Anticoagulated Trauma Patient
November 1, 2023
Reprints
AUTHORS
Philip S. Nawrocki, MD
EMS Medical Director, Allegheny General Hospital, Associate Medical Director, LifeFlight and AHC Critical Care Transport, Core Faculty EMS Fellowship, Allegheny Health Network, Pittsburgh
Sharon Klapec, MD
Emergency Medical Services Fellowship, Pittsburgh
PEER REVIEWER
Colin Kaide, MD, FACEP, FAAEM
Professor of Emergency Medicine, Ohio State University, Wexner Medical Center, Columbus
EXECUTIVE SUMMARY
- Patients on pre-injury oral anticoagulants when compared to patients not on oral anticoagulants showed a higher risk of overall mortality, in-hospital mortality, longer length of hospital stay, and increased incidence of intracranial hemorrhage.
- Emergent reversal of antiplatelet and anticoagulant medications should be considered in all trauma patients, especially those who display major or severe bleeding in critical body areas or those with hemodynamic instability.
- Current guidelines include a critical decisions algorithm for early anticoagulant reversal after trauma from the Western Trauma Association, which recommends reversal of factor Xa inhibitors with andexanet alfa or prothrombin complex concentrate (PCC) for major intracranial, spine, cavitary, or extremity injury, hemorrhagic shock or need for blood transfusion, urgent or emergent surgery or intervention, or significant ocular trauma.
- Idarucizumab should be considered for major bleeding, planned high-risk surgical intervention, and is indicated when the last dose of dabigatran was given within the past 24 hours. Dabigatran also is one of the only direct oral anticoagulants that can be removed by dialysis.
- Protamine sulfate is a reversal agent that directly binds to and inhibits heparins. It should be considered for major or life-threatening hemorrhage in patients who are receiving a heparin infusion or have received a bolus within the past six hours. Patients who have taken enoxaparin within the past 12 hours also should be considered for reversal with protamine, although only approximately 60% of the enoxaparin drug effect is reversed with protamine.
Emergency medicine providers commonly encounter anticoagulated trauma patients, and it is essential to understand the critical aspects of caring for this unique subpopulation, including specific traumatic injury patterns, emergent reversal, and surgical considerations, as well as relevant emerging concepts.
— Ann M. Dietrich, MD, FAAP, Editor
Introduction
Traumatic injuries are the leading cause of death in United States for individuals younger than 45 years of age.1 Hemorrhage following trauma is thought to account for an estimated 30% to 40% of deaths, many of which occur in the prehospital setting.2 Prehospital efforts often are focused on controlling hemorrhage; however, patients often require operative intervention for definitive hemorrhage control. It is estimated that hemorrhage results in an estimated 60,000 deaths in the United States and 1.5 million deaths worldwide annually.3
In response to hemorrhage and injury to blood vessels, the body has natural mechanisms to achieve hemostasis. The first step in the process begins shortly after injury to a blood vessel is detected, with vascular constriction of the injured vessel to limit blood loss. Injured blood vessels also lead to exposed collagen, which helps to activate and aggregate platelets. Von Willebrand factor (vWF) also is exposed, which leads to additional platelet adhesion. Additional factors released by activated platelets, including adenosine diphosphate (ADP), serotonin, and thromboxane A2 (TXA2), further mediate vasoconstriction and platelet aggregation.4 The net result of platelet activation and aggregation is the formation of a platelet plug, which forms a temporary repair of the injured vessel. These first two steps often are referred to as primary hemostasis.
Secondary hemostasis is a physiologic process by which the primary platelet plug is strengthened and stabilized. The primary mechanism for clot stabilization occurs via the coagulation cascade, a sequential system of enzyme activation that ultimately leads to thrombin activation. Thrombin, also known as factor IIa, then catalyzes fibrinogen to fibrin, which is a fibrous protein that adds stability to the developing clot by cross-linking platelets.5 This process commonly is described as two clotting pathways that converge to a final common pathway. The “extrinsic pathway” is activated by tissue factor (TF) that is exposed secondary to damaged vasculature, while the “intrinsic pathway” consists of clotting enzymes that freely circulate throughout the circulatory system within plasma.4 Various steps along the coagulation cascade may be inhibited or affected by pathologic disease or pharmaceutical drugs, as seen in Figure 1.
Figure 1. Secondary Hemostasis (Coagulation Cascade) |
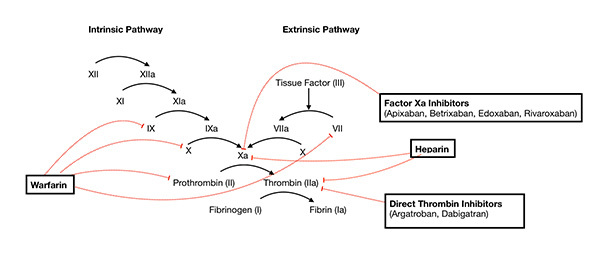 |
Source: https://commons.wikimedia.org/wiki/File:Coagulation_Cascade_and_Major_Classes_of_Anticoagulants.png |
As a balance against excess clot formation and hemostasis, the body additionally has innate mechanisms to break down clot and restore blood flow. This process, termed fibrinolysis, is the fourth and final step of hemostasis and refers to the breakdown of fibrin clot. It occurs concurrently with hemostasis, with the balance between hemostasis and fibrinolysis being determined by local cellular and molecular mediators.
There are several notable hereditary derangements in the primary and secondary hemostasis processes that clinicians should be aware of. Von Willebrand disease refers to a collection of underlying quantitative and qualitative abnormalities in vWF, which leads to impaired platelet adhesion and binding to vascular endothelium following injury.6 Patients are susceptible to various forms of spontaneous bleeding and may have prolonged or excessive bleeding following trauma. Hemophilia A and B occur as a result of decreased activity of factors VIII and IX, respectively. Deficiency in each of these factors leads to impaired secondary hemostasis and formation of fibrin clot, which also may exacerbate hemorrhage from trauma. Factor V Leiden leads to a mutated form of factor V that results in hypercoagulability and has been associated with venous thrombosis.7 There are numerous additional acquired and hereditary conditions that affect hemostasis; however, a full discussion of these is beyond the scope of this article.
Certain patient populations are administered medications with the intent of preventing pathologic clotting and disease burden. Medications may either inhibit platelet activation (antiplatelet agents) or may inhibit various aspects of the coagulation cascade (anticoagulant agents). Certain patient populations who have risk factors, genetic susceptibility, or other underlying factors may be predisposed to develop excess clot burden that can lead to conditions such as ischemic stroke, heart attack, or venous thromboembolism (deep venous thrombosis [DVT] and pulmonary embolism). The specific disease process, patient-specific factors, and evidence-based guidelines assist clinicians in selecting the optimal antiplatelet or anticoagulant agent in each patient to reduce the risk of these clinical conditions. However, tipping the balance away from hemostasis and toward fibrinolysis may have implications in trauma patients, leading to excessive bleeding and risk for hemorrhage.
Epidemiology
Trauma is well known to represent a large proportion of mortality in our population. In fact, unintentional injuries are the fourth leading cause of death in the United States.8 Exsanguination remains the predominant early primary cause of death in trauma.9 Contributing to this is the derangement of the coagulation process by trauma, termed trauma-induced coagulopathy. Trauma-induced coagulopathy may be involved in approximately one-fourth of hemorrhagic deaths due to trauma.10 Indeed, coagulopathy is a component of the widely known “lethal triad” of trauma — coagulopathy, hypothermia, and metabolic acidosis.
Compounding the problem of trauma-induced coagulopathy is the increasing use of oral antiplatelet and anticoagulant medications in the prevention of thrombotic disorders, including stroke, myocardial infarction, and venous thromboembolism, especially in elderly patients. Anticoagulant and antiplatelet therapy in the elderly is a dilemma, since the incidence of both thromboembolic events as well as bleeding increases with advanced age.11 Falls also are more likely with advanced age. Common antiplatelet and anticoagulant agents as well as their mechanisms are listed in Table 1.
