Pediatric Rapid Sequence Intubation: An In-depth Review
Pediatric Rapid Sequence Intubation: An In-depth Review
Pediatric advanced airway management is a critical skill for the emergency medicine practitioner. Safe, effective, emergent intubation can be completed with the use of rapid sequence intubation (RSI). A multitude of alternatives are available with a variety of advantages and disadvantages based on the individual clinical scenario. This article provides a comprehensive review of the process and alternatives associated with pediatric RSI.
— The Editor
Introduction
Pediatric advanced airway management is an important intervention performed for critically ill or injured children in the emergency department (ED). Approximately 270,000 children require endotracheal intubation in the emergency department each year, comprising 0.2% of all ED visits.1 Despite its infrequent occurrence, endotracheal intubation is a potentially lifesaving procedure for children with respiratory compromise and impending respiratory failure. Factors related to pediatric airway anatomy, provider education and training, and the availability of the appropriate equipment can significantly affect patient outcomes.
Safe emergent endotracheal intubation can be achieved through the process of rapid sequence intubation (RSI), using a sequential approach to preparation, sedation, and paralysis prior to intubation.2 RSI must be differentiated from rapid sequence induction used by anesthesiologists to induce general anesthesia. RSI is the preferred method of intubation in the ED, where most patients present with varying levels of consciousness, protective airway reflexes and increased potential for pulmonary aspiration of gastric contents.2-4 While there are no absolute contraindications to RSI, patients in cardiac arrest and those who are comatose without a gag reflex generally do not warrant RSI medications prior to intubation.2 Decisions involving the RSI process are impacted not only by the individual case at hand, but also by a provider’s stylistic approach. As with most things in medicine, practice variation exists and the choices of medications will vary depending on the clinical scenario. This article will serve as a review of standard practice in the ED when caring for the pediatric patient who requires intubation.
Patient Preparation
It is imperative for ED clinicians to obtain a quick history prior to deciding which medications are appropriate for pediatric RSI. A simple mnemonic, AMPLE, serves as a reminder for the hurried clinician: A = allergies & airway history; M = medications; P = past medical history, family history, and anesthetic history; L = last oral intake; and E = events leading up to the intubation.5,6 (See Table 1.) Before preparing for intubation, a rapid but thorough assessment of the patient’s head, neck, neurologic status, airway, and breathing should be performed.7 The patient should be placed in the proper “sniffing” position with the neck in slight extension. Because young children have a prominent occiput, a towel roll may be placed underneath the shoulders to better align the oral and pharyngeal axis and allow maximal laryngeal visualization. The patient should be positioned as close to the edge of the bed as possible, and the bed should be above the waist level of the person performing the intubation to further facilitate correct visualization.
Table 1. History Prior to RSI

Preoxygenation is an essential preparation phase prior to pediatric RSI for achieving maximal pre-intubation oxygen saturation. Many children who require intubation are hypoxic with very poor oxygen reserves or have impending respiratory failure. Children have a higher oxygen consumption rate with a lower functional residual capacity and alveolar volume compared to adults, making children more susceptible to precipitous decline in oxygen saturation.2,8 Preoxygenation allows oxygen to replace nitrogen in the lungs, permitting longer periods of apnea before hypoxemia occurs. Two minutes of administering 100% oxygen to a spontaneously breathing child will wash out approximately 95% of alveolar nitrogen.2,7 Application of 100% oxygen at 15 L/minute via a non-rebreather mask for at least 3 minutes maximizes the oxygen reservoir in the alveoli and the rest of the body.9 While not included in the standard RSI protocol, noninvasive positive pressure ventilation (PPV) is reserved for patients who cannot achieve acceptable oxygen saturation with a nonrebreather mask due to inadequate respiratory effort or advanced lower airspace disease.2,6,9,10 Caution should be taken when administering PPV via manual bag-mask ventilation as a means of preoxygenation because of gastric insufflation and the risk of subsequent regurgitation and aspiration. A trial of PPV also allows the ED clinician to test the adequacy and ease of mask ventilation prior to intubation.10
Cricoid pressure, often referred to as the Sellick maneuver, is commonly used in pediatric RSI to minimize gastric insufflation during PPV and to prevent regurgitation of gastric contents. Using the thumb and index or middle finger, pressure is applied posteriorly over the cricoid cartilage to compress the esophagus once the patient is unconscious. This maneuver may also serve to position the vocal cords in a more direct, aligned view for intubation.2,7 Cricoid pressure should be maintained until the correct position of the ETT is confirmed. The safety and efficacy of the Sellick maneuver has been challenged.10 When performed incorrectly, which is not uncommon, cricoid pressure may interfere with direct visualization of the larynx.5 In rare but severe cases, esophageal rupture may result if cricoid pressure is applied in patients with active vomiting. The decision to use the Sellick maneuver should be made on an individual basis, with gentle force being applied to the cricoid cartilage with caution. Lastly, a nasogastric tube should be placed to deflate excess air in the stomach prior to intubating.
