Treatment of Small Intestinal Bacterial Overgrowth with Botanical Therapies Equivalent to Rifaximin
By Carrie Decker, ND
Founder and Medical Director, Blessed Thistle, Madison, WI
Dr. Decker reports no financial relationships relevant to this field of study.
Synopsis: Small intestinal bacterial overgrowth (SIBO) is a condition associated with symptoms of irritable bowel syndrome and extraintestinal manifestations. This study investigates the effectiveness of the antibiotic rifaximin compared to botanical combination therapies for the treatment of SIBO as diagnosed by lactulose breath testing.
Source: Chedid V, et al. Herbal therapy is equivalent to rifaximin for the treatment of small intestinal bacterial overgrowth. Glob Adv Health Med 2014;3:16-24.
Summary Points
- A retrospective chart review of 104 patients undergoing treatment for small intestinal bacterial overgrowth (SIBO) with a 30-day course of rifaximin or herbal antimicrobials found that herbal treatments were at least equally effective to rifaximin for the treatment of SIBO.
- Herbal antimicrobial combination therapies were similarly effective to triple antibiotic therapy in resolving rifaximin-refractive SIBO.
- Adverse effects were more common and severe in individuals treated with rifaximin.
The expansion of intestinal microbiota from the predominant location in the colon to the small intestine has been studied as one cause of irritable bowel syndrome (IBS).1 The population of microbiota in the small intestine creates gas as a byproduct of fermentation, leading to typical IBS symptoms, and may activate host mucosal immunity, leading to a wide array of systemic symptoms. Lactulose is not absorbed or used by the gastrointestinal tract in humans, and is metabolized by bacteria into hydrogen and methane gas. The amount of gas produced can be measured by a lactulose breath test (LBT) and is a common procedure for diagnosis of small intestinal bacterial overgrowth (SIBO).2 Rifaximin, an antibiotic with little to no systemic absorption, is the most commonly used antibiotic for the treatment of SIBO.3 Many individuals with IBS seek integrative treatment options, but the use of botanical antimicrobials and other oral supplements is common yet not well studied for effectiveness.4
In this retrospective case review, 104 individuals with SIBO symptoms and diagnosis via a positive LBT were treated for SIBO, including a post-treatment LBT. Symptoms suggestive of SIBO included abdominal discomfort, cramping, bloating, flatulence, eructation, diarrhea, symptoms worsened via food consumption, and low serum B12. Exclusion criteria included patients < 18 years of age or > 85 years of age and antibiotic use within 3 months. A subset of 24 patients who had completed initial treatment with rifaximin and were still found to have a positive LBT were further investigated for response to either herbal or triple antibiotic therapies. (See Figure 1.)
|
Figure 1: Retrospective Case Review |
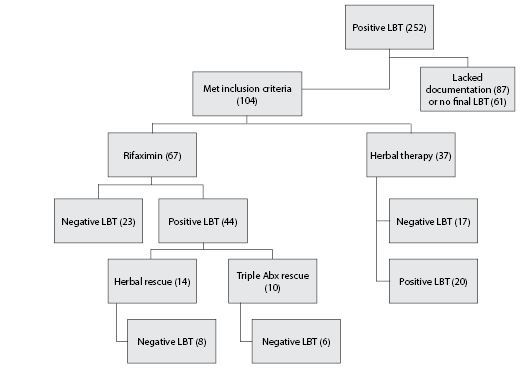 |
Patients were treated for SIBO with rifaximin or herbal antimicrobial protocols based on individual treatment preference. Sixty-seven patients completed treatment with rifaximin and 37 completed treatment with herbal therapies. The herbal products used were the commercially available products Dysbiocide and FC Cidal (Biotics Research Laboratories) or Candibactin-AR and Candibactin-BR (Metagenics). The herbal products were taken at a dosage of two capsules each twice daily. Rifaximin was dosed at two 200 mg tablets three times daily (total 1200 mg/day). Each treatment was for 4 consecutive weeks with a follow-up LBT upon completion. Rifaximin non-responders were further treated by one of the herbal protocols or a triple antibiotic protocol (clindamycin 300 mg, metronidazole 250 mg, and neomycin 500 mg each three times daily) for 4 additional weeks. (See Table 1.)
