Pertussis in the Emergency Department
Pertussis in the Emergency Department
Authors: Sharon G. Humiston, MD, MPH, Associate Professor of Emergency Medicine and Pediatrics, Department of Emergency Medicine, University of Rochester, Rochester, NY; and Anne F. Brayer, MD, Department of Emergency Medicine, University of Rochester, Rochester, NY.
Peer Reviewers: John P. Santamaria, MD, FAAP, FACEP, Chief Medical Officer, After Hours Pediatrics, Associate Professor of Pediatrics, USF School of Medicine, Tampa, FL; and Ronald M. Perkin, MD, Professor and Chairman, Department of Pediatrics, Brody School of Medicine, East Carolina University, Greenville, NC.
Pertussis, or whooping cough, was first described in the 16th century, and the causative agent was isolated in 1906. Nonetheless, pertussis remained a major cause of morbidity and mortality among ch ildren well into the 20th century. During the pre-vaccine era, the average reported annual U.S. pertussis incidence was 157 per 100,000. This dropped to fewer than 1 per 100,000 by the 1970s.1 Pertussis is again a hot topic because of a rise in the number of reported cases in the United States2 and Canada,3 new evidence about the clinical impact of pertussis in adolescents and adults,4 and new opportunities for prevention.5,6
—The Editor
Introduction
Emergency medicine physicians often have misconceptions about pertussis that make the diagnosis easy to miss, and hence, to inadvertently increase the likelihood of disease spread. The case of an unvaccinated child is instructive: A school-aged girl was seen in an urban emergency department (ED) for trauma caused by the kick of a horse. Her past medical history revealed culture-proven pertussis in a household member, no use of antibiotic prophylaxis, and the development of a cough illness. Because the child lived in a religious community that avoided vaccination, she had never been vaccinated. Hospital Infection Control recommended droplet isolation. A sign was posted on the door of the patient’s ED (private) room stating that all personnel should wear a mask and wash their hands before and after patient contact. Nonetheless, the ED attending found several residents closely examining the patient without wearing a mask. The reason repeatedly given was, "I’m immune to pertussis. I was vaccinated when I was a kid." In fact, pertussis vaccination does not confer lifelong immunity. Most fully vaccinated persons are susceptible again by adolescence.7
Other pertussis myths and misunderstandings are illustrated by another case. A coughing child was cultured for pertussis in his primary care physician’s office shortly before a holiday. His family—not realizing the potential severity or communicability of the illness—took the child visiting and stopped in relatives’ homes, hotels, and restaurants in several states. They returned two weeks later just as the pertussis culture was reported to be positive. The state department of health personnel had a frenzied load of contact tracking to do. One relative who certainly deserved prophylaxis was the grandfather who lived with the child. He was told by the department of health personnel to go to his doctor for prophylaxis. Since he did not have a primary care physician, he sought care in an ED. There he was told, "Don’t worry. You don’t need antibiotics. Pertussis is only a children’s disease." The infected child’s grandfather (a household contact) should have received prophylaxis both to prevent disease symptoms and to prevent spread. Adolescents and adults often are the ones to spread Bordetella pertussis to young infants,8 who remain the group most likely to become seriously ill or die from pertussis.9 It has been estimated that the 10-year economic impact of pertussis in the adolescent age group alone is $0.6 billion in direct costs and $2.6 billion in indirect costs.10
In several countries, including Canada,11 France, Australia, and Germany,12 a new pertussis vaccine is being given to adolescents to boost immunity. Until recently in the United States pertussis vaccine was licensed only for children up to the seventh birthday. Recently, the Food and Drug Administration’s vaccine advisory committee licensed two pertussis vaccines for use in older individuals, and the CDC’s Advisory Committee on Immunization Practice recommended a routine pertussis booster during adolescence.13 Some emergency medicine personnel may be interested in receiving the booster as well—especially if they have had to receive antibiotics repeatedly for pertussis exposure.
