Practical Approaches to Insomnia
Practical Approaches to Insomnia
Author: H.P. Attarian, MD, Assistant Professor, Washington University School of Medicine, St. Louis, Mo.
Editor’s Note—Insomnia is the most common sleep-related complaint and the second most common overall complaint (after pain) reported in primary care settings. It affects 35% of the general population, according to the 1984 report of the National Institutes of Mental Health, and is a cause of significant morbidity and mortality. It costs Americans about $100 billion annually in medical expenses, ramifications of accidents, and reduced productivity due to absenteeism and decreased work efficiency.1 The total direct costs in the United States for insomnia in 1995 were estimated to be $13.9 billion.2
Insomnia is the inability to obtain sleep of sufficient length or quality to produce refreshment the following morning. It is not defined by total sleep time per 24 hours. For example, a person who needs only 4 hours of sleep does not have insomnia if he or she is refreshed in the morning after 4 hours of sleep, whereas someone who needs 10 hours of sleep may have insomnia if he or she does not feel refreshed after 8 hours of sleep. Contrary to popular belief, psychiatric or psychological factors are not the most common causes of insomnia. In fact, untreated insomnia is a risk factor for the subsequent development of clinical depression and psychiatric distress.3
Insomnia is not a diagnosis in and of itself. It should be thought of as a constitutional symptom, not unlike pain, fever, or weight loss, requiring identification of an underlying cause before diagnosis and a treatment plan are established.
Types of Insomnia and Underlying Causes
Insomnia can be divided in 2 major categories—primary and secondary. Primary insomnia is when the insomnia is the major or sole symptom of a disorder. Insomnia is considered secondary when it is a symptom of an underlying medical or psychiatric illness. (See Table 1). Patients with primary insomnia have increased heart rate compared to controls and are physiologically more hyperactive.4
| Table 1. Primary and Secondary Insomnias | ||
| Primary Insomnias | Secondary Insomnias | |
| Psychophysiologic or conditioned insomnia | Insomnia in psychiatric illnesses | |
| Idiopathic or childhood-onset insomnia | Insomnia in other sleep disorders | |
| Sleep-state misperception insomnia | Insomnia in neurologic and medical conditions | |
| Poor sleep hygiene | Menopause-related insomnia | |
| Fatal familial insomnia | Medication-induced insomnia Restless legs syndrome Environmentally induced insomnia |
|
Psychophysiologic or Conditioned Insomnia. This is the most common form of insomnia and is typically acquired during a period when other factors (eg, stress) are at work. After a few days of sleeping poorly, the patient becomes concerned and begins trying harder and harder to get to sleep. The result is arousal and aggravation of the insomnia. Stimuli surrounding bedtime (eg, the bedroom, the bed itself) may become triggers to arousal. Thus, such patients may have severe problems with sleep in their own bedroom but sleep remarkably well in other locations (eg, on the living room couch, in a motel, or in a sleep laboratory). The essence of psychophysiologic insomnia is that attention is focused on the inability to sleep. Insomnia is perceived as the only source of distress, and other emotional or mental concerns are minimized. Typically, patients repress or deny awareness of stress factors and see the insomnia as occurring without any reason.
In some patients with psychophysiological insomnia, no precipitating stress is found. Rather, poor sleep may have gradually developed as an occasionally occurring disturbed night leads to increased concern, causing sleep to deteriorate until it becomes the patient’s chief concern. There is clear evidence for the role of familial inheritance in the tendency to develop insomnia.5
Idiopathic or Childhood-Onset Insomnia. This rare condition presents as a chronic, serious inability to initiate and maintain sleep, which can often be traced back to the first few weeks of life. Sleep latency (ie, the time it takes to fall asleep after going to bed) may be long, and sleep is riddled with awakenings. Daytime features typically include decreased attention and vigilance, low levels of energy and concentration, and deterioration of mood that is usually described as grim and subdued rather than obviously depressed or anxious. The presumed underlying neurologic abnormality (either hyperactivity in the arousal system or hypoactivity in the sleep system) may vary from mild to severe, so the range of insomnia encountered also may vary from mild (essentially, the patient is a light sleeper) to severe and incapacitating. In mild or moderate idiopathic insomnia, psychological functioning is remarkably intact. In severe cases, daytime functioning may be severely disrupted, and affected patients may be unable to hold a job. During childhood and adolescence, idiopathic insomnia is often associated with dyslexia and hyperactivity. In many cases, diffuse, nonspecific abnormalities are seen on an electroencephalogram (EEG). There is no direct human evidence for structural neuropathology. Although idiopathic insomnia appears in childhood, not all childhood insomnia is idiopathic.
