Complications of Pregnancy: Part II
Complications of Pregnancy: Part II
Authors: Halleh Akbarnia, MD, FACEP, FAAEM, St. Francis Hospital, Evanston, IL; Teaching Faculty for Resurrection Emergency Department Residency Program, Evanston, IL; and Michelle Mendoza, MD, Chief Resident, Emergency Medicine Residency, Virginia Commonwealth University Health SciencesMedical College of Virginia Campus, Richmond, VA.
Peer Reviewers: Catherine A. Marco, MD, FACEP, Professor, Department of Surgery, Division of Emergency Medicine, University of Toledo College of Medicine, Toledo, OH; and Elaine Thallner, MD, MS, Department of Emergency Medicine, Cleveland Clinic, Cleveland, OH.
I hope you are enjoying this two-part series on complications of pregnancy. In my practice at a public hospital, I see many first and early second trimester pregnant patients, and I think I've gotten good at handling them. It is the last second and third trimester patients that get me nervous, for two reasons. First, pregnant women greater than 20 weeks go directly to the Labor and Delivery unit for their initial assessment, so I don't see that many. Secondly, I am now responsible for two patients: the mother and her potentially viable fetus. I hope this improves your ability to care for and assess these patients.
J. Stephan Stapczynski, MD, Editor
Hypertension in Pregnancy
One of the most common medical disorders affecting pregnancy is hypertension, complicating approximately 5-12% of all pregnancies.1,2 Even more disturbing, in the United States, 18% of maternal deaths can be attributed to this disease.3 Hypertension in pregnancy, a term that is commonly used to describe a wide range of patients with either mild elevation in blood pressure to severe elevations in blood pressure with end organ damage, can also pose potential problems in treatment protocols. Even among practitioners specializing in this disease, there is a wide definition of what hypertension is versus preeclampsia and beyond. According to the practice guidelines established by the Working Group on High Blood Pressure in Pregnancy, preeclampsia is most likely a syndrome, and because of that it is difficult to assign a set definition to the wide attributes of this disease. Although there are many articles that discuss the preeclamptic patient, most reviewed are case studies or small scale studies, and fewer are noted to be large sample size, retrospective reviews that would help establish the newer definitions of hypertension in pregnancy and preeclampsia.1
Normal blood pressure in pregnancy is defined as a systolic pressure of less than 140 and a diastolic pressure of less than 90. Having either number elevated is considered hypertension in the pregnant population. Ninety percent of the cases of hypertension in pregnancy are considered essential, or primary, rather than from a secondary cause. The three most common forms of hypertension are acute gestational hypertension, preeclampsia, and chronic essential hypertension. Eclampsia and hypertensive encephalopathy are severe states that need immediate attention and will be looked at as well.
Gestational hypertension is defined as hypertension in pregnancy that is above 140/90 and is not complicated by the signs and symptoms that may be associated with preeclampsia. This is the most frequent case of hypertension during pregnancy with incidences that range from 6-17% in nulliparous women and between 2-4% in multiparous women. Most cases develop at or after 37 weeks, which leaves the overall outcome of the pregnancy similar to those with normotensive pregnancies.2 However, in gestational hypertensive pregnancies, it has been shown that both birth weight and gestational age at delivery are higher than in the normotensive patient. In the same realm, women with mild gestational hypertension do have higher rates of cesarean sections due to higher rates of induction of labor when compared to their normotensive counterparts.4 The clinical and laboratory signs of patients with gestational hypertension are none other than having an elevated blood pressure. There is no documented proteinuria, no edema, and no laboratory abnormalities associated with it.5,6 Of note, morbidity of both the mother and the neonate are significantly increased in women who have more severe gestational hypertension. These women have even been shown to have higher morbidities than those with mild preeclampsia. Complications can be seen that may be detrimental to the fetus, including abruptio placentae, preterm delivery of less than 37 weeks, and infants that are small for gestational age. These complications are similar to those with severe preeclampsia, and therefore they should be treated as if they do have severe preeclampsia, including bedrest and early delivery if indicated.7
Preeclampsia is defined as hypertension equal or greater than 140/90 seen with proteinuria greater than 300 mg over a 24 hour period or persistent positive results (1+) on a protein dipstick. It can be mild, with a diastolic blood pressure of greater than 90 but less than 110 and not associated with any of the signs of severe preeclampsia, described later in this section. Although edema is still known to be a part of the classic triad of hypertension, proteinuria, and edema, there is now less emphasis given to that since edema tends to be a common finding in normal pregnancies and yet not all preeclamptic women will have edema. In fact, up to one-third of eclamptic women will never show edema, therefore it is now not a part of the newer definitions seen for preeclampsia.8,9
Preeclampsia is a leading cause of maternal mortality and morbidity worldwide, however the etiology is still unknown despite the numerous pathophysiologic abnormalities noted. Some factors that may contribute to preeclampsia include changes in the remodeling process of the uterine arterioles; many do not undergo the remodeling that is supposed to take place. Therefore, they remain thick leading to increasing vascular resistance and placental ischemia since there is decreased blood flow through those vessels. This can cause vasoactive molecules to be released, which act on the maternal vasculature. The changes that follow this lead to increased sensitivity of the maternal vascular endothelium causing more vasoconstriction and maternal hypertension.9,10 At this time also, there are no proven methods to prevent preeclampsia, but perinatal outcome can be improved with good management during pregnancy in that small group of women with severe symptoms less than 32 weeks. There are some risk factors that have been seen with preeclampsia, listed below (see Table 1), which include but are not limited to a younger or older age during pregnancy, obesity, nulliparity, and excessive weight gain. Typically, the onset of the disease occurs after 20 weeks.