Table 1: Herbs in Commercially Available Products |
|||
|
FC Cidal |
Dysbiocide |
Candibactin-AR |
Candibactin-BR |
|
|
|
|
Of the patients completing treatment with rifaximin, 23 of 67 (34%) were found to have a negative LBT with follow-up testing compared to 17 of 37 (46%) of those receiving herbal therapies (P = 0.23). The odds ratio of having a negative LBT after treatment with herbal therapies compared to rifaximin was 1.63 (confidence interval [CI], 0.72-3.70; P = 0.24) and 1.85 (CI, 0.77-4.41; P = 0.17) after adjustment for age, gender, SIBO risk factors (such as known gastrointestinal motility disorder or chronic proton-pump inhibitor use), and IBS status (diagnosed by Rome III criteria). These comparisons show equivalency.
Fourteen of the individuals not responsive to rifaximin treatment were offered herbal therapies, with 8 of 14 (57.1%) having a negative LBT after herbal treatment, while 10 of the individuals non-responsive to rifaximin were offered triple antibiotics with 6 of 10 (60%) having a negative LBT after triple antibiotic treatment (P = 1.0). The population of individuals non-responsive to herbal therapies was not investigated for response to further treatments. The effect of treatments on symptoms was not assessed at completion of treatment.
There was no significant difference in age, gender distribution, IBS-subtype distribution, or risk factors for SIBO in the populations completing treatment with rifaximin vs herbal therapies. However, there was a higher percentage of females than males in both groups (71% in the rifaximin arm and 78% in the herbal arm).
Adverse effects were reported in six individuals in the rifaximin treated arm, including one case of anaphylaxis, two cases of hives, two cases of diarrhea, and one case of Clostridium difficile (post-treatment). One case of diarrhea (non-C. difficile) was reported in the herbal therapy arm. The rates of adverse effects were not analyzed statistically. This study did not assess the number of study dropouts from the two arms separately. Initially 252 individuals with a positive LBT were recommended treatment but only 104 both completed the treatment protocol and had a follow-up LBT. Of the individuals who did not complete both treatment and a follow-up test, 87 individuals lacked proper documentation of treatment and 61 did not have a follow-up LBT.
Commentary
IBS is a common condition, with most prevalence estimates in North America ranging from 10-15%.5 IBS is often seen to be chronic or recurrent in individuals who experience it. Individuals with IBS report lower (worse) health-related quality of life scores than individuals with gastroesophageal reflux disease, asthma, and migraines.6 SIBO has been increasingly of interest as a contributor to symptoms of IBS,7 thereby leading to many investigations of the use of antibiotic therapies for treatment.
The LBT is one of the primary tools used to evaluate for the presence of SIBO,8 and results have shown relationships with IBS. A recent meta-analysis showed the prevalence of a positive LBT of 54% (95% CI, 32%-76%) in individuals with IBS.9 Odds ratios for a positive LBT in individuals meeting the criteria for IBS range from 3.45 to 9.64 when compared to healthy asymptomatic controls.9,10 The treatment and resolution of a positive LBT finding is associated with improvement in symptoms of IBS.11
Counter to this, a study comparing the amount of bacteria cultured from a small intestinal biopsy (the gold standard for diagnosis of SIBO) have found poor correlation with LBT hydrogen and methane results.12 Although various markers from the LBT are evaluated for the diagnosis of SIBO, the findings of this study showed a sensitivity of 0 to 33% depending on which parameter was assessed. A positive biopsy finding indicating the presence of SIBO is when ≥ 10 CFU/mL bacteria are present when cultured. One explanation for this discrepancy is that a bacterial culture is a poor environment to stimulate the small intestinal environment, and in vivo the microbial balance may be far different. However, it does shed light on the fact that there is possibly significant error in LBT for the diagnosis of SIBO.
