Adverse Drug Reactions for the Emergency Physician: Part II
Adverse Drug Reactions for the Emergency Physician: Part II
Authors: Larissa I. Velez, MD, Department of Emergency Medicine, University of Texas Southwestern, Dallas; Javier Caldera, MD, Department of Emergency Medicine, University of Texas Southwestern, Dallas; Sing-Yi Feng, MD, Department of Emergency Medicine, University of Texas Southwestern, Dallas; and Ellen O'Connell, MD, Department of Emergency Medicine, University of Texas Southwestern, Dallas.
Peer Reviewers: Frank LoVecchio, DO, MPH, FACEP, Research Director, Maricopa Medical Center; Medical Director, Banner Poison Center, Phoenix, AZ; and Barry H. Rumack, MD, Director Emeritus, Rocky Mountain Poison and Drug Center, Clinical Professor of Pediatrics, University of Colorado Health Sciences Center, Denver.
This issue is the second part of a two-part series that discusses the pharmacology of adverse drug reactions important to the emergency physician. Part I covered angiotensin-converting enzyme inhibitors and angioedema, gastrointestinal bleeding associated with non-steroidal anti-inflammatory drugs, anti-convulsant hypersensitivity syndrome, drug-induced Stevens-Johnson syndrome and toxic epidermal necrolysis, and drugs associated with the prolonged QT interval syndrome.
An important principle throughout this series is the real potential for adverse drug interactions with drugs used or started from the ED. One very helpful habit is to have an accurate list of all medications the patient is currently taking or has taken within the past two weeks, including prescription, over-the-counter, and herbals. This list should be consulted before using or initiating any medication during an ED visit. The principle of accurately and completely identifying all patient medications is so important, that it has been identified as Goal 8 of the 2006 National Patient Safety Goals: "Accurately and completely reconcile medications across the continuum of care." This medication reconciliation process is required for all patients who access the hospital system—emergency department, outpatient, and inpatient—and will be assessed by JCAHO during their inspection visits. So, in addition to being a good thing to do to minimize adverse drug interactions, medication reconciliation now is a regulatory requirement.
—J. Stephan Stapczynski, MD, Editor
Warfarin
Warfarin (Coumadin) is the most commonly used oral anticoagulant. It was synthesized by and named after the Wisconsin Alumni Research Foundation in 1948. Warfarin is administered as a racemic mixture, with the S-enantiomer being the active form. Warfarin is metabolized mostly by cytochrome P450 2C9 (CYP2C9).1 Its mechanism of action is interfering with the vitamin K-dependent clotting factors (II, VII, IX, X). Warfarin has a narrow therapeutic window and, due to its metabolism through the P450 system, warfarin has many interactions. Individual metabolism, foods, and other drugs can either induce or inhibit the enzymes, which results in either failure of therapy or excessive anticoagulation.1
Emergency physicians must know the most common interactions of warfarin. Whenever a patient using warfarin comes to the ED, a good drug history must be obtained. The history should include the use of antibiotics, anticonvulsants, and herbal remedies. Of the herbal remedies, St. John's Wort, commonly used as an over-the-counter antidepressant, can be particularly problematic. It is known to cause induction of CYP2C9, thus increasing the metabolism of warfarin.2,3 Ginkgo biloba, used for memory enhancement, and ginseng, used for fatigue and anxiety, are both known to increase the risk of bleeding in patients taking warfarin.4 Antifungal drugs (such as miconazole [Monistat-Derm, Micatin, Lotrimin, Micozole] and fluconazole [Diflucan]) and the quinolone antibiotics are potent inhibitors of CYP2C9, thus they increase availability of warfarin and increase the risk of bleeding.5-7 Cimetidine (Tagamet) with some of the selective serotonin reuptake inhibitors (SSRIs) has a similar effect. On the contrary, CYP2C9 inducers such as phenobarbital and rifampin (Rifadin, Rimactane, Rofact) may decrease the effects of warfarin, resulting in an increased risk of thromboembolic events.8 Table 1 summarizes common drug interactions with warfarin and their effect on clotting.
