An Update on Dyspepsia
An Update on Dyspepsia
Authors: Joseph R. Baber, DO, FACP, Gastroenterology Fellow, Wright State University, Boonshoft School of Medicine, Dayton, OH
Ramesh K. Gandhi, MD, FACG, Clinical Associate Professor of Medicine, Wright State University, Boonshoft School of Medicine, Dayton, OH
Peer Reviewer: Sheila E. Crowe, MD, Professor of Medicine, Division of Gastroenterology, Department of Medicine, University of California at San Diego, La Jolla, CA
Introduction
Dyspepsia represents a common yet nebulous symptom complex encountered in the daily clinical practice of primary care physicians, and it is frequently used synonymously with many different sets of symptoms marked by the presence of upper abdominal pain or discomfort. Since most patients do not recognize this term and because many physicians have interpreted the meaning of this symptom differently, controversy persists over its identification as a clinically distinct entity and pursuit of evidence-based management strategies to apply to patients with this heterogeneous disorder. Derived from the Greek words meaning “difficult digestion,” dyspepsia refers to upper abdominal discomfort, but not necessarily pain, ranging from bloating, early satiety, and nausea and vomiting. The Rome III consensus committee has established more specific criteria in an attempt to identify symptoms that originate from the gastroduodenal region. Bothersome postprandial fullness, early satiation, epigastric pain, and epigastric burning are four symptoms considered to be more specific as opposed to heartburn, which was removed from prior definitions.1
Although most patients do not seek medical care, those who do present often are identified for symptoms that fit a particular pattern of known or organic pathophysiology. After an evaluation down one path leads to an unsuccessful clinical outcome, frustration and confusion can result. In this unfulfilling process, patients may sense their symptoms not being validated and pursue care elsewhere or go untreated. It is essential that primary care physicians identify this symptom complex and establish trusting patient-physician relationships before applying a systematic approach to this disorder. Incorporating a comprehensive approach that bears in mind the bio-psychosocial patient care model becomes just as important in validating patients’ concerns and fears while providing a road map for trustworthy continuity of care that avoids additional somatic and/or psychological anguish and minimizes redundant health care expenses.
Epidemiology
Dyspepsia is common in the general population, accounting for a frequency ranging from 10-45% with a slightly higher female to male predominance ratio of 1.4:1. According to a recent case-control analysis obtained in the Southwestern United States, Hispanic persons had a significantly higher risk of dyspepsia and positive Helicobacter pylori testing. The problem with the accuracy in identifying this subgroup of patients revolves around the way in which dyspepsia is defined. Because typical symptoms of gastroesophageal reflux disease (GERD) have been included in some prevalence studies in the past, when heartburn is excluded, the actual frequency of dyspepsia drops to 5-15%. The annual incidence rate in the general population ranges from 1-6%. Health care costs for dyspeptic patients are 54% higher than controls, even before the formal diagnosis is made.2,3
Etiology
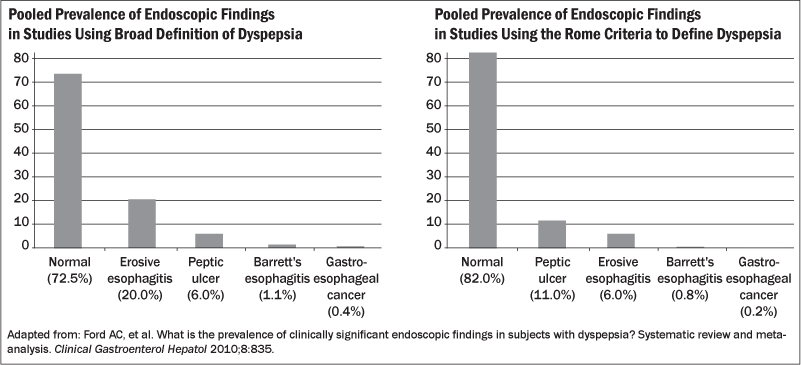
Bearing in mind that dyspepsia is a symptom complex, a rigorous search for the source of this discomfort must be considered. Despite the attention to peptic ulcer disease (PUD) as a cause for dyspeptic symptoms in up to one-quarter of patients by some estimates, recognize that patient symptoms thought to be reflective of ulcer disease hold poor discriminant value. Upper endoscopy finds its niche as a diagnostic test in the workup of uninvestigated dyspepsia. A systematic review and meta-analysis investigating prevalence of endoscopic findings in individuals with broadly defined dyspeptic symptoms have identified erosive esophagitis as the most common finding encountered at endoscopy with a pooled prevalence of 20% followed by gastric or duodenal ulcer rate at 8%. This finding was not replicated when more strict Rome criteria were used to define the same study population, with PUD leading the cause for organic pathology encountered. It is reassuring that both Barrett’s esophagus and upper gastroesophageal malignancy are exceedingly rare sources for patients presenting with dyspeptic symptoms at just about 1% as depicted in Figure 1.4,5 GERD remains another important identifiable source of dyspepsia symptoms, but in the setting of typical reflux-like symptoms, applying the diagnosis of GERD is appropriate and not attributing this as part of the dyspepsia symptom complex as it was in earlier iterations of the dyspeptic definition. Nevertheless sometimes reflux still presents atypically as dyspepsia and this underscores the important overlap between silent GERD and dyspeptic symptoms.
Figure 1: Prevalence of Clinically Significant Endoscopic Findings in Individuals with Dyspepsia
In some people, dyspepsia may develop as a result of a genetic predisposition, whereas others may be exposed to environmental factors including a prior viral infection, inflammation, stress, trauma, surgery, or H. pylori. Hormonal influences play a factor in the symptoms of others (see Table 1).
Table 1: Development of Functional Dyspepsia

The vast majority of patients in the community setting who present to primary care physicians will not have an identifiable systemic or metabolic cause. After an investigative evaluation, including upper endoscopy, is performed among patients who meet Rome III symptomatic criteria, these patients can be identified as having nonulcer dyspepsia (NUD) or more universally termed functional dyspepsia (FD), where a clear pathophysiological mechanism for these symptoms becomes less well defined.