Pertussis Disease
Epidemiology. Incidence and Prevalence. Before the vaccine era, pertussis was a common and serious illness among children. From 1940-1945, more than 1 million cases of pertussis were reported. The vaccine was introduced in the 1940s; thereafter, pertussis incidence fell to fewer than 5000 cases per year in the 1970s. (See Figures 1 and 2.) The nadir was 1976 when 1010 cases of pertussis were reported.14
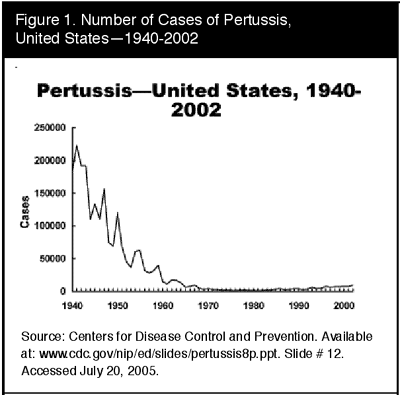
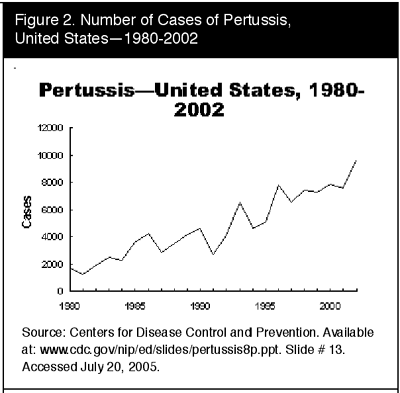
Notably, the number of pertussis cases reported has been gradually increasing. More than 19,000 cases were reported in 2004.13 Experts are debating the reasons for the increase. The increase may be in part a reflection of the 3- to 5-year cycle observed with the disease. Some argue that use of less efficacious pertussis vaccines has contributed to the increase,15 though other experts deny this.12 It must be noted that in 1995 criteria for reporting a pertussis case changed in two important ways: polymerase chain reaction became an accepted method of pertussis confirmation, and data collection began for pertussis cases epidemiologically linked to another pertussis case. These changes primarily affected the reporting among persons 10 years of age or older.16 Overall, the reported incidence of pertussis is estimated to be 1% to 36% of the true incidence.17 In Massachusetts, which has had active surveillance and testing systems with improved sensitivity, the reported incidence of pertussis among adolescents and adults is many fold higher than in the United States overall.18
Among infants in the United States, reported cases of pertussis increased by 49% from the 1980s to the 1990s. Because there was an increased rate of bacteriologic confirmation (not an increase in reliance on PCR) and unchanged severity of illness (not an increase in reporting of mild cases) over this time period, CDC experts believe the increase in pertussis incidence among infants was not a reporting artifact.19
Epidemic Behavior. Outbreaks of pertussis are not uncommon and have been reported in a wide variety of settings such as schools and childcare facilities, institutions for the disabled, health care facilities, workplaces, households, and community wide.20-24 In a recent pertussis outbreak between September 2002 and February 2003 in Yavapai County, Arizona, 485 cases were reported, including 203 associated with schools (56% students, 4% staff, 40% family members, including 9 infants). In this outbreak, the median age was 13 years.25
Geographic Distribution. Pertussis occurs worldwide. For 1999, Crowcroft et al estimated that there were 48.5 million cases of pertussis in children worldwide and 295,000 related deaths. They estimated disability-adjusted life years from pertussis exceeded those from other preventable diseases, including lung cancer and meningitis.26
Temporal Distribution. Pertussis is an endemic disease with 3- to 5-year cycles of increased disease. Pertussis has no distinct seasonal pattern, but may increase in the summer and fall.16
Host Factor. People of any age can be infected, but the reported disease incidence is highest in the first year of life. For example, in 1997-2000 infants younger than 1 year of age had an annual reported pertussis incidence of 55.5 cases per 100,000 population.16 The relative proportion of infected adolescents and adults is growing, though it is not known if this increase is real or simply a reflection of improvements in diagnostic procedures and increasingly complete disease reporting in the older age groups.7
Age also is an important host factor in pertussis with respect to disease severity. Pertussis is most severe for children in the first 6 months of life,27 especially for the premature and unimmunized infants.28 Currently in the United States, the pertussis-related mortality rate among infants is 2.4 deaths per million, and 90% of all fatal cases of pertussis occur in infants.14
Nosocomial Infections. Spread of B. pertussis from infected patients to health care workers and vice versa has been well documented.29 Health care workers in the catarrhal phase of the disease have non-specific common "cold" symptoms so they are unlikely to miss work, yet the disease is highly contagious during this phase.
Case Definition and Classification.16 The Council of State and Territorial Epidemiologists (CSTE) and the CDC have developed the current case definition for pertussis. A clinical case of pertussis is defined as: An acute cough illness lasting at least 2 weeks with either paroxysms of coughing, inspiratory "whoop," or post-tussive vomiting without other apparent cause (as reported by a health professional).
The clinical case definition above is appropriate for endemic or sporadic cases, but in outbreak settings, including household exposures, a case can be defined simply as an acute cough illness lasting at least 2 weeks without other symptoms.
Classification of pertussis into probable and confirmed cases is shown below.
Probable—Meets the clinical case definition, but is not laboratory confirmed and is not epidemiologically linked to a laboratory-confirmed case.
Confirmed—A clinically compatible case that is laboratory-confirmed or epidemiologically linked to a laboratory-confirmed case.
Both probable and confirmed cases should be reported to the National Notifiable Diseases Surveillance System.
Pertussis Surveillance. Statistics on pertussis cases depend on two surveillance systems. Cases are reported to the CDC via two systems: the National Electronic Transmittal System for Surveillance and the more detailed Supplementary Pertussis Surveillance System. The absolute number of cases is not accurate because of underreporting, but the surveillance system serves as a monitor for epidemiologic trends. Though emergency medicine physicians often assume that reporting is a function of the hospital laboratory or the hospital infection control department, clinicians should report suspected cases. Guidelines on pertussis surveillance and outbreak control are available on the National Immunization Program website (http://www.cdc.gov/nip/publications/pertussis/guide.htm).
