Help! My Patient Needs Opioids, or Do They?
Help! My Patient Needs Opioids, or Do They?
Authors: Nirmala Abraham, MD, FABPM, Medical Director, Sycamore Pain Management Center, Miamisburg, OH
Cathy D. Trame, RN, MS, CNS, BC, Manager, Acute and Perioperative Pain Program, Kettering Physician Network, Miamisburg, OH
Peer Reviewer: Clara L. Carls, DO, Program Director, Hinsdale Family Medicine Residency, Hinsdale, IL
Introduction
The national epidemic of substance abuse naturally imparts uncertainty and fear in the decision-making process regarding the prescribing of opioids for patients in pain. Statistics alone substantiate the need to validate our decisions with a documented plan of care. The burden of deciphering which patients are appropriate for ongoing opioid management typically falls to the primary care provider. How best can we manage our patients safely and ethically? A descriptive step-by-step approach to making the decision to prescribe opioids and managing your patients on opioids will be presented.
Background
Statistics released by the Drug Abuse Warning Network (DAWN) tout the drastic upswing in the abuse of prescription opioids and the related deaths.1 Emergency departments (ED) in the United States reported a 156% increase in the non-medical use of opioids from 2004-2010. (See Table 1.) In 2010, the misuse or abuse of drugs accounted for 46.8% of all ED visits related to adverse drug events. The most common drugs of abuse included oxycodone, with a 255% increase, and hydrocodone, with a 149% increase. During the same time period, benzodiazepine abuse increased 139%.
Table 1: Drugs with Increasing Involvement in Emergency Department (ED) Visits for Drug Misuse or Abuse: 2004-2010
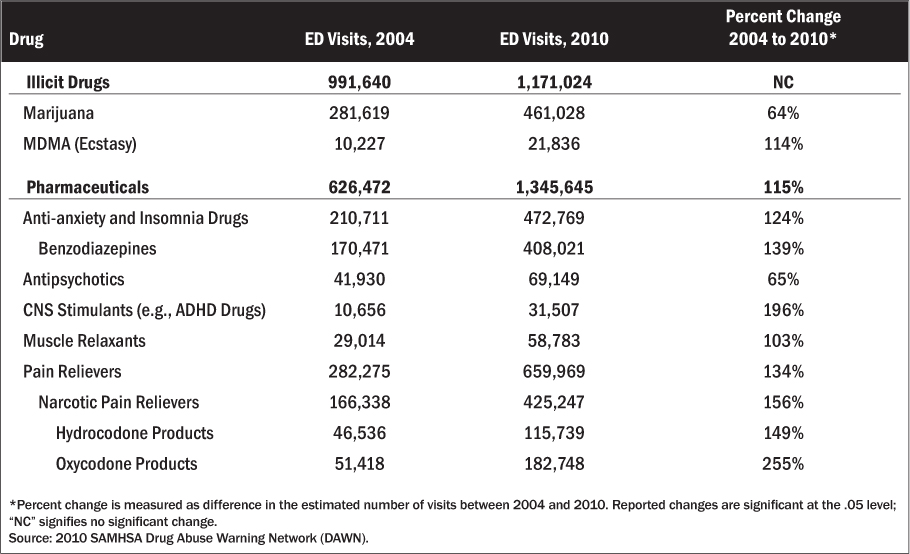
The Centers for Disease Control and Prevention (CDC) has reported equally disturbing data. In the United States, 100 people a day die in the United States from drug overdoses, a rate of death that has tripled since 1990.2 For every four deaths that occur due to prescription drugs, three are related to prescription opioids. The 2009 National Youth Risk Behavior Survey revealed that one in five high school students have taken a prescription drug that has not been prescribed for them,3 while one in 20 people ages 12 and older have admitted to using a prescription opioid within the past year for purely recreational reasons.4 A reported 12 million people in the United States use prescription opioids for the euphoric effect vs pain relief. The abuse of methamphetamines, marijuana, cocaine, steroids, and even alcohol has shown a decline in the time period from 1999-2009.5
While our nation battles the abuse of prescription opioids, the prescribing of these medications has drastically increased. As the population lives longer, with coexisting aging issues related to pain, the demand for analgesic medications increases. The rise in obesity also contributes to the development of chronic pain related to stress on joints and the back. According to the CDC, in the last 15 years, the prescribing of opioids has increased tenfold.6 In 2010, there were enough opioids prescribed to medicate “every American adult around the clock for one month.”7 History reveals that an increase in the availability of a drug increases the risk for abuse.8 Responsible prescribing of opioids, coupled with risk mitigation strategies, has never been more paramount.
Opioid demand has increased as the number of patients with chronic pain has become more prevalent; in fact chronic pain has reached epidemic proportions.
