Aortic Dissection
Aortic Dissection
Authors:
Karis Tekwani, MD, Department of Emergency Medicine, Advocate Christ Medical Center, Oak Lawn, IL.
Paarul Chandra, MD, Department of Emergency Medicine, Advocate Christ Medical Center, Oak Lawn, IL.
Peer Reviewer:
Benjamin Ostrovsky, MD, Senior Clinical Instructor, Department of Emergency Medicine, University of Rochester, Rochester, NY.
Acute aortic dissection is a rare but life-threatening illness in which prompt diagnosis and treatment are critical for the chance of survival.
Introduction
Acute aortic dissection is defined as the rapid development of a false, blood-filled lumen within the tunica media of the aorta. The classic presentation is of an elderly male with a history of chronic hypertension, presenting with abrupt onset of tearing chest pain radiating to the back. Many times, the presenting symptoms can mimic more common conditions, and aortic dissection may not be detected initially in up to 38% of cases.1 Without treatment, nearly 25% of patients die within the first 24 hours and 75% of patients die within 2 weeks.2 In contrast, prompt diagnosis and definitive therapy may increase survival rates to 90% at 1 year.3,4
Incidence
The estimated incidence of aortic dissection is between 2.9 and 4.3 cases per 100,000 persons per year.4-7 A population-based study from Sweden, where autopsies are mandatory for all unexpected deaths, found that between 1987 and 2002, 4425 cases were of aortic dissection were identified with a population of 8.7 million, which comes out to 3.4 per 100,000 people per year.8 The incidence of aortic dissection also appears to be increasing over time, which can likely be attributed to the improvements in diagnostic imaging. In order to assess etiological factors as well as modes of presentation, management, treatment, and hospital outcomes of patients with acute aortic dissection around the world, the International Registry of Aortic Dissection (IRAD)3, a consortium of research centers throughout the world, was established in 1996. The IRAD contains about 2,000 new cases each year from the United States.3
Anatomy
The majority of dissections occur in the thoracic aorta, which is comprised of the ascending aorta, aortic arch, and the descending thoracic aorta. The ascending aorta is made up of the aortic root, which includes the aortic annulus and extends to the sinotubular junction, and the tubular structure, which extends from the sinotubular junction to the innominate (brachiocephalic) artery. The aortic arch (transverse aorta) is a short segment from which the brachiocephalic artery, the left carotid artery, and the left subclavian artery arise. The descending aorta starts immediately distal to the left subclavian artery and continues into the diaphragm, ending at the first branch in the abdominal aorta, the celiac artery.9
The aortic wall is comprised of three distinct layers: the intima, the media, and the adventia. The intima is the innermost layer of the wall and is comprised of endothelial cells and is supported by an internal elastic lamina. The media is composed of smooth muscle cells and is surrounded by elastin, collagen, and proteoglycans. This layer is the strongest of all three and serves as the major structural and elastic properties of the artery. The adventitia is the outermost layer and consists primarily of collagen and vasa vasorum (blood vessels).10
Pathogenesis
The aorta is exposed to a significant amount of pulsatile pressure and sheer stress, making it particularly prone to injury and disease from mechanical force. With each cardiac cycle, blood is ejected into the aorta, and the elastic properties of the aorta allow it to expand and recoil in order to propel forward circulation. With normal aging, the aorta loses some of its elasticity and becomes more vulnerable to these hemodynamic forces.1 Aortic dissection is believed to be caused by degeneration of the aortic media. It is characterized by a loss of smooth muscle cells and elastic tissue and leads to scarring, fibrosis, and hyalin-like changes.11 Sustained hypertension further accelerates degeneration of the aortic media.11
In classic aortic dissection, a small tear forms in the aortic intima, allowing the pulsatile blood to enter between the inner and outer layers of the diseased aortic media. This newly formed blood-filled space creates a false lumen and is separated from the true lumen by the dissecting membrane. It is thought that the intimal tear typically occurs in areas subject to the greatest change in blood pressure or rate of ventricular or aortic pressure change (dP/dT).12 The most common site of the intimal tear is the left posterolateral portion of the aorta.13 The intraluminal pressure serves as a force for the dissection to propagate in an anterograde or retrograde fashion. Due to pressure differences between the true and false lumens, the false lumen may compress or obstruct the true lumen.13 The dissection can reenter the intima or dissect through the adventia into the pericardia, pleural, or peritoneal space. Propagation of the dissection can involve side branches and cause malperfusion syndromes.14-16
An alternative theory of the pathogenesis of aortic dissection includes the formation of an intramural hematoma (IMH) or a penetrating aortic ulcer (PAU). There is some debate whether IMH and PAU are precursor lesions to an acute aortic dissection or distinct entities.
Intramural Hematoma
IMH originates from rupture of the vasa vasorum into the aortic wall, resulting in hemorrhage or hematoma into the media.14,15,17,18 It is characterized by an absence of a detectable intimal tear. They are more commonly located in the descending aorta and are usually associated with hypertension.19 An IMH is found in 5-20% of patients with signs suggesting an aortic dissection.14 IMH can regress in approximately 10% of cases, create a secondary tear causing a classic aortic dissection in 28-47% of cases, or carry a risk of rupture in 20-45% of cases.20,21
Penetrating Aortic Ulcer
PAU is a focal atherosclerotic aortic plaque that ulcerates through the internal elastic lamina into the media,22 with or without associated IMH or pseudoaneurysm. The incidence of PAU in aortic dissection ranges from 2.3% to 11%.21-23 They tend to occur in older men, are associated with extensive atherosclerotic disease, and are commonly located in the descending aorta.22 They can progress to form an aneurysm in 30% of cases24 and may rupture into the media forming an IMH, or result in a classic aortic dissection.