Bordetella pertussis. First isolated in 1906, Bordetella pertussis is the only organism of major clinical importance that causes whooping cough, though B. parapertussis causes a milder form of bronchitis. B. pertussis is a small aerobic gram-negative rod that has affinity for the mucosal layers of the human respiratory tract. It produces pertussis toxin as well as virulence factors such as filamentous hemagglutinin, agglutinogens, adenylate cyclase, pertactin, and tracheal cytotoxin. It is these products that are responsible for the clinical features of pertussis disease. An immune response to one or more of these leads to immunity. Significant antigenic variation has not been demonstrated.30 Because B. pertussis is fastidious, special media are necessary for isolation. B. bronchosepticus is the causative agent of respiratory diseases in cats and swine, but can cause broncho-pulmonary symptoms in severely immunosupressed individuals.31
Mechanism and Route of Transmission. Transmission. Transmission most commonly occurs by the respiratory route through contact with aerosolized droplets. Transmission occurs less frequently through contact with freshly contaminated fomites.
Reservoir. Pertussis is a human disease. B. pertussis is only a pathogen for humans and possibly higher primates.32 Infected adolescents and adults often are the source of pertussis infection for infants and frequently are found to be the first case in a household with multiple pertussis cases.8 Asymptomatic carriers may spread the disease, but the carrier state is thought to be infrequent, transient, and probably not of great importance to disease spread.
Communicability. Pertussis is highly communicable, with a secondary attack rate of 80% among susceptible household contacts. Persons with pertussis are most infectious during the catarrhal period and the first two weeks after cough onset (see the Clinical Features section below). Communicability is longer if the host is younger, unimmunized, immunologically naïve, and untreated. The American Academy of Pediatrics Committee on Infectious Diseases states that a "young unimmunized and untreated infant may be infectious for 6 or more weeks after cough onset; an untreated immunized adolescent may be infectious for only 2 weeks after cough onset."33 Erythromycin eliminates nasopharyngeal carriage within five days of starting treatment.
Pathophysiology. Mechanism of Disease Process. Pertussis disease process is mediated by several toxins. (For details, see reference 31.)
Pertussis Toxin. Pertussis toxin is the major cause of the abnormal cough of pertussis. It is an oligopeptide exotoxin that causes immune system havoc. Its effects include: T cell lymphocytosis, increased IgE synthesis, increased histamine and endotoxin sensitivity, inhibition of many leukocyte functions (e.g., chemotaxis, phagocytosis, and respiratory burst), and impairment of NK cell killing. Additionally, it contributes to bacterial binding to ciliated epithelial cells. It exerts many of its effects by preventing the deactivation of adenylate cyclase, which causes large amounts of cAMP to accumulate; this, in turn, leads to increased mucus secretion and interference with many cellular functions.
Adenylate Cyclase Toxin. Adenylate cyclase toxin penetrates the host cells and catalyzes the conversion of ATP to cAMP, but the cAMP increase caused by this toxin is short-lived. Like pertussis toxin, this exotoxin inhibits phagocyte and NK cell functions.
Tracheal Cytotoxin. Tracheal cytotoxin binds to ciliated epithelial cells and interferes with the movement of the cilia. In higher concentrations, it causes ciliated epithelial cell extrusion and destruction, which contributes to pertussis disease symptoms.
Dermonecrotic (Heat-Labile) Toxin. Dermonecrotic toxin is a very strong vasoconstrictor that causes ischemia and extravasation of leukocytes. Together with tracheal cytotoxin, it causes tracheal tissue necrosis.
Filamentous Haemagglutinins (Agglutinogens). These are implicated in the binding of the organism to ciliated epithelial cells. Antibodies against these molecules are protective, probably acting by preventing bacterial attachment.
Lipopolysaccharide (LPS). Like LPS of other gram-negative bacteria, these endotoxins cause a number of pathophysiological effects. In large quantities—as following bacterial cell lysis—they cause irreversible shock and cardiovascular collapse. In smaller quantities, they activate a variety of inflammatory mediators (e.g., TNF, IL1, IL6, prostaglandins) and generate complement activation products.
Specific Organs Involved. The bacteria attach to the respiratory cilia, produce toxins that paralyze the cilia and kill ciliated cells,34 and cause inflammation of the respiratory tract, thus interfering with the clearing of pulmonary secretions. In addition to the direct respiratory effects, pertussis affects other organ systems. Hypoxia during prolonged attacks may lead to neurologic findings such as seizure, encephalopathy, or coma. Coughing interferes with oral intake. This, combined with vomiting the swallowed mucus, often leads to severe dehydration and weight loss.
Potential Complications. Among infected individuals, serious complications from pertussis are most common in young infants. Three common groupings of pertussis complications are respiratory, neurologic, and gastrointestinal. (See Table 1.) The most common complication and the most common cause of pertussis-related deaths is secondary bacterial pneumonia. From 1997-2000, among all reported cases of pertussis, 5% developed pneumonia. Among infected infants younger than 6 months of age, the incidence was almost 12%. Severe paroxysms of coughing can lead to pressure-related respiratory complications such as pneumothorax and epistaxis.

Neurologic complications of pertussis disease, including seizures and encephalopathy, may occur as a result of hypoxia or may be toxin-mediated. Like secondary bacterial pneumonia, neurologic complications of pertussis are more common among infants. From 1997-2000, among all reported cases of pertussis, 0.8% and 0.1% developed seizures and encephalopathy, respectively. Among infected infants younger than 6 months of age, the incidences for these were 1.4% and 0.2%.
Gastrointestinal problems—anorexia and dehydration—are a common cause of hospitalization for pertussis in infants. In adolescents and adults, weight loss is common. Pressure-related GI complications (from excessive coughing) include hernias and rectal prolapse.
