Evaluation of Acute Chest Pain: An Update on Potential Future Options
August 25, 2013
Reprints
Evaluation of Acute Chest Pain: An Update on Potential Future Options
Authors:
Kevin S. Barlotta, MD, Associate Professor, Assistant Residency Program Director, Head of Quality Improvement, Department of Emergency Medicine, University of Alabama, Birmingham.
James Booth, MD, Department of Emergency Medicine, University of Alabama, Birmingham.
Peer Reviewer:
Brian Tiffany, MD, PhD, FACEP, Medical Staff President, Chandler Regional Medical Center, Mercy Gilbert Medical Center, Phoenix, AZ.
Ischemic heart disease is a leading cause of death in the United States, making the early recognition or exclusion of acute coronary syndrome (ACS) and acute myocardial infarction (AMI) a major focus of the emergency department (ED) evaluation of chest pain.
The spectrum of ischemic heart disease includes the clinical syndromes of AMI and unstable angina (UA), as well as coronary artery disease (CAD), the pathologic process that causes myocardial ischemia. ACS includes both AMI and UA. AMI is recognized by combining information obtained from the patient’s clinical history and physical exam, electrocardiographic (ECG) findings, changes in cardiac biomarker measurements, and cardiac imaging studies. Identifying the patient with UA is primarily done by the clinical history, in that the ECG and cardiac biomarkers usually do not have diagnostic changes. Identifying CAD in a patient presenting with chest pain usually requires a functional test of myocardial perfusion ("stress test") or imaging the coronary arteries. Identifying both UA and CAD is important, in that the untreated patient is at risk for subsequent cardiovascular events, such as AMI and cardiac arrest. Two new ancillary tests are likely to have growing availability to assist in the identification of CAD and risk stratification of patients with chest pain: high-sensitivity cardiac troponin (hs-cTn) and coronary computed tomographic angiography (CCTA).
Despite their intended utility, questions remain regarding the reliability and predictive value of hs-cTn and CCTA. In this article, we will review the literature regarding these tests and discuss their ability to aid in the diagnosis of ischemic heart disease.
High-sensitivity Cardiac Troponin Assay
Introduction. In 1979, the World Health Organization (WHO) defined AMI by the presence of at least two of three criteria: a clinical history indicative of ischemic heart disease, certain ECG abnormalities, and a rise in the serum concentration of cardiac biomarkers.1 At that time, the available assays for cardiac biomarkers lacked specificity for myocardial necrosis. This position changed drastically with the development of cardiac troponin assays.
Troponin Detection in Skeletal and Cardiac Muscle. Troponin is a calcium-binding protein complex found in both skeletal and cardiac muscle. It is composed of three subunits (troponin C, troponin T, and troponin I) that serve to regulate muscle contraction. Similar to the release of muscle enzymes (creatine kinase), troponins are released into the serum during cell injury or death.
Cardiac Troponin Assays. Cardiac troponins (cTn) are expressed exclusively in the myocardium and contain unique structural features that distinguish them from skeletal muscle. Immunologic assays that utilize specialized monoclonal antibodies can distinguish cTn proteins from troponins of skeletal muscle origin. Cardiac troponin assays are engineered to detect either cardiac troponin I (cTnI) or cardiac troponin T (cTnT) proteins. These assays have demonstrated superior sensitivity and specificity compared to conventional myocardial enzyme tests and have provided clinicians with a powerful diagnostic tool to identify cardiac injury.2-5
The Universal Definition of Acute Myocardial Infarction. In 2012, the third universal definition of myocardial infarction was released by a global task force of international cardiology societies establishing the criteria for diagnosis of AMI. (See Table 1.) These guidelines require a clinical setting consistent with myocardial ischemia in association with the detection of cardiac biomarkers, preferably cardiac troponin (cTn). These recommendations emphasize a growing reliance on cTn to diagnose AMI and a clear relationship between detectable cTn levels and poor patient outcomes.
Table 1: Third Universal Definition of Acute Myocardial Infarction
a.) Detection of a rise and/or fall of biomarker values (preferably cardiac troponin) with at least one value above the 99th percentile upper reference limit (URL) and with at least one of the following:
i. Symptoms of ischemia
ii. New or presumed new significant ST-segment-T wave (ST-T) changes or new left bundle branch block (LBBB)
iii. Development of pathologic Q waves on the ECG
iv. Imaging evidence of new loss of viable myocardium or new regional wall motion abnormality
v. Identification of an intracoronary thrombus by angiography or autopsy
b.) Sudden cardiac death with symptoms suggestive of myocardial ischemia and/or presumed new ischemic ECG changes where death occurred before cardiac markers were obtained or before values would be increased
c.) Elevations in biomarkers after percutaneous coronary intervention (PCI)
d.) Stent thrombosis associated with MI
e.) Elevations in biomarkers in patients following coronary artery bypass
grafting (CABG)
*Adapted from Thygesen et al10
Cardiac Troponin: The Preferred Biomarker and a Diagnostic Threshold. In 2000, cTn was identified as the preferred biomarker for the detection of myocardial cell necrosis.6-8 In addition, a decision limit or diagnostic threshold for AMI was established as an increase in the cTn concentration exceeding the 99th percentile upper reference limit for a reference control group.6,7 This cutoff value is unique for each cTn assay and is determined by measuring basal cTn concentrations in a selected (typically healthy) patient population.9
Analytical Performance and the Importance of Precision. The analytical performance of an assay is typically determined by its limit of detection (LoD) and its imprecision profile, as measured by the co-efficient of variation (CV). The LoD is the lowest measurable cTn concentration for a given assay. The CV is a measure of assay precision that varies with cTn concentration.11 The CV reflects an assay’s ability to distinguish between two values and the ability to detect a significant change in measurement. A CV of 10% or less at a given cutoff is considered to be precise.
Many contemporary cTn assays failed to achieve the recommended CV of 10% or less at the 99th percentile upper reference limit and, therefore, used the lowest cTn value at which the assay achieved a CV of 10% or less (i.e., the recommended diagnostic cutoff concentration). In these circumstances, precision was achieved at the expense of sensitivity, resulting in both a reduction and a delay in the number of patients identified with ACS.12 In contrast, many of the newer cTn assays achieve a CV of 10% or less at the 99th percentile upper reference limit and are considered to have optimal precision.12,13 In summary, the interpretation of cTn concentrations in assessing ACS/AMI will depend on an institution’s chosen cTn assay and its particular analytical specifications.