Sleep-State Misperception Insomnia. In this fascinating disorder, complaints of insomnia occur without any objective evidence of sleep disturbance. Patients may report that they have not slept at all in weeks, months, or years. However, on objective sleep studies, they sleep several hours per night.6 When results of sleep evaluation are presented, patients with sleep-state misperception may vehemently insist that the studies are in error, because they are convinced that they sleep very little, if at all.
Poor Sleep Hygiene. In some patients, insomnia is the result of lifestyle. In others, poor sleep hygiene develops as a result of chronic insomnia. For example, in the latter case, patients may begin to drink more and more coffee to remain awake and more and more alcohol to fall sleep. They may stay in bed longer and longer in an attempt to get more sleep. However, such ploys only serve to perpetuate the insomnia. (See Table 2.)
| Table 2. Sleep Hygiene Recommendations | |
| • | Go to bed only when sleepy. |
| • | Use the bed only for sleeping; do not read, watch television, or eat in bed. |
| • | If unable to sleep, get up and move to another room. Stay up until you are definitely sleepy and then return to bed. If sleep does not come easily, get out of bed again. The goal is to associate your bed with falling asleep easily. Repeat this step as often as is necessary throughout the night. |
| • | Set the alarm and get up at the same time every morning, regardless of how much you have slept through the night. |
| • | Do not nap during the daytime. |
| • | Focusing on quiescent tasks that occupy the mind such as reading, watching television, or listening to music promotes relaxation and sleep. |
| • | Sleep induction is associated with a decline in core body temperature so regular exercise in the late afternoon or early evening can promote sleep. Spending 20 minutes in a hot tub a few hours before going to bed may promote sleep also. |
| • | Don’t exercise just before going to bed. |
| • | Don’t engage in stimulating activity just before bed. |
| • | Avoid caffeine in the afternoon. Remember that caffeine is present in chocolate, as well as regular coffee or tea, and caffeinated sodas. |
| • | Don’t use alcohol to help you sleep. |
| • | Avoid nicotine. |
| • | Eliminate clocks in the bedroom. |
| • | Use distracting activities to induce sleep onset. |
Restless Legs Syndrome (RLS). This common condition is found in varying degrees in up to 10% of the population. The 4 cardinal symptoms are a desire to move the legs, accompanying paresthesias that are characterized as uncomfortable or indescribable, motor restlessness, and worsening of symptoms at night and at rest. Symptoms of RLS may worsen with administration of tricyclic antidepressants or selective serotonin reuptake inhibitors and during pregnancy.
Fatal Familial Insomnia. This hereditary condition, with autosomal dominant transmission, is characterized clinically by progressive insomnia, dysautonomia, changes in circadian rhythm of hormone secretion, motor signs, and slight to moderate deterioration of cognition. The usual age of onset is between 35 and 60 years, and the course of the illness is between 7 and 32 months. In this condition, an abnormal prion protein (PrPsc) is present in the brain, and there is mutation of gene coding for this protein. The fatal nature of this illness is due to neurologic degenerative changes, not to the insomnia itself.
Other Sleep Disorders. Occasionally, insomnia is the presenting complaint in obstructive sleep apnea, circadian rhythm abnormalities, and even narcolepsy.
Neurologic and Medical Conditions
Conditions that can cause insomnia, among other symptoms, include neurodegenerative diseases, pain, allergies, gastroesophageal reflux, and asthma.
Menopause-Related Insomnia
There is a high level of sleep disturbance, occurring in about 42% of middle-aged women.7 Although cross-sectional analyses indicate that sleep disturbance may be independent of menopausal status, transition into postmenopausal status is associated with deleterious changes in sleep among women not receiving hormone replacement therapy.7
Psychiatric Conditions
Affective disorders and anxiety disorders can cause insomnia.
Medication-Induced Insomnia
Selective serotonin reuptake inhibitors,8 stimulants, theophylline, prednisone, and 2 of the newer anticonvulsants—felbamate9 (Felbatol) and lamotrigine10 (Lamictal) may cause insomnia. Other medication-related causes of insomnia include withdrawal from sedative agents, idiosyncratic reactions to other medications, and toxin-related reactions (eg, alcohol, carbon monoxide, inorganic mercury, recreational drugs).