Equipment Preparation
When preparing for successful RSI, all necessary monitors, equipment, personnel, and medications must be assembled. A common mnemonic to aid with preparation is SOAPME (see Table 2): Suction with a Yankauer tip; Oxygen; Airway equipment (uncuffed endotracheal tube [ETT] size = 4 + [age/4]; use 0.5 smaller for a cuffed ETT); Pharmacologic agents; and Monitoring Equipment. Laryngoscope blade size is determined based on the patient’s age. (See Table 3.)
Table 2. Equipment Preparation

Table 3. RSI Equipment Recommendations
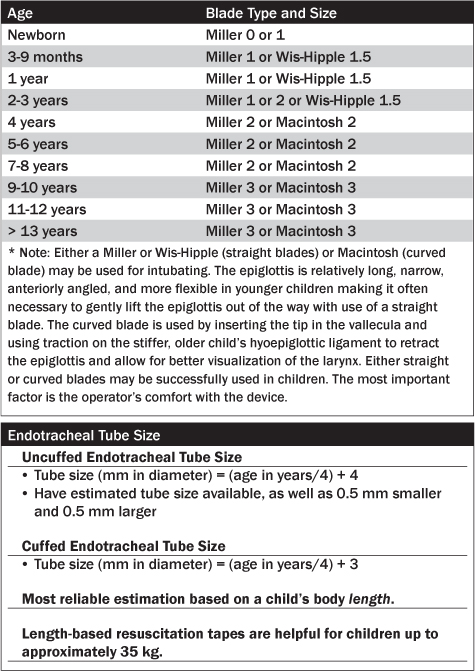
Recently updated Pediatric Advanced Life Support (PALS) guidelines recommend the use of cuffed endotracheal tubes for all ages except the newborn period. Past recommendations guided clinicians to use uncuffed ETTs until age 8 years unless the patient had underlying pulmonary disease.5,6,11-13 The subglottic area is the narrowest part of the pediatric airway and creates an anatomical seal around the ETT at that level. Concerns have been raised about using cuffed ETTs in younger children and include the potential to cause increased airway resistance, laryngeal mucosal injury, and later subglottic stenosis. These concerns, however, are unfounded in the literature.11,12 A prospective cohort study of 597 children younger than 5 years old demonstrated no significant difference between cuffed and uncuffed ETT in the incidence of post-extubation subglottic edema.12 Similarly, a randomized, controlled trial of 2246 children younger than 5 years old showed that cuffed ETTs do not increase the risk of post-extubation stridor.14 Uncuffed ETTs may be associated with air leak, risk of aspiration, difficulty with effective ventilation, and accurate measurement of end tidal CO2 concentration. Cuffed ETTs, on the other hand, are a more reliable way to deliver positive pressure ventilation due to smaller air leak, and they decrease the potential risk of gastric aspiration. Regardless of whether an uncuffed or cuffed ETT is used, it is imperative that clinicians use the correct size ETT to avoid complications such as airway trauma and mucosal ulceration.13
Premedications
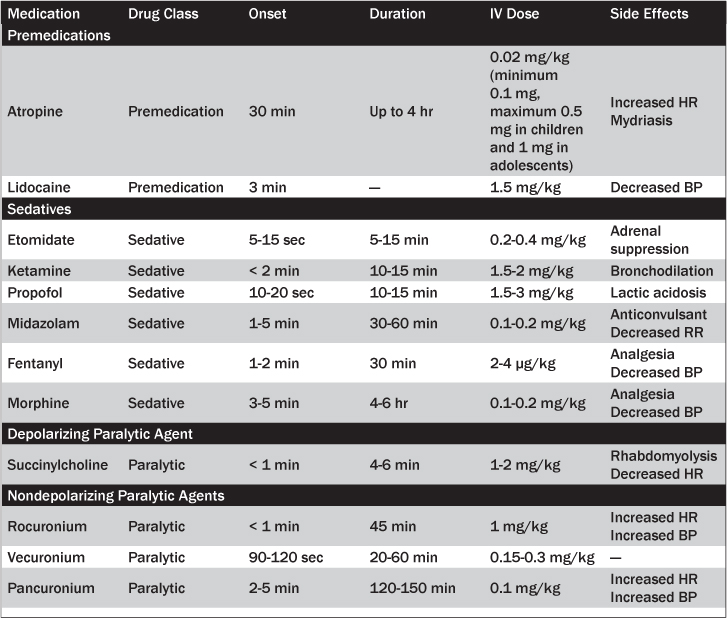
Atropine. Infants and children may experience bradycardia during intubation secondary to hypoxia, vagal nerve stimulation, and effects of medications such as succinylcholine.15,16 Atropine is a nonspecific antagonist of acetylcholine at muscarinic receptors and prevents reflex bradycardia by inhibiting vagal stimulation of the sinoatrial node. In one retrospective cohort study, approximately 10% of emergency intubations were associated with bradycardia (indicated by a pulse of less than 80 beats per minute for children younger than 2 years of age and less than 60 beats per minute for children 2 years of age or older).17 While the literature encourages the routine use of atropine as adjunctive therapy to prevent reflex bradycardia under certain circumstances, such as patients younger than 1 year of age, children younger than 5 years of age receiving succinylcholine, adolescents receiving a second dose of succinylcholine, and patients who are experiencing bradycardia prior to intubation, recent studies have questioned its efficacy.15,16,18,19 One retrospective cohort study of 143 pediatric patients younger than 19 years of age showed no statistically significant difference in the incidence of intubation-induced bradycardia for patients who received atropine compared to those who did not.