 |
Many obstetricians would describe preeclampsia as gestational hypertension plus proteinuria. It would be safe to assume that any gravid patient who presents with hypertension and proteinuria has preeclampsia until proven otherwise. Proteinuria can be seen in the amount of 0.1 g/L or more in at least two random samples, or 300 mg (0.3 g) when collected in a 24-hour time frame. In those patients who do not have proteinuria, preeclampsia should still be considered if more severe symptoms are noted such as cerebral visual disturbances, nausea, vomiting and epigastric pain, oliguria of less than 400 mL in 24 hours, and impaired liver functions such as elevated liver enzymes, or decreased platelets or coagulopathies that cannot be explained.4,6 This brings patients into the more concerning category of severe preeclampsia, defined with a diastolic blood pressure greater than or equal to 110 on at least two occasions or after treatment with antihypertensive medications. The patient can also present with oliguria, pulmonary edema, thrombocytopenia, and severe proteinuria of greater or equal to 3.5 grams in 24 hours or two separate urine protein dipsticks positive at 3+. They may also have right upper quadrant pain and CNS symptoms including blurred vision, headaches, or changes in mental status. The practitioner should also be aware of the possibility of presentation with HELLP syndrome. HELLP syndrome is defined by hemolysis (which can be seen with an increase in bilirubin or LDH, or on a peripheral blood smear), elevated liver enzymes (aminotransferase greater than twice normal), and low platelets (less than 100,000). There have been no studies that can predict which category of women can or will progress into the more serious HELLP syndrome, or eclampsia. Even the mildest cases of hypertension or women with minimal proteinuria have been seen to progress on to very serious forms of this disease. Several studies have been done suggesting that preeclamptic patients with findings of only anuria and oliguria, or having increased headaches and hyperreflexia, were more prone to developing eclampsia than those who did not exhibit these symptoms.11
Chronic essential hypertension is defined arbitrarily as hypertension that can be present before the pregnancy occurs or if diagnosis is made before 20 weeks gestation. It is part of the differential diagnosis for preeclampsia, yet, at the same time, superimposed preeclampsia may be present. If the hypertension persists for more than 42 days postpartum, it would also be known as chronic hypertension.8 In those women who already have a diagnosis of chronic hypertension, diagnosing preeclampsia can be more of a challenge. This, again, can increase both morbidity and mortality for both the mother and the fetus if not diagnosed and monitored carefully. The diagnosis of superimposed preeclampsia with chronic hypertension can be made when there is newly documented proteinuria after 20 weeks of 500 mg in a 24-hour period. In those women with proteinuria before 20 weeks, the diagnosis of superimposed preeclampsia is made along with other abnormalities such as thrombocytopenia, or elevated liver enzymes, or more severe symptoms that did not exist prior to the pregnancy.6,8,12
The definition of eclampsia includes the occurrence of seizure or coma along with hypertensive disease in pregnancy. Although most pregnant women who present with seizure will be eclamptic, other etiologies must be excluded such as previous seizure disorders, brain tumors, HIV disease, infective causes such as meningitis, substance abuse, or recent travel. It is also important to note that seizure can be seen postpartum for up to two weeks, and eclampsia must be first on the differential in those patients.
Another hypertensive entity that can be seen in pregnant females is hypertensive encephalopathy, which may also present with coma or, rarely, seizures. One should always think eclampsia first when deciding if the patient has encephalopathy versus eclampsiaeclampsia is far more common, occurring approximately 80 times more often than encephalopathy.11 Physical examination also can be helpful. Fundoscopic examination can reveal the classic hypertensive crisis findings of papilledema and retinal hemorrhages, which are rarely seen in eclampsia.2
Treatment. Seizure Control. It is well documented that magnesium sulfate is the pharmacologic agent of choice in the management of seizures in eclampsia, although the mechanism is not as well known. It is thought that, in preeclampsia, there is a deficiency of endothelial prostacyclin which is a potent vasodilator, and that magnesium increases the release of this dilator. The theory is that seizures in preeclampsia are therefore due to cerebral ischemia from the constriction of the vessels lacking the endothelial prostacyclin and that magnesium will help dilate the cerebral arteries, thus leading to increased perfusion and, therefore, fewer seizures.13 Multiple studies have been done that support this drug as being superior to any other anticonvulsant, specifically phenytoin and diazepam. Also, lower risks of recurrence of seizures have been documented in those treated with magnesium. The use of magnesium in severe preeclampsia for prevention has been well documented in the literature; however, the evidence is not as supportive for mild preeclamptic patients, although current practice is to not treat these groups of patients any differently and magnesium is administered to both.14 This is because, as mentioned before, there are no known criteria or clinical findings that will determine which of these patients will progress into more severe forms of this disease.15 Of note, if a patient is seizing, and at least two doses of magnesium have been given, benzodiazepines and phenytoin or fosphenytoin should be considered as well. Diazepam can be given at increments of 5 mg, and phenytoin or fosphenytoin can be given at 10-20 mg/kg. The advantage of giving fosphenytoin, although much more costly than phenytoin, is the speed and methods in which it can be given. Phenytoin is given at a rate of 50 mg/min, but fosphenytoin can be given at 150 mg/min and could even be injected if the patient did not have any access.
Magnesium sulfate (20% solution) typically is given during the entire labor and delivery process up to 24 hours postpartum for prevention of seizures. It is given as an initial bolus of 4-6 grams IV over 20 minutes and then maintained at a rate of 2 grams per hour IV. Frequent checks should be made to make sure patients do not develop magnesium toxicity, and those with renal disease should have their levels checked every 2-4 hours to make sure the patients do not exceed the goal of 5-8 mg/dL. In all patients, frequent checks should be made since magnesium can lead to respiratory depression, loss of DTR, and a decrease in urinary output. Once any of these symptoms are evident, magnesium levels should be checked and measures taken to prevent toxicity as well. Patients on magnesium drips will also report feelings of intense heat and "hot flashes," which can be considered normal. If a patient is deemed magnesium toxic, giving her 10 mL IV of 10% calcium gluconate should reverse the symptoms.2,14,16 (See Table 2.)
 |
Hypertension Control. Intravenous hydralazine and labetalol have been well documented in the literature as the primary treatment indicated for severe hypertension in pregnancy, which is a blood pressure with a diastolic over 110. It has not been well documented or supported in previous literature that treatment of the hypertension will lead to decreased maternal or neonatal morbidity and mortality, or even decreases in progression to more severe forms of the hypertensive disease. Despite that, for patients with severe hypertension, treatment is indicated. Options include hydralazine, labetalol, nifedipine and, on the most rare occasions, sodium nitroprusside. Two different very recent studies , including one of more than 200 women, documented that neither drug is superior to the other. Hydralazine was given initially as a slow bolus at 5 mg and repeated every 20 minutes until a maximum dose of 120 mg was reached. Labetalol was given initially at 20 mg and repeated in 20 minutes at 40 mg followed by 80 mg every 20 minutes until the maximum dose of 400 mg was reached. No significant differences were noted for maternal hypotension and persistent hypertension. Although there was some hypotension and bradycardia noted in the neonates from the labetalol group, it was not statistically significant, and the rates of neonatal mortality were the same.16,17 Previous to this large study, hydralazine was thought to be superior to labetalol for first-line therapy. Nifedipine also has been studied as another possible first-line drug. In 2002, a meta-analysis was done involving 21 different trials and more than 800 patients comparing nifedipine to labetalol in some groups and nifedipine to hydralazine in others. It was shown in this paper that more research was needed but that hydralazine was most likely not first-line treatment for hypertension in pregnancy. No significant difference was noted between nifedipine and labetalol, and more trials need to be done comparing the two.18 The goal of treatment, regardless of agent used, is to get the blood pressure to a stable state and not fully normalized for risk of possibly decreasing perfusion to the placenta. This is why hypotension must be fully avoided as well.
It is now documented more clearly that corticosteroids can help in patients who have more severe complications such as HELLP syndrome and in treating preeclamptic patients by increasing platelet count, AST, and LDH. Also, urine output, blood pressure control, and improving lung function in the unborn fetus are all added benefits. Recall that intravenous fluids should be used sparingly since these patients tend to retain fluid.2
Amniotic Fluid Embolus
A potentially deadly syndrome that may be seen during pregnancy or during the immediate postpartum period is the release of amniotic fluid into the circulation of the mother during some form of manipulation of the uterus, including intense contractions. Amniotic fluid embolus (AFE) could also occur at areas of placental separation from the uterine deciduas basalis during placental abruption.