Table 1. Common Antiplatelet and Anticoagulant Drugs and Mechanisms |
||
Drug Class |
Common Drugs Generic Name (Brand Name) |
Mechanism of Action |
Antiplatelet |
Aspirin (Bayer, Ecotrin) |
Nonsteroidal anti-inflammatory drug (NSAID) — Irreversibly inhibits cyclooxygenase (COX)-1 and COX-2, thereby inhibiting platelet aggregation and formation of platelet plug. |
Cangrelor (Kengreal) Clopidogrel (Plavix) Prasugrel (Effient) Ticagrelor (Brilinta) |
Blocks P2Y12 receptors on platelets, which are stimulated by adenosine diphosphate (ADP) to promote platelet activation and aggregation |
|
Abciximab (ReoPro) Eptifibatide (Integrilin) Tirofiban (Aggrastat) |
Inhibits GpIIb-IIIa receptors on platelets, preventing platelet aggregation |
|
Cilostazol (Pletal) Dipyridamole (Persantine) |
Phosphodiesterase inhibitors that elevate cAMP levels and block platelet aggregation |
|
Vitamin K Antagonists (VKA) |
Warfarin (Coumadin, Jantoven) |
Inhibits vitamin K epoxide reductase, which decreases synthesis of clotting factors II, VII, IX, X. |
Direct Thrombin Inhibitor |
Argatroban (Acova) Bivalrudin (Angiomax, Angiox) Dabigatran (Pradaxa) |
Directly binds and inactivates thrombin (factor IIa) |
Factor Xa Inhibitor |
Apixaban (Eliquis) Edoxaban (Lixiana, Savaysa) Rivaroxaban (Xarelto) |
Directly binds to factor Xa and prevents conversion of prothrombin to thrombin |
Antithrombin Dependent |
Unfractionated Heparin (UFH) Heparin |
Binds and activates antithrombin, which then inactivates thrombin and factor Xa |
Low Molecular Weight Heparins Dalteparin (Fragmin) Enoxaparin (Lovenox) Fondaparinux (Arixtra) |
Binds and activates antithrombin, which then selectively inhibits factor Xa |
|
Fibrinolytics |
Alteplase/tPA (Activase, CathFlo) Reteplase (Retavase) Tenecteplase (TNKase) |
Converts plasminogen to plasmin, promoting clot breakdown |
Elderly trauma patients tend to have poorer outcomes than younger patients because of a combination of factors, including decreased physiologic reserve, comorbidities, nutritional deficits, and, notably, pre-medication.12 Elderly trauma deaths are expected to increase as the baby boomers are reaching their sixth and seventh decades of life.13 As the population ages, physicians can expect to be confronted increasingly with treatment decisions for the anticoagulated trauma patient.
Several studies have examined the effect of anticoagulation on the outcomes of trauma patients. Increased mortality among trauma patients taking pre-injury oral anticoagulants when compared to those not anticoagulated has been observed in many recent retrospective studies.14 A National Trauma Data Bank study including more than 1 million trauma patients found increased mortality, increased in-hospital mortality, increased incidence of discharge to a long-term care facility, increased incidence of intracranial hemorrhage, and increased death or need for discharge to a long-term care facility following intracranial hemorrhage in those taking pre-injury anticoagulation.15-18 Similarly, a review and meta-analysis of 19 comparative studies of patients on pre-injury oral anticoagulants compared to patients not on oral anticoagulants showed pre-injury anticoagulation was associated with a higher risk of overall mortality, in-hospital mortality, longer length of hospital stay, and increased incidence of intracranial hemorrhage.19 Anticoagulation status must be appreciated and considered in the care of trauma patients.
This article will discuss important aspects of caring for the anticoagulated trauma patient, including specific traumatic injury, emergent reversal, and surgical considerations, as well as relevant emerging concepts in trauma resuscitation.
Specific Injuries
Traumatic Brain Injury
Traumatic brain injury (TBI) is the most common overall cause of death in trauma.9 Pre-injury anticoagulation is associated with higher risk of mortality, in-hospital mortality, longer intensive care unit (ICU) length of stay, and severe TBI.20-22 The most severe TBI has been seen in those taking aspirin and warfarin in combination.22 On the other hand, while several recent studies have identified that direct oral anticoagulant (DOAC) use is not associated with increased risk of mortality or severity in TBI compared with vitamin K antagonist (VKA) use, numerous studies have further found that in comparison to VKAs, DOACs are associated with reduced risk and mortality in TBI.23-26 Early reversal of antithrombotic medications, including antiplatelet agents, DOACs, and VKAs, in major intracranial injury is recommended; however, recommendations are based largely on expert opinion because of the paucity of strong evidence.27,28 This will be discussed in further detail in later sections of this article.
Hemothorax and Hemopericardium
Thoracic injuries are responsible for up to one-fourth of deaths in trauma, of which traumatic hemothorax and hemopericardium are a subset. Anticoagulated patients experiencing traumatic hemothorax have higher rates of chest tube placement, overall complications, and longer hospital length of stay compared with their non-anticoagulated counterparts.29 Several cases of spontaneous hemothorax have been described as a result of anticoagulant therapy, thought to be secondary to minimal chest trauma or spontaneous rupture of small vessels.30 Traumatic hemorrhagic pericardial effusion is life-threatening but relatively rare. One recent report identified that hemorrhagic pericardial effusion may occur after emergency thoracic surgery for trauma, and coagulopathy portends a worse prognosis.31 Serial echocardiography has been suggested as surveillance for development of hemorrhagic pericardial effusion after blunt chest trauma and thoracic surgery for trauma.31-32 One case report describes development of cardiac tamponade three weeks after sustaining a cardiac contusion when a patient was started on anticoagulant treatment.32 As with other body regions, the clinician should have a higher index of suspicion for serious bleeding in patients who take chronic anticoagulation.
The diagnosis of hemopericardium and cardiac tamponade has been greatly improved through the use of emergency ultrasonography in the trauma bay. Fluid within the pericardial sac typically is identified as a collection of hypoechoic (black) fluid surrounding the heart and generally layers posteriorly. Later ultrasound findings include right atrial systolic collapse and right ventricular diastolic collapse. Clinical findings may include hypotension, diminished heart sounds on auscultation, and jugular venous distention (also termed Beck’s triad). Patients also may display signs of cardiogenic shock due to failure of cardiac output, including respiratory distress, narrow pulse pressure, cool extremities, and altered mental status. Without emergent pericardiocentesis or surgical intervention, this may lead to progressive shock, hemodynamic instability, and death.
Intra-Abdominal Bleeding
Abdominal injuries are common sequelae of both blunt and penetrating trauma. The liver is the most commonly injured organ in blunt trauma, followed by the spleen, while the small bowel and large bowel are most commonly injured with penetrating trauma to the abdomen. A recent study from 2022 found the annual incidence of abdominal injuries in trauma to be 7.2 per 100,000 population with a 30-day mortality of 12.5%.33
The effect of pre-injury anticoagulation in patients with traumatic intra-abdominal bleeding is less clear. One recent retrospective cohort study found higher rates of failure of nonoperative management, cardiac arrest, acute kidney injury, myocardial infarction, mortality, and longer hospital and ICU lengths of stay in anticoagulated blunt abdominal trauma patients compared with those who were not taking anticoagulation.34 However, another recent retrospective cohort study found that compared to the non-anticoagulated patient with blunt hepatic, splenic, or renal injuries, patients taking warfarin had similar needs for surgery. In the same study, those taking warfarin were found to require transarterial embolization more often.35 Another study of patients with splenic injuries found that concomitant anticoagulation was associated with increased mortality risk.36
Pelvic Fracture and Hemorrhage
Pelvic fractures generally occur secondary to high-energy blunt trauma mechanisms. One trauma registry study found that pelvic fractures occurred in 9.3% of patients who had blunt trauma.37 Secondary to the high energy required to fracture the bony pelvis, additional injuries are likely and should be sought out by the trauma clinician. The large number of vascular structures that run through and adjacent to the bony pelvis also puts this region at risk of hemorrhage when exposed to trauma.