Adverse effects include sinus tachycardia, mydriasis, xerostomia, decreased micturition, and hyperthermia.16 It is prudent to perform a pupillary exam before giving atropine because mydriasis can persist for hours after a single dose.
When administered, atropine should be given 1 to 2 minutes prior to any sedative or paralytic agent at a dose of 0.02 mg/kg (minimum 0.1 mg, maximum 0.5 mg in children and 1 mg in adolescents). Doses less than 0.1 mg have been associated with paradoxical bradycardia.
Lidocaine. Lidocaine is used as an adjunct to pediatric RSI to attenuate the increase in intracranial pressure (ICP) associated with laryngoscopy and intubation. The direct mechanism by which it works remains unclear, although purported mechanisms include cough reflex suppression, brain stem depression, and decreased cerebral metabolism.4 Pediatric studies regarding the efficacy of the use of lidocaine are conflicting.4,5,7,10,20,21 Opponents of lidocaine indicate hypotension as a major reason not to administer it. Without strong evidence warranting its use, ED clinicians must decide on a case-by-case basis whether or not to include lidocaine in the RSI algorithm. If used, the recommended lidocaine dose for pediatric RSI is 1.5 mg/kg IV given 3 minutes before intubation.
Sedatives
Many factors should be considered when selecting the appropriate sedative to use for ED-initiated RSI. Practice variation in the choices of sedative agents is to be expected. Each hospital setting varies in its medication availability and provider comfort and experience using the different agents. Minimal adverse side effects are tolerable as long as the medications are easy to administer, rapid in onset, and have a high therapeutic index with few drug interactions.
Etomidate. Etomidate has become the drug of choice for most pediatric intubations in the ED because of its rapid onset of action (5 to 15 seconds), short duration of sedation (5 to 14 minutes), and minimal cardiovascular side effects.10,22-25 Etomidate is favored for critically ill children with tenuous cardiovascular status because of its minimal effects on mean arterial pressure and for children suffering head trauma because it causes a transient decrease in intracranial and intraocular pressures. There are several recent studies evaluating the safe use of etomidate in pediatric RSI. A prospective observational study by Zuckerbraun et al26 demonstrated that when using etomidate at conventional doses (0.3 mg/kg IV), 65.8% of 77 pediatric patients were intubated successfully on the first attempt. Another study of 105 pediatric patients younger than 10 years of age found that 0.3 mg/kg of etomidate caused trivial cardiovascular side effects without the adverse effects of adrenal suppression, myoclonus, status epilepticus, or new-onset seizures.23 These results were similar to those of Sokolove et al,27 who demonstrated a minimal (1 mm Hg) mean drop in systolic blood pressure after etomidate was used in pediatric RSI for 100 children younger than 10 years of age.
One major limitation to the use of etomidate is the dose-dependent, reversible inhibition of adrenocortical activity leading to decreased cortisol production for up to 15 hours.5,22 Etomidate blocks the enzyme 11-β-hydroxylase, which is involved in adrenal steroid production. While there are no absolute contraindications to its use, relative contraindications include patients with the potential for adrenal insufficiency, such as those in septic shock and those with focal seizure disorders, since etomidate tends to lower the seizure threshold.19 Additional adverse effects include the potential to cause myoclonic activity, such as coughing or hiccupping.
The recommended RSI dose range for etomidate is 0.2 to 0.4 mg/kg IV with clinical recovery in 10 to 15 minutes.