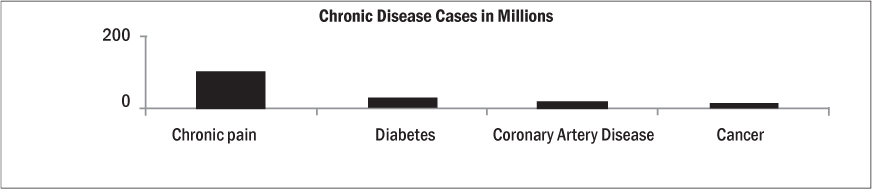
The American Chronic Pain Association describes chronic pain as “... ongoing or recurrent pain, lasting beyond the usual course of acute illness or injury or more than 3 to 6 months, and which adversely affects the individual’s well-being.”9 Chronic pain affects approximately 100 million adults in the United States, costing an average of $2000 for every U.S. resident.10 The National Center for Health Statistics reported 26% of adults over the age of 20 experience chronic pain.11 Chronic pain is four times more prevalent than diabetes,12 nearly six times more prevalent than coronary heart disease,13 and eight times more prevalent than cancer.14 (See Table 2.)
Table 2: Incidence of Chronic Diseases in the United States
According to the Institutes of Medicine, the most common chronic pain treated is low back pain (28.1%), followed by knee pain (19.5%) and migraine or headache pain (16.1%).10 Chronic pain historically has been managed poorly. In the 2006 “Voices of Chronic Pain” survey, respondents revealed that they felt little control over their pain (51%), and that breakthrough pain experienced on a daily basis greatly impacted their quality of life (60%).15 Sadly, 50-75% of cancer patients, at the time of death, have moderate-to-severe pain.16 Clearly, the increase in opioid prescribing has not been effective in alleviating chronic pain. When do these chronic conditions that are frequently evaluated and treated by the primary care physician warrant opioid prescribing?
Management of the Crisis
Government representatives have proposed legislation in an attempt to help mitigate the addiction crisis. Congressman Vern Buchanan (R-FL) proposed federal legislation, H.R. 1065: Pill Mill Crackdown Act of 2011, and Senator Joe Manchin (D-WV) proposed S. 1760: Pill Mill Crackdown Act of 2011 to increase fines and prison terms to individuals who prescribe opioids for monetary gain by running “pill mills.”17 Although the bills are still in committee, it is likely that some federal legislation will be forthcoming. Many state medical boards have enacted legislation regarding rules for the prescribing of scheduled substances, while state legislators have enacted additional monitoring, fines, and imprisonment for illegal prescribing. Thirty-seven states have instituted prescription monitoring systems to help physicians, pharmacists, and law enforcement track the patterns of individuals obtaining controlled substances via prescriptions, while 11 additional states are in the legislative process for implementation.18 Linkage of information among states in nearby geographical locations is being planned in some regions to better monitor those individuals who cross state lines to hide prescription abuse. Primary care physicians have a responsibility to link into their state resources for monitoring prescriptions and to validate or invalidate patient behaviors that are suspicious of substance abuse. The Alliance of States with Prescription Monitoring Programs (www.pmpalliance.org) provides a list for available programs in your state.
Resources from pharmaceutical companies that manufacture and distribute potent opioids are readily available in the form of Risk Evaluation and Mitigation Strategies (REMS) and patient education tools. The FDA began requiring REMS in 2007 for extended-release and long-acting (ER/LA) opioid analgesics.19 General requirements of the manufacturer are to provide patient education materials with clear product labeling regarding the dangers and prescriber education with guidelines for patient monitoring. Prescribers are responsible for implementing the REMS strategies associated with particular medications that they are prescribing for their patients.
Available Tools and Resources
Screening Tools for Abuse Potential. Several easy-to-use assessment tools are available for screening patients for possible substance abuse. For an office-based practice, a tool that is simple to use, reliable, valid, and can be completed fairly quickly would be practical. Tools that aid in predicting abuse potential prior to prescribing include NIDA Drug Use Screening Tool, Opioid Risk Tool (ORT), Screener and Opioid Assessment for Patients with Pain (SOAPP/SOAPP-R), and Diagnosis Intractability Risk Efficacy Tool (DIRE). Many other tools have been studied minimally or are currently being studied; however, all of the tools developed to date are susceptible to deception.
The National Institute on Drug Abuse (NIDA) offers a quick screen via interview that is followed by a more in-depth interview with the NIDA-Modified Assist Tool.20 Since the tool requires an interviewer, it may be more cumbersome for a busy practice. The ORT may be self-administered, has only five questions, can be completed fairly quickly, and shows more sensitivity to low-risk patients than the SOAPP.21 It has been commonly used in the office setting. The SOAPP involves 5-24 questions, is self-administered, and takes a little longer to complete if all 24 questions are administered.22 A newer version, the SOAPP-R, was developed to offer a more subtle approach to questioning, and still has 24 questions.23 Both tools are considered reliable and valid and could be considered if the patient has adequate time to complete the documentation. Finally, the DIRE is seven questions administered by patient interview and is used to screen for appropriateness of placing the patient on opioid therapy.24 Again, the patient must be interviewed so self-administered tools may be more desirable.
Once the patient has been placed on opioids, the Current Opioid Misuse Measure (COMM) could be administered. The COMM helps determine if patients already on opioids are abusing their prescription.25 It includes 17 questions, is self-administered, and will lead to a positive or negative result, thereby lending itself to some false positives. The COMM has been found to be reliable and valid.26 Table 3 lists websites at which the tools can be accessed for clinical use.