The Rise and/or Fall of Cardiac Troponin Levels. As assays became more sensitive and precise, earlier detection of cTn levels at lower concentrations gave rise to questions regarding the significance of measurements below the diagnostic threshold from AMI. Although a clear relationship exists between cTn elevations and myocardial cell injury, it became clear that not all cTn detection was indicative of AMI. Initial cTn measurements represent a single assessment in time and cannot reliably exclude ACS in patients with chest pain. As a result, serial sampling of cardiac biomarkers is essential to differentiate non-ischemic, non-ACS cTn elevations and cTn levels indicative of ACS/AMI.4,13,14 The recommendations set forth in the universal definition of AMI require both a temporal "rise and/or fall of cardiac biomarker values" and symptoms of ischemia.
Although the requirement for dynamic changes in cTn concentrations was clearly established in the universal definition of AMI, the magnitude of change was less clear.5,10 Current guidelines advise that a change in cTn concentration of 20% or greater above baseline is indicative of a rising cTn level.15 Although serial monitoring for changes in cTn levels typically occurs at 36 hour intervals, several novel approaches have been introduced in low-risk patients to shorten reassessment time.16
In contrast to low-risk patients, those at high risk for ACS and minimally elevated cTn levels are at substantially higher risk of death and recurrent ischemic events.17 Therefore, high-risk patients with slightly elevated cTn levels might benefit from more aggressive treatment algorithms, including antiplatelet and antithrombotic regimens, as well as revascularization strategies.
Non-ischemic Troponin Elevation. Elevated cTn has been observed in a range of conditions. (See Table 2.) The key difference from AMI is that cTn levels do not rise during the initial 6-12 hours after symptom onset and do not fall over the subsequent few days as seen with AMI. Acute conditions found to elevate cTn levels include congestive heart failure, pulmonary embolism, and sepsis.13,18 In addition, baseline low-level cTn elevations have been described in patients with chronic cardiac conditions and chronic kidney disease.19-23
Table 2: Non-ischemic Causes of Troponin Elevation
• Congestive heart failure
• Renal failure
• Sepsis
• Pulmonary embolism
• Severe respiratory failure
• Severe anemia
• Myocarditis
• Trauma
• Electrical shock
• Ablation procedures
• Stroke
• Subarachnoid hemorrhage
• Malignancy
• Strenuous exercise
• Takosubo (stress) cardiomyopathy
*Adapted from Thygesen et al.10
Ischemic ACS vs. Non-ACS Myocardial Infarction. The clinical syndrome of AMI can be further classified according to the etiology of myocardial ischemia. (See Table 3.) An important distinction is between type 1, which is due to a primary coronary event, and type 2, which is due to mismatch between myocardial oxygen supply and demand. This differentiation of AMI has important implications in guiding appropriate therapeutic intervention.
Table 3: Classification of Myocardial Infarction
• Type 1: Ischemic myocardial necrosis secondary to ACS
• Type 2: Ischemic myocardial necrosis not due to ACS
• Type 3: Sudden cardiac death
• Type 4: Related to PCI or stent thrombosis
• Type 5: Related to CABG
*Adapted from Newby et al.13
While the treatment of type 1 MI can be based on evidence-based guidelines, there is less structure to the treatment strategies for type 2 MI. Instead, a "more individualized approach" is recommended focusing on the underlying condition responsible for the oxygen supply and demand mismatch.5
Chest Pain Evaluation in the ED
Chest pain is a common presenting complaint in the ED, accounting for more than 5 million ED visits annually in the United States.24 As the preferred cardiac biomarker for myocardial necrosis, cTn testing has become an essential component of the diagnostic workup of patients with suspected ACS.
Pre-test Probability and the Importance of Clinical Context
Patients presenting with chest pain to the ED can be broadly divided into two groups: those with high suspicion for ACS and those with less convincing ACS concerns. It is essential that a clinician consider the clinical context of patient presentation and the pre-test likelihood of ACS before making treatment decisions. Failure to consider pre-test probability often results in increased rates of misdiagnosis.13
The relevance of pre-test probability is seen in scenarios B and C. (See Table 4.) Scenario B involves a patient with high clinical suspicion for ACS and a negative cTn on initial evaluation. Although serial sampling is prudent, despite a negative cTn measurement, the clinical assessment of high pre-test likelihood for ACS should prompt early intervention to avoid a delay in therapy. Scenario C involves a patient with low clinical suspicion for ACS but an elevated cTn level. This patient is unlikely to benefit from aggressive intervention and may be at greater risk of complications that arise from overly invasive therapy. Instead, serial sampling of cTn levels and further assessment to uncover the etiology of cTn elevation is advisable.
Table 4: Management Strategies Based on Pre-test Probability and Troponins
|
Scenario |
Pre-test Probability |
Troponin |
Management |
|---|---|---|---|
|
A |
High |
+ |
Early invasive strategy |
|
B |
− |
Early intervention (conservative) strategy |
|
|
C |
Low |
+ |
Serial sampling and further diagnostic workup to determine underlying cause of troponin elevation |
|
D |
− |
Serial sampling and reassessment |
The Impact of Cardiac Troponin Measurement in Suspected ACS. Patients presenting with a high clinical suspicion for ACS and an elevated cTn concentration (scenario A) have a significant risk of death and recurrent ischemia.17 These patients tend to have more extensive and complex CAD when compared to patients with normal cTn measurements.25,26 They are more likely to benefit from early intervention and treatment, including antiplatelet and anticoagulant therapies coupled with revascularization strategies.27,28
Cardiac Troponin Measurements in Non-ischemic Conditions. Although in many cases the mechanism for cTn elevation is not well understood, elevated cTn concentrations may impart an increased risk of death and poor patient outcome.