Ketamine. Ketamine is a dissociative analgesic derived from phencyclidine (PCP). Patients given ketamine characteristically exhibit a trance-like state with open eyes, a slow nystagmic gaze, and occasional mild respiratory depression despite preserved airway reflexes.5 It is the only agent that causes bronchodilation, a helpful property for use in patients suffering from severe bronchospasm or status asthmaticus, making it the preferred agent for patients with respiratory failure secondary to asthma. An additional side effect of ketamine is the sympathomimetic stimulation of the cardiovascular system, leading to transient tachycardia and hypertension and an increase in ICP. While ketamine may help the cardiovascular status of patients with signs of shock, it is currently contraindicated in patients with increased ICP and uncontrolled hypertension until further research is conducted. Additional relative contraindications to ketamine use include thyrotoxicosis and major depressive disorders because of an emergence phenomenon that can be associated with visual and auditory hallucinations.5
The recommended RSI dose for ketamine is 1.5 to 2 mg/kg IV with clinical recovery in 10 to 15 minutes.
Propofol. Propofol is a lipophilic sedative-hypnotic that produces general anesthesia.2 It has a very rapid onset (10 to 20 seconds) and short duration of action (10 to 15 minutes).7,25 Propofol use is associated with vasodilation, decreases in mean arterial pressure and intracranial pressure, and myocardial depression. It should be given in hemodynamically stable patients without evidence of hypotension.
Propofol contains egg lecithin and soybean oil and, therefore, is unsafe to use in patients with allergies to eggs and/or soybeans. When given in high doses for a prolonged period of time, propofol may be associated with a fatal condition known as propofol infusion syndrome (PIS). This syndrome is characterized by severe lactic acidosis, rhabdomyolysis, acute renal failure, recalcitrant myocardial failure, hypotension, bradycardia, and hyperlipidemia.28 Proposed risk factors include head injury, airway infection, young age (younger than 18 years old), low carbohydrate intake/high fat intake, inborn errors of fatty acid oxidation, high doses of propofol for more than 48 hours, and combined use of catecholamine vasopressors and/ or glucocorticoids.28,29 Serum lactate and creatine phosphokinase (CPK) levels should be monitored closely when infusing propofol.
The recommended RSI dose range for propofol is 1.5 to 3 mg/kg IV with clinical recovery in 10 minutes.
Midazolam. Midazolam is a rapid-acting benzodiazepine with amnestic, anxiolytic, and anticonvulsant properties. Midazolam is the sedative agent of choice for cardiovascularly stable patients presenting with status epilepticus because of its potent anticonvulsant effects.2,20,25 Side effects of midazolam include respiratory depression occasionally associated with apnea, myocardial depression, and hypotension secondary to a dose-related decrease in systemic vascular resistance (SVR). Because of its non-trivial cardiovascular side effects, it should be avoided in hemodynamically unstable patients.
The recommended RSI dose for midazolam is 0.1-0.2 mg/kg IV, with clinical recovery in 30 to 60 minutes.
Narcotics. Narcotics, such as fentanyl and morphine, may be used as sedative agents for RSI. They provide both anesthesia and analgesia while decreasing sympathetic tone, and therefore are good agents for patients exhibiting pain prior to intubation. Compared with morphine, fentanyl is more lipophilic, causing a faster onset (1 to 2 minutes) and shorter duration (approximately 30 minutes) of action. Fentanyl also causes less histamine release but can be associated with chest wall rigidity with compromise in ventilation when rapidly infused. Morphine has a longer onset of action (3 to 5 minutes) and longer duration of action (4 to 6 hours). Increased histamine release associated with morphine may cause significant pruritis. The large doses of both medications often required to achieve significant sedation and analgesia can lead to significant sedative effects, a decrease in SVR, and hypotension.2,25
The recommended RSI dose for fentanyl is 2-4 μg/kg IV and morphine is 0.1-0.2 mg/kg IV.
Table 4. Summary of RSI Medications
Neuromuscular Blocking (Paralytic) Agents
Neuromuscular blocking agents provide muscle paralysis to facilitate endotracheal intubation. They must be used in conjunction with a preceding sedative to achieve full RSI effect.
There are two main classes of neuromuscular blocking agents: depolarizing and nondepolarizing. Both induce motor paralysis by inhibiting acetylcholine stimulation of nicotinic receptors at the neuromuscular junction.
Depolarizing Agents
Succinylcholine. Succinylcholine is a commonly used paralytic agent in pediatric RSI. Its benefits include an extremely fast onset (< 1 minute) and a short duration of action (4 to 6 minutes).2,5,24 It is most often used in previously healthy children and in cases in which brief, transient paralysis is preferred, such as with difficult intubations.
