Drug Criteria and Outcomes: Duloxetine (Cymbalta) Formulary Evaluation
Drug Criteria and Outcomes
Duloxetine (Cymbalta) Formulary Evaluation
Part 1 of 2: Mechanism of Action, Pharmacokinetics, Indication, Dosing, Special Populations, Safety, and Cost
By Andrea Pierce,
PharmD Candidate
McWhorter School of Pharmacy
Samford University, Birmingham, AL
Background
The U.S. Food and Drug Administration (FDA) approved duloxetine (Cymbalta), marketed by Eli Lilly & Co., on Aug. 3, 2004, for the treatment of major depressive disorder (MDD) in adults. Duloxetine is a serotonin-norepinephrine reuptake inhibitor. It not only treats the depressive symptoms, but also reduces the painful physical symptoms of depression (e.g., back pain, headaches, neck pain, etc). Duloxetine was the first drug to receive FDA approval to specifically treat the pain associated with diabetic peripheral neuropathy. It is also being investigated for the treatment of stress urinary incontinence. It already is being marketed in Europe to treat incontinence under the name Yentreve, and the FDA currently is evaluating its efficacy for that indication.
Venlafaxine (Effexor) was chosen for comparison purposes in this review as a representative antidepressant agent having similar pharmacology as duloxetine.
Mechanism of action
Duloxetine is a reuptake inhibitor of both serotonin and norepinephrine. It belongs to the same drug class as venlafaxine, even though structurally it resembles fluoxetine (Prozac). Like venlafaxine, duloxetine is a stronger inhibitor of serotonin reuptake than norepinephrine reuptake. It has no effect on the dopaminergic, histaminergic, muscarinic, alpha 1 adrenergic receptors, 5-HT1A, 5-HT1B, 5-HT1D, 5-HT2A, 5-HT2C, opioid receptors, or monoamine oxidase (MAO). In stress incontinence, duloxetine inhibits the serotonin and the norepinephrine reuptake, causing the detrusor muscle and the urethral sphincter to increase activity, leading to improved bladder control.
Pharmacokinetics
Absorption
- Duloxetine is an oral agent that appears to have a 70% bioavailability based on urinary excretion. For regular-release venlafaxine, 92% is absorbed, but only 12.6% is available to the systemic circulation due to first-pass metabolism. The extended-release venlafaxine is approximately 45% available to the systemic circulation.
- Duloxetine has a two-hour delay after oral administration before absorption begins due to its enteric coating, with the peak concentration occurring six hours after dosing. Peak concentration is achieved in one to two hours with the immediate-release venlafaxine tablets and in about 5.5 hours with the extended-release capsules.
- If given in the evening, absorption of duloxetine occurs within three hours after oral administration, and clearance is increased 30% compared to morning administration.
- When duloxetine is given with food, the time to peak concentration is delayed to six to 10 hours, but it does not affect the peak concentration. With venlafaxine, time of day and food have no effect on absorption. Venlafaxine should be given with food to decrease gastrointestinal adverse effects.
Distribution:
- Duloxetine has greater than 90% protein binding, while venlafaxine has 30% protein binding. Therefore, there is a higher risk of drug interaction with highly bound agents with duloxetine vs. venlafaxine.
- Both are widely distributed throughout the body.
- Steady state is achieved after three days dosing with both duloxetine and venlafaxine.
- Duloxetine has a volume of distribution (Vd) of 1640 L, while venlafaxine’s steady-state Vd is 7.5 ± 3.7 L/hour/kg.
Metabolism
- Duloxetine undergoes hepatic metabolism through the CYP1A2 and CYP2D6. Venlafaxine also is metabolized mainly by the CYP2D6 and undergoes first-pass metabolism to its main active metabolite O-desmethylvenlafaxine.
- Duloxetine has a half-life of approximately 12 hours (ranging from 8 to 17 hours). Venlafaxine has a half-life of five hours with the metabolite having a half-life of 11 hours.
Excretion
- Duloxetine — 70% excreted as metabolites in the urine and 20% in the feces.
- Venlafaxine — 36%-60% excreted through the urine.
- Duloxetine has a clearance of 114 L/hour, while venlafaxine has a mean ± SD steady-state plasma clearance of venlafaxine is 1.3 ± 0.6 L/hour/kg.
Onset of action
- Response to therapy was seen in three weeks in patients treated with duloxetine, while venlafaxine patients showed a response in 2-3 weeks.
Indications
Duloxetine is indicated for the treatment of depression in adults. It also is indicated to treat pain associated with diabetic neuropathy. Studies are currently under way to determine its efficacy in the treatment of stress urinary incontinence.
Venlafaxine is approved to treat anxiety, depression, and social phobia. Unlabeled uses include diabetic neuropathy, neuropathic pain, menopausal symptoms, fibromyalgia, headaches, and premenstrual dysphoric disorder.
Dosing
Duloxetine:
- Duloxetine is available as oral capsules in the strengths of 20 mg, 30 mg, and 60 mg.