Risk Factors
There are many risk factors that predispose patients to an aortic dissection. (See Table 1.) The majority of acquired aortic dissections are seen in the elderly population, with the peak incidence in the sixth and seventh decades of life.1,13 Younger patients who present with an aortic dissection tend to have a congenital disease such as connective tissue disorders, Ehlers-Danlos or Marfan syndrome, congenital heart disease, or giant cell arteritis. Males are more commonly affected, almost two to five times as often as females,13 but women have worse outcomes due to atypical symptoms and delayed diagnosis.25,26
Table 1: Risk Factors
Acquired
Congenital
|
Acquired Causes
Hypertension is the most common risk factor associated with aortic dissection; approximately 75% of patients with aortic dissection have a history of hypertension.9,15,27
Aortic aneurysm is a well-established risk factor for the development of an aortic dissection. The annual rate of acute aortic dissection increases as the aortic diameter increases. The calculated yearly rate of dissection is 1.5% for aortic diameters > 4 cm, 2.5% for diameters > 5 cm, and 3.7% for diameters > 6 cm.28
Cocaine use is a predisposing factor for aortic dissection. It induces an acute catecholamine response, which leads to a sudden and severe elevation of blood pressure, causing an increased sheer stress in the aorta, resulting in an intimal tear.29
There has been some debate about whether pregnancy increases the risk of aortic dissection. The belief now is that pregnant women with Marfan syndrome are at a greater risk of developing an aortic dissection, particularly if they have pre-existing aortic dilatation.30 In patients without connective tissue disease and with an aortic root size of less than 4 cm, pregnancy does not appear to be an independent risk factor.11
In IRAD, 5% of all cases of aortic dissection are iatrogenic, with the most common procedures resulting in dissection being cardiac catheterization and cardiac surgery.3 High-intensity weight lifting or other strenuous exercise may also cause an acute ascending aortic dissection, but this is rare.31
Congenital Causes
Connective tissue disorders such as Marfan or Ehlers-Danlos syndrome also predispose patients to aortic dissection by resulting in cystic medial degeneration. Marfan syndrome, an inherited disorder with autosomal dominant transmission and variable penetration, has an estimated incidence of 1 in 5000.32 It is the most common cause of aortic dissection in patients younger than 40 years and is characterized by cardiovascular, musculoskeletal, and ocular abnormalities. Ehlers-Danlos syndrome is another inherited connective tissue disorder characterized by joint hypermobility, skin hyperelasticity, and tissue fragility. Aortic dissection is more commonly seen in autosomal dominant type IV Ehlers-Danlos.29
Congenital cardiovascular conditions, such as bicuspid aortic valve and aortic coarctation, have an increased incidence of aortic dissection. Bicuspid aortic valve is found in 2-9% of patients with type A aortic dissection and 3% of patients with type B aortic dissection.33,34 While only 1% of individuals affected by aortic coarctation develop an aortic dissection,35 the incidence increases to 8% after the coarctation has been repaired.36
Classification
Aortic dissections are classified based on duration from onset of symptoms and by anatomical location of the injury. If the onset of symptoms at the time of diagnosis is less than 14 days, the dissection is defined as acute, and if the onset of symptoms is greater than two weeks at the time of diagnosis, the dissection is defined as chronic.13
Approximately 60% of aortic dissections occur in the ascending aorta, 10% in the aortic arch, and 30% in the descending aorta.37 The most common sites for an intimal tear are above the aortic valve and just distal to the subclavian artery. The two classification schemes that are most widely recognized are the Debakey system and the Stanford system. (See Figure 1.) The Debakey approach categorizes the dissection into three types based on origin of intimal tear and extent of dissection.13,15 Type I originates in the ascending aorta and typically involves both the aortic arch and the descending aorta. Type II originates in the ascending aorta and is confined there. In type III, the dissection originates in the descending aorta and can propagate distally. Type III dissection is further classified into type IIIa, which refers to dissections that originate in the descending aorta but stop above the diaphragm, or type IIIb, which originate in the descending aorta and extend distally below the diaphragm.13
Figure 1: Classification
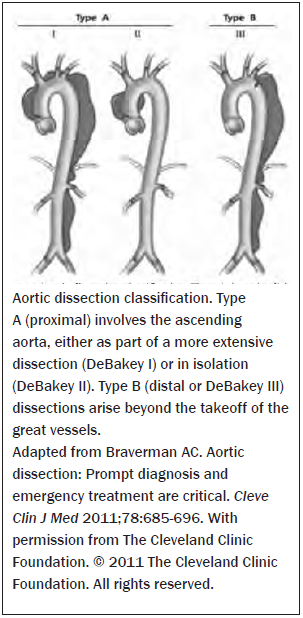
The Stanford Classification system38 simplifies the aortic dissection according to the involvement of the ascending aorta.13 Stanford type A dissections involve the ascending aorta and may extend into the aortic arch and the descending aorta. Stanford type B dissections are defined as all dissections that do not affect the ascending aorta.15 One of the advantages of the Stanford classification system is that it classifies patients into those that require emergent surgery (type A) or those that can be managed without emergent surgery (type B).13 According to IRAD, 62% of dissections are type A and 38% of dissections are type B.3 For the purposes of this paper, the Stanford classification system will be used.
Clinical Presentation
History.The most frequently reported symptom is acute chest or back pain (> 90%).11 The pain occurs abruptly, is most severe at onset, and is more likely to be described as sharp or stabbing than tearing or ripping.11,15 According to IRAD, 84.8% of patients reported pain with abrupt onset and 90.6% of patients reported severe pain as the presenting symptom.3 Clinical presentations are summarized in Table 2.