There are potential significant adverse effects associated with succinylcholine use. Administration of this drug may be associated with bradycardia and, in extreme cases, asystole with repeat doses. For these reasons, although the use of atropine has been questioned, it is typically recommended as premedication in children younger than 5 years old or adolescents receiving a second dose of succinylcholine. Succinylcholine is absolutely contraindicated in children with underlying muscular dystrophy because it interacts with the unstable muscle membranes leading to severe, prolonged rhabdomyolysis and life-threatening hyperkalemia. Succinylcholine can also produce extreme hyperkalemia and should be avoided in the setting of malignant hyperthermia, large body surface area burns, multisystem trauma, and traumatic spinal cord and other denervating injuries. Succinylcholine may lead to an increase in ICP and intraocular pressure and should be avoided in patients with intracranial injuries, brain tumors, and penetrating eye trauma.2,20
It is important to note that defasciculation and priming are no longer recommended when using succinylcholine due to increased medication errors associated with this practice.2
The recommended dose for RSI is 2 mg/kg IV in infants and children younger than 2 years of age, and 1 mg/kg IV in children ages 2 years and older. This age-related dosing discrepancy stems from the fact that succinylcholine is distributed in the extracellular fluid, and younger children tend to have increased extracellular water compared to older children.2
Nondepolarizing Agents
Rocuronium. Rocuronium is a nondepolarizing neuromuscular blocking agent that induces muscle paralysis by competing with the nicotinic cholinergic receptor. It has a rapid onset of action (less than 1 minute) and can last up to 45 minutes. It is recommended in patients with absolute contraindications to succinylcholine such as muscular dystrophy and/or malignant hyperthermia. Rare adverse effects include tachycardia and hypertension.22 The recommended dose for pediatric RSI is 1 mg/kg IV.2,20,22,24
Vecuronium. Vecuronium is an alternative nondepolarizing agent that has a longer onset of action (90 to 120 seconds) and duration of action (20 to 60 minutes). Caution should be taken when using this agent in patients in whom endotracheal intubation may be challenging because its effects tend to last longer than other paralytic options. The recommended dose for pediatric RSI is 0.15 to 0.3 mg/kg IV.
Pancuronium. Pancuronium is another nondepolarizing agent with a much slower onset (2 to 5 minutes) and long duration of action (120 to 150 minutes). It is primarily used for maintenance of paralysis after initial RSI. Significant side effects include a profound vagolytic effect leading to increased heart rate, blood pressure, and cardiac output.2
The recommended dose is 0.1 mg/kg IV for infants and 0.15 mg/kg IV for children.2
Clinical Applications in a Variety of Scenarios
Pediatric emergency medicine (PEM) physicians at Children’s National Medical Center, a busy level-1 pediatric trauma center in Washington, DC, were asked to complete an anonymous online survey of their preferred choice of sedative and paralytic agent in five different case scenarios. Seventeen physicians responded electronically on SurveyMonkey®.
In patients with intracranial injury, 16 of 17 providers (94.1%) recommended using etomidate as the sedative of choice. One physician selected midazolam. The majority, 14/17 (82.4%), chose rocuronium as the paralytic agent of choice, 2/17 (11.8%) chose succinylcholine, and 1/17 (5.9%) chose vecuronium.
In patients with septic shock, 11/17 (64.7%) of the PEM providers recommended ketamine as the preferred sedative. Four of 17 (23.5%) preferred etomidate, 1/17 (5.9%) chose midazolam, and 1/17 (5.9%) chose fentanyl. For paralysis, 14/17 (82.4%) providers recommended rocuronium, 2/17 (11.8%) chose succinylcholine, and 1/17 chose (5.9%) vecuronium.
In patients with status asthmaticus, 17/17 (100%) of the PEM providers recommended ketamine as the sedative agent of choice. Paralytic agent choices varied. Eleven of 17 (64.7%) recommended rocuronium, 5/17 (29.4%) recommended succinylcholine, and 1/17 (5.9%) vecuronium.
In patients with status epilepticus, midazolam was the preferred sedative, with 13/16 (81.3%) in favor of its use. Two of 16 (12.5%) recommended etomidate, and 1/16 (6.3%) suggested ketamine. One provider did not answer the question. The preferred paralytic agent was rocuronium, with 14/17 (82.4%) supporting its use. Two of 17 (11.8%) recommended succinylcholine, and 1/17 (5.9%) recommended vecuronium.