Diagnostic Tools
Polysomnogram and Multiple Sleep Latency Test (MSLT). The polysomnogram is a polygraph of EEG findings, eye movements, electromyography readings, oxygen saturation, limb movements, airflow, and chest and abdominal movements taken during sleep, usually for the entire night. According to the American Sleep Disorders Association (now the American Academy of Sleep Medicine), practice parameters polysomnography is not indicated in routine evaluation of insomnia, except when the diagnosis is uncertain and a primary sleep disorder is suspected and when insomnia does not respond to appropriate behavioral and pharmacologic treatments.
An MSLT is a series of 4 or 5 opportunities, each separated by a 2-hour interval, to take a 15- to 20-minute nap. It is used to assess sleep latency and the possibility of such sleep disorders as obstructive sleep apnea and narcolepsy. In primary insomnia, results of the MSLT are usually normal.11
Sleep Logs. A sleep log is a graph on which, for 2-3 weeks, the patient records bedtime, approximate sleep time, times and duration of awakenings during the sleep period, final awakening time, and naps taken during the day. Although subjective, this record summarizes the patient’s perception of the amount and quality of sleep he or she is getting.
Actigraphy. Actigraphy is a recently developed technique to record activity during waking and sleeping without application of any electrodes. An actigraph is worn on the wrist and is about the size of a watch. It consists of a movement detector and considerable memory, so it can record movement and nonmovement data plotted against time for a week or two. The patient can wear it continuously during sleep and as he or she goes about routine daily activities. Actigraphy is ideal for extended examination of the sleep-wake cycle in the patient’s home environment. It is convenient and is readily accepted by patients. It can be used to supplement sleep logs and to evaluate unusual complaints, such as, "I have not slept for several nights."
In general, patients have fewer limb movements during sleep than while awake. There is a close correlation between the rest-activity findings recorded by the actigraph and the sleep-wake pattern as determined by a polysomnogram. Several investigators used actigraphy in groups of controls of different ages and found minute-by-minute agreement in sleep-wake scoring between polysomnography and actigraphy to exceed 90%.12
Laboratory Evaluation
In patients with RLS, a serum ferritin level of less than 50 m/L is associated with increased severity of symptoms, which may exacerbate insomnia.13
Diagnostic Work-up
Insomnia is usually diagnosed by thorough clinical history taking. In patients with insomnia, if anxiety permeates most aspects of functioning, generalized anxiety disorder is the usual diagnosis rather than primary insomnia. Affective disorders can be differentiated from primary insomnia on the basis of other "vegetative" signs, such as loss of appetite or libido or the typical diurnal fluctuation (worse in the morning) of depression. In these situations, the patient should be first evaluated by a psychiatrist. If a history of significant dysesthesias interfering with sleep is elicited during history taking, then the diagnosis of RLS should be considered, serum ferritin checked, and treatment started accordingly. The complaint of daytime sleepiness is generally indicative of a primary sleep disorder, because patients with insomnia are hyposomnolent and often complain bitterly of the inability to take naps.11 Patients reporting episodes of falling asleep unintentionally during the day should be evaluated with a polysomnogram and a MSLT to rule out primary sleep disorders. Taking a history of the patient’s sleep habits is essential in identifying sleep hygiene issues or circadian rhythm abnormalities. If the patient is unable to fall asleep at a desired time but is able to fall asleep much later and is unable to wake up at a desired time, then the diagnosis of delayed sleep phase should be considered. If the patient is waking up very early in the morning and is unable to go back to sleep but cannot stay awake past early evening, then advanced sleep phase syndrome should be entertained as a diagnosis. In short, if the patient is able to fall asleep but not at socially acceptable times, then the most likely cause of the complaint of insomnia is circadian rhythm abnormalities that can be treated with a combination of chronotherapy, phototherapy, and melatonin. A history of other medical problems, exposure to toxins, and a list of medication a patient is taking is also important since a lot of chemicals—either pharmaceutical or toxic—can affect sleep and cause insomnia. If the diagnosis of persistent primary insomnia is made, then sleep diaries are essential in tailoring treatment to the individual patient’s needs. An actigraph is an essential tool in primary insomnia, both in providing an objective measure of the true extent of the insomnia and for gauging response to treatment. In patients with intermittent situational insomnia, such as Sunday night insomnia, the use of hypnotic medication as the sole method of treatment is encouraged. This is to prevent the intermittent insomnia into perpetuating itself through conditioning and poor sleep hygiene to become persistent and more difficult to treat.