Infants younger than 6 months of age may present with apnea. Adolescents and adults report many other pertussis complications, including problems sleeping, urinary incontinence, rib fractures, and missed school.35
Hospitalization is a common complication of pertussis. From 1997 to 2000, among persons with pertussis, 4% of adults, 2% of adolescents, 10% of children 1-4 years old, 28% of infants 6-11 months old, and 63% of infants younger than 6 months of age were hospitalized. During this same time period, 62 deaths from pertussis were reported; 56 of them in infants younger than 6 months of age.36
Immunity. Pertussis antigens appear to allow the organism to evade host defenses in that lymphocytosis is promoted but chemotaxis is impaired. Until recently it was thought that B. pertussis did not invade the tissues, but the bacterium now has been found in alveolar macrophages. Immunity is difficult to assess because there are no serologic correlates of immunity, although the level of antibodies to pertussis toxin or to the filamentous haemagglutinin often are used as indicators of protection. Evidence suggests that immunity from natural B. pertussis infection wanes after 4-20 years.37 Individuals who were presumed to be immune may develop atypical or unrecognized infection. After pertussis infection, sustained high-level immunity may require occasional exposure to circulating wild B. pertussis. Unfortunately, infants born to mothers who have had pertussis do not necessarily receive protective levels of maternal antibody. Consequently, neonates are particularly vulnerable prior to completion of the primary vaccination series (4 doses, usually given in the first 18 months of life).
Clinical Features. Pertussis’ incubation period usually is 7-10 days, with a range of 4-21 days. In rare cases the incubation period may be as long as 42 days. The subsequent disease is divided into three stages: the catarrhal stage, the paroxysmal stage, and the convalescent stage.
The first or catarrhal stage is indistinguishable from the common cold, beginning insidiously with onset of coryza, sneezing, low-grade fever, and a minor cough. Gradually, the cough worsens over a week or two, signaling the start of the second stage, the paroxysmal stage. Although the culture for B. pertussis is more likely to yield a true positive during the catarrhal stage, the suspicion of pertussis rarely is raised before the paroxysmal stage. Generally, fever is minimal throughout the course of pertussis, which may confuse physicians who assume that cough in the absence of fever is not serious.
In classic cases in pediatric patients, the infected child has paroxysms of rapid coughs caused by difficulty clearing thick mucus from the tracheobronchial tree. Characteristically, the coughing paroxysms are followed by a long, high-pitched inspiratory effort (the "whoop"). Hewlett and Edwards describe the episodes as "rapid forced expirations, followed by gasping inhalation."14 Like older children, infants younger than 6 months of age have paroxysms of coughing but may not have the strength to produce the characteristic whoop. The paroxysmal attacks of pertussis may be associated with cyanosis and general distress and often are followed by post-tussive emesis. The child may look surprisingly normal—though tired—between attacks. Paroxysmal attacks occur an average of 15 times per 24-hour period and more of these occur at night than during the day. The paroxysmal stage usually lasts 1-6 weeks, but may persist for up to 10 weeks. In young children, duration of cough can be greatly influenced by vaccination status.38 The frequency of the paroxysms increases during the first 2 weeks of this stage, remains approximately constant for 2-3 weeks, and then decreases gradually.
It is important for physicians to know that adolescents and adults, as well as partially vaccinated children, may become infected with B. pertussis and spread the disease to infants who remain unimmunized or are too young to be fully immunized. Older infected individuals typically have milder disease than infants do. Frequently, the chief complaint is persistent cough. Studies have shown an association between serologic evidence of B. pertussis and cough illness lasting more than 6 days.39 In adolescents and adults, some pertussis symptoms may not distinguish the illness from other upper respiratory infections—e.g., scratchy throat, sweating. The belief that pertussis need not be considered in the absence of an inspiratory whoop can lead to a missed diagnosis; inspiratory whoop with pertussis is uncommon in adolescents and adults.
Gradual recovery occurs during the convalescent stage. Paroxysms of coughing grow less frequent over the course of a few weeks, but recur with subsequent respiratory infections for many months after the onset of pertussis, hence the traditional name for pertussis, "the 100-day cough."
Diagnostic Studies. Pertussis usually is a clinical diagnosis. Laboratory tests are problematic, particularly in atypical cases that present to the physician late in the clinical course.
Isolation of B. pertussis by culture is the gold standard for confirmation of the diagnosis of pertussis. Because it is fastidious, B. pertussis is difficult to isolate. Isolation of the organism is most successful under the following circumstances: 1) the swab is plated directly at the bedside onto selective Regan-Lowe media; 2) the specimen is obtained during the catarrhal stage of the disease; 3) the specimen is obtained from the posterior nasopharynx, not the throat; and 4) the swab is Dacron or calcium alginate (not cotton). The medium used to grow B. pertussis is Bordet-Gengou medium (potato-glycerol-blood agar) and growth is inhibited by compounds found in ordinary media. Isolation is less successful under circumstances over which the emergency medicine physician has little control: prior use of antibiotics effective against pertussis (e.g., erythromycin, trimethoprim-sulfamethoxazole), prior vaccination, or specimen collection beyond the first two weeks of illness.