Incidence. There is a huge variation reported in the incidence of amniotic fluid embolus. Two recent studies on large populations of women demonstrate the rate of amniotic fluid embolus to be 6 and 14.8 out of 100,000 in primagravid and multigravid pregnancies respectively, with mortality reported as low as 26% and 13%.19,20 However, there are a lot of discrepancies in this number, and multiple other studies have reported much higher rates of mortality, including the well defined United States national registry which examined 46 cases of AFE during a 5-year span and found a 61% mortality (10% of all maternal deaths) with permanent neurological deficits in up to 85% of survivors.21 Although the national registry did not find any demographic risk factors associated with AFE, it did find that 70% of cases happened during the act of labor and 11% immediately after vaginal birth, along with 19% (and reports as high as 60%) of cases occurring during cesarean sections.21,22 There have been reports of increases of AFE found in multiparous women (mean 2.6), those who sustained trauma, and cesarean sections. Increased maternal age (mean of 33), gestational age, a male fetus, and the use of oxytocin have also been suggested as factors, however the support for these theories is minimal and inconsistent.19,23-25
Pathogenesis. The etiology and mechanism for how these emboli occur is still poorly understood. Some early studies were done on post mortem women with unexplained death showing amniotic tissue in the lungs. Another theory describes amniotic debris propelled into the vasculature after being driven by a pressure of electrochemical gradient by way of lacerations and plugging of the vessels, noted in the lower uterine segment, endocervical vessels as well as the placental site. Of note, amniotic fluid has also been found in women who did not show evidence or symptoms of AFE.24 Because some have noted that there are close similarities between the features of AFE and anaphylactic and septic shock, it was suggested at one time to change the name to "anaphylactic syndrome of pregnancy."21 As an alternative theory, the possibility of anaphylaxis could occur with massive mast cell degranulation independent of antigen-antibody mediated classic anaphylaxis.23 Half of the cases of mortality occur during the first two hours when there is vasospasm and mechanical plugging of the vessels as described above as well as release of vasoactive substances.26
Clinical Features/Presentation. As mentioned before, AFE will typically be seen during the immediate postpartum period, or during delivery itself, although there have been some instances where it has been seen up to 48 hours after delivery. On occasion they can be seen during first and second trimester miscarriages; however, this is very rare and has been reported in several case studies.27,28 Cardinal findings that have been shown in approximately 80-100% of cases shortly after initial presentation of amniotic fluid embolus are hypoxia, shock, altered mental status, and disseminated intravascular coagulation, which will be discussed in more detail below. The release of amniotic fluid and the matter in which it is suspended causes a profound immunologic response once it enters the maternal circulation that can trigger the above findings.26 Other findings not as commonly seen include seizure activity, fever, chills, nausea, vomiting, agitation and restlessness, evidence of fetal distress, and headache. It is because of the wide variety of symptoms, and many nonspecific findings, that initially it is difficult to diagnose amniotic fluid embolus and, therefore, that the differential is very large. (See Table 3.)
 |
Cardinal Signs of Amniotic Fluid Embolism. Hypoxia. According to the national registry, 93% of all patients with AFE will present with hypoxia. Many times this severe ventilation perfusion mismatch caused by the initial embolism is the cause of respiratory arrest and initial presentation of cyanosis.21 Severe left ventricular dysfunction, which can cause a cardiogenic pulmonary edema picture, is another theory for this early hypoxia. Less likely, it is thought that bronchospasm may be the cause, accounting for approximately 15% of cases. Although hypoxia typically is present throughout the entire course of amniotic fluid embolism, the underlying etiology seems to vary over time between obstructive, cardiogenic and inflammatory source. Hypoxia in both the initial and later phases has been linked in the development of widespread neurologic deficiencies and brain death. During seizure activity, this hypoxia can be the most severe and can be due to anoxic encephalopathy.
Hypotension/Shock. Another common and early symptom seen in AFE is hypotension. Most often, the etiology is cardiogenic shock seen from left ventricular failure, although other etiologies have been reported as well.29-31 It is hard to know what hemodynamic changes occur in the period immediately following presentation since invasive ongoing hemodynamic monitoring does not occur in the otherwise healthy obstetric patient. Animal models have shown that there is a transient increase in PA pressure shortly after the embolus is seen. This is thought to be due to vasospasm along with left ventricular dysfunction. Blood pressure rises transiently, and this occurs simultaneously with the onset of respiratory distress.21,25 Shortly after the transient increase there is a sudden decrease in blood pressure leading into shock and decreased organ perfusion. Cardiac arrhythmias can be present including bradycardias, tachycardias, and ventricular arrhythmias. Using TEE during this phase can be very helpful and findings may be noted such as right ventricular failure, tricuspid regurgitation, increased right-sided pressures, and bulging of the intraventricular and intraseptal septums from right to left.23,24,32
DIC/Coagulopathy. According the national registry,21 approximately 83% of patients showed laboratory values or clinical features consistent with DIC. Seventy-five percent of these patients who presented with bleeding or isolated coagulation issues died despite all the measures taken to control the symptoms.21,24
Altered Mental Status. Encephalopathy due to hypoxia and impaired oxygen delivery to the brain is a common occurrence. The onset is variable and can be seen with seizure (in 50% of patients with encephalopathy), with cardiopulmonary collapse, or with cardiogenic and hemorrhagic shock. It is possible that all of these can overlap and one would see multiple types of shock, hypoxia, and coagulopathy occurring simultaneously.23
Diagnosis. When suspected, one should obtain a complete blood cell count, ABG, coagulation studies, basic chemistries, and a chest radiograph. Complete intake and output should be monitored with a Foley catheter. Although the true diagnoses can only be made during final autopsy where one would be able to see fetal hairs, debris, and squamous cells, a strong clinical suspicion would be reason to continue treatment for this condition.26 One needs to seriously consider other causes of hypoxia and dyspnea, coagulopathies, and symptoms similar to AFE including PE, anaphylaxis, DIC, HELLP syndrome, septic shock, and seizures that are caused by other etiologies, primarily eclampsia.