Table 2: Clinical Presentation
History
Physical Exam
|
The location of pain reflects the site of initial intimal disruption. Anterior chest pain is more indicative of involvement of the ascending aorta, pain that radiates to the neck, throat, or jaw is associated with dissection of the aortic arch, and pain in the intrascapular, back, or abdomen is more likely to involve the descending aorta.3 Propagation of the dissection can present as migration of pain but only occurs in 17% of cases.11
In addition, patients can have associated systemic symptoms such as diaphoresis, nausea, vomiting, or lightheadedness. They can appear anxious and express a sense of doom.39 Painless aortic dissections are rare, associated with older patients, chronic in nature, and more often occur in type A dissections.1
Physical Exam. Physical exam findings in acute aortic dissection are dependent upon the location and extent of the dissection. While a history of hypertension is common, not all patients are hypertensive on initial presentation.3 The IRAD results indicate that hypertension is found in 70% of patients with a type B dissection, compared to 36% percent with a type A dissection.3 In patients with hypotension, suspect either pericardial tamponade from progression of the dissection into the pericardium or hypovolemia from rupture through the adventitia.11 Pseudohypotension, falsely low or absent blood pressure measurement in the arms, may also occur with aortic dissection. This finding is due to acute compromise of arterial flow to the subclavian artery.11
Pulse deficit, defined as a weak or absent carotid, brachial, or femoral pulse, may be present in 10-30% of patients with an acute aortic dissection.3,39,40 It is caused by impaired or absent blood flow to peripheral vessels and results from two mechanisms: an intimal flap may cover the true lumen of the vessel or the dissecting hematoma may compress an adjacent true lumen.11
Aortic regurgitation is present in about 32% of patients and is more common in type A dissections (44%) than type B dissections (12%).3 The diastolic decrescendo murmur is best auscultated over the right sternal border in patients with an aortic dissection, as compared to the left sternal border in patients with primary aortic valve disease.1
Neurological deficits, including stroke, syncope, or paralysis, can result from extension of the dissection into the carotid arteries and may be seen in up to 40% of patients with type A dissection.41 Neurologic symptoms may dominate the clinical picture and lead to a delay in the diagnosis of dissection.39 Stroke can occur in up to 10% of patients.1 Syncope is more commonly seen in type A dissections (13%) than type B dissections (4%) and is usually associated with poorer outcomes.3 Paraplegia can also occur if there is involvement of the arteries that supply the spinal cord.10
Vascular insufficiency can occur in any branch vessel leading to a variety of characteristic symptoms resulting from ischemia. It is estimated that approximately 30% of patients with acute aortic dissections develop ischemic complications.1 Extension into the abdominal aorta and involvement of the renal artery or the iliac artery can cause renal ischemia/infarction or acute lower extremity ischemia, respectively. Mesenteric ischemia or infarction occurs in about 5% of dissections.39
Acute myocardial infarction presents in up to 5% of patients with a type A dissection, resulting in coronary occlusion.27 The right coronary artery is most commonly involved, leading to an acute inferior myocardial infarction.39
Diagnosis
Electrocardiography. The electrocardiogram (ECG) is a part of the initial diagnostic evaluation of the patient with chest pain but is of limited use in the diagnosis of aortic dissection. Up to 31% of ECGs in patients with acute aortic dissection are normal.11
The more challenging diagnostic dilemma arises in the patient with acute aortic dissection with proximal dissection involving the right coronary artery. The ECG in this case may show an inferior wall myocardial infarction, and the signs and symptoms such as pain, diaphoresis, and hypotension can be difficult to differentiate from acute myocardial infarction.11 Routine interventions for inferior wall myocardial infarction, including anticoagulants and antiplatelet agents as well as thrombolytics could be fatal in a patient with an acute aortic dissection.
Chest Radiography. Chest radiography is a useful study in patients with suspected acute aortic dissection. It is fast, readily available, and is abnormal in 80-90% of patients with acute aortic dissection.11 Unfortunately, the findings are often non-specific and rarely diagnostic.1,11 Up to 12% of patients with acute aortic dissection will have a normal chest radiograph,11 and findings may be so subtle that they cannot be identified on a portable chest radiograph.42
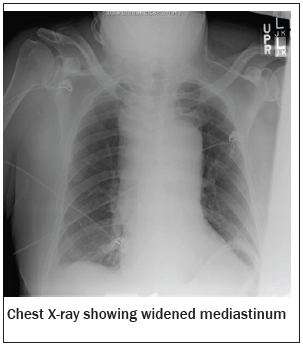
Mediastinal widening (> 8 cm) is the most commonly occurring finding on chest radiograph in patients with acute aortic dissection and is seen in approximately 63% of patients with type A dissections and 56% of patients with type B dissections.3 (See Figure 2.) Other less commonly observed findings include a double shadow of the aortic wall (indicating true and false lumens), disparity in the size of the ascending and descending aorta, a localized bulge along a normally smooth aortic contour, obliteration of the aortic knob, left pleural effusion, or displacement of the trachea or nasogastric tube to the right.1,11,43 The "calcium sign" is a rarely seen radiographic sign in aortic dissection. Normally, if intimal calcification is seen, it is snug against the outer border of the aorta; however, with dissection of the media, this calcification can become separated from the outer wall by more than 5 mm.11
Figure 2: Chest X-ray
Confirmatory Imaging. The diagnosis of acute aortic dissection will almost always require further imaging studies.
Aortography. Historically, retrograde aortography was the standard method of evaluating aortic dissection and was considered the "gold standard" for the diagnosis; however, it has been superseded by other less invasive imaging modalities such as CTA, MRI, and TEE.14 Aortography has a high specificity of 94%, but the sensitivity has been reported to be as low as 88%, which is lower than other imaging modalities.1 Benefits to aortography include the ability to identify the site of origin of the original dissection, branch artery involvement, aortic regurgitation, and coronary involvement. Disadvantages include the invasive nature and length of time to complete the study, administration of contrast dye, and failure to diagnose IMH.1 Aortography may be recommended in patients in whom visceral malperfusion is suspected or percutaneous interventions are being considered.16
Echocardiography. Transthoracic echocardiography (TTE), although it can be done at the patient's bedside and is non-invasive, lacks sufficient sensitivity (77%-80%) to be used as an isolated imaging modality.1,39 TTE suffers from the inability to fully visualize the aortic arch or much of the descending aorta.11 TTE can identify pericardial effusion and tamponade as well as aortic regurgitation, and may be performed at the bedside while awaiting more sensitive imaging tests.11
Transesophageal echocardiography (TEE) is an excellent imaging modality in the diagnosis of aortic dissection, with a sensitivity of 98% and specificity of 95%.44 TEE can detect pericardial effusion and aortic regurgitation similar to TTE and can image the majority of the thoracic aorta. Historically TEE was not as accurate at visualizing the aortic arch and distal ascending aorta; however, advances in probe technology have aided in the detection of this "blind spot."11,39 It can be performed at the bedside with minimal sedation, it does not require contrast agents, and it is the procedure of choice in the unstable patient.11 Diagnosis is confirmed by identifying a mobile intimal flap within the aortic lumen that separates the true and false lumen.1 Disadvantages of TEE are that it is operator-dependent, does not provide any information below the diaphragm, and the ability of obtaining a TEE can be institution-dependent.
