Myocardial Perfusion Imaging: From Diagnosis to Prognosis
Myocardial Perfusion Imaging: From Diagnosis to Prognosis
Author:
Arash Kardan, MD, Nuclear Medicine Physician, Kettering Healthcare Network, Kettering, OH.
Peer Reviewer:
Michael H. Crawford, MD, Professor of Medicine, Chief of Clinical Cardiology, University of California, San Francisco.
This article is adapted from one that originally appeared in the January 2013 issue of Primary Care Reports.
Case 1: You are evaluating a 48-year-old woman for chest pain. The pain is episodic, sometimes with activity and sometimes with rest. The patient has diabetes and smokes cigarettes. The patient is currently pain-free. The physical examination is normal. The ECG has non-specific T-wave changes in the anterior precordium. The initial serum troponin-I is normal. Would a radionuclide stress test be a reasonable and appropriate study in this patient?
Case 2: You are evaluating a 67-year-old man who presents with acute chest pain for the past hour. The onset awoke him from sleep. The pain is moderately severe, located in the center of his chest, but does not radiate. He smokes cigarettes but has no history of diabetes or hypertension. The physical examination is normal. The ECG shows minor J-point elevation of about 1 mm with concave ST-segments in leads V2 to V4. The serum troponin-I is normal. You have treated him with aspirin and morphine. The patient's wife tells you that he was evaluated by a cardiologist three months ago for chest pain. You call the cardiologist and learn that a radionuclide stress test done two months ago was normal. The patient's pain is improving and he is remaining stable. Does the recent stress test provide reassurance? Would it be reasonable to discharge the patient home?
Introduction
Myocardial perfusion imaging (MPI) refers to the utilization of radiotracers to image regional myocardial perfusion from coronary artery blood flow to the heart muscle. Used effectively, MPI can provide the clinician with a noninvasive technique that yields both important diagnostic and powerful prognostic information regarding the functional significance of anatomic coronary artery disease. MPI can help guide therapeutic decision-making by stratifying patients with respect to future risk for adverse outcomes.
Similar to other nuclear medicine imaging techniques, trace amounts of radioactive isotopes are combined with other compounds ("ligands") to form radioactive-labeled chemicals or "radiotracers." These radiotracers localize to specific organs within the body based on the ability of the compound to interact with metabolic pathways or cellular receptors. Thus, radiotracer localization images according to cellular function and physiology rather than physical changes in the tissue anatomy. Nuclear medicine, in a sense, is radiology done inside out, or endoradiology, because it creates images from radiation emitted from within the body rather than radiation that is generated by external sources, such as X-rays.
MPI uses radiotracers that are delivered via blood flow to the heart and then taken up by myocardium. With normal blood flow and metabolic function, healthy myocardium takes in enough radiotracer to be easily visible. Coronary blood flow and myocardial perfusion varies with metabolic demand. With exercise, the heart rate increases, the metabolic needs of the myocytes for oxygen increase, and coronary perfusion rises to increase delivery of oxygenated blood. With blood flow-limiting stenosis in the coronary arteries, the affected myocardium will incorporate less radiotracer that will be detectable as a difference from normal areas. With current technology and protocols, MPI can detect perfusion abnormalities before metabolic, electrical, or anatomic disturbances occur.1 (See Figure 1.)
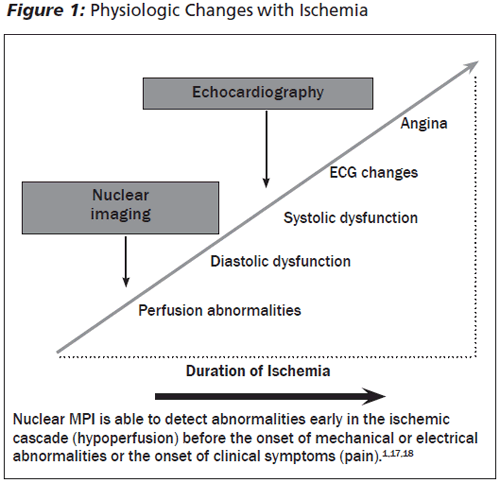
Emergency physicians often review and rely on the results of radionuclide stress tests when evaluating patients in the ED. Some emergency physicians also obtain stress tests on patients in the ED to assist in the disposition of chest pain patients. Thus, it is useful to have knowledge concerning these tests, how they are done, and what the results may mean.
Coronary Anatomy
Two coronary arteries branch from the main aorta just above the aortic valve; the right coronary (RCA) and left main. After a short distance, the left main divides into the left anterior descending (LAD) and left circumflex (LCX) arteries. The most important vascular beds supplied by the coronary arteries are the ventricles, the main pumping chambers of the heart. In general, the RCA supplies blood to the right ventricle, and in 80% of adults, the inferior portion of the left ventricle. The LAD supplies the anterior portion of the left ventricle. The LCX supplies the lateral portion of the left ventricle and in 10% of adults, the inferior portion too. About 10% of adults have a dual supply to the inferior portion of the left ventricle, from the RCA and LCX.
