured before and four to six hours after injection at baseline and at one, three, six, 12, 18, and 24 months.
• Urine calcium and creatinine measured at baseline and at one, six, 12, and 24 months.
• Thoracic and lumbar spine radiograph measured at baseline and 12 months.
• Bone density of spine measured at baseline and at three, six, 12, 18 months, and the last month.
• Bone density of hip, forearm, and total body measured at baseline, 12 months, and the last month.
• Height measured at baseline and 12 months.
• Blood counts and chemical tests measured at baseline and at one, six, 12, and 24 months.
• Urinanalysis measured at baseline and at one, six, 12, and 24 months.
• Tested circulating antibodies to PTH at base- line and at three, 12, and 24 months.
Results
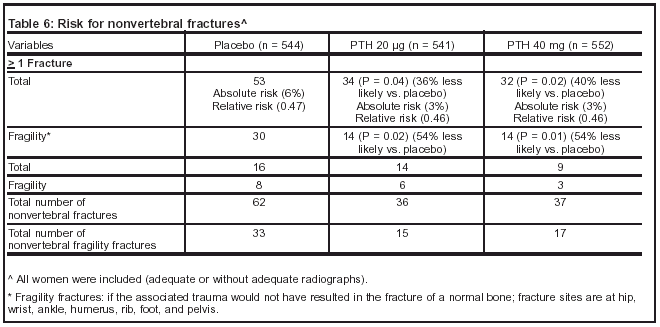
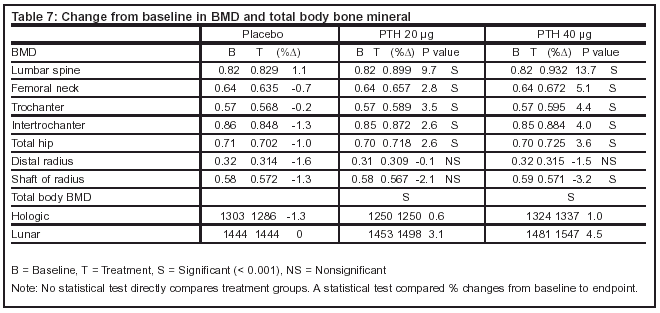
Tables 5-7 summarize the risks of the Neer, et al. study.
Adverse effects
• Cancer: Placebo (4%), PTH 20 mg (2%, P = 0.02), PTH 40 mg (2%, P = 0.07).
• Nausea: Placebo (8%, P < 0.001), PTH 40 mg (18%).
• Headache: Placebo (8%, P = 0.01), PTH 40 mg (13%).
• Dizziness: Placebo (6%, P = 0.05), PTH 20 mg (9%).
• Leg cramps: Placebo (1%, P = 0.02), PTH 20 mg (3%).
• Other adverse effects: Hypercalcemia (> 10.6 mg/deciliter), hypercalciuria (> 300 mg/day), and uric acid and circulating antibodies cross-react with teriparatide.

Strengths
• Prospective, double-blind, randomized, placebo-controlled study.
• Pearson’s chi-square test in the three study groups.
• Methods in study design were appropriate (bone mineral density).
• Participants from 17 countries.
Limitations
• Study subjects predominantly Caucasian women.
• Study period was set for 24 months but was conducted for 12 months.
• No data available from 13-24 months.
• Women with illnesses affecting bone or calcium metabolism, urolithiasis, hepatic insufficiency, renal insufficiency (Scr > 2 mg/deciliter), and drug and alcoholic abusers were not participants of the study
• Some P values were not calculated in the comparison between placebo and PTH-treated associated adverse events.
• Supported by Eli Lilly, and co-author of the study owns Eli Lilly stocks.
• Compliance rate of 79-83%.
Author’s conclusions: PTH in doses of 20 mg and 40 mg daily increases bone mineral density of the spine up to 13% compared to placebo and that reduces the risk of vertebral fractures by 69%. PTH-treated (40 mg) postmenopausal women with osteoporosis had a 54% reduction in nonvertebral fractures.