Polymerase chain reaction (PCR) testing is a recent tool for diagnosing pertussis rapidly with good sensitivity and specificity. PCR can be done on nasopharyngeal swabs or aspirates. Of course, it is not available in every hospital laboratory. Even in settings in which PCR is available, it is recommended that culture accompany PCR so that bacterial isolates can be evaluated for antimicrobial resistance or molecular typing.
Direct fluorescent antibody (DFA) testing of nasopharyngeal specimens may be useful as a pertussis screening test but, because of low sensitivity and variable specificity, it is not sufficiently reliable for laboratory confirmation.
Serological testing is not standardized, is not widely available, and, because there is no clear association between antibody levels and immunity to pertussis, is not easy to interpret. For these reasons, serologic testing should not be used for case confirmation or for the purpose of national reporting. If a patient meets the clinical case definition but is not culture or PCR positive, this patient should be reported as a probable case even if serologically positive.
An elevated white blood cell count with a lymphocytosis is part of the classical disease description,40 but this may be absent in infants and children or in mild or modified cases of pertussis.
Differential Diagnosis. The differential diagnosis for cough is extensive41 and varies by age group; often the patient makes several visits before the correct diagnosis is made.42 Many infectious diseases can cause a similar cough such as certain adenoviruses and influenza. The latter, however, is primarily a fall-winter diagnosis, and there are rapid tests now available to diagnose influenza. Mycoplasma pneumonia must be considered, as well Chlamydia pneumoniae and Chlamydia trachomatis. Among pediatric patients, bronchiolitis (often caused by respiratory synctial virus) and croup or laryngotracheobronchitis are important in the differential diagnosis. Tuberculosis should be considered, especially in high-risk populations. Bordetella parapertussis and Bordetella bronchiseptica also may cause a similar cough.
Non-infectious causes of prolonged cough illness are legion. Asthma, chronic obstructive pulmonary disease, and emphysema could be suspected. A history consistent with gastroesophageal reflux disease, aspiration pneumonia, or malignancy would lead to further evaluation. In pediatric patients, tracheal foreign bodies and cystic fibrosis also are considerations.
Vomiting and dehydration may be the chief complaint, leading to a differential diagnosis headed up by gastroenteritis and intussusception. Neurologic changes may be the chief complaint of viral and bacterial causes of encephalitis. In children, febrile seizures would need to be considered.
Prevention
If there is no known exposure, good hand washing and vaccination are sufficient for prevention. In circumstances in which exposure is likely, more rigorous methods are recommended.
Isolation of Infected and of Susceptible People. Droplet precautions are recommended until the patient is no longer spreading the viable bacteria, that is, 5 days after effective antibiotic treatment is initiated or 21 days after the onset of paroxysmal cough if antibiotics are not given.33 During epidemics, unimmunized children should be excused from school and public gatherings and should be isolated from anyone who is known or suspected to be infected. This should last until 14 days after the last reported case.16
Observation for Symptoms. Exposed individuals should be observed for respiratory tract symptoms for the full possible incubation period of B. pertussis, i.e., 21 days after contact with the infected person.
Prophylactic Antibiotic Use. There have not been controlled clinical trials of prophylaxis of household members and other close contacts (e.g., others in child care), but antibiotics are recommended for these people in an effort to prevent or minimize transmission. Prophylaxis of all household and other close contacts of persons with pertussis should be attempted by administering antibiotics regardless of age and vaccination status.16
Vaccination. Whole-cell pertussis vaccine, composed of a suspension of killed B. pertussis cells, was developed in the 1930s and was widely used by the mid-1940s. Four doses of this vaccine were quite effective, and protection lasted for 5-10 years after the last dose. However, side effects were common and sometimes severe after whole-cell pertussis vaccine. Concern about convulsions and hypotonic hyporesponsive episodes (1 per 1,750 doses), acute encephalopathy (0-10.5 cases per million doses), and even the possibility of permanent neurologic damage spurred the development of more purified (acellular) pertussis vaccines (combined with diphtheria and tetanus toxoids as DTaP) and licensure in the 1990s and 2000s.43 The adverse event profile of acellular pertussis vaccines has been found to be similar to placebo.44
Parent and health care worker confidence in the acellular pertussis vaccine along with much emphasis on improving immunization coverage led to high four-dose DTaP coverage among children aged 19-25 months—almost 85% in 2003,45 but children who are unvaccinated or incompletely vaccinated remain vulnerable. For one of the vaccines, efficacy against World Health Organization-defined pertussis (i.e., more severe disease) was shown to be about 84% after three doses.46 In a different study, 86% of children were protected up to 6 years after three doses.47
With the increase in reported cases of pertussis—particularly among adolescents—and the recognition that pertussis infection in this group is responsible for substantial morbidity and disease spread, there has been a corresponding increase in the desire for a pertussis vaccine for adolescents and adults. Germany, France, and Canada, to name a few countries, already have incorporated adolescent immunization into their routine pertussis vaccination schedules. In 2005 two combination vaccine boosters for older individuals were licensed containing diphtheria and tetanus toxiods along with acellular pertussis vaccine (DTaP). The CDC now has recommended DTaP for routine use in adolescents and subsequently may deliberate recommendations for vaccination of health care workers, parents of newborns,49 and other adults.
Adverse reactions to DTaP include:
• Local reactions—Administration of DTaP causes pain, redness, or swelling at the injection site. Local reactions have been reported frequently after the first three doses of pertussis-containing vaccines.