Four non-ischemic clinical conditions resulting in acute myocardial cell injury in which cTn levels have been useful in guiding risk assessment and patient management include heart failure, pulmonary embolism, sepsis, and chronic kidney disease. Although cTn elevations in these conditions are not necessarily due to coronary atherosclerosis, elevated cTn concentrations in each of these conditions are associated with significant risk of death and poor prognosis.29-32
The Impact of High-sensitivity Cardiac Troponin Assays
High-sensitivity Cardiac Troponin Assays. Cardiac troponin assays were first introduced in the early 1990s. Conventional cTn assays typically refer to first- through fourth-generation assays and represent those currently in use in the United States. Fifth-generation assays are referred to as high-sensitivity cTn assays (hs-cTn) and are capable of measuring a 10-fold lower cTn concentration with high precision (CV 10% or less) when compared to conventional cTn assays.33 The terminology "high-sensitivity cardiac troponin assay" is reserved for assays capable of measuring cTn in at least 50% of a reference population.34-36 The hs-cTn assays are being used clinically throughout the world, and many experts believe it is only a matter of time before they are approved for use in the United States.
High-sensitivity Cardiac Troponin Assays: Analytical Performance and Sensitivity. In comparison to conventional cTn assays, hs-cTn assays demonstrate superior sensitivity, detecting lower levels of cTn with optimal precision. As a result, the 99th percentile upper reference limit and decision limit cutoff for AMI are much lower than for conventional assays.37 (See Figure 1.) These changes enable earlier detection of cTn elevations and will result in many more individuals with detectable cTn levels above the diagnostic threshold for AMI. Thus, sensitivity is vastly improved, but at the cost of specificity.
Figure 1: Troponin Levels
*Elevated but non-diagnostic secondary to decreased precision. (CV >10%)
Wallace and colleagues compared a fourth-generation conventional cTn assay with an hs-cTn assay to assess cTn elevations in the general population. The conventional assay detected cTn levels above the 99th percentile upper reference limit in 0.7% of patients. In the same population, the hs-cTn assay detected concentrations above the 99th percentile upper reference limit in 2% of patients, with only one-half having documentation of cardiac abnormalities.38,39 The prevalence of cTn detection above the 99th percentile upper reference limit is likely to be much higher in the ED and inpatient populations, likely resulting in more patients exceeding AMI threshold values.36
High-sensitivity Cardiac Troponin Assays: Negative Predictive Value. As with most tests demonstrating superior sensitivity, hs-cTn assays possess a high negative predictive value (NPV) and low false-negative rate. Therefore, a negative measurement is reassuring in the appropriate clinical setting and may negate the need for subsequent testing in some patients. Body et al demonstrated that the risk of adverse events in patients with undetectable hs-cTnT levels (below the LoD) was zero (NPV 100%) despite a prevalence of AMI of 19% in the patient population.40 A similar study by Keller et al using an hs-cTnI assay yielded similar results.41 These studies suggest that patients with undetectable hs-cTn levels (below the LoD) may not require serial monitoring and can be safely dispositioned after a single measurement. Although these findings present an appealing alternative to patient assessment, appropriate caution is warranted in patients presenting early after chest pain onset and those with a high clinical suspicion of ACS.
High-sensitivity Cardiac Troponin Assay: Positive Predictive Value. As more patients "rule in" by meeting AMI criteria (high sensitivity), clinicians will face the challenge to differentiate between cTn elevations representing true ACS events and other less acute causes of cTn elevation (low specificity).36 Januzzi et al found that 16.4% of low-to-intermediate risk chest pain patients had hs-cTnT levels meeting AMI criteria, while approximately 3% were later determined to have AMI.42 This lack of specificity and positive predictive value (PPV) presents several significant issues regarding patient care and resource utilization. If patients formerly considered non-ischemic by conventional cTn assays are now found to meet diagnostic criteria for AMI, there arises a potential to initiate overly aggressive and potentially harmful treatment strategies while failing to address the appropriate underlying medical condition. False-positive hs-cTn levels may also lead to increased unnecessary testing.
High-sensitivity Cardiac Troponin Assays: Serial Sampling of hs-cTn. As noted, the AMI definition does not identify a magnitude of change that is required for diagnosis, although a value of 20% is often used to ensure that a given change is not caused by analytic variation alone.4 The application of a relative change is more problematic with hs-cTn assays. At very low concentrations, absolute changes may be very small, while relative changes will be large.43-45 A recent comparison of absolute and relative changes during the serial sampling of hs-cTn demonstrated significant improvement in PPV when utilizing absolute changes.45 When defining the magnitude of change required for diagnosis, a larger change will improve specificity over sensitivity, while smaller changes will improve sensitivity over specificity. As noted by Korely and Jaffe, "it is not clear that all physicians want the same tradeoffs in this regard."36 Emergency room physicians typically prefer higher sensitivity to minimize the rate of missed diagnosis. Cardiologists, on the other hand, have reservations about the use of overly sensitive assays to detect changes in cardiac biomarkers. Many fear that the high sensitivity of cTn assays will detect more myocardial cell injury following cardiac procedures (PCI) and lead to an increased diagnosis of type 4 AMI. As a result, many cardiologists prefer the use of less sensitive CK-MB assays in the post-procedural assessment of serum biomarkers.46-48
High-sensitivity Cardiac Troponin Assays: An Attempted Rapid AMI Protocol. In 2012, Reichlin and colleagues employed a rule-out/rule-in protocol incorporating baseline and absolute changes in hs-cTnT levels at one hour. Four hundred thirty-six patients were divided into three groups based on hs-cTnT measurements. The rule-out group (60%) had a baseline hs-cTnT level of less than or equal to 12 ng/L, or an absolute change of less than or equal to 3 ng/L. The rule-in group (17%) had baseline hs-cTnT concentrations of greater than or equal to 52 ng/L, or an absolute change of 5 ng/L or greater. The observational group (23%) had baseline and absolute changes falling between the rule-in and the rule-out group. In the rule-out group, the sensitivity and NPV were 100%. The specificity for the rule-in group was 97%, with a PPV of 84%. Despite the lack of confirmatory studies, the potential to safely exclude and comfortably include patients over a one-hour time frame is enticing.49
Computed Tomography Angiography in the Evaluation of Chest Pain
Introduction. Computed tomography (CT) angiography is an imaging technique used to evaluate blood vessels throughout the body. This technique combines the use of a CT scanner with a well-timed injection of IV contrast to capture the most optimal view of the blood vessel(s) being evaluated.