Trial two: Orwoll ES, Scheele WH, Paul S, et al. The effect of teriparatide [human parathyroid hormone (1-34)] therapy on bone mineral density in men with osteoporosis. J Bone Miner Res 2003;18:9-18.
Objective: To determine the effect of teriparatide in men with osteoporosis with a bone mineral density at least two standard deviations from the average young men.
Study design: Centralized block randomization, double-blind, placebo-controlled study.
Intervention: 437 men were randomly assigned to receive a placebo (n = 147), 20 mg/day teriparatide (n = 151), or 40 mg/day teriparatide (n = 139). However, only 311 men actually received teriparatide or placebo. All patients received 1,000 mg calcium and between 400 and 1,200 IU vitamin D at least one month before the study.
Patient populations
• Inclusion criteria
• Men ages 30-85, not chronically disabled, with osteoporosis. Recruited from 11 countries.
• Subjects with normal or low levels of testosterone.
• Hypogonadal patients with a stable dose of androgen or anabolic steroids that would be continued during the study for at least six months of therapy.
• Exclusion criteria
• Men diagnosed with metabolic bone diseases including excess glucocorticoid.
• Use of estrogen agonists or antagonists, coumarin and indandione derivatives, anti- convulsants, or calcium- or aluminum- containing antacids.
• Diagnosed with nephrolithiasis or urolithiasis within two years, sprue, inflammatory bowel disease, malabsorption syndrome, poor intestinal absorption of calcium, low urinary calcium excretion, and elevated endogenous parathyroid hormone levels, hepatic or renal insufficiency, drinks more than six alcoholic beverages a day, or abuses drugs.
• Paget’s disease, renal osteodystrophy, or osteomalacia.
• Received androgen, anabolic steroids, progestins, calcitonin, fluorides, oral bisphosphonates, vitamin D of more than 50,000 IU/week, or calcitriol within six months.
• Growth hormone deficiency, previous pituitary surgery, tumor, or radiation.
• History of carcinoma or suspected carcinoma within five years.
• Abnormalities of lumbar spine inhibiting the assessment of bone mineral density.
Statistical analysis
• ANOVA to analyze continuous measures including treatment and country.
• Pearson’s chi-square test (P < 0.05 for significance).
Outcomes measured
• Intact serum PTH (1-84) measured at baseline and 12 months.
• Serum bone alkaline phosphatase measured at baseline and at one, three, six, and 12 months.
• Lumbar spine bone mineral density measured at baseline and at three, six, and 12 months.
• Hip, whole body, and radial bone mineral density measured at baseline and 12 months.
• Serum calcium levels measured at one, six, and 12 months.
• Calcium and creatinine excretion measured at baseline and at one, six, and 12 months.
• An elevation in serum calcium or urinary calcium excretion rates of greater than 350 mg/ day, and increased urinary calcium to creatinine ratio in patients treated with teriparatide received a reduced dose of calcium supplement or were discontinued.
• Testosterone levels were measured to establish a baseline value using radioimmunoassay.
• Serum estradiol, complete blood count, urinalysis, and serum chemistry were measured at baseline and at six and 12 months.
• Serum parathyroid hormone (1-84) was assessed at baseline and 12 months.
• Radioimmunoassay was used to measure serum antibodies to teriparatide at baseline and 12 months.
• Smoking and consumption of alcoholic beverages were noted and documented at baseline.
Results
• Mean serum Ca concentration was higher in the teriparatide-treated groups than placebo throughout the 12 months of study, but only 6.2 % and 16.8% of the patients receiving 20 mg and 40 mg teriparatide, respectively, were hypercalcemic. Four patients in the 20 mg group and 12 patients in the 40 mg group needed to reduce their calcium supplementa- tion due to hypercalcemia, elevated urinary Ca excretion, and symptoms of headache or nausea. Seven patients in the 40 mg group reduced their dose to 20 mg while three patients in the 20 mg group and six patients in the 40 mg group withdrew from the study.
