Drug Criteria & Outcomes: Ticlid/Plavix formulary evaluation
Drug Criteria & Outcomes: Ticlid/Plavix formulary evaluation
By Jamie Hayes, PharmD*
* Written as a PharmD candidate at Auburn University School of Pharmacy, Auburn, AL.
Platelet aggregation inhibitors
Clopidogrel (Plavix) — Bristol-Myers-Squibb/Sanofi
Ticlopidine (Ticlid) — Roche
Mechanism of action
Both drugs selectively inhibit adenosine diphosphate (ADP)-induced platelet aggregation with no direct effect on arachidonic acid metabolism. Blocking ADP receptors prevents fibrinogen binding at the site and thereby reduces the possibility of platelet adhesion and aggregation.
Pharmacokinetics
The pharmacokinetic profiles of clopidogrel and ticlopidine are shown in Table 1, below.

Dosage
Ticlopidine 250 mg bid. Dose adjustment in renal failure patients and possible dose adjustment in the elderly may be necessary. Ticlopidine is contraindicated in hepatic failure patients.
Clopidogrel 75 mg QD. Dose adjustment in renal failure patients or the elderly is not required, but caution is advised in hepatic failure patients. A loading dose of 300 mg is optional.
Indications
Both drugs are used to treat the underlying condition of atherothrombosis, which can manifest clinically in many different ways in any part of the cardiovascular system. There also is significant overlap between disease manifestations in cardiovascular disease. For example, approximately 5% of this population has coronary, peripheral arterial, and cerebrovascular disease.
Clopidogrel
Clopidogrel is approved by the Food and Drug Administration to treat atherosclerotic events, including: myocardial infarction (MI), stroke, and vascular death in patients with atherosclerosis that is documented by stroke, recent MI, or established peripheral arterial disease.
Unlabeled indications for clopidogrel that currently are being investigated include Buerger’s disease, acute MI, diabetic nephropathy, ulcers due to venous stasis, and intermittent claudication.
Ticlopidine
Ticlopidine is labeled to reduce the risk of thrombotic stroke (fatal or nonfatal) in patients who have experienced stroke precursors, and in patients who have had a completed thrombotic stroke.
Ticlopidine has been used in various other conditions where further study is needed; these unlabeled uses are described in Table 2, below.
Both drugs are used after coronary stenting (unlabeled). This indication is being studied actively.

Safty
Ticlopidine has a block-box warning regarding life-threatening hematological adverse reactions, including neutropenia/agranulocytosis and thrombotic thrombocytopenic purpura (TTP). The incidence of ticlopidine-associated TTP is estimated to be 1 per 1,600-5,000 patients.
More than 3 million patients have taken clopidogrel, and approximately 11 cases of TTP have been identified. This rate is similar to background frequency of TTP reported in the general population and approximately 100 times less frequent than ticlopidine-induced TTP. Several of these reports were not believed to be caused by clopidogrel. In 10 of the 11 cases, the effect was seen in less than 14 days. Table 3, below, provides a list of common adverse drug reactions and their frequencies.

Monitoring
Ticlopidine requires monitoring for neutropenia and TTP, with baseline and serial complete blood counts every two weeks for the first three months of treatment.
In clinical trials with clopidogrel, neutropenia frequency was no higher than that associated with aspirin. Serial blood tests, therefore, are not required with clopidogrel.
Drug interactions
Clopidogrel inhibits the P-450 CYP2C9 system at high concentrations and, therefore, may interfere with metabolism of other drugs metabolized by this system, requiring dose adjustment. Ticlopidine inhibits the P-450 CYP2C19 system and may require dose adjustment. Table 4, below, shows the most important, currently known drug interactions.
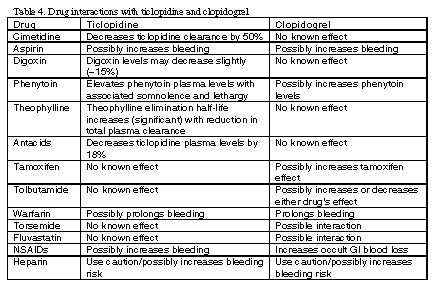
Cost and usage
Ticlopidine: $0.88/d (generic); five days of therapy/month.