• Limb swelling—Unexplained swelling of the entire thigh or upper arm has been reported after DTaP booster doses.50 There may be erythema and pain, but painless swelling that does not limit activity also has been reported. Limb swelling resolves without treatment or sequelae and is not considered a contraindication to boosters. Emergency medicine physicians may be called upon to examine children with extensive swelling reactions and may be tempted to incorrectly diagnose and treat these as cellulitis.48
• Mild systemic reactions—Fever, drowsiness, and fretfulness may follow vaccination, but usually are self-limited. Acetaminophen and/or ibuprofen are useful symptomatic treatment of these.
• Moderate or severe systemic events—High fever, febrile seizures, persistent crying for three or more hours, and hypotonic hyporesponsive episodes rarely have been reported after DTaP.
Emergency physicians should be aware that individuals may be susceptible to pertussis for a host of reasons including that the pertussis vaccine may have been withheld from children with contraindications to the vaccine. (See Table 2.) Of course, some children are under-vaccinated because of missed visits, parental refusal, or office errors. Additionally, some vaccinated children remain susceptible because of mishandling of the vaccine, improper vaccine administration, or vaccine failure. As mentioned earlier, vaccine-induced immunity may last 5-10 years.

After exposure to pertussis, all close contacts younger than 7 years of age who have not completed a four-dose series should complete the series with the minimal intervals between doses. Similarly, after exposure those who have completed a primary series but have not received a pertussis vaccine within three years of exposure should be given a booster dose.16
Management
Supportive Treatment. Pertussis is treated supportively with supplemental oxygen as well as intravenous fluids and nutrition as needed. Hospitalization may be needed for pulmonary toilet and monitoring. Infants often require hospitalization for supportive care to manage apnea. Extracorporeal life support has been offered to infants with pertussis and respiratory failure refractory to mechanical ventilation.51
Antibiotics. Antibiotics only modify the illness’ course if early in the course of disease symptoms. During the paroxysmal phase of the disease, eradication of the bacteria by antibiotics may not significantly change the clinical course for the patient, but is warranted to limit spread—even up to 8 weeks after disease onset if the patient has contact with high-risk persons. Erythromycin has been listed as the drug of choice for pertussis, but courses of erythromycin often are discontinued because of nausea or diarrhea.52 The standard treatment course is 14 days (40-50 mg/kg/d in 4 divided doses, maximum 2 g/d), although a 7-day course may be equally effective.53 Erythromycin estolate achieves higher concentrations in respiratory tract secretions, and should be used in preference to erythromycin ethylsuccinate when possible.33 In comparison with erythromycin, azithromycin (10-12 mg/kg/d in a single dose, maximum 600 mg/d, for 5 days) and clarithromycin (15-20 mg/kg/d in 2 divided doses, maximum 1 g/d, for 7 days) have similar efficacy, fewer side effects, and a more patient-friendly schedule.53 Trimethoprim-sulfamethoxazole is another acceptable option. Penicillins as well as first- and second-generation cephalosporins are not effective against pertussis. Resistance to erythromycin or the macrolides by B. pertussis has been reported rarely.
Infants younger than 2 weeks of age who receive oral erythromycin may be at increased risk for hypertrophic pyloric stenosis (IHPS). Because of the severity of pertussis in this age group55,56 and because the risk of IHPS after use of macrolides is not known, erythromycin remains the drug of choice for newborns for prophylaxis and treatment of B. pertussis, though parents should be informed about potential risks and signs of IHPS.33
Investigational Treatment. Pertussis-specific Immune Globulin is an investigational product not licensed for use. Conclusive studies on the value of corticosteroids and beta 2-agonists (e.g., albuterol) to control pertussis symptoms are not available.57 A Cochrane review of existing studies concluded that use of dexamethasone did not show a clear decrease in hospital stay, and salbutamol showed no change in the frequency of coughing paroxysms.58
Conclusions
Misconceptions that pertussis disease is rare or inconsequential beyond childhood and that it is completely controlled by early childhood immunization have been common among health care workers. This has contributed to low levels of suspicion with consequent infrequent testing. Inherent difficulties in culturing for this fastidious organism led to many false negatives, contributing to physician indifference to pertussis infection. Aggressive surveillance and newer modes of testing are pointing to the presence of more circulating pertussis than previously was recognized. Studies of the distribution and effects of the disease are spurring new interest in protecting adolescents and adults to limit their own morbidity and their contribution to disease spread to vulnerable infants. Two new tetanus-diphtheria-acellular pertussis vaccines licensed for older age groups make prevention possible in a way it previously was not. Active surveillance in coming years will help judge the effects of the increased preventive efforts in the second half of this decade.
References
1. Cherry JD. The epidemiology of pertussis and pertussis immunization in the United Kingdom and the United States: A comparative study. Curr Probl Pediatr 1984;14:1-78.
2. Centers for Disease Control and Prevention. Notice to readers: Final 2003 reports of notifiable diseases. MMWR Morb Mortal Wkly Rep 2004;53:687-697.
3. Ntezayabo, DeSerres G, Duval B. Pertussis resurgence in Canada largely caused by a cohort effect. Pediatr Infect Dis J 2003;22:22-27.