Table 3: Screening Tool Samples for Clinical Use – Websites

Toxicology Screening. Random toxicology screening is recommended for any patient prescribed opioids longer than 3 months. The American Pain Society (APS) guidelines for screening include testing every 3-6 months for low-risk patients and up to weekly for patients at high risk of aberrant behaviors.27 Patients should be made aware that random screening will occur if they are maintained on an opioid regimen. Urine is the most common method of screening in an office setting, but saliva, hair, blood, or stomach contents also may be screened. Provisions should be made to ensure that the patient’s own urine is what is tested.
Laboratories may vary regarding which medications are routinely included in their screens, so the prescriber should be familiar with the standard inclusions in the random screen. If unsure, the specific drug(s) that the provider is looking for should be identified on the order. In general, “ordering and interpreting urine drug screenings requires an understanding of the different testing modalities, the detection times for specific drugs, and the common reasons for false-positive and false-negative test results.”28 A good reference on urine drug screening, including false positives, timeframe for reliability, and other variables, usually can be provided by the laboratory that is contracted for screening.
Opioid Agreements. An opioid agreement should delineate specific rules that the patient must follow while being prescribed opioids. According to APS and American Academy of Pain Medicine (AAPM) clinical guidelines,29 the following components should be included:
- A discussion of the risks and benefits of opioid therapy.
- The types of common side effects and risks of adverse effects including abuse, addiction, and overdose.
- The risk of hyperalgesia, sexual, and endocrine dysfunction.
- Goals of opioid therapy.
- How the opioids should be taken; scheduled vs as needed, and frequency with guidelines for tapering and weaning. Patient should be informed when they might need to discontinue the medication, including signs of aberrant behavior.
- Expectations for office follow-up, refill process, pill counts, and random drug screening.
- Use of one prescriber and one pharmacy for opioid therapy.
- Guidance on safe storage of prescriptions to guard against theft and policy on theft replacement.
- How to properly dispose of opioids.
- Alternatives to opioid therapy.
Informed consent and a sample opioid agreement can be found in Appendix 6 and 7 at www.jpain.org/article/S1526-5900(08)00831-6/fulltext#appseca6.
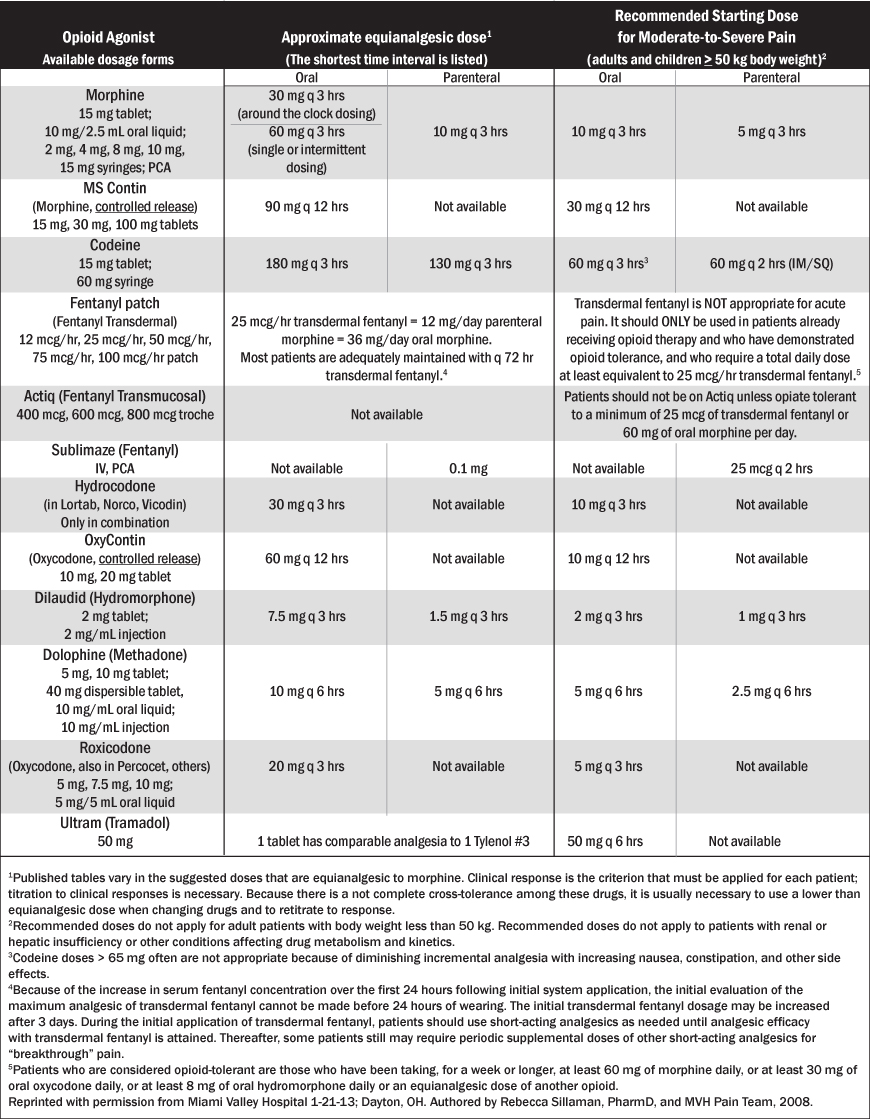
Equianalgesic Dosing Charts. Equianalgesic dosing charts provide dosing guidelines when switching from one opioid to another. (See Table 4.) Calculating an accurate conversion more likely provides a safe dose with analgesic efficacy. Making a switch in opioids without a calculation may cause oversedation or respiratory depression, or on the other end of the spectrum, inadequate analgesia. Dose conversions are most easily calculated by totaling the past 24-hour usage of all opioids into a morphine equivalent. The sum can then be used to convert to an equianalgesic amount of the newly prescribed opioid. Most references suggest using a one-third to one-half total reduction in the sum before conversion, due to the patient’s incomplete cross tolerance, resulting in an unpredictable response.29 Additionally, most references caution against initiating methadone unless experienced in its use, such as a pain specialist or palliative care specialist. The long half-life of methadone, with the cumulative effect, and the unpredictability of equianalgesic conversions, can be dangerous. Patients maintained on a stable dose for pain management could be managed by a primary care provider; special licensing to prescribe methadone only applies to opioid detoxification or rehabilitation maintenance programs.30