Clopidogrel: $2.60/d; 1,200 days of therapy/ month.
Clinical trials
Few head-to-head clinical trials prove superior efficacy of ticlopidine vs. clopidogrel. However, three important trials (two of ticlopidine and one of clopidogrel) compare the safety and efficacy of these drugs to aspirin or placebo. The differences in these trials make them very difficult to compare. The CLASSICS trial compared the safety of the two drugs against each other, but lacked sufficient power to prove efficacy.
CATS trial
The CATS trial was a multicenter, randomized, placebo-controlled trial designed to assess the effect of ticlopidine (250 mg bid) in reducing the risk of secondary stroke, MI, or vascular events in 1,053 patients who had a recent thromboembolic event.1 Study results are listed in Table 5, below.

P values were not given for actual results. The results demonstrate a trend toward lower event rates with ticlopidine, but significance cannot be assessed. Data were listed in terms of relative risk, which showed a 30.2% reduction in the incidence of stroke, MI, or vascular death associated with the use of ticlopidine (P = 0.006).
Severe adverse events occurred in 8.2% of the ticlopidine group and 2.8% of the placebo group (P < 0.001), including one case of moderate neutropenia in the placebo group and four cases of severe neutropenia in the ticlopidine group.
The CATS trial showed efficacy of ticlopidine and its potential for causing serious adverse events. The study was well-designed and had adequate sample size; however, the absence of P values supporting actual events is a major limitation.
TASS trial
The TASS trial was a multicenter, randomized, triple-blind, controlled trial comparing ticlopidine vs. aspirin for prevention of secondary atherothrombotic stroke or death in 3,069 patients.2 Patients were randomized to receive ticlopidine 250 mg bid or aspirin 650 mg bid. The primary results of this trial are listed in Table 6, below.
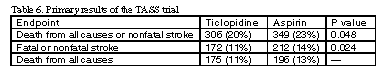
Gastrointestinal side effects were the most common adverse drug reaction in both treatment groups. The dose of aspirin was 1,300 mg/d, which is much higher than the typical 325 mg/d dose prescribed today. It was not stated whether the aspirin supplied was enteric-coated. Thus, gastrointestinal safety of ticlopidine in comparison to the lower standard dose of aspirin cannot be assessed. The incidence of diarrhea and rash in the ticlopidine group was twice that of the aspirin group. A total of 13 cases of neutropenia developed in the ticlopidine group; no cases of neutropenia developed in the aspirin group.
The TASS study had a high dropout rate that was similar between treatment groups. The trial was well-designed. Not all P values were given, and most of the data were reported in terms of relative risk. This study did show slightly greater efficacy of ticlopidine compared to aspirin, but ticlopidine also resulted in serious side effects.
CAPRIE trial
The CAPRIE trial was a randomized, blinded, international trial comparing the safety and efficacy of clopidogrel and aspirin for the prevention of secondary stroke in patients at risk of ischemic events.3 This study was designed to investigate the risk of a composite outcome cluster of ischemic stroke, MI, or vascular death. The results are summarized in Table 7, below.
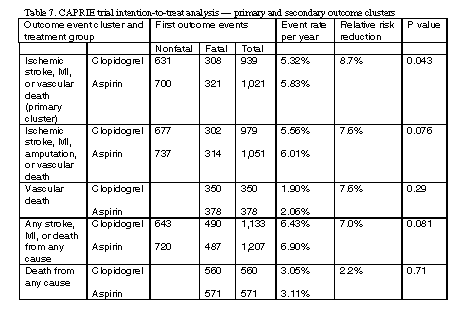
These results show a consistently lower event rate for clopidogrel vs. aspirin, although statistical significance is unknown. The primary endpoint events, including ischemic stroke, MI, or vascular death, showed a total event rate of 939 (9.8%) in the clopidogrel group and 1,021 (10.7%) in the aspirin group. Therefore, this trial shows a less than 1% difference in the efficacy of clopidogrel and aspirin.
