A Wake-Up Call: African Trypanosomiasis Strikes Back
Special Feature
A Wake-Up Call: African Trypanosomiasis Strikes Back
By Stan Deresinski, MD, FACP
Two cases of East African trypanosomiasis occurring in travellers who had briefly visited the Serengeti Park in Tanzania were reported to the annual meeting of the American Society of Tropical Medicine and Hygiene last autumn in Houston, Tex. These reports were disturbing because, according to David Freedman, the GeoSentinel network database covering 26 global sites had had no reports of East African trypanosomiasis in the previous 3 years. These occurrences were not totally unexpected, however, since trypanosomiasis is present throughout Tanzania and, within that country, trypanosomiasis is most prevalent in a region from Kigoma at Lake Tanganyika to Arusha in the northern part of the country, areas commonly visited by tourists. Furthermore, the incidence of African trypanosomiasis has been increasing for decades. Thus, it is not surprising that new cases of trypanosomiasis in travelers continue to be reported with the recent total now reaching at least 8.1
Seven of the 8 were Europeans, including residents of Holland, Italy, Norway, and the United Kingdom. Most had been in or near the Serengeti and most reported tsetse bites, although the infection may have been acquired in other areas visited. The interval from onset of symptoms to diagnosis appears to have ranged from 2 to 4 days in most cases. At least 1 patient, who did not receive therapy until 10 days after the onset of symptoms, died.
These cases in western travellers are, of course, of concern and should alert physicians to consideration of this infection in febrile individuals returning from endemic areas. However, it is more a reflection of another in a long line of African tragedies—the resurgence of a once almost eradicated disease that causes uncounted economic and human devastation.
Human African trypanosomiasis (HAT) is caused by 1 of 2 subspecies of the hemoflagellate protozoan, Trypansoma brucei, that are endemic in areas of sub-Saharan between 14º north and 15º south latitudes.2,3 T b gambiense is transmitted in central and west Africa and in general causes chronic disease with an incubation period in indigenous people of months to years. T b rhodesiense is acquired in southern and east Africa and causes acute infection after a relatively briefer incubation period. Uganda is currently the only country in which both subspecies are reportedly present.
HAT is transmitted by the bite of the tsetse, a word meaning "fly" in Tswana, the language of the former Bechuanaland and, since 1966, Botswana. The tsetse is 6-14 mm in length, with a proboscis projecting straight from the head (see Figure 1).4 Tsetse are found in vegetation near rivers and lakes, gallery-forests, and wooded savannah. The flies that transmit T b gambiense, for which there is no known animal reservoir, usually reside along the banks of shaded streams in proximity to human habitation. In contrast, the flies that transmit T b rhodesiense dwell on lightly covered bush land and feed on game animals as well as domesticated animals. In cattle, the disease is called "naganà." The presence of an animal reservoir makes eradication potentially more difficult for this subspecies.
 |
The usually prolonged incubation period seen in native populations in Gambian HAT is generally much briefer in non-natives. The initial symptoms include intermittent fevers that correspond to successive surges of antibodies produced in response to the remarkable antigenic variation of surface proteins of the parasite as it attempts to escape immune control. Lymphadenopathy is commonly present (Winterbottom’s sign is occipital adenopathy seen in this infection), and a trypanosomal chancre may still be detectable in those who present after shorter incubation periods. The episodes of fever generally last several days and recur within several weeks. They may be accompanied by a multiform skin eruption and pruritus and occasionally edematous swelling, particularly of the face. After months of chronic illness accompanied by weight loss, symptoms of central nervous system disease appear, with somnolence developing in most. In Rhodesian HAT, the time course is more compressed and the patients more acutely ill. The diagnosis of HAT is made by the demonstration of trypanosomes in lymph node "juice," blood, or cerebrospinal fluid (CSF). All patients with HAT should have examination of CSF.
Treatment depends on both the infecting subspecies (identified by geographic origin of the infection) and the stage of the disease, with Phase II reflecting evidence of neurological involvement (see Tables 1 and 2). However, treatment is associated with significant adverse effects as well as increasing evidence of inefficacy suggesting the development of resistance in the organism. For instance, treatment with melarsoprol, an arsenical discovered in 1949 and the only available drug that has been effective in the presence of neurological involvement by either subspecies, is often accompanied by severe side effects, including a reactive encephalopathy reported to be fatal in 3-10% of cases. In addition, it has recently been reported that the relapse rate after treatment in patients with T b gambiense infection in northwestern Uganda after treatment with melarsoprol is > 25%.5
| Table 1-WHO Case Definitions | |||||||
| • Confirmed: Trypanosomes observed in blood, lymph gland fluid, or CSF. |
|||||||
| • Stage I: Trypanosomes in blood or lymph gland fluid, but not CSF and with < 5 WBC. |
|||||||
| • Stage II: One or more of the following: Trypanosomes in CSF and/or CSF WBC > 5. |
|||||||
| • Suspected relapse: after appropriate treatment of confirmed disease, develops either/or an increase in CSF WBC of > 2-fold with a total CSF WBC > 5. |
|||||||
| • Confirmed relapse: After appropriate treatment of confirmed infec- tion has trypanosomes observed in CSF, blood, or lymph gland fluid within a 2-year post-treatment follow-up. |
|||||||
| Table 2-WHO Treatment Recommendations | |||||||
| Stage I: | |||||||
| T b gambiense-pentamidine | |||||||
| T b rhodesiense-suramin | |||||||
| Stage II: | |||||||
| T b gambiense-melarsoprol or eflornithine hydrochloride | |||||||
| T b rhodesiense-melarsoprol | |||||||
The therapeutic picture has been further muddied as the result of drug shortages resulting from economic decisions of pharmaceutical companies. Perhaps the most disgusting story, albeit one with a favorable outcome, was that of eflornithine, the only available drug for those who fail treatment with melarsoprol. The pharmaceutical company with the rights to eflornithine abandoned production of this drug in 1994 due to low profitability. In 2000, however, the company resumed production of the drug—not out of the goodness of their hearts and concern for their fellow man, but because they were now marketing it as a topical agent for removal of unwanted hair.