Table 4: Equianalgesic Opioid Table (mg)
Managing Pain with Opioids
The first step in prescribing opioids is the decision to initiate the therapy. Some decision-making steps have already been described, including the use of a screening tool and checking prescription history via a state website. Other assessment questions that have demonstrated predictive value may be utilized. Past or current use or abuse of tobacco, alcohol, or other illicit drugs all have been shown to increase the risks for future abuse, while existence of addictive behaviors, such as gambling, sex addiction, Internet addiction, kleptomania, or other impulse control disorders, are directly correlated with an increased risk of substance abuse.31,32,33 According to Grant, gamblers have been shown to have a tenfold increase in risk of substance abuse as compared with the normal population.31 Other addiction disorders demonstrate similar behaviors to substance abuse including uncontrolled use despite harm and diminished self-control due to cravings.31 Neurobiologists believe that the similarities of addictive disorders are attributable to abnormalities in serotonin production and availability.31 A discussion regarding past or current abuse behaviors should be included as part of the risk stratification for routine screening prior to opioid initiation.
Other predictors may include mental health disorders or a history of physical or sexual abuse as a child.32,33 Substance abusers report high rates of major depression and anxiety (23.5%) as well as high rates of sexual abuse (25.4%).34 As the incidence of anxiety disorders, mood disorders, or behavior disorders increases, there is a direct linear correlation with an increase in substance abuse.35 As the addiction develops, unemployment or frequent job changes, failed relationships or marital problems, involvement in litigation, seeing multiple medical providers, or “doctor shopping” may emerge.33 Successful recovery from addiction results most often from strong social and family relationships, ongoing employment with possible vocational training, and development of leisure activities.36 Without these variables present at the initiation of opioid therapy, the patient may be more vulnerable to misuse.
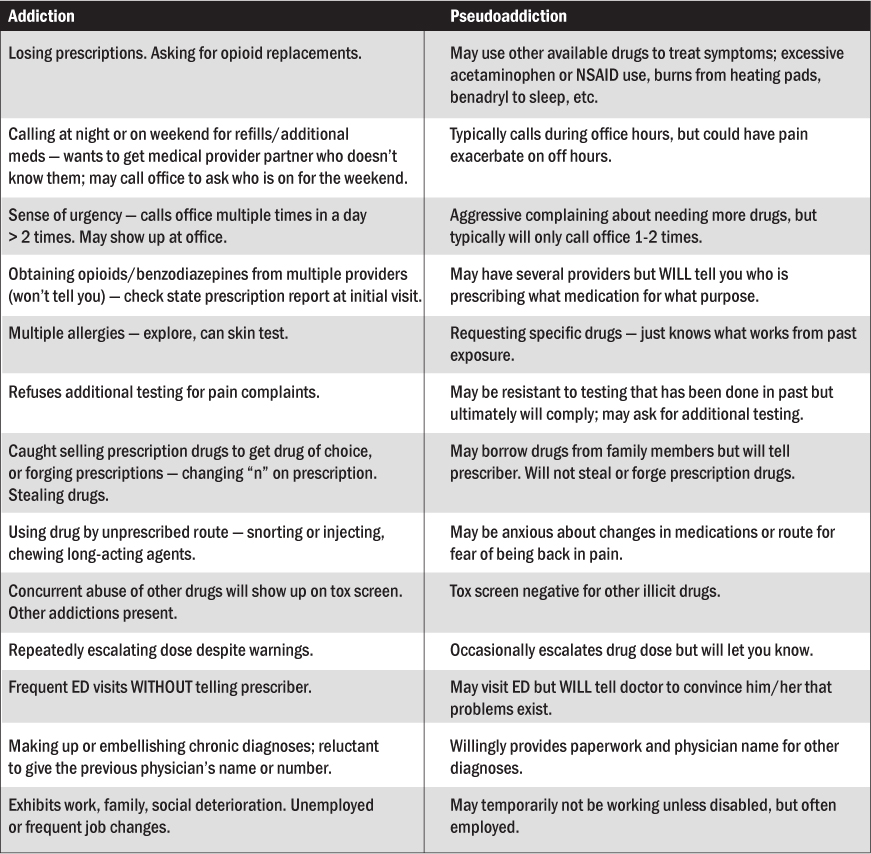
After the decision is made to initiate opioids, careful monitoring of the patient must ensue. An opioid agreement should be reviewed with the patient and signed with a copy included on the chart and a copy given to the patient. The prescriber should be alert to any developing signs of addiction. Patients with undermanaged pain may develop pseudoaddictive behaviors, which can be difficult to differentiate from addictive behaviors. Although both behaviors are not 100% consistent with each category, a comparision of the most likely categorized addictive and pseudoaddictive behaviors is included in Table 5.37
Table 5: Addiction vs Pseudoaddiction
If aberrant behaviors develop that are more indicative of addiction, the prescriber should follow the guidelines issued by the Association of Healthcare Research and Quality (AHRQ) and AAPM described later in this article, and refer the patient to an addictionologist. Patients who have altered prescriptions or sold their prescriptions have committed a felony. They should be reported to law enforcement and immediately discharged from your practice.