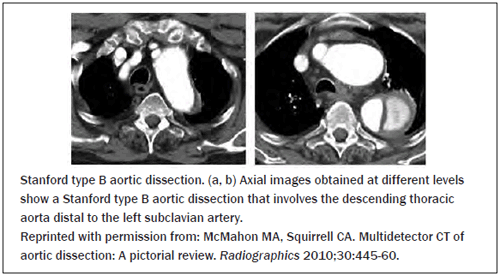
CT Angiography. CTA is a reliable imaging modality for diagnosing aortic dissection. It has a sensitivity of 95%-100% and a specificity of 98%.14,44 It is the diagnostic test of choice in most institutions due to the ease of obtaining it, the fast turn-around time, and the ability to image the full aorta. In order to eliminate pulsation artifacts, it is best performed with electrocardiographic gating or multidetector scanning.39,45,46 Findings on CTA that suggest aortic dissection include dilation of the aorta, identification of an intimal flap, and demonstration of true and false lumina.11,45 (See Figures 3 and 4.) CTA can also visualize pericardial effusion, aortic rupture, and branch vessel involvement.39 Additionally, it can aid surgeons in delineating the extent of dissection and in planning operative management. The CTA protocol for aortic dissection should include unenhanced scans that provide information about the presence of calcifications as well as the ability to detect an IMH.1,14 The main disadvantages to CTA are the administration of contrast agents as well as the need to leave the ED for imaging.15
Figure 3: Stanford Type A Aortic Dissection
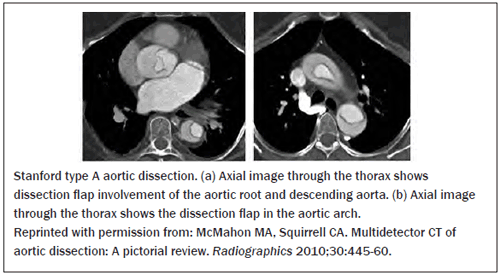
Figure 4: Stanford Type B Aortic Dissection
A "triple-rule-out" CT is an appealing protocol used to visualize the aorta, coronary arteries, and pulmonary arteries during a single scan with the use of several optimally timed boluses of contrast material and ECG gating.46
Magnetic Resonance Imaging. Of all the imaging modalities, MRI has consistently been shown to have the highest sensitivity (98%) and specificity (98%).44 It provides a wealth of information, including the site of intimal tear, type and extent of dissection, aortic insufficiency, and differential flow velocities in the true and false channels and in the aortic side branches.11 Diagnosis is confirmed by visualization of a double lumen with intimal flap.1 MRI avoids the need for administration of contrast agents and saves the patient ionizing radiation.16 This imaging modality, however, is of limited use in the ED. MRI requires patients to leave the ED for extended periods of time for image acquisition, it is unsuited for the unstable patient due to limited monitoring capability, and it may not be available after hours. Additionally, it is contraindicated in patients with implantable devices and aneurysm clips.14 MRI is better suited to following patients after aortic dissection, in patients with chronic aortic dissection, and in those with preexisting aortic disease.
Laboratory Tests
Laboratory tests are of little value in the diagnosis of aortic dissection. Unless the dissection extends into the coronary circulation, cardiac enzymes are often normal to mildly elevated.
Recently, there have been studies of serum biomarkers to exclude aortic dissection. Three markers under investigation are smooth muscle myosin heavy-chain protein (a protein released from damaged aortic media), soluble elastin fragments, and d-dimer levels.
The d-dimer assay has been suggested as a useful screening tool to exclude acute aortic dissection and is attractive to the emergency physician due to the ease of obtaining it and its familiarity. Unfortunately, there is a subset of patients who develop a thrombosed false lumen and fail to stimulate the clotting cascade, creating a false-negative d-dimer level.47 The sensitivity has been reported to be between 94%-97%, but the lower limit of the confidence interval can approach 85%, which is unacceptable for excluding a life-threatening illness.43,47 Several meta-analyses have suggested that a d-dimer value of 500 ng/mL (similar cutoff to pulmonary embolism) provides an acceptable sensitivity in ruling out acute aortic dissection.48,49 Given the limited sample sizes, inconsistent methodology, and different d-dimer cut-off values, it is currently recommended that the d-dimer not be used to exclude acute aortic dissection.
Management
Initial Management. The initial management of type A and type B aortic dissections is similar: stabilize the patient, decrease the blood pressure, and diminish the shearing forces on the aorta. The target blood pressure in aortic dissection is 100 to 120 mm Hg systolic with a heart rate of less than 60.11 Early pain management with opioids will decrease the sympathetic tone and assist in blood pressure control. Morphine sulfate is recommended for pain relief, but fentanyl is preferred for patients with labile blood pressures. A beta-blocker is always first line in treating aortic dissection, since administration of vasodilators can reflexively increase the heart rate. (See Table 3.) Beta-blockers also reduce the shear stresses on the aorta and play a critical role in managing aortic dissection.39
Table 3: Anti-hypertensive Medications
Name |
Starting Dose |
Maintenance Dose |
Comments |
1st Line |
|||
Esmolol (beta-blocker) |
500 mcg/kg IV |
Continuous IV 50-300 mcg/kg/min |
Short acting, may test to see if patient tolerates beta-blocker |
Propranolol (beta-blocker) |
1-3 mg IV |
1-3 mg IV every 4 hours |
No IV infusion available |
Metoprolol (beta-blocker) |
5 mg IV every 2 min x 3 |
5 mg IV every 6 hours or 50 mg PO every 6 hours |
No IV infusion available |
Labetalol (alpha and beta blocker) |
20 mg IV, then 40-80 mg IV every 10 min (max 300 mg) |
Continuous IV 1-2 mg/min, titrate to 6 mg/min |
Good first-line treatment Watch for prolonged hypotension and bradycardia due to its long half-life |
Morphine (opioid analgesic) |
0.1 mg/kg IV |
4-8 mg IV every 4-6 hours |
|
Hydromorphone (opioid analgesic) |
1 mg IV |
1-4 mg IV every 3-6 hours |
|
Fentanyl (opioid analgesic) |
1-2 mcg/kg IV q30-60 min |
1-3 mcg/kg/hr continuous IV infusion |
Least potential to cause hypotension, good for patients with labile BPs |
2nd Line |
|||
Sodium nitroprusside (vasodilator) |
0.5-3 mcg/kg/min IV (max 10 mcg/kg/min x 10 min) |
Continuous IV 1-3 mcg/kg/min |
Preferred vasodilator, only to be used if additional blood pressure control is required after beta-blockade, can cause cyanide toxicity |
Nitroglycerin (vasodilator) |
5 mcg/min IV |
Continuous IV 5 mcg/min, increase 5-20 mcg/min every 3-5 minutes (max 200 mcg/min) |
Non-preferred vasodilator, no evidence to support its use |
Diltiazem (calcium channel blocker) |
0.25 mg/kg IV (max 25 mg) |
Continuous IV 5-10 mg/hour |
Used primarily if patient has major contraindications to beta-blockers (allergy, lung disease, heart failure) |
Verapamil (calcium channel blocker) |
2.5-5 mg IV |
May repeat 5-10 mg IV in 30 min (max 20 mg/total dose) |
|
Resuscitation should proceed as with any unstable patient. If the patient is hypotensive secondary to pericardial tamponade or aortic rupture, IV fluids are the mainstay of therapy, as well as immediate transport to the operating room. Pericardiocentesis is not indicated in aortic dissection, as the procedure may actually cause a rebound in intra-aortic pressure and raise the blood pressure, thus worsening outcomes.50
Esmolol and labetalol are commonly used first-line anti-hypertensive agents in the treatment of aortic dissection. The benefit to esmolol is that it is easily titrated and has a fast-on and fast-off mechanism of action. Labetalol has both alpha- and beta-blocking activity. The selective beta-blockers such as metoprolol or atenolol should be considered in patients with chronic obstructive pulmonary disease or asthma. Calcium channel blockers (diltiazem or verapamil) may be used if beta-blockers are contraindicated.
