The Evidence-based Approach to Neurologic Emergencies: Part I: Acute Stroke
The Evidence-based Approach to Neurologic Emergencies: Part I: Acute Stroke
Author:
Ademola Adewale, MD, FAAEM, Assistant Professor of Emergency Medicine, Florida State University College of Medicine, Director of Research and Medical Simulation, Florida Hospital Emergency Medicine Residency Program, Orlando, FL.
Peer Reviewer:
Bentley J. Bobrow, MD, Associate Professor of Emergency Medicine, Emergency Medicine Department, Maricopa Medical Center, Phoenix, AZ.
Stroke remains a leading cause of death, but the disability associated with a stroke can be devastating and costly. In past decades, little could be done to reduce the morbidity and mortality of stroke. But over the past decade, use of thrombolytics by specialized stroke centers has reduced the morbidity of survivors. However, the reduction of morbidity comes at a cost of an increase in intracerebral hemorrhage, often associated with death.
Some emergency physicians have been reluctant to use thrombolytics in their patients with stroke. In large part that is due to the risk of intracranial hemorrhage, but it is also often due to the lack of a neurology specialist 24/7 to assist with the interpretation of the CT scan and back-up by neurosurgical specialists in case something untoward should occur. Many are concerned about the medical liability risk that may occur if they either order or fail to order thrombolytics.
Despite these concerns, there are now clear guidelines for the use of thrombolytics in stroke patients. This paper will review the most recent guidelines to assist physicians who work in centers with sufficient support to offer thrombolytic therapy for stroke.
Sandra M. Schneider, MD, FACEP, Editor
Introduction
Neurologic emergencies are common presentations to the emergency department (ED). Emergency physicians should have a broad-based understanding of the disease process and the best evidence-based approach to care for these patients to minimize morbidity and potential mortality. A neurologic emergency encompasses a broad variety of disease pathology; thus, it is impossible to discuss the entirety in a single paper. This paper will focus on one of the most common life-threatening entities: acute stroke.
Stroke in the United States is the leading cause of death; more than 800,000 people die each year from stroke and cardiovascular disease.1
Someone has a stroke every 40 seconds. Someone dies of stroke every 4 minutes.2
On a yearly basis, about 795,000 people have a stroke.
More than 600,000 of these are first time or new strokes.
Almost 185,000 people who survive a stroke have a second stroke.2
Stroke is an important cause of disability, and in 2005 almost 1.1 million survivors reported having difficulty performing daily tasks.3
The cost of stroke care is staggering. In 2010 alone, the estimated cost of stroke care was $53.9 billion.2
Incidence
The incidence of stroke varies widely depending on age, sex, ethnicity, and socioeconomic status. The American Heart Association (AHA) data4 suggest that blacks have a 3-fold higher multivariate–adjusted risk of lacunar stroke compared to whites. While typically stroke occurs in older adults, stroke can be seen in younger adults and even children, especially those with chronic disease such as sickle cell and those with cardiac defects such as patent ductus arteriousis. According to the Centers for Disease Control (CDC), about 85% of strokes are ischemic, while 10-12% are hemorrhagic. Transient ischemic attacks (TIA) and cryptogenic stroke are the remaining 3-5%.4
Risk Factors
The risk factors (see Table 1) for stroke are similar for all stroke types: hypertension, advanced age, previous history of a stroke, and illicit drug use. However, in hemorrhagic stroke, certain factors such as anticoagulation therapy, arteriovenous malformation, cerebral amyloidosis, coagulopathies, intracranial aneurysm, neoplasm, vasculitis, and thrombolytic therapy are significant contributors.
Table 1: Risk Factors for Stroke
|
Modifiable |
Not Modifiable |
|
|
The Common Blood Vessels in Stroke
Stroke presentation varies widely depending on the cerebral blood vessel involved. (See Table 2.) The major blood vessels of the brain are the right and left common carotids and the vertebral arteries. These two blood vessels merge to form the circle of Willis at the base of the brain. Vessels that emanate from the circle are the anterior cerebral artery (ACA), the middle cerebral artery (MCA), and the posterior cerebral artery (PCA). Emanating from the brainstem are the vertebral arteries that give off the anterior inferior cerebellar artery (AICA). It continues as the basilar artery (BA), which gives off the posterior inferior cerebellar artery (PICA) and superior cerebellar artery, before merging with the circle.