In patients with non-asthma-related respiratory failure, 10/16 (62.5%) chose ketamine, and 6/16 (37.5%) chose etomidate. One provider did not answer the question. The choice of paralytic agents was equal for succinylcholine and rocuronium at 8/17 (47.1%), respectively. One of 17 (5.9%) recommended vecuronium.
Confirmation of Endotracheal Intubation
Upon successful endotracheal intubation, the ETT should be secured and proper positioning confirmed. The ETT depth should be gauged by the equation: ETT depth at lips = 3 x tube size (mm in diameter). Breath sounds must be evaluated by auscultation over bilateral axillary areas with visualization of adequate, symmetric chest rise during bag insufflation. End-tidal carbon monoxide monitoring is essential to ensure adequate ventilation. This can be achieved continuously with mainstream monitoring directly from the ETT or as a one-time immediate confirmation using a colorimetric device. Lastly, a chest radiograph should be performed to confirm appropriate ETT placement. Documentation of vital signs, including temperature, heart rate, blood pressure, pulse oximetry, and end-tidal carbon monoxide level, is recommended every 5 minutes.
Complications of Endotracheal Intubation
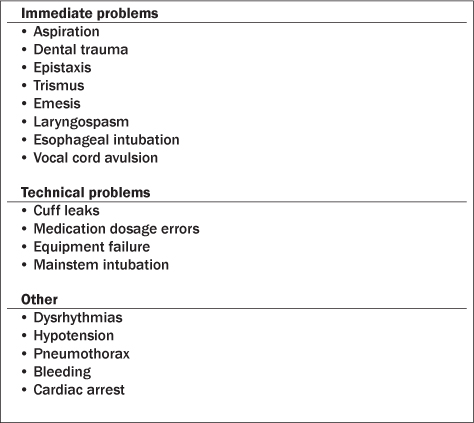
Respiratory complications of pediatric intubation are much more common than in adults, particularly in the setting of younger patients, a non-fasting state, and underlying comorbidities.5 The frequency of complications associated with endotracheal intubation in pediatric patients outside the operating room has been estimated to range between 15% and 38%.19 Complications often stem from characteristics of the pediatric airway that are markedly different from the adult airway and pose challenges for the ED clinician caring for pediatric patients.7,8,17 Children have proportionally larger heads and occiputs, causing passive anterior neck flexion and airway obstruction when lying supine. They have relatively larger tongues with less muscle tone, allowing for less space in the mouth for intubation. The epiglottis in children is shorter, narrower, and softer, and the larynx is located more anteriorly, making visualization of the cords more difficult. The trachea in children is shorter and located more anteriorly, increasing the risk of right mainstem intubation. Finally, the airway in children is narrower with increased airway resistance.
Complications with endotracheal intubation are often unpredictable and can be quite frightening even to the experienced clinician. Immediate problems include aspiration, dental trauma, emesis, epistaxis, laryngospasm, lip laceration, trismus, esophageal intubation, and vocal cord avulsion. Technical problems involve cuff leaks, medication dosage errors, equipment failure, mainstem intubation, and self-extubation. Patients may develop dysrhythmias, hypotension, pneumothorax, bleeding, and even cardiac arrest during endotracheal intubation. (See Table 5.) ED providers need to prepare for the aforementioned risks and know how to manage them.3,30,31 A retrospective, observational study using video review in a tertiary pediatric ED by Kerrey et al32 demonstrated that complications associated with pediatric RSI may be more prevalent than previously thought. Of the 114 pediatric patients requiring RSI on video, 70 (61%) experienced at least one adverse event.32 The most common complications were oxyhemoglobin desaturation, right mainstem intubation, and non-airway intubation. Two children had physiologic deterioration necessitating CPR. Lastly, 11 minutes is considered to be the gold standard by the Manual of Emergency Airway Management for time to RSI completion — from medication decision-making to confirmation of ETT placement. This time was achieved in 45% of the patients in the study, implying there is much room for improvement.