Clinical Features
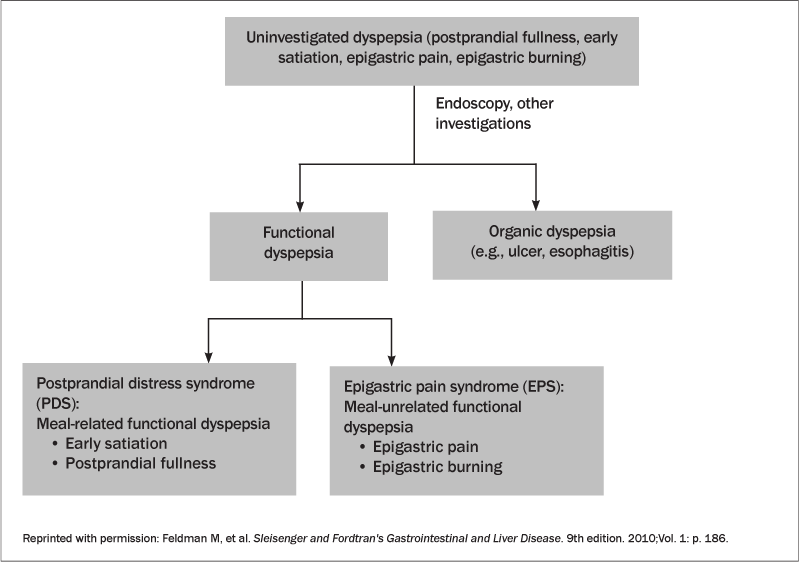
Patients with FD present in a variety of ways. Although their symptoms are marked by the presence of upper abdominal discomfort, they may not necessarily report pain. They may describe their discomfort as either a bothersome postprandial fullness, describe early satiation, or even report epigastric burning. The onset of symptoms is not associated with a change in bowel habits, distinguishing it from another functional disorder, irritable bowel syndrome (IBS). There must be a component of chronicity associated with these symptoms occurring for the last 3 months and with at least a 6-month time frame before the diagnosis of functional dyspepsia is applied (see Table 2). The astute physician can sometimes tease out either a predominant meal-time component or reflux component to the symptoms patients report. FD has been subcategorized into post-prandial distress syndrome (PDS) with meal-related symptoms involving repetitive fullness to ordinary sized meals or early satiation that prevents finishing a regular meal. Alternatively, it can be categorized as epigastric pain syndrome (EPS) with intermittent burning or pain localized to the epigastrium occurring at least weekly as the predominant symptom that may or may not be related to food. It would be nice to define all patients in this way, but many times patients exhibit overlapping symptoms of both subgroups (see Figure 2). This not only helps the current understanding of this heterogeneous disorder for research methodology but also offers implications on selecting therapeutic treatment modalities.6
Table 2: Diagnostic Criteria of Functional Dyspepsia

Figure 2: Classification of Dyspepsia according to Rome III Criteria
Pathophysiology
The pathogenesis of functional dyspepsia is diverse. Like IBS and many other functional disorders that are defined by symptom criteria, a broad array of contributing factors have been proposed, and all likely interplay in varying degrees among patients who report dyspeptic symptoms (see Table 1).
The most robust research exists on alterations in gastroduodenal motor and reflex function. The mechanical digestive process within the stomach is well regulated. The distal stomach grinds food contents until they are churned to be small enough to pass easily though the pylorus. The proximal stomach acts as a reservoir during and after ingestion, which allows more complete distension to accommodate large food boluses without a rise in intragastric pressure. This process is tightly regulated by the vagus nerve and is thought to be adversely affected in approximately 40% of patients with functional dyspepsia.7,8 Compensatory activation of tension-sensitive mechanoreceptors in either proximal or distal stomach is felt to account for the perceived discomfort that some patients experience.
A delay in gastric emptying has been noted to occur in roughly 30% of patients with dyspepsia symptoms. It is plausible to understand how residual gastric contents can result in early satiation and postprandial fullness followed by nausea and emesis. A large U.S. multicenter study, however, found no association between impaired gastric motility and patient symptoms, suggesting that delayed gastric emptying may be an epiphenomenon rather than a pathophysiologic mechanism for disease.9 Accelerated gastric emptying also may occur, but again it remains difficult to select those who become symptomatic from these gastric emptying derangements on either end of the spectrum. In a recent publication, Lacy contends that because of the significant overlap of symptoms of gastric neuromuscular dysfunction exhibited in FD with idiopathic gastroparesis, the pathophysiologic processes are more similar than they are currently defined in the literature.10
Impaired visceral sensation has been strongly proposed in the pathogenesis of functional dyspepsia. Post-infectious bowel irritation has been cited in recent studies contributing to selected patients with IBS. Similar findings have been reported in patients following a Salmonella gastroenteritis infectious outbreak, with the thought that residual mucosal inflammation along with alterations in gut immune function and changes in the enteric nervous system contribute to altered visceral sensation.11 The mast cell mucosal proliferative response seen in some patients with H. pylori-negative FD have suggested a mechanism by altering signaling in the brain-gut axis.12 Tack et al found that patients were hypersensitive to isobaric gastric distension, and as a result of this we recognize and target visceral hypersensitivity as a modulating factor in treating those with FD. This phenomenon is amplified in patients afflicted with underlying psychosocial stressors, most notably affective disorders, somatoform disorders, and prior history of physical or sexual abuse.7
It has been known that the response to chemical stimuli including acid, nutrients, and capsaicin at the mucosal surface of the duodenum play an important role in enhancing gastroduodenal mechanosensitivity and resultant dyspeptic symptoms.13 It has been postulated that serotonergic signaling pathways may be involved in the gastric sensorimotor function; however, recent randomized controlled studies of selective serotonin reuptake inhibitors and selective serotonin and norepinephrine reuptake inhibitors have not substantiated this postulation clinically.14,15 A Japanese group was able to replicate dyspeptic symptoms by duodenal acidification using transnasal endoscopy and reported the induction of dyspeptic symptoms more significantly in patients with FD than in healthy volunteers.16 A recent investigation sought to determine whether visceral hypersensitivity in FD is a manifestation of a central sensitization also encompassing somatic sensitization. Capsaicin was used as a chemical stimulant to trigger the transient receptor potential vanilloid-1 pathway involved in gastric mechanosensory physiology, and in a small, double-blind, randomized study, the dose required to trigger moderate pain in the FD arm was half that required to do the same in the control arm.17 This interesting physiologic study points to a specific pathway that provides a rationale for the treatment of dyspeptic symptoms, verifies the mechanism of action of current agents that have been studied, and is thought to decrease visceral hypersensitivity via this mechanism.