There is clearly a need for new therapeutic approaches. In the absence of interest from the pharmaceutical industry, WHO has identified a number of rather limited therapeutic research priorities. These include the use of nifurtimox as a single agent or in combination as well as the evaluation of other drug combinations, the evaluation of a short pentamidine protocol, and the evaluation of efficacy of 2 newer drugs, berenil and megazol.
Control of the disease involves low-altitude spraying, trapping, and case finding with treatment. The only means by which travellers can avoid infection is to avoid the bite of the tsetse. Areas of heavy infestation tend to be sporadically distributed and are usually well known to local inhabitants. Avoidance of such areas is the best means of protection. These flies are attracted to moving vehicles and dark, contrasting colors. They are not affected by insect repellents and can bite through lightweight clothing. Travellers at risk should wear clothing of wrist and ankle length that is made of medium-weight fabric in neutral colors that blend with the background environment.6
Three major epidemics of HAT have occurred over the last century. The first lasted a decade, ending in 1906, the year after the first therapeutic agent, the arsenical Atoxyl had been introduced. In the same year that the epidemic ended, Colonial Undersecretary Winston Churchill reported to the British House of Commons that "the disease had reduced the population of Uganda from 6.5 million to 2.5 million."7
The second epidemic, in 1920, was rapidly controlled as the result of systematic screening of millions at risk by mobile teams and treatment with an arsenical. In 1930, the French publication "L’illustration" reported that "Our doctors have vanquished the tsetse fly."8 In a common theme of political and public health failure (think, eg, of malaria, TB, etc), the disease became infrequent between 1960 and 1965, resulting in dismantling of control programs. The result: a third epidemic starting in 1970, which continues at this time and shows no signs of abatement. The failure of public health can be seen by the fact that only 3-4 million of the more than 60 million people at risk are currently under surveillance. While 45,000 cases were reported in 1999, it is believed that the true case incidence is 300,000 to 500,000. Despite inadequate surveillance, the WHO indicates that the prevalence of trypanosomiasis in some villages in Angola, the Democratic Republic of Congo, and southern Sudan is between 20% and 50%.9 For instance, screening of 1400 persons in 16 villages of Tambura County, Western Equatoria, Sudan in 1997 found prevalences as high as 45%, with a mean prevalence of 19.3%.2 The mean prevalence in this same area in 1988 had been only 0.3%.
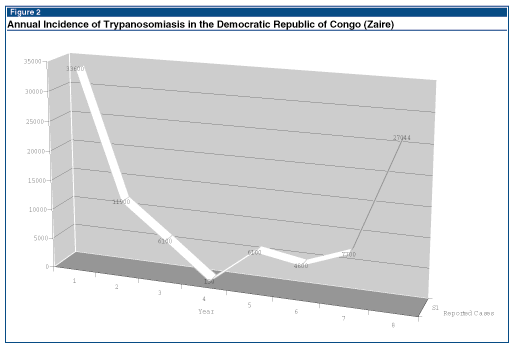
Figure 2, taken from data reported to Promed, demonstrates the incidence of trypanosomiasis in the Democratic Republic of Congo (formerly Zaire), at approximately 10-year intervals, over most of the last century (see Figure 2).1
 |
The human and animal forms of African trypanosomiasis are a major obstacle to development of rural sub-Saharan Africa as a consequence of loss of cattle and human disease leading to abandonment of potentially fertile lands. While much of the resurgence of disease can be attributed to population displacement, political instability, civil war, and the collapse of health systems, the indifference of the developed world contributes mightily.
References
2. African trypanosomiasis or sleeping sickness. WHO Fact Sheet No. 258, March 2001;1-5. http://www.who.int/inf-fs/en/fact259.html .
3. Pepin J, Donelson JE. African trypanosomiasis (sleeping sickness). In: Guerrant RL, Walker DH, Weller PF, eds. Tropical Infectious Diseases. Philadelphia, Pa: Churchill Livingstone; 1999:774-784.
4. http://www.roberth.u-net.com/tsetse.htm.
5. 3rd Meeting of the WHO Steering Committee of Human African Trypanosomiasis Treatment and
Drug Resistance Network, Bruges, Belgium, May
28-30, 2000.
6. http://www.mdtravelhealth.com/infectious/who_trypanyogeo_4.html.
7. Zimmer C. www.discover.com/august_issue/sleep.html.
8. http://www.tulane.edu/~dmsander/WWW/224/Trypano.html .
9. West African trypanosomiasis resurging in southern Sudan. CDC/NCID FOCUS. 1997; 6/ September-October 1997. http://www.cdc.gov/ncidod/focus/vol6no5/trypan.htm.
Additional Reading
1. Sinha A, et al. African trypanosomiasis in two travelers from the United States. Clin Infect Dis. 1999;29: 840-844.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