Table 2: Stroke Syndromes
|
Vessel |
Territory |
Clinical Presentation |
|
Adapted from www.strokecenter.org. Accessed April 2012. |
||
|
Anterior cerebral |
Frontal lobes Personality, voluntary movement |
Uncommon stroke. Unilateral may be silent Contralateral leg weakness Bilateral: akinetic mutism (inability to move or speak) Confusion |
|
Middle cerebral |
Some frontal lobe Lateral surface of temporal and parietal lobes Basal ganglia Anterior/posterior internal capsules |
Contralateral ataxia and hemiparesis Inability to write, calculate, right-left confusion Contralateral homonymous hemianopia, receptive aphasia, apraxia Contralateral hemiparesis, facial weakness, sensory loss, expressive aphasia |
|
Posterior cerebral |
Temporal lobe Occipital lobe |
Lethargy/agitation, hemiplegia hemisensory loss, ophthalmoplegia Homonymous hemianopia Visual agnosia Inability to interpret vision, poor hand eye coordination, apraxia of gaze Inability to recognize faces Alexia (inability to read) Disorder of color vision Memory impairment Contralateral ataxia, oculomotor palsy Cortical blindness (Anton syndrome) Contralateral hemisensory loss or pain (thalamic syndrome) Contralateral hemiparesis with lateral gaze weakness (Weber syndrome) Locked in |
|
Basilar |
Cerebellum, pons, brainstem |
Cortical blindness Contralateral hemiparesis, ipsilateral facial and lateral gaze weakness |
|
Vertebral artery |
Brainstem, cerebellum |
Ipsilateral facial sensory loss and pain, ataxia, nystagmus, Horner's syndrome, contralateral hemisensory loss to pain and temperature (Wallenberg syndrome) |
|
Lenticulostriate arteries |
Deep branches off MCA |
Lacunar infarctions |
The anterior cerebral artery supplies the frontal lobes (responsible for the control of logical thoughts, personality, and voluntary movements), and most of the medial surface of the cerebral cortex. Unilateral infarct involving this region results in contralateral sensory and motor deficits, which preferentially involves the lower extremities. The bilateral involvement of the ACA at their stems results in infarct involving the anterior medial aspect of the cerebral cortex. This infarct results in paraplegia of the lower extremities, incontinence, and a phenomenon known as akinetic mutism that manifests as motor aphasia and abulia (lack of ability to act or make decisions) and a profound frontal lobe release symptoms (personality change).
The middle cerebral artery (MCA) is the largest of the cerebral arteries and it is by far the most common blood vessel involved in stroke. The cerebral cortex supplied by the MCA is vast. It supplies the majority of the outer convex of the brain, almost all the basal ganglia, and the anterior and posterior internal capsules. Simply put, it supplies the primary motor areas of the face, throat, hand, arm, and the area of speech in the dominant hemisphere. Infarct involving this vessel manifests as a myriad of neurologic sequelae, which include contralateral hemiplegia involving the face, arms, and legs, homonymous hemianopsia, and global aphasia if the infarct involves the left cerebral hemisphere.5
The posterior cerebral artery (PCA) often emanates from the basilar artery or terminal branch of the basilar artery in most individuals, but sometimes from the ipsilateral internal carotid arteries.5 This vessel supplies the posteromedial temporal lobes and occipital lobes of the cerebral hemispheres. Infarcts in this area account for approximately 5-10% of ischemic stroke and often result as a consequence of an embolism from the vertebral basilar system. PCA infarcts often manifest as thalamic or thalamic perforate syndrome (contralateral hemianesthesia, loss of sense of position resulting in ataxia, excruciating pain on affected side), Weber's syndrome (contralateral upper and lower extremity weakness, and ipsilateral lateral gaze weakness), hemianopsia, and a myriad of other symptoms, such as acute visual loss, dizziness, nausea, and language disorder. However, occipital lobe infarct is the most common finding and it presents as a contralateral visual field defect.5
Infarcts involving lenticulostriate arteries and the penetrating branches of the circle of Willis, vertebral, and basilar arteries result in lacunar-type strokes. Lacunar type strokes account for about 20% of all strokes, and the most common risk factor is hypertension.6
Case Example
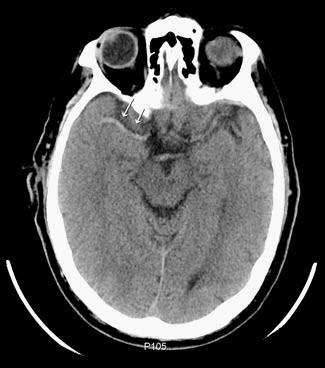
A 64-year-old man arrived via EMS with difficulty speaking and left side weakness. He was last seen well two hours ago. He is currently unable to speak or move his left side. His initial vital signs reveal a blood pressure of 224/121, pulse of 98, oxygen saturation of 99%, respiratory rate of 18, GCS 12, and the EMS blood sugar of 228 mg/dL. A nonenhanced CT scan of the brain shows no hemorrhage but does show the presence of a clot within the right MCA (the hyperdense MCA sign). (See Figure 1.)