The coronary arteries divide to encircle the heart, covering its surface with a lacy network that looks like a slightly crooked crown. There is overlap between the areas perfused by the different arteries. Also, collateral circulation can develop to supply an area from one artery when the other is obstructed. All combined, the coronary arteries carry about 130 gallons of blood through the heart daily.2
Atherosclerosis, Coronary Artery Disease, and Myocardial Infarction
Current theories of the pathogenesis of the lesions of atherosclerosis start with low-grade injury to the artery wall that elicits an inflammatory response, which in turn causes accumulation of blood cells and plasma constituents in the intima of the artery.3 Encrustation of small mural thrombi at sites of arterial injury are organized by the growth of smooth muscle cells into them. These sites are where the lesions then subsequently grow.4,5 This hypothesis, termed the "response to injury hypothesis of atherosclerosis," has been modified to take into account aspects of risk factors associated with atherogenesis, including hyperlipidemia, hormone dysfunction, hypertension, cigarette smoking, and diabetes.6,7
Almost all ischemic heart disease is due to atherosclerosis of the coronary arteries, termed coronary artery disease, or CAD. Almost all myocardial infarctions (MIs) result from CAD, generally with superimposed coronary thrombosis. The end result is luminal narrowing of the coronary artery that impairs blood flow. Below a critical level of blood flow, myocardial cells develop ischemic injury, and if the impairment is severe and/or prolonged, irreversible damage with cell necrosis (i.e., infarction) occurs.8 A coronary artery lesion that results in a 50% reduction or greater in the luminal diameter is considered significant, likely to impair blood flow during periods of stress, resulting in myocardial ischemia.
Atherosclerotic coronary obstructions are an easily understood explanation of why patients have exertional angina — the increased metabolic demand associated with exercise creates a need for increased oxygen, and the ability of coronary flow in that region to increase is limited by the blockage. At rest, oxygen delivery is adequate for demand. It is only with exercise that ischemia develops, but is reversible when demand drops.
The pathogenesis for ischemia to occur at rest, unstable angina or myocardial infarction (two processes encompassed in the concept of acute coronary syndrome), is complex and related to a change in the heretofore stable atherosclerotic obstructions. These lesions may become unstable, promoting the development of overlying thrombosis. This may turn a patient with stable, predictable, and reversible ischemia into one with an unstable and potentially progressive course.
Myocardial Perfusion Imaging
The two radionuclides used for MPI are thallium-201 and technetium-99m. Both emit radioactivity in the form of gamma rays or photons. These photons are detected by devices ("gamma cameras") as two-dimensional images, termed planar MPI. Radioactivity can also be detected by devices that utilize multiple gamma cameras, with the results analyzed using computed tomographic reconstruction principles to create a three-dimensional representation of radionuclide distribution. This can then be used to generate tomographic images in different planes of view, similar to CT or MR imaging. The overall technology is called single photon emission computed tomography, or SPECT.
Thallium-201 and technetium-99m have important differences (see Figures 2-4); thallium-201 emits a lower energy photon (70-80 keV) and longer half-life (73 hours) compared to technetium-99m (140 keV and 6 hours, respectively). The higher energy of the technetium-99m-emitted photon means fewer photons are stopped by the body and more arrive at the gamma camera, resulting in better image quality.9,10 The shorter half-life of technetium-99m reduces the duration that the patient is exposed to radioactivity and allows for higher doses to be administered. Thus, technetium-99m has become the predominant radionuclide used for MPI.