Sodium nitroprusside can be used in conjunction with beta-blockers for additional blood pressure control if systolic blood pressure remains above 120 mm Hg. Nitroprusside should not be used without first controlling the heart rate with beta-blockade, since vasodilation may trigger a reflex activation of the sympathetic nervous system leading to enhanced ventricular contraction and increased aortic wall shear stress.1
Hydralazine should be avoided, as it can increase aortic wall shear stress and its effect is less predictable than that of other drugs mentioned. If hypertension is refractory to medications, consider renal artery hypertension secondary to the dissection causing renal malperfusion.39
Acute Type A Aortic Dissection. In addition to resuscitation and blood pressure stabilization, acute type A aortic dissections require prompt surgical treatment in which the intimal tear is resected and replaced with a graft to redirect blood into the true lumen.11,51 When aortic regurgitation complicates acute type A dissection, it is necessary to either repair the valve through resuspension or replace the valve during surgery.39
There are several circumstances in which exclusive medical management may be considered, including completed stroke with Glasgow Coma Scale ≤ 5, multiple comorbid conditions (cancer, multi-organ system failure, advanced age), and prior aortic valve replacement.50,52,53 Completed stroke is considered a relative contraindication to surgical repair because of the concern that anticoagulation and reperfusion may convert ischemic stroke to hemorrhagic stroke.14,50
Currently, research is being conducted to determine the role of endovascular treatment or a combined surgical and endovascular approach.14,54
Occasionally, patients with type A aortic dissections will present to the ED or be transferred to a tertiary care center more than 48 hours after the initial event. These cases are deemed subacute dissections and have already passed through the initial most deadly period. It may be reasonable in such cases, after discussing with the cardiothoracic surgeon, to initiate medical management and delay nighttime surgery until the next morning.42
Acute Type B Aortic Dissection. Current standard of care for acute uncomplicated type B aortic dissections is medical management with strict blood pressure control.55-57
Open surgical treatment for type B dissections is generally reserved for complicated presentations and has a mortality of 21-50%.58 Patients to consider for surgical or endovascular treatment of acute type B dissections include those with signs of malperfusion (organ or limb ischemia), progressive dissection, intractable pain, uncontrolled hypertension, or impending aortic rupture.13,25,59 Surgical repair can be complicated by spinal cord ischemia, renal ischemia, dysrhythmias, stroke, or end organ ischemia.57
Endovascular repair with aortic stent grafting has become increasingly used instead of open surgical repair in patients with complicated acute type B dissections, with the goal being to redirect the blood flow into the true lumen and cause thrombosis of the false lumen.58 Current evidence suggests that this technique yields similar mortality (11%) to that of patients with complicated type B aortic dissections treated medically (9%).60
Asymptomatic Thoracic Aortic Aneurysm. It is not uncommon that an asymptomatic thoracic aortic aneurysm is found incidentally on CT. Studies have shown that after an aneurysm reaches 6 cm in the ascending and 7 cm in the descending aorta, the complication rates are greatly increased.61,62 Therefore, it is recommended that patients with an aneurysm of the ascending aorta greater than 5.5 cm or of the descending aorta greater than 6 cm undergo surgical intervention.62 The cutoff is lower in patients with genetic conditions such as Marfan syndrome or Ehlers-Danlos. Any patient with even a mildly dilated aorta (> 3.5 cm) needs annual screening by their primary care physicians.