Other studies have evaluated the use of rifaximin as a therapy for patients with SIBO, as demonstrated by positive LBT. Standard dosing strategies are between 1200 mg/day and 550 mg three times a day for 10-14 days.13 Rifaximin lacks FDA approval for the treatment of SIBO and therefore is costly, ranging from $600-1250 per month. Other antibiotics that have been studied for the purpose of treating SIBO include neomycin, tetracycline, amoxicillin clavulanate, metronidazole, and fluoroquinolones.14
This study had some limitations, in addition to the major methodological flaw that it was a retrospective chart review with self-selected treatment choices rather than a double-blind, randomized, controlled trial. Although this study provided a general look at the treatment of SIBO with herbal antimicrobials and saw positive results compared to rifaximin, the results did not distinguish the success rate with the two different herbal antimicrobial protocols. The herbal antimicrobials each were comprised of more than four different herbs with known antimicrobial effects, but there were very little similarities in the formulations.
There also was no report of whether symptoms associated with SIBO were resolved at the end of the treatment course, or if the individuals treated were able to maintain a symptom-free/negative LBT status after discontinuing rifaximin or herbal antimicrobial treatment. A high recurrence rate of SIBO has been demonstrated, with rates of 12.6%, 27.5%, and 43.7% at 3, 6, and 9 months, respectively, after successful (breath test negative) treatment with rifaximin for 1 week.15 Another study found that after a 14-day treatment course with rifaximin, IBS-associated symptoms (including bloating, flatulence, diarrhea, and pain) were improved for a period of 3 months after treatment.16 The 30-day treatment course with rifaximin used in the study was longer than in both of these studies, and it is unclear if the longer treatment duration would be more effective in reducing recurrence rates.
Additionally, the positive findings of the lactulose breath test did not distinguish whether positive findings were due to elevated hydrogen, methane gas, or both. It has been observed that constipation-type IBS is associated with methanogens and is more difficult to resolve with rifaximin.17,18 Rifaximin has been studied more extensively for the treatment of non-constipation type IBS.19 Significant improvements in constipation-type IBS have been seen with neomycin,20 one of the triple antibiotics used in individuals who did not have a negative LBT after rifaximin treatment. The combination of neomycin with rifaximin has been shown to be most effective for normalization of a high methane-positive LBT.21
The authors did not state whether other diseases that may have similar gastrointestinal symptoms had been ruled out in the participants in this study. The symptoms suggestive of SIBO (abdominal discomfort, cramping, bloating, flatulence, eructation, diarrhea, symptoms worse with eating, and low serum B12) also may be symptoms of conditions such as celiac disease or inflammatory bowel disease. Although the positive LBT has been shown to demonstrate SIBO, there may be other underlying problems in individuals non-responsive to treatment, or the LBT can be a false positive.
Finally, the authors did not note whether participants followed specific dietary interventions that are commonly recommended for individuals with IBS or SIBO. A diet low in fermentable oligosaccharides, disaccharides, monosaccharides, and polyols (FODMAPs) is commonly recommended for individuals with IBS and has been shown to have efficacy in the reduction of symptoms,22 while a high FODMAPs diet has been shown to affect the amounts of breath hydrogen and methane.23 Individuals who are motivated to seek treatment for IBS often follow gluten-free or dairy-free diets, or diets with low amounts of starch and complex sugar-sourced carbohydrates. The use of other treatments also was not controlled in the study participants. Supplement and over-the-counter medication use is common among individuals with IBS and includes probiotics, digestive enzymes, and antidiarrheal or laxative agents.
Overall, this study was the first to compare the use of rifaximin to herbal antimicrobials for the treatment of SIBO. However, as it is not a prospective study and many variables that may have had an effect were not controlled, it is difficult to come to a clinically relevant conclusion. That said, as adverse effects were minimal with herbal treatments, and resolution of a positive LBT test was found to be equivalent with both therapies, if individuals are interested in using botanical treatments for this purpose, there is no evidence to indicate that this should be discouraged. Further investigation of the efficacy of herbal antimicrobials and a 30-day treatment course of rifaximin that addresses the aforementioned items will offer further insight into what may be most effective for the treatment of SIBO and prevention of IBS recurrence.
REFERENCES
- Lin HC, Pimentel M. Bacterial concepts in irritable bowel syndrome. Rev Gastroenterol Disord 2005;5(Suppl 3):S3-9.