Figure 1: Hyperdense MCA Sign
A CT angiogram of the head and neck (CTA) and CT perfusion were ordered, and the stroke neurologist was promptly consulted. The stroke neurologist confirmed the diagnosis of acute ischemic stroke.
Ischemic Stroke
Ischemic stroke results from vessel occlusion that reduces the blood flow to the perfused area of the brain. Embolic stroke accounts for 15-30% of all strokes. In a study by Moulin et al,7 cardio-embolism accounted for about 50% of MCA strokes, 34% of deep MCA strokes, and 41% of cortical strokes. In embolic stroke, a dislodged plaque or embolus travels to distal arteries or arterioles eventually disrupting the blood supply. In a thrombotic stroke, the obstruction occurs from a formed thrombus or a plaque that slowly propagates until large enough to completely occlude a vessel. The resultant manifestation is a cascade of cellular or microcellular events that lead to cerebral infarction. The pathophysiology of these cellular and microcellular cascades involves the release of excitatory amino acids, neurotransmitters, calcium influx into the cells, generation of free radicals, membrane depolarization, and the eventual loss of membrane integrity of the blood-brain barrier.7
The presentation of a stroke varies with the vessel involved. For the purpose of this section, emphasis will be placed on MCA infarct since this is the most common blood vessel involved in ischemic stroke.
Management
The management of stroke patients starts pre-hospital. The initial identification of a stroke patient and rapid transport to the appropriate facility may improve outcome. Several scoring systems have been developed for the prehospital personnel to aid in rapid identification of potential stroke patients. These scores include the ABCD score, the Los Angeles Pre-hospital Stroke Scale (LAPSS), and the Cincinnati Stroke Scale. (See Table 3.)
Table 3: Cincinnati Prehospital Stroke Scale
|
Normal |
Abnormal |
|
|
Facial Droop |
Both sides of face move equally |
One side of face does not move |
|
Arm Drift |
Both arms move equally |
One arm drifts compared to the other |
|
Speech |
Patient uses correct words with no slurring |
Slurred or inappropriate words or mute |
A noncontrast CT scan of the patient showing thrombus within the right MCA. This is referred to as the hyper-dense MCA sign. This is one of the early signs of ischemia.
Table 4: NIH Treatment Guideline/Sequence Considering the "Golden Hour"
Emergency department arrival to initial physician evaluation: 10 minutes
Emergency department arrival to stroke team notification: 15 minutes
Emergency department arrival to CT scan initiation: 25 minutes
Door to CT scan interpretation: < 45 minutes
On arrival of this patient to the emergency department, the following sequence of events should occur rapidly and simultaneously with the understanding that the goal to reperfusion is 60 minutes from arrival (see Table 4):
Determine and confirm the last time seen normal;
Establish IV access;
Draw routine laboratory studies, order noncontrast CT scan;
Perform detailed neurological examination using the screening assessment tools such as the NIH stroke scale (NIHSS);
Monitor blood pressure;
Obtain a 12-lead electrocardiogram (ECG) and assess for arrhythmia;
Review CT scan to exclude hemorrhage;
Exclude the presence of abnormal lucency on the CT scan;
Consult stroke neurologists;
Review laboratory test results;
Review inclusion and exclusion criteria for initiation of thrombolytic (see Table 5).
Table 5: Inclusion and Exclusion Criteria for Intravenous tPA Therapy in Patients with Acute Ischemic Stroke
tPA Indications
- Ischemic stroke onset within 3 hours of drug administration.
- Measurable deficit on NIH Stroke Scale examination.
- Patient's computed tomography (CT) does not show hemorrhage or non stroke cause of deficit.
- Patient's age is > 18 years.
Do NOT administer tPA if any of these statements are true:
- Patient's symptoms are minor or rapidly improving.
- Patient had seizure at onset of stroke.
- Patient has had another stroke or serious head trauma within the past 3 months.
- Patient had major surgery within the last 14 days.
- Patient has known history of intracranial hemorrhage.
- Patient has sustained systolic blood pressure > 185 mmHg.
