Heart Failure
Heart Failure
Authors: Robert E. Hobbs, MD, and RogerM. Mills, MD, Cleveland Clinic Foundation, Department of Cardiovascular Medicine, Cleveland, Ohio.
Editor’s Note—Heart failure is on the rise and reaching epidemic proportions.1-2 Approximately 5 million Americans have heart failure (2% of the population), and the number is expected to increase during the next 30 years. Hospital admissions for heart failure have tripled over the last 3 decades and now approach 1 million per year. Most hospitals sustain financial loss with these admissions.
The incidence of heart failure increases rapidly with age. It is the No. 1 diagnosis-related group in the elderly and the single largest expense for Medicare. Heart failure has the highest readmission rate of any hospital discharge diagnosis, largely due to patient noncompliance and failure to optimize patient management. Long-term prognosis with heart failure is poor, worse than most malignant tumors, with a 5-year survival of only 25-50%. Heart failure costs were estimated to be a staggering $38 billion in 1994 and probably are higher today. Two-thirds of these costs are hospital charges.
Because of the huge health and economic burden of heart failure, we now have several sets of guidelines, each focused on a different aspect of the heart failure syndrome: prevention, staging, workup, standard therapies, unresolved issues, and future therapies.3-8 These guidelines are based on evidence from clinical trials. Despite this published information, slightly more than half of heart failure patients receive ACE inhibitors and less than half are treated with a beta-blocker.
This article reviews multiple aspects of heart failure, providing direction for workup and management of heart failure patients. All recommendations follow current published guidelines. Unresolved and controversial issues are reviewed, and future therapies are proposed.
Definition
Heart failure occurs when impaired cardiac function is inadequate to meet the metabolic needs of the body. It is a syndrome characterized by dyspnea, fatigue, fluid retention, and objective evidence of systolic or diastolic cardiac dysfunction. Heart failure is the end result of many diseases, including coronary artery disease, idiopathic dilated cardiomyopathy, hypertension, valvular heart disease, congenital heart disease, arrhythmias, and toxins. End-stage coronary artery disease is the most common cause, accounting for more than half of the cases.
Heart failure may be characterized in several ways: acute vs chronic, systolic vs diastolic, left vs right sided, and high vs low output. Acute heart failure refers to rapid cardiac decompensation leading to dyspnea, acute pulmonary edema, or fluid retention. Chronic heart failure refers to prolonged impairment in functional capacity due to dyspnea, fatigue, and fluid retention. The most common clinical presentation is chronic with acute exacerbations. Systolic heart failure refers to left ventricular contractile dysfunction. Diastolic heart failure occurs in the setting of normal left ventricular contractility and is associated with abnormal ventricular relaxation, left ventricular hypertrophy, diastolic dysfunction, and a small left ventricular cavity.9-10 Diastolic heart failure, seen more commonly in the elderly and women, occurs with hypertension, coronary artery disease, aortic stenosis, and myocardial fibrosis. Frequently, diastolic heart failure occurs in association with systolic failure, each contributing to cardiac dysfunction. Left-sided heart failure refers to symptoms of effort intolerance and dyspnea. Right-sided heart failure produces congestion in the systemic veins manifested by ascites and edema. Right-sided heart failure does not distinguish between which ventricle is more severely damaged, because right heart failure often is caused by left heart failure. Low-output heart failure is characterized by marked fatigue and effort intolerance, often in the absence of fluid retention. High-output failure is uncommon, occurring with thyrotoxicosis, anemia, arteriovenous fistula, and beri-beri. Heart failure symptoms may be described in terms of New York Heart Association Functional Class (NYHA FC) (see Table 1 below). Heart failure should always be designated as a syndrome secondary to a known cardiovascular disease and never as the final diagnosis.
| Table 1. New York Heart Association Heart Failure Symptom Classification | |
|
NYHA
|
Level of Impairment |
|
I |
No symptoms with ordinary activities |
|
II
|
Symptoms with moderate activities |
|
III
|
Symptoms with mild activities |
|
IV
|
Symptoms at rest or with minimal activities |
Epidemiology
Some 550,000 new cases of heart failure are diagnosed annually. The prevalence, increasing with age, will become more common during the next 30 years. The mean age of patients with heart failure is 74 years with a male to female ratio of 1:1. The number of hospitalizations has increased steadily as a result of an aging population, improved therapeutics, and the inevitable progression of underlying cardiac diseases. Heart failure has the highest readmission rate of any discharge diagnosis. The prognosis of heart failure generally is poor. Half of the patients die suddenly, and most others die of progressive pump failure. Heart failure is responsible for approximately 280,000 deaths each year. Half of the heart failure population will die within 4-5 years of diagnosis; half of the patients with severe heart failure will die in 1-2 years. Prognosis is related to the severity of the syndrome in most cases. Markers of severe cardiac dysfunction include impaired exercise tolerance, reduced left ventricular ejection fraction, elevated B-type natriuretic peptide levels, wide QRS complex, hyponatremia, hypocholesterolemia, and cardiac cachexia.
| Table 2. ACC/AHA Classification of Chronic Heart Failure | |
| Stage | Description |
|
A. High risk for developing heart failure |
Hypertension, diabetes mellitus, CAD, family history of cardiomyopathy. |
| B. Asymptomatic heart failure | Previous MI, LV dysfunction, valvular heart disease. |
| C. Symptomatic heart failure | Structural heart disease, dyspnea and fatigue, impaired exercise tolerance. |
| D. Refractory end-stage heart failure | Marked symptoms at rest despite maximal medical therapy |
Recent guidelines have concentrated on prevention of heart failure and management of end-stage patients.6 In this classification, heart failure is divided into 4 stages (see Table 2 above). Stage A heart failure refers to patients who are at risk of developing heart failure but have no structural heart disease at present. These include patients with hypertension, coronary artery disease, diabetes mellitus, valvular heart disease, congenital heart disease, and use of cardiac toxins. Stage B heart failure refers to patients who have structural heart disease but are asymptomatic, corresponding to NYHA FC I. Stage C heart failure includes patients with symptoms and evidence of structural heart disease (NYHA FC II-III). This group of patients has received the most attention from clinicians. Stage D heart failure refers to patients with end-stage heart disease refractory to medical therapy. This stage corresponds to NYHA FC IV.
Pathophysiology
The pathophysiology of heart failure remains incompletely understood. There is a complex interrelationship between precipitating etiologies, compensatory pathophysiology, and progressive left ventricular dysfunction. Hemodynamic, neurohormonal, structural, and noncardiac abnormalities occur in heart failure. The classic hemodynamic abnormalities are low cardiac output and high intracardiac filling pressures. Low cardiac output leads to fatigue and effort intolerance, whereas high intracardiac filling pressures result in dyspnea and edema.