Thallium-201 behaves as a potassium analog and, therefore, can shift in and subsequently out of myocardiocytes.11,12 Thallium-201 is readily taken up by the myocytes in a linear relationship to perfusion over a wide range of flows.13,14 Because thallium-201 was the first agent used for MPI, even today many physicians refer to all MPI studies as "thallium scans." As noted, due to its long half-life, which limits the dose that can be administered, and its low energy gamma radiation, which limits picture quality, thallium-201 has largely been replaced by technetium-99m-labeled radiotracers for evaluation of myocardial perfusion. However, due to its behavior as a potassium-like analog, thallium continues to be used as a cost-effective radiotracer alternative in evaluation of myocardial viability.10
As an isolated element, technetium-99m does not participate in intracellular metabolic processes or bind to cell surface receptors. In order to localize this radionuclide to the myocardium, it must be attached to ligands to form radiotracers. The two commercially available radiotracers using technetium-99m are sestamibi (Cardiolite®) and tetrofosmin (Myoview®). Both of these ligands are cationic lipophilic molecules that diffuse through the cell membrane and subsequently localize in the mitochondria. Unlike thallium, which shifts back out of myocardial cells analogous to potassium transport, both the technetium-99m radiotracers get permanently "locked" in the mitochondria and do not redistribute. This stability of localization, along with higher energy resulting in clearer imaging, allows for the acquisition of gated images synchronized utilizing electrocardiogram leads to take pictures of the heart during different stages of contraction. With this information, wall motion and ejection fractions can be evaluated, something not possible with thallium.1,9,10
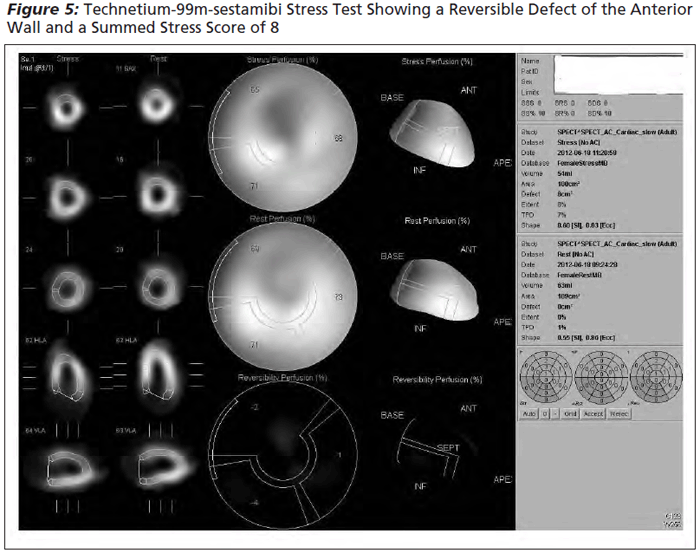
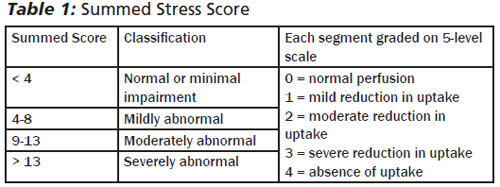
Once the radionuclide images are acquired and reconstructed in three dimensions, they can be superimposed on a model of the left ventricle, divided into segments or regions perfused by each of the major coronary arteries. (See Figure 5.) Perfusion in each segment or region can be qualitatively assessed and graded by visual inspection. Alternatively, perfusion can be quantitatively assessed by the intensity of radioactivity emanating from each region. A numerical score can be calculated to quantify the impairment to myocardial perfusion. The Summed Rest Score (SRS) is the numerical score representing myocardial perfusion during rest and the Summed Stress Score (SSS) is the score calculated with exercise. The SRS identifies impaired perfusion at rest, generally areas of myocardium that are infarcted and not recoverable. The SSS identifies poor perfusion in both areas that have diminished perfusion at rest in addition to areas that are now ischemic with exercise. The difference between the two scores is the amount of reversibility between stress and exercise or the extent of reversible ischemic burden to the left ventricle. 16 (See Table 1.) The semi-qualitative visual approach divides the left ventricle into 17 segments and grades perfusion in each segment using a five-level scoring system: 0 = normal perfusion and 4 = absence of perfusion in the area of interest. The individual segment scores are then totalled, with a sum greater than 13 indicating severe abnormality.
Software applications have been created to calculate the SSS and SRS by automatic segmentation, quantification, analysis, and display of myocardial perfusion SPECT images with minimal operator intervention. The results are as good if not better than the semi-qualitative visual approach.
Stress Testing
MPI is typically done when the patient is at rest ("rest view") and during exercise ("stress view"). The principle behind this approach is that a partial coronary obstruction may not impair blood flow and oxygen delivery during rest when the metabolic demand of the myocardium is low, but when oxygen demand is high — as during exercise — the partial obstruction now limits perfusion to that vascular bed, and decreased amounts of radiotracer reach that area of myocardium compared to the other areas of the heart. This is detected comparing the rest view with the stress view, noting a region that has normal radionuclide uptake during rest but decreased uptake during exercise.
Patient preparation is important to maximize results. Ideally, the patient should be fasting. The patient should discontinue beta-blockers and calcium channel blockers for at least 24 hours. Long-acting nitrates should be discontinued for at least 4 hours. Contraindications to stress testing include active unstable angina, recent myocardial infarction, severe aortic stenosis, and uncontrolled hypertension.
Exercise is typically done usually a treadmill but can also be done with a bicycle. Treadmill testing uses a defined protocol in which the rate and slope of the device is periodically increased to place increasing demands on the heart. The end-point of the exercise is when the heart rate reaches a predefined level, usually adjusted according to the patient's age, typically 85% of the age-adjusted maximal heart rate. If the exercise portion of the study is halted before the patient's heart rate reaches the predefined level, the resulting images may be inadequate to detect CAD.
Exercise stress testing is done with continuous electrocardiographic (ECG) monitoring and periodic blood pressure measurements. The ECG monitoring is done for practical purposes (to assess heart rate during the test), for testing purposes (to detect ST-segment depression that may be a sign of myocardial ischemia), and for safety purposes (to detect arrhythmias or cardiac conduction abnormalities provoked during exercise). Exercise stress testing can also provide an assessment of the patient's physical condition and identify symptoms such as chest pain, dyspnea, or lightheadedness provoked by exertion.