Management. Because of how uncommon amniotic fluid embolisms are, the treatment options are mostly anecdotal and mostly include supportive care. High flow oxygen, continuous end tidal CO2 monitoring, continuous cardiac telemetry, supportive ventilation and oxygenation with intubation and ventilator control and the anticipation complications of coagulopathies all must be considered and implemented.23,26 For the patient who has not delivered, a continuous fetal monitoring device should be placed.23 It has also been suggested that plasma exchange may be helpful to remove triggering cytokines.33 These patients, because of how labile and critical they are, should always be monitored in an intensive care unit setting and hemodynamically monitored as well, which can be done through central line placement. There are no measures that one can take to prevent the development of amniotic fluid embolism except for a constant watch of the expectant mother and to note when she becomes agitated and restless and shows signs of sudden dyspnea. It has been shown that the onset of AFE can be extremely rapid, ranging from 45 minutes post delivery to death by 1-7 hours. When presenting within 1-2 hours, the mortality has been seen to be close to 50%, due to vasospasm, release of vasoactive substances, and physical plugging of vessels especially in the maternal pulmonary vasculature. Those that survive can then go into seizures, DIC, ARDS, and left ventricular dysfunction; this can cause full cardiopulmonary collapse and death.19 Again, treatment is supportive and can also include TTE or TEE to evaluate for left ventricular filling, and vasopressor therapy for refractory hypotension.23
Bleeding in Late Pregnancy
Placental abruption and placenta previa are important causes of bleeding in late pregnancy (> 20 weeks) and during labor that can cause significant morbidity and mortality for both fetus and mother.
Placenta Previa. Previously, there were three variations of placenta previa that were described in the literature: complete, partial, and marginal, with the differences between marginal and partial difficult to differentiate. With the availability and use of ultrasound, it has become easier to assess the precise location of the placenta in relation to the os, thus redefining the terms. The more contemporary definition of complete placenta previa is the implantation of the placenta over the entire cervical os with the definition of marginal placental previa being within 2 cm of the internal os. A low lying placenta can be defined as one that has its edge within 2 to 3.5 cm of the os.34-36 Overall, all three forms are responsible for approximately 20% of bleeding episodes after 20 weeks gestation.35,37 Placenta previa was detected in approximately 42% of pregnancies at 11-14 weeks, but dramatically decreased to 3.9% at 20-24 weeks, and 1.9% at term.38 One study found that the probability of placenta previa at term is 8% when the lower edge of the placenta is within 23 mm of the internal os between 11-14 weeks. The resolution of most cases of placenta previa prior to delivery is thought to be secondary to trophotropism, the development of the placenta preferentially in the more highly vascularized fundus. The portion of the placenta overlying, or in close proximity to, the os is thought to undergo atrophy, rather than "migrate" as some authors have suggested in the past.39 The incidence of previa increases with prior caesarian, presence during later gestation, chronic hypertension, multiparity, multiple gestations, advanced maternal age, tobacco use, and prior uterine curettage.36,40 There is also a linear risk seen with the number of prior caesarian sections as well.41
Typically, placenta previa presents with painless bright red bleeding, usually after sexual intercourse or minor trauma, in the second or third trimester.40 Many times there can be a "sentinel bleed," when the patient will state that she noted a small amount of painless bleeding around the end of the second trimester that resolved spontaneously. This can be an indication that more bleeding will be likely at time of labor. Uterine contractions may be present in 20% of cases. Fetal positioning is also likely to be abnormal.42,43 Diagnosis can be confirmed safely with a careful sterile speculum exam to exclude other sources of bleeding and transvaginal ultrasonography. Neither should enter the cervical canal more than 3 cm and preferably should be done after consultation with an obstetrician.44,45 A digital vaginal examination should not be undertaken.46 It has been suggested that the patient should be prepped in an OR with anesthesia available in anticipation of an emergent caesarian section before doing a speculum examination and even ultrasonagraphy. Also, with the recent advances seen in ultrasound technology, many cases of placenta previa are now being diagnosed far before the patient would present with their first episode of bleeding and precautions can be made in advance.
Assessment of the bleeding patient should include maternal vital signs, uterine contraction and fetal heart rate monitoring, obtaining adequate intravenous access and baseline laboratory values (complete blood count, type and cross match, coagulation studies).47 Rh immune globulin should be administered if that patient is Rh negative. In a stable patient, observation with fetal and uterine monitoring is indicated for approximately 48 hours after the bleeding has stopped.48 Complications related to prematurity are the major causes for neonatal morbidity and mortality.49 Therefore, attempts to delay delivery and prolong the gestation to allow for fetal lung maturity are encouraged, including administration of corticosteroids. The use of tocolytics can be considered in consultation with an expert in maternal-fetal medicine if bleeding is considered to be secondary to contractions. b-mimetics should be avoided due to the blunting of the maternal response to hypotension.47 Consideration should be given to transferring a patient with placenta previa to a facility with the capabilities for maternal-fetal medicine and neonatal consultation.
Placental Abruption. Placental abruption is a life-threatening condition involving the separation of the placenta from the uterine wall before delivery. Separation may be partial, or rarely, complete, which leads to prostaglandin production causing uterine contraction and a further decrease in placental perfusion.50 On occasion a disruption of the fetal-placental tissues causes the separation, and these damaged vessels will cause the bleeding, leading to a decidual hematoma. This hematoma is the cause of placental separation, destruction of the tissue in the placenta, and subsequent loss of surface area for gas and nutrient exchange between the mother and fetus.34,51 This is why, in late pregnancy, it is imperative to monitor the fetus after a traumatic event involving blunt abdominal injuries where the incidence of placental abruption becomes much higher. When injured pregnant victims alone were looked at in one study, the incidence of abruptio placentae was 20-50% when major maternal trauma was involved, and 1-5% with minor trauma.52,53
Abruption occurs in 1-2% of pregnancies and is associated with neonatal death in approximately 10-30% of those cases due to prematurity of the fetus. Approximately 50% of patients with placental abruption will also deliver prematurely. In previous studies, those rates were reported as being lower (from 0.3% to just above 1%), however, the increase in rate seen is most likely due to increased recognition of the disease by providers.43,54 In a large retrospective study performed in Finland in 2006, risk factors for the development of placental abruption were shown to include previous history of placental abruption (recurrence seen in 8.8% of cases), smoking and previous cesarean section. Other risk factors have been identified but after multivariate analysis was performed, it was difficult to assess the percentage risk associated with them. These include paternal smoking, alcohol use, placenta previa, chorioamnionitis, preeclampsia, and other hypertensive disorders of pregnancy. Previously it was noted that increasing maternal age and black race were risk factors, but recent literature has shown this not to be true.55
The most common presenting signs of placental abruption are vaginal bleeding, abdominal pain, bloody amniotic fluid, uterine contractions and tenderness, and fetal heart rate abnormalities or fetal death. Of note, in 19% of the cases, neither bleeding nor abdominal pain were present clinically. Disseminated intravascular coagulation is also a serious consequence that can occur with severe abruption.51,56,57
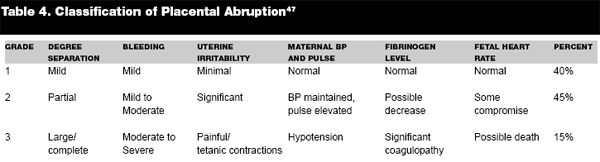
Diagnosis of placental abruption is primarily made clinically and should be considered in all women who present in unexplained preterm labor, with abdominal pain and/or vaginal bleeding, or a history of trauma. It is also important to note that not all patients with placental abruption will present with bleeding. Ultrasound has been shown to have some utility when in the past it had none. Ultrasound cannot rule out the presence of abruption due to the similar sonographic appearance of the acute hemorrhage to normal placental tissue. There are some other signs that can be seen, including the "jello" sign in which the ultrasonagrapher applying pressure with the transducer can cause the placenta to "jiggle." One study found the sensitivity, specificity, and positive and negative predictive values of sonography were 24%, 96%, 88%, and 53%, respectively; therefore, at least half of all cases of abruption will not be detected. However, when there seems to be an abruption on ultrasound, the likelihood that there truly is one is extremely high.58,59 Also, many abruptions are not associated with DIC. (See Table 4.)