Neurohormonal abnormalities include activation of the sympathetic nervous system, stimulation of the renin-angiotensin-aldosterone system, release of vasopressin, and elevated levels of endothelin, natriuretic peptides, and cytokines.11 Sympathetic activation is associated with increased plasma levels of norepinephrine, and patients with the highest levels have the worst prognosis.12 Abnormalities caused by sympathetic activation include resting tachycardia, vasoconstriction, beta-receptor dysfunction (which also results from a change in phenotype of B receptor as well as receptor down-regulation), myocardial toxicity, adverse remodeling, cardiac arrhythmias, and activation of other neurohormones. Activation of the renin-angiotensin-aldosterone system leads to elevated levels of angiotensin-II and aldosterone. Angiotensin-II is a potent vasoconstrictor and growth factor, which causes sodium and water retention and release of aldosterone. Aldosterone, released from the adrenal cortex, induces sodium retention, potassium loss, sympathetic activation, baroreceptor dysfunction, and myocardial fibrosis. Vasopressin is released from the hypothalamus as a result of osmotic and baroreceptor stimuli. Vasopressin is a vasoconstrictor, which promotes sodium and water retention. Endothelin arises from multiple sources with the cardiovascular system. Elevated endothelin levels in heart failure are associated with growth and remodeling, vasoconstriction, and neurohormonal augmentation. Tumor necrosis factor, an inflammatory cytokine, arises from macrophages and causes cardiac cachexia, skeletal muscle dysfunction, and apoptosis in the failing heart. The altered loading conditions and myocardial changes mediated by this neurohormonal response to impaired cardiac function lead to short-term hemodynamic improvements but long-term deleterious effects through ventricular remodeling. In contrast, natriuretic peptides are volume regulatory hormones secreted by the atria and ventricles in response to elevated filling pressures. They are arterial and venous dilators, which promote sodium and water excretion, and modulate other neurohormones.
Cardiac remodeling, resulting from myocardial injury and neurohormonal activation, is characterized by left ventricular hypertrophy, dilatation, abnormal beta-receptor function, expression of fetal phenotype, interstitial fibrosis, and apoptosis. Remodeling causes the heart to assume a spherical rather than an elliptical shape. This altered geometry results in mitral and tricuspid regurgitation. Noncardiac organ dysfunction is common in heart failure. Low cardiac output decreases perfusion to the kidneys leading to sodium and water retention, as well as renal dysfunction. Skeletal muscle dysfunction impairs exercise capacity and causes further debilitation.
Symptoms
Patients with left ventricular dysfunction may be asymptomatic or may experience dyspnea, fatigue, and fluid retention. Common symptoms of heart failure include effort intolerance, lack of stamina, dyspnea, orthopnea, paroxysmal nocturnal dyspnea, cough, weight gain, fluid retention, edema, abdominal bloating, nausea and vomiting, ascites, lightheadedness, anorexia, and muscle wasting. The clinician should seek evidence of underlying heart disease when interviewing the heart failure patient. Inquire about angina pectoris, previous myocardial infarctions, catheterizations, or cardiac surgery. Ask about hypertension, diabetes mellitus, heart murmurs, lung disease, thyroid problems, family history, tobacco abuse, hyperlipidemia, and anemia. Determine if there is a family history of cardiomyopathy, heart failure, or sudden death. It is also important to search for a precipitating cause of decompensated heart failure, although the most common cause is patient noncompliance with dietary restrictions or medications. Other causes of decompensation include inadequate medical treatment, arrhythmias (atrial fib/flutter), worsening renal function, other drugs (NSAIDS, diabetic TZD agents), infections, pulmonary embolism, myocardial ischemia, valvular regurgitation, anemia, hypothyroidism, or alcohol abuse. There is poor correlation between symptoms and left ventricular ejection fraction or prognosis. Mild symptoms do not necessarily equate with mild dysfunction. The initial severity of symptoms do not predict prognosis. Symptoms are best described in terms of NYHA functional class. Although this scheme is quite subjective, it correlates well with outcome.
Signs
Blood pressure, heart rate, and respiratory rate are variable in heart failure. Blood pressure may be normal, low, or high, although pulse pressure frequently is narrow. Heart rate may be normal or increased. Pulsus alternans, a beat-to-beat variation in the intensity of the pulse, indicates severe heart failure. Respirations may be normal or rapid with decompensation. Sleep disordered breathing is common. A patient with decompensated heart failure often is diaphoretic. Jugular venous distention and hepatojugular reflux (elicited by sustained elevation in JVP while pressing on the liver and noting further distention of the neck veins) indicate congestion and fluid overload. Pulmonary examination in chronic heart failure usually reveals clear lungs. Rales are heard with acute decompensation, and decreased breath sounds at the bases are noted with pleural effusions. Cardiac palpation frequently demonstrates a diffuse, displaced apical impulse. Rhythm may be regular or irregularly irregular with atrial fibrillation. Often the decompensated patient has resting tachycardia. The first heart sound usually is normal; the second heart sound may be paradoxically split from delayed mechanical or electrical activation of the left ventricle. A fourth heart sound reflects a noncompliant left ventricle, whereas a third heart sound often indicates elevated LV filling pressures. With heart rates over 90 beats/min in sinus rhythm, these sounds merge into a summation gallop. Murmurs of mitral and tricuspid regurgitation are common. Abdominal examination may reveal hepatosplenomegaly and ascites. The legs often are edematous. Cool hands and feet signal low cardiac output. The presence of multiple physical signs increases the likelihood that heart failure is indeed the correct diagnosis.
Diagnostic Testing
The initial evaluation should define the etiology of the syndrome. The electrocardiogram may show Q waves or left bundle branch block, both good predictors of left ventricular dysfunction. Left ventricular hypertrophy may reflect either systolic or diastolic heart failure. Atrial fibrillation and atrial flutter are common. A normal electrocardiogram has a 90% negative predictive value in excluding heart failure. The chest radiograph often is abnormal, showing cardiomegaly and pulmonary congestion. Specific radiographic abnormalities include increased cardiothoracic ratio, prominent pulmonary arteries, increased pulmonary vascularity with redistribution of blood flow to upper lobes, pleural effusions, Kerley B lines, fluid in the fissures, interstitial edema, and alveolar edema. Recommended laboratory studies in heart failure include complete blood count, chemistry panel, thyroid function tests, lipid profile, and B-type natriuretic peptide (BNP) assay. Measurement of BNP level is an important screening test for heart failure.13 BNP is released from the ventricles in response to pressure or volume overload, in an attempt to maintain sodium and fluid homeostasis. Normal levels are < 100 pg/mL. Decompensated heart failure is associated with levels of 700 to > 1300 pg/mL.14 High normal values are seen in the elderly. Levels of BNP are elevated in chronic heart failure and correlate with severity and prognosis. Measurement of norepinephrine, renin, angiotensin II, aldosterone, vasopressin, endothelin, and cytokine levels are not recommended for routine patient management.