CT Angiogram Quality. In general, the quality of the study is dependent on the quality of the CT scanner. Most hospitals have some form of multi-detector CT (MDCT) scanner ranging from 16 to 64 slices. One analogy for understanding the significance of MDCT slices is cutting a loaf of raisin bread. The chance of slicing through a raisin increases with the number of slices. Similarly, a greater number of slices increases the sensitivity of detecting a pulmonary embolism. Newer scanners are now capable of acquiring 128 to 320 slices corresponding to the number of images that can be acquired during a certain time frame. Consequently, the quality of time-dependent studies, including CT angiograms, improves with a greater number of slices.
There are different scanning protocols depending on the vessel to be imaged. Some are simple, while others are technically challenging and complex. "Bolus tracking" and "test boluses" are two common techniques used to time the image acquisition with the IV contrast.50 This involves using the CT scanner to detect when the IV contrast reaches the target area or using a test bolus to time the delay between the injection and the scan. More complex protocols, such as those for coronary CT angiography, include the use of medications such as beta-blockers to slow the heart rate in order to reduce motion artifact. Similarly, a technique called ECG gating may be used to time the patient’s heart beat with the image capture and further reduce motion artifact.51
Important Considerations for Performing CT Angiography. While emergency physicians generally are not involved in the specifics of these protocols, they should be aware of factors that may affect the quality of the results or the ability to perform the exam. To reduce motion artifact and increase the quality of the images, patients must be able to tolerate the supine position, remain still, and hold their breath for several seconds. Caution should be used when sending hemodynamically unstable patients to the CT scanner, where they may decompensate and where proper equipment and personnel are unavailable.
Renal function is another important consideration when ordering a CT angiogram. Contrast-induced nephropathy (CIN) is a form of acute renal failure occurring after a patient is given an IV contrast bolus. The risk of CIN increases in patients with repeat contrast dosing, renal insufficiency, diabetes, and heart failure.52,53 IV hydration before CT angiography can significantly reduce the chance of CIN, and lower-dose studies may also be performed.53
Intravenous access is a common stumbling block, as CT angiograms require large-bore IV catheters to achieve the fast injection rates necessary to vessel visualization. A power injector is commonly used to control the rate of injection. If the IV catheter is too small, there is a risk of subcutaneous infiltration with extravasation of contrast. Ideally, a large-bore peripheral IV larger than 20-gauge in the antecubital fossa is required. Of note, not all central venous lines (CVLs) are adequate for power injection; therefore, a physician should confirm whether the type of CVL being placed is compatible with power injection. Additionally, some institutions prohibit power injection through an external jugular vein regardless of the IV catheter size because of the risk of extravasation into the neck.
Adverse reactions to IV contrast have generated significant confusion among health care providers. Contrary to a common myth, iodine itself is not a true allergen, and a shellfish allergy does not increase the likelihood of a reaction to IV contrast any more than other allergies do (e.g., peanuts, latex).54 Reactions to IV contrast are termed anaphylactoid and do not convey the same risk of escalation with repeat exposures as IgE-mediated reactions typically do. Patients at increased risk of adverse reaction may be pre-medicated with an antihistamine such as diphenhydramine. Steroids have not been shown to decrease the severity of reactions unless given greater than 3 hours prior to the study.55 Patients should be questioned about the severity of any previous reactions to IV contrast. Emergency physicians should consider pre-medication with a history of mild reactions and avoid contrast if reactions were severe.
Radiation exposure can vary significantly between different institutions and types of CT scanners.56 A routine chest CT with contrast may have a median dose range of 711 mSv, while a chest CT angiography may have a median dose range of 821 mSv.57 Repeat CT angiograms throughout a patient’s lifetime may increase the risk of developing a malignancy, including breast cancer. It is estimated that 1 in 270 40-year-old women will develop a radiation-induced cancer after undergoing pulmonary CT angiogram. For 20-year-old women, it is estimated to be 1 in 150.57 For reasons such as this, physicians should be judicious with their use CT angiography.58
Evaluation of Chest Pain in the ED. CT angiography offers a means of rapidly evaluating for several life-threatening causes of acute chest pain in the ED. These causes include pulmonary embolism, aortic dissection, and ACS. Different types of CT angiograms with distinct protocols are used for each of the three disease processes. A limited number of institutions are using a technique known as the "triple rule out" (TRO-CTA). This is a CT angiography protocol for evaluating the pulmonary arteries, coronary arteries, and aorta with a single scan. The concept is appealing: rule out three of the deadliest causes of chest pain with a single study. In reality, there are significant issues that have kept this type of study from becoming more widely used. It typically requires at least a 64-slice CT scanner, longer patient breath holds, and higher radiation doses. This study is technically challenging; the most significant issue is timing of the contrast injection. The time between peak contrast enhancement of the pulmonary arteries versus the aorta ranges between 10-12 seconds apart. Consequently, maximizing the enhancement of the different vessels simultaneously is difficult and may result in decreased overall sensitivity of the exam. Most TRO-CTA protocols use larger contrast boluses or two separate boluses to improve the quality.59,60 The TRO-CTA should be used judiciously and not as a routine study for chest pain evaluation. Current recommendations are for patients deemed to be low-to-intermediate risk for ACS whose histories are unclear and have sufficiently high probability for both aortic dissection and pulmonary embolism.61,62
Comparing Anatomical Studies: CCTA and Invasive Coronary Angiography
Similar to cardiac catheterization (which traditionally is considered the gold standard), CCTA is an anatomical study that evaluates the presence and degree of coronary artery disease. (See Figure 2.)
Figure 2: Coronary CT Angiography Images
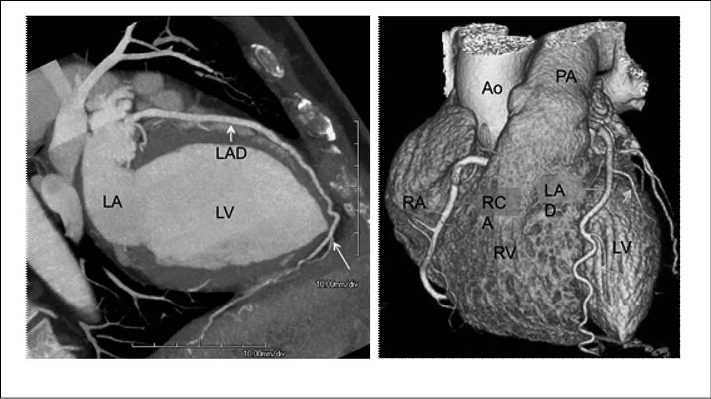
Courtesy of Dr. Satinder Singh, UAB Department of Radiology
ACCURACY is a multicenter prospective clinical trial published in 2008 that evaluated CCTA performed on 64-slice CT scanners in patients referred for non-emergent invasive coronary angiography (ICA).63 Patients received both CCTA and ICA. The results of this study demonstrated that CCTA had 99% negative predictive value (NPV) for excluding obstructive coronary stenosis. The results suggested that CCTA might be used as an effective screening tool in the right patient population.