The CAPRIE trial found clopidogrel’s safety to be similar to that of a 325 mg aspirin. There was a significantly greater incidence of severe adverse reactions in the clopidogrel group vs. the aspirin group for rash (0.26% vs. 0.10%, P = 0.017) and diarrhea (0.23% vs. 0.11%, P = 0.080), and a greater incidence of severe gastrointestinal discomfort (P = 0.096), intracranial hemorrhage (P = 0.080), and gastrointestinal hemorrhage (P = 0.05) with the aspirin group. It is not known whether the aspirin was enteric-coated. The frequency of neutropenia was 0.1% for clopidogrel and 0.17% for aspirin.
The CAPRIE trial was well-designed; 19,185 patients with parallel demographics were evaluated for 1-3 years. The number of patients discontinuing this study due to adverse events was similar between treatment groups, and the overall safety was comparable between the two drugs. Efficacy was reported as relative risk reduction; no P values for actual events were provided. Differentiation between subgroups cannot be evaluated due to overlapping disease states; 2,144 patients in ischemic stroke and peripheral artery disease had a history of MI. In addition, the trial did not achieve power to detect differences in subgroups. The conclusions of this trial are that clopidogrel is safe and effective.
CLASSICS trial
The CLASSICS trial was a double-blind study comparing the safety of clopidogrel with and without a loading dose and in combination with aspirin vs. ticlopidine in combination with aspirin after coronary stenting. The trial showed the safety and tolerability of clopidogrel to be superior to ticlopidine.4 Patients were randomized into three treatment groups: 300 mg clopidogrel (loading dose) and 325 mg/d aspirin on day 1, followed by 75 mg/d clopidogrel and 325 mg/d aspirin (days 2-28); 75 mg/d clopidogrel and 325 mg/d aspirin (days 1-28); and 250 mg bid ticlopidine and 325 mg/d aspirin (days 1-28). The 300 mg loading dose of clopidogrel vs. ticlopidine was well-tolerated, with no increase in the risk of bleeding. Difference in efficacy of clopidogrel vs. ticlopidine could not be evaluated in this trial due to insufficient power. Results are listed in Table 8, below.
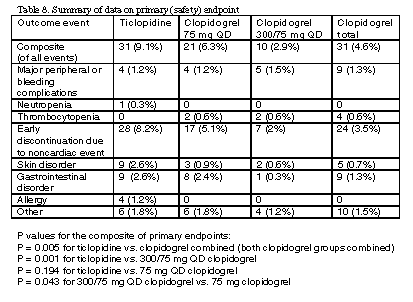
Thrombocytopenia was observed in four clopidogrel patients. These events were reported as transient and without clinical significance. There were no reports in the other treatment groups.
The large sample size and the design of the CLASSICS trial were its strengths. This trial showed safety of clopidogrel with or without a loading dose to be greater than ticlopidine. The loading dose regimen of clopidogrel had a greater statistical significance than the clopidogrel regimen without a loading dose. The limitations of the CLASSICS trial were a short duration (28 days), a lack of statistical power to establish efficacy of one drug vs. the other, and strict inclusion/exclusion criteria that made the results of this trial difficult to apply to the entire cardiovascular disease population. Also, one of clopidogrel’s advantages is its faster onset of action; this trial allowed a six-hour time frame to give the study drug. If this trial had demonstrated enough power to prove superior efficacy of either drug, this time frame possibly could affect the results.
Several other studies have been conducted with these two agents in patients undergoing coronary stent placement.5 With current information, it appears that clopidogrel is associated with a very low rate of stent thrombosis, with efficacy similar to that of ticlopidine, and clopidogrel is better tolerated.
