2010 Salary Survey Results: IRBs ride out tough times, see hope for turnaround
IRBs ride out tough times, see hope for turnaround
Fewer raises, rising workloads cited
As institutions continue to weather the economic downturn, the toll on IRB offices is showing. The trends of previous years fewer raises, more job cuts, increasing workloads continued in 2010, according to responses to IRB Advisor's annual Salary Survey.
But there's room for optimism in some of the results. Some previous concerns, including pay cuts, seem to have subsided, and more respondents in this year's survey reported higher salaries than in the past.
At Vanderbilt University, Julie Ozier, MHL, CHRC, CIP, associate director of the Human Research Protection Program, says she's cautiously optimistic after signs of a modest recovery this year. In 2010, IRB staff members in her office were eligible for raises of up to 3%, after a year in which no raises were given. Another ½% increase was scheduled in January.
"Our medical center feels like they're recovering a little bit," Ozier says. "I don't know if we're starting to see a turn, but we're very hopeful."
The 2010 salary survey drew responses from 52 people IRB coordinators, administrators and managers, directors of human research protection and other professionals involved in the oversight of human subjects research.
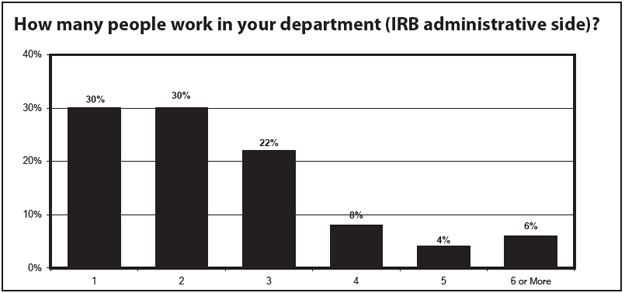
Those reporting were primarily from smaller offices 82% reported that three or fewer people worked in IRB administration in their departments. Most institutions reporting were in hospitals (54%) or academia (40%), and were located in either medium-sized cities (48%) or urban areas (31%).
The salary spread in this year's survey appears larger than in the past. Over the past several years, the largest percentage of respondents made between $40,000 and $49,999. This year, there were as many people in that group as there were making $70,000-79,999 and $100,000-$129,999. Overall, a larger percentage of 2010 respondents made more than $50,000 (71%) and more than $70,000 (46%) than had been reported in previous years.
Mixed signals on raises
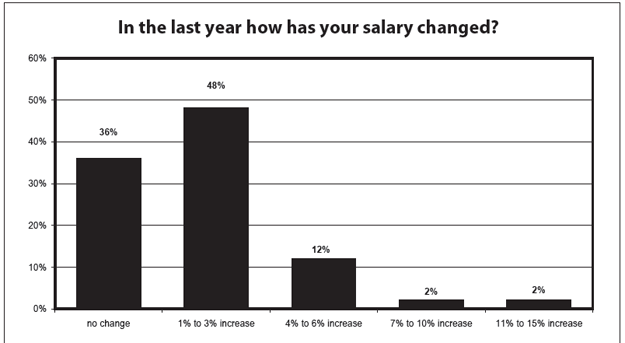
Nearly half of all respondents reported this year that they received a 1% to 3% raise about the same percentage who reported a similar raise in 2009 and more than the 44% who received it in 2008. And unlike the previous two years, no 2010 respondents reported a decline in salary.
But the percentage of IRB professionals who reported no change in salary has climbed steadily since 2007, when just 16% said they'd had no change, to 36% this year.
At the University of Southern California in Los Angeles, staff members have alternated between raises and no raises over the past few years, says Susan Rose, PhD, executive director of the Office for the Protection of Research Subjects.
"Last year, we had a 2% raise and the year before that, no raise," she says. "This year, we're looking at none again. But we also kept staff."
Rose says her office has cut back in other areas, including curtailing travel, in order to save jobs. Both she and Ozier say they're trying to continue to provide professional development for their staff, but are focusing on conferences closer to home instead of sending people across the country.
Rose says she was surprised by one of the findings in the salary survey reports of increased workloads in IRB offices.
Three-quarters of all the IRB professionals surveyed in 2010 reported that their workload had increased in the past year up from 68% in 2009. Sixty-seven% of 2010 respondents said they worked more than 40 hours a week.
At USC, they've actually seen fewer studies in the past year, particularly big national studies funded by pharmaceutical companies, Rose says.