Cognitive Behavioral Treatment
Behavioral methods include sleep restriction consolidation, sleep hygiene education, relaxation therapy, and stimulus control therapy.14,15 These behavioral methods are effective in increasing the total sleep time by reducing sleep latency, by increasing sleep efficiency, and by reducing the wake time after sleep.16-18
Relaxation therapy includes teaching patients how to recognize physiological tension and then learn to control it. A series of exercises that consist of first tensing and the relaxing each muscle group in a systemic fashion is taught to the patient in a 1-hour training session. Additional sessions are scheduled on an as needed basis. The patient must practice the exercises twice per day just before bedtime.
Stimulus Control Therapy. In patients suffering from primary insomnia, an association between the sleep environment and wakefulness can develop. Stimulus control attempts to break this association by teaching the patient not to engage in "sleep incompatible" behavior in the bedroom and to be in the bedroom only when drowsy or asleep.
Sleep Restriction Consolidation Therapy. This treatment seeks to increase sleep drive through partial sleep deprivation. A strict schedule for bedtime and arising time is prescribed to consolidate sleep and decrease amount of time spent awake during the night. Patients initially reduce their sleep time in bed to the amount of time they are actually sleeping according to their sleep logs but no less than 4.5 hours. If 2-3 weeks later their sleep efficiency is increased to 90% or better, according to the sleep logs, then 15 minutes is added to either end of their sleep cycle. This continues until the patient is sleeping the amount of time needed for him or her to feel refreshed during the day.
Sleep Hygiene Advice
Insomnia can develop or be perpetuated because of poor sleep hygiene (see Table 2). It is important to advise patients of all rules of good sleep hygiene, but in order to prevent the advised changes to be overwhelming, ask them to concentrate on 2-3 rules that are most pertinent to their situation. Caffeine, ethanol, and nicotine are the 3 most common ingested chemicals that can have deleterious effects on sleep. Good sleep hygiene teaching should include advice on how and when to consume these chemicals to avoid their deleterious effects on sleep.
Alcohol may be mildly stimulating in some people, but more commonly it has a transient, sedative effect. It is the most commonly used sleeping aid in the general population. Alcohol shortly before bedtime tends to shorten sleep latency. It also tends to increase the relative proportion of NREM sleep and reduce that of REM sleep. Alcohol, however, is metabolized rapidly: about 1 glass of wine or half a pint of beer per hour. Therefore, after 4 drinks in the hours before bedtime, alcohol concentration approaches zero about halfway through the night.
As a result, the person is in withdrawal and experiences shallow, disrupted sleep, increased proportion of REM sleep, increased dream recalls, nightmares, and physiological signs of agitation and arousal, such as sweating and faster heart rates. Stomach irritation, headache, and a full bladder, all due to alcohol, may also interrupt sleep. The adverse effects of alcohol on sleep continue some time after the blood alcohol levels have become undetectable. Moderate drinking during the so-called happy hour in the late afternoon may disrupt sleep and, hence, reduce its quality during the last half of the night, long after alcohol has disappeared from the blood.
Although alcohol increases sleep at the beginning of the night, it decreases sleep at the end of the night. The effect of nicotine on the sleep of nonsmokers is not well defined. There is some evidence to suggest that at low doses of nicotine can be sedating, and high doses alerting. Overall, active smokers tend to have a harder time falling sleep and staying asleep than nonsmokers. During withdrawal from nicotine, sleep deteriorates even more with more difficulty falling sleep and multiple arousals at night. This in turn results in sleepiness during the day. The nicotine patch does not seem to improve the sleep complaints associated with cessation of smoking.
|
|||||||||||||||||||||||||||
Caffeine and related substances called methylxanthines are stimulants found not only in coffee but also in tea, hot chocolate, cocoa, over-the-counter pain and cold medications, and colas. (See Table 3.) Caffeine promotes wakefulness and can cause insomnia. Many people, however, develop tolerance and apparently sleep well per subjective reports. The effects of caffeine last for 8-14 hours and therefore may have significant effects on sleep at night, causing insomnia, even if it is consumed late afternoon and early evening. Caffeine has an important role in combating fatigue and sleepiness in the average person. The combination of caffeine and alcohol can cause insomnia synergistically many hours later. Initially they have opposing effects. The sedative effects of alcohol counteract the arousing effect of caffeine. Three to 4 hours into the sleep period, however, because the effects of alcohol last for a shorter time, the individual goes into the arousing, withdrawal effects of alcohol while the caffeine blood levels are still high.