When the distribution of radiotracer is the same during rest and stress, the study is considered normal, or in the parlance of reporting, considered as showing no evidence of reversible ischemia. (See Figure 2.) If an area of underperfusion is visible during exercise but not with rest, this is deemed evidence of reversible ischemia. (See Figure 3.) If there is a region of decreased radiotracer uptake identical in both the rest and stress views, this is termed a fixed defect, usually due to prior myocardial infarction. (See Figure 4.)
A variety of protocols for timing (one or two day) and sequencing (rest or stress first, followed by the other) exist. A typical one-day protocol using technetium-99m-sestamibi (Cardiolite®) is to intravenously inject a low dose (6 to 10 millicuries) of the radiotracer with the patient at rest and obtain the SPECT images 45 minutes later. Image acquisition takes place after 30 minutes. After that, the patient exercises with injection of a larger dose (20-30 millicuries) of technetium-99m-sestamibi when the target heart rate is achieved. The exercise is stopped so that the SPECT images are obtained 30 minutes later. The larger dose for the stress portion is given so that myocardial perfusion with exercise can be distinguished from the residual radioactivity left over from the rest study. This is usually successful, but for clearer distinction, the rest and stress studies can be separated by 24 hours to allow for reduction in residual radioactivity from the initial study.
A simplified protocol is to perform only the exercise stress study, and if normal, a rest study is not performed. The principle is that if an adequate stress is achieved and no myocardial perfusion defects are seen, the patient is very unlikely to have significant CAD, and comparison with a rest study does not add additional information.
Like other diagnostic tests, stress MPI is not perfect, and patients with CAD can have normal studies and patients with clean coronary arteries on cardiac catheterization can have a positive stress test. The accuracy of exercise stress MPI testing is affected by several factors, some of which can account for false-negative MPI studies in patients with CAD. (See Table 2.) If the patient cannot exercise long enough to place an adequate demand on the heart, flow-limiting coronary obstructions may not manifest. Body habitus may affect the acquisition of images. Drugs and medications may influence the test, especially beta-blockers, long-acting nitrates, and calcium-channel antagonists. Since MPI compares blood flow between different regions of the heart, if all regions are equally affected, the difference will not be detectable. A worrisome cause for false-negative results is in patients with multivessel disease, with the obstruction balanced in the vascular beds ("3-vessel disease"), which may have equal radiotracer uptake during both rest and exercise so that no perfusion difference will occur between rest and stress imaging.

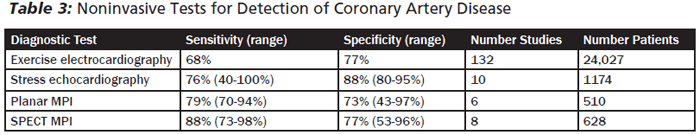
The reported sensitivity and specificity of exercise stress MPI testing varies according to the population studied, the technique utilized, and the gold-standard applied for comparison. Overall, exercise stress MPI testing is reasonably accurate. A meta-analysis of 33 studies, which included thallium-201 and technetium-99m tracers, found that SPECT MPI with treadmill exercise stress for the detection of significant coronary artery disease, defined as a stenosis of more than 50%, had an average sensitivity of 87% and specificity of 73%.9 A positive test increased the likelihood of the patient having CAD by about three times (LR+ = 3.2), and a negative test decreased the likelihood by about five times (LR- = 0.18).
With the addition of gated (synchronized with the ECG) SPECT imaging in MPI with technetium-99m radiotracers in patients with symptoms suggestive of typical or atypical angina, exercise stress SPECT MPI yielded even more accurate results; a sensitivity of 85-90% and a specificity of 80-90%.9,19-21
A meta-analysis of 1405 patients compared stress echocardiography with stress MPI for detection of coronary artery disease. MPI yielded higher sensitivity (84% for MPI vs 80% for stress echo), but had lower specificity (77% for MPI vs 86% for stress echo).17 Compared to other techniques to detect CAD, exercise stress MPI is one of the most accurate. 9,19-21 (See Table 3.) The studies included in this meta-analysis primarily utilized older techniques, whereas studies utilizing more contemporary techniques for MPI have yielded specificities similar to stress echocardiography.9,19-21
Alternatives to Exercise Testing
For patients incapable of adequate exercise, pharmacologic means can be used to increase heart rate and myocardial oxygen demand (e.g., dobutamine) or induce coronary vasodilation and increase blood flow (e.g., dipyridamole, adenosine, or regadenoson).
In a dobutamine stress test, the drug is administered as an intravenous infusion starting at 5 micrograms/kg per minute. The infusion rate is increased every three minutes until the target heart rate is achieved, at which time the radiotracer is injected and the infusion is discontinued. Contraindications to the use of dobutamine include severe aortic stenosis, unstable angina, recent myocardial infarction, history of tachyarrhythmias, severe hypertension, and poor left ventricular function. If the patient experiences adverse side effects during the dobutamine infusion, treatment with intravenous beta-blockers can be beneficial.