Referral to a pain specialist should be considered when the current medical provider becomes uncomfortable with the opioid dosing, is not seeing functional improvement with the analgesic regimen and/or intolerable side effects, and is unsure of the next steps for optimal safe care of the patient. The referral should be made with a direct phone call to the receiving pain specialist with the following information: the expectations for the referral, whether it is confirmation of the current regimen, suggestions for improvement in the regimen, further diagnostics, interventional strategies that the current provider does not offer, and the current plan.38 Frequent communication between the pain specialist and the referring physician is the key to a successful outcome.
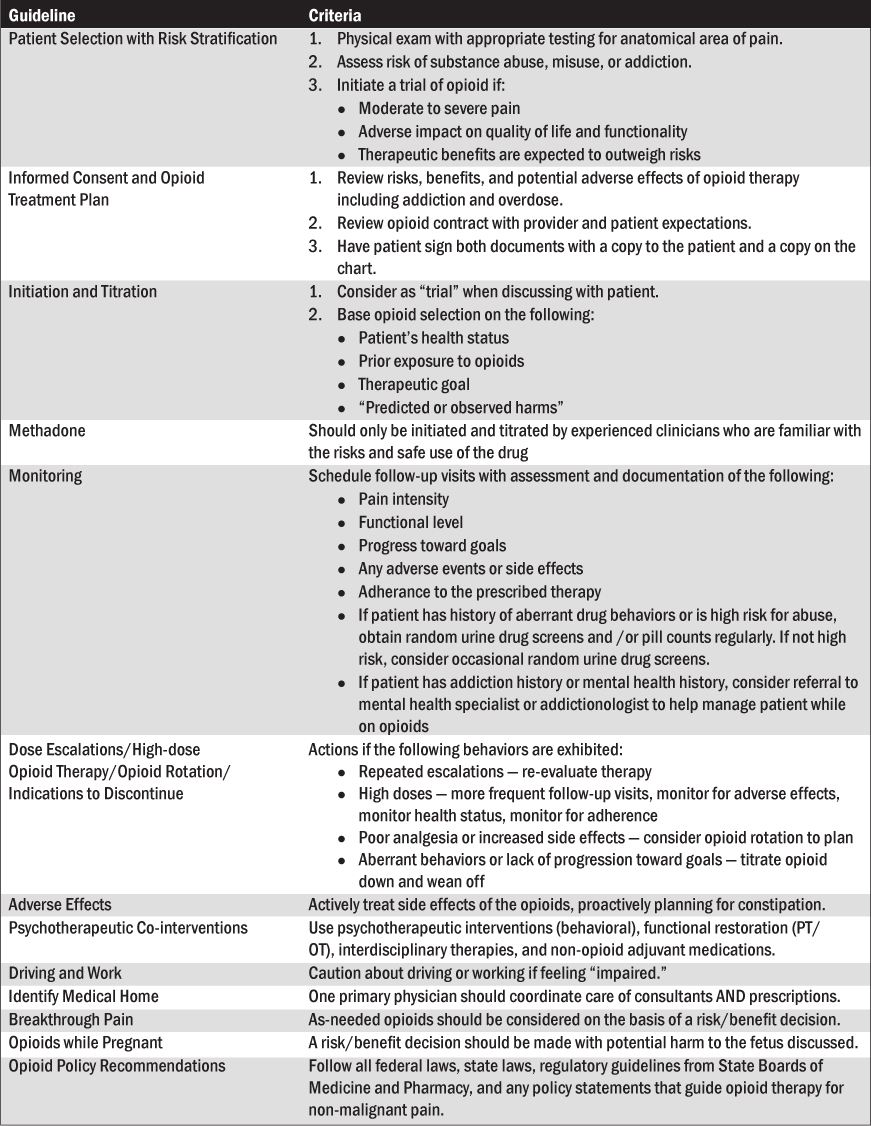
Several organizations have provided guidelines on how to manage the non-cancer patient on long-term opioid therapy based on clinical guidelines published by Chou in 2009.39 A summary of those guidelines is listed in Table 6.