Lipid digestion slows gastric emptying to enable further breakdown and absorption of these ingested particles. Once sensed by the duodenum, cholecystokinin is released and the perception of gastric distension normally should be appreciated. Altered duodenal sensitivity to lipids exacerbates this process in some, which results in discomforting symptoms. Data suggest similar duodenal hypersensitivity to acid, which also induced nausea in patients with FD.18
The only known genetic link to dyspepsia has been identified as polymorphisms of the GNB3 gene that encodes guanine nucleotide binding protein, beta polypeptide 3. Homozygous variants (CC and TT) have been identified in one U.S. community study as an associated factor in meal-unrelated dyspepsia, but clinical testing for this is not practical or cost effective in the primary care setting.19
Differential Diagnosis
A thorough history to tease out GERD, IBS, biliary pain, and other organic diseases, in addition to iatrogenic causes for dyspepsia caused by commonly prescribed medications, is a useful starting point in the office. In the presence of typical reflux symptoms, a provisional diagnosis of GERD should be made, and only after a sufficient trial of unsuccessful antisecretory therapy should the diagnosis be challenged by upper endoscopy and confirmed by 24-hour pH testing if necessary. IBS can overlap with FD making these two functional disorders difficult to identify at times, but reviewing the Rome III symptomatic diagnostic criteria for each will help delineate these two disorders. Keep in mind that the presence of IBS does not exclude the diagnosis of any functional gastroduodenal disorder, including FD.6 Although the pain of biliary colic also can be meal-related like dyspepsia, it is usually severe, unpredictable, and prolonged as it builds in intensity over 15 minutes before slowly abating. History alone should suffice to separate these two disorders and physicians should avoid routinely ordering abdominal ultrasounds for this purpose.
Drug-induced dyspepsia by nonsteroidal anti-inflammatory drugs (NSAIDs) is exceedingly common and often overlooked if patients are not asked specifically about over-the-counter (OTC) medication uses. Calcium channel blockers, alendronate, iron and potassium supplements, oral contraceptives, orlistat, methylxanthines, and selected antibiotics including metronidazole and erythromycin can be culpable pharmacologic agents prescribed in the primary care setting.20 Consider stopping the suspected drug while assigning a clinical diagnosis of drug-induced dyspepsia and reserve endoscoping for those whose symptoms fail to respond. Chronic NSAID use, particularly when associated with epigastric tenderness, is more suggestive of PUD than classic historical ulcer symptoms obtained by history itself. The importance of obtaining a diligent social history to exclude the OTC use of NSAIDs cannot be overemphasized as an initial diagnostic workup for FD.
Foods also can cause dyspeptic symptoms and history of allergic reactions should be ascertained with consideration for a competing diagnosis of eosinophilic gastroenteritis. Lactose intolerance can present with abdominal discomfort and bloating that can be misdiagnosed as dyspepsia. Celiac disease can present with dyspepsia, and bloating may be the initial manifestation of this autoimmune enteropathy even in the absence of iron deficiency, transaminitis, or coexistent autoimmune diseases like type 1 diabetes. Given the relative frequency of celiac disease, the cost of serologic screening, and the potential for treatment, many experts would recommend screening for this elusive disease in unexplained dyspepsia either by serologic testing or by obtaining duodenal biopsies should the patient go on to an esophagogastroduodenoscopy (EGD). Some diabetics have associated autonomic dysfunction that can affect normal gastroduodenal motility and sensation. Nondiabetic (idiopathic) gastroparesis has significant overlap with functional dyspepsia; however, quantifying the delay in gastric emptying may not change management as prokinetic therapy-altering emptying does not always correlate with symptoms.
Aside from simple metabolic abnormalities of the thyroid or hypercalcemia, rarer causes of upper abdominal discomfort include heavy metals, acute intermittent porphyria, angioneurotic edema, familial Mediterranean fever, as well as chronic intestinal angina, superior mesenteric artery syndrome, steatohepatitis, IBS, or connective tissue diseases, which may rarely masquerade as dyspepsia and should be considered when subsequent management is required after initial therapy treatment approaches have been unsuccessful. The chance of chronic pancreatitis or small bowel pathology is rare but may be identified on abdominal imaging.
Careful inquisition for alarm features that raise the suspicion of gastric malignancy should be performed. Unintended weight loss generally exceeding 10% is concerning. Persistent vomiting, hematemesis, or progressive dysphagia or odynophagia can be presenting features of organic esophagogastric pathology. Further features include anemia, bleeding, a history of peptic ulcer, jaundice, an abdominal mass, lymphadenopathy, a family history of upper GI tract cancer, previous gastric surgery, or malignancy (see Table 3). In the United States, any patient 55 years of age or older manifesting new symptoms of dyspepsia also should undergo EGD to exclude malignancy as well as those younger patients presenting with alarm features. The value of these alarm features lie in their strong negative predictive value. A large meta-analysis of 15 studies including more than 57,000 patients found that patients without any alarm symptoms had more than a 97% chance of not having an underlying malignancy discovered on endoscopy.21
Table 3: Indications for EGD in Patients with Dyspepsia

Strategies to Investigate Dyspepsia
After obtaining a history and physical from an initial patient encounter and possibly performing some preliminary testing, the pretest probability of the differential diagnosis will help delineate the diagnostic strategy forward. Empiric antisecretory therapy can be offered to young patients without alarm features where the prevalence of H. pylori is low. For younger patients residing in an area with high prevalence of H. pylori, a test-and-treat strategy can be employed as long as there is a plan to reassess symptoms within 8 weeks. A third costly and invasive strategy involves pursuing upper endoscopy, serving as the gold standard diagnostic test for establishing a specific cause in patients with upper abdominal pain.
Empiric antisecretory therapy with proton pump inhibitors (PPI) for uninvestigated dyspepsia offers particular advantages in the primary care setting. It is relatively inexpensive and provides symptomatic relief when dosed for a sufficient duration of time (at least 8 weeks) while healing lesions in those with erosive esophagitis or PUD. This is an effective option provided the patient resides in an exceedingly low (< 10%) prevalence area for H. pylori.23 They also provide benefit to up to one-third of patients with FD. The disadvantage to this approach is that they can mask pathologic symptoms by promoting partial healing of peptic ulcers leaving them prone to recur when H. pylori is a confounding yet unsuspected actor. Reflux symptoms may recur rapidly after cessation of therapy in some as a result of acid rebound leaving them reluctant to wean. Concerns exist regarding the theoretical side effect of vitamin B12 deficiency arising from longstanding acid suppressive therapy as well as the proper absorption of other vitamins and minerals. While iron sulfate and calcium carbonate require acid for absorption, calcium citrate and calcium in dairy products remain bioavailable in the setting of achlorhydria and can be advised to patients who require acid suppressive therapy. The increased association of Clostridium difficile infection remains a real threat, particularly to hospitalized patients receiving PPI therapy and efforts should continue to be made to review their indication in this setting.