| Table 1. Drug Interactions with Warfarin |
|
Serotonin Syndrome
Serotonin syndrome (SS) is an iatrogenic drug-induced "toxidrome" that results from9 the overstimulation of the 5-HT1A receptors in the central grey nuclei and the medulla, with perhaps some overstimulation of the 5-HT2 receptors.10 This overstimulation may be due to excess precursors of serotonin or its receptor agonists, lower reuptake of serotonin from the synaptic cleft, or slowdown of serotonin metabolism.10,11
The diagnosis of SS includes a history of use of a serotonergic agent(s), the presence of recognized signs and symptoms, and the exclusion of other conditions. Since it may be hard to distinguish from neuroleptic malignant syndrome, the diagnosis of SS often includes the lack of use of a neuroleptic agent. The syndrome mainly affects the autonomic, GI, and neurological systems. The onset of symptoms is sudden, and it usually manifests itself less than 24 hours after treatment initiation or overdose.10 It seems that the more powerful the serotonergic agent and the higher the dose, the more pronounced are the SS symptoms.12 The diagnosis of SS is guided by the Sternbach criteria, listed in Table 2, although other less utilized diagnostic criteria exist.13-15
| Table 2. Sternbach Criteria for the Diagnosis of Serotonin Syndrome |
|
There is no specific test to diagnose the serotonin syndrome—elevation of total creatine kinase and leukocyte count, elevated transaminases, and lower bicarbonate levels have been used, but lack accuracy. Disseminated intravascular coagulation, kidney failure, metabolic acidosis, and adult respiratory distress syndrome (ARDS) are reported complications.10
Meticulous supportive management is the mainstay of care. All serotonergic agents must be discontinued. Isotonic crystalloids should be administered to optimize urine output and avoid renal failure from myoglobin deposition in the kidneys. Benzodiazepines should be given to treat agitation. Although their effectiveness has not been demonstrated in prospective studies, cyproheptadine (Periactin), chlorpromazine (Thorazine), and methylsergide (Sansert) have been described as possible antidotal therapies for SS.16-18
Cyproheptadine (4 mg po q2-4h up to 0.5 mg/kg/d) is a histamine-1 receptor antagonist with anticholinergic and antiserotonergic characteristics. Chlorpromazine (25 mg IM) is a neuroleptic with 5-HT1A and 5-HT2 receptor antagonism that also has anticholinergic effects. One case of partial improvement has been reported with the use of propranolol (20 mg po q8h).19
Neuroleptic Malignant Syndrome
Neuroleptic malignant syndrome (NMS) is an uncommon but life-threatening condition with a reported mortality of 30-50%.20 It was first described in the 1960s by Delay.21 NMS is an idiosyncratic condition that is thought to occur in 1% of the patients receiving neuroleptics.22 The syndrome usually occurs 3-9 days after the neuroleptic therapy is initiated. NMS is associated more frequently with the high potency neuroleptics haloperidol and thiothixene.22 However, most of the newer antipsychotics also have been reported to cause NMS.23-26
NMS has been associated with reduced signaling in the dopaminergic pathways due to sudden withdrawal of dopaminergic drugs (such as Parkinson's disease drugs) and drugs that block dopamine signaling.27 In both of these scenarios, the culprit is thought to be dopaminergic blockade in the basal ganglia and in the hypothalamus.22
The clinical features of NMS include alteration in mental status, autonomic instability, hyperthermia, and rigidity.28 The primary motor component is the lead pipe rigidity. The patient's mental status can range from delirium to coma. The cardiovascular instability results in labile blood pressure, diaphoresis, tachycardia, and tachydysrhythmias. Hyperthermia is key to the diagnosis, although isolated case reports describe the syndrome without elevated temperatures.27,28
The diagnosis is made on clinical grounds in a patient with the signs and symptoms described above and who is using a neuroleptic agent. The most widely accepted diagnostic criteria were published by Levenson in 1985.29 (See Table 3.)
| Table 3. Levenson's Criteria for NMS |
|
Though not diagnostic, laboratory abnormalities that can be seen include leukocytosis with a left shift, and an elevated creatinine kinase due to rhabdomyolysis from muscle rigidity.22 Myoglobinuria may also be present and renal function may be compromised due to the rhabdomyolysis. Electrolyte imbalances, such as hyperkalemia and a low bicarbonate, also may be found.