 |
Another finding that in the past has been reported more in obstetric and radiology literature is the finding of intrauterine hematomas, primarily subchorionic hematomas. Typically they are diagnosed with ultrasound when evaluating for causes of early pregnancy bleeding. Approximately 11% of patients with threatened miscarriage have a subchorionic hematoma. Many of these can lead to early fetal demise; however, if they progress into the second and third trimester, they can be thought of as a significant risk factor for placental abruption.60-62
Management goals of patients with placental abruption include preventing mortality in the fetus/neonate and mother, significant hemorrhage, DIC, infection, and Couvelaire uterus (where retroplacental blood may peentrate through the myometrium and into the peritoneal cavity).50 In cases of abruption when the pregnancy is near term and there is a live fetus, the goal should be prompt delivery. Careful consideration should be made to minimize fetal or maternal delivery, especially when attempting vaginal delivery. If there is any evidence of distress, cesarean delivery should be performed immediately to prevent total detachment of the placenta, which many times can occur without any warning. If both the mother and the fetus are doing well, management may be more conservative, but hesitantly. The mother should be placed on tocodynometry with the fetus on a heart rate monitor allowing the progression of labor. Baseline laboratory values should be obtained after sufficient intravenous access is obtained including complete blood count, coagulation studies, fibrin split products, fibrinogen, blood type, and cross match, with blood products available if necessary. Administration of Rh immune globulin is warranted in the Rh-negative patient. Any time there are signs of distress such as persistent decelerations when monitoring the fetal heart rate or bradycardia, prompt delivery is required. As in patients with placenta previa, consideration must be given regarding the need to transfer the patient to a facility equipped to handle high-risk obstetrics and neonatal compromise.
Placenta Accreta, Placenta Increta, Placenta Percreta. When the placenta is abnormally attached to the uterine wall it is known as placenta accreta, increta or percreta, depending on how deep and severe it is attached to the wall.
The most life-threatening clinical problem, massive hemorrhage, is seen at delivery and, when it occurs, can cause up to 10% increased risk of maternal and perinatal mortality.63 Although it was considered very rare at one point, the incidence in the past 50 years has increased ten-fold and now can be seen in one out of every 2500 deliveries.64,65 The specific cause of these conditions is unknown, however they are thought to be caused by the absence of the deciduas basalis and incomplete development of the layer beneath. Accreta is the most common presentation of abnormal attachment and does not penetrate through the uterine muscle, accounting for more than 75% of cases. Otherwise, placenta increta and percreta are seen very rarely and refer to more severe extension of the placenta into the uterine myometrial layer with extension into surrounding organs percreta; they can respectively occur approximately 15% and 5% of the time.65 The most significant risk factors seen with these abnormal implantations include previous placental previa and previous cesarean deliveries. Other risk factors have been shown as well, including increased maternal age, previous uterine surgeries such as fibroid removal, and defects in the endometrium.63,65,66 The risk of accreta in a patient with placenta previa alone and no previous surgeries is 3%. In a woman with placenta previa and a history of one previous cesarean delivery, the risk goes up to 11% and increases with each cesarean section. (With four cesarean deliveries, it is 67%.)67-69
Presentation is similar to placenta previa with painless vaginal bleeding noted most prominently. Again, with the newer ultrasonagraphic technology available, this can be diagnosed more frequently antenatal when in the past it was diagnosed with the difficult placental removal at birth, or incomplete removal and hemorrhage. MRI can be used as well, especially in those patients who have equivocal ultrasound findings, or a placenta that is located posteriorly and may be difficult to visualize.70-72
Management of placenta accreta can be quite dramatic. Because of the risk of massive hemorrhage postpartum, the patient may require a hysterectomy performed at the time of cesarean delivery. This delivery should be done at facilities that are equipped with multidisciplinary teams to handle any other complications that may arise. There are other more conservative treatments described including delayed packing of the placenta, methotrexate, and uterine artery embolization that can save the uterus and preserve future fertility options.68,69,73
Vasa Previa. Vasa previa is a condition in which the blood vessels in the placenta and the umbilical cord are trapped between the presenting part of the fetus and the cervical os, and may accompany velamentous insertion (the condition in which the umbilical cord inserts on the chorioamniotic membranes rather than on the placental mass) of the fetal vessels over the cervical os. This carries a high fetal mortality during birth due to massive hemorrhage, since the vessels now lack protection from Wharton's jelly and can be prone to rupture. In the past, this was diagnosed in women during birth when vessels would tear during the act of delivery. Massive hemorrhage would occur and perinatal mortality rates have been reported between 58% and 73%.74-76 Often, the risk factors associated with vasa previa include previous placental previa, or an unusually formed placenta (bilobed, succenturiate-lobed), and with in vitro fertilization or multiple gestations. It can be detected as early as the 16th week of pregnancy with the use of transvaginal ultrasonography and color Doppler.77 Final management should be similar to placenta previa, and should include a cesarean section to save the fetus at approximately 36 weeks.78
Postpartum Hemorrhage. Typically, after a normal vaginal delivery, up to 500 mL of blood is lost, and during a normal cesarean section, approximately 1000 mL. Anything above that can be defined as postpartum hemorrhage and can affect between 5 and 8% of all deliveries. Of all maternal deaths, it can account for 25% of them. Immediate hemorrhage can be seen most commonly right after delivery and up to 24 hours after. Delayed postpartum hemorrhage can be seen 24 hours after and up to 6 weeks post delivery. Commonly, this blood loss is underestimated by the practitioner and may not be calculated correctly due to many reasons, primarily the mother not reporting her own loss.
A simple solution can be as easy as two handed manual stimulation of the very soft and boggy uterus when there is uterine atony. Uterine atony, a diagnosis of exclusion, has been shown to be the cause of up to 50% of cases of postpartum hemorrhage. This can also be done along side of pharmacologic treatments with oxytocics (including oxytocin [Pitocin] and methylergonovine [Methergine]) as well. These two maneuvers should be enough to stop the bleeding. Spontaneous delivery of the placenta has also been shown to reduce blood loss by up to 30% and decrease uterine atony as well as postpartum endometritis.79 However, as a precaution in case these fail, typed and crossmatched blood should be available for resuscitation of the mother if needed. The physician must also decide if trauma played a role in the delivery and if that may be the reason for the blood loss.