The test-and-treat approach enables a logical and evidence-based approach to the management of uninvestigated dyspepsia. It is predicated on the inherent prevalence of H. pylori within the population in question. The American College of Gastroenterology (ACG) and the American Gastroenterological Association (AGA) prefer either the fecal antigen or urea breath test for determining active H. pylori infection over the serologic test. Cost-effective models have not determined that there is a significant difference between a test-and-treat approach vs empiric antisecretory therapy, but is more than likely to be beneficial in areas where the H. pylori infection rate is high. The disadvantages of this approach include exposure to antibiotic resistance and C. difficile. Furthermore, eradication of H. pylori does not always abolish symptoms and, ironically, at least one-third of patients will remain or become symptomatic because of unmasking of underlying GERD or because their symptoms were caused by underlying FD and not the H. pylori.20
Although EGD is the most expensive approach, some argue that the upfront expense may be justified in a patient with significant anxiety and fears of malignancy who may ultimately return for care after completion of alternative strategies or seek care from other providers and ultimately end up with endoscopy. It simply is not practical to send all patients with dyspepsia symptoms to upper endoscopy due to its associated cost and the associated risks with the invasiveness of this procedure, as well as the limitations in health care resources. A meta-analysis of five trials comparing initial endoscopy with a test-and-treat approach showed that endoscopy may reduce the risk of recurrent dyspeptic symptoms but at the expense of cost effectiveness.22 The evidence-based value of patient reassurance with this strategy remains largely unsubstantiated.
Diagnostic Approach and Considerations
After a thorough history and physical are obtained to develop a tiered differential diagnosis, a reasonable and cost-effective workup can be pursued. The physical exam for a patient thought to have FD is unrevealing and serves to rule out alarm features such as jaundice, lymphadenopathy, and abdominal mass as well as to examine for competing biliary or musculoskeletal conditions. Routine laboratory testing is limited to a CBC to complete the alarm feature assessment for anemia that cannot be asked directly during the patient encounter. Based on the pretest probability of the differential diagnosis obtained on the history and physical, additional labs and studies may be appropriate.
Age remains one of the most important determinant risk factors for the probability of identifying a malignancy. In the United States, where the prevalence of gastric carcinoma is far less than in certain Asian countries like Japan, using an age cutoff of 55 years in those lacking alarm symptoms, there remains only a 0.5% risk of missing an occult malignancy, which is deemed to be the threshold for acceptable risk by the guidelines put forth by the ACG.23 Patients > 55 years of age or < 45 years of age with risk factors for gastric malignancy — including those with a family history, those who emigrated from a country with a high rate of gastric cancer, or those who had a prior partial gastrectomy — will require upper endoscopy as the first-line diagnostic study, considering that most patients with newly diagnosed gastric cancer are already incurable at the time of diagnosis (see Table 4).
Table 4: Treatment Approach to Patients with Functional Dyspepsia

Conversely, the widespread use of EGD for GERD recently has been reviewed by the Clinical Guidelines Committee of the American College of Physicians to describe the indications for and yield of this study in helping primary care physicians make high-value patient care decisions.24 Because of the significant overlap between symptoms of dyspepsia with those who describe reflux disease, the judicious use of EGD is important to prevent the overall higher health care costs without improving patient outcomes. White men older than age 50 who are obese and smoke with longstanding symptoms for more than 5 years’ duration and nightly reflux symptoms represent the highest risk for developing Barrett’s esophagus and may benefit from a one-time screening upper endoscopy. Clear evidence demonstrates that men who are younger than 50 or women of any age with longstanding classic reflux symptoms should not be screened in this fashion, as they represent such a low absolute risk for esophageal adenocarcinoma.
In patients with risk factors and suspicion for malignancy or PUD but who either decline or are not good candidates based on risk to undergo upper endoscopy, a barium swallow with upper gastrointestinal series remains a formidable second-line test to assess for overt luminal abnormalities such as large ulcers or mass lesion causing gastric obstruction. Malignant ulcers tend to have irregular margins while benign ulcers appear smooth. This test also enables the primary care physician to make a rudimentary determination regarding gastric emptying. A large obstructive mass creating gastric outlet obstruction will be evident to the naked eye, but the radiologist reading the sequence of fluoroscopically timed films also will be able to make a comment on the amount of time the barium spends in the stomach and determine whether a motility disorder is present. Determining whether this gastric motility derangement is accountable for the patient’s symptoms is debatable, and the position from Rome III is that a gastric-emptying study currently is not recommended as a routine clinical test because the results uncommonly alter management.6
Delayed gastric emptying is commonly identified in patients with dyspepsia. One study estimates the overlap to be between 30% and 80%.25 Gastroparesis is a well-known and documented entity in patients with underlying diabetes and is sometimes not considered in nondiabetic patients presenting de novo with dyspepsia symptoms. Gastric emptying studies can document and quantify the severity of gastric emptying delay by measuring the half-life of a radioactively labeled egg sandwich consumed. The most reliable method to establish the diagnosis of gastroparesis is gastric retention of solids at 4 hours measured by scintigraphy, whereas studies of shorter duration or based on a liquid challenge result in decreased sensitivity.26 This must be considered in centers that offer only a 90-minute or 2-hour study. Subsequent liquid phase gastric emptying studies still may hold clinical value to help primary care physicians determine if a patient is able to tolerate a liquid diet to maintain adequate nutrition.
