Black Cohosh: The "Hot" Treatment for Hot Flashes
Black Cohosh: The "Hot" Treatment for Hot Flashes
By Melinda Ring, MD
The top prescription hormone replacement therapy (HRT), Prempro (medroxyprogesterone/conjugated estrogen), suffered a marked decline in popularity following the July 9, 2002, Women’s Health Initiative (WHI) announcement that HRT increased the relative risk of breast cancer and cardiovascular disease. The early halt to the Prempro arm of the WHI study dropped the number of Prempro prescriptions filled weekly from 379,581 to 211,249 in just the first two months.1
This abrupt loss of confidence in HRT stimulated tremendous interest in the already fashionable field of "natural" alternatives for the management of climacteric symptoms. More than any other herbal remedy, black cohosh (Cimicifuga racemosa) shows promise in being an effective and acceptable treatment for vasomotor symptoms.
Historical Perspective
Black cohosh is an indigenous herb of the buttercup (Ranunculaceae) family that produces 3- to 8-foot spikes of bright white flowers. Many of its synonyms refer to its insect-repellent characteristics. (See Table 1 below.)

Native Americans used the herbal roots to assist in labor/parturition and for musculoskeletal complaints and respiratory problems. It was adopted rapidly by early settlers for its sedative and muscle-relaxant effects. In the 1800s the Eclectic medical practitioners, major players in the medical world of their era, endorsed its use for many conditions, especially those related to women’s health.
In the late 19th century, Lydia E. Pinkham’s Vegetable Compound, which contained black cohosh, unicorn root, and fenugreek, was a best-seller for, as the label proclaimed, "all those painful complaints and weaknesses so common to our best female population." Lydia’s family made a fortune with her concoction, peaking in 1925 with annual revenues of $3.8 million. Although black cohosh was the chief "active" ingredient of the vegetable compound, the 36-proof alcohol content may have accounted for some of its renowned "therapeutic" properties, especially during Prohibition.
In the 1930s, herbal use abated in America; however, black cohosh was developed and investigated further in Europe, primarily Germany.
Pharmacology/Mechanism of Action
The black cohosh root contains many substances believed to play an active therapeutic role. Of primary importance are the triterpene glycosides, including actein, 27-deoxyactein, and cimifugoside. The most often studied preparation, Remifemin, is standardized to the 27-deoxyactein content. Other compounds include resins (cimicifugin), caffeic and isoferulic acids, and fukinolic acid.
The mechanism of action remains a mystery. There has been considerable debate about whether black cohosh is a true phytoestrogen: a plant-derived substance exhibiting estrogenic activity. In vitro and in vivo studies to assess estrogenic properties have yielded contradictory results. Sample studies on the estrogenicity of black cohosh, with their rationales and pros and cons are detailed in Table 2.2,4
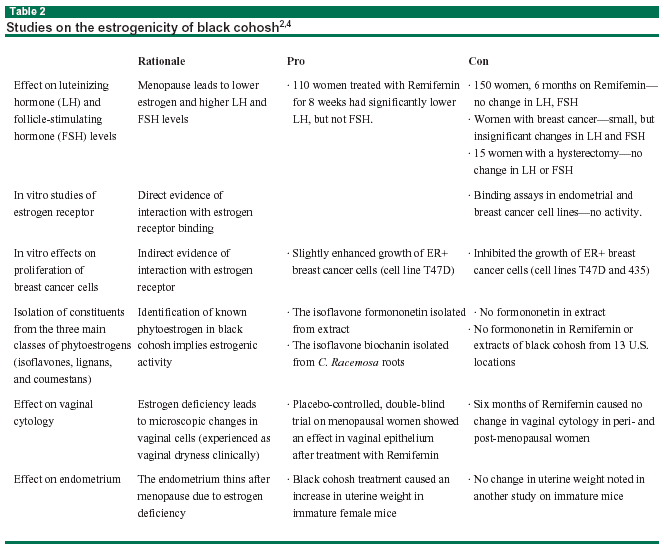
Clinical Trials
Studies on vasomotor symptoms uniformly have recorded improvements in response to placebo in 30-50% of women, calling into question any study that supplies treatment response rates without a control group for comparison. For a study on hot flashes to be truly meaningful, it should include a lead-in period prior to therapy, double-blind and crossover designs, a placebo control, and stringent measures of frequency and severity of hot flashes.3
Kronenberg and Fugh-Berman recently published a review limited to randomized controlled trials (RCTs) for menopausal symptoms.4 Searches of the MEDLINE and AMED (allied and complementary medicine database) yielded four RCTs on black cohosh performed between 1985 and 2001.5-8 (See Table 3 below.)