In conclusion, alcohol should not be used as a sleep aid and caffeine should be avoided in the afternoon in individuals who are sensitive to its stimulant effects. (See Table 4.)
| Table 4. Program to Achieve Sleep Restriction and Consolidation and Control Stimuli | |
| • | Restrict time in bed to achieve some degree of sleep deprivation. |
| • | Do not spend more than 15 minutes awake in bed, either at the beginning of sleep or during awakenings. After 15 minutes, get up and leave the bedroom. |
| • | Avoid productive, accomplishment-achieving activities (eg, balancing checkbook, doing housework) at bedtime, since they may be subconsciously rewarding for being awake. |
| • | Do not take naps. |
| • | Get out of bed at a predetermined time, no matter how little sleep you have gotten. Once you are sleeping for 90% of the time in bed, you may add 15 minutes to either end of the sleep period. |
| • | Use medications (eg, benzodiazepines) as prescribed. |
Pharmacological Treatment
RLS and the resulting insomnia respond only to medication. Behavioral therapies have no role in RLS treatment. Cessation of aggravating medication and iron replacement, in those patients who have low serum ferritin, helps. Most RLS patients, however, need medication. The most effective medications for treating symptoms of RLS belong to 3 distinct classes: 1) the dopaminergic agents (eg, pramipexole19,20 [Mirapex], ropinirole hydrochloride21,22 [Requip], pergolide mesylate23 [Permax], bromocriptine mesylate24 [Parlodel], and levodopa24 [Dopar, Larodopa]; 2) the opiates;25 and 3) the benzodiazepines (especially clonazepam26 [Klonopin]. Recent studies have shown that the newer anticonvulsant gabapentin27 (Neurontin) and Clonidine28 (especially in patients who do not have a large amount of periodic limb movements) are also effective in controlling RLS. The doses of the above medication used in the treatment of RLS are usually much lower than the doses used in the treatment of other conditions such as Parkinson’s disease, pain, anxiety, seizures, and other medical conditions for which the above medication are effective. In rare instances, higher doses are reached by titrating to the patients’ symptoms.
Menopause-Related Insomnia. Estrogen replacement therapy7,29 may control insomnia along with other menopause-related symptoms.
Benzodiazepines. Until the advent of the imidazopyridines (which are discussed below), benzodiazepines were the only hypnotics of choice for insomnia. They were clinically proven to be efficacious and had rare adverse reactions. Benzodiazepines differ from each other mainly by speed of absorption and elimination half-life. (See Table 5.)
| Table 5. Benzodiazepines Approved in the United States for the Treatment of Insomnia | |||
| Drug | Elimination Half-Life | Peak plasma level | Active metabolites |
| quazepam | 39 h | 2 h | Yes |
| flurazepam | 2.3 h | 20 min | Yes |
| estazolam | 10-15 h | 0.5-2 h | No |
| temazepam | 8-15 h | 30- 60 min | No |
| triazolam | 1.5-5.5 h | 15-30 min | No |
Most benzodiazepine adverse reactions are viewed as extensions of the therapeutic effect beyond the desired time.30 The use of benzodiazepines has been generally restricted in the treatment of insomnia because of concerns of addiction, dependence, and tolerance. The risk of habituation and abuse, however, is lower than previously thought in patients who are properly diagnosed and use these medications for medicinal purposes.31,32 However, there are exceptions. Alcoholics and drug abusers have a higher tendency to abuse benzodiazepines.33 So benzodiazepines should be prescribed with caution in this population of patients. Benzodiazepines are also contraindicated in pregnancy since they are FDA pregnancy category X.
Imidazopyridines. Zolpidem and zaleplon are hypnotics belonging to a new chemical class, the imidazopyridines. They are structurally unrelated to benzodiazepines. Zolpidem is an effective and safe hypnotic with minimal adverse effects and no dependence, withdrawal, tolerance, or rebound insomnia over long-term use.34 Its elimination half-life is 2.2 hours and peak plasma levels are reached in 90 minutes. The dose needs to be adjusted in the setting of hepatic impairment but not with altered renal function. Zaleplon also is an effective and safe hypnotic.35 The onset of action is approximately 30 minutes and the duration of action is about 4 hours. Peak zaleplon serum concentrations occur in about 1 hour and its elimination half-life is also about 1 hour. Similar to zolpidem, its dose needs to be adjusted with altered hepatic function but not with renal impairment. Because of its very short half-life, there is no residual sedation when zaleplon is administered in the middle of the night, hence it is the ideal medication for sleep maintenance insomnia.36 Zolpidem and zaleplon are safe hypnotics with minimal adverse effects and no dependence, withdrawal, tolerance or rebound insomnia over long-term use.34,35
Antidepressants. Due to concerns of tolerance, addiction, and dependence antidepressants (specifically, amytriptyline and trazodone) have been used more and more in the treatment of insomnia. There has been an upward trend in the number of antidepressant prescriptions that were written for insomnia in the last decade and a half.37 Unfortunately, there are no good data regarding the efficacy of most antidepressants as hypnotics and the presence of lingering daytime sedation after nighttime administration and their other adverse effects (with tricyclics aggravation of RLS, anticholinergic effects, etc) makes them poor choices for the treatment of insomnia. Trazodone is the only antidepressant on which there are some clinical data regarding efficacy in insomnia. Trazodone does have the daytime sedation but does not tend to aggravate RLS and periodic limb movements.