Management of pelvic fractures focuses on stabilizing the bony pelvic ring, reducing the internal volume of the pelvis, and controlling associated hemorrhage. Stabilization of the fracture provides benefit by reducing further bony injury, reducing patient pain, and reducing local soft tissue injury. Use of pelvic binders is thought to decrease the pelvic volume and reduce hemorrhage through tamponade. In 2011, the Eastern Association for the Surgery of Trauma (EAST) updated their recommendations on the management of pelvic fracture hemorrhage.38 Emergent angiography is recommended in trauma patients who have evidence of active bleeding and/or hemodynamic instability secondary to pelvic fracture. Pelvic binder use may limit hemorrhage but does not seem to affect mortality. EAST suggests that preperitoneal packing be used in instances where angiography is unavailable or delayed, or as a salvage technique after angiographic embolization.
Anticoagulated patients exposed to pelvic fracture warrant special consideration. Bleeding may be more robust and prolonged in those who are taking anticoagulant medications. Thorough assessment, close monitoring, and temporary cessation of anticoagulation should be considered in patients with a pelvic fracture and associated hemorrhage. Significant arterial injury is possible following a low-energy mechanism, such as ground level fall, particularly in the elderly or frail patient.39 Reversal of anticoagulation should be strongly considered in the event of hemorrhage or hemodynamic instability. Aggressive resuscitation measures and early angiographic or surgical intervention is likely to benefit this population. Delayed bleeding also has been reported following low-energy blunt trauma in an elderly anticoagulated patient and should be monitored for carefully.40 No studies comparing specific outcomes of patients on anticoagulation to those who were not could be found.
In the absence of hemorrhage or hemodynamic instability, there is strong evidence that patients with pelvi-acetabular trauma benefit from venous thromboembolism prophylaxis.41 Commonly used modalities include mechanical compression devices as well as pharmaceutical prophylaxis with low molecular weight heparins, oral vitamin K antagonists, factor-Xa inhibitors, or aspirin.
Long Bone Fracture
Long bone fractures are common injuries that generally occur following blunt traumatic mechanisms. However, these injuries also may occur secondary to penetrating trauma from gunshot wounds. In these cases, fractures are more commonly comminuted, open, and associated with significant soft tissue injuries. In 2019 there were 178 million total bone fractures reported worldwide.42 One population-based study found the incidence of long bone fracture to be 406 per 100,000 per year, while another reported the incidence of all bone fractures to be 1,229 per 100,000 per year.43,44 Long bone fractures are more common in younger males and elderly females. Socioeconomic burdens are common following fracture and can include loss of work or occupation, physical disability, impaired quality of life, financial costs, and chronic pain.
Management of traumatic long bone fractures relies on several key principles. Splinting should be performed to reduce blood loss, prevent soft tissue injury, and alleviate pain. A thorough assessment also should be performed for additional injuries, particularly for concomitant nerve, vascular, tendon, or muscular injuries that have the potential to affect limb function. An assessment for bleeding associated with long bone fracture also should be performed, although severe hemorrhage and hemodynamic instability are uncommon in simple long bone fractures. Tourniquets and direct pressure may be used in the event of life-threatening bleeding from an open long bone fracture.
In the anticoagulated trauma patient, special consideration should be given to those with long bone fracture. Compartments of the upper and lower extremities generally provide adequate tamponade to control bleeding from long bones, although the clinician should monitor for excessive bleeding that may lead to compartment syndrome. Estimated blood loss for a closed femur fracture is 1,000 mL to 1,500 mL, and 500 mL to 1,000 mL for a closed tibial fracture.45 These volumes may double if the fracture is open or if the patient takes an anticoagulant. Significant blood loss also commonly is described during surgical fixation of hips and long bones.46 Tranexamic acid (TXA) was found to be safe and effective in reducing blood loss for trochanteric fractures in one randomized study.47 No studies comparing specific outcomes of patients with long bone fractures taking anticoagulation to those who were not could be found.
Pharmacologic interventions may be used for those undergoing surgical intervention for pelvic or long bone fractures in the perioperative period to reduce blood loss. These may include use of antifibrinolytics (tranexamic acid, etc.), desmopressin, factor VIIa and XIII, fibrinogen, fibrin sealants, and non-fibrin sealants. A recent Cochrane meta-analysis found insufficient evidence to draw specific conclusions because of a lack of data.48
Soft Tissue Injury and Hematoma
Soft tissue injuries are common occurrences following trauma. These injuries primarily include skin lacerations and abrasions and hematomas of the skin and deeper structures.
Similar to other traumatic injuries, bleeding from skin and other soft tissue lacerations and injuries produces excessive and prolonged bleeding in the anticoagulated trauma patient. Topical therapies likely are more beneficial in this patient population and may be used to assist with hemostasis. External agents may come as specialized preformed gauze and dressings. QuikClot is a dressing that contains kaolin, a substance that activates factor XII. This may be used for control of external or internal hemorrhage during myriad surgical procedures.49 Chitosan-based dressings, such as Celox, HemCon, and ChitoFlex, interact with platelets and aid primary hemostasis. NuStat is a relatively newer product that uses cellulose and silica-based fibers to locally concentrate and activate platelets and coagulation factors, as well as apply compression to bleeding vessels.
Several other specialized products assist hemostasis by forming a scaffold for thrombus deposition. These products additionally help concentrate coagulation factors at the site and apply direct pressure to areas of bleeding. Gelfoam and Surgifoam are provided as a sponge or powder and consist of an absorbable gelatin matrix. Surgicel is a similar product that comes in mesh form and is based on an absorbable cellulose matrix. Several of the available topical hemostatic agents are shown in Figure 2.
Figure 2. Topical Hemostatic Agents |
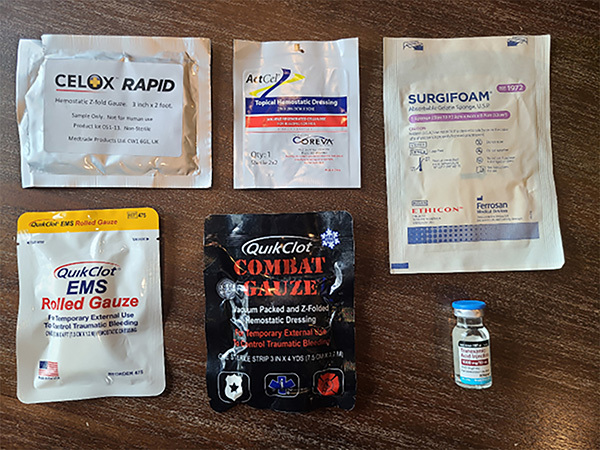 |
Several other topical agents can assist with uncontrolled soft tissue bleeding. Topical thrombin is supplied as a liquid and can help convert tissue fibrinogen to fibrin, strengthening clots produced by primary hemostasis. Fibrin also may be applied topically and may be formulated as a powder, liquid, or preapplied to a gauze dressing. TXA is relatively inexpensive and readily available as a liquid that may be applied to topical bleeding. During the physiology anticoagulation process (also termed fibrinolysis), plasminogen is converted to plasmin by tissue plasminogen activator. This normally serves as a balance to the clotting process and protects against excessive clot formations. TXA prevents this conversion of plasminogen to plasmin, thus inhibiting fibrinolysis and tipping the balance toward forming and strengthening blood clots. This has the benefit of helping to control and prevent excessive pathologic bleeding. A Cochrane meta-analysis found that topical TXA reduces bleeding and blood transfusion in surgical patients.50
In patients who are therapeutically anticoagulated, soft tissue hematomas may occur following minor trauma or may even occur spontaneously. Hemorrhage into muscular spaces may be significant because of the potential for arterial bleeding pressures and the ability to extend between and dissect the muscle fascia and other subcutaneous tissue layers.51 Clinical examination reveals a firm, tender, often ecchymotic mass beneath the skin. Computed tomography angiography commonly is used to diagnose significant hematomas and can help determine whether active hemorrhage is occurring. Treatment generally occurs via interventional radiology, although open surgical management may be used if interventional radiology is unavailable or for salvage therapy.
Emergent Reversal
Emergent reversal of antiplatelet and anticoagulant medications should be considered in all trauma patients. Generally, those who display major or severe bleeding in critical body areas or those with hemodynamic instability may be candidates for reversal.
