Unapproved abbreviations most problematic patient safety goal
Unapproved abbreviations most problematic patient safety goal
Read-back of verbal orders also a common challenge for pharmacy
Almost half of hospitals trying to be compliant with patient safety goals of the Joint Commission on Accreditation of Healthcare Organizations (Joint Commission) still struggle with using unapproved abbreviations, says a field surveyor with the Joint Commission.
Sonja A. Nisson, PharmD, also regional manager of pharmacy and diabetes at Asante Health System in Medford, OR, spoke about the top non-compliant medication-related National Patient Safety Goals (NPSG) in 2005. Her remarks were made at the American Society of Health-System Pharmacists Midyear Clinical Meeting held in early December in Las Vegas. (For list of top noncompliant medication-related NPSG in 2005, see below.)
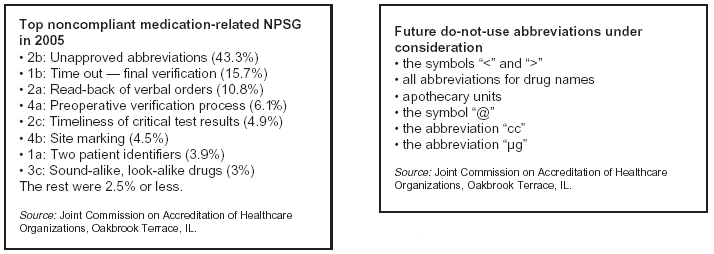
Using unapproved abbreviations is challenging for health care organizations, Nisson says. The Joint Commission found that health care organizations still are noncompliant with NPSG 2b ("Standardize a list of abbreviations, acronyms, and symbols that are not to be used throughout the organization") 43.3% of the time. "Organizations are having difficulty with it, but it’s also paramount to safety," she notes.
Most compliance problems are with the abbreviations "QD" and "MS," Nisson says. Other information related to this NPSG includes:
- It applies to all types of orders. "It is a medication-related documentation," she says.
- It does not apply to trade names.
- Once you have an abbreviation, clarification has no impact on compliance with this NPSG.
- Preprinted forms should be in 100% compliance.
"Certain physicians have preprinted orders that are standard for them, and they have to come in and sign them. You have to have periodic review of those," Nisson says.
Some hospitals do a good job of reviewing the forms and removing all the abbreviations. Then somehow older versions seem to surface. "Once they show up, they seem to proliferate everywhere," she says. "While this has a number of other serious implications, one of the problems is they almost always contain unapproved abbreviations."
One problem Nisson frequently sees in pharmacy or nursing areas is free-text keyboard entry where abbreviations have been commonly used. "People who have been in practice longer seem to have more trouble than new practitioners of including the abbreviation instead of the whole word."
At this time, this NPSG doesn’t apply to computer-generated documentation where the abbreviations are hard-wired into the system. Nisson, however, thinks this will change in the future.
The third top noncompliant medication-related NPSG in 2005 was 2a: "For verbal or telephone orders or for telephonic reporting of critical test results, verify the complete order or test result by having the person receiving the order or test result read-back the complete order or test result." Health care organizations were found to be noncompliant with this NPSG 10.8% of the time.
Try to minimize the verbal orders in your organization, Nisson says. One problem has been with on-site physicians giving medication orders to nurses as they go past the nursing station. A nurse may be on the telephone or handling another task; by the time the nurse is able to read back the order, the physician has left the area. For this reason, some hospitals are not allowing on-site physicians to do verbal orders.
Although the emphasis on this NPSG has been on nursing, pharmacists have problems with this, too, Nisson says. This can relate to the hospital pharmacy, pharmacists on decentralized units, and throughout the organization with other specialties. "When I walk through pharmacies, even during a survey, I have heard pharmacists [repeat back an order on the telephone to a physician] and hang up the phone without writing it down," Nisson says. "In some cases, they have then turned and verbally told the technician [the order]. Sometimes it will be 10-15 minutes before that order is committed to writing."