Antihistamines and over-the-counter hypnotics. These are the most used and most recommended hypnotics, despite absence of good data supporting their efficacy and the presence of several studies that show significant adverse effects. Diphenhydramine, the active ingredient in most over-the-counter hypnotics, impairs driving performance more than alcohol.38 There are no good studies showing efficacy of antihistamines in insomnia. A few studies have recommended not using them as hypnotics due to lack of efficacy and poor side-effect profile.39,40
Dietary Supplements and Other Natural Products
Melatonin. Several studies have shown efficacy of melatonin in the treatment of circadian rhythm problems. The data available, however, do not support its use as a hypnotic for the treatment of insomnia.41,42
Valerian Root. Over the past few years, multiple small trials, both open-label and double-blind (placebo-controlled and using oxazepam as an active control), have shown clinically significant polygraphic and subjective improvement in the sleep of patients with primary insomnia who were given different valerian products at doses ranging from 460 mg to 600 mg.43-46 Earlier studies, however, did not show clinically significant hypnotic benefit of valerian at similar doses.47,48 The evidence for valerian as a treatment for insomnia is inconclusive. There is a need for large-scale rigorous trials to determine its efficacy and safety. Moreover, there is no FDA-regulated standard formulation of neither valerian nor melatonin.
The ideal method of treatment of primary insomnia is a combination of different behavioral methods and hypnotic medication. (See Algorithm.)
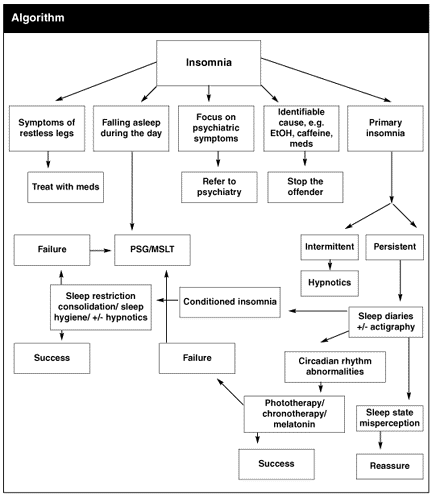 |
Methods of Treatment for Circadian Rhythm Abnormalities
Chronotherapy. The desirable total sleep time is determined by sleep logs during a "free running" period. The patient then delays or advances the sleep onset by a few hours every day, sleeping only the predetermined number of hours until the sleep onset time is at the desired time. The patient thereafter attempts to maintain that time. This method requires several days of free time.
Phototherapy. Exposure to bright light has a potent effect upon the biological clock: phototherapy uses exposure to light at strategic times in the wake/sleep cycle to induce a change in the underlying rhythm. The timing of phototherapy and its duration depend on diagnosis and individual response. The patient sits at a prescribed distance from a bright, full spectrum light, which provides an illuminance of more than 2500 Lux at that distance for 30 minutes at a time.
Melatonin 5 mg administered 3-5 hours prior to desired bedtime is effective in advancing sleep time in delayed sleep phase syndrome.42
Prognosis and Complications
If untreated, insomnia can continue for decades. In some cases, it gradually worsens because a vicious cycle develops. The complications are: excessive use of hypnotics, self treatment with alcohol, treatment of ensuing daytime somnolence by stimulants, and daytime tension with tranquilizers.49 Psychological complications include a passive and defeatist attitude, as well as cognitive and memory impairment.50,51 Untreated insomnia is a risk factor for the subsequent development of clinical depression and psychiatric distress.3,52 Chronic insomnia is also associated with increased motor vehicle accidents, decreased job performance and decreased quality of life.53 Tension related disorders (headaches, gastric upset, vasoconstrictions leading to cold hands and feet, etc) are also complications.