There are several diagnostic tests that may help to monitor antiplatelet and anticoagulant activity in patients. Various diagnostic tests are able to evaluate different portions of the entire clotting process. The test used depends on what part of the clotting pathway the antiplatelet or anticoagulant agent affects. Several drugs also have specific diagnostic tests to monitor for drug activity. The international normalized ratio (INR) is calculated from prothrombin time (PT) and is used to monitor vitamin K antagonists (for example, warfarin). Factor-Xa levels are helpful in monitoring Factor-Xa inhibitor drug activity, although they may not be available at smaller clinical laboratories. When unavailable, partial thromboplastin time (PTT) or PT/INR may be used to detect the presence of Factor-Xa inhibitors, but they are not effective in detecting the actual drug activity level. Heparins typically are monitored using the PTT, while the presence of dabigatran can be detected using the thrombin time. Thromboelastography (TEG) is gaining traction as a tool to monitor all phases of the clotting process, from initial platelet plug formation to fibrinolysis, and will be discussed in a later section.
Aspirin and P2Y12 Inhibitors (Antiplatelets)
Aspirin is a cyclooxygenase (COX) inhibitor that inhibits thromboxane A2, a substance that accelerates platelet aggregation. Other antiplatelet agents, including clopidogrel, prasugrel, ticagrelor, and ticlodipine, inhibit platelet aggregation by blocking the platelet ADP receptor (P2Y12) and, therefore, are referred to as P2Y12 inhibitors. As previously discussed, these agents inhibit the primary coagulation process and may worsen outcomes in trauma. Because of the complexity of platelet activation and aggregation, there are no specific reversal agents for antiplatelet agents.
Evidence for emergent reversal of antiplatelet agents in the setting of trauma is lacking; however, antiplatelet reversal has been studied in the setting of traumatic intracranial hemorrhage. In 2016, the Neurocritical Care Society and Society of Critical Care Medicine offered guidelines for antithrombotic reversal in intracranial hemorrhage. The authors recommend against platelet transfusion in antiplatelet-associated intracranial hemorrhage in patients not undergoing a neurosurgical procedure; however, they do recommend for platelet transfusion in patients with antiplatelet-associated intracranial hemorrhage who will undergo a neurosurgical procedure.52 One frequently cited study of platelet transfusion in antiplatelet-associated spontaneous intracranial hemorrhage in 2016 (the Patch trial) showed increased mortality and complications in patients who received platelet transfusion compared to those who did not.53 Since that time, platelet transfusion in antiplatelet-associated intracranial hemorrhage has been studied specifically in the trauma population, and these studies have yielded mixed results. A 2019 systematic review identified a lack of clear mortality benefit of platelet transfusion in patients with traumatic intracranial hemorrhage taking antiplatelet therapy, and further included three studies that showed significant evidence of harm with platelet transfusion.54 A 2020 systematic review similarly identified no association between platelet transfusion and risk of death, no significant reduction in hemorrhage progression, and no significant reduction in need for neurosurgical procedures with platelet transfusion in antiplatelet-associated traumatic intracranial hemorrhage.55 However, since that time, one retrospective cohort study found that transfusion of two units of platelets may decrease hemorrhage progression, neurosurgical intervention, and mortality in patients with TBI taking pre-injury antiplatelet agents.56 The 2016 guidelines additionally recommend platelet function testing prior to platelet transfusion.52
Some investigation has focused on laboratory measurement of platelet function in the setting of antiplatelet-associated TBI and further emphasized the complexity of the role of antiplatelet agents. One study found that while platelet reactivity testing for aspirin did detect platelet inhibition related to aspirin use, clinically significant worsening of traumatic intracranial hemorrhage did not differ between patients who received a platelet transfusion for platelet inhibition and those who did not.57 Additional studies found that a significant proportion of patients taking pre-injury antiplatelet agents did not have laboratory-demonstrated platelet inhibition.58-59 One of these studies found that, furthermore, more than half of patients taking antiplatelets with laboratory-demonstrated platelet inhibition did not demonstrate normalization of platelet inhibition with transfusion of platelets.59 The other study found that progression of traumatic intracranial hemorrhage was not related to laboratory-demonstrated platelet inhibition or platelet transfusion.58
Desmopressin, or DDAVP, is another agent that has been used for reversal of pre-injury antiplatelet therapy in the setting of traumatic intracranial hemorrhage. DDAVP increases endothelial release of factor VIII and may increase platelet adhesion by increasing membrane expression of glycoproteins. The 2016 guidelines recommend consideration of a single dose of DDAVP in intracranial hemorrhage associated with COX inhibitors and P2Y12 inhibitors.52 One recent small retrospective cohort study found DDAVP was associated with a lower incidence of hematoma expansion in patients with mild TBI taking pre-injury antiplatelet agents.60 Another recent retrospective cohort study found no difference in traumatic intracranial hemorrhage progression in patients who received DDAVP and/or platelets for laboratory-demonstrated platelet inhibition in the setting of antiplatelet agent use vs. those who demonstrated no antiplatelet inhibition and therefore did not receive DDAVP or platelets. However, this study found worse neurologic outcomes (worse Glasgow Coma Scale scores) in patients receiving DDAVP and/or platelets.61
TXA is another therapy that has been suggested to play a role in managing antiplatelet-associated bleeding.62 TXA acts by inhibiting conversion of plasminogen to plasmin, which helps prevent the inhibition of platelets by plasmin.
Factor-Xa Inhibitors
Factor Xa inhibitors, such as apixaban, rivaroxaban, and edoxaban, are DOACs that inhibit factor Xa conversion of prothrombin to thrombin, causing their anticoagulant effect. There are no recommended blood products for the reversal of factor Xa inhibitors; however, three- and four-factor prothrombin complex concentrate (PCC) have been used. The only Food and Drug Administration (FDA)-approved reversal agent for rivaroxaban and apixaban reversal is andexanet alfa, a recombinant inactivated factor Xa decoy molecule that binds to circulating factor Xa inhibitor and removes it. Andexanet alfa use is considered off-label for edoxaban. The Andexanet Alfa for Bleeding Associated with Factor Xa Inhibitors (ANNEXA-4) trial found that patients with acute major bleeding after factor Xa inhibitor administration (apixaban or rivaroxaban) treated with andexanet alfa showed markedly reduced anti-factor Xa activity and 82% had hemostatic efficacy at 12 hours following therapy. However, this study is limited by its lack of a comparison and lack of focus on patient-oriented outcomes. Additionally, patients requiring emergency surgery and patients with severe traumatic intracranial hemorrhage were excluded, limiting the trial’s generalizability. Also, notably, 10% of patients experienced thrombotic events.63 A systematic review and meta-analysis of 21 studies comparing andexanet alfa or PCC for factor Xa-associated acute, major bleeding found no significant difference between reversal agents.64 One recent retrospective study found significantly lower in-hospital and 30-day mortality in patients with acute major bleeding associated with factor Xa inhibitor use treated with andexanet alfa compared to those treated with PCC.65 However, another recent retrospective cohort study found no difference in clinical outcomes in patients with acute major bleeding associated with factor Xa inhibitor use treated with andexanet alfa vs. PCC. However, this study did find that patients treated with andexanet alfa faced a four times greater cost than the PCC group, highlighting the major drawback of andexanet alfa — cost.66 One study estimated the median cost for andexanet alfa was $22,120 per patient vs. $5,670 per patient for four-factor PCC. This study additionally found thromboembolic events occurred in 4% of patients treated with four-factor PCC, which is less than half of the 10% identified in the ANNEXA-4 trial.67 Current guidelines include a critical decisions algorithm for early anticoagulant reversal after trauma from the Western Trauma Association, which recommends reversal of factor Xa inhibitors with andexanet alfa or PCC for major intracranial, spine, cavitary, or extremity injury, hemorrhagic shock or need for blood transfusion, urgent or emergent surgery or intervention, or significant ocular trauma.27
In patients who take factor-Xa inhibitors, the American Heart Association recommends four-factor PCC to reverse anticoagulation in patients with major bleeding or who require major surgery.68 However, this is an off-label use since it is not an FDA-approved indication.
Another emerging therapy is ciraparantag, also known as aripazine, which binds directly to factors Xa and IIa inhibitors, and has been shown to normalize clotting assays. In two randomized, placebo-controlled, dose-ranging trials in healthy elderly subjects taking apixaban and rivaroxaban, 60 mg and 180 mg, respectively, ciraparantag was found to normalize whole blood clotting time.69 Additional investigation is required before this drug is used clinically.