Other means of management of postpartum bleeding include uterine tamponade, which can be done with large packing placed up to the bleeding surface, and selective arterial embolization, which is becoming more common in controlling patients with stable postpartum bleeding and has shown a success rate of up to 97%. This procedure has the added benefit of keeping future fertility options open.80 When these options fail, surgical intervention should include arterial ligation, suturing techniques, and, in the end, hysterectomy.81
Other causes of postpartum hemorrhage that carry less morbidity, are described below.
During actual birth, factors relating to the fetus and to delivery itself can result in maternal birth trauma, such as macrosomia, or malpresentations, and delivery that is uncontrolled. This is the second most common cause of postpartum hemorrhage, accounting for 20% of all cases. Lacerations and tears can include the anatomy of the birthing canal, as well as the urethra, cervix, and rectum. Large hematomas can be formed without bleeding, and if left undiagnosed, can expand and eventually lead to large amounts of bleeding hours later. Full examinations must be completed in these women who experience signs of shock and blood loss but with no apparent source of bleeding.
Another source of uterine bleeding, approximately 10% of cases, is caused by retained placental tissue that can be caused by traction done inappropriately on the placenta during delivery, causing tears and retained products of conception. If there is remaining placental tissue in the uterus, the uterus will most likely have a hard time contracting down and further measures may need to be taken. Retained particles result in a decrease in myometrial constriction, and can result in significant hemorrhage. Initially, treatment can include digital exploration of the uterus with attempts to blunt dissect the fragments from the myometrium and allow myometrial contraction. This should not be too difficult since normal placental tissue comes apart easily. Abnormal placental attachment as described earlier with placenta accreta and its variation would not be freed as easily and can cause more bleeding.82,83
Uterine Inversion. Uterine inversion is very uncommonapproximately 1 in 2500and can be seen during the last stage of labor and is associated with immediate life-threatening postpartum hemorrhage.47 Many times, it is associated with aggressive traction on the umbilical cord during delivery as well as placing pressure on the fundus when it is relaxed. However, it can occur spontaneously as well to result in the collapse of the uterine fundus into the uterine cavity.47,82,84 Some risk factors that have been seen associated with it include primiparity, short umbilical cord, fundal pressure during delivery, use of oxytocin, overdistention, prolonged labor, manual extraction of the placenta, and fundal implantation of the uterus.47,85 The patient typically notices sudden onset of abdominal pain and, on examination, there is an absence of the uterine corpus and a bulging noted from the vaginal introitus. Ultrasound can be used to help determine diagnosis as well.86 Efforts should be made, after diagnosis, to properly correct the anatomy which is best done immediately after initial inversion. There are different ways of describing uterine inversion, including complete, incomplete, and prolapsed. A complete inversion involves the internal lining of the fundus passing through the cervical os, which forms a rounded mass in the vagina and no fundus felt abdominally. With an incomplete inversion, part of the fundus passes and part remains, therefore the practitioner may still feel an intraabdominal fundus. If the entire uterus prolapses the cervical os with the fundus coming out of the vaginal opening, it is called a prolapsed uterine inversion.47 Once the cervical uterine segments start to contract, a muscular ring can establish and placement of the uterus is much more difficult. Pharmacologic attempts, which include terbutaline, magnesium sulfate, and tocolytics, can then be made to help relax the uterus if cervical rings develop. Even general anesthesia has been shown to create enough uterine relaxation to promote manipulation of the uterus. If all of these methods fail, then surgical replacement may be necessary.87
Uterine Rupture. Another devastating complication that has been reported in women who choose to have vaginal birth after cesarean section (VBAC) is uterine rupture. With VBAC births becoming more and more common, the success rate and safety has dramatically increased. The risk of having a uterine rupture is relatively small, but it does exist at approximately 0.3% to 1.7%.88,89 As more and more women choose to consider VBAC deliveries, it is anticipated that this rate will start to rise, and physicians should be aware of this risk and how to manage it. Since there is significant fetal mortality associated with rupture, it needs to be diagnosed very quickly. Partial extrusion of the fetus is associated with perinatal mortality rates of less than 1%, however, complete ruptures can be seen at 10-20% fetal mortality. Risk factors primarily include previous cesarean section, mostly with the prior classic vertical incision; also, the risk increases with each cesarean delivery.82 Diagnosis needs to be considered if there are signs of fetal distress or intrapartum vaginal bleeding (although the differential diagnosis includes many other disorders). Ultrasound can certainly help with diagnosis, but it is not always fully accurate. Delivery is imminent so that the fetus can be prevented from exposure to hypoxia, with the window being less than 30 minutes to preserve fetal outcome. Pharmacologic oxytocic agents should be avoided since they can increase the rupture.83,89,90
Conclusion
The primary goal of the emergency physician when treating pregnant patients is to consider any potential disease process that could be life-threatening to both the mother and the unborn fetus. With the use of more modern ultrasound techniques, laboratory tests, higher clinical suspicion of various disease states, and better outpatient follow-up of patients, these patients should not be missed.
References
1. Report of the National High Blood Pressure Education Program Working Group on High Blood Pressure in Pregnancy. Am J Obstet Gynecol 2000;183:S1-S22.
2. Vidaeff A, Carroll M, Ramin SM. Acute hypertensive emergencies in pregnancy. Crit Care Med 2005;33(10 Suppl):S307-12.
3. Koonin LM, MacKay AP, Berg CJ, et al. Pregnancy-related mortality surveillance-United States, 1987-1990. Morb Mortal Wkly Rep CDC Surveill Summ 1997;46:17-36.
4. Sibai BM. Diagnosis and management of gestational hypertension and preeclampsia. Obstet Gynecol 2003;102:181.
5. Hauth JC, Ewell MG, Levine RL, et al. Pregnancy outcomes in healthy nulliparas who developed hypertension. Calcium for Preeclampsia Prevention Study Group. Obstet Gynecol 2000;95:24-28.
6. Brown MA, Hague WM, Higgins J, et al. The detection, investigation, and management of hypertension in pregnancy. Full consensus statement of recommendations from the Council of the Australian Society for the Study of Hypertension in pregnancy. Aust N Z J Obstet Gynaecol 2000;40:139-155.
7. Buchbinder A, Sibai BM, Caritis S, et al. Adverse perinatal outcomes are significantly higher in severe gestational hypertension than in mild preeclampsia. Am J Obstet Gynecol 2002;186:66-71.
8. Sibai BM. Hypertension. In: Gabbe SG, et al, eds. Obstetrics: Normal and Problem Pregnancies, 5th ed. Philadelphia, Churchhill Livingstone Elsevier 2007:Chapter 33.
9. Granger JP, Alexander BT, Llinas MT, et al. Pathophysiology of hypertension during preeclampsia linking placental ischemia with endothelial dysfunction. Hypertension 2001;38(3 Pt 2):718-722.