The echocardiogram is the single most useful test in assessing the heart failure patient. The study should document left ventricular size, hypertrophy, and ejection fraction in addition to visualizing valvular abnormalities. An echo study also provides an estimate of the right ventricular systolic pressure, size, and contractility. In experienced hands, Doppler echocardiography provides a fairly accurate assessment of diastolic dysfunction. Transesophageal echocardiography (TEE) is not recommended for routine assessment of the heart failure patient but may be useful in patients with an inadequate echo window, complicated valvular disease, prosthetic valves, or suspected intracardiac thrombi. Stress or dobutamine echocardiograms may be performed to screen for myocardial ischemia.
Cardiac catheterization is indicated in many patients. Right heart catheterization may clarify perplexing clinical situations by determining intracardiac pressures and cardiac output, thereby providing direction for tailored therapy. Left heart catheterization defines coronary anatomy and determines whether ischemic heart disease is the cause of heart failure. The clinician should have a low threshold for requesting catheterization studies to exclude coronary artery disease and provide hemodynamic information, because the findings may offer clues as to interventions that improve prognosis.
Metabolic stress testing (cardiopulmonary exercise testing), usually performed at larger centers, provides objective data on the level of functional impairment. Maximal oxygen consumption (peak VO2) > 25 mL/kg/min is normal for middle-aged adults. Maximal oxygen consumption < 14 mL/kg/min indicates severe functional impairment and usually is the threshold for considering cardiac transplantation as a management option. The cardiopulmonary exercise test may also be used to differentiate cardiac from pulmonary impairment in patients with unexplained dyspnea.
Pulmonary function tests may be obtained to assess patients with dyspnea. Often, restrictive ventilatory impairment is seen in heart failure. Sleep apnea is common in heart failure, and polysomnography should be considered in patients with a history of nocturnal breathing difficulties and sleep disturbances.
Ambulatory Holter monitoring frequently detects ventricular arrhythmias in the heart failure population. These arrhythmias are nonspecific, but may be predictors of severe heart failure and subsequent mortality. In most cases, Holter monitoring will not identify candidates for antiarrhythmic or defibrillator (ICD) therapy. Sustained ventricular tachycardia, when detected, may indicate the need for an implantable defibrillator.
Management
The management of heart failure is challenging, because counseling, pharmacotherapy, procedures, devices, and rehabilitation are required. Specific aims of treatment are to prevent cardiac decompensation, slow progression of the disease, improve quality of life, prevent complications, and prolong survival. Patient education is important for successful heart failure management. Patients should be instructed to follow a low-sodium diet (2-3 g sodium daily) and restrict fluids (2 L daily). Overweight patients are encouraged to achieve ideal body weight. Heart failure patients should record their weights daily and keep a diary for review. Home blood pressure monitoring is useful in many patients. Cigarette smoking and excess alcohol consumption should be discouraged. An exercise program of mild to moderate intensity improves well being and functional status. Cardiac rehabilitation is helpful when available. Patients are instructed to recognize signs and symptoms of worsening heart failure and seek help if these develop. Close follow-up appointments with monitoring of compliance reduce hospitalizations. Patients should avoid taking nonsteroidal antiinflammatory drugs (NSAIDs), insulin-sensitizing TZD agents, decongestants, and herbal remedies. Because heart failure is a complex syndrome, successful management involves several strategies in combination. A heart failure management program with its team approach provides multiple services, reduces costs, and prevents hospitalizations.15
The new ACC/AHA guidelines emphasize measures to prevent or delay the onset of heart failure and also discuss management of the end-stage patient. For patients with stage A heart failure (at risk but no structural abnormalities), treatment of hypertension, diabetes, coronary artery disease, and avoidance of cardiac toxins is important. For patients with stage D heart failure (end stage with refractory symptoms), a discussion about aggressive high-tech management options (transplant, LVAD, investigational therapies) vs end-of-life care should be conducted with the patient and family.
Drug Therapy For Long-term Management
Angiotensin-converting Enzyme (ACE) Inhibitors. ACE inhibitors probably are the most important drugs used in the management of heart failure.16 Multiple randomized, controlled trials have reported an overall 20% improvement in mortality for all stages of heart failure, as well as improvement in symptoms, exercise tolerance, quality of life, and left ventricular ejection fraction.17-20 ACE inhibitors reduce the risk of hospitalizations and emergency department visits. These agents inhibit ACE, blocking the conversion of angiotensin I to angiotensin II, thereby reducing the detrimental effects of this hormone. ACE inhibitors also prevent the degradation of bradykinin, augmenting kinin-meditated prostaglandin synthesis and vasodilation. Other actions of ACE inhibitors include antihypertensive, anti-inflammatory, anti-fibrotic, reverse remodeling, and renal protective effects. ACE inhibitors should be initiated at low doses and titrated to achieve target doses or the maximally tolerated dose (see Table 3 below).21
| Table 3. ACE Inhibitor Dosing Table | |||
| Agent |
Initial
Dose (mg)
|
Target
Dose (mg)
|
Frequency
|
|
Captopril* (Capoten) |
6.25 |
50 |
TID |
| Enalapril* (Vasotec) |
2.5
|
20
|
BID
|
| Lisinopril* (Prinivil, Zestril) |
2.5
|
40
|
QD
|
| Ramipril** (Altace) |
1.25
|
5
|
BID
|
| Quinapril* (Accupril) |
5
|
20
|
BID
|
| Fosinopril* (Monopril) |
2.5
|
20
|
BID
|
| Benazepril* (Lotensin) |
2.5
|
20
|
BID
|
| Trandolapril** (Mavik) |
1
|
4
|
QD
|
| *
FDA approved for heart failure ** FDA approved for post-myocardial infarction heart failure |
|||
The benefits of ACE inhibitors appear to be a class effect, shared among many agents. Six ACE inhibitors currently are FDA approved for heart failure treatment: captopril, enalapril, lisinopril, quinapril, fosinopril, and benazepril. Ramipril and trandolapril are approved for heart failure following myocardial infarction. The main side effect of ACE inhibitors, intractable cough, occurs in 10-20% of patients and may necessitate substitution of an angiotensin receptor blocker or hydralazine/nitrate. ACE inhibitor cough is persistent, nonpositional and nonproductive. Before stopping an ACE inhibitor, the clinician should be certain that the ACE cough is the correct diagnosis. Other side effects of ACE inhibitors include hypotension, hyperkalemia, renal dysfunction, angioedema, and agranulocytosis. ACE inhibitors should not be prescribed to patients with acute renal failure, a history of angioedema, or during pregnancy. ACE inhibitors must be used cautiously in patients with relatively low blood pressure, creatinine > 3, renal artery stenosis, or high normal serum potassium levels. All patients with systolic or diastolic heart failure (or asymptomatic LV dysfunction) should be treated with an ACE inhibitor.
