Treatment of Neurocysticercosis
Treatment of Neurocysticercosis
Abstract & Commentary
Synopsis: A 10-day treatment course of albendazole and dexamethasone for patients with seizures and neurocysticercosis with viable cysts significantly reduced the incidence of subsequent seizures.
Source: Garcia HH, et al. A trial of antiparasitic treatment to reduce the rate of seizures due to cerebral cysticercosis. N Engl J Med. 2004;350:249-258.
Garcia and colleagues randomized 120 adults with neurocysticercosis with viable cysts and a history of seizures to receive either placebo or treatment with albendazole (400 mg twice daily) and dexamethasone (2 mg every 8 h) for 10 days. The patients were a highly selected group (see inclusion and exclusion criteria in Table 1). Patients with cysts with signs of inflammation as determined by the presence of edema or contrast enhancement were included only if intracystic contents had a CT density similar to that of cerebrospinal fluid with confirmation of its liquid nature by MRI. Inflamed lesions were analyzed separately.

The primary end point of the study was seizure control. All patients received adequate anticonvulsant therapy, with monitoring of serum concentrations. Anticonvulsant therapy was discontinued after a 2-month taper in patients who were seizure-free for 1 year. Study participation was discontinued after 6 seizure-free months after discontinuation of therapy.
The frequency of adverse effects did not differ between the 2 treatment arms except for abdominal pain, which was reported by 8 albendazole recipients and no placebo recipient. Two albendazole recipients and 1 placebo recipient had seizures with generalization during therapy; partial seizures occurred in 8 and 5, respectively.
Albendazole recipients had 46% fewer total seizures during the 2-30 months post-therapy follow-up period, but this difference was not statistically significant (P = .30). There was, however, a significant, 67% reduction in the rate of seizures with generalization in albendazole recipients (P = .01).
MRI performed 6 months after placebo administration demonstrated that 243 of 279 cysts (87%) in 54 patients persisted unchanged. In contrast, only 79 of 192 (41%) were unchanged in the 55 albendazole recipients who underwent imaging, a difference that was significant (P < .0001). Cysts showing signs of inflammation at study entry were also more likely to have resolved in the albendazole recipients (49% vs 21%; P = .013). Twenty-one (38%) in the albendazole group and 8 (15%) in the placebo group were free of active lesions (P = .007).
Patients with no active brain lesions 6 months after treatment had 62% fewer seizures than in those with at least one active lesion (P = .24).
Comment by Stan Deresinski, MD, FACP
Neurocysticercosis is the most common parasitic infection of the central nervous system affecting humans and the most frequent cause of post-childhood-onset seizures in most of the developing world. Cerebral cysticercosis may be the most common cause of severe burns in parts of New Guinea and Irian Jaya as a consequence of contact with fire during seizures.1,2
Cysticercosis is commonly encountered in the United States as well. A study involving 11 university-affiliated emergency departments in the United States found that 2.1% of patients presenting with seizures who underwent cerebral imaging had cysticercosis as the cause.3 The infection was associated with Hispanic ethnicity, immigrant status, and exposure to areas where cysticercosis is endemic.3 The crude mortality over 12 years attributed to cysticercosis in California is 3.9 per million population.4
Entry into this study required a history of seizures. Between 50% and 80% of patients with parenchymal brain cysts or calcifications have seizures,5 but not all patients present with seizure. Some cases are discovered serendipitously when an individual undergoes brain imaging for unrelated reasons. In addition, neurocysticercosis may also present with headache. Some patients may have intracranial hypertension, hydrocephalus, or both due to mass effect from a cyst, from inflammation, from fibrosis, or from some combination. Rarely, a free-floating intraventricular cyst may intermittently obstruct the third ventricle.
Massive cerebral infection may lead to presentation with acute encephalitis. Approximately 1% of patients have cystic involvement of the spinal cord due to either an intramedullary or subarachnoid location. Meningitis may occur, often with an eosinophilic pleocytosis. Extraneural cysts may present in muscle and other soft tissues, as well as in ocular tissues.
The imaging diagnosis of cysticercosis is easy when multiple cysts are present and confirmed by the identification of a scolex within a cyst by MRI. Multiple calcifications are consistent with old, resolved infection. Multiple stages may be simultaneously present. Incipient death of a cyst is heralded by thickening of the cyst wall, contrast enhancement with or without surrounding edema, and increasing density of the cyst fluid.
The serological test used in this study, EITB, has been demonstrated to have very high sensitivity and specificity in patients with multiple cerebral lesions. The test, unfortunately, has significantly lower sensitivity in patients in whom the diagnosis may be most difficultthose with a single cerebral lesion.
No previous study has convincingly demonstrated benefit from anticysticercal therapy. A clear benefit appears to accrue with regard to a reduction of seizure occurrence from treatment with albendazole and dexamethasone in patients with viable cerebral cysts. This trial does not provide data by which to judge therapeutic benefit in a variety of other circumstances, such as in the patient who presents with headache alone. It also provides no evidence to support longer or shorter courses of therapy, therapy without adjunctive dexamethasone, or retreatment in cases in which patients have apparently failed previous courses of therapy. It also does not address the efficacy of praziquantel therapy or the possibility that the combination of praziquantel and albendazole may provide better results than the use of one or the other alone.
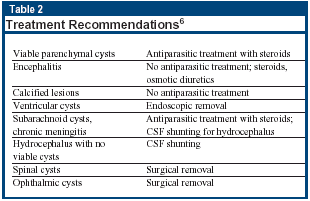
A consensus panel has recently provided recommendations on the management of other manifestations of cysticercosis (see Table 2).6
Dr. Deresinski, Clinical Professor of Medicine, Stanford; Associate Chief of Infectious Diseases, Santa Clara Valley Medical Center, is Editor of Infectious Disease Alert.
References
1. Bending JJ, Catford JC. Epidemic of burns in New Guinea due to cerebral cysticercosis. Lancet. 1983;1:922.
2. Margono SS, et al. Cysticercosis in Indonesia: Epidemiological aspects. Southeast Asian J Trop Med Public Health. 2001;32(Suppl 2):79-84.
3. Ong S, et al. Neurocysticercosis in radiographically imaged seizure patients in U.S. emergency departments. Emerg Infect Dis. 2002;8:608-613.
4. Sorvillo FJ, et al. Cysticercosis-related deaths, California. Emerg Infect Dis. 2004. Online.
5. Garcia HH, et al. Taenia solium cysticercosis. Lancet. 2003;362:547-556.
6. Garcia HH, et al. Current consensus guidelines for treatment of neurocysticercosis. Clin Microbiol Rev. 2002;15:747-756.
A 10-day treatment course of albendazole and dexamethasone for patients with seizures and neurocysticercosis with viable cysts significantly reduced the incidence of subsequent seizures.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