10. Hayman R, Warren A, Brockelsby J, et al. Plasma from women with pre-eclampsia induces an in vitro alteration in the endothelium-dependent behaviour of myometrial resistant arteries. BJOG 2000;107:108-115.
11. Witlin AG, Friedman SA, Egerman RS, et al. Cerebrovascular disorders complicating pregnancy--beyond eclampsia. Am J Obstet Gynecol 1997;176:1139-1145.
12. ACOG Committee on Practice Bulletins-Obstetrics. Diagnosis and Management of Preeclampsia and Eclampsia. Obstet Gynecol 2002;99:159.
13. Watson KV, Moldow CF, Ogburn PL, et al. Magnesium sulfate: Rationale for its use in preeclampsia. Proc Natl Acad Sci USA 1986;83:1075-1078.
14. Livingston JC, Livingston LW, Ramsey R, et al. Magnesium sulfate in women with mild preeclampsia: A randomized controlled trial. Obstet Gynecol 2003;101:217-220.
15. Lain KY, Roberts JM. Contemporary concepts of the pathogenesis and management of preeclampsia. JAMA 2002;287:3183-3186.
16. Vigil-De Gracia P, Lasso M, Ruiz E, et al. Severe hypertension in pregnancy: Hydralazine or labetalol. A randomized clinical trial. Eur J Obstet Gynecol Reprod Biol 2006;128:157-162.
17. Fenakel K, Fenakel G, Appelman Z, et al. Nifedipine in the treatment of severe preeclampsia. Obstet Gynecol 1991;77:331-337.
18 Rosenberg VA, Lockwood CJ. Thromboembolism in pregnancy. Obstet Gynecol Clin North Am 2007;34:481-500.
19. Gilbert WM, Danielsen B. Amniotic fluid embolism: Decreased mortality in a population-based study. Obstet Gynecol 1999;93:973-977.
20. Kramer MD, Rouleau J, Baskett TF, et al. Amniotic-fluid embolism and medical induction of labour: A retrospective, population-based cohort study. Lancet 2006;368:1444-1448.
21. Clark SL, Hankins G, Dudley DA, et al. Amniotic fluid embolus: Analysis of the national registry. Am J Obstet Gynecol 1995;172(4 Pt 1):1158-1167.
22. Lau G, Chui PP. Amniotic fluid embolism: A review of 10 fatal cases. Singapore Med J 1994;35:180-183.
23. Moore J, Baldisseri MJ. Amniotic fluid embolism. Crit Care Med 2005;33(10 Suppl):S279-S285.
24. Stafford I, Sheffield J. Amniotic fluid embolism. Obstet Gynecol Clin North Am 2007;34:545-553.
25. Clark SL. New concepts of amniotic fluid embolus: A review. Obstet Gynecol Surv 1990;45:360-368.
26. Houry DE, Abbott JT. Acute Complications of PregnancyAmniotic Fluid Embolism. In: Marx JA, et al, eds. Rosen's Emergency Medicine: Concepts and Clinical Practice, 6th ed. Philadelphia, Mosby Elsevier; 2006:Chapter 177.
27. Cromley MG, Taylor PJ, Cumming DC. Probable amniotic fluid embolism after first-trimester termination. A case report. J Reprod Med 1983;28:209-211.
28. Meier PR, Bowes WA Jr. Amniotic fluid embolus-like syndrome presenting in the second trimester of pregnancy. Obstet Gynecol 1983;61:31S-34S.
29. James CF, Feinglass NG, Menke DM, et al. Massive amniotic fluid embolism: Diagnosis aided by emergency transesophageal echocardiography. Int J Obstet Anesth 2004;13:279-283.
30. Staten RD, Iverson LI, Daugharty TM et al. Amniotic fluid embolism: Causing catastrophic pulmonary vasoconstriction: Diagnosis by transesophageal echocardiogram and treatment by cardiopulmonary bypass. Obstet Gynecol 2003;102:496-498.
31. Koegler A, Sauder P, Marolf A, et al. Amniotic fluid embolism: A case with non-cardiogenic pulmonary edema. Intensive Care Med 1994;20:45-46.
32. Price TM, Baker VV, Cefalo RC. Aminiotic fluid embolus: Three case reports with a review of the literature. Obstet Gynecol Surv 1985;40:462-465.
33. Tuffnell DJ. Amniotic fluid embolism. Curr Opin Obstet Gynecol 2003;15:119-122.
34. Clark SL. Placenta previa and abruptio placentae. In: Creasy RK, Resnik R, ed. Maternal-Fetal Medicine: Principles and Practice, 5th ed. Philadelphia: WB Saunders Company; 2004:707-714.
35. Bhide A, Prefumo F, Moore J, et al. Placental edge to internal os distance in the late third trimester and mode of delivery in placenta praevia. BJOG 2003;110:860-864.
36. Faiz AS, Ananth CV. Etiology and risk factors for placenta previa: An overview and meta-analysis of observational studies. J Matern Fetal Neonatal Med 2003;13:175-190.
37. Tintinalli, J, et al, eds. Emergency Medicine: A Comprehensive Study Guide, 6th Edition. McGraw-Hill 2003. Chapter 106.
38. Mustafa SA, Brizot ML, Carvalho MH, et al. Transvaginal ultrasonography in predicting placenta previa at delivery: A longitudinal study. Ultrasound Obstet Gynecol 2002;20:356-359.
39. Benirschke K, Kaufmann P. Pathology of the Human Placenta, 4th ed. New York, NY: Springer; 2000.
40. Sakornbut E, Leeman L, Fontaine P. Late pregnancy bleeding. Am Fam Physician 2007;75:1199-1206.
41. Hendricks MS. Previous cesarean section and abortion as risk factors for developing placenta previa. J Obstet Gynaecol Res 1999;25:137-142.
42. Abbrescia K, Sheridan B. Complications of second and third trimester pregnancies. Emerg Med Clin North Am 2003;21:695-710.
43. Hladky K, Yankowitz J, Hansen WF. Placental abruption. Obstet Gynecol Surv 2002;57:299-305.
44. Leerentveld RA, Gilberts EC, Arnold MJ, et al. Accuracy and safety of transvaginal sonographic placental localization. Obstet Gynecol 1990;76:759-762.
45. Timor-Tritsch IE, Yunis RA. Confirming the safety of transvaginal sonography in patients suspected of placenta previa. Obstet Gynecol 1993;81:742-744.
46. Oyelese Y, Smulian JC. Placenta previa, placenta accreta, and vasa previa. Obstet Gynecol 2006;107:927-941.
47. Francois KE, Foley MR. Antepartum and Postpartum Hemorrhage. In: Gabbe SG, et al, eds. Obstetrics: Normal and Problem Pregnancies, 5th ed. Philadelphia, Churchhill Livingstone Elsevier 2007. Chapter 18.
48. Chamberlain G, Steer P. ABC of labour care: Obstetric emergencies. BMJ 1999;318:1342-1345.
49. Cotton DB, Read JA, Paul RH, et al. The conservative aggressive management of placenta previa. Am J Obstet Gynecol 1980:137:687-695.