4. DeSerres G, Shadmani R, Duval B, et al. Morbidity of pertussis in adolescents and adults. J Infect Dis 2000;182: 174-179.
5. Product Approval Information—Licensing Action (Boostrix). Available at: http://www.fda.gov/cber/products/ tdapgla050305.htm. Accessed July 20, 2005.
6. Product Approval Information—Licensing Action (Adacel). Available at: http://www.fda.gov/cber/products/tdapave061005.htm. Accessed July 20, 2005.
7. Wirsing von Konig CH, Halperin S, Riffelmann M, et al. Pertussis of adults and infants. Lancet Infect Dis 2002 Dec; 2:744-750.
8. Bisgard KM, Pascual FB, Ehresmann KR, et al. Infant pertussis—Who was the source? Pediatr Infect Dis J 2004;23: 985-989.
9. Centers for Disease Control and Prevention. Pertussis deaths—United States, 2000. MMWR Morb Mortal Wkly Rep 2002;51:616.
10. Purdy KW, Hay JW, Botteman MF, et al. Evaluation of strategies for use of acellular pertussis vaccine in adolescents and adults: A cost-benefit analysis. Clin Infect Dis 2004;39: 20-28.
11. Halperin SA. Canadian experience with implementation of an acellular pertussis vaccine booster-dose program in adolescents: Implications for the United States. Pediatr Infect Dis J 2005;24(6 Suppl):S141-146.
12. Wharton M. Prevention of pertussis among adolescents by vaccination: Taking action on what we know and acknowledging what we do not know. Clin Infect Dis 2004;39:29-30.
13. ACIP Recommends Adolescent Vaccination for Tetanus, Diphtheria, and Pertussis Vaccine. Available at: http://www.cdc.gov/nip/pr/pr_tdap_jun2005.htm. Accessed July 20, 2005.
14. Hewlett EL, Edwards KM. Pertussis—Not just for kids. N Engl J Med 2005;352:1215-1222.
15. Cherry JD. The epidemiology of pertussis: A comparison of the epidemiology of the disease pertussis with the epidemiology of Bordetella pertussis infection. Pediatrics 2005;115: 1422-1427.
16. Epidemiology and Prevention of Vaccine-Preventable Diseases. 8th edition. Chapter 7—Pertussis. Available at: http://www.cdc.gov/nip/pr/pr_tdap_jun2005.htm. Accessed July 20, 2005.
17. Strebel P, Nordin J, Edwards K, et al. Population-based incidence of pertussis among adolescents and adults, Minnesota, 1995-1996. J Infect Dis 2001;183:1353-1359.
18. Yih WK, Lett SM, des Vignes FN, et al. The increasing incidence of pertussis in Massachusetts adolescents and adults, 1989-1998. J Infect Dis 2000;182:1409-1416.
19. Tanaka M, Vitek C, Pascual FB, et al. Trends in pertussis among infants in the United States, 1980-1999. JAMA 2003;290:2969-2975.
20. Addiss DG, Davis JP, Meade BD, et al. A pertussis outbreak in a Wisconsin nursing home. J Infect Dis 1991;164:704-710.
21. Christie CD, Marx ML, Daniels JA, et al. Pertussis containment in schools and day care centers during the Cincinnati epidemic of 1993. Am J Publ Health 1997;87:460-462.
2. Sprauer MA, Cochi SL, Zell, et al. Prevention of secondary transmission of pertussis in households with early use of erythromycin. Am J Dis Child 1992;146;177-181.
23. Steketee RW, Wassilak SG, Adkins WN Jr, et al. Evidence for a high attack rate and efficacy of erythromycin prophylaxis in a pertussis outbreak in a facility for the developmentally disabled. J Infect Dis 1988;157:434-440.
24. Tanaka Y, Fujinaga K, Goto A, et al. Outbreak of pertussis in a residential facility for handicapped people. Dev Biol Stand 1991;73:329-332.
25. Centers for Disease Control and Prevention. School-associated pertussis outbreak—Yavapai County, Arizona, September 2002—February 2003. MMWR Morb Mortal Wkly Rep 2004:53:216-219.
26. Crowcroft NS, Stein C, Duclos P, et al. How best to estimate the global burden of pertussis? Lancet Infect Dis 2003;3: 413-418.
27. Vitek CR, Pascual FB, Baughman AL, et al. Increase in deaths from pertussis among young infants in the United States in the 1990s. Pediatr Infect Dis J 2003;22:628-634.
28. Mikelova LK, Halperin SA, Scheifele D, et al. Predictors of death in infants hospitalized with pertussis: A case-control study of 16 pertussis deaths in Canada. J Pediatr 2003;143: 576-581.
29. Centers for Disease Control and Prevention. Outbreaks of pertussis associated with hospitals—Kentucky, Pennsylvania, and Oregon, 2003. MMWR Morb Mortal Wkly Rep 2005; 54:67-71.
30. World Health Organization. Immunization, Vaccines, and Biologicals—Pertussis vaccine. The pathogen. Available at: http://www.who.int/vaccines/en/pertussis.shtml. Accessed July 20, 2005.
31. Ghaffar A. Bordetella and Haemophilus. Available at http://pathmicro.med.sc.edu/2003-bacpdf/03BPERT.pdf. Accessed July 20, 2005.