Table 5. Complications with Endotracheal Intubation
Video Laryngoscopy
Video laryngoscopy is becoming a popular tool to aid clinicians with RSI in both prehospital and hospital settings. Video laryngoscopy uses a camera associated with a laryngoscope blade to allow for both direct and indirect visualization of the glottis during intubation. Real-time visualization of the intubation on a video monitor improves the view of the larynx and the success of endotracheal tube placement. The use of video laryngoscopy has increased the success rates of both standard intubations and those with difficult or failed attempts by traditional direct visualization.33,34
Safety of RSI
Numerous studies confirm the safety and efficacy of pediatric RSI in the ED. According to recent literature, pediatric RSI is associated with fewer attempts at laryngoscopy30,35 and better success at achieving endotracheal intubation.35 RSI remains the method of choice for managing an emergent airway in the ED. The Pediatric Emergency Medicine Committee of the American College of Emergency Physicians advocates for the use of RSI “in every emergency intubation involving a child with intact upper airway reflexes.”6,7,35 When conducted properly with appropriate RSI medications, intubation can be achieved with minimal complications.3
Conclusion
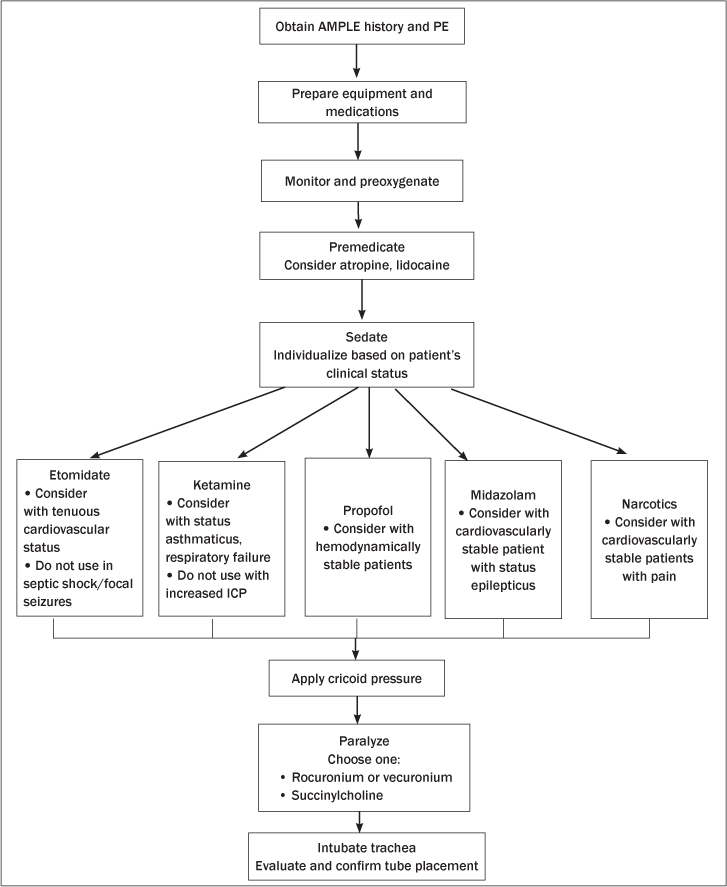
Pediatric RSI is a valuable process used to guide the successful advanced airway management in critically ill children. Patient safety is paramount when undertaking potentially life-saving procedures. Knowledge of pediatric-specific anatomy challenges, patient and equipment pre-intubation preparation, RSI medication recommendations, means of confirming proper ETT placement, and potential complications will give guidance to any clinician preparing for emergency pediatric intubation. (See Figure 1.)
Figure 1. Algorithm
References
1. National Hospital Ambulatory Medical Care Survey: 2009 Emergency Department Summary Tables. Center for Disease Control.
2. Agrawal D. Rapid sequence intubations (RSI) in children. UpToDate June 2012.
3. Tayal VS, Riggs RW, Marx JA, et al. Rapid-sequence intubation at an Emergency Medicine residency: Success rate and adverse events during a two-year period. Academic Medicine 1999;6:31-37.
4. Zelicof-Paul A, Smith-Lockridge A, Schnadower D, et al. Controversies in rapid sequence intubation in children. Curr Opin Pediatrics 2005;17:355-362.
5. McAllister JD, Gnauck KA. Rapid sequence intubation of the pediatric patient: Fundamentals of practice. Pediatric Clin North Am 1999;46:1249-1284.
6. Bledsoe GH, Schexnayder SM. Pediatric rapid sequence intubation: A review. Pediatric Emerg Care 2004;20:339-344.
7. Gerardi MJ, Sacchetti AD, Cantor RM, et al. Rapid sequence intubation of the pediatric patient. Pediatric Emergency Medicine Committee of the American College of Emergency Physicians. Ann Emerg Med 1996;28:55–74.
8. Bingham RM, Proctor LT. Airway management. Pediatric Clin North Am 2008;55:873-886.
9. Weingart SD, Levitan RM. Preoxygenation and prevention of desaturation during emergency airway management. Ann Emerg Med 2011; 1-12.
10. El-Orbany M, Connolly LA. Rapid sequence induction and intubation: Current controversy. Anesthesia Analgesia 2010;110:1318-1325.
11. Weber T, Salvi N, Wolf A. Cuffed vs non-cuffed endotracheal tubes for pediatric anesthesia. Pediatric Anesthesia 2009;19:46-54.
