Candida glabrata
Candida glabrata
Abstract & Commentary
Synopsis: Caspofungin and flucytosine were the active antifungals against C. glabrata, while fluconazole was least active. Isolates with high level resistance to fluconazole demonstrated reduced susceptibility to voriconazole.
Source: Pfaller MA, et al. Geographic Variation in the Susceptibilities of Invasive Isolates of Candida glabrata to 7 Systemically Active Antifungal Agents: A Global Assessment From the ARTEMIS Antifungal Surveillance Program Conducted in 2001 and 2002. J Clin Microbiol. 2004;42:3142-3146.
Pfaller and colleagues at the University of Iowa evaluated the in vitro susceptibility of 601 worldwide isolates of Candida glabrata against 7 antifungal agents by the NCCLS broth microdilution method. North American isolates represented 55% of the total. NCCLS susceptibility criteria for fluconazole (S, < 8 mcg/mL; S-dose dependent, 16 to 32 mcg/mL; R, > 64 mcg/mL) and flucytosine (S, < 4 mcg/mL; intermediate, 8 to 16 mcg/mL; R, > 32 mcg/ml) were utilized. For the other drugs (for which no NCCLS criteria exist), < 1 mcg/mL was used as the criterion for susceptibility.
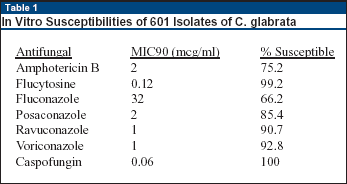
Caspofungin and flucytosine were the most active agents, with 100% and 99.2% isolates being judged susceptible (see Table 1, below). The least active agent was fluconazole, with an MIC90 of 32 mcg/mL and only 66.2% being fully susceptible. By the criteria utilized here, only 75.2% were susceptible to amphotericin B; 92.8% were susceptible to voriconazole.
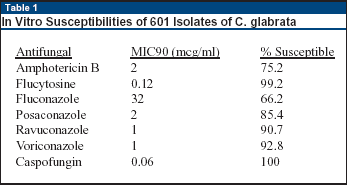
Forty-six (7.7%) isolates were resistant to fluconazole (MIC > 64 mcg/mL). All 46 were susceptible to caspofungin and to flucytosine, but cross-resistance to triazoles (voriconazole, itraconazole, posaconazole, and ravuconazole) was the rule. While only 78.3% of these fluconazole-resistant isolates were susceptible to < 1 mcg/mL amphotericin B, all were susceptible to < 2 mcg/mL (see Table 2, below). However, among the total of 601 isolates, both fluconazole-susceptible and fluconazole-resistant, 4.4% of European isolates had an amphotericin B MIC > 2 mcg/mL, a result found in < 1% of North American isolates.
Comment by Stan Deresinski, MD, FACP
C. glabrata is often the second most frequently isolated Candida species isolated from bloodstream infections, surpassed only by C. albicans. This is true despite the fact that C. glabrata may be misidentified as other species of Candida.1-2 Furthermore, its isolation may be delayed because of its slower growth in culture in the laboratory.3 The species identification engenders further delay and antifungal susceptibility test results often take a week or more before they are available to the clinician. All of this points to the importance of the initial choice of antifungal therapy in the patient with presumed or identified candidemia. This choice must necessarily be based on presumptions regarding the potential likelihood of antifungal resistance, as well as drug toxicity.
Fluconazole is the most widely used antifungal agent. Unfortunately, C. glabrata is often either intermediately susceptible ("susceptible dose-dependent"—SDD) or resistant to this azole. In this study, only 66.2% were fully susceptible to fluconazole. While the reduced susceptibility of the 27.1% SDD isolates can be overcome, this requires the use of higher doses of fluconazole than are commonly used by many clinicians. The Infectious Disease Society of America recommends administration of 400-800 mg daily.4 Pharmacokinetic analysis would suggest that the 800 mg dose is a better choice for adequate coverage of the range of MICs in this susceptibility category.
On the other hand, fluconazole at any dose is believed to be ineffective against Candida isolates with full resistance (MIC > 64 mcg/mL). While many SDD isolates remain susceptible to newer azoles, such as voriconazole, this is not the case with the isolates with these very high MIC levels. In addition, amphotericin B appears to be less active against C. glabrata than against C. albicans and most other species of Candida. This has led to the recommendation that the dose of amphotericin B deoxycholate, used in the treatment of C. glabrata infections, be at least 1 mg/kg daily.4 It should be noted that the susceptibility breakpoints chosen for this study for these drugs, for which there are as yet no NCCLS guidelines, tended toward the conservative.
The most active agents tested were caspofungin and flucytosine. The latter drug has been underutilized for fear of its toxicity, and because of the necessity to use it in combination with another antifungal to reduce the likelihood of selection of flucytosine resistant mutants. Caspofungin is safe and effective. In a randomized trial of treatment of invasive candidiasis, the response rates to caspofungin and amphotericin B deoxycholate, among the patients whose infection was due to C. glabrata, were 76.9% and 80%, respectively.5
References
1. Coignard C, et al. Resolution of Discrepant Results for Candida Species Identification By Using DNA Probes. J Clin Microbiol. 2004;42:858-61.
2. Deresinski SC. Misidentification of Candida Species By Clinical Laboratories Not a Good Thing! Infect Dis Alert. 2004;
3. Horvath LL, et al. Detection of Simulated Candidemia By the BACTEC 9240 System With Plus Aerobic/F and Anaerobic/F Blood Culture Bottles. J Clin Microbiol. 2003;41:4714-4717.
4. Pappas PG, et al. Guidelines for Treatment of Candidiasis. Clin Infect Dis. 2004;38:161-189.
5. Mora-Duarte J, et al. Comparison of CaspoFungin and Amphotericin B for Invasive Candidiasis. N Engl J Med. 2002;347:2020-2029.
Stan Deresinski, MD, FACP, Clinical Professor of Medicine, Stanford; Associate Chief of Infectious Diseases, Santa Clara Valley Medical Center, is Editor of Infectious Disease Alert.
Caspofungin and flucytosine were the active antifungals against C. glabrata, while fluconazole was least active. Isolates with high level resistance to fluconazole demonstrated reduced susceptibility to voriconazole.Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