Angiotensin Receptor Blockers (ARBs). These agents block the effects of angiotensin II at the tissue level.22 They are useful alternative drugs in heart failure therapeutics for patients who are ACE intolerant because of cough or angioedema (see Table 4 below).23 These agents probably are not as effective as ACE inhibitors in heart failure but have fewer side effects.24 ARBs are not associated with cough and may cause renal dysfunction or hyperkalemia. The benefits of "add-on" therapy are uncertain. When ARBs are added to either an ACE inhibitor or a beta blocker, patients may experience improvement in morbidity but not mortality.25 When ARBs are added to an ACE inhibitor plus a beta-blocker, outcomes may worsen.
| Table 4. Angiotensin Receptor Blocker Dosing Table | ||
| Agent |
Initial
Dose (mg)
|
Maximal
Dose (mg)
|
|
Valsartan (Diovan)* |
80 |
320 |
| Candesartan (Atacand) |
16
|
32
|
| Losartan (Cozaar) |
25
|
100
|
| Irbesartan (Avapro) |
75
|
300
|
| Telmisartan (Micardis) |
40
|
80
|
| Eprosartan (Tevetan) |
400
|
800
|
| Olmesartan (Benicar) |
20
|
40
|
| * FDA approved for heart failure in ACE intolerant patients | ||
Hydralazine and Nitrates. These agents are arterial and venous dilators, respectively, and together they reduce afterload and preload. In addition, hydralazine has antioxidant properties, and nitrates inhibit growth and remodeling. Because multiple doses are required throughout the day, patient compliance with these drugs is difficult (see Table 5 below). This combination of vasodilators is inferior to ACE inhibitors in terms of survival, but they may actually improve ejection fraction and exercise tolerance more than ACE inhibitors. Hydralazine/nitrates may be added to ACE inhibitors for additional hemodynamic benefit. Hydralazine/nitrates in combination are particularly useful in ACE-intolerant patients, especially those with renal dysfunction or hyperkalemia.
| Table 5. Other Heart Failure Drugs | |||
| Agent |
Initial
Dose
|
Maximal
Dose
|
Guidelines
|
|
Digoxin |
0.125 mg QD |
0.25 mg QD |
Reduce dose in renal dysfunction and in patients on amiodarone |
| Hydralazine |
25
mg QID
|
100
mg QID
|
Use with nitrates |
| Isosorbide |
20
mg TID
|
80
mg TID
|
Also useful for angina pectoris |
Beta-Blockers. Large, randomized control trials have shown that the beta-blockers carvedilol, long-acting metoprolol, and bisoprolol improve survival and symptoms in heart failure patients.26-30 This is not a class effect; other beta-blockers do not have FDA approval for treating heart failure. Beta-blockers have antihypertensive, antiarrhythmic, anti-ischemic, and negative chronotropic properties. Beta-blockers modulate neurohormones, restore beta receptor function, and reverse remodeling.31 Beta-blockers improve ejection fraction, quality of life, exercise capacity, and survival, in addition to reducing blood pressure, ischemia, and arrhythmias and hospitalizations.
Beta-blockers may be prescribed to euvolemic patients with left ventricular dysfunction and any class of heart failure.32 Beta-blockers are initiated at low doses and slowly titrated to target levels over weeks to months (see Table 6 below). Initially, blocking sympathetic stimulation may worsen left ventricular ejection fraction and symptoms, but after several months, ejection fraction and symptoms improve. Side effects from beta-blockers include hypotension, lightheadedness, fluid retention, cardiac decompensation, and bradycardia. Caution must be used in prescribing beta-blockers to patients with reactive airway disease, heart block, sick sinus syndrome, hyponatremia, or hypotension.
| Table 6. Beta Blocker Dosing Table | ||
| Beta Blocker |
Initial
Dose
|
Target
Dose
|
|
Carvedilol* (Coreg) |
3.125 mg BID |
50
mg BID if > 75 kg |
| Metoprolol * succinate (Toprol XL) |
12.5
mg QD
|
200
mg QD
|
| Metaprolol tartrate (Lopressor) |
12.5
mg BID
|
50
mg TID
|
| Bisoprolol* (Zebeta) |
2.5
mg QD
|
10
mg QD
|
| * FDA approved for heart failure | ||
Diuretics. Most patients with heart failure require diuretics to control symptoms of congestion. Diuretics have not been shown to improve survival but are important for success with other drugs, especially beta-blockers. Diuretics inhibit sodium resorption at various sites in the renal tubules.33 Loop diuretics (furosemide, bumetanide, and torsemide) inhibit sodium resorption in the thick ascending segment in the loop of Henle. Thiazides, including metolazone, inhibit sodium resorption in the distal convoluted tubule. Spironolactone acts in the collecting duct. Intravenous diuretics often are the first drugs given to patients with acute pulmonary edema, providing rapid symptomatic relief. Intravenous diuretics are used to treat hospitalized patients with fluid overload. Continuous infusion of loop diuretics may be needed for volume overloaded patients with diuretic resistance. Chronically, the dosing of diuretics requires careful attention to avoid intravascular volume contraction, while providing adequate drug effect to achieve euvolemia (see Table 7 below). Diuretics should not be prescribed as monotherapy, and diuretic doses should be reduced as patients achieve compensation. In severe heart failure, a loop diuretic may be given in combination with a thiazide such as metolazone to promote diuresis. A flexible, patient-guided diuretic regimen is recommended for chronic outpatient management of heart failure. Diuretics side effects include hyponatremia, hypokalemia, hypomagnesemia, hypotension, azotemia, and neurohormonal activation.
| Table 7. Diuretic Dosing Table | ||||
| Generic Name | Trade Name | Class |
Initial
Dose (mg)
|
Special Considerations |
|
Furosemide
|
Lasix |
Loop |
20 |
Can be given intravenously. PO equivalent twice IV dose. |
| Bumetanide | Bumex | Loop |
0.5
|
Good oral bioavailability. Can be given intravenously.Oral and IV dose the same. |
| Torsemide | Demadex | Loop |
5-10
|
Best oral availability. |
| Ethacrynic acid | Edecrin | Loop |
50
|
Only diuretic with no sulfhydryl group. Used if allergic to furosemide. |
| Hydrochlorothiazide | Hydrodiuril | Thiazide |
12.5
|
Weak diuretic, used mainly for hypertension. |
| Metolazone | Zaroxolyn | Thiazide |
2.5
|
Give 1/2 hour before furosemide. Only available orally. High risk of hypokalemia. |
| Spironolactone | Aldactone | K+ sparing |
12.5
|
Weak diuretic. Risk of hyperkalemia. Avoid in patients with renal failure. Gynecomastia in men. Only available orally. |
Digoxin. Digoxin is indicated for the treatment of symptomatic heart failure (NYHA FC 2-4). It improves exercise capacity and ejection fraction and prevents decompensation and hospitalizations but has a neutral effect on mortality.34 Digoxin has beneficial effects in heart failure regardless of the ejection fraction. Digoxin is classified as a neurohormonal modulating agent.35 It inhibits the enzyme Na+/K+ ATPase. In the heart, this shifts calcium into the myocyte, enhancing contractility. In the central nervous system, this effect reduces sympathetic outflow. In the kidney, it decreases the release of renin. Digoxin also slows conduction through the AV node, controlling ventricular rate in atrial fibrillation. Digoxin has a low therapeutic-toxic range and a potential for drug toxicity. Use a low dose of digoxin (0.125 mg daily) in most patients, maintaining a serum digoxin level < 1 mg/dL. Reduce the dose with renal dysfunction and with concomitant use of amiodarone (see Table 5).