- Depression: Initially 40-60 mg per day as a single dose or in two divided doses. The maximum dose has been determined to be 120 mg, but no increase in efficacy is seen with the higher dose.
- Diabetic neuropathy: 60 mg given once a day; 120 mg per day has been used, but no increase in efficacy was noted.
Venlafaxine:
- Venlafaxine is available in immediate-release (IR) tablets (25 mg, 37.5 mg, 50 mg, 75 mg, 100 mg) and extended-release (XR) capsules (37.5 mg, 75 mg, 150 mg).
- Depression: Venlafaxine IR — 75 mg/day in 2-3 divided doses and may be increased by 75 mg/day every 4 days to a maximum of 225 mg/day. XR — Initially 75 mg per day; can begin at a dose of 37.5 mg/day and increase to 75 mg/day in 4-7 days. Increase dose by 75 mg every 4 days to goal dose, not to exceed 225 mg day.
- Both agents can be dosed daily.
Special populations
Duloxetine should be avoided in patients with hepatic failure. In patients with mild-to-moderate renal failure, no dosing adjustment is needed. It is recommended to avoid use of duloxetine in patients with severe renal impairment or end-stage renal disease.
The dose of venlafaxine should be decreased 50% in patients with moderate hepatic impairment. In patients with renal impairment, the dose is individualized based on creatinine clearance.
Administration of duloxetine and venlafaxine
- Swallow capsules whole.
- Do not cut, chew, or crush capsules (duloxetine capsules or extended-release venlafaxine products).
Nausea, dry mouth, sweating, weight loss, and gastrointestinal problems occurred more frequently with both drugs compared to placebo (see Table 1). Nausea was more prevalent during the first week of therapy with duloxetine. It can be decreased in patients by starting with the 30 mg per day capsules the first week and titrating to 60 mg per day by the end of the week. Venlafaxine XR had higher rates of dizziness, somnolence/insomnia, hypertension, sweating, abnormal dreams, and nervousness when compared to placebo than duloxetine. The risk of seizures is also higher in patients taking venlafaxine than in the duloxetine-treated patients. Both groups had sexual adverse effects, with venlafaxine XR having a substantially elevated rate of delayed ejaculation compared to placebo. However, caution must be used in comparing these values, as they are not derived from directly comparative trials.
Drug interactions
Duloxetine is metabolized by the CYP2D6 and CYP1A2 enzymes. There is a moderate risk of drug interactions with duloxetine because it is both a CYP2D6 substrate and an inhibitor there (see Table 2).
- Inhibitors of 1A2 — Duloxetine concentration is increased (e.g., fluvoxamine, cimetidine, quinolones).
- Inhibitors of 2D6 — Duloxetine concentration is increased (e.g., paroxetine, fluoxetine, quinidine).
- Use caution in drugs metabolized through the CYP2D6 enzyme that have a narrow therapeutic index.
- Do not use duloxetine and venlafaxine or other serotonin agents in combination with each other due to risk of serotonin syndrome.
- Do not use with monoamine oxidase inhibitors (MAOIs) or with drugs that have MAOI activity (procarbazine or linezolid). Wait at least two weeks after MAOI agent discontinued before starting either duloxetine or venlafaxine.
- Do not use with tricyclic antidepressants (TCAs).
- Due to its metabolism through the CYP2D6 enzymes, venlafaxine has similar drug interactions as duloxetine.
Pregnancy rating
- Both duloxetine and venlafaxine have a pregnancy rating of category C.
- Avoid use in lactation if possible with both agents.
Contraindications
Similar in both duloxetine and venlafaxine.
- Hypersensitivity to duloxetine/venlafaxine.
- Use with MAOIs.
- Duloxetine is contraindicated in patients with narrow angle glaucoma due to increased risk of mydriasis.
Storage
Capsules should be stored at 25°C (77°F) and away from sunlight. Excursions are permitted to 15°-30°C (59°-86°F).
Potential for medication error
There is a potential for duloxetine and venlafaxine to be confused, especially during verbal communication. Another possible error could occur if duloxetine and fluoxetine were confused with each other, especially with written orders because they look alike and also are available in similar strengths. Educating the staff on the potential of these errors occurring is key in maintaining patient safety.
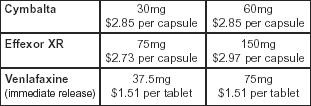
Cost
The cost of the duloxetine and the extended-release venlafaxine is similar (see Table 3, below). The immediate-release tablets are not as expensive, but more tablets per day will have to be taken to get the equivalent dose of the extended-release capsules, causing the cost to increase, and compliance may not be as good.
Patient information for duloxetine and venlafaxine
- If a dose is missed, take as soon as you remember and continue the next day with the regular dosing schedule. Do not double doses.
- Avoid alcohol.
- Drowsiness may occur, so use caution in activities that require alertness (driving, operating heavy machinery, etc.).
- Notify physician if you become pregnant or are breast-feeding.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