50. Ferentz KS, Nesbitt LS. Common Problems and Emergencies in the Obstetric Patient. Prim Care 2006;33:727-750.
51. Gillen-Goldstein J, Lockwood CJ. Abruptio placentae. Accessed November 2007. www.uptodate.com
52. Towery R, English TP, Wisner D. Evaluation of pregnant women after blunt injury. J Trauma 1993;35:731-735.
53. Pearlman MD, Tintinalli JE, Lorenz RP. Blunt trauma during pregnancy. N Engl J Med 1990;323:1609-1613.
54. Ananth CV, Wilcox AJ. Placental abruption and perinatal mortality in the United States. Am J Epidemiol 2001;153:332-337.
55. Rathore SS, McMahon MJ. Racial variation in the frequency of intrapartum hemorrhage. Obstet Gynecol 2001;97:178-183.
56. Tikkanen M, Nuutila M, Hiilesmaa V, et al. Prepregnancy risk factors for placental abruption. Acta Obstet Gynecol Scand 2006;85:40-44.
57. Tikkanen M, Nuutila M, Hiilesmaa V, et al. Clinical presentation and risk factors of placental abruption. Acta Obstet Gynecol Scand 2006;85:700-705.
58. Glantz C, Purnell L. Clinical utility of sonography in the diagnosis and treatment of placental abruption. J Ultrasound Med 2002;21:837-840.
59. Oyelese Y, Ananth CV. Placental abruption. Obstet Gynecol 2006;108:1005-1016.
60. Gilling-Smith C, Toozs-Hobson P, Potts DJ, et al. Management of bleeding in early pregnancy in accident and emergency departments. BMJ 1994;309:574–575.
61. Jouppila P, Huhtaniemi I, Tapanainen J. Early pregnancy failure: Study by ultrasonic and hormonal methods. Obstet Gynecol 1980;55:42–47.
62. Pearlstone M, Baxi L. Subchorionic hematoma: a review. Obstet Gynecol Surv 1993;48:65-68.)
63. Bennett MJ, Sen RC. Conservative management of placenta praevia percreta: Report of two cases and discussion of current management options. Aust N Z J Obstet Gynaecol 2003;43:249-251.
64. Cunningham FG, et al, eds. Williams Obstetrics, 22nd ed. Norwalk, CT: Appleton & Lange; 2005. Chapter 35.
65. Thia EW, Lee SL, Tan HK, Tan LK. Ultrasonographical features of morbidly-adherent placentas. Singapore Med J 2007;48:799-802.
66. Bhide A, Thilaganathan B. Recent advances in the management of placenta previa. Curr Opin Obstet Gynecol 2004;16:447-451.
67. Silver RM, Landon MB, Rouse DJ, et al. Maternal morbidity associated with multiple repeat cesarean deliveries. Obstet Gynecol 2006;107:1226-1232.
68. Resnik R. Diagnosis and management of placenta accreta. Accessed November 2007. www.uptodate.com
69. Miller DA, Chollet JA, Goodwin TM. Clinical risk factors for placenta previa-placenta accreta. Am J Obstet Gynecol 1997;177:210-214.
70. Comstock CH, Love JJ, Bronsteen RA, et al. Sonographic detection of placenta accreta in the second and third trimesters of pregnancy. Am J Obstet Gynecol 2004;190:1135-1140.
71. Chou MM, Ho ES, Lee YH. Prenatal diagnosis of placenta previa accreta by transabdominal color Doppler ultrasound. Ultrasound Obstet Gynecol 2000;15:28-35.
72. Mazouni C, Gorincour G, Juhan V, et al. Placenta accreta: A review of current advances in prenatal diagnosis. Placenta 2007;28:599-603.
73. Kayem G, Davy C, Goffinet F, et al. Conservative versus extirpative management in cases of placenta accreta. Obstet Gynecol 2004;104:531-536.
74. Lee W, Lee VL, Kirk JS, et al. Vasa previa: Prenatal diagnosis, natural evolution, and clinical outcome. Obstet Gynecol 2000;95:572-576.
75. Oyelese KO, Turner M, Lees C, et al. Vasa previa: An avoidable obstetric tragedy. Obstet Gynecol Survey 1999;54:138-145.
76. Francois K, Mayer S, Harris C, et al. Association of vasa previa with a history of second-trimester placenta previa. J Reprod Med 2003;48:771-774.
77. Catanzarite V, Maida C, Thomas W, et al. Prenatal sonographic detection of vasa previa: Ultrasound findings and obstetric outcome in ten cases. Ultrasound Obstet Gynecol 2001;18:109-115.
78. Oyelese Y, Catanzarite V, Prefumo F, et al. Vasa previa: The impact of prenatal diagnosis on outcomes. Obstet Gynecol 2004;103(5 Pt 1):937-942.
79. McCurdy CM, Magann EF, McCurdy CJ, et al. The effect of placental management at cesarean delivery on operative blood loss. Am J Obstet Gynecol 1993;167:1363-1367.
80. Dildy GA. Postpartum hemorrhage: New management options. Clin Obstet Gynecol 2002;45:330-344.
81. Mousa HA, Walkinshaw S. Major postpartum haemorrhage. Curr Opin Obstet Gynecol 2001;13:595-603.
82. Oyelese Y, Scorza WE, Mastrolia R, et al. Postpartum hemorrhage. Obstet Gynecol Clin North Am 2007;34;421-441.
83. Landon MB. Caserean Delivery. In: Gabbe SG, et al, eds. Obstetrics: Normal and Problem Pregnancies, 5th ed. Philadelphia, Churchhill Livingstone Elsevier 2007: Chapter 19.
84. Lipitz S, Frenkel Y. Puerperal inversion of the uterus. Eur J Obstet Gynecol Reprod Biol 1988;27:271-274.
85. Silverman F. Management of the third stage of labor. Accessed Nov 2007. www.uptodate.com.
86. Hseih TT, Lee JD. Sonographic findings in acute puerperal uterine inverson. J Clin Ultrasound 1991;19:306.
87. Baskett TF. Acute uterine inversion: A review of 40 cases. J Obstet Gynaecol Can 2002;24:953-956.
88. Guise JM, McDonagh MS, Osterweil P, et al. Systematic review of the incidence and consequences of uterine rupture in women with previous caesarean section. BMJ 2004;329:19-25.
89. Landon MB, Hauth JC, Leveno KJ, et al. National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network. Maternal and perinatal outcomes associated with a trial of labor after prior cesarean delivery. N Engl J Med 2004;351:2581-2589.
90. Chauhan SP, Martin JN Jr, Henrichs CE, et al. Maternal and perinatal complications with uterine rupture in 142,075 patients who attempted vaginal birth after cesarean delivery: A review of the literature. Am J Obstet Gynecol 2003;189:408-417.
Two-part series on complications of pregnancy.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