Response to reversal should be monitored, ideally using a specific anti-Xa level. If unavailable, INR is a less preferable measurement that can be used alternatively. The authors of this text believe that, in alignment with FDA approval and several existing guidelines, andexanet alfa is the reversal agent of choice for major or life-threatening bleeding in patients who take apixaban or rivaroxaban.
Direct Thrombin Inhibitors
Direct thrombin inhibitors (DTIs) bind directly to thrombin and prevent the conversion of fibrinogen to fibrin as well as the conversion of several clotting factors to their activated state. Therefore, patients taking DTIs who experience trauma may have difficulty with secondary hemostasis through inability to strengthen the platelet plug formed during primary hemostasis, and impaired activation of the clotting cascade enzymes.
Patients who take DTIs and who have serious or life-threatening bleeding secondary to trauma warrant close monitoring and aggressive intervention. Idarucizumab is a monoclonal antibody fragment that directly binds to dabigatran, inhibiting its action. It should be noted that it has no hemostatic properties in and of itself, so additional interventions are warranted to stop bleeding. Idarucizumab should be considered for major bleeding, planned high-risk surgical intervention, and is indicated when the last dose of dabigatran was given within the past 24 hours. Dabigatran also is one of the only DOACs that can be removed by dialysis.
Other DTIs, including argatroban and bivalirudin, have no specific antidote to reverse their action. However, the half-life is reported to be around 45 minutes, so in the event of serious bleeding, the drug should be discontinued, and supportive therapy pursued. Pharmacologic effect should correct without intervention in two to six hours in the absence of renal or hepatic impairment.70 For major bleeding, fresh frozen plasma or prothrombin concentrate can be considered. American Heart Association (AHA) guidelines recommend the use of four-factor PCC for DTI-related bleeding, although this is a non-FDA approved indication.68 There is insufficient evidence to suggest PCC use in patients who have received idarucizumab to reverse dabigatran. Response to reversal therapy ideally should be monitored using thrombin time (TT), although if unavailable at the local laboratory, PTT may be used instead.
Heparins
Heparins are a category of drugs that bind to antithrombin and ultimately act to inactivate both thrombin and factor Xa (unfractionated heparins) or factor Xa selectively (low molecular weight heparins). Therefore, patients exposed to heparins have limited ability for secondary hemostasis and strengthening the platelet plug formed during primary hemostasis.
Patients taking heparins with major bleeding also warrant close attention and urgent consideration for reversal. Protamine sulfate is a reversal agent that directly binds to and inhibits heparins. It should be considered for major or life-threatening hemorrhage in patients who are receiving a heparin infusion or have received a bolus within the past six hours. Patients who have taken enoxaparin within the past 12 hours also should be considered for reversal with protamine, although only approximately 60% of the enoxaparin drug effect is reversed with protamine. Dosing is dependent on the amount of heparin received as well as the time of last bolus. Local pharmacy guidelines should be consulted. The risks of protamine have been reported to include anaphylaxis, hypotension, and pulmonary hypertension.
Vitamin-K Antagonists (Coumarins)
Vitamin K antagonists (VKAs), including warfarin, are agents that inhibit the synthesis of vitamin K, a key agent that allows for the synthesis of active functional forms of clotting factors II, VII, IX, and X. Therefore, patients who take VKAs may have difficulty with secondary hemostasis, leading to prolonged and excessive bleeding. The INR is a common laboratory test used to monitor anticoagulation. INR should be measured urgently in the trauma patient taking VKAs.
Management of bleeding in patients who take VKAs is multifaceted. As with other agents, basic therapeutic options should be employed to include direct pressure, local wound hemostatic agents, and consideration of specific reversal agents. Vitamin K may be administered orally or intravenously for the reversal of warfarin. However, since time is required to synthesize new clotting factors using vitamin K, it may take a minimum of four to six hours before any therapeutic effect occurs.71 If a patient taking warfarin requires a semi-urgent procedure or surgery, guidelines recommend oral vitamin K the day prior to the intervention to correct INR and avoid the need for transfusion.72
Fresh frozen plasma (FFP) contains all clotting factors and historically has been used to reverse severe or life-threatening bleeding in patients on warfarin. Since FFP contains naturally occurring factors, there is less lag-time to achieve INR correction compared with vitamin K. Newer agents termed PCC contain only the factors inhibited by VKAs and include KCentra (inactivated factors) and FEIBA (activated factors). Recent guidelines recommend the use of PCCs because they have less risk of infection transmission, lower volume of administration, and less time to administration. They also have been shown to improve INR more rapidly.52,73 Four-factor PCC also is recommended by the AHA as the preferred agent to reverse warfarin-associated bleeding.68
Although recombinant factor VIIa (rFVii or Novoseven) has been used for VKA reversal, a Cochrane review found that the effectiveness is unproven and there is an increased risk of arterial thromboembolic events.74 It is no longer recommended for use in warfarin-associated bleeding.
Thrombolytic Agents
Trauma following thrombolytic administration is less common, although it has been described.75 These medications are indicated for the acute management of pathologic blood clots, including select cases of acute myocardial infarction, acute pulmonary embolism, acute stroke, acute limb ischemia, and occasionally for severe DVT. Common thrombolytics, also referred to as fibrinolytics, include tissue plasminogen activator (TPA), alteplase, reteplase, tenecteplase, and streptokinase. These medications work by catalyzing the conversion of plasminogen to plasmin, the primary enzyme that works to break down clots.
One key consideration for thrombolytic use is the potential for trauma that occurred before administration. Contraindications to thrombolytic use commonly include significant head trauma in the preceding three months and serious trauma within the prior 14 days. However, occult or unreported trauma remains a possible risk. One database review found four of 329 patients who received thrombolytic therapy had occult acute traumatic fractures, including three with rib fractures and one with a pelvic fracture.76 All patients required blood transfusion, three required emergent surgery, and two suffered in-hospital death. This risk likely is more pronounced in elderly patients receiving thrombolytic therapy.
Another potential risk for patients who have received thrombolytic medications is falls. Patients with stroke may have ambulatory dysfunction. They also are more likely to be elderly with additional comorbidities, with less functional capacity at baseline.
Management of bleeding following thrombolytic administration is controversial. Guidelines recommend the use of cryoprecipitate to provide additional clotting factors and aid hemostasis.77 Administration of TXA also may help since this drug inhibits the further conversion of plasminogen to plasmin.78 Evidence supporting the use of FFP, platelet, and PCCs is controversial and not recommended.
General Reversal Considerations
Strong consideration for the reversal of antiplatelet and anticoagulant medications should be given in the trauma patient, particularly those who have uncontrolled hemorrhage, major bleeding in critical areas of the body (particularly the brain), as well as those patients who require emergent surgical interventions. Vital sign and mental status changes often are later signs of hemorrhage and warrant particular attention and anticoagulant reversal. Specific antidotes should be used where available, with consideration of adjunct therapies, such as topical hemostatic agents, direct pressure, and other nonselective medications. Although a specific reversal agent and dosing should be administered in collaboration with pharmacy and trauma clinicians, commonly used reversal strategies for specific antiplatelets and anticoagulants are outlined in Table 2.
Table 2. Antiplatelet and Anticoagulant Reversal Agents |
||
Drug Class |
Common Drugs Generic Name (Brand Name) |
Reversal Agents |
Antiplatelet |
|
|
|
||
|
||
|
||
Vitamin K Antagonists (VKA) |
|
|
Direct Thrombin Inhibitor |
|
|
Factor Xa Inhibitor |
|
|
Antithrombin Dependent |
|
|
|
|
|
|
||
|
||
Fibrinolytics |
|
|
Emerging Concepts in Resuscitation
TEG-Directed Resuscitation
Because conventional coagulation studies do not reliably reflect coagulopathy associated with antiplatelet use, DOAC use, or trauma-induced coagulopathy, viscoelastic tests, such as TEG and rotational thromboelastometry (ROTEM), and associated goal-directed therapies have been a recent focus of interest. TEG and ROTEM measure tension during the various stages of clot formation to provide measurements of clot time, maximum clot firmness, and clot lysis time in a whole blood sample. This is in contrast to static coagulation assays, such as prothrombin time and activated partial thromboplastin time, which measure the function of coagulation factors in plasma.79
A growing body of evidence surrounding viscoelastic testing in the setting of traumatic hemorrhage has yielded mixed results. While some studies suggest resuscitation guided by viscoelastic testing improves outcomes and reduces blood product administration, others suggest no benefit. One recent large, multicenter, randomized controlled trial, the ITACTIC trial, compared trauma outcomes in patients receiving massive transfusion protocols augmented by either interventions guided by viscoelastic testing or conventional coagulation testing and found no difference in overall outcomes.80 More specifically, regarding anticoagulation-associated traumatic hemorrhage, a recent systematic review of 53 studies examining the impact of DOACs on viscoelastic tests concluded that DOAC-specific viscoelastic test assays can provide rapid information regarding DOAC activity; however, DOAC-nonspecific viscoelastic testing was not sensitive enough for identification or quantification of DOAC plasma concentration compared to anti-Xa laboratory measurements.81 Given the paucity of evidence for TEG or ROTEM to guide anticoagulation reversal, specific guidelines cannot be made until additional evidence emerges.