12. Newth CJL, Rachman B, Patel N, et al. The use of cuffed versus uncuffed endotracheal tubes in pediatric intensive care. Journal of Pediatrics 2004;144:333-337.
13. James I. Cuffed tubes in children. Paediatric Anesthesia 2001;11: 259-263.
14. Weiss M, Dullenkopf A, Fischer JE, et al. Prospective randomized controlled multi-centre trial of cuffed or uncuffed endotracheal tubes in small children. Br J Anesthesia 2009;103:867-873.
15. Fastle RK, Roback MG. Pediatric rapid sequence intubation: Incidence of reflex bradycardia and effects of pretreatment with atropine. Pediatric Emerg Care 2004;20:651-655.
16. Jones P, Dauger S, Peters MJ. Bradycardia during critical care intubation: Mechanisms, significance and atropine. Arch Dis Child 2012;97: 39-144.
17. Carroll CL, Spinella PC, Corsi JM. Emergent endotracheal intubations in children: Be careful if it’s late when you intubate. Pediatric Crit Care Med 2010;11:343-348.
18. PALS Provider Manual. American Academy of Pediatrics/American Heart Association; 2002:361.
19. McAuliffe G, Bissonnette B, Boutin C. Should the routine use of atropine before succinylcholine in children be reconsidered? Canadian Journal of Anesthesia 1995;42:724-729.
20. Frakes MA. Rapid sequence intubation medications: An update. J Emergency Nursing 2003;29: 553-540.
21. Robinson N, Clancy M. In patients with head injury undergoing rapid sequence intubation, does pretreatment with intravenous lignocaine/Lidocaine lead to an improved neurological outcome? A review of the literature. Emerg Med J 2001;18:453-457.
22. Ching KY, Baum CR. Newer agents for rapid sequence intubation: Etomidate and Rocuronium. Pediatric Emerg Care 2009;25: 200-210.
23. Guldner G, Schultz J, Sexton P, et al. Etomidate for rapid-sequence intubation in young children: Hemodynamic effects and adverse events. Acad Emerg Med 2003;10:134-139.
24. Mallon WK, Keim SM, Shoenberger JM, et al. Rocuronium versus succinylcholine in the emergency department: A critical appraisal. J Emerg Med 2009;37:183-188.
25. Yamamoto LG. Emergency airway management — rapid sequence intubation. In: Fleisher GR, Ludwig S, eds. Textbook of Pediatric Emergency Medicine, 6th edition. Lippincott Williams and Wilkins; Philadelphia: 2010:74-84.
26. Zuckerbraun NS, Pietti RD, Herr SM, et al. Use of etomidate as an induction agent for rapid sequence intubation in a pediatric emergency department. Acad Emerg Med 2006;13:602-609.
27. Sokolove PE, Price DD, Okada P. The safety of etomidate for emergency rapid sequence intubation of pediatric patients. Pediatric Emerg Care 2000;16:18-21.
28. Testerman GM, Chow TT, Easparam S. Propofol infusion syndrome: An algorithm for prevention. Brief Reports 2011;12:1714-1715.
29. Wong JM. Propofol infusion syndrome. American Journal of Therapeutics 2010;17:487-491.
30. Sagarin MJ, Chiang V, Sakles JC et al. Rapid sequence intubation for pediatric emergency airway management. Pediatric Emerg Care 2002;18:417-423.
31. Reid C, Chan L, Tweeddale M. The who, where, and what of rapid sequence intubation: Prospective observational study of emergency RSI outside the operating theatre. Emerg Med Journal 2004;21: 296-301.
32. Kerrey BT, Rinderknecht AS, Geis GL, et al. Rapid sequence intubation for pediatric emergency patients: higher frequency of failed attempts and adverse effects found by video review. Ann Emerg Med 2012;60:251-259.
33. Nagler J, Bachur RG. Advanced airway management. Curr Opin Pediatrics 2009;21:299-305.
34. Aziz MF, Healy D, Kheterpal S, et al. Routine clinical practice effectiveness of the Glidescope in difficult airway management: An analysis of 2,004 Glidescope intubations, complications and failures from two institutions. Anesthesiology 2011;114:34-41.
35. Gnauck K, Lungo J, Scalzo A, et al. Emergency intubation of the pediatric medical patient: Use of anesthetic agents in the emergency department. Ann Emerg Med 1994;23: 1242–1247.
Pediatric advanced airway management is a critical skill for the emergency medicine practitioner. Safe, effective, emergent intubation can be completed with the use of rapid sequence intubation (RSI). A multitude of alternatives are available with a variety of advantages and disadvantages based on the individual clinical scenario. This article provides a comprehensive review of the process and alternatives associated with pediatric RSI.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