CURE trial
The recently published CURE trial evaluated the efficacy and safety of the antiplatelet agent clopidogrel with aspirin vs. aspirin alone in more than 12,000 patients with acute coronary syndromes (ACS) without ST-segment elevation.6
The trial was a randomized, double-blind, placebo-controlled study enrolling ACS patients who presented within 24 hours following symptom onset; patients received either a clopidogrel 300 mg loading dose followed by 75 mg/d plus aspirin (75-325 mg/d), or placebo plus aspirin for 3-12 months.
The first primary outcome was the composite of death from cardiovascular causes, nonfatal MI, or stroke. The second primary outcome was the composite of the first primary outcome or refractory ischemia.
The secondary outcomes were the need for revascularization, health failure, and severe anemia. The safety outcomes were major and minor bleeding. The triple composite, first primary outcome occurred in 9.3% of the clopidogrel group and 11.4% of the placebo group (P < 0.001).
The second primary outcome occurred in 16.5% of the clopidogrel group and 18.8% of the placebo group (P < 0.001). All secondary endpoints occurred significantly less often in the clopidogrel group. Major bleeding occurred in 3.7% of the clopidogrel group and 2.7% of the placebo group (P = 0.001).
The rate of the first primary outcome was lower with clopidogrel within the first 30 days, and clopidogrel’s benefit was apparent within the first few hours after randomization. Study drug was discontinued permanently in 21.1% and 18.8% of clopidogrel and placebo patients, respectively.
The CURE study demonstrates the benefit of the clopidogrel-aspirin combination in the trial population, as well as an increased risk of bleeding.
Other selected studies
In another trial on patients undergoing coronary artery stenting, 243 study patients were randomized to ticlopidine 500 mg/d with and without aspirin 100 mg/d.7 The primary endpoint was the absence of death, cardiac events, and vascular access-site complications during hospitalization.
Follow-up clinical exams and angiography were performed at three months. Stent thrombosis occurred in two combined-treatment patients and in none in the monotherapy group. No significant differences in the primary endpoint were demonstrated between the two groups. There are several important weaknesses with this study, and the results need to be confirmed in larger multicenter trials.
In a randomized, unblended trial in 700 patients undergoing coronary stenting, patients received either clopidogrel 75 mg/d or ticlopidine 250 mg bid for 30 days, with no loading doses. Both regiments included aspirin 100 mg/d.8
Glycoprotein IIb/IIIa receptor blockers were administered in 7% and 11% of the ticlopidine and clopidogrel patients, respectively. The primary endpoint was the incidence of noncardiac events at 30 days (i.e., noncardiac death, stroke, leukopenia/thrombocytopenia, hemorrhage, or vascular complication or intolerance). Occurrence rates were 4.5% for clopidogrel and 9.6% for ticlopidine (P = 0.01).
The secondary endpoint of cardiac-related events at 30 days (i.e., thrombotic stent occlusion, target vessel revascularization, nonfatal MI, and cardiac death) was lower with ticlopidine (1.7% vs. 3.1%) but did not prove to be statistically significant. The authors proposed that a clopidogrel loading dose might be beneficial.
Several nonrandomized, observational studies indicate that clopidogrel may be similar in efficacy to ticlopidine but causes fewer adverse effects when used as prophylaxis with coronary stent thrombosis, when both drugs are combined with aspirin.9-11 Investigators recommend large controlled trials be done to study these issues.
Clopidogrel doses of either 50 mg, 75 mg, or 100 mg daily or ticlopidine 250 mg bid were started the day following coronary artery bypass surgery in 62 patients studied.12 At postoperative day 9, clopidogrel did not significantly inhibit platelet aggregation vs. baseline, but ticlopidine exerted a significant antiplatelet effect. At 28 days post-op, all three clopidogrel doses demonstrated a significant antiplatelet effect, and short-term outcomes were similar in both groups. The authors speculated that a clopidogrel loading dose may be necessary in this population.
Summary
The CAPRIE, TASS, and CATS trials showed both drugs to be effective. The CLASSICS trial showed clopidogrel to have a better safety profile than ticlopidine. Although neutropenia and TTP are less common with clopidogrel than ticlopidine, these reactions can occur. Patients and health care providers need to be aware and watchful for signs and symptoms of these adverse events. Both drugs have demonstrated efficacy with coronary stent placement.