- Patient has sustained diastolic blood pressure > 110 mmHg.
- Aggressive treatment is necessary to lower the patient's blood pressure.
- Patient has symptoms suggestive of subarachnoid hemorrhage.
- Patient has had gastrointestinal or urinary tract hemorrhage within the last 21 days.
- Patient has had arterial puncture at noncompressible site within the last 7 days.
- Patient has received heparin with the last 48 hours and has elevated PTT.
- Patient's prothrombin time (PT) is > 15 seconds.
- Patient's platelet count is < 100,000 μL.
- Patient's serum glucose is < 50 mg/dL or > 400 mg/dL.
tPA Relative Contraindications
If either of the following statements is true, use tPA with caution:
- Patient has a large stroke with NIH Stroke Scale score > 22.
- Patient's CT shows evidence of large middle cerebral artery (MCA) territory infarction (sulcal effacement or blurring of gray-white junction in greater than one-third of MCA territory).
Adapted from the clinical policy of the American College of Emergency Physicians.
Therapeutic Intervention
Intravenous Recombinant Tissue Plasminogen Activator (tPA). While the inclusion and exclusion criteria are reviewed, the family asks about the possibility of "clot-busting" drugs. The patient's family members are informed of the risks involved and believe the patient would want thrombolytic therapy.
Thrombolysis with tPA is the indicated treatment for appropriate patients with acute stroke. While there is substantial evidence that it improves outcomes, there are potential serious side effects, most importantly the risk of intracranial bleeding.
The early recommendation for tPA utilization was for patients whose last time seen normal was less than 3 hours ago. However, recent studies suggest that the time window can be extended from 3 to 4.5 hours in some patients. The observational data from the SITS-ISTR study (Safe Implementation of Thrombolysis in Stroke-International Stroke Treatment Registry)8 suggest there are no differences in outcomes for patients who received tPA within 3 hours compared to those who received the drug between 3-4.5 hours after the onset of stroke symptoms.
Other studies9,10 have supported the extension of the therapeutic window from 3 to 4.5 hours. The ECASS 3 study10 (European Cooperative Acute Stroke Study 3) was a multicenter, placebo-controlled RCT. It enrolled a total of 821 patients and utilized the current tPA regimen guidelines with the following exclusions: NIH stroke scale > 25, current use of oral anticoagulants, age greater than 80 years, and history of stroke and diabetes. The study found that disability at 90 days was significantly better in the treatment group, although the incidence of symptomatic intracranial bleed was higher. The mortality in both groups (treatment and placebo) were similar. Based on the results of the SITS-ISTR and ECASS 3 study, some advocate extending the window for tPA to 4.5 hours, although this is not FDA approved.
The recommended dosing guideline for tPA in acute ischemic stroke is 0.9 mg/kg (maximum of 90 mg) single dose, with an initial 10% of the calculated dose (not to exceed 90 mg) given as a bolus over 1 minute and the rest infused over one hour.10
Thrombolysis should be considered for patients that meet the criteria in those hospitals that have systems in place for its safe administration. Despite the evidence supporting thrombolytics in both the 3 and extended 4.5 hour window, there is still some resistance to giving the drug. Clearly there needs to be institutional support available for the emergency physician, particularly in smaller, more rural hospitals. Where there is support, use of thromboly-tics has become common practice.
In the case example, the initial blood pressure of the patient was 224/121. Reviewing the inclusion and exclusion criteria, this is a contraindication to initiation of intravenous thrombolytics until the blood pressure is controlled.
Blood Pressure Management in Acute Ischemic Stroke
For patients receiving tPA, control of the blood pressure is very important. Studies have shown that markedly increased blood pressure may increase the risk for intracerebral hemorrhage.11 The current treatment guidelines stipulate treatment of blood pressure to achieve a systolic of 185 mm Hg or lower and a diastolic of 110 mm Hg or lower before tPA is administered.