Conclusion
Insomnia is a common and easily treated condition. It may be the presenting symptom of many other medical conditions and usually is not a sign of psychological or psychiatric illness. Untreated, insomnia can cause significant economic hardship, morbidity, and mortality, and it may be a risk factor for development of depression or anxiety. Actigraphy and sleep logs are essential tools in evaluating insomnia. Their use usually results in a specific causal diagnosis, with effective therapeutic implications. Treatment includes both pharmacologic and behavioral interventions.
References
1. Stoller MK. Economic effects of insomnia. Clin Ther. 1994;16(5):873-897.
2. Walsh JK, Engelhardt C. The direct economic costs of insomnia in the United States for 1995. Sleep. 1999;22(2 Suppl):S386-393.
3. Katz DA, McHorney C. Clinical correlates of insomnia in patients with chronic illness. Arch Intern Med. 1998;158(10): 1099-1107.
4. Bonnet MH, Arand DL. Heart rate variability in insomniacs and matched normal sleepers. Psychosom Med. 1998;60(5):610-615.
5. Bastien CH, Morin CM. Familial incidence of insomnia. J Sleep Res. 2000;9(1):49-54.
6. Bonnet MH, Arand DL. Physiological activation in patients with sleep state misperception. Psychosom Med. 1997;59(5):533-540.
7. Owens JF, Matthews KA. Sleep disturbance in healthy middle-aged women. Maturitas. 1998;30(1):41-50.
8. Zajecka J, et al. Changes in adverse events reported by patients during 6 months of fluoxetine therapy. J Clin Psychiatry. 1999;60(6):389-394.
9. Leppik IE. Felbamate. Epilepsia. 1995;36(Suppl 2):S66-S72.
10. Sadler M. Lamotrigine associated with insomnia. Epilepsia. 1999;40(3):322-325.
11. Bonnet MH, Arand DL. Activity, arousal, and the MSLT in patients with insomnia. Sleep. 2000;23(2):205-212.
12. Sadeh A, et al. Actigraphic home-monitoring sleep-disturbed and control infants and young children: A new method for pediatric assessment of sleep-wake patterns. Pediatrics. 1991;87(4): 494-499.
13. Sun ER, et al. Iron and the restless legs syndrome. Sleep. 1998; 21(4):371-377.
14. Beullens J. Determinants of insomnia in relatively healthy elderly. A literature review. Tijdschr Gerontol Geriatr. 1999;30(1):31-38.
15. Morin CM, et al. Nonpharmacological treatments of late life insomnia. J Psychosom Res. 1999;46(2):103-116.
16. Edinger JD, et al. Cognitive behavioral therapy for the treatment of chronic primary insomnia. JAMA. 2001;285(14):1856-1864.
17. Hoch CC, et al. Protecting sleep quality in later life: A pilot study of bed restriction and sleep hygiene. J Gerontol B Psychol Sci Soc Sci. 2001;56(1):52-59.
18. Espie CA, et al. Predicting clinically significant response to cognitive behavior therapy for chronic insomnia in general medical practice: analysis of outcome data at 12 months posttreatment. J Consult Clin Psychol. 2001;69(1):58-66.
19. Lin SC, et al. Effect of pramipexole in treatment of resistant restless legs syndrome. Mayo Clin Proc. 1998;1998;73(6):497-500.
20. Becker PM, et al. Encouraging initial response of restless legs syndrome to pramipexole. Neurology. 1998;51(4):1221-1223.
21. Saletu M, et al. Sleep laboratory studies in restless legs syndrome patients as compared with normals and acute effects of ropinirole. 2. Findings on periodic leg movements, arousals and respiratory variables. Neuropsychobiology. 2000;41(4):190-199.
22. Saletu, et al. Sleep laboratory studies in restless legs syndrome patients as compared with normals and acute effects of ropinirole. 1. Findings on objective and subjective sleep and awakening quality. Neuropsychobiology. 2000;41(4):181-189.
23. Wetter TC, et al. A randomized controlled study of pergolide in patients with restless legs syndrome. Neurology. 1999;52(5): 944-950.
24. Walters AS, et al. A double-blind randomized crossover trial of bromocriptine and placebo in restless legs syndrome. Ann Neurol. 1988;24(3):455-458.
25. Walters AS, et al. Successful treatment of the idiopathic restless legs syndrome in a randomized double-blind trial of oxycodone versus placebo. Sleep. 1993;16(4):327-332.
26. Peled R, Lavie P. Double-blind evaluation of clonazepam on periodic leg movements in sleep. J Neurol Neurosurg Psychiatry. 1987;50(12):1679-1681.