Clopidogrel has a number of advantages over ticlopidine, including once-daily dosing, better safety and tolerability, no serial blood testing required, and faster onset of action. Ticlopidine recently has become available as a generic drug, thus decreasing the cost significantly; however, the cost of laboratory monitoring may offset these savings. These characteristics give clopidogrel an advantage over ticlopidine, along with the fact that prescribing practices have trended toward clopidogrel (five patient therapy days/month of ticlopidine use vs. 1,200 patient therapy days/ month of clopidogrel use). Ticlopidine has no significant advantage over clopidogrel, other than possibly cost; therefore, clopidogrel can be recommended as the formulary "workhorse drug." Considering the current prescribing trend, this interchange will be relatively simple.
General recommendations
- Patients entering the hospital on ticlopidine should be changed to clopidogrel, if there are no contraindications.
- Patients prescribed one of these drugs while in the hospital should be started on clopidogrel.
- Educate clinicians and patients on clopidogrel-induced TTP and neutropenia, which occurs with low frequency. Although no specific laboratory monitoring is required for clopidogrel, practitioners should be watchful for signs and symptoms of TTP and neutropenia.
Interchange doses
Ticlopidine (Ticlid) 250 mg bid å clopidogrel (Plavix) 75 mg QD
Steps: Initiating clopidogrel into pharmacy
- Establish pharmacist interchange for these antithrombotic agents.
- Educate on clopidogrel formulary interchange program prior to implementation.
- Evaluate program once in place to assess problems, compliance, and outcomes.
- Address any problems that arise and re-educate, as needed.
References
1. Gent M, et al. The Canadian American Ticlopidine Study (CATS) in thromboembolic stroke. Lancet 1989; 1:1215-1220.
2. Hass WK, et al. A randomized trial comparing ticlopidine with aspirin for the prevention of stroke in high-risk patients. N Engl J Med 1989; 321:501-507.
3. CAPRIE Steering Committee. A randomized, blinded trial of clopidogrel vs. aspirin in patients at risk of ischaemic events (CAPRIE). Lancet 1996; 348:1329-1338.
4. Bertrand ME, et al. Double-blind study of the safety of clopidogrel with and without a loading dose in combination with aspirin, compared with ticlopidine in combination with aspirin after coronary stenting. The Clopidogrel Aspirin Stent International Cooperative Study (CLASSICS). Circulation 2000; 102:624-629.
5. Berger P. Clopidogrel instead of ticlopidine after coronary stent placement: Is the switch justified? Am Heart J 2000; 140:354-358.
6. CURE Study Investigators. Effects of clopidogrel in addition to aspirin in patients with acute coronary syndromes without ST-segment elevation. N Eng J Med 2001; 345:494-502.
7. Machraoui A, et al. Efficacy and safety of ticlopidine monotherapy vs. ticlopidine and aspirin after coronary artery stenting: Follow-up results of a randomized study. J Invasive Cardiol 2001; 13:431-436.
8. Muller C, et al. A randomized comparison of clopidogrel and aspirin vs. ticlopidine and aspirin after the placement of coronary-artery stents. Circulation 2000; 101:590-593.
9. Berger PB, et al. Clopidogrel vs. ticlopidine after intracoronary stent placement. J Am Coll Cardiol 1999; 34:1891-1894.
10. Mishkel GJ, et al. Clopidogrel as an adjunctive anti-platelet therapy during coronary stenting. J Am Coll Cardiol 1999; 34:1884-1890.
11. Moussa I, et al. Effectiveness of clopidogrel and aspirin vs. ticlopidine and aspirin in preventing stent thrombosis after coronary stent implantation. Circulation 1999; 99:2364-2366.
12. David JC, Limet R. Antiplatelet activity of clopidogrel in coronary artery bypass graft surgery patients. Thromb Haemost 1999; 82:1417-1421.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
