Special Feature: Biological Determinism: Time for a Paradigm Shift in Cancer Therapeutics?
Special Feature
Biological Determinism: Time for a Paradigm Shift in Cancer Therapeutics?
By Robert L. Coleman, MD, Associate Professor, University of Texas; M.D. Anderson Cancer Center, Houston, is Associate Editor for OB/GYN Clinical Alert
In the November 20th issue of the Journal of Clinical Oncology, two important articles appear, demonstrating the clinical impact of a novel therapeutic, bevacizumab, in women with recurrent ovarian cancer.1,2 Bevacizumab is a fully humanized monoclonal antibody that targets vascular endothelial growth factor (VEGF)—a principle compound secreted by tissues (tumors) to gain access to new endogenous vasculature. The drug, often referred to as a "biologic" because of its primary focus on functional interruption of the tumor microenvironment, has been shown to be of significant merit in combination with chemotherapy in several solid tumors, such as colon, lung and breast cancer—each with substantive improvement in progression-free and, in the former two, in overall survival.3 The current articles represent seminal events for ovarian cancer therapeutics because they are the first two completed Phase II clinical studies in this disease site and demonstrate clinical efficacy as single agents. A summary of the two trials is presented in the Table below.
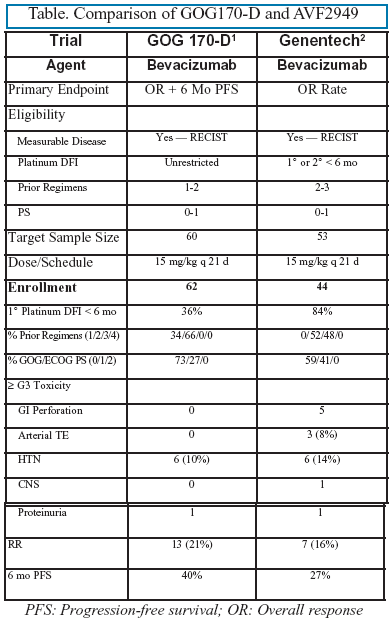
In the study conducted by the Gynecologic Oncology Group, 62 evaluable women with recurrent ovarian cancer (no more than 2 prior regimens) were given bevacizumab every three weeks until progression. Since it was anticipated that patients may experience "non-progression" as a reflection of the targeted therapy's effect, the design uniquely incorporated consideration of both objective response (traditional measures—RECIST) and percentage of patients being progression-free at 6 months in its statistical decision algorithm for efficacy. In all, clinical response was seen in 21%, including 2 patients remarkably achieving complete response; 40% were progression-free at 6 months. Severe treatment-related toxicity was uncommon. In contrast, the second trial, initiated by the pharmaceutical sponsor was conducted in a more heavily pretreated population (up to 3 prior regimens) and demonstrated sufficient toxicity, predominately gastrointestinal perforation, to warrant early termination. Overall response (primary endpoint) was 16%—all partial responses. While the same dose and schedule of bevacizumab was used in this study, the unanticipated toxicity and lack of easily identifiable risk factors reminds one of the importance of using investigational agents in the clinical trial setting. Currently, randomized clinical studies in the setting of primary and recurrent disease are underway and represent mature clinical development of this novel compound.
On the whole, validation of efficacy for this "non-cytotoxic" agent has encouraged discovery, and has ushered in an explosion of new compounds with new targets and/or combinations of targets. Many of these have or are now poised to enter the clinic. The expansion of targets and "targetables" also raise speculation (and optimism) of the next evolutionary iteration in cancer therapy: "biological determinism." While the term may assume various interpretations (eg, intrinsic biology defining outcome), what piques our imagination is the ability to accurately describe an individual patient's specific tumor biology and concocting a tumor-directed lethal cocktail for treatment. Customized or individualized therapy is not a new clinical concept; yet, the ubiquitous prevalence of "standard therapy" studies highlights how far the body of mature literature is from this endpoint. A comical surrogate marker of our progress could be prevalence of such treatment depicted in science fiction features; if customized therapy was commonplace, we wouldn't see it in Hollywood. Nonetheless, the catalogue of relevant targets is expanding and with it an arsenal of new compounds that may be of relevance for therapy. Our difficulty is matching the two.
On a superficial level, customization of therapy is part of our current strategic practice. For example, patients can be identified with low or reasonable probability of response to certain agents through clinical evaluation (prior lack of response or lack of exposure to agents within a certain class) or through in vitro testing. Several companies now offer chemoresistance or sensitivity testing in sampled tissues where several agents and/or combination of agents are tested in cell cultures of an individual's tumor. Unfortunately, validation of these assays as a predictive test for choosing the optimal drug at a specific time in therapy is very difficult and requires many thousands of patients.4 Prognostic scoring algorithms based on clinical and molecular data have now also become available and are currently being used to triage patients into different treatment programs (eg, chemotherapy vs hormonal therapy for breast cancer).