A recent meta-analysis by Salavati et al showed a pooled sensitivity of 99% and a specificity of 89% for CCTA performed by dual-source CT scanners.64 With this advanced technology, both a high sensitivity and NPV were maintained despite faster heart rates. The CONFIRM registry, an ongoing multinational prospective study enrolling more than 25,000 patients, was established to assess the utility of CCTA to evaluate the presence of CAD. Study results thus far have supported the role of CCTA as an effective screening tool for ICA.65 These studies demonstrate that CCTA may exclude the need for ICA in many patients undergoing non-emergent evaluation for CAD.
Evaluation of ACS in the ED
During the past five years, several important studies have been published concerning the utility of CCTA in the ED.
In 2009, Hollander et al conducted a prospective trial with 568 patients undergoing CCTA for possible ACS at a single university hospital ED.66 Patients were enrolled if they had a TIMI score of 0 to 2 and if the physician’s clinical suspicion was high enough to otherwise warrant a rule-out admission. Eighty-four percent of patients were discharged home after CCTA with no subsequent death or MI within 30 days. Hollander later published a second study in 2009 evaluating one-year outcomes in the same cohort and found no AMIs or revascularization procedures in patients with CCTA results showing coronary stenosis less than 50%.67 These studies suggested that CCTA could play a role in ED chest pain.
Following the Hollander studies, a multicenter prospective trial called CT-STAT was published in 2011 evaluating 749 ED patients randomized to receive CCTA (anatomic study) or rest-stress myocardial perfusion imaging (MPI) (functional study) as part of a low-risk chest pain protocol.68 There was no significant difference in clinical outcomes between the two groups. Eight percent of patients who received CCTA subsequently underwent ICA, and approximately 50% of these patients (4% of total) went on to PCI or CABG. Similarly, 7.4% of patients who received MPI underwent ICA, and 36% of these patients underwent revascularization.
A year later in 2012, two multicenter prospective trials were published in the New England Journal of Medicine evaluating the use of CCTA in the ED for acute chest pain: the ACRIN-PA trial69 and the ROMICAT II trial.70 In both trials, patients at low-to-intermediate risk for ACS were randomized to CCTA or standard care (which typically included MPI). In both trials, clinically significant coronary artery stenosis (a "positive" CCTA result) was defined as stenosis of 50% or greater.
The ACRIN-PA trial enrolled 1,370 low-to-intermediate risk patients. Inclusion criteria included patients older than the age of 30 years with TIMI scores of 0 to 2, one set of negative cardiac biomarkers, and a non-ischemic ECG. Nine hundred eight patients underwent CCTA. Of this group, approximately 70% had "negative" CCTA results (less than 50% stenosis) and were safely discharged with no subsequent death or MI within 30 days. There was a small (statistically insignificant) increase in revascularization procedures in the CCTA group.
The ROMICAT II trial enrolled 1,000 patients between the ages of 40 and 74 with no previous history of CAD, one set of negative cardiac biomarkers, and a non-ischemic ECG. Four hundred ninety-seven patients underwent CCTA. Of this group, patients with "negative" CCTA results had no subsequent death or MI within 28 days. In comparison to previous studies, ROMICAT II showed a statistically significant increase in subsequent diagnostic testing in the CCTA group. Twelve percent of CCTA patients underwent subsequent ICA, as opposed to 8% in the MPI group, and 6% of CCTA patients went on to PCI or CABG, as opposed to 4% in the MPI group.
The ACRIN-PA and ROMICAT II trials further demonstrated that CCTA could be safely used in low-to-intermediate chest pain evaluations with a notable trend toward an increased number of invasive procedures.
Short-term Benefits and Long-term Consequences
In addition to studies demonstrating CCTA as an effective screening tool for coronary disease, implementation of CCTA protocols may reduce ED lengths of stay (LOS) and ED costs compared to usual care. A 2013 meta-analysis by Hulten et al71 looked at four prospective clinical trials (Goldstein, CT-STAT, ACRIN-PA, and ROMICAT II) and showed reductions in times to diagnosis ranging from 4477% compared to usual care. Hospital LOS was reduced overall by 2427%, and approximately 4750% of patients who underwent CCTA were discharged directly from the ED, compared to 1223% of patients receiving usual care. ED cost-savings of 1538% were also demonstrated in the CCTA group.71 These findings suggest that there may be several advantages of using CCTA in the evaluation of low-to-intermediate chest pain evaluations. (See Table 5.)
Table 5: Current Evidence Concerning CCTA-based Strategy in the ED*
• An acceptable method of objective testing for patients with low-to-intermediate risk of ACS
• Reduced ED length of stay
• Reduced ED cost
• Higher rate of direct ED discharge
• Higher rate of subsequent cardiac catheterizations
• Unclear effect on overall patient outcomes
*Compared to standard care strategies typically involving a stress test
Rapid dispositions and decreased ED costs are naturally attractive to emergency physicians; however, there are potential downstream consequences of CCTA to consider. As suggested by several studies mentioned previously, the meta-analysis by Hulten et al showed that CCTA was associated with an overall 2% increase in the number of invasive coronary angiograms and a similar 2% increase in revascularization compared to MPI.71 The ongoing CONFIRM trial suggests that patients discovered to have mild CAD on CCTA tend to undergo an increased number of subsequent ICA as well.72 The corresponding increase in both ICA and revascularization might suggest that CCTA detects a higher number of patients with clinically significant CAD; however, long-term data showing improved patient outcomes is lacking. Additionally, the long-term health care cost-savings of CCTA are debatable. While evidence suggests there are short-term savings for the ED, studies evaluating downstream costs for patients are limited and have mixed results.72,73
It should also be mentioned that critics argue a significant percentage of the patient population being randomized to studies such as CCTA and MPI might be safely discharged from the ED without receiving either test.74 Both tests typically involve additional radiation exposure and added health care cost as well as an increased likelihood of invasive procedures. Patients at low risk of ACS with a normal ECG and negative serial cardiac biomarkers may be at an extremely low risk to negate the need for tests such as CCTA or MPI. Emergency physicians should be prudent when ordering CCTA. (See Table 6.)