Testing for active H. pylori plays an important role in the test-and-treat strategy as described above and requires clear guidance on the interpretation of positive studies at the time in which they are ordered. H. pylori is second to only NSAIDs as the leading culprit of PUD in the United States. While PUD constitutes a minority (5-15%) of cases of chronic dyspepsia, most of these patients have H. pylori infection.27 If the decision is made to test for H. pylori in patients with uninvestigated dyspepsia, the prevalence of this infection should first be determined because rates vary widely among populations within the United States. Asian, black and Hispanic Americans have higher rates of H. pylori infection than white Americans.3 Immigrant communities also may contribute to a rise in H. pylori prevalence. Primary care physicians practicing in populations exceeding 10% prevalence should consider this strategy, whereas physicians who practice in less prevalent communities may proceed with empiric antisecretory trial.23
Furthermore, the type of test utilized to determine H. pylori infection makes a difference. The most accurate noninvasive method to assess active infection is by stool antigen testing or with either nonradioactive C13 or radioactive C14 urea testing. When performing a urea breath test, expired breath is collected for measurement of labeled carbon dioxide. These tests accurately detect active infection either before or after treatment of H. pylori, unlike the IgG serology that signifies infection at any time in the past and not necessarily a current active infection. It is important to note that stool or breath testing can yield false negative results in patients who recently took antibiotics, bismuth-containing compounds, PPIs, or possibly H2-receptor antagonists. Patients should be instructed to stop PPIs at least 2 weeks before a urea test is scheduled. Physicians must remember that follow-up testing also should be delayed at least 4 weeks after completing eradication therapy to optimize accurate results. The serologic test has lower sensitivity in all patients and low specificity, especially in the elderly or those with cirrhosis. Results from one of the largest randomized trials, which included 500 patients with uninvestigated dyspepsia, revealed that after 1 year, 60% fewer endoscopies were performed without a statistically significant difference in symptoms, quality-of-life scores, or recurrent physician or hospital visits.28 The gold standard remains histology with special stains obtained only after EGD with biopsy.
Ultrasonography holds no role in the evaluation or management of dyspepsia in the absence of typical biliary pain or suggestive biochemical test results, and is not recommended as a routine clinical test in the evaluation of dyspepsia because of its low diagnostic yield. This is an important clinical pearl to avoid being led down the biliary pathway, given the propensity for incidental findings resulting in unintended consequences that may not affect the presenting symptom of dyspepsia. Nevertheless, it sometimes can be difficult to ascertain typical biliary pain by history, particularly among patients with language barriers or other communication gaps. Although clinical evidence to correlate with cholecystitis warrants surgical referral, incidental findings of cholelithiasis do not. It becomes even more imperative after gallstones are detected to illicit a good history of biliary pain rather than bloating or other symptoms of dyspepsia that will not improve following cholecystectomy.
Management
Uninvestigated Dyspepsia. The initial management strategy for uninvestigated dyspepsia involves assessing risk for malignancy or PUD and symptoms consistent with reflux disease in all patients. Those with alarm features or who are ≥ 55 years of age should proceed directly to upper endoscopy. Patients taking chronic NSAIDs, even with concomitant antisecretory therapy, also are at increased risk to develop ulcer disease with complications such as gastric outlet obstruction, which is very hard to ascertain by history alone. These patients also should proceed with structural examination if a trial off therapy does not abate symptoms.
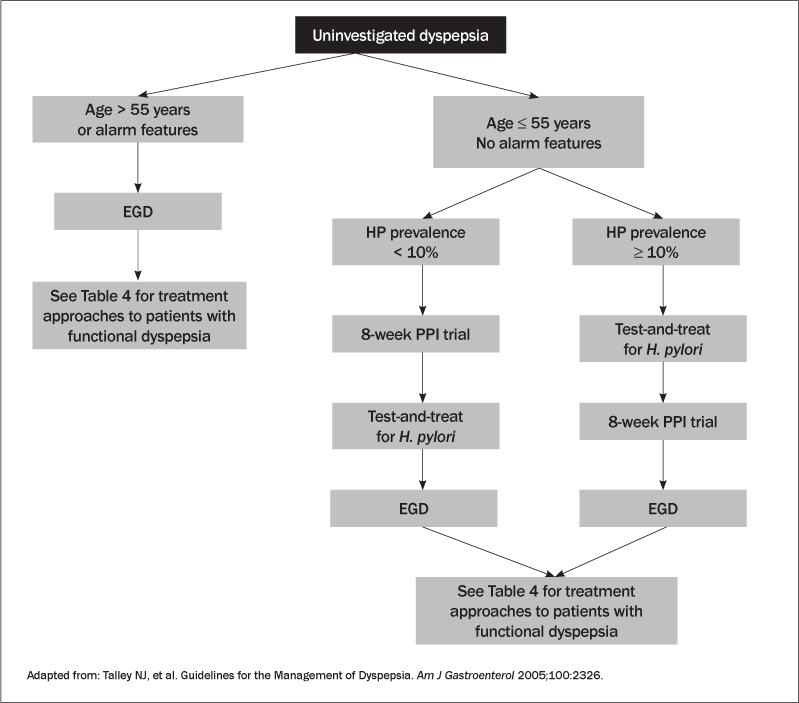
The management of younger patients with uninvestigated dyspepsia without risk factors for PUD becomes more challenging. The ACG and AGA recommend a test-and-treat approach followed by acid suppression therapy trial if symptoms persist. The American Society of Gastrointestinal Endoscopy also agrees with this initial approach with endoscopy offered electively to those who are found to be H. pylori negative. All three societies agree that upper endoscopy should be recommended after a trial of unsuccessful antisecretory therapy, preferably with PPIs. Where the prevalence of H. pylori is exceedingly low (< 10%), the ACG advises forgoing H. pylori testing altogether and proceeding with a 4- to 8-week trial of PPI first (see Figure 3).
Figure 3: Algorithm for the Management of Uninvestigated Dyspepsia
Investigated (Functional) Dyspepsia. The algorithmic approach above is well-defined to correctly identify those at risk for life-threatening symptoms or treatable ulcer disease, but the vast majority of patients with resultant nonulcerative FD may be left in the same predicament with symptoms unchanged. FD remains one of the most common forms of dyspepsia in the primary care setting and requires added evidence-based attention. The present paradigm to management involves overlapping antisecretory therapies, H. pylori eradication therapies, and prokinetic therapies.
Establish Strong Patient-Physician Relationship. A holistic approach to the psychosocial stressors affecting patients with functional dyspepsia as well as biomedical factors that afflict the patient is important. Careful anticipatory guidance is important to establish a tiered approach to address this condition with the expectation set that ruling out serious disease in a well-planned approach is important even if a trial of acid suppressive medicines and/or combination antibiotic therapy has failed. Explaining to the patient that the current diagnostic Rome III criteria requires 3 months duration of dyspeptic symptoms with symptom onset at least 6 months prior to the diagnosis without structural explanation for their symptoms can help frustrated patients realize that this is a chronic, involved disorder that applies to them and requires time to fulfill the criteria. Re-establishing new goals and validating patient symptoms is crucial to maintaining the trust of patients when they may face the crux of seeking care elsewhere or disregarding further medical evaluation altogether.