Table 6: Clinical Guidelines for the Use of Opioid Therapy in Chronic Non-Cancer Pain
Managing the Palliative Care Patient on Opioids
Managing the pain of a terminally ill patient seems an obvious ethical responsibility, yet > 50-90% of cancer patients report moderate-to-severe pain at end of life.40 As many as 75% of heart failure patients in the last 6 months of life and 50% of AIDS patients describe pain related to the disease itself, comorbities, or related treatment.40 A large study conducted from 1994-2006 concluded that about 26% of patients in the last 2 years of life experienced “clinically significant” or moderate pain on a regular basis, which increased to 46% in the last month of life.41 The American Medical Association Code of Ethics is clear by stating, “Physicians have an obligation to relieve pain and suffering and to promote the dignity and autonomy of dying patients in their care. This includes providing effective palliative treatment even though it may foreseeably hasten death.”
There are many myths surrounding pain management at the end of life, including saving strong analgesics until near death.42 The reality is that strong analgesics may be needed early in diagnosis to enhance the patient’s quality of life until death.
The World Health Organization published an analgesic ladder for cancer pain, familiar to most clinicians, suggesting initiation of therapy with an oral non-opioid with adjuvant for mild pain, progressing to opioids with non-opioids (nonsteroidal anti-inflammatory drugs [NSAIDS] or acetaminophen) and adjuvants for severe pain.43 Delay in aggressive pain therapy may occur if the patient’s pain is severe, and the clinician conservatively begins treatment at the base of the ladder. In an article written by Fine in 2012, it was noted that 20% of patients with advanced disease are not adequately managed with the analgesic ladder.44 Since the undertreatment of pain is widely published, related to the palliative population, multimodal therapies including neuropathic agents and opioids should be employed.
The American College of Physicians (ACP) published practice guidelines for end-of-life care in 2008 and recommended a combination of radiotherapy/radiopharmacology, opioids, NSAIDs, adjuvant medications, and particularly biophosphonates for bone cancer from breast cancer or myeloma.45 Clinical guidelines developed regarding opioids for palliative care still recommend morphine as the primary mainstay of opioid therapy.46 A long-acting version of morphine for sustained pain, along with breakthrough dosing of a short-acting agent, is recommended. As with all opioid therapy, individual variances may require use of another opioid such as oxycodone or hydromorphone, particulary if the patient has renal dysfunction. Transdermal fentanyl preparations are not recommended for initial therapy as a level of opioid tolerance should be established prior to initiating. If the patient cannot tolerate oral therapy, subcutaneous injection of opioid should be considered.46 Fear of respiratory depression from potent opioids should not outweigh the benefit of effective analgesia.47 Many states have compassionate care laws in place that protect the physician from litigation for treating the dying patient.
The emotional component of the experience of dying cannot be ignored when managing the pain of a palliative care patient. Multiple literature sources cite depression as a major component at end of life.48 Clearly undermanaged emotional distress can exacerbate physical pain. Therapies recommended based on systematic review of the literature include treatment with tricyclic antidepressants or selective serotonin reuptake inhibitors (SSRIs), in combination with psychosocial therapies.48 Interdisciplinary management of the patient is paramount for optimizing pain control. The ACP clinical guidelines include the following recommendations (summarized)45:
- Pain, dyspnea, and depression should be regularly reassessed.
- Pain therapies utilized at end of life should be clinically proven; for cancer pain this includes the use of NSAIDs, opioids, and biophosphonates.
- Dyspnea should be actively managed with opioids and oxygen.
- Depression should be treated with tricyclic antidepressants, SSRIs, and psychosocial support.
- Advance care planning, including completion of Advance Directives, should be completed.
Risks of addiction in the palliative care population in patients who have no previous history of addiction is extremely rare.49 Presenting signs of aberrant behaviors in terminal patients are often manifestations of pseudoaddiction and undertreated pain.49 Patients who have a prior history of substance abuse will need to be monitored more closely, just as any patient with an addiction history. Since substance abuse lends itself to a higher risk of chronic disease and cancer, patients should be actively screened regardless of terminal diagnosis.
Summary
The decision to prescribe opioids for a patient with non-cancer pain is a difficult and calculated one. Patients with a substance abuse or mental health history should not be denied opioids when experiencing a co-existing pain condition that is affecting their quality of life. Careful screening with a pre-determined pain management plan is imperative for optimal management of all patients, including palliative care. When viewed as a “trial,” the patient is informed with initiation, that the use of opioids requires ongoing monitoring and follow-up. The use of available screening tools and opioid prescribing guidelines are recommended to enhance safe, quality patient outcomes. The complete text for the clinical guidelines for prescribing is available at http://www.painmed.org/Library/Clinical_Guidelines.aspx.