Table 6: Considerations for CCTA Patient Selection
• 30 years old or older
• Hemodynamically stable
• Low-to-intermediate risk of ACS*
• No previous history of coronary artery disease
• Sinus rhythm with no ischemic changes on ECG
• Normal troponin
• Normal renal function
• No prior IV contrast reactions
Consider the attributable lifetime risk of radiation-induced cancer, particularly in younger women.
*Deemed by the physician to warrant further risk stratification
Coronary Artery Calcium Scoring and ACS in the ED
Coronary artery calcium scoring (CACS) is frequently cited in discussions concerning CCTA and is often a point of confusion. While both CACS and CCTA screen for the presence of coronary artery disease using computed tomography, there are several important distinctions to note. CACS is a non-contrasted study that looks specifically at the degree of detectable calcium in the coronary arteries. Higher calcium scores are correlated with a higher likelihood of atherosclerosis and coronary artery disease. However, CACS does not assess the patency of the coronary vessels and, compared to CCTA, research regarding CACS in patients presenting to the ED with chest pain is sparse. CACS is used most frequently in the risk stratification of asymptomatic patients and has a limited role in patients with suspected ACS. In 2010, Nabi et al reported that patients with a low pre-test probability of ACS and a calcium score of 0 have a high likelihood of normal MPI and safe short-term outcomes.75 As a result, CACS may have a role in limiting further cardiac imaging in low-risk chest pain patients. Further research regarding CACS utility in the ED will be required to help delineate its possible role in the assessment of ACS.
Conclusion
Emergency physicians should be familiar with the diagnostic studies available to aid in the risk assessment and diagnosis of ACS and AMI. Two newly developed modalities, the hs-cTn biomarker assay and CCTA, are likely to find increased use in the ED.
High-sensitivity cardiac troponin assays have superior sensitivity and analytic precision at low cTn levels. There is legitimate concern that a greater number of patients will be mistakenly labeled as AMI by meeting criteria defined by the universal definition of MI (developed with the use of conventional cTn assays). This may lead to more invasive testing strategies and subject patients to potential harm. Perhaps with the introduction of the hs-cTn assay into common clinical use, we will need to once again redefine the criteria for AMI.
CCTA is an imaging technique used with increasing frequency in the evaluation of patients presenting to the ED with chest pain. CCTA shows promise for being a safe and faster alternative to more traditional chest pain protocols; however, it is associated with an increase in subsequent invasive procedures with questionable long-term benefits.
It is interesting to consider the impact that these two technologies may have on each other. Both the hs-cTn assay and CCTA have the potential to detect more CAD and lead to more frequent initiation of early, invasive treatment strategies. However, as cTn measurements often precede other functional (MPI) and anatomic (CCTA) studies, the early detection of low-level cTn concentrations may prompt clinicians to bypass less invasive risk assessment and proceed directly to invasive testing (i.e., decrease the utility of CCTA). Alternatively, patients with very low levels of cTn detection (of questionable clinical significance) may require further characterization before invasive examination (i.e., increase the utility of CCTA). In either scenario, the most important factor driving decision-making should be the clinical context of patient presentation and the pre-test likelihood of disease. Ultimately, these technologies must be part of a larger "clinically driven management algorithm."13
References
1. Nomenclature and Criteria for Diagnosis of Ischemic Heart Disease. Report of the Joint International Society and Federation of Cardiology/World Health Organization Task Force on Standardization of Clinical Nomenclature. Circulation 1979;59:607-609.
2. Collinson PO, Boa FG, Gaze DC. Measurement of cardiac troponins. Ann Clin Biochem 2001;38:423-429.
3. Jaffe AS, Ravkilde J, Roberts R, et al. It’s time for a change to a troponin standard. Circulation 2000;102:1216-1220.
4. Thygesen K, Alpert JS, Jaffe AS, et al. Joint ESC/ACCF/AHA/WHF Task Force for the Universal Definition of Myocardial Infarction. Third universal definition of myocardial infarction. Circulation 2012;126(16):2020-2035.
5. de Lemos JA. Increasingly sensitive assays for cardiac troponins. A review. JAMA 2013;309(21):2262-2269.
6. Alpert JS, Thygesen K, Bassand JP, et al. Myocardial infarction redefined. A consensus document of the Joint European Society of Cardiology/American College of Cardiology Committee for the Redefinition of Myocardial Infarction.
J Am Coll Cardiol 2000;36:959-969.
7. Myocardial infarction redefined — a consensus document of the Joint European Society of Cardiology/American College of Cardiology Committee for the redefinition of myocardial infarction. Eur Heart J 2000;21:502-513.
8. Jesse RL. On the relative value of an assay verus that of a test: A history of troponin for the diagnosis of myocardial infarction. J Am Coll Cardiol 2010;55:2125-2128.
9. Tovey JA, Brain A, Penney MD. Calculation of the 99th percentile upper reference limit and clinical decision concentration of the Abbott STAT TnI assay. Ann Clin Biochem 2010;47(6):576-577.
10. Thygesen K, Alpert JA, Jaffe AS, et al. Third universal definition of myocardial infarction. J Am Coll Cardiol 2012;60:1581-1598.
11. Jaffe AS, Apple FS, Morrow DA, et al. Being rational about (im)-precision: A statement from the Biochemistry Subcommittee of the Joint European Society of Cariology/American College of Cardiology Foundation/American Heart Association/World Heart Federation Task Force for the definition of myocardial infarction. Clin Chem 2010;56:941-943.
12. Apple FS. What is the 99th percentile and the 10% coefficient of variation and how do they related to troponin testing? Biomarkers.cardiosource.org/Ask-the-expert/2010/07/99th-percentileaspx?p=1.
13. Newby LK, Jess RL, Babb JD, et al. ACCF 2012 expert consensus document on practical clinical considerations in the interpretation of troponin elevations: a report of the American College of Cardiology Foundation taskforce on Clinical Expert Consensus Documents.
J Am Coll Cardiol 2012;60:2427-2463.
14. Jesse RL. On the relative value of an assay versus that of a test: A history of troponin for the diagnosis of myocardial infarction. J Amer Coll Cardiol 2010;55;2125-2128.