Frame the conversation in this context: To achieve the best success, a multidisciplinary approach will be instituted together with adjustments based on patient feedback along the way. This impresses upon the patient the need to replace the single-treatment, trial-and-error approach with a comprehensive plan to address multiple factors including the psyche. Set a collaborative environment from the start that involves the patient as part of the team so that he or she becomes more actively involved in the care plan and feels comfortable bringing up new factors that may have been subconsciously suppressed. Recruit the patient to document his/her symptoms by a daily written log to serve as an objective reference point to determine future improvement in addition to what is documented in the medical record. Establish a reasonable and achievable goal of achieving 50% symptomatic improvement 50% of the time rather than eliminating symptoms altogether.29
Lifestyle and Dietary Modification. Considering the high rate of delayed gastric emptying in patients with functional dyspepsia, this becomes an easy empiric and risk-free starting point to adjust lifestyle habits. Applying the same physiologic concepts understood about digestion and treatment of gastroparesis makes good sense. Educating patients on limiting fat and fiber consumption, which delays gastric emptying, is important. Patients should avoid aggravating foods, similar to the dietary modifications recommended for GERD. A food diary may be used to identify specific foods that trigger symptoms. Foods frequently reported to worsen dyspeptic symptoms include onions, peppers, citrus fruit, coffee, carbonated beverages, and spices.30 Despite the lack of formally assessed clinical evidence to demonstrate causation with specific foods and functional dyspepsia, this is a low-cost, low-risk therapeutic option that may provide symptomatic benefit.
Altering eating habits to smaller, more frequent meals is a necessary part of any treatment plan for gastroparesis and can easily be employed for those who have FD with meal-related symptoms. Primary care physicians play a pivotal role in actively managing the medicine chest for their patients and can make tremendous strides to limit polypharmacy with drug interactions that delay gastric emptying. Wean and discontinue opiods and benzodiazepines wherever possible. Recruit the assistance of physiatrists for creative, nonopiod analgesic alternatives or interventional therapies where applicable. Recognize that electrolyte homeostasis is a requirement for proper gastric motility and ensure potassium, calcium, magnesium, and phosphorus serum levels are intact. A few metabolic syndromes, including hypothyroidism, can contribute to impaired gastric emptying. Adrenal insufficiency manifests in subtle ways and includes early chronic nausea and vomiting with gastrointestinal symptoms before frank hypotension and multisystem decompensation ensue. When symptoms persist, many authorities support obtaining a 4-hour, solid-phase gastric emptying scan, although this cannot distinguish a dyspeptic patient from a nondiabetic patient with gastroparesis, given that 30% of patients with FD have delayed gastric emptying.10
Pharmacotherapy. Drug treatment for nonulcer or FD is limited and only marginally effective; however, recent randomized trials have established efficacy of certain agents over placebo. Effective treatments include tricyclic antidepressants, H2-receptor antagonists, and PPIs, even if patients had little to no response to prior trials before their diagnostic EGD. The evidence on antacid, sucralfate, and bismuth has not met statistical significance and should be abandoned as ineffective for this indication. Investigational therapies, including gastric fundus relaxation techniques with buspirone and sumatriptan as well as prokinetic agents and visceral analgesic medications, hold limited promise.
Tricyclic Antidepressants. Tricyclic antidepressants or trazodone prescribed in low doses both have been useful in functional gastrointestinal disorders to target visceral hypersensitivity. Their sedating side effect may serve as a positive influence in younger patients with concomitant insomnia and impaired visceral perception in those with FD.31,32 Amitriptyline 10-25 mg at night or trazodone 25-50 mg at night can be initiated and uptitrated to 2-3 times. These starting doses are based on side effects with the realization that 4-6 weeks will be required to assess for positive clinical effect. Secondary amines, such as desipramine or nortriptyline, have fewer anticholinergic side effects than tertiary amines, such as amitriptyline or imipramine, and may be desirable in an older patient or in those who cannot tolerate other agents starting at 25 mg by mouth nightly.33 The effects of other anti-depressants, including sertraline and the selective serotonin and norepinephrine reuptake inhibitor venlafaxine, have not proved to be effective in randomized, controlled trials when compared to placebo on improving symptoms and cannot be recommended in the absence of concomitant affective symptomatology with any strong evidence.14,15
Buspirone. A recent randomized, placebo-controlled, crossover trial demonstrated efficacy of the 5-hydroxytryptamine 1A receptor agonist buspirone over placebo in the symptomatic treatment of patients with FD. Based on impaired accommodation and hypersensitivity to gastric distension believed to be involved in the development of FD, buspirone relaxes the proximal stomach in healthy individuals and, when applied to a small population of patients meeting Rome III criteria for FD, had statistically significant reduction in overall severity of symptoms of dyspepsia (P < 0.005) as well as less individual symptoms of postprandial fullness, early satiation, and upper abdominal bloating. Buspirone significantly increased gastric accommodation compared to placebo, although gastric emptying of liquids was delayed. This proof-of-concept study is exciting as it builds on the observations of a recent larger, placebo-controlled study in Japan using tandospirone with the same mechanism of action. The oral administration of buspirone 10 mg taken three times daily for 4 weeks had no statistical change in adverse events when compared to placebo, but when used at higher doses as indicated for anxiolytic purposes, it has been associated with dizziness, lightheadedness, and nausea. While the comorbidity between functional gastrointestinal disorders and psychiatric disorders, namely anxiety disorders, is high, further research into targeting pathways involving this pathway may offer a potential therapeutic advantage to patients with FD.34
Antisecretory Therapy. Convincing patients to return to antisecretory therapy after a prior failed trial may be difficult, but both H2-receptor antagonists and PPI therapy have been studied and deemed more effective than placebo. A meta-analysis of eight placebo-controlled, randomized trials of PPIs for FD noted that for every 10 patients treated with a PPI, one patient will derive benefit. Interestingly, the benefits were greatest in those with reflux-like symptoms and less effective in those with only epigastric pain, but there was no significant benefit in those with dysmotility-like symptoms. Moayyedi’s subsequent meta-analysis of 10 randomized, controlled trails including 3347 patients demonstrated again that PPIs were effective with more than a third of patients improved with PPI therapy compared to placebo.35,36
Esomeprazole similarly was compared to placebo and no benefit was found for twice daily dosing for FD compared to once daily; however, there was a statistically significant benefit seen at 8 weeks that was not evident at 4 weeks.37 It is important that PPIs be used for at least 8 weeks prior to abandoning this therapy as ineffective, because it takes a prolonged time period to restore remodeling of visceral sensation in FD based on what is known from physiologic studies of gastric acid hypersensitivity to duodenal exposure. The take-home point for primary care physicians is that if empiric antisecretory trial with PPI therapy is pursued, once daily dosing is fine, but a 1- to 4-week trial is not predictive of an 8-week trial of therapy. Know this before counseling patients on what to expect and when to expect it.