- Park JS, et al. [Usefulness of lactulose breath test for the prediction of small intestinal bacterial overgrowth in irritable bowel syndrome]. Korean J Gastroenterol 2010;56:242-248.
- Cremonini F, Lembo A. Rifaximin for the treatment of irritable bowel syndrome. Expert Opin Pharmacother 2012;13:433-440.
- Kong SC, et al. The incidence of self-prescribed oral complementary and alternative medicine use by patients with gastrointestinal diseases. J Clin Gastroenterol 2005;39:138-141.
- Saito YA, et al. The epidemiology of irritable bowel syndrome in North America: A systematic review. Am J Gastroenterol 2002;97:1910-1915.
- Frank L, et al. Health-related quality of life associated with irritable bowel syndrome: Comparison with other chronic diseases. Clin Ther 2002;24:675-689.
- Wall GC, et al. Irritable bowel syndrome: A concise review of current treatment concepts. World J Gastroenterol 2014;20:8796-8806.
- Ghoshal UC. How to interpret hydrogen breath tests. J Neurogastroenterol Motil 2011;17:312-317.
- Ford AC, et al. Small intestinal bacterial overgrowth in irritable bowel syndrome: Systematic review and meta-analysis. Clin Gastroenterol Hepatol 2009;7:1279-1286.
- Shah ED, et al. Abnormal breath testing in IBS: A meta-analysis. Dig Dis Sci 2010;55:2441-2449.
- Pimentel M, et al. Normalization of lactulose breath testing correlates with symptom improvement in irritable bowel syndrome. A double-blind, randomized, placebo-controlled study. Am J Gastroenterol 2003;98:412-419.
- Ghoshal UC, et al. Breath tests in the diagnosis of small intestinal bacterial overgrowth in patients with irritable bowel syndrome in comparison with quantitative upper gut aspirate culture. Eur J Gastroenterol Hepatol 2014;26:753-60.
- Saadi M, McCallum RW. Rifaximin in irritable bowel syndrome: Rationale, evidence and clinical use. Ther Adv Chronic Dis 2013;4:71-75.
- Basseri RJ, et al. Antibiotics for the treatment of irritable bowel syndrome. Gastroenterol Hepatol (N Y) 2011;7455-493.
- Lauritano EC, et al. Small intestinal bacterial overgrowth recurrence after antibiotic therapy. Am J Gastroenterol 2008;103:2031-2035.
- Meyrat P, et al. Rifaximin treatment for the irritable bowel syndrome with a positive lactulose hydrogen breath test improves symptoms for at least 3 months. Aliment Pharmacol Ther 2012;36:1084-1093.
- Hwang L, et al. Evaluating breath methane as a diagnostic test for constipation-predominant IBS. Dig Dis Sci 2010;55:398-403.
- Majewski M, McCallum RW. Results of small intestinal bacterial overgrowth testing in irritable bowel syndrome patients: Clinical profiles and effects of antibiotic trial. Adv Med Sci 2007;52:139-142.
- Pimentel M, et al. Rifaximin therapy for patients with irritable bowel syndrome without constipation. N Engl J Med 2011;364:22-32.
- Pimentel M, et al. Neomycin improves constipation-predominant irritable bowel syndrome in a fashion that is dependent on the presence of methane gas: Subanalysis of a double-blind randomized controlled study. Dig Dis Sci 2006;51:1297-1301.
- Low K, et al. A combination of rifaximin and neomycin is most effective in treating irritable bowel syndrome patients with methane on lactulose breath test. J Clin Gastroenterol 2010;44:547-550.
- de Roest RH, et al. The low FODMAP diet improves gastrointestinal symptoms in patients with irritable bowel syndrome: A prospective study. Int J Clin Pract 2013;67:895-903.
- Ong DK, et al. Manipulation of dietary short chain carbohydrates alters the pattern of gas production and genesis of symptoms in irritable bowel syndrome. J Gastroenterol Hepatol 2010;25:1366-1373.
Small intestinal bacterial overgrowth is a condition associated with symptoms of irritable bowel syndrome and extraintestinal manifestations. This study investigates the effectiveness of the antibiotic rifaximin compared to botanical combination therapies for the treatment of the condition as diagnosed by lactulose breath testing.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