15. Morrow DA, Cannon CP, Jesse RL, et al. National Academy of Clinical Biochemistry. National Academy of Clinical Biochemistry Laboratory Medicine Practice Guidelines: Clinical characteristics and utilization of biochemical markers in acute coronary syndromes. Clin Chem 2007;53(4):552-574.
16. Than M, Cullen L, Aldous S, et al. 2-Hour accelerated diagnostic protocol to assess patients with chest pain symptoms using contemporary troponins as the only biomarker; the ADAPT trial. J Am Coll Cardiol 2012;59(23):2091-2098.
17. Bonaca M, Scirica B, Sabatine M, et al. Prospective evaluation of the prognostic implications of improved assay performance with a sensitive assay for cardiac troponin I. J Am Coll Cardiol 2010;55(19):2118-2124.
18. Jeremias A, Gibson CM. Narrative review: Alternative causes for elevated cardiac troponin levels when acute coronary syndromes are excluded. Ann Intern Med 2005;142(9):786-791.
19. Eggers KM, Lagerqvist B, Oldgren J, et al. Pathophysiologic mechanism of persistent cardiac troponin I elevation in stabilized patients after an episode of acute coronary syndrome. Am Heart J 2008;156(3):588-594.
20. Sato Y, Yamada T, Taniguchi R, et al. Persistently increased serum concentrations of cardiac troponin T in patients with idiopathic dilated cardiomyopathy are predictive of adverse outcomes. Circulation 2001;103(3):369-374.
21. Wallace TW, Abdullah SM, Drazner MH, et al. Prevalence and determinants of troponin T elevation in the general population. Circulation 2006;113:1958-1965.
22. Eggers KM, Lind L, Ahlstrom H, et al. Prevalence and pathophysiological mechanisms of elevated cardiac troponin I levels in a population-based sample of elderly subjects. Eur Heart J 2008;29(18):2252-2258.
23. Lestner DM, Klotsche J, Pieper L, et al. DETECT Study Group. Circulating troponin as measured by a sensitive assay for cardiovascular risk assessment in primary prevention. Clin Chem 2012;58:200-208.
24. Bhuiya FA, Pitts SR, McCaig LF. Emergency department visits for chest pan and abdominal pain: United States, 199-2008. NCHS Data Brief 2010;(43):1-8.
25. Lindahl B, Diderholm E, Lagerqvist B, et al; FIRSC II (Fast Revascularization during Instability in CAD) Investigators. Mechanisms behind the prognostic value of troponin T in unstable coronary artery disease: A FRISC II substudy. J Am Coll Cardiol 2001;38(4):979-986.
26. Wong GC, Morrow DA, Murphy S, et al. Elevations in troponin T and I are associated with abnormal tissue-level perfusion: A TACTICS-TIMI 18 substudy. Circulation 2002;106(2):202-207.
27. Lindahl B, Venge P, Wallentin L; Fragmin in Unstable Coronary Artery Disease (FRISC) Study Group. Troponin T identifies patients with unstable coronary artery disease who benefit from long-term antithrombotic protection. J Am Coll Cardiol 1997;29(1):43-48.
28. Bavry AA, Bumbhani DJ, Quiroz R, et al. Invasive therapy along with glycoprotein IIb/IIIa inhibitors and intracoronary stents improves survival in non-ST-segment elevation acute coronary syndromes: A meta-analysis and review of the literature. Am J Cardiol 2004;93(7):830-835.
29. Peacok WF IV, De Marco T, Fonarow GC, et al. ADHERE Investigators. Cardiac troponin and outcome in acute heart failure. N Engl J Med 2008;358(20):2117-2126.
30. Giannitsis E, Muller-Bardorff M, Kurowski V, et al. Independent prognostic value of cardiac troponin T in patient’s with confirmed pulmonary embolism. Circulation 2000;102(2):211-217.
31. Ammann P, Maggiorini M, Bertel O, et al. Troponin as a risk factor for mortality in critically ill patients without acute coronary syndromes. J Am Coll Cardiol 2003;41(11):2004-2009.
32. Apple FS, Murakami MM, Pearce LA, et al. Predictive value of cardiac troponin I and T for subsequent death in end-stage renal disease. Circulation 2002;106(23):2941-2945.
33. Daubert MA, Jeremaias A. The utility of troponin measurement to detect myocardial infarction: Review of the current findings. Vasc Health Risk Manag 2010;6:691-699.
34. Apple FS, Collinson PO. Analytical characteristics of high-sensitivity cardiac troponin assays. Clin Chem 2012;58:54-61.
35. Apple FS. A new season for cardiac troponin assays: It’s time to keep a scorecard. Clin Chem 2009;55:1303-1306.
36. Korley FK, Jaffe AS. Preparing the United States for high-sensitivity cardiac troponin assays. J Am Coll Cardiol 2013;61:
1753-1758.
37. Apple FS, Parvin CA, Buechler KF, et al. Validation of the 99th percentile cutoff independent of assay imprecision (CV) for cardiac troponin monitoring for ruling out myocardial infarction. Clin Chem 2005;51:2198-2200.
38. de Lemos JA, Drazner MH, Omland T, et al. Association of troponin T detected with a highly sensitive assay and cardiac structure and mortality risk in the general population. JAMA 2010;304:2503-2512.
39. Wallace TW, Abdullah SM, Drazner MH, et al. Prevalence and determinants of troponin T elevation in the general population. Circulation 2006;113:1958-1965.
40. Body R, Carley S, McDowell G, et al. Rapid exclusion of acute myocardial infarction in patients with undetectable troponin using a high-sensitivity assay.
J Am Coll Cardiol 2011;58:1332-1339.
41. Keller T, Zeller T, Ojeda F, et al. Serial changes in highly sensitive troponin I assay and early diagnosis of myocardial infarction. JAMA 2011;306:2684-2693.
42. Januzzi JL Jr, Bamberg F, Lee H, et al. High sensitivity troponin T concentrations in acute chest pain patients evaluated with cardiac computed tomography. Circulation 2010;121(10):1227-1234.
43. Turer AT, Addo TA, Martin JL, et al. Myocardial ischemia induced by rapid atrial pacing causes troponin T release detectable by a highly sensitive assay: Insights from a coronary sinus sampling study. J Am Coll Cardiol 2011;57(24):2398-2405.