• Mean serum concentrations of 1,25 (OH)2 vita- min D increased in both teriparatide treatment groups.
• A negative test on serum antibody against teriparatide was demonstrated.
• Uric acid levels were above normal in 0.7%, 0%, and 2.3% in the placebo, 20 mg group, and 40 mg group, respectively.
• Serum chloride levels were below normal in 2.1%, 1.4%, and 1.5% in the placebo, 20 mg group, and 40 mg group, respectively.
• Serum magnesium levels were below normal in 2.3% of the 40 mg group.
• Changes in bone mineral density were not significantly affected by free testosterone and estradiol levels (P = 0.398).
• Of the 39 patients withdrawn, 4.8%, 9.3%, and 12.9 % were in the placebo, 20 mg group, and 40 mg group, respectively.
• The teriparatide 40 mg group experienced a statistically significant increased rate (P < 0.001) of nausea compared to placebo.
• The teriparatide 40 mg group experienced a statistically insignificant increased rate (P = 0.053) of headache compared to placebo.
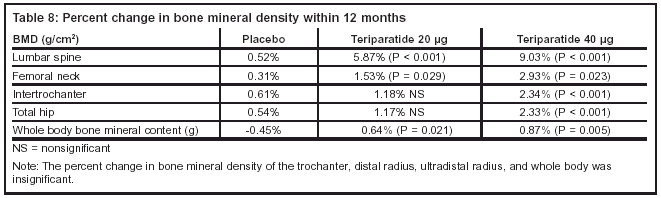
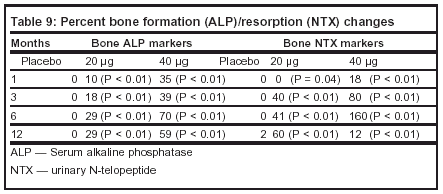
Tables 8 and 9 summarize the the changes in bone mineral density and bone formation reported in the Orwoll et al study.
Strengths
• Centralized block randomization, double- blind, placebo-controlled study.
• Inclusion and exclusion criteria were appropriate.
• Men from 11 countries.
• Intention-to-treat analysis performed.
• Standard deviation reported.
• a value set at 0.05 at the beginning of the study.
• A study conducted in men with osteoporosis although less common than in women.
Limitations
• Compliance rate was 79% and no statistical analysis was performed.
• All data were not presented in this article.
• Initial intact serum PTH (1-84) was not measured.
• Patient population consisted of mainly Caucasians.
• Percentage of patients with headaches in the placebo and teriparatide 20 mg group were not given.
• Eli Lilly supported the study, and the authors were consultants or employees of Eli Lilly.
• The baseline T-scores in the placebo, teriparatide 20 mg group, and 40 mg group were more representative of patients with osteopenia than osteoporosis (National Society of Osteoporosis Guidelines not followed).
Author’s conclusions: Teriparatide increases lumbar spinal and femoral neck bone mineral density in men with osteoporosis during a period of 11 months. Teriparatide-treated patients had an increase in serum calcium levels and an increase in 1,23 (OH)2 vitamin D levels.
Other clinical trials: A third randomized double-blind, placebo-controlled trial examined the effects of teriparatide on the skeletal changes and adverse effects in rats over a 24-month period. There was a substantial increase in bone mineral density after the administration of teriparatide. One unique finding from this study was that rats developed osteoblastoma, osteoma, focal osteoblast, and osteosarcoma in a dose-dependent and duration-dependent manner. Although the findings in this study cannot directly be extrapolated to humans, this clinical data should be factored into the drug’s profile of benefits vs. risks. This significant finding has prompted the U.S. Food and Drug Administration to require Eli Lilly to place a black box warning in the package insert that teriparatide may cause osteosarcoma.