NMS can be misdiagnosed as sepsis. For this reason, it is important to consider other causes for the patient's signs and symptoms, such as meningitis or encephalitis. A head computerized tomography (CT) scan and lumbar puncture should be performed, and empiric treatment with antibiotics should be considered. The syndrome also is difficult to differentiate from SS.30
As with the most adverse drug reactions (ADRs), the mainstay of treatment consists of aggressive supportive measures and discontinuing the offending agent. Patients should be hydrated aggressively to maintain an adequate urine output. Hyperthermic patients must be cooled. Intravenous benzodiazepines should be used to decrease muscle rigidity and to control the agitation. Antipyretics generally are not beneficial, since the hyperthermia is due to excessive muscle contractions, not hypothalamic disturbances.
In addition to the above-mentioned interventions, some pharmacologic agents can be used. Dantrolene sodium (Dantrium), bromocriptine (Parlodel), and L-dopa (Dopar, Larodopar) are the most common agents used.20,27,30,31 Dantrolene sodium causes muscle relaxation via inhibition of the release of calcium from the sarcoplasmic reticulum and is the classic antidote for malignant hyperthermia.27,32 The recommended dose of dantrolene sodium is 3-5 mg/kg/day IV divided in 3-4 doses.22 Bromocriptine, a central dopamine agonist, also has been used successfully. It should be administered orally or by nasogastric tube at 5 mg every 6 hours. In severe cases, patients may need to be paralyzed with non-depolarizing agents and intubated. Electroconvulsive therapy is cited in various articles as an alternative treatment.22,27,28 The time to recovery from NMS has been reported to be 9-15 days.27
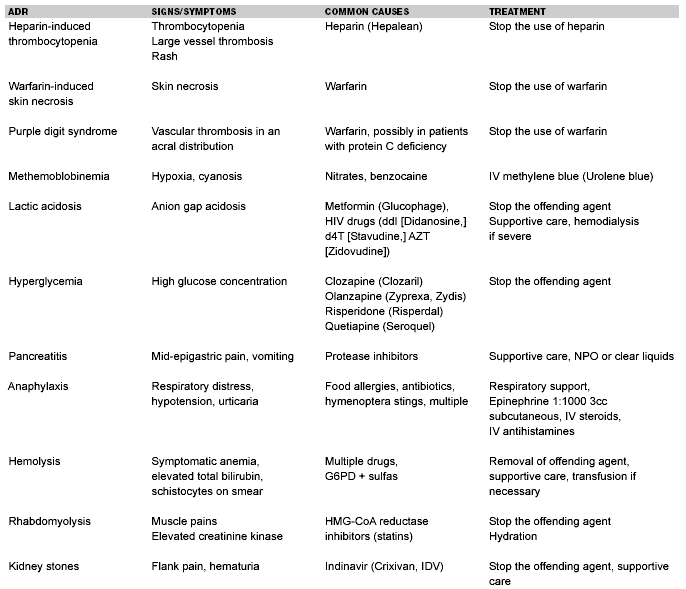
| Table 4. Other Adverse Reactions |
|
| Table 5. Adverse Drug Reaction Resources | ||||||||||||||||
|
Malignant Hyperthermia (MH)
MH is a rare autosomal dominant disorder of the skeletal muscle, specifically at the ryanodine receptor complex.33 This receptor controls calcium release from the sarcoplasmic reticulum. When triggered, there is an excessive release of calcium from the sarcoplasmic reticulum in the skeletal muscle and increased permeability of the cell membranes.33,34 Its incidence is estimated to be between 1 in 50,000 to 1 in 100,000 people.33 MH is associated with the use of halogenated anesthetic gases such as halothane (Fluothane) and succynylcholine (Suxamethonium, Anectine), and as such, it generally occurs in the intraoperative setting.33 The condition is characterized by rapid onset of fever, changes in blood pressure, and hyperkalemia followed by a metabolic acidosis and hypercarbia.28,34,35 Masseter muscle rigidity is one of the most common findings.36 Treatment consists of immediate cessation of the exposure to the offending agent and the use of dantrolene sodium.30 Dantrolene inhibits the ryanodine receptor, and its introduction resulted in a significantly decreased mortality rates, from 80% to less than 10%.32
Extrapyramidal Symptoms (EPS)
Drug-induced movement disorders, also called extrapyramidal symptoms (EPS), usually are associated with a blockage of the dopamine receptor, with an unopposed cholinergic tone.28,37 Dystonia and akathisia will be discussed, since these are the most likely EPSs encountered by emergency physicians.