Intramural Hematoma. IMH is classified similar to aortic dissection and is considered a precursor to aortic dissection. The treatment principles are similar in that patients with uncomplicated type B IMH are treated medically. The evidence for surgical treatment of type A IMH is less robust, but mortality may be slightly lower with surgical intervention.14,15
Penetrating Aortic Ulcer. A PAU develops as a result of atherosclerotic plaque rupture and can be associated with IMH. They are predominately located in the descending aorta. No generally accepted therapeutic regimens have been established for the treatment of PAU apart from tight blood pressure control.14,22
Pregnancy and Aortic Dissection
Treatment of acute aortic dissection during pregnancy is dependent upon the type of dissection as well as gestational age. Pregnant patients with type B dissections are treated similar to non-pregnant patients with IV beta-blockers and nitroprusside.63 Type A dissections require surgical intervention. If the fetus is less than 28 weeks, it is recommended to perform surgical repair with the fetus in utero, and after 32 weeks, surgical repair is performed concomitantly with cesarean section.30,64 Between 28 and 32 weeks gestation, the risk to the fetus being delivered premature is weighed against maternal condition.64
Cocaine and Aortic Dissection
Management principles are similar in patients with cocaine-related aortic dissection. The drugs of choice, however, vary slightly for those with cocaine-related aortic dissection. There is a long-standing fear that beta-blockade in patients with cocaine use will lead to unopposed alpha adrenergic receptor-mediated vasoconstriction and severe hypertension.65 Therefore, benzodiazepines, either in bolus doses or continuous infusion, are the first-line treatment of cocaine-induced hypertension. Phentolamine may be considered for refractory hypertension.65 The role of labetalol and calcium channel blockers in cocaine-related aortic dissection is still uncertain.65
Chronic Aortic Dissection
Chronic aortic dissection is primarily managed with long-term blood pressure management and close follow-up. Indications for resection include diameter greater than 6 cm, enlargement of more than 7 to 10 mm in one year, recurrent persistent pain, and localized saccular dilation.42
Follow-up
Patients with aortic dissection should be maintained indefinitely on antihypertensives including beta-blockers and ACE inhibitors as tolerated. They require periodic monitoring for complications, including aortic insufficiency, aneurysm formation, re-dissection, and rupture. Patients who have survived acute aortic dissection should be re-imaged at 1, 3, 6, and 12 months after the event and then every 1-2 years thereafter, with MRI being the imaging modality of choice.65
Recurrent Symptoms After Surgical Repair or Endovascular Stenting
Patients who present to the ED after surgical repair or endovascular stenting represent a unique group of patients, with the most common cause of death in long-term survivors being rupture of the aorta due to a subsequent dissection or aneurysm formation.1 Once the aorta is greater than 6 cm in diameter, there is a 12% annual mortality rate secondary to false lumen rupture.51 Approximately 15% of operative survivors will require late reoperation at an average of 5 years postoperatively.42
Outcomes/Mortality
Outcomes in aortic dissection still remain poor. An estimated 21% of patients with aortic dissection die before reaching the hospital.1 Acute type A dissection has a mortality of 1-2% per hour during the first 24-48 hours and a 62-91% mortality if untreated within one week.14 Even with surgical correction, the 14-day mortality rate for acute type A dissections is about 25%, and reported survival rates after hospitalization range from 52% to 94% at one year and 45% to 88% at 5 years.39
The early mortality rate for acute type B dissections treated medically is around 10% compared to 25% to 50% for those treated surgically.39 Long-term survival rates after acute type B dissections range from 56% to 92% at 1 year and 48% to 82% at 5 years.39 Patients who present with side-branch vessel involvement, paraplegia, and shock have a poorer prognosis.
Medicolegal Considerations
There are no reliable history or physical examination findings to exclude aortic dissection, and these patients are often misdiagnosed with other clinical conditions presenting with chest pain, including acute coronary syndrome.66 Most cases of missed dissection that prompted legal action involved death or stroke, with the majority of cases centered on delay in diagnosis.67
Critical components of the history and physical exam that should prompt the treating physician to consider the diagnosis of aortic dissection include: abrupt onset of pain, pulse or blood pressure differentials, abnormal chest radiograph findings, unexplained syncope, acute-onset congestive heart failure, those patients who have chest pain in addition to another complaint such as weakness, neurologic complaints, extremity or abdominal pain, and patients who are younger than the typical patient with acute coronary syndrome.45,73
Disposition
Disposition of all patients with acute aortic dissections needs to be to an intensive care unit skilled in handling such patients. Prompt diagnosis and transfer to a tertiary care center with cardiothoracic capability can be life-saving.
Figure 5: Diagnostic Algorithm for Patients with Suspected Aortic Dissection
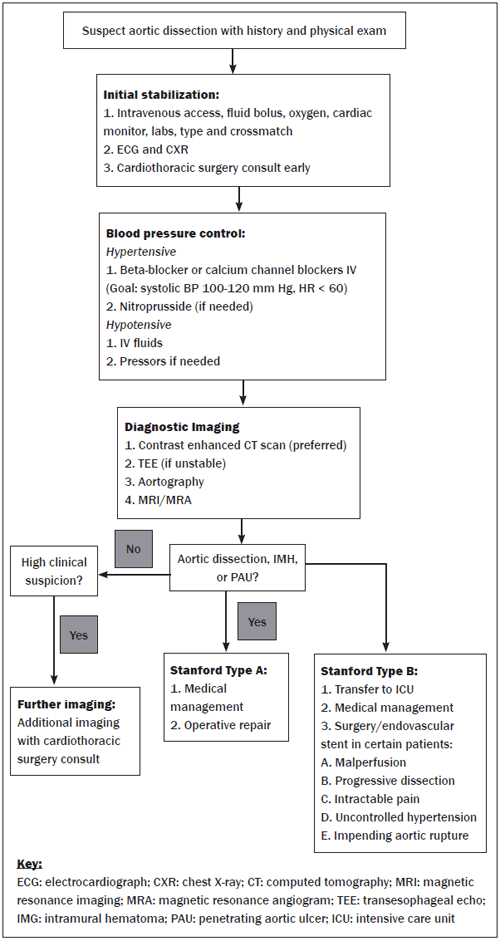
Conclusion
Aortic dissection is an uncommon but potentially fatal disease. Early diagnosis and treatment is essential. All patients with aortic dissection require strict blood pressure control, and those with type A dissections require prompt surgical intervention. Mortality rates are time-dependent, and despite advances in technology, morbidity and mortality in aortic dissection remain high.
References
- Patel PD, Arora RR. Pathophysiology, diagnosis, and management of aortic dissection. Therapeutic Advances in Cardiovascular Disease 2008;2:439–468.
- Chen K, Varon J, Wenker OC, et al. Acute thoracic aortic dissection: The basics. J Emerg Med 1997;15:859–867.
- Hagan PG, Nienaber CA, Isselbacher EM, et al. The international registry of acute aortic dissection (IRAD). JAMA 2000;283:897–903.
- Kouchoukos NT, Dougenis D. Surgery of the thoracic aorta. N Engl J Med 1997;336:1876–1889.
- Clouse WD, Hallett Jr JW, Schaff HV, et al. Acute aortic dissection: Population-based incidence compared with degenerative aortic aneurysm rupture. Mayo Clinic Proceedings 2004;79:176–180.