Aldosterone Antagonists. Aldosterone is secreted by the adrenal cortex in response to angiotensin II.36 It promotes sodium and water retention with potassium and magnesium loss. Aldosterone activates the sympathetic and inhibits the parasympathetic nervous system. It causes myocardial fibrosis and remodeling. Spironolactone, a weak diuretic and aldosterone antagonist, decreases mortality and hospitalizations in patients with severe heart failure.37 This agent is indicated in NYHA Class III and IV patients with severe heart failure. Prescribe it at low doses (12.5-25 mg daily), and monitor serum potassium levels regularly. Its major side effects are hyperkalemia and gynecomastia. Spironolactone should be avoided in patients with serum creatinine > 2.0 mg/dL or potassium > 5.3 mmo/L.
Calcium Channel Antagonists. Short-acting calcium channel blockers are contraindicated in heart failure because they depress left ventricular function, worsen symptoms, activate neurohormones, and increase the risk of death. Amlodipine has a neutral effect on cardiac function and mortality.38 It may be prescribed adjunctively to treat angina pectoris or hypertension associated with heart failure. Long-acting calcium channel blockers may be useful for treating some patients with diastolic heart failure with normal left ventricular ejection fraction, but objective data are lacking.
Antiarrhythmic Drugs. Amiodarone is the preferred antiarrhythmic agent for patients with heart failure and low ejection fraction. It may be used to manage atrial fibrillation or high-grade ventricular arrhythmias. Class IA agents such as procainamide, quinidine, and disopyramide should be avoided. Beta-blockers, which have multiple beneficial effects in heart failure, also have antiarrhythmic properties. Implantable defibrillators are superior to drug therapy in preventing sudden cardiac death.
Anticoagulation. The risk of thromboembolic events in heart failure ranges from 1-3% annually. The prophylactic use of warfarin in these patients is controversial based on risk/benefit ratio. Specific indications for warfarin anticoagulation in heart failure include atrial fibrillation, left ventricular thrombus, left ventricular aneurysm, hypercoagulable state, history of prior thromboembolism, and patent foramen ovale. Aspirin has beneficial effects in patients with ischemic cardiomyopathy; no evidence supports its use in patients with normal coronary arteries. Clinical trials are now under way to determine whether anticoagulant agents should be given to all patients with heart failure.
Other Therapies. Statins are recommended for patients with ischemic cardiomyopathy because of multiple benefits. Dietary supplements such as coenzyme Q-10, carotene, antioxidants, thyroid hormone, and growth hormone are not recommended. Exercise training and cardiac rehabilitation improve the clinical status in heart failure patients. Diagnosis and treatment of sleep apnea is an important adjunctive measure and may help reduce elevated pulmonary artery pressures. Treatment of chronic anemia improves functional status. Heart failure management programs, guided by nurse specialists, improve quality of life, reduce hospital admissions, and decrease heart failure costs.
Intravenous Vasodilators for Decompensated Heart Failure (see Table 8 below)
| Table 8. Intravenous Agents Used in Heart Failure | ||
| Drug | Dose | Special Considerations |
|
Nitroprusside |
10-500 mcg/min |
Thiocyanate accumulation in renal failure; may provoke ischemia by coronary steal; vasodilator; ICU only. |
| Nesiritide | 2 mcg/kg/bolus then 0.01 mcg/kg/min | Fixed weight-based dose; vasodilator; occasional hypotension. |
| Nitroglycerin | 10-500 mcg/min | Anti-ischemic, vasodilator; limited by vascular headache; hypotension; tolerance develops rapidly. |
| Dobutamine | 2-20 mcg/kg/min | Beta receptor agonist; pro-arrhythmic; ^HR; ^ Ishemia. |
| Milrinone | 0.25-0.75 mcg/kg/min | Phosphodiesterase inhibitor; vasodilator; may improve pulmonary hypertension; used in patients taking beta blockers; pro-arrhythmic. |
Nitroprusside. Sodium nitroprusside is a potent short-term arterial and venous vasodilator metabolized by the liver to nitric oxide and cyanide. This afterload reducing agent is to treat acutely decompensated heart failure in patients with adequate systemic blood pressure. It is administered by continuous infusion in an ICU setting; invasive hemodynamic monitoring is required for safe use (see Table 8 above). It should be avoided in patients with active ischemia because it may cause coronary steal syndrome. It is associated with rebound worsening hemodynamic effects when the infusion is discontinued. During the infusion, patients should be optimized on oral vasodilator regimen (ACE inhibitors, nitrates/hydralazine) for long-term benefits. Prolonged infusions, especially in patients with renal dysfunction, may be associated with thiocyanate toxicity.
Nesiritide. Nesiritide, synthetic B-type natriuretic peptide, is a balanced venous and arterial vasodilator with modest natriuretic effects.39 Nesiritide may be used to treat patients with decompensated heart failure who have dyspnea at rest or with minimal activities associated with fluid overload. It may be initiated in the emergency department or in the hospital. It normalizes hemodynamics, rapidly improves heart failure symptoms, and promotes diuresis. Nesiritide acts via specialized natriuretic peptide receptors on the cell surface of smooth and endothelial cells. Its primary action is smooth muscle cell relaxation and vasodilation. The onset of action occurs within 15 minutes and the half-life is 18 minutes. Nesiritide is administered by bolus followed by a continuous infusion (see Table 8). In most cases, invasive hemodynamic monitoring is not required. However, telemetry monitoring, blood pressure recordings, and electrolyte determinations are mandatory. The usual length of infustion is 1-2 days. During the infusion, intravenous diuretics are administered, and vasodilators are up-titrated.
Nitroglycerin. Intravenous nitroglycerin is a venous vasodilator at low doses and an arterial vasodilator at high doses. It reduces ventricular preload and improves myocardial ischemia. Nitroglycerin is started at low doses and titrated upward to achieve hemodynamic and clinical benefits (see Table 8). Its effectiveness is limited by tachyphylaxis, which develops within hours after initiation of therapy. Headache occurs in 20% and hypotension in 5% of patients treated with this agent.