Measuring critical test results
Another problematic NPSG has been 2c: "Measure, assess and, if appropriate, take action to improve the timeliness of reporting, and the timeliness of receipt by the responsible licensed caregiver, of critical test results and values." Health care organizations were found to be noncompliant with this NPSG 4.9% of the time in 2005.
Health care organizations must determine what they consider to be "critical test results," Nisson says. Also, they should determine which test results are the responsibility for the pharmacist to report.
"There have to be data to measure," she says. Knowing how to measure, however, can be a sticky point.
Nisson has seen people measure the time interval from when the specimen got to the lab to the time the final test result was available. "That really isn’t the intent here," she says. "The intent is to get real information to the people who need it when they need it." Staff instead should be measuring the time it takes from when the lab is ordered and is completed to when it gets to the person who makes the decision about the patient’s care. "That person can be the pharmacist, if he or she is doing pharmacokinetics and that type of thing. That is the interval you want to measure."
This NPSG also has an emphasis on improving the process. "How do you know when you have an opportunity to improve it?" Nisson asks. One way is to look at this as a risk point analysis. Another way is to ask when has this delay of having the results to the person who needs it been a factor in the outcome or the delay in treatment of the patient. "You can use your own data to see if the time frames you have are working."
Patients should have necessary care all the time, and some hospitals find their system works well 8 to 5, but not as well on evenings and weekends when reaching a person responsible for a patient’s care is more difficult. That’s another way to look at this, Nisson says. Does it take longer in the off-hours to make critical changes based on test results?
Three percent of surveyed organizations had difficulty being compliant with NPSG 3c: "Identify and, at a minimum, annually review a list of look-alike/sound-alike drugs used in the organization, and take action to prevent errors involving the interchange of these drugs."
In surveys conducated as recently as September, Nisson found organizations that still did not have a list. Others looked at the list, found errors, put measures in place to correct errors, and then did not repeat the process. "Sustain what you are going to do," she says.
Having the list is only step one, she explains. "The second step is to put those safety measures in place. Some people do segregation where they have [the look-alike/sound-alike drugs] in different boxes. It’s up to you what is necessary to do."
The safety measures set up in the pharmacy, however, are not always in place in night lockers or medication storage areas on nursing units. "I have even gone to satellite pharmacies outside the main pharmacy and not seen the same safety measures employed. Decide your safety strategies and employ them consistently, " Nisson advises.
She also spoke of the importance of NPSG 3b: "Standardize and limit the number of drug concentrations available in the organization."
"This requires that we only have concentrations that have a therapeutic necessity," she says. "There have been many sentinel events related to inadvertent administration of the wrong concentration of drugs. So when you have titration, weight-based dosing, or other types of dosing, it is important to vary the rate and not the concentration."
Pharmacists can assist in units such as pediatrics and the intensive care, both of which typically have more challenges regarding this issue. "Pharmacy needs to help where we have rate variances, to develop the rate charts for nurses. We are the experts in that area and it is something we can do well." She mentioned the Broselow Cart Medication System, in which excellent modifications have been made to have pre-mixed IVs and set concentrations and to vary the rate based upon the weight and size of the patient.
Organizations shouldn’t have trouble finding out how compliant they are with this and other NPSGs, Nisson says. "It isn’t really rocket science to find out what is going on. If you do your own tracers, if you follow patients through all of their venues of care on a day-to-day basis, you’ll know if you are doing these things."
On her surveys, Nisson often talks to patients and asks their opinion about the organization’s patient safety. "Believe me, with the education out there about patient safety, and how much our patients want and expect to be safe, they will give you answers about how well you are doing in your own organization."
(Editor’s note: In the next issue of Drug Formulary Review, Nisson will give advice on complying with NPSG 8: "Accurately and completely reconcile medications across the continuum of care.")
Almost half of hospitals trying to be compliant with patient safety goals of the Joint Commission on Accreditation of Healthcare Organizations (Joint Commission) still struggle with using unapproved abbreviations, says a field surveyor with the Joint Commission.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