- Sato F, Kitamura T, Kongo M, et al. Newly diagnosed acute aortic dissection. Int Heart J 2005;46:1083–1098.
- Yu HY, Chen YS, Huang SC, et al. Late outcome of patients with aortic dissection: Study of a national database. Eur J Cardio-thoracic Surg 2004;25:683–690.
- Olsson C, Thelin S, Ståhle E, et al. Thoracic aortic aneurysm and dissection. Circulation 2006;114:2611–2618.
- LeMaire SA, Russell L. Epidemiology of thoracic aortic dissection. Nature Reviews Cardiology 2010;8:103–113.
- Tintinalli JE, Stapczynski JS, Ma OJ, et al. Tintinalli's Emergency Medicine: A Comprehensive Study Guide, 7th ed. McGraw-Hill; 2011.
- Rosen AF. Rosen's Emergency Medicine: Concepts and Clinical Practice, 7th ed. Mosby Elsevier; 2010.
- Williams GM, Gott VL, Brawley RK, et al. Aortic disease associated with pregnancy. J Vascular Surgery 1988;8:470–475.
- Lin PH, Huynh TT, Kougias P, et al. Descending thoracic aortic dissection: Evaluation and management in the era of endovascular technology. Vascular and Endovascular Surgery 2009;43:5–24.
- Nordon IM, Hinchliffe RJ, Loftus IM, et al. Management of acute aortic syndrome and chronic aortic dissection. Cardiovascular and Interventional Radiology 2011;34:890–902.
- Nienaber CA, Powell JT. Management of acute aortic syndromes. Eur Heart J 2012;33:26–35.
- Golledge J, Eagle KA. Acute aortic dissection. Lancet 2008;372:55–66.
- Setacci F, Sirignano P, de Donato G, et al. Acute aortic dissection: Natural history and classification. J Cardiovascular Surgery 2010;51:641–646.
- Macura KJ, Corl FM, Fishman EK, et al. Pathogenesis in acute aortic syndromes: Aortic dissection, intramural hematoma, and penetrating atherosclerotic aortic ulcer. AJR 2003;181:309–316.
- Maraj R, Rerkpattanapipat P, Jacobs LE, et al. Meta-analysis of 143 reported cases of aortic intramural hematoma. Am J Cardiology 2000;86:664–668.
- Yamada T, Tada S, Harada J. Aortic dissection without intimal rupture: Diagnosis with MR imaging and CT. Radiology 1988;168:347–352.
- Nienaber CA, von Kodolitsch Y, Petersen B, et al. Intramural hemorrhage of the thoracic aorta: Diagnostic and therapeutic implications. Circulation 1995;92:1465–1472.
- Lansman SL, Saunders PC, Malekan R, et al. Acute aortic syndrome. J Thoracic and Cardiovascular Surgery 2010;140:92–97.
- Coady MA, Rizzo JA, Elefteriades JA. Pathologic variants of thoracic aortic dissections: Penetrating atherosclerotic ulcers and intramural hematomas. Cardiology Clin 1999;17:637–657.
- Harris JA, Bis KG, Glover JL, et al. Penetrating atherosclerotic ulcers of the aorta. J Vascular Surgery 1994;19:90-98.
- Nienaber CA, Kische S, Ince H, et al. Thoracic endovascular aneurysm repair for complicated type B aortic dissection. J Vascular Surgery 2011;54:1529–1533.
- Grubb KJ, Kron IL. Sex and gender in thoracic aortic aneurysms and dissection. Semin Thoracic d Cardiovascular Surgery 2011;23:124–125.
- Mészáros I, Mórocz J, Szlávi J, et al. Epidemiology and clinicopathology of aortic dissection. Chest 2000;117:1271–1278.
- Elefteriades JA. Natural history of thoracic aortic aneurysms: Indications for surgery, and surgical versus nonsurgical risks. Ann Thoracic Surgery2002;74:1877–1880.
- Alli O, Jacobs L, Amanullah A. Acute aortic syndromes: Pathophysiology and management. Reviews in Cardiovascular Medicine 2008;9:111–124.
- Houston L, Tuuli M, Macones G. Marfan syndrome and aortic dissection in pregnancy. Obstetrics and Gynecology 2011;117:956–960.
- Hatzaras I, Tranquilli M, Coady M, et al. Weight lifting and aortic dissection: More evidence for a connection. Cardiology 2007;107:103–106.
- De Paepe A, Devereux RB, Dietz HC, et al. Revised diagnostic criteria for the Marfan syndrome. Am J Medical Genetics 1996;62:417–426.
- Barbetseas J, Alexopoulos N, Brili S, et al. Atherosclerosis of the aorta in patients with acute thoracic aortic dissection. Circulation Journal 2008;72:1773-1776.
- Ando M, Okita Y, Morota T, et al. Thoracic aortic aneurysm associated with congenital bicuspid aortic valve. Cardiovascular Surgery 1998;6:629–634.
- Oliver JM, Gallego P, Gonzalez A, et al. Risk factors for aortic complications in adults with coarctation of the aorta. J Am Coll Cardiol 2004;44:1641–1647.
- von Kodolitsch Y, Aydin MA, Koschyk DH, et al. Predictors of aneurysmal formation after surgical correction of aortic coarctation. J Am Coll Cardiol2002;39:617–624.
- Prêtre R, Von Segesser LK. Aortic dissection. Lancet 1997;349:1461–1464.
- Daily PO, Trueblood HW, Stinson EB, et al. Management of acute aortic dissections. Ann Thoracic Surgery 1970;10: 237-247.
- Braverman AC. Aortic dissection: Prompt diagnosis and emergency treatment are critical. Cleveland Clinic J Med 2011;78:685–696.
- Hiratzka LF, Bakris GL, Beckman JA, et al. 2010 ACCF/AHA/AATS/ACR/ASA/SCA/SCAI/SIR/STS/SVM guidelines for the diagnosis and management of patients with thoracic aortic disease. J Am Coll Cardiol 2010;55:1510-1539.
- Gaul C, Dietrich W, Erbguth FJ. Neurological symptoms in aortic dissection: A challenge for neurologists. Cerebrovascular Diseases 2008;26:1–8.
- Moon MR. Approach to the treatment of aortic dissection. Surgical Clin North Am 2009;89:869–893.