Vanderbilt's IRB, by comparison, has seen steadily rising numbers of new submissions more than 800 a year since 2008, Ozier says.
Workload concerns cited in the survey may be partly attributable to lost staff. In 2010, about 22% of all of those surveyed said their offices lost staff, up from 15% in 2009 and 12% in 2008.
Other results from the 2010 salary survey:
- Demographics 85% of respondents were women. Age distribution was fairly wide, with about half of those surveyed between the ages of 26 and 50.
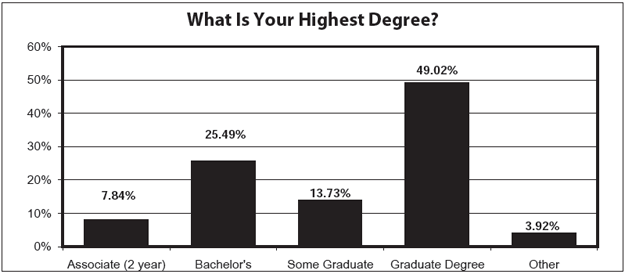
- Education and experience Nearly half of all the respondents have a graduate degree. For another 25%, the highest degree obtained was a bachelor's degree. The most common certification held is a Certified IRB Professional (CIP). More than three-quarters of all those surveyed have been in the human subjects protection field 12 years or less.
Personnel issues abound
Those who responded to the survey also listed some of the biggest personnel issues they face. Among the most common themes:
- Maintaining qualified, educated staff during lean times;
- Lack of resources, even as research programs grow and workloads increase;
- Burnout and lack of morale due to decreasing raises.
Ozier and Rose say there are steps that IRB offices can take to combat these problems.
At Vanderbilt, administrators are looking at ways to shift resources in order to use protocol analysts more efficiently.
"Currently, analysts are assigned to a committee and work only with that committee," Ozier says. "We're looking at maybe creating a pool of protocol analysts to spread the volume (of work) out more equitably."
To deal with morale issues, she schedules team-building exercises and retreats. When it's not possible to give raises, providing employees with more work options can help soften the financial blow.
"We allow them to do flexible schedules for example, we allow them to do four 10-hour shifts in a week instead of five eight-hour shifts," Ozier says. "We're doing more things electronically, so it's allowing us to be able to work from home. All the protocol analysts can set a schedule where once a week they can work from home, and that's working pretty well."
Rose says that USC provides free tuition for employees who are working on higher level degrees. "There's a wonderful master's degree in regulatory science that a lot of them take."
Identify the unnecessary
As far as managing an IRB office in tough economic times, Rose says the key is learning not to do what you don't have to do.
For example, at USC, the IRB no longer looks at every adverse event report that comes in.
"If they are internal ones, if they meet the criteria for an unanticipated problem, look at it," she says. "If it's from another site and doesn't (meet those criteria) don't."
While automating IRB operations can streamline the process and save staff time, Rose notes that it doesn't necessarily result in fewer employees.
"We now have two full-time IT (information technology) people so it's a trade of types of employees," she says.
Rose advises looking carefully at the regulations and ensuring that the IRB is reviewing studies at the right level.
"If you review everything at full board or expedited when you don't have to, you don't know what you're doing," she says.
She's also working on changing policies regarding exempt studies so that the office doesn't waste time on activities that aren't really human subjects research. In many cases, she says, boards are reviewing things like polls and program evaluations that don't require IRB review.
"For example, USC and The L.A. Times have been involved in polls and none of the exempt categories use the word 'polls,'" Rose says. "We're creating a category for things that are clearly no risk, where there's no federal funding and are clearly exempt."
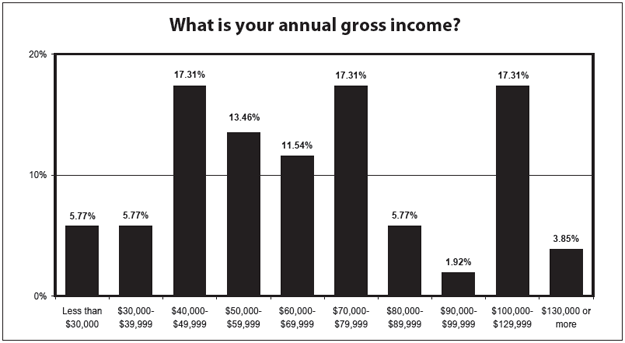
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