Interestingly, two clinical trials compared the bisphosphonate alendronate to teriparatide in postmenopausal women with osteoporosis. The study design and outcomes measured were similar to the first two trials described in this evaluation. Teriparatide increased lumbar spine bone mineral density, femoral neck bone mineral density, and total hipbone mineral density significantly more than 10 mg alendronate daily. The Body, et al. study showed that postmenopausal women treated with teriparatide for 12 months had a greater than a 150% change in bone resorption and a 50% change in bone formation; however, alendronate reduced biochemical markers of bone resorption and formation by 50%. There was no explanation given as to why alendronate reduced bone resorption and formation in 12 months and statistical tests were not conducted to determine if bone resorption was significantly different from bone formation in the two treatment groups (teriparatide and alendronate). However, other studies have demonstrated that alendronate inhibits bone resorption and not bone formation, and increases bone mineral density.
Summary and recommendations: Clinical trials of teriparatide provided data that it increases bone mineral density at certain bone sites. No study has been conducted to test the efficacy of teriparatide on humans beyond the two years. Furthermore, the risk of developing osteosarcoma by using teriparatide was found to be dose- and duration-dependent in animals. Formulary recommendations are as follows: 1) do not stock as formulary drug; 2) defer starting drug in hospital for chronic drug therapy, and start in outpatient setting where therapy costs can be correlated with insurance reimbursement; and 3) patients on chronic outpatient therapy should use their home supply for in-hospital use.
Resources
• Eli Lilly & Co. Forteo AMCP Formulary Submission Dossier. Indianapolis; 2002.
• Neer R, Arnaud C, Zanchetta J, et al. Effect of parathyroid hormone (1-34) on fractures and bone mineral density in postmenopausal women with osteoporosis. N Engl J Med 2001;344:1434-1441.
• Vahle J, Sato M, Long G, et al. Skeletal changes in rats given daily subcutaneous injections of recombinant human parathyroid hormone (1-34) for 2 years and relevance to human safety. Toxicol Pathol 2002;30:312-321.
• Tashjian A, Chabner B. Commentary on clinical safety of recombinant human parathyroid hormone 1-34 in the treatment of osteoporosis in men and postmenopausal women. J Bone Miner Res 2002;17:1151-1161.
• Orwoll E, Scheele W, Paul S, et al. The effect of teriparatide [human parathyroid hormone (1-34)] therapy on bone density in men with osteoporosis. J Bone Miner Res 2003;18:9-16.
• Kuijpers G, Schneider B, Stadel B, et al. Recombinant human parathyroid hormone: Preclinical data on rat osteosarcoma were not dismissed. Br Med J 2002;324:1218-1220.
• Reeve J. Recombinant human parathyroid hormone: Author’s reply. Br Med J 2002;324:1218-1219.
• Body J-J, Gaich G, Scheele W, et al. A randomized double-blind trial to compare the efficacy of teriparatide [Recombinant human parathyroid hormone (1-34)] with alendronate in postmenopausal women with osteoporosis. J Clin Endocrinol Metab 2002;87:4528-4535.
• Guyton AC, Hall JE. Textbook of Medical Physiology. Ninth ed. Philadelphia: W.B. Saunders Co.; 1996:992-995.
• Haynes RC, Murad F. Agents affecting calcification: Calcium, parathyroid hormone, calcitonin, vitamin D, and other compounds. In: Gilman AG, et al, eds. Goodman and Gilman’s The Pharmacological Basis of Therapeutics. Seventh ed. New York City: MacMillan Publishing Co.; 1985: 1517-1543.
• Merck & Co. Fosamax Package Insert Literature. 2003; Available at: www.fosamax.com.
• Personal Communication. R. Cramer, PharmD, Drug Information Coordinator. Department of Pharmacy, Huntsville (AL) Hospital; July 2003.
Teriparatide is the first synthetic, recombinant human parathyroid hormone (1-34) [rhPTH(1-34)] to stimulate new bone growth and increase bone mineral density (BMD) of the hip and lumbar spine by accelerating osteoblastic activity and restore bone integrity and architecture.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