Dystonia
Dystonia is an idiosyncratic reaction defined as a tonic muscular contraction localized to one or several groups of muscles. It usually involves the mouth and/or the neck.38 The clinical effects are oculogyric crisis, jaw fixation, torticollis, and even opisthotonus.37 Although not life-threatening, the symptoms are distressing to the patient. Dystonias usually occur during the first week of treatment with an antipsychotic.37 It is estimated that 2-3% of the patients on an antipsychotic medication may develop a dystonic reaction. The acute treatment of a dystonic reaction includes the use of IV anticholinergic agents such as diphenhydramine (1mg/kg IV or IM) or benztropine (Cogentin, Bensylate) (2 mg IM or PO) followed with by oral doses of the medications for up to a week.28 The patients should not resume the use of the offending agent.
Akathisia
The term akathisia refers to the sensation of restlessness, usually in the lower extremities, that causes patients to feel an urge to move around.38,39 Akathisia is caused by dopamine receptor blockers such as the antipsychotics and antiemetics but also has been reported with the use of selective serotonin reuptake inhibitors (SSRIs).40 It is estimated to occur in 20-30% of patient treated with neuroleptics, although the number varies widely in the literature.41 It usually occurs in patient within the first week of treatment with these medications.37 Anticholinergic drugs such as benztropine 1-2 mg/day have been used as treatment, but there are no prospective studies addressing their efficacy.42
Disulfiram-Like Reactions
Disulfiram (tetraethylthiuram disulfide) (Antabuse) has been used as an aid to treat alcohol dependence for more than 40 years.43 It acts by inhibiting the hepatic aldehyde dehydrogenase, resulting in an elevation of acetaldehyde in the blood when alcohol is ingested.44 The clinical effects of this disulfiram-ethanol reaction include nausea, vomiting, headache, flushing, and palpitations.44,45 In severe instances this can cause myocardial infarctions, respiratory failure, congestive heart failure, respiratory depression, and even death.43 There are multiple substances known to cause disulfiram-like reactions: mushrooms (Coprinus atramentarius), sulfonylureas, industrial solvents (tricholorethylene), scombroid toxicity, and antimicrobial agents such as trimethroprim-sulfamethoxazole (Bactrim, Septra, Sulfatrim), cephalosporins, and metronidazole (Flagyl).44,45
There are no specific tests to diagnose disulfiram-like reactions. The mainstay of treatment consists of supportive measures. IV crystalloids will treat the hypotension and fluid loss due to the vomiting. Rarely, vasopressors may be needed.46 Hypoglycemia should be treated with D50W. Thiamine and folate should be repleted in the malnourished patient. Antihistamines can be used to treat the flushing and pruritus.47
Contrast-Induced Nephropathy
While not exactly an adverse therapeutic drug reaction, contrast-induced nephropathy (CIN) is an adverse event due to contrast-requiring studies, which frequently are a part of the ED patient work-up.
CIN usually is defined as a rise in serum creatinine of 0.5 mg/dL or a 25% increase above baseline, which usually occurs within 2-3 days after receiving parenteral radiographic contrast for a study, and for which there is no alternative explanation.48,49 The exact mechanism of the nephropathy is unknown. Etiologies that have been postulated are a direct reduction in renal perfusion caused by the contrast medium, and direct toxicity to renal tubular cells.48
The exact incidence of CIN is unknown. In a group of patients undergoing cardiac angiography, Rudnick found that an increase in serum creatinine of 0.5 mg/dL in 8% of patients with a baseline creatinine of less than 1.5. None of these patients had an increase greater than 1 mg/dL.50 However the incidence of CIN in patients with baseline renal impairment has been found to be 12-27% and may actually be higher.48
There are several identified risk factors for developing CIN. From the contrast standpoint, factors are the amount of contrast used and the type of contrast. There is a statistically significant increased incidence of CIN in patients receiving ionic vs. non-ionic contrast medium.50 Also, high-osmolar contrast is worse than low osmolar contrast.48
Multiple patient factors contribute to an increased risk for CIN. The patients at highest risk are those with pre-existing renal insufficiency alone or in combination with diabetes.50 Other conditions that put patients at risk are diabetes alone, hypertension, age older than 70 years, heart failure, dehydration, NSAID use, and aminoglycoside use.48,49 Particular caution must be taken with diabetics who are using biguanides such as metformin, as a slight decrease in renal function due to contrast use may bring about a severe, potentially fatal metabolic (lactic) acidosis.49