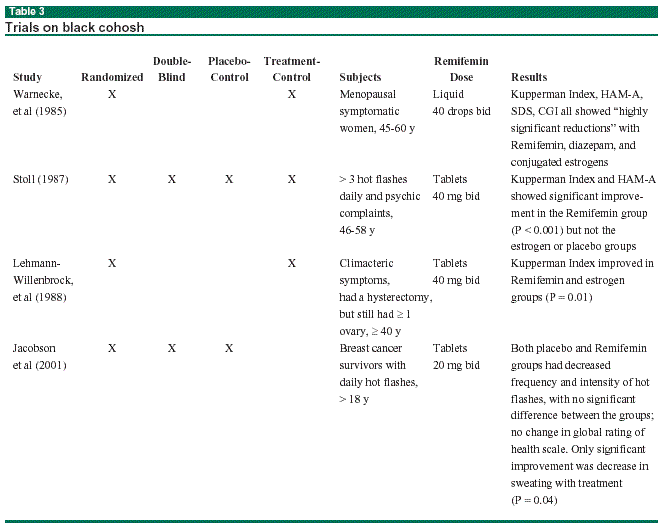
The first three trials were performed in Germany in the 1980s.5-7 Each trial studied 60-80 women with climacteric symptoms, and used validated outcome measures. These scales assess both somatic and psychometric complaints, and include the Kupperman menopausal index, the Self-assessment Depression Scale (SDS), the Hamilton Anxiety Scale (HAM-A), and the Clinical Global Impression Scale (CGI). Significant improvement was noted in all three trials. All studies employed black cohosh extracts standardized to 27-deoxyactein, although there was some variation in formulation and dosing. (See Table 3 above.) The results of the most rigorous of these trials, the Stoll trial, have been questioned because standard estrogen doses did not show an effect on hot flashes over that of placebo (both showed similar reductions).
The most recent study, and the only RCT published in an English language journal, involved a different population.8 Eighty-five women (69 completed the trial) who were breast cancer survivors with daily hot flashes took either placebo or black cohosh for two months. No significant difference in frequency or duration of hot flashes was noted between the two groups. The findings of this study are compromised by the brief duration of the study, and may not apply to menopausal women without breast cancer. The fact that 59 of the women were taking the anti-estrogen tamoxifen also clouds the results.
The effect of black cohosh on menopausal symptoms has been examined through clinical trials involving more than 2,000 women. The vast majority of these trials suffer from some design flaw that limits the utility of the findings, and from variations in the dosing recommendations and extraction techniques of Remifemin, the black cohosh extract used in nearly all clinical trials.
Side Effects, Drug Interactions, and Contraindications
Side effects. A very low incidence of side effects has been noted in clinical trials, primarily frontal headaches and mild gastrointestinal complaints.
Contraindications. Given the inconsistent evidence about black cohosh and phytoestrogen activity, women with a history of estrogen-dependent breast tumors should be advised that the real risk is unclear at this time. The German Commission E and manufacturers of Remifemin do not feel a history of breast cancer is a contraindication to black cohosh use. Black cohosh is contraindicated in pregnancy and lactation, except when used by medical professionals to assist labor.
Drug interactions. The combination of tamoxifen and black cohosh appears to act synergistically in preclinical studies to prevent proliferation of estrogen-dependent breast cancer cells. Concurrent use with HRT is not recommended due to possible potentiation of estrogen effects. No other drug interactions or interference with lab tests have been recorded.
Dosage and Formulations
The most commonly studied formulation of black cohosh is Remifemin. The German herb company Schaper & Brümmer initially manufactured Remifemin in liquid form. Over time the extraction process changed, along with a shift to tablets. Recently, GlaxoSmithKline has been producing the equivalent Remifemin Menopause in the United States.
Each tablet contains 20 mg of dried rhizome (underground stem) and root extract. Patients are advised to take one tablet twice daily (a change from previous manufacturer recommendations to take two equivalent tablets twice daily). Inactive ingredients include cellulose powder, lactose monohydrate, potato starch, magnesium stearate, and peppermint oil. Each 20 mg tablet is standardized to contain 1 mg of 27-deoxyactein.9
Unlike prescription HRT, the beneficial effect of black cohosh is a gradual process. The first therapeutic effects generally are seen within two weeks of treatment initiation, with maximal symptom relief occurring in most women by 8-12 weeks.