44. Sabatine MS, Morrow DA, de Lemos JA, et al. Detection of acute changes in circulating troponin in the setting of transient stress test-induced myocardial ischemia using an ultrasensitive assay: Results form TIMI 35. Eur Heart J 2009;30:162-169.
45. Reichlin T, Irfan A, Twerenbold R, et al. Utility of absolute and relative changes in cardiac troponin concentrations in the early diagnosis of acute myocardial infarction. Circulation 2011;124(2):136-145.
46. Califf RM, Abdelmeguid AE, Kuntz RE, et al. Myonecrosis after revascularization procedures. J Am Coll Cardiol 1998;31:241-251.
47. White HD. The prequel. Defining prognostically important criteria in the peri-procedural PCI troponin saga. Circ Cardiovasc Interv 2012;5:142-145.
48. Jaffe AS, Apple FS, Lindahl B, et al. Why all the struggle about CK-MB and PCI? Eur Heart J 2012;33:1046-1048.
49. Reichlin T, Schindler C, Drexler B, et al. One hour rule-out and rule-in of acute myocardial infarction using high-sensitivity cardiac troponin T. Arch Intern Med 2012;172(16):1211-1218.
50. Bae KT. Test-bolus versus bolus-tracking technqiues for CT angiographic timing. Radiology 2005;236:369-370.
51. Wu W, Budovec J, Foley WD. Prospective and retrospective ECG gating for thorasic CT angiography: A comparative study. Am J Roentgenol 2009;193(4):955-963.
52. Toprak O. Risk markers for contrast-induced nephropathy. Am J Med Sci 2007;334(4):283-290.
53. Balemans CE, Reichert LJ, van Schelven BI, et al. Epidemiology of contrast material-induced nephropathy in the era of hydration. Radiology 2012;263:706-713.
54. Schabelman E, Witting M. The relationship of radiocontrast, iodine, and seafood allergies: A medical myth exposed.
J Emerg Med 2010;39:701-707.
55. American College of Radiology. Manual on Contrast Media. Version 8. Reston, VA: American College of Radiology; 2012. Available at: http://www.acr.org/~/media/ACR/Documents/PDF/QualitySafety/Resources/Contrast%20Manual/FullManual.pdf. Accessed May 15, 2013.
56. Hausleiter J, Meyer T, Hermann F, et al. Estimated radiation dose associated with cardiac CT angiography. JAMA 2009;301(5):500-507.
57. Smith-Binder R, Lipson J, Marcus R, et al. Radiation dose associated with common computed tomography examinations and the associated lifetime attributable risk of cancer. Arch Intern Med 2009;169(22):2078-2086.
58. Parker MS, Hui FK, Camacho MA, et al. Female breast radiation exposure during CT pulmonary angiography. Am J Roentgenol 2005;185:1228-1233.
59. Halpern EJ, et al. Comparison of image quality and arterial enhancement with a dedicated coronary CTA protocol versus a triple rule-out coronary CTA protocol. Acad Radiol 2009;16(9):1039-1048.
60. Rahmani N, Jeudy J, White CS. Triple rule-out and dedicated cornary artery CTA: Comparison of coronary image quality. Acad Radiol 2009;16:604-609.
61. Harpern EJ. Triple-rule-out CT angiography for evaluation of acute chest pain and possible acute coronary syndrome. Radiology 2009;252:332-345.
62. Cury RC, et al. Cardiac CT in the emergency department: Convincing evidence, but cautious implementation. J Nucl Cardiol 2011;18:331-341.
63. Budoff MJ, Dowe D, Jollis JG, et al. Diagnostic performance of 64-multidetector row coronary computed tomographic angiography for evaluation of coronary artery stenosis in individuals without known coronary artery disease. J Am Coll Cardiol 2008;52(21):1724.
64. Salavati A, Radmanesh F, Heidari K, et al. Dual-source computed tomography angiography for diagnosis and assessment of coronary artery disease: Systematic review and meta-analysis. J Cardio Comp Tomography 2012;6:78-90.
65. Shaw LJ, Hausleiter J, Achenbach S, et al. Coronary computed tomographic angiography as a gatekeeper to invasive diagnostic and surgical procedures. J Am Coll Cardiol 2012;60(20):2103.
66. Hollander JE, Chang AM, Shofer FS, et al. Coronary computed tomographic angiography for rapid discharge of low-risk patients with potential acute coronary syndromes. Ann Emerg Med 2009;53:295304.
67. Hollander JE, Chang AM, Shofer FS, et al. One-year outcomes following coronary computerized tomographic angiography for evaluation of emergency department patients with potential acute coronary syndrome. Acad Emerg Med 2009;16:693698.
68. Goldstein JA, Chinnaiyan KM, Abidov A, et al. The CT-STAT (Coronary Computed Tomographic Angiography for Systematic Triage of Acute Chest Pain Patients to Treatment) trial. J Am Coll Cardiol 2011;58:14141422.
69. ACRIN-PA Trial: Litt HI, et al. CT angiography for safe discharge of patients with possible acute coronary syndromes.
N Engl J Med 2012;366:1393-1403.
70. ROMICAT II Trial: Hoffmann U, et al; ROMICAT-II Investigators. Coronary CT angiography versus standard evaluation in acute chest pain. N Engl J Med 2012;367:299-308.
71. Hulten E, Pickett C, Bittencourt MS, et al. Outcomes after coronary computed tomography angiography in the emergency department: A systematic review and meta-analysis of randomized, controlled trials. J Am Coll Cardiol 2013;61(8):880-892.
72. Shreibati JB, Baker LC, Hlatky MA. Association of coronary CT angiography or stress testing with subsequent utilization and spending among medicare beneficiaries. JAMA 2011;306(19):2128.
73. Min JK, Gilmore A, Budoff MJ, et al. Cost-effectiveness of coronary CT angiography versus myocardial perfusion SPECT for evaluation of patients with chest pain and no known coronary artery disease. Radiology 2010;254(3):801.
74. Redberg RF. Coronary CT angiography for acute chest pain. N Engl J Med 2012;367(4):375-376.
75. Nabi N, Chang SM, Pratt C, et al. Coronary artery calcium scoring in the emergency department: Identifying which patients with chest pain can be safely discharged home. Ann Emerg Med 2010;56(3):220-229.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