Much is known about the relevant biological pathways in human cancer. Certainly, that knowledge is only a fraction of what is truly needed for individualized cancer therapy. However, tumor biology has and is currently being leveraged in clinical trials. For example, P53, a housekeeping gene for many tumors, is frequently mutated in ovarian cancer. While the prognostic impact of P53 status on outcome is variably reported, the importance of functional P53 to cellular processing is substantial.5 In vitro and in vivo preclinical studies demonstrated that insertion of a normal P53 gene could signal tumor cell death in ovarian cancer models.5 These data paved the way for the development of a number of "gene therapy" trials where, among other strategies, normal P53 was inserted into cells by viral vector constructs delivered intraperitoneally.6,7 In fact, at one point, a randomized clinical trial of P53 gene therapy in combination with chemotherapy was enrolling patients after surgery with primary ovarian cancer. The trial did not meet it accrual goals—largely due to toxicity concerns. Still of therapeutic interest, the strategy has been revisited now with extensive work being done in vector technology.8
Treatment directed at the epidermal growth factor receptor (EGFR) is another example of tumor-specific targeted therapy. EGFR, the first biological target selected for therapy, is overexpressed in most solid tumors including ovarian cancer. Activation of the growth receptor occurs when two individual like receptors or related receptors bind (dimerize), signalling a number of downstream functions including cellular proliferation, survival, adhesion and migration. The activation can be through ligand dependent and/or independent mechanisms and is the focus of several agents including monoclonal antibodies, dimerization inhibitors and tyrosine kinase small molecule inhibitors (TKIs).9 Some of these have been very successful (trastuzumab, lapatinib) and others more variable. However, of great interest has been the recent recognition that certain individual characteristics of the target might actually predict in whom these agents may work. This was highlighted by the initial FDA accelerated approval and then retraction of approval of the agent gefitinib in patients with lung cancer. Initial work demonstrated that selected patients had dramatic responses to single-agent gefitinib; however the agent did not reach its survival endpoints in mature phase III investigation. Subsequent studies demonstrated a candidate reason for the mixed observation was due the presence of certain mutations in the binding site of the receptor. Patients (and cell lines) with these mutations were substantially more likely to respond to gefitinib than those without.10,11 Unfortunately, the presence of this factor is only a subset of the total lung cancer population but it draws light into how customization of therapy can be levied by understanding the key characteristics of a specific patient's tumor.
Globally, more than 360 biologically-based agents are in clinical development, many with specific biomarkers in the hopes of identifying in whom the agent works best. Putting these together in novel combinations and sequences is the true representation of biological determinism. However, tests to demonstrate which factors should be targeted (in vitro testing, tissue microarrays, gene profiling, etc) need validation, as does the pairing of identifiable targets with specific agents. In early September 2007, one "proof of concept" trial was presented, demonstrating "better than expected" outcomes in 4 of 7 refractory and heavily pretreated patients with solid tumors in which their individual tumors were profiled and matched with drugs targeting these pathways. The prospective allocation of agents (chemotherapeutics and biologics) was based on profiling computational analysis of the interaction of multiple targets and signalling pathways, and represents a first step in individualization of therapy. However, much more work in understanding which pathway/pathways is/are critical, can be targeted by specific-enough agents, can be administered with acceptable toxicity, and will genuinely identify the "Achilles heel" of an individual's tumor is needed before "biologic determinism" can be allocated more merit than just "pie in the sky."
References
- Burger RA, et al. Phase II trial of bevacizumab in persistent or recurrent epithelial ovarian cancer or primary peritoneal cancer: a Gynecologic Oncology Group Study. J Clin Oncol. 2007;25:5165-5171.
- Cannistra SA, et al: Phase II study of bevacizumab in patients with platinum-resistant ovarian cancer or peritoneal serous cancer. J Clin Oncol. 2007;25:5180-5186.
- Jain RK, et al: Lessons from phase III clinical trials on anti-VEGF therapy for cancer. Nat Clin Pract Oncol. 2006;3:24-40.
- Sargent DJ, et al. Clinical trial designs for predictive marker validation in cancer treatment trials. J Clin Oncol. 2005;23:2020-2027.
- Hogdall EV, et al. P53 mutations in tissue from Danish ovarian cancer patients From the Danish "MALOVA" ovarian cancer study. Gynecol Oncol. 2005
- Wolf JK, et al: A phase I study of Adp53 (INGN 201; ADVEXIN) for patients with platinum- and paclitaxel-resistant epithelial ovarian cancer. Gynecol Oncol. 2004;94:442-448.
- Von Gruenigen VE, et al. Efficacy of intraperitoneal adenovirus-mediated p53 gene therapy in ovarian cancer. Int J Gynecol Cancer. 1999;9:365-372.
- Tsuruta Y, et al. A mosaic fiber adenovirus serotype 5 vector containing reovirus sigma 1 and adenovirus serotype 3 knob fibers increases transduction in an ovarian cancer ex vivo system via a coxsackie and adenovirus receptor-independent pathway. Clin Cancer Res. 2007;13:2777-2783.
- Lo HW, Hung MC. Nuclear EGFR signalling network in cancers: linking EGFR pathway to cell cycle progression, nitric oxide pathway and patient survival. Br J Cancer. 96 Suppl:R16-20, 2007
- Lynch TJ, et al: Activating mutations in the epidermal growth factor receptor underlying responsiveness of non-small-cell lung cancer to gefitinib. N Engl J Med. 2004;350:2129-2139.
- Paez JG, et al. EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy. Science. 2004;304:1497-500.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