27. Adler CH. Treatment of restless legs syndrome with gabapentin. Clinical Neuropharmacology. 1997;20(2):148-151.
28. Wagner ML, et al. Randomized, double-blind, placebo-controlled study of clonidine in restless legs syndrome. Sleep. 1996; 19(1):52-58.
29. Moe KE. Reproductive hormones, aging, and sleep. Semin Reprod Endocrinol. 1999;17(4):339-348.
30. Mendelson WB TC, Franko T. Adverse reactions to sedative/hypnotics: Three years’ experience. Sleep. 1996;19(9): 702-706.
31. Schenck CH, Mahowald MW. Long-term, nightly benzodiazepine treatment of injurious parasomnias and other disorders of disrupted nocturnal sleep in 170 adults. Am J Med. 1996;100(3): 333-337.
32. Doghramji K. The need for flexibility in dosing of hypnotic agents. Sleep. 2000;23(Suppl 1):S16-S20.
33. Strang J, et al. Survey of use of injected benzodiazepines among drug users in Britain. BMJ. 1994;308(6936):1082.
34. Saletu-Zyhlarz G, et al. Placebo-controlled sleep laboratory studies on the acute effects of zolpidem on objective and subjective sleep and awakening quality in nonorganic insomnia related to neurotic and stress-related disorder. Neuropsychobiology. 2000; 41(3):139-148.
35. Fry J, et al. M. Zaleplon improves sleep without producing rebound effects in outpatients with insomnia. Zaleplon Clinical Study Group. Int Clin Psychopharmacol. 2000;15(3):141-152.
36. Walsh JK, et al. Lack of residual sedation following middle-of-the-night zaleplon administration in sleep maintenance insomnia. Clin Neuropharmacol. 2000;23(1):17-21.
37. Walsh JK, Schweitzer PK. Ten-year trends in the pharmacological treatment of insomnia. Sleep. 1999;22(3):371-375.
38. Weiler JM, et al. Effects of fexofenadine, diphenhydramine, and alcohol on driving performance. Ann Intern Med. 2000;132: 354-363.
39. Gilhooly TC, et al. What happens when doctors stop prescribing temazepam? Use of alternative therapies. Br J Gen Pract. 1998; 48(434):1601-1602.
40. Sproule BA, et al. The use of non-prescription sleep products in the elderly. Int J Geriatr Psychiatry. 1999;14(10):851-857.
41. Stone BM, et al. Hypnotic activity of melatonin. Sleep. 2000; 23(5):663-669.
42. Kayumov L, et al. A randomized, double-blind, placebo-controlled crossover study of the effect of exogenous melatonin on delayed sleep phase syndrome. Psychosom Med. 2001;63(1): 40-48.
43. Dominguez RA, et al. Valerian as a hypnotic for Hispanic patients. Cultur Divers Ethni Minor Psychol. 2000;6(1):84-92.
44. Donath F, et al. Critical evaluation of the effect of valerian extract on sleep structure and sleep quality. Pharmacopsychiatry. 2000;33(2):47-53.
45. Dorn M. Efficacy and tolerability of Baldrian versus oxazepam in non-organic and non-psychiatric insomniacs: A randomised, double-blind, clinical, comparative study. Forsch Komplementarmed Klass Naturheilkd. 2000;7(2):79-84.
46. Fussel A, et al. Effect of a fixed valerian-Hop extract combination (Ze 91019) on sleep polygraphy in patients with nonorganic insomnia: A pilot study. Eur J Med Res. 2000;5(9):385-390.
47. Balderer G, BorbelyAA. Effect of valerian on human sleep. Psychopharmacology. 1985;87(4):406-409.
48. Schulz H, et al. The effect of valerian extract on sleep polygraphy in poor sleepers: A pilot study. Pharmacopsychiatry. 1994;27(4): 147-151.
49. Roehrs T, et al. Ethanol as a hypnotic in insomniacs: self administration and effects on sleep and mood. Neuropsychopharmacology. 1999;20:279-286.
50. Szelenberger W, Niemcewicz S. Severity of insomnia correlates with cognitive impairment. Acta Neurobiologiae Experimentalis. 2000;60(3):373.
51. Roehrs T, Roth T. Sleep-wake state and memory function. Sleep. 2000;23(Suppl 3):S64-S68.
52. Chang PP, et al. Insomnia in young men and subsequent depression: The Johns Hopkins Precursors Study. Am J Epidemiol. 1997;146(2):105-114.
53. Leger D, et al. SF-36: Evaluation of quality of life in severe and mild insomniacs compared with good sleepers. Psychosom Med. 2001;63(1):49-55.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