The 2007 American Heart Association (AHA) guideline suggests intravenous labetalol 10-20 mg (bolus) as a first-line drug for rapid control of blood pressure. This could be followed by repeated boluses (up to 300 mg) or the initiation of labetalol drip at 2-8 mg/hour until the desired blood pressure is accomplished. The 2010 AHA guidelines list intravenous nicardipine and labetalol as the first-line agents for rapid blood pressure control. When using nicardipine, start the infusion at 5 mg/hour and titrate up 2.5 mg/hour every 5-15 minutes as needed to a maximum of 15 mg/hour. When the desired blood pressure is reached, the dose can be reduced by 3 mg/hour.12 The blood pressure should be monitored every 15 minutes during the treatment, and then for another 2 hours. For the next 6 hours, it should be monitored every 30 minutes, and every one hour for the next 16 hours.12
In the case example, the patient's blood pressure was controlled with intravenous nicardipine, as per guidelines, to achieve a systolic blood pressure of less than 185 mm Hg and a diastolic BP less than 110 mm Hg. Aggressive treatment of blood pressure also comes at a cost and could be detrimental to the patient. The ischemic penumbra is defined as the area of the brain tissue that is perfused but at the threshold of functional impairment and morphologic integrity that has the capacity to recover if perfusion is promptly restored.13 Aggressive control of blood pressure can compromise the flow to the ischemic penumbra by creating an environment of relative hypotension or a state of hypoperfusion. This eventually leads to the death of potentially salvageable brain tissue.14
In the case example, while preparing the tPA, the remainder of the patient's laboratory study returned and revealed a blood glucose of 265 mg/dL in a nondiabetic patient. The patient was started on insulin drip as per institutional protocol, with tight monitoring of the serum glucose.
Glycemic Control. Hyperglycemia in stroke may be due to stress response with catecholamine release and it has been shown to be an independent predictor of poor outcome by inducing secondary brain injury.15 Hyperglycemia has been shown to be associated with worse prognosis in the patients receiving thrombolytic therapy.16 A study by Alvarez-Sabin et al17 evaluated the impact of admission hyperglycemia on stroke outcome after thrombolysis demonstrated that the effect of admission hyperglycemia on stroke outcome varied depending on the time to tPA reperfusion. The authors stated that the detrimental effect of acute hyperglycemia is higher after early reperfusion than after delayed or no reperfusion, and suggested that ultra early glycemic control before reperfusion may improve the efficacy of thrombolytic therapy.
While it is appropriate to control hyperglycemia in stroke patients, the ideal glucose level is unknown. The Glycemia in Acute Stroke (GLIAS) study18 evaluated the prognostic value of capillary glucose level in acute stroke. The study demonstrated that hyperglycemia ≥ 155 mg/dL at any time within the first 48 hours of stroke onset, and not only the isolated value of admission glycemia is associated with poor outcome independently of stroke severity, infarct volume, diabetes, or age. A post-hoc analysis of the GLIAS study19 evaluated persistent hyperglycemia ≥ 155 mg/dL in acute stroke patients. The study demonstrated that almost 40% of patients maintained levels > 155 mg/dL despite corrective treatment, and it was associated with poorer outcome and increased mortality. According to ASA guidelines, a reasonable approach would be to initiate treatment among acute stroke patients with blood glucose > 200 mg/dL.
The available body of evidence confirms the deleterious effect of hyperglycemia in acute stroke patients and also demonstrates the threshold that correlates well with an increased mortality (greater than or equal to 155 mg/dL). The goal is to attain a target blood glucose of at least < 155 mg/dL using insulin therapy with caution to avoid over shooting and inducing a state of hypoglycemia. Baker et al20 recommended that patients receiving thrombolytic therapy should be started on an established and standardized intravenous insulin protocol to improve glucose control for at least the first 24 to 48 hours of hospitalization.
In the case example, the patient received intravenous tPA with adequate monitoring of the blood pressure while the infusion was ongoing in the ED. Mid-way through the infusion, the patient indicated he was having a headache. At this time, the infusion was stopped and an emergent CT scan of the head was obtained. The scan revealed no hemorrhage, and the infusion was resumed. The patient was subsequently admitted to the neuro-critical stroke care unit.
The management of acute ischemic stroke hinges on early identification of the stroke patient, utilizing a validated screening tool, understanding the time-line for evaluation and reperfusion, and adequately screening using screening scales and the inclusion and exclusion criteria for thrombolytic intervention. In addition, following the guideline for the blood pressure control and post-tPA care relating to BP while in the emergency department is very important. Stroke-capable centers offer the benefit of a multidisciplinary approach to stroke care. If the patient presents to an ED without thrombolytic capability, consideration should be given to rapid transfer to a stroke capable center.
Situations might arise when, despite the best efforts, the last time "seen normal" cannot be delineated. In this event, a non-contrast CT scan, CT angiogram of the head and neck, and CT perfusion may be utilized to aid in the identification of patients who are candidates for acute intervention. (See Figures 2 and 3.) The CT scan perfusion enables the identification of ischemic penumbra that may be a criterion for intra-arterial thrombolytic in academic centers that have the availability of interventional neuroradiologist or neurosurgeons on staff. In the case that these resources are not available, transfer of appropriate patients should be considered.