An example TEG result is shown in Figure 3. Table 3 outlines common TEG parameters with an explanation of each one, as well as a description of the therapeutic intervention commonly used for abnormal values at each stage.
Figure 3. Thromboelastography Parameters |
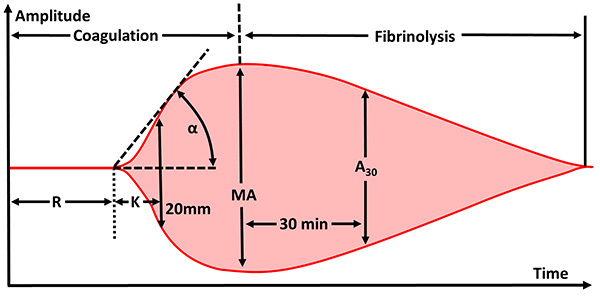 |
Source: https://commons.wikimedia.org/wiki/File:Thromboelastography_parameters.png |
Table 3. Thromboelastography Parameters |
|||
Parameter |
Description |
Dependent on |
Treatment if Abnormal |
R Time |
Time to start of clot or fibrin formation |
Clotting factors |
FFP or PCC |
K Time |
Time to achieve a specific clot strength |
Fibrinogen |
Cryoprecipitate |
Alpha angle |
The speed of fibrin accumulation and cross-linking (rate of clot formation) |
Fibrinogen |
Cryoprecipitate |
Maximum amplitude (MA) |
Maximum clot strength |
Platelets and fibrin |
Platelets ± desmopressin |
Lysis at 30 minutes (LY30) |
Clot strength 30 minutes after maximum clot strength achieved |
Fibrinolysis |
Tranexamic acid |
FFP: fresh frozen plasma; PCC: prothrombin complex concentrate |
|||
Tranexamic Acid
TXA for traumatic hemorrhage has received attention in the past several years. TXA is a synthetic lysine derivative that inhibits plasminogen from binding to and lysing formed plasmin and fibrin. It thereby stabilizes the fibrin clot, exerting its procoagulant effect.82 In 2010, the CRASH-2 trial was published, which was a randomized, placebo-controlled trial of more than 20,000 trauma patients with significant hemorrhage. It was the first study to find a reduced all-cause mortality and reduced risk of death due to bleeding in trauma patients treated with TXA vs. placebo.83 In 2019, the CRASH-3 trial emerged, which was a randomized, placebo-controlled trial that examined more specifically the effects of TXA in TBI patients. It found a significantly reduced risk of head injury-related death in patients treated with TXA vs. placebo. Additionally, this trial showed a similar risk of vascular occlusive events between TXA and placebo groups.84 Despite these positive results among trauma patients as a whole, the effect of TXA has not been studied specifically among trauma patients taking anticoagulant medications. Further research is required before recommendations can be made for the use of TXA in the anticoagulated trauma population specifically.
Whole Blood
A shift from blood component transfusion therapy to whole blood transfusion for massive traumatic hemorrhage has occurred recently. Advantages to whole blood transfusion over component therapy include maintenance of coagulation factors, lack of subsequent thrombocytopenia, and reduction of infused anticoagulant. Several studies suggest the utility of whole blood transfusion in trauma resuscitation.85 A recent systematic review of five studies comparing the efficacy and safety of resuscitation with whole blood vs. blood components identified one study showing significant benefit on 24-hour and 30-day survival, while four others showed no statistically significant difference in survival. All studies demonstrated a benefit in blood product utilization.86
One recent prospective cohort study of 1,377 injured patients receiving emergency-release blood products found whole blood was associated with a four-fold increased survival compared to blood component therapy. Additionally, whole blood patients were observed to have a 60% reduction in overall transfusions compared to patients receiving component therapy.87 There is little available evidence on the effect of whole blood transfusion for traumatic hemorrhage in the setting of anticoagulation specifically, and it is an area requiring further investigation.
Summary
As the population continues to age, the prevalence of anticoagulation use is expected to continue to increase. Clinicians who are involved in providing care for traumatically injured patients should be familiar with common antiplatelet and anticoagulant medications that patients frequently take. Once a patient has experienced trauma, the clinician should aggressively work to evaluate for and manage specific injuries, particularly those at high risk of hemorrhage that may be worsened with concomitant anticoagulant use. Use of specific reversal and treatment options for the anticoagulated trauma patients should strongly be considered. Specific agents and dosing protocols and strategies should be created in consultation with pharmacy and other local subject matter experts. Adjunct therapies, such as direct pressure, topical hemostatic agents, and emerging treatments such as TXA, should strongly be considered as part of the management plan.
REFERENCES
- Centers for Disease Control and Prevention. Injury Prevention and Control. Injuries and violence are leading causes of death. https://www.cdc.gov/injury/wisqars/animated-leading-causes.html
- Sauaia A, Moore FA, Moore EE, et al. Epidemiology of trauma deaths: A reassessment. J Trauma 1995;38:185-193.
- Lozano R, Naghavi M, Foreman K, et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012;380:2095-2128.
- Chang R, Holcomb JB, Leibner E, et al. Hemostasis, Surgical Bleeding, and Transfusion. In: Brunicardi FC, Andersen DK, Billiar TR, et al, eds. Schwartz’s Principles of Surgery, 11e. McGraw Hill; 2019.
- Cico S. Hemostasis. In: Tintinalli JE, Ma O, Yealy DM, et al, eds. Tintinalli’s Emergency Medicine: A Comprehensive Study Guide, 9e. McGraw Hill; 2020.
- Sabih A, Babiker HM. Von Willebrand Disease. In: StatPearls [Internet]. StatPearls Publishing; 2023 Jan-. Updated Aug. 29, 2022. https://www.ncbi.nlm.nih.gov/books/NBK459222/
- Juul K, Tybjaerg-Hansen A, Steffensen R, et al. Factor V Leiden: The Copenhagen City Heart Study and 2 meta-analyses. Blood 2002;100:3-10.
- Xu J, Murphy SL, Kochanek KD, et al. Mortality in the United States, 2021. NCHS Data Brief 2022;456:1-8.
- Callcut RA, Kornblith LZ, Conroy AS, et. al. The how and why our trauma patients die: A prospective multicenter Western Trauma Association study. J Trauma Acute Care Surg 2019;86:864-870.
- Moore EE, Moore HB, Kornblith LZ, et al. Trauma-induced coagulopathy. Nat Rev Dis Primers 2021;7:30.
- Torn M, Bollen WL, van der Meer FJ, et al. Risks of oral anticoagulant therapy with increasing age. Arch Intern Med 2005;165:1527-1532.
- Sartini S, Spadaro M, Cutuli O, et. al. Does antithrombotic therapy affect outcomes in major trauma patients? A retrospective cohort study from a tertiary trauma centre. J Clin Med 2022;11:57-64.
- Rhee P, Bell J, Pandit V, et al. Increasing trauma deaths in the United States. Ann Surg 2014;260:13-21.
- LaDuke ZJ, Hecht JP, Cain-Nielsen AH, et al. Association of mortality among trauma patients taking preinjury direct oral anticoagulants versus vitamin K antagonists. Surgery 2019;166:564-571.