- Wittels K. Aortic emergencies. Emerg Med Clin North Am 2011;29:789–800.
- Shiga T, Wajima Z, Apfel CC, et al. Diagnostic accuracy of transesophageal echocardiography, helical computed tomography, and magnetic resonance imaging for suspected thoracic aortic dissection: Systematic review and meta-analysis. Arch Intern Med 2006;166:1350–1356.
- Chung JH, Ghoshhajra BB, Rojas CA, et al. CT angiography of the thoracic aorta. Radiologic Clin North Am 2010;48:249–264.
- McMahon MA, Squirrell CA. Multidetector CT of aortic dissection: A pictorial review. Radiographics 2010;30:445–460.
- Sutherland A, Escano J, Coon TP. D-dimer as the sole screening test for acute aortic dissection: A review of the literature. Ann Emerg Med 2008;52:339–343.
- Shimony A, Filion KB, Mottillo S, et al. Meta-analysis of usefulness of D-dimer to diagnose acute aortic dissection. Am J Cardiology 2011;107:1227–1234.
- Marill KA. Serum D-dimer is a sensitive test for the detection of acute aortic dissection: A pooled meta-analysis. J Emerg Med 2008;34:367–376.
- Feldman M, Shah M, Elefteriades JA. Medical management of acute type A aortic dissection. Ann Thorac Cardiovasc Surg 2009;15:286–293.
- Karthikesalingam A, Holt P, Hinchliffe R, et al. The diagnosis and management of aortic dissection. Vascular and Endovascular Surgery 2010;44:165–169.
- Elefteriades JA, Feldman M. Acute Type A aortic dissection: Surgical intervention for all: CON. Cardiology Clinics 2010;28:325–331.
- Estrera AL, Safi HJ. Acute Type A aortic dissection: Surgical intervention for all: PRO. Cardiology Clinics 2010;28:317–323.
- Bonser RS, Ranasinghe AM, Loubani M, et al. Evidence, lack of evidence, controversy, and debate in the provision and performance of the surgery of acute Type A aortic dissection. J Am Coll Cardiol 2011;58:2455–2474.
- Booher AM, Isselbacher EM, Nienaber CA, et al. Ascending thoracic aorta dimension and outcomes in acute type B dissection (from the International Registry of Acute Aortic Dissection [IRAD]). Am J Cardiology 2011;107:315–320.
- Bogdan Y, Hines GL. Management of acute complicated and uncomplicated Type B dissection of the aorta: Focus on endovascular stent grafting. Cardiology in Review 2010;18:234–239.
- Apostolakis E, Baikouss NG, Georgiopoulos M. Acute Type-B aortic dissection: The treatment strategy. Hellenic J Cardiol 2010;51:338–347.
- Meisner RJ, Labropoulos N, Bilfinger T. The current state of type B aortic dissection. Acta Chirurgica Belgica 2011;111:57–62.
- Tran TP, Khoynezhad A. Current management of type B aortic dissection. Vascular Health and Risk Management2009;5:53–63.
- Fattori R, Tsai TT, Myrmel T, et al. Complicated acute type B dissection: Is surgery still the best option? A report from the International Registry of Acute Aortic Dissection. JACC: Cardiovascular Interventions 2008;1:395–402.
- Elefteriades JA, Farkas EA. Thoracic aortic aneurysm: Clinically pertinent controversies and uncertainties. J Am Coll Cardiol 2010;55:841–857.
- Booher AM, Eagle KA. Diagnosis and management issues in thoracic aortic aneurysm. Am Heart J 2011;162:38–46.
- Zeebregts CJ, Schepens MA, Hameeteman TM, et al. Acute aortic dissection complicating pregnancy. Ann Thoracic Surgery 1997;64:1345–1348.
- Parlakgumus HA, Haydardedeoglu B, Alkan O. Aortic dissection accompanied by preeclampsia and preterm labor. J Obstetrics and Gynaecology Research 2010;36:1121–1124.
- Singh A, Khaja A, Alpert MA. Cocaine and aortic dissection. Vascular Medicine 2010;15:127–133.
- Woo KC, Schneider JI. High-risk chief complaints I: Chest pain-the big three. Emerg Med Clin North Am 2009;27:685–712.
- Elefteriades JA. Physicians should be legally liable for missing an atypical aortic dissection: CON. Cardiology Clinics 2010;28:245–252.
Physician CME Questions
Which of the following is the most common risk factor for the development of aortic dissection?- Marfan syndrome
- hypertension
- bicuspid aortic valve
- cocaine use
- pregnancy
- Aortic dissections only propagate in the anterograde fashion.
- The most common site of intimal tear is the right posterolateral aorta.
- Penetrating aortic ulcers carry a poorer prognosis than intramural hematoma or classical aortic dissection.
- Aortic dissection is believed to be caused from the degeneration of the intimal layer.
- hypertension
- hemorrhage
- atherosclerosis
- connective tissue disorder
- Stanford type A dissection
- De Bakey type I
- De Bakey type II
- Stanford type B dissection
- Aortic regurgitation is more common in type B dissections.
- The location of pain can help localize the area of injury.
- Lack of hypertension essentially rules out the diagnosis of aortic dissection.
- Patients describe the pain more as a stabbing than sharp.
- 65-year-old male with acute onset of crushing back pain
- 70-year-old male with acute onset of stabbing chest pain with a weak femoral pulse
- 30-year-old male with Marfan syndrome and chest pain
- 25-year-old male with acute onset of chest pain and history of cocaine use
- normal chest X-ray
- calcium sign
- obliteration of the aortic knob
- left pleural effusion
- mediastinal widening
- A negative transthoracic echocardiography effectively rules out an aortic dissection.
- Laboratory tests are of great value in the diagnosis of aortic dissection.
- CTA has a high sensitivity and specificity and is the diagnostic test of choice in most institutions.
- Aortography has the highest sensitivity of all the diagnostic modalities.
- esmolol
- nitroprusside
- hydralazine
- hydrochlorothiazide
- young age
- ongoing stroke
- prior aortic valve replacement
- patient on anticoagulation
Click here to take your CME/CNE test online.
Acute aortic dissection is a rare but life-threatening illness in which prompt diagnosis and treatment are critical for the chance of survival.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