Prokinetic Therapy. Prokinetic therapy has a limited niche in patients with FD with concomitant delayed gastric emptying, which affects up to a third of all patients. These patients may have been subgrouped as postprandial distress syndrome (PDS) with meal-related symptoms involving early satiation and postprandial fullness. Moayyedi also performed a meta-analysis of studies on domperidone and cisapride for FD including 19 randomized, controlled trials including 3178 patients. This study demonstrated a significant prokinetic superiority in alleviating symptoms with a number needed to treat of 6.38 The problem is that neither drug in this study is available in the United States because of QT prolongation and cases of fatal cardiac arrhythmias, respectively.
Metoclopramide is a dopaminergic and serotonergic receptor antagonist with a stimulatory effect on upper gastrointestinal motility based on poor-quality, older studies that could be reserved for difficult cases and for a limited treatment duration considering their potential for developing irreversible neurologic side effects. There are no studies with metoclopramide in patients with FD. Routine gastric emptying studies are not recommended because improvements in emptying do not correlate well with symptom improvement.39 This drug can prolong QT intervals and increase serum prolactin levels. Central nervous system side effects can be seen in 20% of patients treated with standard doses of metoclopramide. This includes agitation, anxiety, drowsiness, or insomnia. Metoclopramide currently holds a black box warning for irreversible tardive dyskinesia and mandates deliberate informed consent prior to initiation of this medication with clear stop instructions conveyed to the patient and close interval reassessment of ongoing use beyond 12 weeks.
Both PPIs and prokinetics can be used as initial empiric pharmacotherapy based on symptom patterns. PPIs are more likely to be effective among patients reporting reflux component in the presence of retrosternal or epigastric burning or pain. Prokinetic therapy may be of benefit among those subgrouped as PDS with meal-related fullness and early satiation. Combination therapy may have additive effects, but single-drug therapy is preferred initially.40
Helicobacter pylori Eradication. The effect of H. pylori eradication in FD remains controversial. It remains part of the diagnostic evaluation of uninvestigated dyspepsia and if diagnosed it should be treated. Separate meta-analyses have fallen on either side of the issue in the past, though a more recent Cochrane meta-analy- sis has reported a 10% relative risk reduction with H. pylori eradication compared to placebo at 12 months of follow-up. This may provide symptom resolution in a small percentage of patients.
Bio-Psycho-Social Model. Psychological therapies — including hypnosis, cognitive behavioral therapy, and relaxation therapy — generally are not helpful in patients with FD.41 Despite this, recent patient survey data suggest a strong relationship between dyspepsia and psychiatric comorbidity with a twofold increased risk of generalized anxiety disorder and a threefold increased risk of major depressive episodes in the general population.42 Physicians should screen for these affective disorders as well as somatoform disorders and provide necessary treatment.
Integrative Medical Therapies. Outside the scope of pharmacotherapy, integrative medicine is gaining more attention in functional disorders, with many patients seeking nontraditional care on their own. A systematic review of studies suggests a benefit from peppermint oil.43 A recently published study involving acupuncture treatment significantly improved symptoms and quality-of-life scores of patients with FD.44 Interestingly, incorporating state-of-the-art PET-CT imaging of the brain, this study indicated that acupuncture treatment markedly decreased glycometabolism in the insula, ACC, and hypothalamus, which are considered the key regions of “gut-brain communication” and which play important roles in processing and modulating pain, emotion, and visceral sensation.
Vigilance to Differential Diagnosis. Overall, up to 86% of primary care patients with dyspepsia continue to be symptomatic up to 1 year after initial presentation, despite multiple therapeutic options.45 Developing a strategy of care similar to that used in managing patients with other unexplained physical symptoms is appropriate. Monitor symptoms with regular follow-up visits, address psychosocial stressors, and enable patients to cope with their illness while remaining attentive to the development of other coexisting disorders such as IBS, GERD, or any other functional gastroduodenal disorder as well as new psycho-social stressors affecting patient’s state of wellness (see Table 4).
Summary
Dyspepsia remains a common complaint in the United States, and although only a quarter of those with symptoms actually present for this, dyspepsia represents a very large number of patients in primary care. Those who seek out care need an appropriate, evidence-based clinical evaluation. Initial treatment strategies should be pursued after assessment for alarm features to a strategy of antisecretory therapy, H. pylori eradication therapy, or upper endoscopy with a plan for subsequent reassessment, realizing that no one approach is expected to yield complete remission of symptoms if FD ultimately is discovered. Reassurance and lifestyle advice may be sufficient in patients with mild or intermittent symptoms, whereas drug or integrative therapies may be useful in those with more severe symptoms. An individually tailored, holistic treatment model is required for those with FD after establishing rapport to help patients manage and remain productive while living with this chronic disorder.
References
1. Ikenberry SO, et al. The role of endoscopy in dyspepsia. Gastrointest Endosc 2007;66:1071-1075.
2. Feldman M, et al. Sleisenger and Fordtran’s Gastrointestinal and Liver Disease. 9th Ed. 2010;1:183-195.
3. Mapel D, et al. The epidemiology, diagnosis, and cost of dyspepsia and Helicobater pylori gastritis: A case-control analysis in the Southwestern United States. Helicobacter 2013;18:54-65.
4. Moayyedi P, et al. Can the clinical history distinguish between organic and functional dyspepsia? JAMA 2006;295:1566-1576.
5. Ford AC, et al. What is the prevalence of clinically significant endoscopic findings in subjects with dyspepsia? Systematic review and meta-analysis. Clin Gastroenterol Hepatol 2010;8:830-837.