In an adenosine stress test, the drug is infused at a rate of 0.14 milligrams/kg per minute for 6 minutes and the technetium-99m radiotracer is injected 3 minutes after the start of the infusion. For a regadenoson (Lexiscan®) stress test, the drug is administered as a 0.4 mg IV push over 10-15 seconds, with administration of the technetium-99m radiotracer 10-20 seconds later. Both adenosine and regadenoson produce coronary vasodilation and increase blood flow by stimulating adenosine receptors in the cardiac circulation. Adenosine receptor antagonism may also cause bronchoconstriction and atrioventricular block. Contraindications to the use of adenosine or regadenoson for stress testing include acute asthma exacerbation with wheezing, severe chronic obstructive pulmonary disease, active unstable angina, recent myocardial infarction, and hypotension. Patients with second- or third-degree atrio-ventricular block or sick sinus syndrome with a functioning cardiac pacemaker may undergo stress testing with these agents. Severe adverse side effects are treated with aminophylline. With both adenosine and regadenoson, patients should not ingest caffeine for 12 to 24 hours prior to the stress test using these agents, as caffeine interferes with the degree of coronary vasodilation and reduces the ability to detect CAD.
With either of these three pharmacologic agents, images are acquired 45 minutes after the radiotracer injection. SPECT MPI with pharmacologic stress demonstrates similar results to exercise testing, with an average sensitivity of 89% and specificity of 75%.9
Risk Stratification
The application of prognostic testing is based on the premise that patients can be stratified with respect to risk for adverse outcomes and can receive intervention to change the natural history of their disease so that their subsequent risk is reduced. Several studies have historically shown that patients with stable coronary disease can be stratified into different risk groups based on demographics, exercise, MPI, and catheterization information.22-31
Schinkel and colleagues found that half of 650 patients with known or suspected CAD with normal exercise ECGs had abnormal SPECT myocardial perfusion studies. In 20% of these studies, the abnormalities were related to ischemia and, therefore, reversible. The degree of abnormality on myocardial perfusion SPECT was predictive of cardiovascular death or morbidity out to 10 years. The SSS, which measures both infarcted and ischemic myocardium, was an independent predictor of major adverse cardiac events. The SRS, which measures the amount of infarcted myocardium, was an independent predictor of death.
Studies that utilized planar imaging techniques with treadmill exercise stress found that a normal study was a significant predictor of lower risk for MI or death at 1 year.24-26 Compared with planar imaging, SPECT imaging has better image quality, improved sensitivity in detecting individual diseased vessels, and is more quantitative in nature with respect to describing the severity or extent of disease.32,33 Iskandrian and colleagues examined the independent and incremental prognostic value of exercise SPECT thallium imaging in patients with angiographically defined coronary artery disease. Data were obtained in 316 medically treated patients with coronary artery disease who underwent coronary angiography over a mean follow-up time of 28 months. The primary endpoints were cardiac death or non-fatal MI. Univariate analysis showed that gender, exercise work load, extent of coronary artery disease and left ventricular ejection fraction, and thallium SPECT variables were prognostically important. Multivariate analysis demonstrated that thallium SPECT data provided incremental prognostic value to catheterization data, with the extent of the perfusion abnormality being the single best predictor of prognosis.34
The important concept for emergency physicians is the low risk of death for patients with a normal stress test. Shaw and colleagues enrolled 4728 consecutive patients who underwent SPECT MPI with yechnetium-99m-tetrofosmin with either exercise or pharmacologic stress. The observed annualized survival rate for those patients with a normal study was 99.4%.35 Other investigators have demonstrated that event rates associated with normal or low-risk myocardial perfusion SPECT with thallium or technetium agents are less than 1% per year of follow-up.37-43
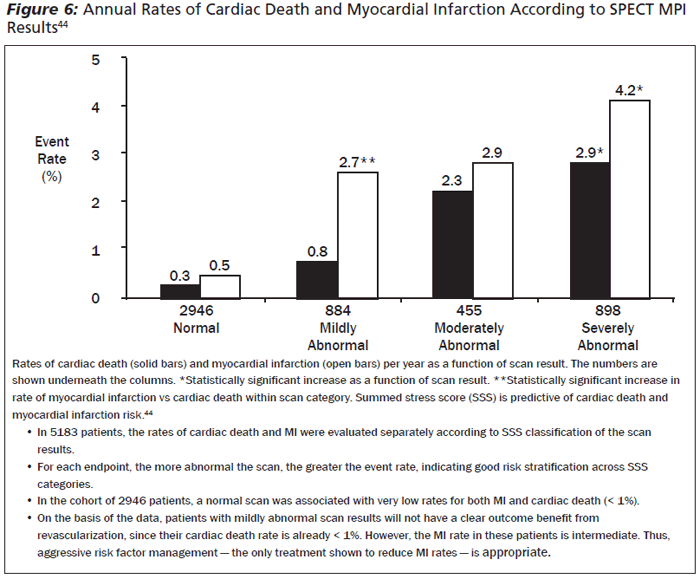
Hachamovitch and colleagues performed a study to examine differences in the rates of MI and cardiac death after SPECT myocardial perfusion testing.44 They identified 5183 consecutive patients who underwent stress/rest SPECT and were followed up for the occurrence of cardiac death or MI over a mean follow-up of 642 ± 226 days. Patients with normal scans were at low risk for myocardial infarction (0.5% per year) or cardiac death (0.3% per year). Rates of both outcomes increased significantly with worsening scan abnormalities. (See Figure 6.)