References
1 Drug Abuse Warning Network. Trends in Emergency Department Visits Involving Non-Medical Use of Narcotic Pain Relievers. Available at: www.samhsa.gov/data/2k10/DAWN016/OpioidED.htm. Accessed Dec. 21, 2012.
2. Centers for Disease Control and Prevention. Unintentional Poisoning. Available at: www.cdc.gov/HomeandRecreationalSafety/Poisoning/index.html. Accessed Dec. 21, 2012.
3. Centers for Disease Control and Prevention. National Youth Risk Behavior Survey, 2009. Available at: www.cdc.gov/mmwr/pdf/ss/ss5905.pdf. Accessed Dec. 21, 2012.
4. Centers for Disease Control and Prevention. Vital Signs: Overdoses of Prescription Opioid Pain Relievers — United States, 1999-2008. Morbidity and Mortality Weekly Report (MMWR), November 4, 2011;60:1487-1492. Available at: www.cdc.gov/mmwr/preview/mmwrhtml/mm6043a4.htm?s_cid=mm6043a4_w. Accessed Dec. 21, 2012.
5. Centers for Disease Control and Prevention. Illegal Drug Use. FastStats. Available at: www.cdc.gov/nchs/fastats/druguse.htm. Accessed Dec. 21, 2012.
6. Centers for Disease Control and Prevention. Policy Impact: Prescription Painkiller Overdoses. Available at: www.cdc.gov/homeandrecreationalsafety/rxbrief/index.html. Accessed Dec. 21, 2012.
7. Centers for Disease Control and Prevention. Policy Impact: Prescription Painkiller Overdoses. Available at: www.cdc.gov/homeandrecreationalsafety/rxbrief/index.html. Accessed Dec. 21, 2012.
8. Okie S. A flood of opioids, a rising tide of death. N Engl J Med 2010;363:1981-1985.
9. American Chronic Pain Association (ACPA). Glossary. Available at: http://theacpa.org/Glossary. Accessed Dec. 21, 2012.
10. National Academies Press. Relieving Pain in America: A Blueprint for Transforming Prevention, Care, Education, and Research. Washington DC: The National Academies Press. Available at: http://books.nap.edu/openbook.php?record_id=13172&page=62. Accessed Dec. 21, 2012.
11. U.S. Department of Health and Human Services. National Centers for Health Statistics. Health, United States, 2006: Chartbook on Trends in the Health of Americans 2006. Special Feature: Pain. Available at: www.cdc.gov/nchs/data/hus/hus06.pdf. Accessed Dec. 21, 2012.
12. American Diabetes Association. Diabetes Statistics. Available at: www.diabetes.org/diabetes-basics/diabetes-statistics/. Accessed Dec. 21, 2012.
13. Roger VL, et al. Heart disease and stroke statistics—2011 update: A report from the American Heart Association. Circulation 2011;123:e18-e209, page 20.
14. American Cancer Society. Cancer Prevalence: How Many People Have Cancer? Available at: www.cancer.org/docroot/CRI/content/CRI_2_6x_Cancer_Prevalence_How_Many_People_Have_Cancer.asp. Accessed Dec. 21, 2012.
15. 2006 Voices of Chronic Pain Survey. American Pain Foundation.
16. A controlled trial to improve care for seriously ill hospitalized patients. JAMA 1995;274:1591-1598.
17. H.R. 1065 (112th): Pill Mill Crackdown Act of 2011. Available at: www.govtrack.us/congress/bills/112/hr1065. Accessed Dec. 21, 2012.
18. U.S. Department of Justice. Drug Enforcement Administration. Office of Diversion Control. State Prescription Drug Monitoring Programs. October 2011. Available at: www.deadiversion.usdoj.gov/faq/rx_monitor.htm#4. Accessed Dec. 21, 2012.
19. U.S. Food and Drug Administration. Approved Risk Evaluation and Mitigation Strategies (REMS). Available at: www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformation-forPatientsandProviders/cm111350.htm. Accessed Dec. 21, 2012.
20. National Institute on Drug Abuse. Resource Guide: Screening for Drug Use in General Medicine Settings. Available at: www.drugabuse.gov/publications/resource-guide/nida-quick-screen. Accessed Dec. 21, 2012.
21. National Institute of Health. ORT: Opioid Risk Tool. Available at: www.opioidrisk.com/node/884. Accessed Dec. 21, 2012.
22. PainEDU. Opioid Risk Management. Screener and Opioid Assessment for Patients with Pain. Available at: www.painedu.org/soapp-development.asp. Accessed Dec. 21, 2012.
23. Inflexxion, Inc. SOAPP®. Available at: www.inflexxion.com/SOAPP. Accessed Dec. 21, 2012.
24. Diagnosis Intractability Risk Efficacy Tool. Available at: http://www.opioidrisk.com/node/1202; http://www.sciencedirect.com/science/article/pii/S1526590006006262. Accessed Dec. 21, 2012.