Radiographic Evaluation of Stroke Patients
Imaging of a stroke patient should occur promptly to avoid delays in door-to-reperfusion. The clinician should understand the implications of knowing the last time the patient was seen normal and the type of imaging needed to avoid potential delays in therapeutic intervention.
The AHA scientific statements21 recommend that the imaging study decided upon should definitely address the following: the presence or absence of hemorrhage; the presence of intravascular thrombus that can be treated with thrombolytic or thrombectomy; the presence and size of a core that is irreversibly infarcted; and the presence of hypoperfused tissue at risk for infarction if reperfusion is not promptly restored.
The initial radiographic study to evaluate an acute stroke patient is a nonenhanced CT scan (NECT) of the brain. Although the average sensitivity of CT in detecting early ischemia is 66%,22 it is the preferred study for initial triaging for patients being considered for thrombolytic therapy. The NECT helps detect the presence or absence of hemorrhage or cerebral mass, or alternative diagnosis of the patients' symptoms. NECT creates a good contrast between blood and cerebrospinal fluid. In addition, it is widely available with minimal contraindications to its utility.
When interpreting the NECT, the emergency physician should be conversant with some terminologies representing the early signs of stroke on the CT scan. These include the insular ribbon sign (loss of gray and white matter interface in the proximal region of the MCA distribution), obscuration of lentiform nucleus (loss of the outline of the lentiform nucleus of the basal ganglia), and the hyperdense MCA sign (presence of acute thrombus in the MCA). (See Figure 1.) In addition to the early ischemic signs, the clinician should understand the signs of contraindication to thrombolytics in the absence of hemorrhage.
Figure 4: PCA Infarct
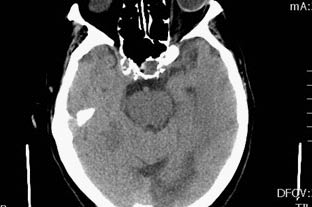
This image shows an unenhanced CT scan of a patient with a PCA infarct. Because of the hypo-attenuation in the posterior circulation, and the risk for hemorrhagic transformation, the patient was not a tPA candidate.
One of these signs is obvious hypo-attenuation on the NECT. This is indicative of irreversible infarct and carries a significant risk for hemorrhagic transformation. (See Figure 4.) According to the ECASS I and II study, the involvement of more than one third of the MCA territory on NECT is an exclusion from thrombolytic therapy due to the risk of hemorrhagic transformation.23,24
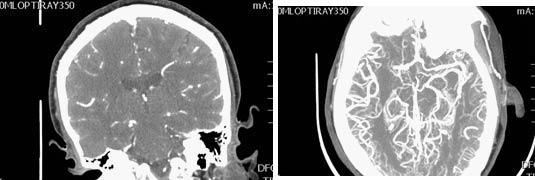
CT angiography (CTA) (from aorta to top of head) can easily be performed immediately after NECT, thus reducing delays. (See Figure 2.) The 3-D reconstruction of the images and blood vessels allows for the visualization of areas of stenosis, aneurysms, bleed, and other vascular abnormalities such as AVM. When imaging is obtained from the aorta to the top of the head, it will help detect the presence or absence of aortic or carotid dissection as the source of presenting symptoms. At the same time, the source images obtained from the CTA can help identify qualitative information regarding the cerebral blood volume (CBV) map that establishes the core of infarction and the tissue at risk for infarction.
Figure 2: CT Angiogram of a Patient Showing Flow Pattern of the Right MCA Branch
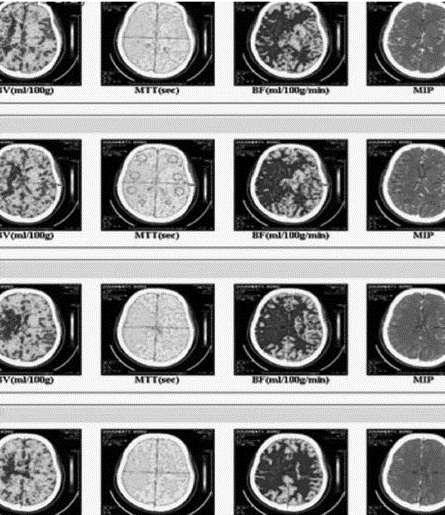
CT perfusion (CTP) is used to examine the blood flow and identify potentially salvageable hypoperfused region or the ischemic penumbra, and areas that are completely infarcted. (See Figure 3.) Parameters such as cerebral blood flow (CBF relates to the volume of blood flowing per brain mass in a minute), cerebral blood volume (CBV relates to volume per unit of brain), the mean transit time (MTT), and the time to peak flow (time difference between arterial inflow and venous outflow) can all be obtained. CTP is usually performed immediately after the CTA to avoid double contrast injection.