- Bläsius FM, Laubach M, Andruszkow H, et al. Impact of anticoagulation and antiplatelet drugs on surgery rates and mortality in trauma patients. Sci Rep 2021;11:15172.
- Narula N, Tsikis S, Jinadasa SP, et al. The effect of anticoagulation and antiplatelet use in trauma patients on mortality and length of stay. Am Surg 2022;88:1137-1145.
- Yamaji F, Okada H, Kamidani R, et al. Retrospective cohort study to determine the effect of preinjury antiplatelet or anticoagulant therapy on mortality in patients with major trauma. Front Med (Lausanne) 2023;9:1089219.
- Nguyen RK, Rizor JH, Damiani MP, et al. The impact of anticoagulation on trauma outcomes: A National Trauma Data Bank Study. Am Surg 2020;86:773-781.
- Lee ZX, Lim XT, Ang E, et al. The effect of preinjury anticoagulation on mortality in trauma patients: A systematic review and meta-analysis. Injury 2020;51:1705-1713.
- Lim XT, Ang E, Lee ZX, et al. Prognostic significance of pre-injury anticoagulation in patients with traumatic brain injury: A systematic review and meta-analysis. J Trauma Acute Care Surg 2021;90:191-201.
- Yorkgitis BK, Tatum DM, Taghavi S, et al. Eastern Association for the Surgery of Trauma Mulitcenter Trial: Comparison of pre-injury antithrombotic use and reversal strategies among severe traumatic brain injury patients. J Trauma Acute Care Surg 2022;92:88-92.
- Scotti P, Séguin C, Lo BW, et al. Antithrombotic agents and traumatic brain injury in the elderly population: Hemorrhage patterns and outcomes. J Neurosurg 2019;Jul 5:1-10.
- Nederpelt CJ, van der Aalst SJM, Rosenthal MG, et al. Consequences of pre-injury utilization of direct oral anticoagulants in patients with traumatic brain injury: A systematic review and meta-analysis. J Trauma Acute Care Surg 2020;88:186-194.
- Kobayashi L, Barmparas G, Bosarge P, et al. Novel oral anticoagulants and trauma: The results of a prospective American Association for the Surgery of Trauma Multi-Institutional Trial. J Trauma Acute Care Surg 2017;82:827-835.
- Santing JAL, Lee YX, van der Naalt J, et al. Mild traumatic brain injury in elderly patients receiving direct oral anticoagulants: A systematic review and meta-analysis. J Neurotrauma 2022;39:458-472.
- Nishimura T, Guyette FX, Naito H, et al. Comparison of direct oral anticoagulant and vitamin K antagonists on outcomes among elderly and nonelderly trauma patients. J Trauma Acute Care Surg 2020;89:514-522.
- Peck KA, Ley EJ, Brown CV, et al. Early anticoagulant reversal after trauma: A Western Trauma Association critical decisions algorithm. J Trauma Acute Care Surg 2021;90:331-336.
- Iaccarino C, Carretta A, Demetriades AK, et al. Management of antithrombotic drugs in patients with isolated traumatic brain injury: An intersociety consensus document. Neurocrit Care 2023; Apr 7. [Online ahead of print].
- Bhogadi SK, Nelson A, El-Qawaqzeh K, et al. Does preinjury anticoagulation worsen outcomes among traumatic hemothorax patients? A nationwide retrospective analysis. Injury 2023;54:110850.
- Yıldız I, Aksu E, Yildix PO, et al. A case of rivaroxaban associated spontaneous hemothorax. Turk Gogus Kalp Damar Cerrahisi Dert 2019;27:118-120.
- Niziolek GM, Dowzicky P, Joergensen S, et al. Brief report on the development of hemorrhagic pericardial effusion after thoracic surgery for traumatic injuries. Trauma Surg Acute Care Open 2023;8:e001090.
- Valette S, Nouette-Gaulain K, Chardon P, et al. [Delayed tamponade and traumatic myocardial contusion: Evaluate the risk after blunt chest trauma.] Ann Fr Anesthesia Reamin 2007;26:593-595.
- Larsen JW, Søreide K, Søreide JA, et al. Epidemiology of abdominal trauma: An age- and sex-adjusted incidence analysis with mortality patterns. Injury 2022;53:3130-3138.
- Reina R, Anand T, Bhogadi SK, et al. Nonoperative management of blunt abdominal solid organ injury: Are we paying enough attention to patients on preinjury anticoagulation? Am J Surg 2022;224:1308-1313.
- Huang JF, Hsu CP, Fu CY, et al. Preinjury warfarin does not cause failure of nonoperative management in patients with blunt hepatic, splenic or renal injuries. Injury 2022;53:92-97.
- Bhattacharya B, Becher RD, Schuster KM, et al. Anticoagulation is associated with increased mortality in splenic injuries. J Surg Res 2021;266:1-5.
- Demetriades D, Karaiskakis M, Toutouzas K, et al. Pelvic fractures: Epidemiology and predictors of associated abdominal injuries and outcomes. J Am Coll Surg 2002;195:1-10.
- Cullinane DC, Schiller HJ, Zielinski MD, et al. Eastern Association for the Surgery of Trauma practice management guidelines for hemorrhage in pelvic fracture hemorrhage — update and systematic review. J Trauma 2011;71:1850-1868.
- Weber CD, Herren C, Dienstknect T, et al. Management of life-threatening arterial hemorrhage following a fragility fracture of the pelvis in the anticoagulated patient: Case report and review of the literature. Geriatr Orthop Surg Rehabil 2016;7:163-167.
- Sandri A, Regis D, Bizzotto N. Delayed bleeding and pelvic haematoma after low-energy osteoporotic pubic rami fracture in a warfarin patient: An unusual cause of abdominal pain. Case Rep Emerg Med 2014;2014:783268.
- Aggarwal S, Patel S, Vashisht S, et al. Guidelines for the prevention of venous thromboembolism in hospitalized patients with pelvi-acetabular trauma. J Clin Orthop Trauma 2020;11:1002-1008.
- GBD 2019 Fracture Collaborators. Global, regional, and national burden of bone fractures in 204 countries and territories, 1990-2019: A systematic analysis from the Global Burden of Disease Study 2019. Lancet Healthy Longev 2021;2:e580-e592.
- Meling T, Harboe K, Søreide K. Incidence of traumatic long-bone fractures requiring in-hospital management: A prospective age- and gender-specific analysis of 4890 fractures. Injury 2009;40:1212-1219.
- Bergh C, Wennergren D, Möller M, Brisby H. Fracture incidence in adults in relation to age and gender: A study of 27,169 fractures in the Swedish Fracture Register in a well-defined catchment area. PLoS One 2020;15:e0244291.
- Lee C, Porter KM. Prehospital management of lower limb fractures. Emerg Med J 2005;22:660.
- Foss NB, Kehlet H. Hidden blood loss after surgery for hip fracture. J Bone Joint Surg Br 2006;88:1053-1059.
- Baruah RK, Borah PJ, Haque R. Use of tranexamic acid in dynamic hip screw plate fixation for trochanteric fractures. J Orthop Surg (Hong Kong) 2016;24:379-382.
- Gibbs VN, Geneen LJ, Champaneria R, et al. Pharmacological interventions for the prevention of bleeding in people undergoing definitive fixation or joint replacement for hip, pelvic and long bone fractures. Cochrane Database Syst Rev 2023;6:CD013499.
- Johnson D, Westbrook DM, Phelps D, et al. The effects of QuikClot Combat Gauze on hemorrhage control when used in a porcine model of lethal femoral injury. Am J Disaster Med 2014;9:309.
- Ker K, Beecher D, Roberts I. Topical application of tranexamic acid for the reduction of bleeding. Cochrane Database Syst Rev 2013;23:CD010562.
- Dohan A, Darnige L, Sapoval M, Pellerin O. Spontaneous soft tissue hematomas. Diag and Int Imaging 2015;96:789.
- Frontera JA, Lewin JJ 3rd, Rabinstein AA, et al. Guideline for reversal of antithrombotics in intracranial hemorrhage: A statement for healthcare professionals from the Neurocritical Care Society and Society of Critical Care Medicine. Neurocrit Care 2016;24:6-46.