32. Todar’s Online Textbook of Bacteriology. Bordetella pertussis and Whooping Cough. Available at: http://textbookofbacteriology.net/pertussis.html. Accessed July 20, 2005.
33. American Academy of Pediatrics. Pertussis. In: Pickering LK, ed. Red Book: 2003 Report of the Committee on Infectious Diseases. 26th ed. Elk Grove Village, IL: American Academy of Pediatrics, 2003:472-486.
34. Preston A, Maskell DJ. A new era of research into Bordetella pertussis pathogenesis. J Infect 2002;44:13-16.
35. Lee GM, Lett S, Schauer S, et al; Massachusetts Pertussis Study Group. Societal costs and morbidity of pertussis in adolescents and adults. Clin Infect Dis 2004;39: 1572-1580.
36. Centers for Disease Control and Prevention. Pertussis—United States, 1997-2000. MMWR Morb Mortal Wkly Rep 2002; 51:73-76.
37. Wendelboe AM, Van Rie A, Salmaso S, et al. Duration of immunity against pertussis after natural infection or vaccination. Pediatr Infect Dis J 2005;24(5 Suppl):S58-61.
38. Tozzi AE, Ravà L, Ciofi degli Atti ML, et al. Clinical presentation of pertussis in unvaccinated and vaccinated children in the first 6 years of life. Pediatrics 2003;112:1069-1075.
39. Senzilet LD, Halperin SA, Spika JS, et al, for the Sentinel Health Unit Surveillance System Pertussis Working Group. Pertussis is a frequent cause of prolonged cough illness in adults and adolescents. Clin Infect Dis 2001;32:1691-1697.
40. Fung KS, Yeung WL, Wong TW, et al. Pertussis—a re-emerging infection? J Infect 2004;48;145-148.
41. Pediatrics, Pertussis. Available at: http://www.emedicine.com/emerg/topic394.htm. Accessed July 20, 2005.
42. Lee LH, Pichichero M. Costs of illness due to Bordetella pertussis in families. Arch Fam Med 2000;9:989-996.
43. LeSaux N, Barrowman NJ, Moore DL, et al. Decrease in hospital admissions for febrile seizures and reports of hypotonic-hyporesponsive episodes presenting to hospital emergency departments since switching to acellular pertussis vaccine in Canada: A report from IMPACT. Pediatrics 2003; 112:e348. Available at: http://www.pediatrics.org/cgi/content/full/112/5/e348. Accessed July 20, 2005.
44. Jefferson T, Rudin M, DiPietrantonj C. Systematic review of the effects of pertussis vaccines in children. Vaccine 2003;21: 2012-2023.
45. Centers for Disease Control and Prevention. National, state, and urban area vaccination coverage among children aged 19-35 months —United States, 2003. MMWR Morb Mortal Wkly Rep 2004;53:658-661.
46. Greco D, Salmaso S, Mastrantonio P, et al. A controlled trial of two acellular vaccines and one whole-cell vaccine against pertussis. N Engl J Med 1996;334:341-348.
47. Salmaso S, Mastrantonio P, Tozzi AE, et al. Sustained efficacy during the first six years of life of 3-component acellular pertussis vaccines administered in infancy: The Italian experience. Pediatrics 2001;108:e81.
48. Rennels MB. Extensive swelling reactions occurring after booster doses of diphtheria-tetanus-acellular pertussis vaccines. Semin Pediatr Infect Dis 2003;14:196-198.
49. Scuffham PA, McIntyre PB. Pertussis vaccination strategies for neonates—an exploratory cost-effectiveness analysis. Vaccine 2004;22:2953-2964.
50. Gold MS, Noonan S, Osbourn M, et al. Local reactions after the fourth dose of acellular pertussis vaccine in South Australia. Med J Aust 2003;179:191-194.
51. Pooboni S, Roberts N, Westrope C, et al. Extracorporeal life support in pertussis. Pediatr Pulmonol 2003 Oct;36:310-315.
52. Purcell BK, Dooley DP, Gray PJ, et al. Experience with directly observed prophylaxis using erythromycin in military trainees exposed to pertussis. Mil Med 2004;169:417-420.
53. von Konig CH. Use of antibiotics in the prevention and treatment of pertussis. Pediatr Infect Dis J 2005;24(5 Suppl):S66-68.
54. Altunaiji S, Kukuruzovic R, Curtis N, et al. Antibiotics for whooping cough (pertussis). Cochrane Database Syst Rev 2005;(1):CD004404.
55. Colletti JE, Homme JL, Woodridge DP. Unsuspected neonatal killers in emergency medicine. Emerg Med Clin North Am 2004;22:929-960.
56. Crowcroft NS, Booy R, Harrison T, et al. Severe and unrecognised: pertussis in UK infants. Arch Dis Child 2003; 88:802-806.
57. Kerr JR, Preston NW. Current pharmacotherapy of pertussis. Expert Opin Pharmacother 2001;2:1275-1282.
58. Pillay V, Swingler G. Symptomatic treatment of the cough in whooping cough. Cochrane Database Syst Rev 2003;(4):CD003257.
Pertussis, or whooping cough, was first described in the 16th century, and the causative agent was isolated in 1906. Nonetheless, pertussis remained a major cause of morbidity and mortality among ch ildren well into the 20th century.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
