ACE-Inhibitor Angioedema: How Common? How Serious?
Abstract & Commentary
Source: Sondhi D, et al. Airway compromise due to angiotensin-converting enzyme inhibitor-induced angioedema: Clinical experience at a large community teaching hospital. Chest. 2004;126:400-404.
In this retrospective study from a community teaching hospital in Philadelphia, Sondhi and colleagues identified all patients whose admissions had been assigned the diagnosis code for angioedema (AE) during the five academic years starting in 1996. They reviewed these patients’ medical records to determine the presenting symptoms and signs, treatments and outcomes, as well as whether the patients were taking angiotension-converting enzyme inhibitors (ACEIs) when they developed symptoms. All patients on ACEIs were assumed to have ACEI-induced AE. During the five-year study period, 70 patients were admitted with AE, among whom this was considered related to ACEI therapy in 45 (64%). In the other 25 patients, AE was ascribed to food allergy, antibiotics, or infection in 23. No precipitating factor could be identified in two cases.
Of the 45 patients with ACEI-related AE, the mean age was 62 years and 41 (91%) were African-American. Twenty-nine (67%) were women. No specific ACEI appeared more likely to cause AE than the others. Duration of ACEI use prior to presentation ranged from 1 day to 5 years; 21 of 45 (45%) patients presented within two months of starting the drug, while 11 (24 patients) had been on it for six months to five years.
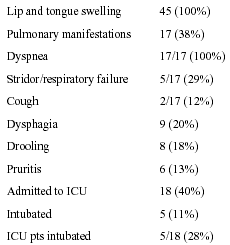
Presenting symptoms and signs are shown in the Table. All patients were treated in the ED with H1 blockers, methylprednisolone, and intravenous fluids, and 15 of them received epinephrine. Eighteen patients (40%) were admitted to the ICU, and five (28% of those requiring intensive care) required intubation, one of whom underwent emergency cricothyrotomy. Mean duration of intubation in these patients was 2.2 days, and patients admitted to the ICU spent an average of 2.2 days there. Care in the ICU was supportive. There were no deaths or serious complications in the study cohort.
Table.
Findings and Interventions
Among
45 Patients
Presenting with ACE-
Inhibitor-Related Angioedema
Commentary by David J. Pierson, MD
Angioedema produces asymmetrical, non-pitting edema of the skin and mucus membranes. Typically presenting with facial swelling, AE may progress to involve the tongue and supraglottic soft tissues, and fatal upper airway obstruction can ensue. Although it often is associated with allergy to foods (e.g., nuts or shellfish), stings by insects of the genus Hymenoptera, or administration of other medications (e.g., antibiotics), AE is increasingly encountered in patients taking ACEIs. It has been estimated that 0.1% to 0.2% of patients receiving ACEIs develop AE. With more than 40 million people taking these agents in the United States today, this seemingly low incidence of a potentially life-threatening complication is important to clinicians working in EDs and ICUs.
The present study has a number of limitations. As with all retrospective studies, the true incidence of the entity being investigated cannot be determined. Cases of AE may have been miscoded or misdiagnosed. The association of AE with ACEI use may have been missed, and patients taking ACEIs may have had AE caused by something else. Clinicians should be cautious in generalizing these findings to other practice settings. For example, the finding that 91% of the patients in this series were African-American has uncertain implications; African-Americans compose 64% of inpatient admissions to the study hospital.
These cautions notwithstanding, this study is important in reminding us of a potentially life-threatening, but entirely treatable, complication whose frequency is substantial: At the study’s 600-bed hospital, an average of nine patients with ACEI-induced AE were admitted each year during the study period.
In this retrospective study from a community teaching hospital in Philadelphia, Sondhi and colleagues identified all patients whose admissions had been assigned the diagnosis code for angioedema during the five academic years starting in 1996.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