Prevention of CIN has provoked much study. However in general, there remains no clear strategy for prevention. Using a non-ionic, low- or iso-osmolar contrast medium is a first step. Maintaining adequate hydration is a generally accepted prevention measure.49 Recently there has been great excitement over the use of IV sodium bicarbonate (NaHCO3) to prevent CIN. A 2004 study compared IV NaCl to IV NaHCO3 as pretreatment, given from 1 hour prior to the use of the contrast until 6 hours after. The majority of patients (110/119) were undergoing cardiac catheterization. In the NaHCO3 group, 1/60 developed CIN, whereas 8/59 in NaCl group did.51
Another described prevention strategy is the use of N-acetylcysteine (NAC) pre- and post-treatment. NAC is thought to function as a vasodilator and free radical scavenger.49
In the past, forced diuresis with mannitol (Osmitrol, Resectisol) or furosemide (Lasix) had been recommended. However, these treatments have proven only to exacerbate dehydration, thus worsening the situation and should be avoided.48
The primary treatment for CIN is prevention. Usually, most elevations in serum creatinine are transient, peaking at 3-4 days post procedure, and returning to baseline in 1-2 weeks.48,49 Rarely is hemodialysis necessary, and it remains the only true treatment for CIN. Hydration remains the mainstay, as well as using least-toxic media in the smallest amounts necessary. NaHCO3 pre- and post-treatment, and/or NAC also may be helpful. However, more data are needed before either strategy can be recommended routinely as prevention for CIN. Monitoring post-procedure for a rise in serum creatinine or symptoms of decreased renal function is recommended for patients most at risk for CIN, particularly those with preexisting renal insufficiency or diabetes.
Conclusions
ED patients are at risk of adverse drug events. Some of the risk factors for this are multiple medical problems, advanced age, polypharmacy, and the frequent use of herbal products and over-the-counter medications not reported to the physician.8,52,53-55 Emergency physicians should be aware of the most common ADRs and must know how to prevent and manage them. In the ED, a careful history of the patient's medical conditions, a complete list of medicines and herbal remedies might help reduce the risk for ADRs. However, in the hectic environment of the ED physicians sometimes might overlook potentially dangerous drug interactions.
Prescription errors that may result in ADRs might be reduced by hospital protocols to track ADRs.56 Having a dedicated ED pharmacist has been proven to reduce medication errors by having someone dedicated to analyzing physicians' orders and the patients' lists of medications.57 Other useful resources to recognize potential drug interactions and ADRs in the ED are computer programs, PDAs, and the Internet.58,59 Computerized order entries can help not only in making orders understandable and thus reducing errors, but also can be used to help identify potential drug interactions.60
Another way to reduce ADRs is to report all events encountered in clinical practice. Wood and Stein suggest that a drug safety and ADR reporting program independent from the FDA might be useful.61 Finally Vinay in 2005 lists a number of different web sites that are available to report and read about ADRs associated with different drugs.62
References:
1. Wilkinson GR. Drug metabolism and variability among patients in drug response. N Engl J Med 2005;352:2211-2221.
2. Xie HG, Kim RB. St. John's wort-associated drug interactions: Short-term inhibition and long-term induction? Clin Pharmacol Ther 2005;78:19-24.
3. Dostalek M, Pistovcakova J, Jurica J, et al. Effect of St. John's wort (Hypericum perforatum) on cytochrome P-450 activity in perfused rat liver. Life Sci 2005;78:239-44.
4. Pal D, Mitra AK. MDR- and CYP3A4-mediated drug-herbal interactions. Life Sci 2006.
5. Niwa T, Shiraga T, Takagi A. Effect of antifungal drugs on cytochrome P450 (CYP) 2C9, CYP2C19, and CYP3A4 activities in human liver microsomes. Biol Pharm Bull 2005;28:1805-1808.
6. Delafuente JC. Understanding and preventing drug interactions in elderly patients. Crit Rev Oncol Hematol 2003;48:133-143.
7. Prybys KM. Deadly drug interactions in emergency medicine. Emerg Med Clin North Am 2004;22:845-863.
8. Ogu CC, Maxa JL. Drug interactions due to cytochrome P450. Proc Bayl Univ Med Cent 2000;13:421-423.
9. Gillman PK. A review of serotonin toxicity data: Implications for the mechanisms of antidepressant drug action. Biol Psychiatry 2006.
10. Birmes P, Coppin D, Schmitt L, et al. Serotonin syndrome: A brief review. CMAJ 2003;168:1439-1442.
11. Mason PJ, Morris VA, Balcezak TJ. Serotonin syndrome. Presentation of 2 cases and review of the literature. Medicine (Baltimore) 2000;79:201-209.