Because no long-term studies have been conducted to date, it is recommended that consumption of black cohosh not exceed six consecutive months without physician guidance. If symptoms return after a month-long cessation of therapy, treatment may be resumed for another six months.
Lydia Pinkham’s herbal compound is still available, and has been reformulated with iron, calcium, vitamins E and C, black cohosh, motherwort, dandelion, licorice, Jamaican dogwood, pleurisy root, and gentian (Numark Laboratories, NJ). It no longer has a 36-proof alcohol content.
Other dosage approaches include: dried root/rhizome (0.5-1 g 3-4 times per day); liquid extracts (1.5-3 mL/d of 1:2 extract; 3.5-7 mL/d of 1:5 extract); or tinctures (6-12 mL/d of 1:10 tincture).
Regulatory/Professional Status
In the 1980s, black cohosh was approved by Germany’s Commission E for "neurovegetative complaints of premenstrual, dysmenorrheic, or climacteric origin." In the United States, it is available over the counter as a dietary supplement, although it does not have GRAS (generally regarded as safe) status.
In 2001, the American College of Obstetricians and Gynecologists (ACOG) released a bulletin indicating that black cohosh may be helpful in short-term (less than six months) management of vasomotor symptoms, based on general consensus and expert opinion.10
Current Research
Currently in progress, the National Center for Complementary and Alternative Medicine-funded Phase II study at the University of Illinois College of Medicine is a year-long randomized, double-blind study of 112 women comparing HRT, black cohosh, red clover, and placebo. The Phase I single-dose pharmacokinetic study of these two herbs was implemented to assist in determining dosage requirements. Columbia University also is conducting a 12-month trial to observe the effect of black cohosh on hot flashes, bone metabolism, and cognitive function. The results of these well-designed studies will help elucidate the role black cohosh should play in symptom management of the climacteric.
Conclusion
Although only four RCTs involving black cohosh have been published to date, the three that involved the general perimenopausal population showed significant improvement in vasomotor symptoms compared to placebo. Respected organizations such as ACOG have given a preliminary "seal of approval" for limited duration of use. The utility and safety of black cohosh for women with more complex hormonal environments, such as breast-cancer survivors (who may be taking tamoxifen), are unclear.
Recommendation
For now, black cohosh is an herb with minimal side effects, and at least some suggestion of benefit in the management of vasomotor symptoms in menopause. Recommend it to your patients who are looking for an alternative to HRT, reminding them to use reputable brands and follow the dosing guidelines. Unlike estrogen, black cohosh has no known effect on bone or cardiovascular health. Treatment of women with a history of breast cancer should be individualized.
Dr. Ring is Clinical Training Attending Physician and Coordinator, CAM Curriculum, Internal Medicine Residency Training Program, St. Joseph Hospital, Chicago, IL.
References
1. NDCHealth: A health care information services company.
2. Questions and answers about black cohosh and the symptoms of menopause. Office of Dietary Supplements Web site. Available at: http://ods.od.nih.gov/factsheets/blackcohosh_pf.html. Accessed Feb. 12, 2003.
3. Fitzpatrick LA, Santen RJ. Hot flashes: The old and the new, what is really true? Mayo Clin Proc 2002;77:1155-1158.
4. Kronenberg F, Fugh-Berman A. Complementary and alternative medicine for menopausal symptoms: A review of randomized, controlled trials. Ann Intern Med 2002;137:805-813.
5. Warnecke G. Beeinflussing Klimakterischer Beschwerden durch ein Phytoitherapeutikum: Erfolgreiche Therapie mit Cimicifuga-Monoextrakt. Med Welt 1985;36:871-874.
6. Stoll W. Phytopharmacon influences atrophic vaginal epithelium-double-blind study—Cimicifuga vs. estrogenic substances [in German]. Therapeutikon 1987;1:23-31.
7. Lehmann-Willenbrock E, Riedel HH. Clinical and endocrinologic studies of the treatment of ovarian insufficiency manifestations following hysterectomy with intact adnexa [in German]. Zentralbl Gynakol 1988;110:611-618.
8. Jacobson JS, et al. Randomized trial of black cohosh for the treatment of hot flashes among women with a history of breast cancer. J Clin Oncol 2001;19:2739-2745.
9. GlaxoSmithKline. Remifemin. Available at: www.remifemin.com/professional. Accessed Feb. 12, 2003.
10. ACOG: Use of Botanicals for management of menopausal symptoms. ACOG Pract Bull 2001;28:1-11.
Ring M. Black cohosh: The "hot" treatment for hot flashes. Altern Ther Women's Health 2003;5(3):17-21.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