Recommended Uses for Nuclear MPI
The Appropriate Use Criteria (AUC) for Cardiac Radionuclide Imaging were developed by the American College of Cardiology Foundation (ACCF), along with key cardiology societies, to serve as a guide for the responsible use of radionuclide MPI.
In general, the use of MPI is appropriate for the diagnosis and risk assessment of intermediate- and high-risk CAD patients, according to the AUC. (See Figure 7.) MPI testing for low-risk patients, routine repeat testing, and general screening in certain clinical scenarios are considered less appropriate.45 If the pretest probability for CAD is low, analysis indicates that a stress test with ECG analysis alone would be more cost-effective and, thus, the use of radionuclide MPI in low-risk patients would be inappropriate.
Radiation from Nuclear Myocardial Perfusion Imaging
Procedures that utilize ionizing radiation should be performed in accordance with the as low as reasonably achievable (ALARA) philosophy. Thus, physicians ordering and performing cardiac imaging should be very familiar with the dosage of radiation from cardiac diagnostic tests and ways in which the dose can be minimized.
Radiation dosimetry from a study using a radiopharmaceutical is typically estimated on the basis of a mathematical biokinetic model that quantifies the distribution and metabolism of that agent in the body. Such models incorporate biokinetic data from animal and human models.
Effective doses of MPI procedures are nontrivial and vary greatly between protocols. Substantial differences exist between procedures with the use of different radiopharmaceuticals and between different procedures with the use of the same agent. While the typical effective dose of a posteroanterior chest X-ray is 0.02 mSv, and the annual background radiation in the United States is 3.0 mSv, typical values for MPI studies range from 2.2-31.5 mSv. Of the most commonly performed studies, a rest-stress technetium-99m-sestamibi study averages 11.3 mSv, and a rest-stress technetium-99m-tetrofosmin study averages 9.3 mSv. Single-injection protocols are associated with a dose that is approximately 30% lower. Doses are much higher for studies using thallium-201. A single-injection thallium-201 MPI study has an average value of 22 mSv. Dual isotope studies have the highest effective doses, with an average value of 29.2 mSv for a combination thallium-201 and technetium-99m- sestamibi study or approximately three times that of a single-injection protocol using a technetium-99m containing agent.46 Thus, ALARA considerations appear to favor the use of technetium-99m agents rather than thallium-201.46
Biological effects of ionizing radiation can be classified as deterministic or stochastic. Deterministic effects such as skin injuries and cataract formation occur predictably when the dose exceeds a certain threshold, whereas stochastic effects such as cancer incidence and germ cell mutations occur with a probability that increases with the dose.
In cardiac imaging, the only deterministic effect that occurs with any frequency in patients is skin injury. Stochastic risks of potential concern include heritable genetic effects and cancer. Risks for all classes of genetic diseases occur at a rate estimated at 0.30% to 0.47% per Gy per first-generation progeny. Even with the highest gonadal doses found in cardiac imaging using thallium-201, the testicular absorbed dose of approximately 60 mGy, corresponded to a risk of genetic diseases of only 0.02% to 0.03% per first-generation progeny.46
Ionizing radiation causes numerous types of DNA damage and it is hypothesized that damaged sites such as double-strand breaks are oncogenic. For the type of radiation used in cardiac imaging, i.e., low levels (≤ 100 mSv) of low linear energy transfer ionizing radiation, the relationship between dose and lifetime risk of cancer is a controversial one. The estimated lifetime risk for all cancer attributable to a single radiation exposure increases with the intensity of radiation and decreases with age. Thus, the estimated lifetime risk of cancer for a 40-year-old due to a 100 mSv exposure is less than 1%, whereas in a newborn, it is 2-4%.
Conclusion
Case 1: The patient has two major risk factors for CAD and is symptomatic with a mixed picture of exertional and rest chest pain. The pretest risk stratification for this patient would be moderate risk for CAD, and stress MPI is appropriate.
Case 2: This patient has four major risk factors for CAD and with new-onset chest pain has entered an unstable phase. The patient's recent stress test result is irrelevant when acute symptoms or instability occurs. The patient underwent coronary angiography the next day and was found to have 3-vessel CAD with acute obstruction of the LAD. The relatively balanced obstructions in the three major coronary arteries accounted for the normal stress test.
References
- Mieres JH, Makaryus AN, Redberg RF, et al. Noninvasive cardiac imaging. Am Fam Physician 2007;75:1219-1228.