12. Gillman PK. The serotonin syndrome. N Engl J Med 2005;352:2454-2456; author reply 2454-2456.
13. Sternbach H. The serotonin syndrome. Am J Psychiatry 1991;148:705-713.
14. Dunkley EJ, Ibister GK, Sibbritt D, et al. The Hunter Serotonin Toxicity Criteria: Simple and accurate diagnostic decision rules for serotonin toxicity. QJM 2003;96:635-642.
15. Radomski JW, Durun SM, Reveley MA, et al. An exploratory approach to the serotonin syndrome: an update of clinical phenomenology and revised diagnostic criteria. Med Hypotheses 2000;55:218-224.
16. Graham PM. Successful treatment of the toxic serotonin syndrome with chlorpromazine. Med J Aust 1997;166:166-167.
17. McDaniel WW. Serotonin syndrome: Early management with cyproheptadine. Ann Pharmacother 2001;35:870-873.
18. LoCurto MJ. The serotonin syndrome. Emerg Med Clin North Am 1997;15:
665-675.
19. Guze BH, Baxter LR Jr. The serotonin syndrome: Case responsive to propranolol. J Clin Psychopharmacol 1986;6:119-120.
20. Bismuth C, de Rohan-Chabot P, Goulon M, et al. Dantrolene—a new therapeutic approach to the neuroleptic malignant syndrome. Acta Neurol Scand Suppl 1984;100:193-198.
21. Delay J, Pichot P, Lemperiere F, et al. [A non-phenothiazine and non-reserpine major neuroleptic, haloperidol, in the treatment of psychoses.]. Ann Med Psychol (Paris) 1960;118:145-152.
22. Carbone JR. The neuroleptic malignant and serotonin syndromes. Emerg Med Clin North Am 2000;18:317-325, x.
23. Moltz DA, Coeytaux RR. Case report: Possible neuroleptic malignant syndrome associated with olanzapine. J Clin Psychopharmacol 1998;18:485-486.
24. Nyfort-Hansen K, Alderman CP. Possible neuroleptic malignant syndrome associated with olanzapine. Ann Pharmacother 2000;34:667.
25. Spalding S, Alessi NE, Radwan K. Aripiprazole and atypical neuroleptic malignant syndrome. J Am Acad Child Adolesc Psychiatry 2004;43:1457-1458.
26. Ananth J, Parameswaran S, Gunatilake S, et al. Neuroleptic malignant syndrome and atypical antipsychotic drugs. J Clin Psychiatry 2004;65:464-470.
27. Bhanushali MJ, Tuite PJ. The evaluation and management of patients with neuroleptic malignant syndrome. Neurol Clin 2004;22: 389-411.
28. Kipps CM, Fung VS, Grattan-Smith P, et al. Movement disorder emergencies. Mov Disord 2005;20:322-334.
29. Levenson JL. Neuroleptic malignant syndrome. Am J Psychiatry 1985;142:1137-1145.
30. Rusyniak DE, Sprague JE. Toxin-induced hyperthermic syndromes. Med Clin North Am 2005;89:1277-1296.
31. Tsutsumi Y, Yamamoto K, Matsuura S, et al. The treatment of neuroleptic malignant syndrome using dantrolene sodium. Psychiatry Clin Neurosci 1998;52:433-438.
32. Krause T, Gerbershagen MU, Fiege M, et al. Dantrolene—a review of its pharmacology, therapeutic use and new developments. Anaesthesia 2004;59:364-373.
33. McNeil B. Malignant hyperthermia. Br J Perioper Nurs 2005;15:376-377, 379-382.
34. McCarthy EJ. Malignant hyperthermia: pathophysiology, clinical presentation, and treatment. AACN Clin Issues 2004;15:231-237.
35. Chang KY, ting CK, Chan K, et al. Malignant hyperthermia with excellent response to small dose of dantrolene. Acta Anaesthesiol Taiwan 2004;42:241-245.
36. Christiansen LR, Collins KA. Pathologic findings in malignant hyperthermia: A case report and review of literature. Am J Forensic Med Pathol 2004;25:327-333.