Intravenous Inotropic Agents for Hypotension and Low Cardiac Output
Dobutamine. Dobutamine is an intravenous inotropic agent that acts directly on cardiac beta receptors.40 It increases adenylate cyclase activity, converting ATP to cyclic AMP, which releases calcium from the sarcoplasmic reticulum and leads to enhanced contractility. Dobutamine increases cardiac output, heart rate, and myocardial oxygen consumption. It has the potential of provoking arrhythmias and aggravating ischemia. Dobutamine may be administered by continuous infusion in a hospital setting for hypotensive or low output heart failure (see Table 8). The onset of action is 1-2 minutes and the half-life is 2 minutes. Continuous outpatient dobutamine infusions sometimes are administered as palliative therapy to improve symptoms and decrease hospitalizations in patients with end-stage heart failure, but survival may be shortened with this therapy. Intermittent outpatient infusions are not recommended.
Milrinone. Milrinone, a phosphodiesterase III inhibitor, is a positive inotropic and vasodilating agent. Milrinone prevents the degradation of cyclic AMP, the second messenger for improving contractility. Milrinone may be more potent as a pulmonary artery vasodilator than dobutamine. It has minimal chronotropic properties but may increase the risk for cardiac arrhythmias. Milrinone is infused for hypotensive or low-output heart failure (see Table 8). A recent study showed that routine inpatient milrinone infusions for decompensated heart failure were associated with arrhythmias and hypotension but did not reduce length of stay.41 Milrinone (or dobutamine) may stabilize patients waiting in the hospital for cardiac transplantation. Milrinone occasionally is given as an outpatient continuous infusion for palliation of end-stage heart failure. Intermittent outpatient milrinone infusions are not recommended.
Dopamine. Dopamine is a sympathomimetic amine and the immediate precursor of norepinephrine. It exerts dose-dependent physiologic effects when administered intravenously to heart failure patients.42 At low doses, dopamine activates dopaminergic receptors in the mesenteric arteries and kidneys, producing renal vasodilation. At medium doses, it increases the release of norepinephrine from sympathetic neurons, stimulating cardiac beta-receptors and producing a positive inotropic effect. At high doses, it activates alpha-receptors in the peripheral vasculature causing vasoconstriction (see Table 8). In most centers, dopamine has been replaced by dobutamine and milrinone. It is controversial whether "renal dose dopamine" has any benefit; clinical trial data failed to show a renal protective effect with this agent.
Electronic Devices for Heart Failure
Biventricular Pacing. Conduction disturbances occur in a third of patients with advanced heart failure, usually left bundle branch block. The QRS widens as heart failure progresses, and patients with the widest QRS have the poorest prognosis. A wide QRS usually reflects left ventricular dyssynchrony, which causes inefficient contractility, low cardiac output, decreased ejection fraction, prolonged mitral regurgitation, and cardiac remodeling. Biventricular pacing, also known as resynchronization therapy, uses a 3-lead pacing system consisting of a right atrial lead, a right ventricular lead, and a left ventricular lead placed via the coronary sinus into a left lateral cardiac vein.43 Left ventricular lead placement, technically difficult and tedious, has a 10% failure rate. Indications for biventricular pacing are symptomatic heart failure (NYHA FC III-IV), despite a good medical regimen and a wide QRS (> 130 ms). The pacemaker is synchronized by echocardiography to improve contractility. Benefits of resynchronization therapy include improvement in left ventricular ejection fraction, cardiac output, mitral regurgitation, quality of life, exercise tolerance, NYHA FC, and survival. Approximately 67% of patients treated with biventricular pacing experience improvement in heart failure symptoms. However, it is difficult if not impossible to identify patients prospectively who will benefit from this therapy.
Implantable Defibrillators. Approximately half of all heart failure patients die suddenly. Placement of a defibrillator is indicated for survivors of a cardiac arrest, sustained ventricular tachycardia, inducible sustained ventricular tachycardia in the electrophysiology laboratory in patients with an ischemic CM and an EF < 35%, and post-myocardial infarction patients with low ejection fraction.44-45 It is not known whether ICD placement should be recommended for all patients with heart failure and low ejection fraction. Devices are superior to antiarrhythmic drugs in preventing sudden death.
Surgical Therapies
Bypass surgery, valve repair, and ventricular reconstruction. Screening for underlying coronary artery disease is an important aspect of heart failure management. Physicians should have a low threshold for performing cardiac catheterization in this population. If coronary artery disease is detected and myocardial ischemia or hibernation is present, coronary artery bypass surgery or percutaneous coronary intervention (stent/angioplasty) should be performed if suitable target vessels are present. Although surgery is associated with higher risk in patients with low ejection fraction, improvement in left ventricular function and heart failure symptoms may occur. Following revascularization, these patients should continue with a comprehensive heart failure medical management program, including ICD implantation if LVEF is < 35%.
In some patients, coronary arteries are normal but severe mitral or tricuspid valvular regurgitation is present. In experienced centers, these patients can successfully undergo valve repair or replacement and have improvement in heart failure symptoms. In patients with chronic atrial fibrillation, this approach may include a maze procedure to restore sinus rhythm. Medical therapy should be continued postoperatively in all instances.
Left ventricular reconstruction is performed in selected centers for patients with ischemic cardiomyopathy.46 Surgical repair consists of resection of left ventricular aneurysm or scar, followed by reconstruction of the left ventricle to change its shape from spherical to elliptical. The mitral and tricuspid valves are repaired and coronary arteries bypassed. Often a left ventricular pacing lead is attached at the time of surgery and used for resynchronization pacing if the patient remains symptomatic postoperatively. These patients also require continuation of medical therapy after surgical reconstruction.
Left Ventricular Assist Devices (LVADs). Left ventricular assist devices are mechanical pumps used to support the failing heart in patients with cardiogenic shock.47 These pumps are electrically powered and are implanted surgically. Several LVADs are FDA approved for clinical use: HeartMate, Novacor, Thoratec, and Abiomed. The HeartMate and the Novacor are implanted in the abdomen and connected via an external cable to an electrical power source. The inflow cannula is attached to the apex of the left ventricle and separated from the pumping chamber by a bioprosthetic valve. An outflow cannula connects the pumping chamber with the aorta. These devices provide pulsatile blood flow at a rate of 4-10 L/min. The Thoratec and the Abiomed are external devices connected via large cannulae to the circulatory system. The Abiomed often is used as a means of stabilizing patients with cardiogenic shock following cardiac surgery or acute MI. The Thoratec device provides left ventricular and right ventricular support via external pumping chambers. In general, an LVAD is used as a "bridge to cardiac transplantation" in suitable individuals. Very few patients with dilated cardiomyopathy recover sufficient cardiac function to allow explantation of an LVAD. These devices are limited by a high infection rate, thromboembolic complications, and mechanical failures. Smaller LVADs with continuous nonpulsatile flow are undergoing clinical trials.