- Clark J. A Visual Guide to the Human Anatomy: A Comprehensive Atlas of the Structures of the Human Body. New York: Sterling Publishing; 2001:119.
- Virchow R. Phlogose und Thrombose im Gefäβsystem, Gesammelte Abhandlungen zur Wissenschaftlichen Medicin. Frankfurt am Main: Meidinger, Sohn, and Co.; 1856:458.
- Rokitansky C. A Manual of Pathological Anatomy. Translated by: Day GE. London: The Sydenham Society; 1852:vol 4.
- Duguid JB. Thrombosis as a factor in the pathogenesis of coronary atherosclerosis. J Pathol Bacteriol 1946;58:207.
- Ross R, et al. The pathogenesis of atherosclerosis. In: Braunwald E (ed.) Heart Disease: A Textbook of Cardiovascular Medicine. 4th ed. Philadelphia: WB Saunders Company; 1992:1113.
- Ross R, et al. The pathogenesis of atherosclerosis. In: Braunwald E (ed.) Heart Disease: A Textbook of Cardiovascular Medicine. 4th ed. Philadelphia: WB Saunders Company; 1992:1106.
- Pasternak, et al. Acute myocardial infarction. In: Braunwald E (ed.) Heart Disease: A Textbook of Cardiovascular Medicine. 4th ed. Philadelphia: WB Saunders Company; 1992:1201.
- Klocke FJ, Baird MG, Lorell BH, et al. ACC/AHA/ASNC guidelines for the clinical use of cardiac radionuclide imaging — Executive summary: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (ACC/AHA/ASNC Committee to Revise the 1995 Guidelines for the Clinical Use of Cardiac Radionuclide Imaging). Circulation 2003;108:1404-1418.
- Zeissman HA, et al. Cardiac system. In: Thrall J (ed.) Nuclear Medicine: The Requisites in Radiology. 3rd ed. Philadelphia: Elsevier Mosby; 2006:450-507.
- Kawana M, Krizek H, Porter J, et al. Use of 199 Thallium as a potassium analog in scanning. J Nucl Med 1970;11:333.
- Britten J, Blank M. Thallium activation of Na-K ATPase of rabbit kidney. Biochem Biophys Acta 1968;159:160-166.
- Bradley-Moore PR, Lebowitz E, Greene MW, et al. Thallium-201 for medical use. II. Biological behavior. J Nucl Med 1975;16:156-160.
- Strauss HW, Harrison K, Langan JK, et al. Thallium-201 for myocardial imaging. Relation of thallium-201 to regional myocardial perfusion. Circulation 1975;51:641.
- Cerqueira MD, Weissman NJ, Dilsizian V, et al; American Heart Association Writing Group on Myocardial Segmentation and Registration for Cardiac Imaging. Standardized myocardial segmentation and nomenclature for tomographic imaging of the heart: A statement for healthcare professionals from the Cardiac Imaging Committee of the Council on Clinical Cardiology of the American Heart Association. Circulation 2002;105:539-542.
- [No authors listed.] Imaging guidelines for nuclear cardiology procedures: Part 2. American Society of Nuclear Cardiology. J Nucl Cardiol1999;6:G47-G84.
- Schinkel AF, Bax JJ, Geleijnse ML, et al. Noninvasive evaluation of ischemic heart disease: Myocardial perfusion imaging or stress echocardiography? Eur Heart J 2003;24:789-800.
- Schuijf JD, Shaw LJ, Wijns W, et al. Cardiac imaging in coronary artery disease: Differing modalities. Heart 2005;91:1110-1117.
- Taillefer R, DePuey EG, Udelson JE, et al. Comparative diagnostic accuracy Tl-201 and Tc-99m sestamibi SPECT imaging (perfusion and ECG-Gated SPECT) in detecting coronary artery disease in women. J Am Coll Cardiol 1997;29:69-77.
- Amanullah AM, Berman DS, Hachamovitch R, et al. Identification of severe or extensive coronary artery disease in women by adenosine technetium-99m sestamibi SPECT. Am J Cardiol 1997;80:132-137.
- Santana-Boado C, Candell-Riera J, Castell-Conesa J, et al. Diagnostic accuracy of technetium-99m SPECT in women and men. J Nucl Med 1998;39:751-755.
- Bobbio M, Pollock BH, Cohen I, et al. Comparative accuracy of clinical tests for diagnosis and prognosis of coronary artery disease. Am J Cardiol 1988;62:896-900.
- Morris CK, Ueshima K, Kawaguchi T, et al. The prognostic value of exercise capacity: A review of the literature. Am Heart J 1991;122:1423-1431.
- Brown KA, Boucher CA, Okada RD, et al. Prognostic value of exercise thallium-201 imaging in patients presenting for evaluation of chest pain. J Am Coll Cardiol 1983;1:994-1001.