37. Dressler D, Benecke R. Diagnosis and management of acute movement disorders. J Neurol 2005;252:1299-1306.
38. Houltram B, Scanlan M. Extrapyramidal side effects. Nurs Stand 2004;18:39-41.
39. Janno S, Holi MM, Tuisku K, et al. Actometry and Barnes Akathisia Rating Scale in neuroleptic-induced akathisia. Eur Neuropsychopharmacol 2005;15:39-41.
40. Lane RM. SSRI-induced extrapyramidal side-effects and akathisia: Implications for treatment. J Psychopharmacol 1998;12:192-214.
41. Sachdev P. The epidemiology of drug-induced akathisia: Part I. Acute akathisia. Schizophr Bull 1995;21:431-449.
42. Lima AR, Weiser KV, Bacaltchuk J, et al. Anticholinergics for neuroleptic-induced acute akathisia. Cochrane Database Syst Rev 2004(1):CD003727.
43. Williams SH. Medications for treating alcohol dependence. Am Fam Physician 2005;72:1775-1780.
44. Visapaa JP, Tillonen JS, Kaihovaara PS, et al. Lack of disulfiram-like reaction with metronidazole and ethanol. Ann Pharmacother 2002;36:971-974.
45. Williams CS, Woodcock KR. Do ethanol and metronidazole interact to produce a disulfiram-like reaction? Ann Pharmacother 2000;34:255-257.
46. Motte S, Vincent JL, Gillet JB, et al. Refractory hyperdynamic shock associated with alcohol and disulfiram. Am J Emerg Med 1986;4:323-325.
47. Stowell A, Johnsen J, Ripel A, et al. Diphenhydramine and the calcium carbimide-ethanol reaction: A placebo-controlled clinical study. Clin Pharmacol Ther 1986;39:521-525.
48. Thomsen HS, Morcos SK. Contrast media and the kidney: European Society of Urogenital Radiology (ESUR) guidelines. Br J Radiol 2003;76:513-518.
49. Barrett BJ, Parfrey PS. Preventing nephropathy induced by contrast medium. N Engl J Med 2006;354:379-386.
50. Rudnick MR, Goldfarb S, Wexler L, et al. Nephrotoxicity of ionic and nonionic contrast-media in 1196 patients—A randomized trial. Kidney International 1995;47:254-261.
51. Merten GJ, Burgess WP, Gray LV, et al. Prevention of contrast-induced nephropathy with sodium bicarbonate: A randomized controlled trial. JAMA 2004;291:2328-2334.
52. Malhotra S, Karan RS, Pandhi P, et al. Drug related medical emergencies in the elderly: Role of adverse drug reactions and noncompliance. Postgrad Med J 2001;77:703-707.
53. Heininger-Rothbucher D, Bischinger S, Ulmer H, et al. Incidence and risk of potential adverse drug interactions in the emergency room. Resuscitation 2001;49:283-288.
54. Goldberg RM, Mabee J, Chan L, et al. Drug-drug and drug-disease interactions in the ED: Analysis of a high-risk population. Am J Emerg Med 1996;14:447-450.
55. Dennehy CE, Kishi DT, Louie C. Drug-related illness in emergency department patients. Am J Health Syst Pharm 1996;53:1422-1426.
56. Cohen MM, Kimmel NL, Benage MK, et al. Medication safety program reduces adverse drug events in a community hospital. Qual Saf Health Care 2005;14:169-174.
57. Cut medication errors in half with ED pharmacist. ED Manag 2002;14:88-90.
58. Langdorf MI, Fox JC, Marwah RS, et al. Physician versus computer knowledge of potential drug interactions in the emergency department. Acad Emerg Med 2000;7:1321-1329.
59. Szarfman A, Machado SG, O'Neill RT. Use of screening algorithms and computer systems to efficiently signal higher-than-expected combinations of drugs and events in the US FDA's spontaneous reports database. Drug Saf 2002;25:381-392.
60. Benjamin DM. Reducing medication errors and increasing patient safety: Case studies in clinical pharmacology. J Clin Pharmacol 2003;43:768-783.
61. Wood AJ, Stein CM, Woosley R. Making medicine safer—the need for an independent drug safety board. N Engl J Med 1998;339:1851-1854.
62. Gopalani V. Web sites on adverse drug reactions and drug interactions. Indian J Dermatol Venereol Leprol 2005;71:60-62.
Warfarin (Coumadin) is the most commonly used oral anticoagulant. It was synthesized by and named after the Wisconsin Alumni Research Foundation in 1948.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.