Cardiac Transplantation. Cardiac transplantation is a management option for a very small subset of heart failure patients.48 In general, these patients are younger and have disabling heart failure symptoms despite maximal medical therapy. Patients are otherwise healthy except for heart failure but have poor estimated survival. The number of donor hearts in the United States is steadily decreasing despite altruistic efforts to increase organ donation. Approximately 2000 heart transplants are performed nationwide annually.49 Contraindications to cardiac transplantation include recent malignancy, morbid obesity, systemic disease limiting survival or rehabilitation, active smoking, medical noncompliance, renal or hepatic failure, active infection, or recent pulmonary infarction. Patients referred to a heart transplant center undergo extensive medical and psychosocial evaluation. If selected for listing, the patients are prioritized according to severity of illness based on United Network for Organ Sharing (UNOS) criteria. Status I patients require inotropic or mechanical support for the failing heart. Status II patients remain at home on oral medical therapy. Mortality associated with cardiac transplantation is approximately 10% during the first year and 50% by year 10. Complications include infection, rejection, allograft coronary artery disease, renal dysfunction, and malignancy. All patients are maintained on multiple immunosuppressive medications. Complications frequently are related to over- or under-immunosuppression.
Clinical Decision Making
|
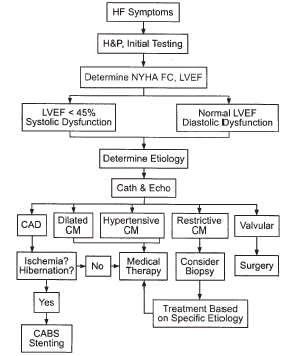
Figure
1 . Heart Failure Diagnostic Algorithm
|
 |
|
Figure
2 . Heart Failure Management Algorithm
|
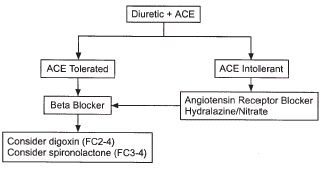 |
Figure 1 (above) provides a diagnostic algorithm for evaluating patients with heart failure. The echocardiogram and cardiac catheterization are the most important diagnostic techniques for determining etiology and structural abnormalities. Figure 2 (above) is a management algorithm in which a diuretic and an ACE inhibitor are given as initial therapy. The clinician is provided with guidelines for additional pharmacological agents. Indications for hospitalization include severe dyspnea at rest or with minimal activities, marked fluid retention and weight gain, worsening renal or hepatic function, hypotension, cardiac arrhythmias or syncope, unstable angina pectoris, or need for IV diuretics or vasodilators. Indications for referral to a tertiary heart failure center include NYHA FC 3-4 symptoms despite maximally tolerated medications, complex cases with frequent decompensations, possible candidates for biventricular pacing, surgical therapies, cardiac transplantation, investigational therapies, or palliative home inotropic therapy.
Future Directions
The outcomes of several recent clinical trials in heart failure have favored device and surgical therapies over conventional pharmacological approaches. However, several new classes of investigational drugs are undergoing clinical trial evaluation: oral inotropics, newer diuretics, vasopressin antagonists, and aldosterone antagonists.50 Clinical trials of newer LVADs and surgical devices (cardiac support and shape devices) currently are under way.
Summary
Heart failure has become a common clinical problem associated with high cost, morbidity, and mortality. In describing heart failure, it is important to determine the etiology, NYHA functional class, systolic vs diastolic dysfunction, and acute vs chronic decompensation. All patients with left ventricular dysfunction should be treated with an ACE inhibitor initially and then a beta-blocker. Although diuretics do not improve mortality, they are necessary for symptom relief in most patients. Digoxin may be added to the regimen for symptomatic heart failure, and an aldosterone antagonist is used for patients with severe heart failure. Biventricular pacing and cardiac defibrillators should be considered in selected cases. Surgical therapies may help to reverse heart failure symptoms, and surgical management should be considered as an important adjunctive measure.
References
1. National Heart Lung and Blood Institute. Morbidity and Mortality; 2002 Chart Book on Cardiovascular, Lung, and Blood Diseases. Bethesda, MD: 2002.
2. American Heart Association. 2001 Heart and Stroke Update. Dallas, Texas: 2000.
3. ACC/AHA Task Force On Practice Guidelines: Guidelines for the evaluation and management of heart failure. Circulation. 1995;92:2764-2784.
4. Heart Failure Society of America. HFSA Guidelines for management of patients with heart failure caused by left ventricular systolic dysfunction—Pharmacological approaches. J Cardiac Fail. 1999;5:357-382.
5. Advisory Council to Improve Outcomes Nationwide in Heart Failure (Action HF). Consensus recommendations for heart failure. Am J Cardiol. 1999;83:1A-38A.
6. Hunt SA, et al. ACC/AHA Guidelines for the evaluation and management of chronic heart failure in the adult: Executive summary: A report of the American College of Cardiology/American Heart Association Task Force on practice guidelines (committee to revise the 1995 guidelines for the evaluation and management of heart failure). Circulation. 2001;104:2996-3007.
7. Gomberg-Maitland M, Baran DA, Fuster V. Treatment of congestive heart failure: Guidelines for the primary care physician and the heart failure specialist. Arch Intern Med. 2001;161: 342-352.
8. Task Force for the Diagnosis and Treatment of Chronic Heart Failure, European Society of Cardiology. Guidelines for the diagnosis and treatment of chronic heart failure. Eur Heart J. 2001; 22:1527-1560.
9. Working Group Report. How to diagnose diastolic heart failure. European study of diastolic heart failure. Eur Heart J. 1998;19:990-1003.
10. Chen HH, et al. Diastolic heart failure in the community: Clinical profile, natural history, therapy, and impact of proposed diagnostic criteria. J Cardiac Fail. 2002;8:279-287.
11. Swedberg K. Importance of neuroendocrine activation in chronic heart failure. Impact of treatment strategies. Eur J Heart Fail. 2000;2:229-233.
12. Cohn J. Sympathetic nervous system in heart failure. Circulation. 2002;106:2417-2418.
13. Maisel A. B-type natriuretic peptide levels: A potential novel "white count" for congestive heart failure. J Cardiac Fail. 2001; 7:183-193.
14. Dao Q, et al. Utility of B-type natriuretic peptide in the diagnosis of congestive heart failure in an urgent-care setting. J Am Coll Cardiol. 2001;37:379-385.
15. Fonarow GC, et al. Impact of a comprehensive heart failure management program on hospital readmission and functional status of patients with advanced heart failure. J Am Coll Cardiol. 1997;30: 725-732.
16. Brown NJ, Vaughn DE. Angiotensin-converting enzyme inhibitors. Circulation. 1998;97:1411-1420.
17. Consensus Trial Study Group. Effects of enalapril on mortality in severe congestive heart failure. Results of the cooperative North Scandinavian enalapril survival study (CONSENSUS). N Engl J Med. 1987;316:1429-1435.