- Abraham RD, Freedman SB, Dunn RF, et al. Prediction of multivessel coronary disease and prognosis early after acute myocardial infarction by exercise electrocardiography and thallium-201 myocardial perfusion scanning. Am J Cardiol 1986;58:423-427.
- Ladenheim ML, Pollock BH, Rozanski A, et al. Extent and severity of myocardial infarction as predictors of prognosis in patients with suspected coronary artery disease. J Am Coll Cardiol 1986;7:464-471.
- Kemp HG, Kronmal RA, Vlietstra RE, et al. Participants in the coronary artery surgery study. Seven-year survival in patients with normal or near normal coronary arteriograms: A CASS registry study. J Am Coll Cardiol 1986;7:479-483.
- Mock MB, Ringqvist I, Fisher LD, et al. Survival of medically treated patients in the Coronary Artery Surgery Study (CASS) registry. Circulation 1982;66:562-568.
- De Feyter PJ, van Eenige MJ, Dighton DH, et al. Prognostic value of exercise testing, coronary angiography, and left ventriculography 6-8 weeks after myocardial infarction. Circulation 1982;66:527-536.
- Califf RM, Phillips HR 3rd, Hindman MC, et al. Prognostic value of a coronary artery jeopardy score. J Am Coll Cardiol 1985;5:1055-1063.
- Proudfit WJ, Bruschke AV, MacMillan JP, et al. Fifteen year survival study of patients with obstructive coronary artery disease. Circulation 1983;68:986-997.
- Heo J, Iskandrian AS. Stress thallium imaging. Am J Noninvasive Cardiol 1991;5:173-184.
- Iskandrian AS, Heo J, Askenase A, et al. Thallium imaging with single photon emission computed tomography. Am Heart J 1987;114:852-865.
- Iskandrian AS, Chae SC, Heo J ,et al. Independent and incremental prognostic value of exercise single-photon emission computed tomographic (SPECT) thallium imaging in coronary artery disease. J Am Coll Cardiol 1993;3:665-670.
- Shaw LJ, Hendel R, Borges-Neto S, et al. Prognostic value of normal exercise and adenosine 99mTc-tetrofosmin SPECT imaging: Results from the multicenter registry of 4,728 patients. J Nucl Med 2003;44:134-139.
- Hachamovitch R, Berman DS, Kiat H, et al. Exercise myocardial perfusion SPECT in patients without known coronary artery disease: Incremental prognostic value and use in risk stratification. Circulation 1996;93:905-914.
- Berman DS, et al. Nuclear cardiology. In: Fuster V, et al. (eds.) Hurst's: The Heart, 10th ed. New York: McGraw Hill; 2001:525-565.
- Berman DS, Hachamovitch R, Lewin H, et al. Risk stratification in coronary artery disease: Implications for stabilization and prevention. Am J Cardiol 1997;79:10-16.
- Berman DS, Kiat H, Friedman JD, et al. Separate acquisition rest thallium-201/stress technetium-99m sestamibi dual-isotope myocardial persusion single-photon emission computed tomography: A clinical validation study. J Am Coll Cardiol 1993;22:1455-1464.
- Berman DS, Hachamovitch R, Kiat H, et al. Incremental value of prognostic testing in patients with known or suspected ischemic heart disease: A basis for optimal utilization of exercise technetium-99m sestamibi myocardial perfusion single-photon emission computed tomography. J Am Coll Cardiol 1995;26:639-647.
- Shaw LJ, Hachamovitch R, Heller GV, et al. Noninvasive strategies for the estimation of cardiac risk in stable chest pain patients. The Economics of Noninvasive Diagnosis (END) Study Group. Am J Cardiol 2000;86:1-7.
- Gibbons RJ, Hodge DO, Bermand DS, et al. Long-term outcome of patients with intermediate-risk exercise electrocardiograms who do not have myocardial perfusion defects on radionuclide imaging. Circulation 1999;23:2140-2145.
- Iskander S, Iskandrian AE. Risk assessment using single-photon emission computed tomographic technetium-99m sestaimibi imaging. J Am Coll Cardiol 1998;32:57-62.
- Hachamovitch R, Berman DS, Shaw LJ, et al. Incremental prognostic value of myocardial perfusion single photon emission computed tomography for the prediction of cardiac death: Differential stratification for risk of cardiac death and myocardial infarction. Circulation 1998;97:535-543.
- Fromer L. Appropriate use criteria for cardiac radionuclide imaging: Part I. J Fam Pract online Cardiowatch Issue 1. Available at: www.jfponline.com/cardiowatch/CardioWatch-Issue1.html. Accessed Dec. 14, 2012.
- Einstein AJ, Moser KW, Thompson RC, et al. Radiation dose to patients from cardiac diagnostic imaging. Circulation 2007;116:1290-1305.
Subscribe Now for Access
You have reached your article limit for the month. We hope you found our articles both enjoyable and insightful. For information on new subscriptions, product trials, alternative billing arrangements or group and site discounts please call 800-688-2421. We look forward to having you as a long-term member of the Relias Media community.