6. Tack J, et al. Functional gastroduodenal disorders. Gastroenterology 2006;130:1466-1479.
7. Tack J, et al. Pathophysiology and treatment of functional dyspepsia. Gastroenterology 2004;127:1239-1255.
8. Kindt S, Tack J. Impaired gastric accommodation and its role in dyspepsia. Gut 2006;55:1685-1691.
9. Talley NJ, et al. Functional dyspepsia, delayed gastric emptying, and impaired quality of life. Gut 2006;55:933-939.
10. Lacy BE. Functional dyspepsia and gastroparesis: One disease or two? Am J Gastroenterol 2012;107:1615-1620.
11. Mearin F, et al. Dyspepsia and irritable bowel syndrome after a Salmonella gastroenteritis outbreak: One-year follow-up cohort study. Gastroenterology 2005;129:98-104.
12. Hall W, et al. Gastric mucosal mast cells are increased in Helicobacter pylori-negative functional dyspepsia. Clin Gastroenterol Hepatol 2003;1:363-369.
13. van Boxel OS, et al. Role of chemical stimulation of the duodenum in dyspeptic symptom generation. Am J Gastroenterol 2010;105:803-811.
14. van Kerkhoven LA, et al. Effect of the antidepressant venlafaxine in functional dyspepsia: A randomized, double-blind, placebo-controlled trial. Clin Gastroenterol Hepatol 2008;6:746-752.
15. Tan VP, et al. Treatment of functional dyspepsia with sertraline: A double-blind randomized placebo-controlled pilot study. World J Gastroenterol 2012;18:6127-6133.
16. Ishii M, et al. Duodenal hypersensitivity to acid in patients with functional dyspepsia—pathogenesis and evaluation. J Smooth Muscle Res 2010;46:1-8.
17. Li X, et al. Visceral and somatic sensory function in functional dyspepsia. Neurogastroenterol Motil 2012; Nov 21. [Epub ahead of print].
18. Samsom M, et al. Abnormal clearance of exogenous acid and increased acid sensitivity of the proximal duodenum in dyspeptic patients. Gastroenterology 1999;116:515-520.
19. Camilleri CE, et al. A study of candidate genotypes associated with dyspepsia in a U.S. community. Am J Gastroenterol 2006;101:581-592.
20. Talley NJ. Dyspepsia. Gastoenterology 2003;125:1219-1226.
21. Vakil N, et al. Limited value of alarm features in the diagnosis of upper gastrointestinal malignancy: Systemic review and meta-analysis. Gastroenterology 2006;131:390-401.
22. Ford AC, et al. Helicobater pylori “test and treat” or endoscopy for managing dyspepsia: An individual patient data meta-analysis. Gastroenterology 2005;128:1838-1844.
23. Talley NJ, et al. Guidelines for the management of dyspepsia. Am J Gastroenterol 2005;100:2324-2337.
24. Shaheen NJ, et al. Upper endoscopy for gastroesophageal reflux disease: Best practice advice from the clinical guidelines committee of the American College of Physicians. Ann Intern Med 2012;157:808-816.
25. Bytzer P, Talley NJ. Dyspepsia. Ann Intern Med 2001;134(9 PT 2):815-822.
26. Camilleri M, et al. Clinical guideline: Management of gastroparesis. Am J Gastroenterol 2013;108:18-37.
27. Silverstein MD, et al. Initial endoscopy of empiric therapy with or without testing for Helicobacter pylori for dyspepsia: A decision analysis. Gastroenterology 1996;110:72-83.
28. Lassen AT, et al. Helicobacter pylori test-and-eradicate versus prompt endoscopy for management of dyspeptic patients: A randomised trial. Lancet 2000;356:455-460.
29. Longstreth GF. Functional dyspepsia—Managing the conundrum. N Engl J Med 2006;354:791-793.
30. Saad RJ, Chey WD. Review article: Current and emerging therapies for functional dyspepsia. Aliment Pharmacol Ther 2006;24:475-492.
31. Jackson JL, et al. Treatment of functional gastrointestinal disorders with antidepressant medications: A meta-analysis. Am J Med 2000;108:65-72.
32. Mertz H, et al. Effect of amitriptyline on symptoms, sleep, and visceral perception in patients with functional dyspepsia. Am J Gastroenterol 1998; 93:160-165.
33. Max MB, et al. Effects of desipramine, amitriptyline, and fluoxetine on pain in diabetic neuropathy. N Engl J Med 1992;326:1250-1256.
34. Tack J, et al. Efficacy of buspirone, a fundus-relaxing drug, in patients with functional dyspepsia. Clin Gastroenterol Hepatol 2012;10:1239-1245.
35. Moayyedi P, et al. The efficacy of proton pump inhibitors in nonulcer dyspepsia: A systematic review and economic analysis. Gastroenterology 2004;127:1329-1337.
36. Moayyedi P, et al. Cochrane Collaboration Feb 2006.
37. Talley NJ, et al. Randomized-controlled trial of esomeprazole in functional dyspepsia patients with epigastric pain or burning: Does a 1-week trial of acid suppression predict symptom response? Aliment Pharmacol Ther 2007;26:673-682.
38. Moayyedi P, et al. Cochrane Collaboration 2007.
39. Talley NJ, et al. Can symptoms discriminate among those with delayed or normal gastric emptying in dysmotility-like dyspepsia? Am J Gastroenterol 2001;96:1422-1428.
40. Camilleri M, Stanghellini V. Current management strategies and emerging treatments for functional dyspepsia. Nat Rev Gastroenterol Hepatol 2013; Feb 5 [Epub ahead of print].
41. Soo S, et al. A systematic review of psychological therapies for nonulcer dyspepsia. Am J Gastroenterol 2004;99:1817-1822.
42. Mak AD, et al. Dyspepsia is strongly associated with major depression and generalised anxiety disorder – A community study. Aliment Pharmacol Ther 2012;36:800-810.
43. Koretz RL, Rotblatt M. Complementary and alternative medicine in gastroenterology: The good, the bad, and the ugly. Clin Gastroenterol Hepatol 2004;2:957-967.
44. Zeng F, et al. Influence of acupuncture treatment on cerebral activity in functional dyspepsia patients and its relationship with efficacy. Am J Gastroenterol 2012;107:1236-1247.
45. Kurata JH, et al. A prospective study of dyspepsia in primary care. Dig Dis Sci 2002;47:797-803.
Dyspepsia represents a common yet nebulous symptom complex encountered in the daily clinical practice of primary care physicians, and it is frequently used synonymously with many different sets of symptoms marked by the presence of upper abdominal pain or discomfort.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