18. Captopril-Digoxin Multicenter Research Group. Comparative effects of captopril and digoxin in patients with mild to moderate heart failure. JAMA. 1988;259:539-544.
19. Cohn JN, et al. A comparison of enalapril with hydralazine—isosorbide dinitrate in the treatment of congestive heart failure. N Engl J Med. 1991;325:303-310.
20. The SOLVD Investigators. Effect of enalapril on survival of patients with reduced left ventricular ejection fractions and congestive heart failure. N Engl J Med. 1991;325:293-302.
21. Packer M, et al. Comparative effects of low and high doses of the angiotensin-converting enzyme inhibitor, lisinopril, on morbidity and mortality in chronic heart failure. ATLAS Study Group. Circulation. 1999;100:2312-2318.
22. Burnier M. Angiotensin II type 1 receptor blockers. Circulation. 2001;103:904-912.
23. Jamali AH, et al. The role of angiotensin receptor blockers in the management of chronic heart failure. Arch Intern Med. 2001; 161:667-672.
24. Jong P, et al. Angiotensin receptor blockers in heart failure: Meta analysis of randomized controlled trials. J Am Coll Cardiol. 2002;39:463-470.
25. Cohn JN, Tognoni G. A randomized trial of the angiotensin-receptor blocker valsartan in chronic heart failure. N Engl J Med. 2001;345:1667-1675.
26. Packer M, et al. The effect of carvedilol on morbidity and mortality in patients with chronic heart failure. N Engl J Med. 1996; 334:1349-1355.
27. MERIT-HF Study Group. Effect of metoprolol CR/XL in chronic heart failure: Metoprolol CR/XL randomized intervention trial in congestive heart failure (MERIT-HF). Lancet. 1999;353: 2001-2007.
28. CIBIS-II Investigators and Committees. The cardiac insufficiency bisoprolol study II (CIBIS-II): A randomized trial. Lancet. 1999; 353:9-13.
29. Packer M, et al. Effect of carvedilol on survival in severe chronic heart failure. N Engl J Med. 2001;344:1651-1658.
30. Brophy JM, Joseph L, Rouleau JL. Beta blockers in congestive heart failure: A Bayesian meta-analysis. Ann Intern Med. 2001; 134:550-560.
31. Bristow MR. Beta-adrenergic receptor blockade in chronic heart failure. Circulation. 2000;101:558-569.
32. Eichhorn EJ. Clinical use of beta-blockers in patients with heart failure. J Cardiac Fail. 2000;6(Suppl 1):40-46.
33. Brater DC. Diuretic therapy. N Engl J Med. 1998;339:387-395.
34. Digitalis Investigation Group. The effect of digoxin on mortality and morbidity in patients with heart failure. N Engl J Med. 1997; 336:525-533.
35. Hauptman PJ, Kelly RA. Digitalis. Circulation. 1999;99: 1265-1270.
36. Weber KT. Aldosterone in congestive heart failure. N Engl J Med. 2001;345:1689-1697.
37. Pitt B, et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. N Engl J Med. 1999; 341:709-717.
38. Packer M, et al. Effect of amlodipine on morbidity and mortality in severe chronic heart failure. N Engl J Med. 1996;335: 1107-1114.
39. Hobbs RE, Mills RM, Young JB. An update on nesiritide for treatment of decompensated heart failure. Exp Opin Invest Drugs. 2001;10:935-942.
40. Felker GM, O’Connor CM. Inotropic therapy for heart failure: An evidence-based approach. Am Heart J. 2001;142:393-401.
41. Cuffe MS, et al. Short-term intravenous milrinone for acute exacerbation of chronic heart failure. A randomized controlled trial. JAMA. 2002;287:1541-1547.
42. Van De Borne P, Somers VK. Dopamine and congestive heart failure: Pharmacology, clinical use, and precautions. Congest Heart Fail. 1999;5:216-221.
43. Leclercq C, Kass DA. Retiming the failing heart: Principles and current status of cardiac resynchronization. J Am Coll Cardiol. 2002;39:194-201.
44. Collob MH, Seger JJ. Current status of the implantable cardioverter-defibrillator. Chest. 2001;119:1210-1221.
45. Moss AJ, et al. Prophylactic implantation of a defibrillator in patients with myocardial infarction and reduced ejection fraction. N Engl J Med. 2002;346:877-883.
46. McCarthy PM, et al. Left ventricular reduction surgery with mitral valve repair. J Heart Lung Transplant. 2000;19:S64-S67.
47. Delgado DH, et al. Mechanical circulatory assistance: State of the art. Circulation. 2002;106:2046-2050.
48. Hunt SA. Current status of cardiac transplantation. JAMA. 1998; 280:1692-1698.
49. Hertz MI, et al. The registry of the International Society for Heart and Lung Transplantation: Nineteenth Official Report—2002. J Heart Lung Transplant. 2002;21:950-970.
50. McMurray J, Pfeffer MA. New therapeutic options in congestive heart failure: Part I. Circulation. 2002;105:2099-2106. Part 2. Circulation. 2002;105:2223-2228.
CME Questions
7. The most common etiology of heart failure is:
a. alcohol abuse.
b. atrial fibrillation.
c. hypertension.
d. coronary artery disease.
8. The pathophysiologic response to heart failure:
a. restores many patients to good health.
b. improves cardiac hemodynamics and heart size.
c. worsens cardiac function and prognosis.
d. is a temporary response to acute decompensation.
9. Most of the expense associated with heart failure:
a. is associated with diagnostic testing.
b. is related to prescription drugs.
c. is due to hospitalizations.
d. is used to finance heart transplants.
10. Current guidelines emphasize the importance of ACE inhibitors and beta-blockers in heart failure management:
a. because diuretics work so well already.
b. because these drugs may help when digoxin fails.
c. because these agents have minimal side effects.
d. because these drugs have proven survival benefits.
11. Which statement about intravenous drugs for heart failure is true?
a. Vasodilators improve hemodynamics quickly.
b. Milrinone and dobutamine are not proarrhythmic.
c. Nitroglycerin has sustained vasodilator effect.
d. Nesiritide improves cardiac output by positive inotropic actions.
12. Approximately half of all heart failure patients die suddenly. The most effective intervention for preventing this complication is:
a. amiodarone therapy.
b. quinidine and digoxin in combination.
c. implantable defibrillators.
d. class 1-C antiarrhythmic agents.
13. Patients with heart failure should be:
a. reassured because transplant is always an option.
b. referred for surgical evaluation if severe mitral regurgitation and atrial fibrillation are present.
c. always managed medically because surgical risk in heart failure patients is prohibitive.
d. treated initially with dietary supplements and herbal remedies.
This article reviews multiple aspects of heart failure, providing direction for workup and management of heart failure patients. All recommendations follow current published guidelines. Unresolved and controversial issues are reviewed, and future therapies are proposed.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