Figure 3: CT Perfusion of the Patient with Right MCA Stroke Demonstrating the Blood Volume, Mean Transit Time, and Blood Flow
Several factors often affect the type of imaging obtained. These may include cost of the study, institutional capability (if not a designated comprehensive stroke center), last time seen normal, the patient hemodynamic stability, and the impact on door to reperfusion. These studies could be performed simultaneously in less than 20 minutes. Viewing the NECT simultaneously as it is performed allows one to proceed immediately with the CTA and CTP.
Occasionally, the CTA will identify carotid dissection extending to the thoracic aorta, thus precluding the initiation of thrombolytic therapy with a potential deadly consequence. The turn-around time for these imaging studies should not delay therapeutic interventions. It is important that the emergency physician have baseline foundation on CT imaging interpretations and the ability to identify the presence or absence of hemorrhage, mass, or dissection. This knowledge will allow therapeutic intervention to proceed while awaiting the official radiologist interpretation.
The diffusion-weighted magnetic resonance imaging (MR-DWI) is the most sensitive and specific technique for identifying acute infarction within minutes of occurrence. The combination of DWI with MR–perfusion will enable the identification of viable from nonviable hypoperfused brain tissues. Despite the sensitivity and specificity of MRI, obtaining the study could be time consuming and can also create a logistical nightmare, especially when time is of essence. The role or MRI in acute stroke triage is still debatable, although a study by Schellinger et al25 demonstrated that the diagnostic evaluation consisting of the DWI, MRP, and intracranial MRA could all be performed within 10 minutes.
Protocols for MRI utilization vary from different institutions. The typical MRI stroke protocol consist of T2/FLAIR, T2 weighted, diffusion (DWI), and perfusion (PWI). The FLAIR image is very sensitive in identifying acute thrombosis and subarachnoid hemorrhage, while the T2 weighted image is very sensitive in detecting loss of arterial flow signal in the presence of hyperacute stroke within minutes. According to Wintermark et al,26 these studies could be performed in less than 30 minutes.
The AHA recommendation for imaging for patients within 3 hours of symptoms is either NECT or MRI before intravenous tPA administration to exclude ICH and to determine the presence or absence of hypodensity (CT) or hyperintensity (MRI) of ischemia. Also, the presence of hypointensity on CT scan that involves more than a third of the MCA territory is a contraindication to thrombolytic therapy (class I, level of evidence A).
Summary
Neurologic emergency from acute stroke carry a significant morbidity and potential mortality. If approached in a systematic faction, emergency physicians can improve the chances for a good neurologic outcome from this critical illness. Emergency physicians need to be aware of the most current recommendations and guidelines that would lead to a favorable patient outcome. With the many advances in basic and clinical research for acute stroke, there will be more clarity to some of these current controversies regarding the best care for patients with stroke.
References
1. Miniño AM, Murphy SL, Xu J, et al. National Vital Statistics Reports; vol 59 no 10. Hyattsville, MD: National Center for Health Statistics; 2011.
2. Roger VL, Go AS, Lloyd-Jones DM, et al. Heart disease and stroke statistics 2011 update: A report from the American Heart Association. Circulation 2011;123:e18-e209. Epub 2010 Dec 15.
3. Centers for Disease Control and Prevention. Prevalence and most common causes of disability among adults United States, 2005. MMWR 2009;58:421–426.
4. Lloyd-Jones D, Adams R, Carnethon M, et al. Heart disease and stroke statistics 2009 update. A report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation 2009;119:480-486.
5. National Stroke Association. Stroke/Brain Attack Reporter's Handbook. Englewood, CO: National Stroke Association; 1995.
6. Moulin T, Tatu L, Vuillier F, et al. Role of a stroke data bank in evaluating cerebral infarction subtypes: Patterns and outcome of 1,776 consecutive patients from the Besançon stroke registry. Cerebrovasc Dis 2000;10:261-271.
7. Garcia JH, et al. In: Barnett HJM, et al, eds. Stroke Pathophysiology, Diagnosis, and Management. New York: Churchill Livingstone; 1992:125.