25. Pain EDU. Current Opioid Misuse Measure. Available at: www.painedu.org/soap.asp. Accessed Dec. 21, 2012.
26. Butler SF, et al. Cross validation of the current opioid misuse measure to monitor chronic pain patients on opioid therapy. Clin J Pain 2010;26:770-776.
27. Chou R, et al, American Pain Society and American Academy of Pain Medicine. Clinical Guidelines for the Use of Opioid Therapy in Chronic Non-Cancer Pain. J Pain 2009;10:113-130.e22. Available at: http://www.jpain.org/article/S1526-5900(08)00831-6/fulltext. Accessed Dec. 21, 2012.
28. Standridge JB, et al. Urine drug screening: A valuable office procedure. Am Fam Physician 2010;81:635-640.
29. American Pain Society. Principles of Analgesic Use, 6th ed. Glenview, IL: APS; 2008.
30. Medical College of Wisconsin. Gazelle G, Fine PG. Methadone for the Treatment of Pain, 2nd ed. Fast Facts and Concepts. 2006. Available at: www.eperc.mcw.edu/EPERC/FastFactsIndex/ff_075.htm. Accessed Dec. 21, 2012.
31. Grant JE, et al. The neurobiology of substance and behavioral addictions. CNS Spectr 2006;11:924-930.
32. National Institute on Drug Abuse. Preventing Drug Abuse among Children and Adolescents. Chapter 1: Risk factors and protective factors. Available at: www.drugabuse.gov/publications/preventing-drug-abuse-among-children-adolescents/chapter-1-risk-factors-protective-factors. Accessed Dec. 21, 2012.
33. National Institutes of Health. Appendix C DSM-IV-TR Material. Criteria for Substance Abuse. Opioid Dependence. Available at: www.ncbi.nlm.nih.gov/books/NBK64247/. Accessed Dec. 21, 2012.
34. Fergusson DM, et al. Exposure to childhood sexual and physical abuse and adjustment in early adulthood. Child Abuse Negl 2008;32:607-619.
35. Merikangas KR, et.al. Lifetime prevalence of mental disorders in U.S. adolescents: Results from the national comorbidity survey replication-Adolescent supplement (NCS-A). J Am Acad Child Adolesc Psychiatry 2010;49:980-989.
36. Scherbaum N, Specka M. Factors influencing the course of opioid addiction. Int J Methods Psychiat Res 2008;17(Suppl 1):539-544.
37. Portenoy RK, et al. Pain and Addiction; Chapter 17: p. 367. In: Clinical Textbook of Addiction Disorders, 3rd ed. Frances RJ, et al., eds. New York: Guilford Publications; 2009.
38. Fine PG, et al. Protect your patients, protect your practice: Practical risk assessment in the structuring of opioid therapy in chronic pain. J Fam Pract 2010;59(9 Suppl 2):S1-S16.
39. Chou R, et al. Clinical guidelines for the use of chronic opioid therapy in chronic non-cancer pain. J Pain 2009;10:113-130.
40. Broglio K, Cole BE. Pain management and terminal illness. Pract Pain Mmgt. 2008. Available at: www.practicalpainmanagement.com/resources/hospice/pain-management-terminal-illness. Accessed Dec. 21, 2012.
41. Smith AK, et al. The epidemiology of pain during the last 2 years of life. Ann Intern Med 2010;153:563-569.
42. Hospice Foundation of America. Myths about pain. Available at: www.hospicefoundation.org/painmyths. Accessed Dec. 21, 2012.
43. World Health Organization. Cancer. WHO’s pain ladder. Geneva, Switzerland. Available at: www.who.int/cancer/palliative/painladder/en/. Accessed Dec. 21, 2012.
44. Fine PG. Pain management and end-of-life care. Pain Mgmt 2012;2:325-328.
45. Qaseem A, et al. Evidence-based interventions to improve the palliative care of pain, dyspnea, and depression at the end of life: A clinical practice guideline from the American College of Physicians. Ann Intern Med 2008;148:141-146.
46. National Institute for Health and Clinical Excellence, 2012. Opioids in palliative care: Safe and effective prescribing of strong opioids for pain in palliative care of adults. NICE Clinical Guideline: Available at: www.nice.org.uk/nicemedia/live/12953/58519/58519.pdf. Accessed Dec. 21, 2012.
47. Jackson V, Nabati L. 2012. End of life care: Ethical considerations in pain management. Up To Date Available at: www.uptodate.com/contents/end-of-life-care-ethical-considerations-in-effective-pain-management. Accessed Dec. 21, 2012.
48. Lorenz KA, et al. Evidence for improving palliative care at the end of life: A systematic review. Ann Intern Med 2008;148:147-159.
49. National Cancer Institute, 2011. Substance abuse issues in cancer. Available at: http://www.every-dayhealth.com/health-center/substance-abuse-issues-in-cancer-pdq-supportive-care-health-professional-information-nci.aspx. Accessed Dec. 21, 2012.
The national epidemic of substance abuse naturally imparts uncertainty and fear in the decision-making process regarding the prescribing of opioids for patients in pain. Statistics alone substantiate the need to validate our decisions with a documented plan of care.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