- Baharoglu MI, Cordonnier C, Salman, RA, et al. Platelet transfusion versus standard care after acute stroke due to spontaneous cerebral hemorrhage associated with antiplatelet therapy (PATCH): A randomized, open-label, phase 3 trial. Lancet 2016;387:2605-2613.
- Thorn S, Güting H, Mathes, et al. The effect of platelet transfusion in patients with traumatic brain injury and concomitant antiplatelet use: A systematic review and meta-analysis. Transfusion 2019;59:3536-3544.
- Alvikas J, Myers SP, Wessel CB, et al. A systematic review and meta-analysis of traumatic intracranial hemorrhage in patients taking prehospital anti platelet therapy: Is there a role for platelet transfusions? J Truama Acute Care Surg 2020;88:847-854.
- Lokhandwala AM, Asmar S, Khurrum M, et al. Platelet transfusion after traumatic intracranial hemorrhage in patients on antiplatelet agents. J Surg Res 2021;257:239-245.
- Eastman DK, Spilman SK, Tang K, et al. Platelet reactivity testing for aspirin patients who sustain traumatic intracranial hemorrhage. J Surg Res 2021;263:186-192.
- Peleaz CA, Spilman SK, Bell CT, et al. Not all head injured patients on antiplatelet drugs need platelets: Integrating platelet reactivity testing into platelet transfusion guidelines. Injury 2019;50:73-78.
- Choi PA, Parry PV, Bauer JS, et al. Use of aspirin and P2Y12 response assays in detecting reversal of platelet inhibition with platelet transfusion in patients with traumatic brain injury on antiplatelet therapy. Neurosurgery 2017;80:98-104.
- Barletta JF, Abdul-Rahman D, Hall ST. The role of desmopressin on hematoma expansion in patients with mild traumatic brain injury prescribed pre-injury antiplatelet medications. Neurocrit Care 2020;33:405-413.
- Glass NE, Riccardi J, Horng H, et al. Platelet dysfunction in patients with traumatic intracranial hemorrhage: Do desmopressin and platelet therapy help or harm? Am J Surg 2022;223:131-136.
- Fischer K, Bodalbhai F, Awudi E, Surani S. Reversing bleeding associated with antiplatelet use: The role of tranexamic acid. Cureus 2020;12:e10290.
- Connolly SJ, Crowther M, Eikelboom JW, et al. Full study report of andexanet alfa for bleeding associated with factor Xa inhibitors. N Engl J Med 2019;380:1326-1335.
- Nederpelt CJ, Naar L, Krijnen P, et al. Andexanet alfa or prothrombin complex concentrate for factor Xa inhibitor reversal in acute major bleeding: A systematic review and meta-analysis. Crit Care Med 2021;49;e1025-e1036.
- Sutton SS, Magagnoli J, Cummings TH, et al. Real-world clinical outcomes among US veterans with oral factor Xa inhibitor-related bleeding treated with andexanet alfa or 4-factor prothrombin complex concentrate. J Thromb Thrombolysis 2023;56:137-146.
- Keinath JJ, Lekura J, Hauser CD, et al. Deterioration free discharge comparison of andexanet-alfa and prothrombin complex concentrates (PCC) for reversal of factor Xa inhibitor associated bleeds. J Thromb Thrombolysis 2023;56:315-322.
- Frontera JA, Boat P, Lalchan R, et al. Cost comparison of andexanet versus prothrombin complex concentrates for direct factor Xa inhibitor reversal after hemorrhage. J Thromb Thrombolysis 2020;49:121-131.
- Tomaselli GF, Mahaffey KW, Cuker A, et al. 2017 ACC Expert Consensus Decision Pathway on Management of Bleeding in Patients on Oral Anticoagulants: A report of the American College of Cardiology Task Force on Expert Consensus Decision Pathways. J Am Coll Cardiol 2017;70:3042-3067.
- Angel J, Baker S, Laulicht BE, et al. Ciraparantag reverses the anticoagulant activity of apixaban and rivaroxaban in healthy elderly subjects. Eur Heart J 2022;43:985-992.
- Yee AJ, Kuter DJ. Successful recovery after an overdose of argatroban. Ann Pharmacother 2006;40:336-339.
- Vang ML, Hvas AM, Ravn HB. Urgent reversal of vitamin K antagonist therapy. Acta Anaesthesiol Scand 2011;5:507-516.
- Garcia DA, Crowther MA. Reversal of warfarin: Case-based practice recommendations. Circulation 2012;125:2944-2947.
- Holbrook A, Schulman S, Witt DM, et al. Evidence-based management of anticoagulant therapy: Antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest 2012;141(2 Suppl):e152S-e184S.
- Simpson E, Lin Y, Stanworth S, et al. Recombinant factor VIIa for the prevention and treatment of bleeding in patients without haemophilia. Cochrane Database Syst Rev 2012;14:CD005011.
- Meguro T, Higashi H, Nishimoto K. Acute subdural hematoma after intra-arterial thrombolysis for acute ischemic stroke—case report. Neurol Med Chir (Tokyo) 2005;45:627-630.
- Sultan-qurraie A, Previti M, Alden E, et al. Abstract TP48: IV-tPA in elderly with occult trauma is associated with high risk for hemorrhagic complication. Stroke 2022;53:ATP48.
- Yaghi S, Willey JZ, Cucchiara B, et al. Treatment and outcome of hemorrhagic transformation after intravenous alteplase in acute ischemic stroke: A scientific statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2017;48:e343-e361.
- Yaghi S, Eisenberger A, Willey JZ. Symptomatic intracerebral hemorrhage in acute ischemic stroke after thrombolysis with intravenous recombinant tissue plasminogen activator: A review of natural history and treatment. JAMA Neurol 2014;71:1181-1185.
- Nalezinski S. Methods to correct drug-induced coagulopathy in bleeding emergencies: A comparative review. Lab Med 2022;53:336-343.
- Baksaas-Aasen K, Gall LS, Stensballe J, et al. Viscoelastic haemostatic assay augmented protocols for major trauma hemorrhage (ITACTIC): A randomized, controlled trial. Intensive Care Med 2021;47:49-59.
- Sahli SD, Castellucci C, Roche TR, et al. The impact of direct oral anticoagulants on viscoelastic testing — A systematic review. Front Cardiovasc Med 2022;9:991675.
- Cai J, Ribkoff J, Olson S, et al. The many roles of tranexamic acid: An overview of the clinical indications for TXA in medical and surgical patients. Eur J Haematol 2020;104:79-87.
- CRASH-2 trial collaborators; Shakur J, Roberts I, Bautista R, et al. Effects of tranexamic acid on death, vascular occlusive events, and blood transfusion in trauma patients with significant hemorrhage (CRASH-2): A randomized, placebo-controlled trial. Lancet 2010;376:23-32.
- CRASH-3 trial collaborators. Effects of tranexamic acid on death, disability, vascular occlusive events and other morbidities in patients with acute traumatic brain injury (CRASH-3): A randomized, placebo-controlled trial. Lancet 2019;394:1713-1723.
- Weymouth W, Long B, Koyfman A, et al. Whole blood in trauma: A review for emergency clinicians. J Emerg Med 2019;56:491-498.
- Malkin M, Nevo A, Brundage SI, Schreiber M. Effectiveness and safety of whole blood compared to balanced blood components in resuscitation of hemorrhaging trauma patients — A systematic review. Injury 2021;52:182-188.
- Brill JB, Tang B, Hatton G, et al. Impact of incorporating whole blood into hemorrhagic shock resuscitation: Analysis of 1,377 consecutive trauma patients receiving emergency-release uncrossmatched blood products. J Am Coll Surg 2022;234:408-418.
- Baskaran J, Lopez RA, Cassagnol M. Prothrombin complex concentrate. StatPearls. StatPearls Publishing; 2023 Jan. 2022 Dec. 19. https://pubmed.ncbi.nlm.nih.gov/30969538/
- Pollack CV Jr, Reilly PA, Eikelboom J, et al. Idarucizumab for dabigatran reversal. N Engl J Med 2015;373:511-520.
Emergency medicine providers commonly encounter anticoagulated trauma patients, and it is essential to understand the critical aspects of caring for this unique subpopulation, including specific traumatic injury patterns, emergent reversal, and surgical considerations, as well as relevant emerging concepts.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