8. Wahlgren N, Ahmed N, Davalos A, et al. Thrombolysis with alteplase 3-4.5 h after acute ischaemic stroke (SITS-ISTR): An observational study. Lancet 2008;372:1303-1309.
9. del Zoppo GJ, Saver JL, Jauch EC, et al. Expansion of the time window for treatment of acute ischemic stroke with intravenous tissue plasminogen activator. Stroke 2009;
10. Hacke W, Kaste M, Bluhmki E, et al. Thrombolysis with alteplase 3 to 4.5 hours after acute ischemic stroke. N Engl J Med 2008;359:1317-1329.
11. Larrue V, von Kummer RR, Müller A, et al. Risk factors for severe hemorrhagic transformation in ischemic stroke patients treated with recombinant tissue plasminogen activator: A secondary analysis of the European-Australasian Acute Stroke Study (ECASS II). Stroke 2001;32: 438-441.
12. Jauch EC, Cucchiara B, Adeoye O, et al. Part 11: Adult stroke. 2010 American Heart Association guidelines for cardiopulmonary resuscitation and emergency cardiovascular care. Circulation 2010;122(suppl 3):S818-S828.
13. Paciaroni M, Caso V, Agnelli G. The concept of ischemic penumbra in stroke and therapeutic opportunities. Eur Neurol 2009;61:321-330.
14. Owen B. Blood pressure control in acute cerebrovascular disease. J Clin Hypertens (Greenwich) 2011;13:205-211.
15. Garg R, Chaudhuri A, Munschauer F, et al. Hyperglycemia, insulin, and acute ischemic stroke. A mechanistic justification for a trial of insulin infusion therapy. Stroke 2006;37:267-273.
16. Ribo M, Molina CA, Delgado P, et al. Hyperglycemia during ischemia rapidly accelerate brain damage in stroke patients treated with tPA. J Cereb Blood Flow Metab 2007; Feb. 14;Epub ahead of print.
17. Alvarez-Sabin J, Molina CA, Ribo M, et al. Impact of admission hyperglycemia on stroke outcome after thrombolysis: Risk stratification in relation to time to reperfusion. Stroke 2004;35:2493-2498.
18. Fuentes B, Castillo J, San Jose B, et al. The prognostic value of capillary glucose levels in acute stroke: The Glycemia in Acute Stroke (GLIAS) study. Stroke 2009;40:562-568.
19. Fuentes B, Ortega-Casarrubios MA, SanJose B, et al. Persistent hyperglycemia > 155 mg/dL in acute ischemic stroke patients: How well are we correcting it? Implications for outcome. Stroke 2010;41:2362-2365.
20. Baker L, Juneja R, Bruno A. Management of hyperglycemia in acute stroke. Curr Treat Options Neurol 2011;13:616-628.
21. Latchaw RE, Albert MJ, Lev MH, et al. AHA scientific statement: Recommendations for imaging in acute stroke. Stroke 2009;40:3646-3678.
22. De La Paz RL, Wippold FJ, Cornelius RS, et al. American College of Radiology ACR Appropriateness Criteria: Cerebrovascular disease. Available at: http://www.acr.org/ac. Accessed January 20, 2012.
23. Hacke W, Kaste M, Fieschi C, et al. Intravenous thrombolysis with recombinant tissue plasminogen activator for acute hemispheric stroke. The European Cooperative Acute Stroke Study (ECASS). JAMA 1995;274:1017-1025.
24. Larrue V, von Kummar RR, Muller A, et al. Risk factors for severe hemorrhagic transformationin ischemic stroke patients treated with tissue plasminogen activator: A secondary analysis of the European Cooperative Acute Stroke Study (ECASS II). Stroke 2001;32:438.
25. Schellinger PD, Jansen O, Fiebach JB, et al. Feasibility and practicality of MR imaging of stroke in the management of hyperacute cerebral ischemia. AJNR Am J Neuroradiol 2000;21:1184-1189.
26. Wintermark M, Albers GW, Alexandrov AV, et al. Acute stroke imaging research roadmap. AJNR Am J Neuroradiol 2008;29:e23-30.
Stroke remains a leading cause of death, but the disability associated with a stroke can be devastating and costly. In past decades, little could be done to reduce the morbidity and mortality of stroke. But over the past decade, use of thrombolytics by specialized stroke centers has reduced the morbidity of survivors. However, the reduction of morbidity comes at a cost of an increase in intracerebral hemorrhage, often associated with death.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
